| Original Article | ||
Open Vet J. 2022; 12(6): 956-964 Open Veterinary Journal, (2022), Vol. 12(6): 956–964 Original Research Vitamin D inhibited endometriosis development in mice model through interleukin-17 modulationAlfi Ruham Burjiah1, Ashon Sa’adi2, and Widjiati Widjiati3*1Postgraduate Reproductive Health Science Study Program, Faculty of Medicine, Universitas Airlangga, Surabaya, Indonesia 2Department of Obstetrics and Gynecology, Faculty of Medicine, Universitas Airlangga, Surabaya, Indonesia 3Department of Veterinary Science, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia Submitted: 22/09/2022 Accepted: 11/11/2022 Published: 09/12/2022 *Corresponding Author: Widjiati Widjiati. Department of Veterinary Science, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: widjiati [at] fkh.unair.ac.id © 2022 Open Veterinary Journal
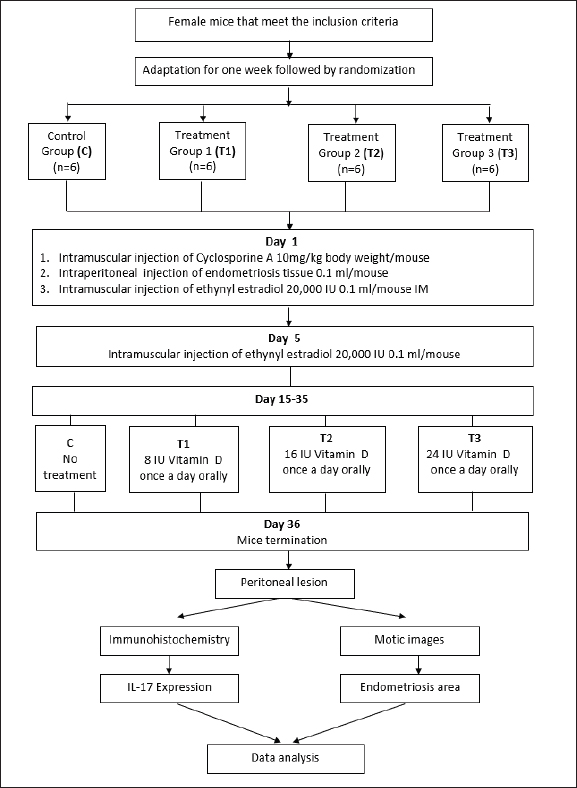
AbstractBackground: Endometriosis is a common, benign, estrogen-dependent, and chronic gynecological disease. Immune system disturbance and inflammatory abnormalities were involved in the pathogenesis of endometriosis. Therefore, it is logical to use vitamin D, which has an immunomodulatory capacity, as supportive therapy for endometriosis. Aim: This research aimed to study the effect of different doses of vitamin D on Interleukin-17 (IL-17) expression in endometriosis mice models. Methods: Endometriosis was induced in 24 mice divided into 4 groups of 6. Group C received no treatment, while groups T1, T2, and T3 received graded doses of oral vitamin D, sequentially 8, 16, and 24 IU, for 3 weeks. IL-17 expression and the extent of endometriotic peritoneal lesions were then measured and analyzed. Statistical tests were performed to see the difference in the mean area of endometriosis lesions and IL-17 expression between the control and treatment groups, as well as the correlation between the extent of endometriosis lesions and IL-17. Results: Endometriosis lesions decreased after 16 and 24 IU of vitamin D administration (p 0.023 and 0.009). Endometriosis lesion also tends to be smaller after 8 IU of vitamin D supplementation, although insignificant (p > 0.05). IL-17 expression was significantly lower after 24 IU vitamin D administration compared to the untreated group (p=0.004). Lower IL-17 expressions were also observed after 8 and 16 IU vitamin D administration, although insignificant (p=0.452 and p=0.645). IL-17 expression was moderately and positively correlated with the extent of endometriosis lesions (p=0.012, rho=0.505). Conclusion: By modulating the expression of IL-17 in endometriotic lesions, vitamin D inhibited the development of endometriotic lesions in the endometriosis mice model. The recommended vitamin D dose in this study was 24 IU. Keywords: Endometriosis lesion, Interleukin-17, Reproductive, Vitamin D. IntroductionEndometriosis is a relatively common benign, estrogen-dependent, and chronic gynecological disorder (Giudice and Kao, 2004; Zondervan et al., 2020). The prevalence ranges from 10% in the reproductive-age female to 50% in infertile women (Hendarto, 2015; Zondervan et al., 2020). Endometriosis clinical manifestations include dysmenorrhea, dyspareunia, chronic pain, and infertility. This disease affects the body and mind, causing depression, anxiety, and impaired social relationships. Endometriosis also reduces work productivity due to fatigue, psychological burden, and excessive health resource usage (Della Corte et al., 2020; Mińko et al., 2021; Eisenberg et al., 2022). Eisenberg et al. (2022) stated that endometriosis patients were 1.6 times more likely to see a gynecologist and twice as likely to be hospitalized annually. Endometriosis patients also significantly used more analgesics, particularly narcotic-like medications (Eisenberg et al., 2022). Endometriosis was also