| Case Report | ||
Open Vet J. 2022; 12(4): 519-524 Open Veterinary Journal, (2022), Vol. 12(4): 519–524 Case Report Ovarian granulosa cell tumor in a Persian cat: Case report from Tripoli, LibyaAsma M. Elbahi1*, Shaima A. M. Mahgiubi2, and Abdurraouf O. Gaja31Department of Anatomy, Histology and Embryology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 2Department of Pathology and Clinical Pathology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 3Department of Surgery and Theriogenology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya *Corresponding Author: Asma M. Elbahi. Department of Anatomy, Histology and Embryology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya. Email: a.elbahi [at] uot.edu.ly Submitted: 12/04/2022 Accepted: 17/07/2022 Published: 10/08/2022 © 2022 Open Veterinary Journal
AbstractBackground: Ovarian granulosa cell tumors (GCTs) are common neoplasms in domestic animals that originate from the sex-cord stromal cells of the ovary. The aim of the present paper was to report a case of ovarian GCT in a 5-year-old Persian female cat and to describe the histopathological pattern of this tumor. Case Description: The cat was brought for a routine diagnostic examination for pregnancy at Albaittar clinic in Tripoli, Libya with a history of 1 month mating before it was brought to the clinic for investigation of progressive behavioral changes. The cat external examination showed noticeable enlargement in the abdomen that was potentially suspected of pregnancy; however, the abdominal ultrasonography showed a great mass located on the right ovary. A therapeutic ovariohysterectomy was performed. The cat’s clinical signs resolved, 6 months later it was diagnosed with a mass on the right kidney suspected as metastasis and one week later the cat died. Conclusion: In this case, macroscopic and microscopic studies are discussed and histopathological examination confirmed ovarian GCT with cystic endometrial hyperplasia and suspected metastasis in one kidney. Keywords: GCT, Ovary, Histopathological finding, Persian cat, Tripoli. IntroductionGranulosa cell tumors (GCTs) are the most common sex cord–stromal tumors in animals (McEntee, 2002), and predominately occur in older cats (Saba and Lawrence, 2013). These tumors arise from ovarian granulosa cells from the specialized stromal of the ovary and include GCTs, luteomas (also called interstitial gland, lipid, or interstitial cell tumors), thecomas, and retiform Sertoli-Leydig cell tumor (Gomez-Laguna et al., 2008; Solano-Gallego and Masserdotti, 2016). In cats, nearly, 50% of GCTs are malignant (metastasis) and appear unilateral, smooth surfaced, and round with a diameter between 20 and 30 cm (Foster, 2006). The metastatic sites of these tumors may involve the peritoneum, lumbar lymph nodes, omentum, diaphragm, kidney, pancreas, spleen, liver, and lungs (McEntee, 2002). Macroscopic and histological reports on feline GCTs are scarce. Typically, these tumors are described as round to cuboidal cells with indistinct boundaries. The cells form various histologic patterns, including micro and macrofollicular arrangements, nests, sheets, trabecular to ribbon-like cords (palisading), and diffuse. The cytoplasm of cells is scarce to moderate with variable amounts of vacuolated spots, and the nuclei are round to oval with indistinct nucleoli and mild to moderate cellular atypia. There are also spindle cell or “Sertoli cell” variants. (Kennedy et al., 1998). Cells may be visualized and arranged in an acinar pattern surrounding amorphous eosinophilic extracellular mucin material called Call-Exner bodies which is a useful diagnostic feature of GCT (MacLachalan and Kennedy, 2002; Foster, 2006; Baba and Câtoi, 2007). Mitoses and cellular atypia are obvious and Leydig cells may appear with lipid, round, uniform, and well-delimited cells (Kennedy et al., 1998; Baba and Câtoi, 2007). Cystic endometrial hyperplasia (CEH) is commonly reported in association with GCT and reflects the effects of estrogen or progesterone on the endometrium (Sivacolundhu et al., 2001). In cats, macroscopically, endometrial hyperplasia is thick and convoluted and the corkscrew shape of the lumen is exaggerated with visible cysts, especially on cross sections. Histologically, the