associated with higher total healthcare costs (Della Corte et al., 2020; Eisenberg et al., 2022). Overall, endometriosis reduces the quality of life and burdens patients, their partners, and their families (Nnoaham et al., 2011; Della Corte et al., 2020). Endometrial cells can implant and grow in ectopic locations due to retrograde menstruation, hormonal factors, immune system abnormalities, and chronic inflammation (Parasar et al., 2017). Interleukin-17 (IL-17) is one of the cytokines that contribute to the development of endometrial lesions and is associated with the severity of endometriosis (Izumi et al., 2018). IL-17 has several known roles in endometriosis, including macrophage chemotaxis, macrophage polarization to a pathogenic M2 phenotype, and inflammatory factor stimulation (Hirata et al., 2008; Kuwabara et al., 2017; Miller et al., 2020). It can be concluded that IL-17 plays an essential role in the development of the disease. Therefore, therapy that regulates the action of IL-17 has the potential to inhibit the development of endometriosis. Endometriosis is a chronic condition with a high recurrence rate (Brosens et al., 2013). Ideas for alternative and supportive therapies are being developed to overcome therapeutic limitations (Bedaiwy et al., 2017). Because endometriosis patient has immune system disorders and inflammatory abnormalities, it is logical to use Vitamin D, which has immunomodulatory property, as supportive therapy. Vitamin D deficiency is linked with endometriosis and is inversely related to the extent and severity of endometriosis (Miyashita et al., 2016; Anastasi et al., 2017; Ciavattini et al., 2017). Studies showed that Vitamin D had anti-mitogenic effects, inhibited angiogenesis through regulation of Vascular Endothelial Growth Factor (VEGF) expression, and decreased ectopic endometrial cell invasion (Abbas et al., 2013; Yildirim et al., 2014; Delbandi et al., 2016; Miyashita et al., 2016; Cermisoni et al., 2018). It is thought that vitamin D can modulate endometriosis through other modulating pathways. Vitamin D has been reported to decrease the number of IL-17-producing cells, inhibit the differentiation of CD4+ T cells into Th17 cells, and repress IL-17 expression (Bruce et al., 2011; Joshi et al., 2011). Although vitamin D is known to improve the profile of various autoimmune diseases, the effect of vitamin D on IL-17 expression in endometriosis has not been researched yet (Bruce et al., 2011; Joshi et al., 2011; Bhargava et al., 2015; Aly et al., 2017; Xia et al., 2019; Ghorbani et al., 2021; Mahmoud et al., 2021). Based on those studies, it can be extrapolated that Vitamin D can regulate IL-17 expression in endometriosis. Therefore, this study proposed the regulatory pathway of endometriosis by modulation of IL-17 expression. So far, there have been pitfalls in translating in vitro and in vivo research into applied clinical research. These flaws include the inappropriate way of administering vitamin D when translated to human subjects and the use of supraphysiological doses. Therefore, the design of this research also focused on overcoming these weaknesses. This research used vitamin D administered as physiologically as possible via the oral route. Although the optimal dose for immune system modulation in endometriosis is still unknown, vitamin D deficiency is related to endometriosis (Anastasi et al., 2017; Ciavattini et al., 2017; Delbandi et al., 2021). Thus, this study estimated that the effective dose modulating the expression of IL-17 in endometriosis refers to the deficiency correction dose recommended by the Endocrine Society (Holick et al., 2011). These served as the foundation for using graded vitamin D doses to explain their effects on the expression of IL-17, the extent of endometriosis lesions, and the relationship between these variables. Materials and MethodsResearch animalsThis study was conducted in the Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia, with 24 female Mus musculus. This research used 3-month old mice weighing between 20 and 25 g. They were divided into four groups, each consisting of six mice. The animals were fed ad libitum and caged in a controlled environment. Endometriosis model inductionHuman endometrial tissue was injected into the intraperitoneal cavity of mice to induce endometriosis. Cyclosporine A 10 mg/kg body weight was administered intramuscularly to mimic the impaired immune clearance condition. Estrogen 20,000 IU was injected intramuscularly on the first and fifth days to create an estrogen-dependent condition in endometriosis. On the 14th day, the endometriosis model emerged. Research design and procedureThe mice were randomized into four groups of six mice. Group Control (C) served as a group of untreated endometriosis mice. The treatment (T) groups T1, T2, and T3 were endometriosis mice receiving 8, 16, and 24 IU oral vitamin D via a gastric probe. The treatment began on the 15th day of the study and lasted for 3 weeks. The peritoneum was isolated after the mice were sacrificed to examine the lesion area and the expression of IL-17. The research flowchart guided the study’s procedures (Fig. 1). Endometriosis lesion measurementEndometriosis lesions were obtained by performing abdominal surgery on mice and opening the peritoneum. The goal was to measure the endometriosis lesion using a cross-section of the peritoneum. The Motic image program was then used to include all lesions in the form of white/brown cysts on the peritoneum. IL-17 immunohistochemistry procedureEndometriosis lesion samples were fixed in 10% formalin and made prior to immunohistochemical examination for 2 hours minimum. The tissues were dehydrated by placing them in cassettes and soaking them in 70%, 80%, and 96% alcohol. The tissue was cleared three times for 60 minutes each with xylol. The tissue was then immersed in liquid paraffin with a melting point of 58°C at 45°C for 24 hours in an incubator. Afterward, the tissue paraffin blocks were cut with a microtome to a thickness of 5–7 m. The cut tissue was placed on a poly L-lysine slide and incubated overnight at 37°C. The slides that had been incubated were mounted with 5% gelatin. The glass object was submerged in xylol four times for 5 minutes each time. The slides were rehydrated with alcohol (70%, 80%, and 96%) and rinsed for 5 minutes with distilled water. The slides were then washed twice with PBS for 5 minutes. After 15 minutes of dripping with 0.3% H2O2, the slides were washed with distilled water for 5 minutes. The slides were washed twice in PBS for 5 minutes each, dripped with blocking serum, and drained. Afterward, the slides were dripped with the prepared IL-17 antibody and incubated overnight at 4°C. Afterward, they were washed twice with PBS for 5 minutes each time. Secondary antibodies were placed on slides and incubated at room temperature for 1 hour. The slides were then treated with chromogen and left at room temperature for 10 minutes. Slides were washed for 5 minutes with running water before being counterstained if necessary. A light microscope was used to examine the slides, and a DSLR camera was used to record observations. Measurements were taken using an immunoreactive scoring score calculated by multiplying the percentage score of immunoreactive cells by the color intensity score. The data for each sample were the mean value of the immunoreactive score observed in 10 different fields of view at 100× and 400× magnification.
Fig. 1. Research design flow chart.
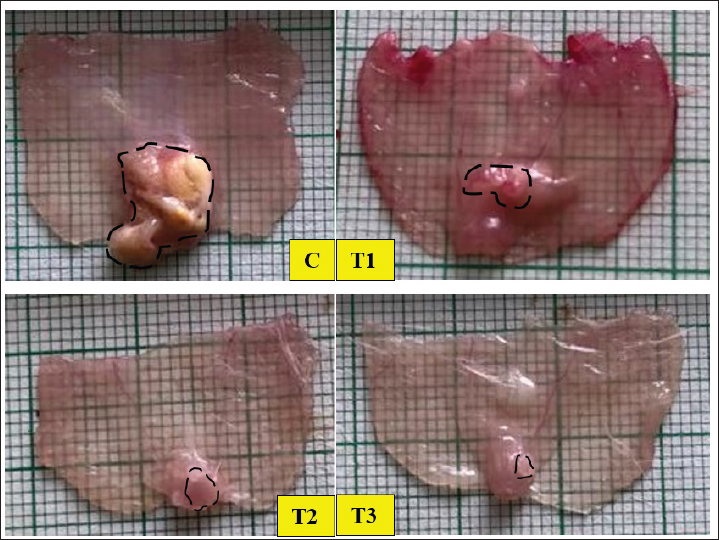
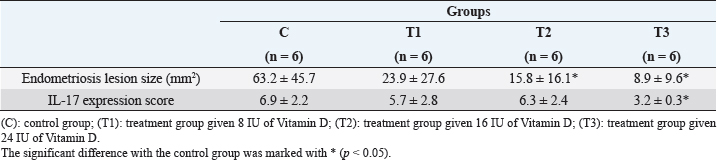
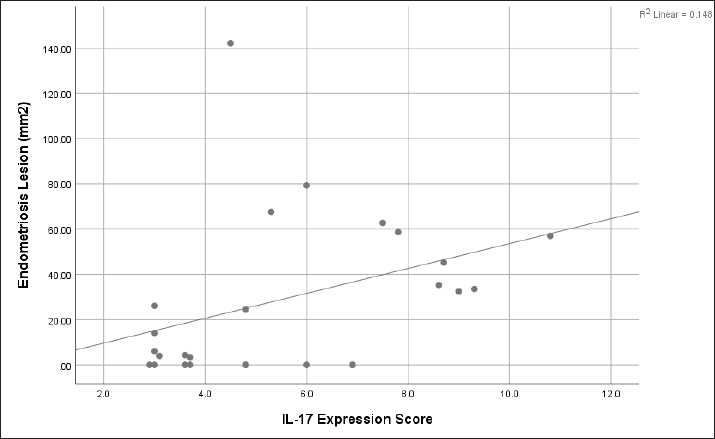