changes vary from mild to severe in the endometrium with hyperplasia (Foster, 2006). Cats with CEH sometimes show no clinical signs; however, it could be associated with implant failure and infertility as well as small littermates and early embryonic death (Agudelo, 2005). This presented case report investigate the macroscopic and microscopic findings of ovarian GCT associated with CEH in a 5-year-old Persian cat from Tripoli-Libya. Case DetailsA 5-year-old Persian, female cat was brought for investigation of progressive behavioral changes (loss appetite, loss weight, imbalance, and lethargy) at Albaittar clinic in Tripoli, Libya. The cat was mated 1 month before it was brought to the clinic. It showed vigorous behavior in a male during estrus. The cat’s owner visited the clinic for suspecting pregnancy. At presentation, the cat had a body weight of 2.5 kg and a body temperature of 38°C. Physical examination revealed an enlargement of the abdomen, with a palpable irregular-shaped mass on the right side of the mesogastric region. Abdominal Ultrasound examination (DRAMIŃSKI ANIMALprofi L, DRAMIŃSKI, Poland) revealed the presence of an echogenic irregular mass in the right ovary. The cat subsequently underwent therapeutic ovariohysterectomy (OHE). Ovaries, uterus, contralateral ovary, and fallopian tube were fixed in 10% neutral buffered formalin within 48 hours and submitted for macroscopic and histopathological examination. The cat received 20 mg/kg of long-acting Amoxicillin for five consecutive days after the operation and the wound was dressed with wound disinfectant and antibiotic powder every 2 days. The cat started to eat on the next day of the operation. Macroscopic examination revealed a uterus measured 11 cm in length, convoluted with corkscrew appearance, soft in consistency, and intact serosal layer (Fig. 1a). The right ovary showed an attached ovarian cystic tissue mass measured 6 × 5 × 3 cm, rounded in shape, dark red to tan in color, soft to firm in consistency with multilobulation and intact capsule (Fig. 1b). The specimen was inked and serially cut. The contralateral ovary measured 0.9 cm and fallopian tube measured 0.5 cm in length. On cut section of the ovarian tissue mass, a well-circumscribed multilobular tumor mass, solid heterogeneous, greyish white to tan in colored areas with multiple foci of necrosis and cystic spaces were detected. The capsule is intact grossly. The uterine cavity showed thickened wall with variable cystic dilatations. The other ovary and fallopian tube showed no obvious pathological abnormality. For histopathological examination, serial sections were taken from the ovarian tumor mass and from the uterus, as well as, from the contralateral ovary and fallopian tube, and were fixed in 10% buffered formalin for later processing and examination. All histological procedures were performed according to Bancroft and Gamble (2008). The tissue sections were stained with hematoxylin and eosin (H&E) following the standard procedures. The stained tissue sections were examined under a light microscope (ZEISS, Germany). Histopathological examination of the ovarian tumor mass revealed a malignant tumor composed of diffuse atypical granulosa cells with spherical to oval nuclei, distinct nucleoli, and scant eosinophilic cytoplasm arranged in different patterns (micro follicles, small nests, acinar formation, and alveolar-like) (Fig. 2a–d). Regions of the tumor exhibited Sertoli-pattern (Luteinization), while others showed sarcomatous spindle cells arranged in tight fascicles (Fig. 2e and f). The cells were separated by connective tissue septa along with areas of hemorrhage and necrosis (Fig. 3a). Mitotic count according to active foci of the tumor showed 2–3 mitosis / 10 hpF. The fibroconnective tissue capsule (FCC) revealed evidence of vascular invasion which showed blood vessels lined by endothelial cells (Fig. 3b).
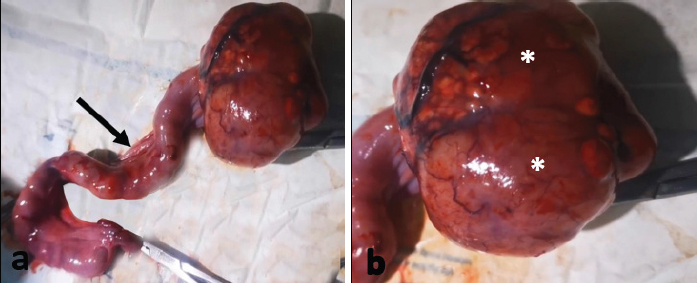
Fig. 1. Gross appearance of the uterus and the right ovarian GCT in a 5- year-old Persian cat. (a) The uterus appears thickened and convoluted with cork screw appearance (arrow). (b) The right ovary shows an attached ovarian cystic tissue mass which is a rounded in shape with multilobulation and intact capsule (asterisks).