Fig. 2. Cross-section of the peritoneum and endometriotic lesions in endometriosis models of mice in groups C, T1, T2, and T3 at termination. The black dotted line indicates an endometriosis lesion. C: control group; T1: treatment group given 8 IU of Vitamin D; T2: treatment group given 16 IU of Vitamin D; T3: treatment group given 24 IU of Vitamin D. Ethical approvalThe Animal Care and Use Committee, Faculty of Veterinary Medicine, Universitas Airlangga, had ethically approved this study (number 2.KE.143.12.2021). All procedures followed the research policy on Animal Ethics. ResultsEndometriosis lesionEndometriosis lesions were observed in the peritoneal cross-section slide prepared from mice. Figure 2 shows endometriosis lesions from each group. The average area of endometriosis lesions is shown in Table 1. The mean area of endometriosis in the treatment groups T1 (23.9 ± 27.6 mm2), T2 (15.8 ± 16.1 mm2), and T3 (8.9 ± 9.6 mm2) was lower compared to the untreated group (63.2 ± 45.7 mm2). Analysis of variances test showed p: 0.008, meaning a significant difference between the control and treatment groups. Tukey’s HSD post-hoc test showed significant differences between T2 and T3 compared to the C group (p: 0.023 and p: 0.009). IL-17 expressionThe image of endometriosis cells expressing IL-17 is shown in Figure 3 and is indicated by red arrows at 400× magnification. IL-17 was expressed in all mice group slides, with the strongest expression in group C and the weakest in group T3. Although they were weaker than the control group, the T2 group slides provided a stronger expression picture than the other treatment groups. The average score of IL-17 expression is shown in Table 1. The mean IL-17 expression scores in group T1 (5.7 ± 2.8), T2 (6.3 ± 2.4), and T3 (3.2 ± 0.3) are lower than the group C (6.9 ± 2.2). Kruskal–Wallis test showed a significant difference between the treatment and control group (p: 0.029). Mann–Whitney test showed a significant difference in the P3 group (p: 0.004) but not in P1 and P2 (p: 0.452 and p: 0.645). Relationship between IL-17 expression and endometriosis lesion areaThe Spearman correlation test examined the relationship between the IL-17 expression and the extent of endometriosis lesions. The scatter plot image in Figure 4 shows the linearity between variables. The test yielded a p-value of 0.012 and a correlation coefficient of 0.505. Table 1. The mean and standard deviation of endometriosis lesion area and IL-17 expression in endometriosis mice model in the control and vitamin D treatment groups.
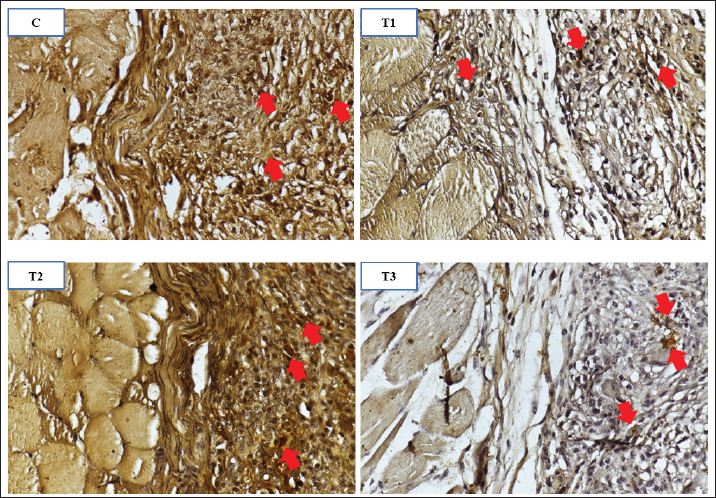
Fig. 3. Comparison of IL-17 expression in endometriosis cells in each treatment group. Red arrows indicate the presence of IL-17 expression in endometriosis cells. C: control group; T1: treatment group given 8 IU of Vitamin D; T2: treatment group given 16 IU of Vitamin D; T3: treatment group given 24 IU of Vitamin D. DiscussionThis research evaluated the effect of vitamin D on the endometriosis mice model. The extent of endometriosis area and IL-17 expression was assessed. We divided 24 mice equally into 4 groups, 3 treatment groups, and 1 control group. The treatment groups were given graded doses of 8, 16, and 24 IU. The results indicated that 16 and 24 IU vitamin D decreased endometriosis lesions in mice. A smaller area of endometriosis in mice given 8 IU of vitamin D versus those not treated was also observed, though they were insignificant. IL-17 expression is significantly lower in the endometriosis mice model given 24 IU of vitamin D versus the untreated group. The IL-17 expression also showed decreased tendencies with the administration of 8 and 16 IU of vitamin D, although insignificant. This study also observed a positive correlation between IL-17 expression and the extent of endometriosis lesions in the endometriosis mice model.