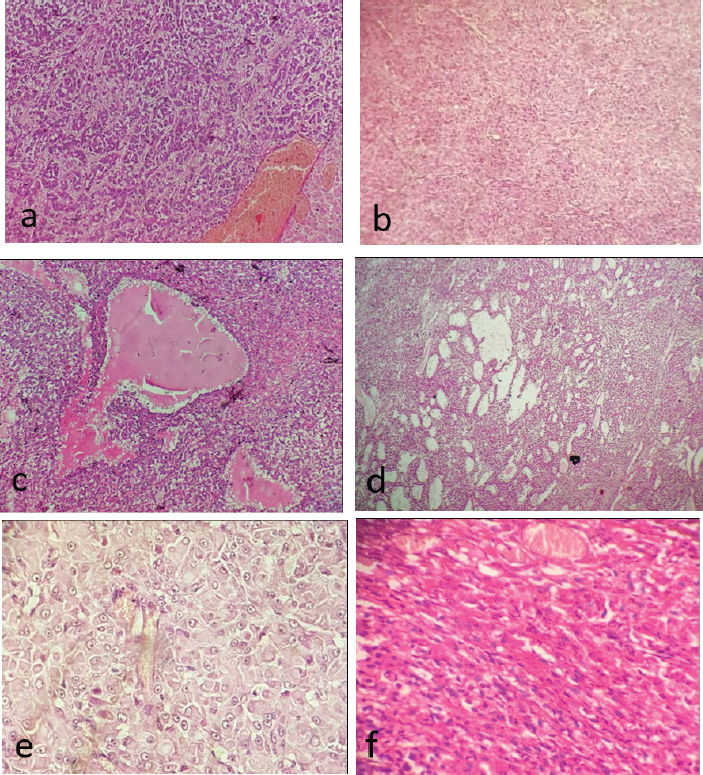
Fig. 2. Various histopathological patterns of ovarian GCT in a 5-year-old Persian cat. (a) Microfollicular growth pattern. (b) Tumor cells are arranged in small nests. (c) Acinar formation. (d) Alveolar like growth pattern. (e) Sertoli cell like growth pattern. (f): Spindle cells arranged in tight fascicles. (H&E stain; a– f: 100×; d: 40×). The endometrium examination revealed thickened endometrial lining with elongated glands and prominent stroma. There was also a dilatation of glands with formation of variable sized cysts (Fig. 4). No cytological atypia was seen, nor evidence of complexity or tumor deposits or invasion. The contralateral ovary and fallopian tube showed normal histological findings, with no evidence of malignant infiltrations.
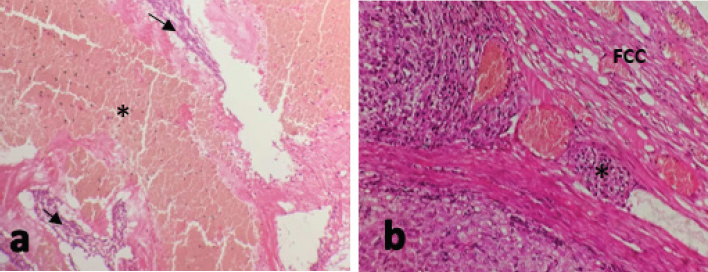
Fig. 3. (a): The tumor cells show areas of hemorrhages (asterisk) and necrosis (arrows). (b): The (FCC shows blood vessel lined by endothelial cells (vascular invasion) (asterisk). (H&E stain; a: 100×; b: 200×).