Fig. 4. The scatter-plot graph showing the correlation between IL-17 expression score and endometriosis lesions. The heterologous endometriosis induction method was used in this study, which involved injecting human endometrial tissue into the intraperitoneal cavity of mice. This study’s model was a mild endometriosis type. Lesions caused by this method are typically white or red to brownish cystic lesions or dark solid peritoneal tumors. This method excels at studying disease in its early stages, such as angiogenesis, apoptotic defects, endometrial proliferation, and inflammation. This method is similar to the pathophysiology of retrograde menstruation in humans. Another advantage is that because this heterologous model is based on human endometrial tissue, its development is thought to mimic human endometriosis lesions (Taniguchi et al., 2021). Because it depends on the amount of tissue and the size of the injected endometrial fragments, this method had limitations regarding the number, distribution, and phenotype of endometrial lesions (Burns et al., 2022). Another disadvantage is that mice, as a nonmenstruating species, may not accurately reflect all aspects of endometriosis pathophysiology (Taniguchi et al., 2021). This study demonstrates that the untreated group of endometriosis mice model had the largest endometriosis lesion area. These data are consistent with the theory and other studies mentioned before. The extent of endometriotic lesions reflects the severity of endometriosis according to the ASRM category. Endometriotic lesions in the peritoneal cavity implant are closely associated with various immune cells (Izumi et al., 2018). The recruitment of immune cells, cell adhesion, and an increase in the inflammatory process promote endometrial cell implantation and survival (Agostinis et al., 2021). The ability of ectopic endometrial cells to invade the basement membrane, as well as angiogenesis originating from nearby vascularization or incorporation of circulating endothelial precursor cells near the vascularization site, all influence the development of endometrial lesions (Burney and Giudice, 2012; Capobianco and Rovere-Querini, 2013). Endometrial lesions have thus been linked to uncontrolled growth, invasion of surrounding tissues, defective apoptosis, neoangiogenesis, and a persistent inflammatory response. The vitamin D-treated endometriosis mice model consistently had a smaller area of endometriosis lesions. The smallest to largest mean lesion area in the endometriosis mice model given vitamin D was obtained at doses of 24, 16, and 8 IU, respectively. According to the analysis, the data were statistically significant at 16 and 24 IU doses. In this study, IL-17 expression was highest in untreated endometriosis models. This result is consistent with other research that has found an increase in the number of Th-17 cells and IL-17 levels in endometriosis (Hirata et al., 2008; Ahn et al., 2015; Miller et al., 2020). IL-17 has been linked to many autoimmune disorders and chronic inflammatory conditions such as endometriosis (Kuwabara et al., 2017; Miller et al., 2020). This cytokine is made up of six homologous molecules known as IL-17A–IL-17F. The IL-17 receptor (IL-17R) is made up of five homologous sequences (IL-17Ra, IL-17RB, IL-17RC, IL-17RD, and IL-17RE) (Kuwabara et al., 2017). Chemotactic, macrophage polarization induction, cytokine stimulation, angiogenic factor stimulation, COX2 expression induction, and endometriotic stromal cell proliferation are all roles of IL-17A in the pathogenesis of endometriosis (Hirata et al., 2008; Ahn et al., 2015; Miller et al., 2020). Early disease onset is linked to endometriosis progression via macrophage recruitment and polarization and increased cytokine production (Miller et al., 2020). Thus, IL-17 is linked to impaired immune clearance, endometrial cell adhesion at ectopic sites, and invasion of these cells into tissues at ectopic sites. These processes allow endometrial cells at ectopic sites to develop into endometrial lesions. The administration of vitamin D in this study’s treatment group reduced the expression of IL-17 compared to the control group. The trend of decreasing IL-17 expression was consistent across all vitamin D groups. Statistical analysis revealed a significant difference in IL-17 expression between the endometriosis mice model given vitamin D at a dose of 24 IU and mice