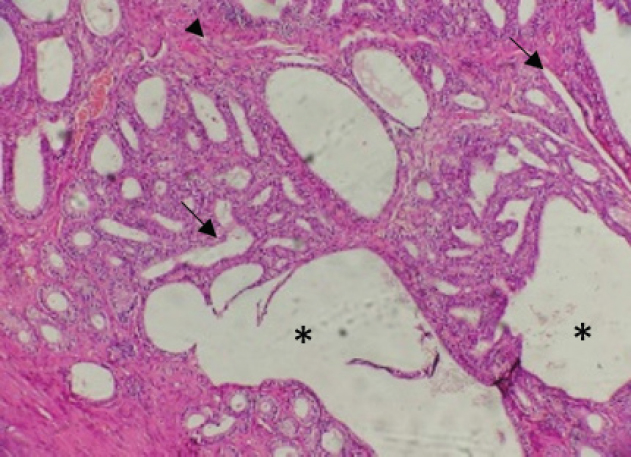
Fig. 4. CEH. The endometrium showed thickened endometrial lining with elongated glands (arrows) and prominent stroma (arrowhead), dilatation of the glands with formation of variable sized cysts (asterisks). (H&E stain; 100×). Based on histological findings, the GCT of the ovary with evidence of vascular invasion associated with CEH was made and the contralateral ovary and fallopian tube were free. The surgical intervention resulted in the complete resolution of clinical signs. However, 6 months following surgery the cat was brought to the Albittar clinic again with complaints of weakness, cachexia, ascites, off food, and lethargy with the presence of abdominal mass. Sings of pain were determined on palpation of the right abdominal region and ultrasonography examination was performed. According to the ultrasonography examination, a clear mass at the mesogastric region attached to the right kidney was determined and was suspected as a sequela of past tumor metastasis. Since the poor prognosis surgery was not performed, the cat had paralysis in hind limbs and massive weight loss after one week and eventually died. The owner of the cat rejected the euthanized decision and the postmortem examination. DiscussionGCTs are the most common sex-cord stromal tumors in many animal species and the most common ovarian tumors in felines (Norris et al., 1969; Aliakbrai and Ivoghli, 1979; McEntee, 2002). In the current case, the histopathological findings in accordance with the previous studies that described feline GCT (Kennedy et al., 1998; Gunay Ucmak et al., 2018). These features correspond with different reported cases which document the aggressive behavior of this tumor in cats (Baker, 1956; Norris et al., , 1969; Giacóia et al., 1999). The mean age preference of GCTs in cats is generally 11 years old (Holzworth, 1987), however, the presented case was only 5 years old. The reappearance of the lesion in kidney after 6 months of surgery supported the severe malignancy of the GCTs case (Gelberg and McEntee, 1985). Previously, kidney was reported among metastatic sites for GCTs associated with old cats (McEntee, 2002). In addition, Oviedo-Peñata et al. (2020) reported that GCTs in dogs frequently affect the left ovary; however, this was not the case in cats which some authors described GCTs in both ovaries (Giacóia et al., 1999; Heaps et al., 2017). Ultrasonographic imaging of the ovaries is typically used for the diagnosis of GCTs and supports the surgery intervention (Gunay Ucmak et al., 2018). This technique was useful to reveal the existence of echogenic irregular mass in the right ovary in the current case, and also was helpful to supplement histological findings of GCTs and further support our diagnosis and surgical intervention. The current case of GCT was also diagnosed with mild to severe lesions of CEH. Endometrial hyperplasia with GCTs has been previously reported in some cats and dogs associated with estrogenic effects on the uterus (Giacóia et al., 1999; Heaps, et al., 2017). Endometrial hyperplasia was mostly seen in older cats and was related to age and the effect of estrogen stimulation but not progesterone (Potter et al.,1991). Typically, GCT is usually associated with clinical signs in particular the repeated estrus considered an important differential diagnostic sign (Giacóia et al., 1999). Such association was evident, showing clinical signs like vigorous and progressive behavioral changes after 1 month of mating. In addition, the current case had CEH with no evidence of inflammation corresponding with other findings that reported hyperplasia but no evidence of inflammation (Foster, 2006). Persian cats appear to be particularly sensitive to low light levels and more prone to infrequent estrous cycles. This breed will not exhibit regular estrous cycle even during daylight, which is the common cause of infertility in queens (Little, 2012). CEH influenced by chronic estrogenic stimulation from recurrent estrous cycle that does not result in pregnancy (Potter et al., 1991). Pets with a clinical history of infertility and changes in the time of proestrus or estrus should be examined and a GCT should be included in the differential diagnosis (Toledo et al., 2021). In this case, the cat was of Persian breed with clinical history of infertility and exhibited vigorous behavior during the estrous cycle, all these signs suggest that the breed may play a role to develop a GCT and CEH. OHE remains the basis and recommended treatment for ovarian tumors. The current case complains of weakness, loss of appetite, massive weight loss, cachexia, ascites, lethargy, abdominal enlargement at mesogastric region and a mass attached to the right kidney after 6 months of surgery. This is widely reported that surgical procedures do not improve the prognosis of these types of tumors (Gunay Ucmak et al., 2018). Due to poor prognosis of such cases, no further intervention was performed and the cat eventually developed paralysis in its hind limbs and died. The cause of ovarian tumors in cats is not fully understood but the condition is closely associated with certain risk factors such as age, spaying, hormonal instability, and infection (McEntee, 2002). Nevertheless, ovarian tumors in pets are mainly linked to different risk factors including environmental and genetic factors, e.g., hereditary and prone to metastasizing (Dobson, 2013; Cannon, 2015). In the current case, the breed is possibly a potential risk factor that may play a role to develop a GCT and CEH; however, the pathogenesis of GCT and CEH may be multifactorial and requires further study. A GCT should be included in the differential diagnosis in cats with a clinical history of infertility and changes in the time of proestrus or oestrus, particularly not spayed Persian cat. To conclude, this paper reports a GCT and CEH in a cat. Although GCTs in cats were reported by many researchers around the world, this is the first report from Libya according to the authors of this study. AcknowledgmentThe authors would like to acknowledge the valuable contribution of Prof. Mohamed O. Ahmed from the Department Microbiology and Parasitology, Faculty of Veterinary Medicine, University of Tripoli, in the preparation of this manuscript. Conflict of interestThe authors declare that there is no conflict of interest. ReferencesAgudelo, C. 2005. Cystic endometrial hyperplasia-pyometra complex in cats. A review. Vet. Q. 27, 173–182. Aliakbrai, S. and Ivoghli, B. 1979. Granulosa cell tumor in a cat. J. Am. Vet. Med. Assoc. 174, 1306–1308. Baba, A.I. and Câtoi, C. 2007. Comparative oncology. Bucharest, Romania: Publishing House of the Romanian Academy Bucharest. Baker, E. 1956. Malignant granulosa cell tumor in a cat. J. Am. Vet. Med. Assoc. 129, 322–324. Bancroft, J.D. and Gamble, M. 2008. Theory and practice of histological techniques. London, UK: Elsevier health sciences. Cannon, C.M. 2015. Cats, cancer and comparative oncology. Vet. Sci. 2, 111–126. Dobson J.M. 2013. Breed-predispositions to cancer in pedigree dogs. ISRN Vet. Sci, 2013, 941275; doi: 10.1155/2013/941275. Foster, R.A. 2006. Female Reproductive System. In Pathologic Basis of Veterinary Diseases. Eds., McGavin, M.D.and Zachary, J.F. 4th ed. Mosby, China, pp: 1280-1282. Gelberg, H. and McEntee, K. 1985. Feline ovarian neoplasms. Vet. pathol. 22, 572–576. Giacóia, M.R., Maiorka, P.C., Oliveira, C.M., Sinhorini, I.L. and Dagli, M.L.Z. 1999. Granulosa cell tumour with metastasis in a cat. Braz. J. Vet. Res. Anim. Sci. 36, 250–252. Gomez-Laguna, J., Millan, Y., Reymundo, C., Domingo, V. and de Las Mulas, J.M. 2008. Bilateral retiform sertoli–leydig cell tumour in a bitch. Alpha-inhibin and epithelial membrane antigen as useful tools for differential diagnosis. J. Comp. Pathol. 