not given treatment. At serum vitamin D levels greater than 20 ng/ml, vitamin D supplementation is known to regulate IL-17 and improve the profile of various autoimmune diseases (Bruce et al., 2011; Joshi et al., 2011; Bhargava et al., 2015; Aly et al., 2017; Xia et al., 2019; Ghorbani et al., 2021; Mahmoud et al., 2021). Roffe-Vazquez et al. (2019) discovered a negative relationship between serum vitamin D levels and IL-17A. This happened in long-term systemic inflammatory conditions and was accompanied by vitamin D deficiency in adult heart failure patients (Roffe-Vazquez et al., 2019). Vitamin D immunomodulation of IL-17 can occur through a decrease in the number of IL-17-producing cells, inhibition of CD4+ T cell differentiation into Th17 cells, and a repressive effect on IL-17 expression (Bruce et al., 2011; Joshi et al., 2011). The repressive effect is caused by the vitamin D receptor’s transcription suppression mechanism, which inhibits the Il-17/Il-17R pathway (Joshi et al., 2011; Xia et al., 2019). IL-17 is one factor contributing to the overall development of endometrial lesions. This study showed a moderate relationship between IL-17 and the severity of endometriosis lesions. In this study, administering 24 IU of Vitamin D resulted in the lowest IL-17 expression and the smallest endometriosis lesion area. The study demonstrated that vitamin D inhibits the development of endometriosis lesions via IL-17 expression regulation. Other studies have shown that endometrial lesions can be prevented from spreading through other pathways. Miyashita et al. (2016) observed that vitamin D inhibits NF-kB activation, thereby reducing the number and DNA synthesis of ectopic endometrial cells (Miyashita et al., 2016). Vitamin D administration inhibits VEGF-A gene expression, VEGF, and MMP-9 expression in the ectopic endometrial stroma, thereby inhibiting angiogenic capacity and endometrial lesion growth (Yildirim et al., 2014; Delbandi et al., 2016). Delbandi et al. (2016) also demonstrated vitamin D’s benefits in slowing the invasion and proliferation of ectopic endometrial stromal cells. Vitamin D reduces the severity of endometrial lesions by modulating angiogenesis through decreased VEGF expression and MMP-9 activity (Abbas et al., 2013). Vitamin D modulates endometrial cell adhesion, the number of peritoneal macrophages, cytokine and angiogenic factor secretion, extracellular matrix degradation, and endometrial cell invasion in ectopic sites via its receptors on immune cells (Abbas et al., 2013; Yildirim et al., 2014). Yildirim et al. (2014) showed that vitamin D could induce fibrosis and apoptosis in endometrial tissue, resulting in the regression of endometriosis implants. This study showed a relationship between IL-17 expression and the extent of endometriosis lesions. Statistically, the correlation was significant with a moderate positive direction. A decrease in IL-17 was thus associated with reducing endometrial lesions and vice versa. The pathway for broad regulation of endometrial lesions through the modulation of IL-17 by vitamin D was thus evident in this study. This study used a vitamin D dose based on the per-oral correction of vitamin D deficiency conditions. The findings of this study are intriguing and have the potential to be translated into clinical research. This is due to the method of administration being similar to the recommended vitamin D deficiency correction and supplementation method in humans. More clinical research should be conducted to increase the potential of Vitamin D as an adjuvant supportive therapy for endometriosis. There were several limitations to this study. When mice were fed ad libitum, the condition of their gastrointestinal tract was unknown until vitamin D was administered. When taken orally, vitamin D is better absorbed from the gastrointestinal tract when combined with fat-containing foods. With a fat/oil-based carrier, vitamin D can be absorbed even if the gastrointestinal tract is empty, although not optimal (Silva and Furlanetto, 2018). This condition could potentially cause the vitamin D given in this study to be poorly absorbed. The following limitation was that vitamin D levels before and after treatment were not assessed. The assessment of vitamin D levels before treatment could serve as baseline data. Evaluating vitamin D levels after treatment could be used to determine post-supplementation levels. Genetic polymorphisms associated with endogenous synthesis or vitamin D metabolism in mice were unknown. These factors may contribute to differences in the vitamin D status of mice in this study. This study also did not evaluate estrogen levels in mice, although vitamin D could influence estrogen production. ConclusionCompared to the endometriosis mice model that was not given vitamin D, vitamin D significantly decreased IL-17 expression. In this study, the effective oral dose of vitamin D for regulating IL-17 expression was 24 IU. Vitamin D administration was associated with smaller endometriosis lesions. In this study, the effective oral doses of vitamin D for controlling the area of endometriosis lesions were 16 and 24 IU. The IL-17 expression positively correlated with the extent of endometriosis lesions. Thus, the researcher concluded that vitamin D administration could inhibit the development of endometrial lesions by modulating the expression of IL-17 in peritoneal endometriosis lesions of mouse endometriosis models. Human clinical trials are needed to investigate the potential role of vitamin D as adjuvant supportive therapy in endometriosis. This study is significant because it adds to our understanding of the modulating mechanism of vitamin D in endometriosis and serves as a translational basis for vitamin D as supportive therapy for endometriosis in humans. Conflict of interestAll authors declare that there is no conflict of interest. FundingThe authors received no financial support for this article’s research, authorship, or publication. Authors’ contributionA.R.B., W.W., and A.S. designed this research. A.R.B., W.W., and A.S. conducted a survey and took samples at the samples field. All authors examined samples in the research laboratory. All authors compiled, read, revised, and approved the final manuscript. ReferencesAbbas, M.A., Taha, M.O., Disi, A.M. and Shomaf, M. 2013. Regression of endometrial implants treated with vitamin D3 in a rat model of endometriosis. Eur. J. Pharmacol. 715, 72–75. Agostinis, C., Balduit, A., Mangogna, A., Zito, G., Romano, F., Ricci, G., Kishore, U. and Bulla, R. 2021. Immunological basis of the endometriosis: the complement system as a potential therapeutic target. Front. Immunol. 11, 599117. Ahn, S.H., Edwards, A.K., Singh, S.S., Young, S.L., Lessey, B.A. and Tayade, C. 2015. IL-17A contributes to the pathogenesis of endometriosis by triggering proinflammatory cytokines and angiogenic growth factors. J. Immunol. 195, 2591–2600. Aly, D., Mohammed, F., Sayed, K., Gawdat, H., Mashaly, H., Abdel Hay, R., Elias, T. and Agaiby, M. 2017. Is there a relation between vitamin D and interleukin-17 in vitiligo? A cross-sectional study. Dermatology 233, 413–418. Anastasi, E., Fuggetta, E., De Vito, C., Migliara, G., Viggiani, V., Manganaro, L., Granato, T., Benedetti Panici, P., Angeloni, A. and Porpora, M.G. 2017. Low levels of 25-OH vitamin D in women with endometriosis and associated pelvic pain. Clin. Chem. Lab. Med. 55(12), e282–e284. Bedaiwy, M.A., Alfaraj, S., Yong, P. and Casper, R. 2017. New developments in the medical treatment of endometriosis. Fertil. Steril. 107, 555–565. Bhargava, P., Sotirchos, E., Eckstein, C., Ntranos, A., Gocke, A., Mowry, E. and Calabresi, P. 2015. High-dose vitamin D supplementation reduces IL-17-producing CD4+ T-cells and effector-memory CD4+ T-cells in multiple sclerosis patients (S38.001). Neurology 84, S38.001. Brosens, I., Gordts, S. and Benagiano, G. 2013. Endometriosis in adolescents is a hidden, progressive and severe disease that deserves attention, not just compassion. Hum. Reprod. 28, 2026–2031. Bruce, D., Yu, S., Ooi, J.H. and Cantorna, M.T. 2011. Converging pathways lead to overproduction of IL-17 in the absence of vitamin D signaling. Int. Immunol. 23, 519–528. Burney, R.O. and Giudice, L.C. 2012. Pathogenesis and pathophysiology of endometriosis. Fertil. Steril. 98, 511–519. Burns, K.A., Pearson, A.M., Slack, J.L., Por, E.D., Scribner, A.N., Eti, N.A. and Burney, R.O. 2022. Endometriosis in the mouse: challenges and progress toward a ‘Best Fit’ Murine Model. Front. Physiol. 12, 806574. Capobianco, A. and Rovere-Querini, P. 2013. Endometriosis, a disease of the macrophage. Front. Immunol. 