139, 137–140. Gunay Ucmak, Z., Uçmak, M., Tek, Ç., Koenhemsi, L., Bamac, Ö.E., Gürel, A. and Yildar, E. 2018. Granulosa cell tumor in a spayed young queen. J. Hellenic. Vet. Med. Soc. 69, 1010–1015. Heaps, L.A., Scudder, C.J., Lipscomb, V.J., Steinbach, S.M., Priestnall, S.L., Martineau, H., Szladovits, B., Fowkes, R.C., and Garden, O.A. 2017. Serum anti-Müllerian hormone concentrations before and after treatment of an ovarian granulosa cell tumour in a cat. JFMS Open Reports, 3(2), 2055116917722701; doi: 10.1177/2055116917722701. Holzworth, J. 1987. Tumours of the female reproductive system In Diseases of the cat: medicine and surgery, 1st ed. Philadelphia, PA: W.B. Saunders, pp: 520-521. Kennedy, P., Cullen, J., Edwards, J., Goldschmidt, M., Larsen, S., Munson, L. and Nielsen, S. 1998. Histological classification of tumors of the genital system of domestic animals second series, 2nd ed. WashaningtonWashington, DC: Armed Forces Institute of Pathology. Little, S.E. 2012. Chapter 40 - Female Reproduction. Editor(s): Susan E. Little. The Cat, W.B. Saunders, pp: 1195-1227. MacLachalan, N.J. and Kennedy, P.C. 2002. Tumors of the genital systems. In Tumors in domestic animal. Eds., Meuten D.J. Ames, IowaA: Iowa State University Press, pp: 547–573. McEntee, M.C. 2002. Reproductive oncology. Clin. Tech. Small Anim. Pract. 17, 133–149. Norris, H.J., Garner, F. and Taylor, H.B. 1969. Pathology of feline ovarian neoplasms. J. Pathol. ´97, 138–143. Oviedo-Peñata, C.A., Hincapie, L., Riaño-Benavides, C. and Maldonado-Estrada, J.G. 2020. Concomitant presence of ovarian tumors (teratoma and granulosa cell tumor) and pyometra in an english bulldog female dogː a case report. Front. Vet. Sci. 6, 500. Potter, K., Hancock, D. and Gallina, A. 1991. Clinical and pathologic features of endometrial hyperplasia, pyometra, and endometritis in cats: 79 cases (1980-1985). J. Am. Vet. Med. Assoc. 198, 1427–1431. Saba, C.F. and Lawrence, J.A. 2013. Tumors of the female reproductive system. In Withrow and Macewen’s small animal clinical oncology. Eds., Vail, D.M. 5th ed. Saint Louis, MOo: W.B. Saunders, pp: 532–537. Sivacolundhu, R., O’Hara, M. and Read, R. 2001. Granulosa cell tumour in two speyed bitches. Aust. Vet. J. 79, 173–176. Solano-Gallego, L. and Masserdotti, C. 2016. Reproductive system. In Canine and feline cytology. Eds., Raskin, R.E. and Mayer, D.J. St. louisLouis, MO: Elsevier, pp: 313–352. Toledo, G.N., Rolemberg, D.S., Silva, T., Bertolo, P.-H.L., Coutinho, A.R., Moreira, S.H., Pelógia, M.E., Lima, P.R., Pavan, L. and Moraes, P.C. 2021. Mesenteric metastasis of granulosa cell tumor in a bitch: a case report. Rev. Colomb. de Cienc. Pecu. 34, 231–239. | ||
| How to Cite this Article |
| Pubmed Style Elbahi AM, Mahgiubi SA, Gaja AO. Ovarian granulosa cell tumor in a Persian cat: Case report from Tripoli, Libya. Open Vet J. 2022; 12(4): 519-524. doi:10.5455/OVJ.2022.v12.i4.15 Web Style Elbahi AM, Mahgiubi SA, Gaja AO. Ovarian granulosa cell tumor in a Persian cat: Case report from Tripoli, Libya. https://www.openveterinaryjournal.com/?mno=10497 [Access: April 25, 2024]. doi:10.5455/OVJ.2022.v12.i4.15 AMA (American Medical Association) Style Elbahi AM, Mahgiubi SA, Gaja AO. Ovarian granulosa cell tumor in a Persian cat: Case report from Tripoli, Libya. Open Vet J. 2022; 12(4): 519-524. doi:10.5455/OVJ.2022.v12.i4.15 Vancouver/ICMJE Style Elbahi AM, Mahgiubi SA, Gaja AO. Ovarian granulosa cell tumor in a Persian cat: Case report from Tripoli, Libya. Open Vet J. (2022), [cited April 25, 2024]; 12(4): 519-524. doi:10.5455/OVJ.2022.v12.i4.15 Harvard Style Elbahi, A. M., Mahgiubi, . S. A. & Gaja, . A. O. (2022) Ovarian granulosa cell tumor in a Persian cat: Case report from Tripoli, Libya. Open Vet J, 12 (4), 519-524. doi:10.5455/OVJ.2022.v12.i4.15 Turabian Style Elbahi, Asma Mohamed, Shaima A.M Mahgiubi, and Abdurraouf Omar Gaja. 2022. Ovarian granulosa cell tumor in a Persian cat: Case report from Tripoli, Libya. Open Veterinary Journal, 12 (4), 519-524. doi:10.5455/OVJ.2022.v12.i4.15 Chicago Style Elbahi, Asma Mohamed, Shaima A.M Mahgiubi, and Abdurraouf Omar Gaja. "Ovarian granulosa cell tumor in a Persian cat: Case report from Tripoli, Libya." Open Veterinary Journal 12 (2022), 519-524. doi:10.5455/OVJ.2022.v12.i4.15 MLA (The Modern Language Association) Style Elbahi, Asma Mohamed, Shaima A.M Mahgiubi, and Abdurraouf Omar Gaja. "Ovarian granulosa cell tumor in a Persian cat: Case report from Tripoli, Libya." Open Veterinary Journal 12.4 (2022), 519-524. Print. doi:10.5455/OVJ.2022.v12.i4.15 APA (American Psychological Association) Style Elbahi, A. M., Mahgiubi, . S. A. & Gaja, . A. O. (2022) Ovarian granulosa cell tumor in a Persian cat: Case report from Tripoli, Libya. Open Veterinary Journal, 12 (4), 519-524. doi:10.5455/OVJ.2022.v12.i4.15 |