4, 9. Cermisoni, G.C., Alteri, A., Corti, L., Rabellotti, E., Papaleo, E., Viganò, P. and Sanchez, A.M. 2018. Vitamin D and endometrium: a systematic review of a neglected area of research. Int. J. Mol. Sci. 19, 2320. Ciavattini, A., Serri, M., Delli Carpini, G., Morini, S. and Clemente, N. 2017. Ovarian endometriosis and vitamin D serum levels. Gynecol. Endocrinol. 33, 164–167. Delbandi, A.-A., Mahmoudi, M., Shervin, A. and Zarnani, A.H. 2016. 1,25-Dihydroxy vitamin D3 modulates endometriosis-related features of human endometriotic stromal cells. Am. J. Reprod. Immunol. 75, 461–473. Delbandi, A.A., Torab, M., Abdollahi, E., Khodaverdi, S., Rokhgireh, S., Moradi, Z., Heidari, S. and Mohammadi, T. 2021. Vitamin D deficiency as a risk factor for endometriosis in Iranian women. J. Reprod. Immunol. 143, 103266. Della Corte, L., Di Filippo, C., Gabrielli, O., Reppuccia, S., La Rosa, V.L., Ragusa, R., Fichera, M., Commodari, E., Bifulco, G. and Giampaolino, P. 2020. The burden of endometriosis on women’s lifespan: a narrative overview on quality of life and psychosocial wellbeing. Int. J. Environ. Res. Public Health 17, 4683. Eisenberg, V.H., Decter, D.H., Chodick, G., Shalev, V. and Weil, C. 2022. Burden of endometriosis: infertility, comorbidities, and healthcare resource utilization. J. Clin. Med. 11, 1133. Ghorbani, Z., Rafiee, P., Haghighi, S., Razeghi Jahromi, S., Djalali, M., Moradi-Tabriz, H., Mahmoudi, M. and Togha, M. 2021. The effects of vitamin D3 supplementation on TGF-β and IL-17 serum levels in migraineurs: post hoc analysis of a randomized clinical trial. J. Pharm. Heal. Care Sci. 7, 9. Giudice, L.C. and Kao, L.C. 2004. Endometriosis. Lancet 364, 1789–1799. Hendarto, H. 2015. Endometriosis dari Aspek Teori Sampai Penanganan Klinis. Surabaya, Indonesia: Airlangga University Press. Hirata, T., Osuga, Y., Hamasaki, K., Yoshino, O., Ito, M., Hasegawa, A., Takemura, Y., Hirota, Y., Nose, E., Morimoto, C., Harada, M., Koga, K., Tajima, T., Saito, S., Yano, T. and Taketani, Y. 2008. Interleukin (IL)-17A stimulates IL-8 secretion, cyclooxygensase-2 expression, and cell proliferation of endometriotic stromal cells. Endocrinology 149, 1260–1267. Holick, M.F., Binkley, N.C., Bischoff-Ferrari, H.A., Gordon, C.M., Hanley, D.A., Heaney, R.P., Murad, M.H. and Weaver, C.M. 2011. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 96, 1911–1930. Izumi, G., Koga, K., Takamura, M., Makabe, T., Satake, E., Takeuchi, A., Taguchi, A., Urata, Y., Fujii, T. and Osuga, Y. 2018. Involvement of immune cells in the pathogenesis of endometriosis. J. Obstet. Gynaecol. Res. 44, 191–198. Joshi, S., Pantalena, L.-C., Liu, X.K., Gaffen, S.L., Liu, H., Rohowsky-Kochan, C., Ichiyama, K., Yoshimura, A., Steinman, L., Christakos, S. and Youssef, S. 2011. 1,25-Dihydroxyvitamin D3 ameliorates Th17 autoimmunity via transcriptional modulation of interleukin-17A. Mol. Cell. Biol. 31, 3653–3669. Kuwabara, T., Ishikawa, F., Kondo, M. and Kakiuchi, T. 2017. The role of IL-17 and related cytokines in inflammatory autoimmune diseases. Mediators Inflamm. 2017, 1–11. Mahmoud, S.B., Anwar, M.K., Shaker, O.G. and El Sharkawy, D.A. 2021. Possible relation between Vitamin D and interleukin-17 in the pathogenesis of lichen planus. Dermatology 237, 896–901. Miller, J.E., Ahn, S.H., Marks, R.M., Monsanto, S.P., Fazleabas, A.T., Koti, M. and Tayade, C. 2020. IL-17A modulates peritoneal macrophage recruitment and M2 polarization in endometriosis. Front. Immunol. 11, 108. Mińko, A., Turoń-Skrzypińska, A., Rył, A., Bargiel, P., Hilicka, Z., Michalczyk, K., Łukowska, P., Rotter, I. and Cymbaluk-Płoska, A. 2021. Endometriosis a multifaceted problem of a modern woman. Int. J. Environ. Res. Public Health 18, 8177. Miyashita, M., Koga, K., Izumi, G., Sue, F., Makabe, T., Taguchi, A., Nagai, M., Urata, Y., Takamura, M., Harada, M., Hirata, T., Hirota, Y., Wada-Hiraike, O., Fujii, T. and Osuga, Y. 2016. Effects of 1,25-Dihydroxy Vitamin D3 on Endometriosis. J. Clin. Endocrinol. Metab. 101, 2371–2379. Nnoaham, K.E., Hummelshoj, L., Webster, P., D’Hooghe, T., de Cicco Nardone, F., de Cicco Nardone, C., Jenkinson, C., Kennedy, S.H. and Zondervan, K.T. 2011. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertil. Steril. 96, 366–373.e8. Parasar, P., Ozcan, P. and Terry, K.L. 2017. Endometriosis: epidemiology, diagnosis and clinical management. Curr. Obstet. Gynecol. Rep. 6, 34–41. Roffe-Vazquez, D.N., Huerta-Delgado, A.S., Castillo, E.C., Villarreal-Calderón, J.R., Gonzalez-Gil, A.M., Enriquez, C., Garcia-Rivas, G. and Elizondo-Montemayor, L. 2019. Correlation of Vitamin D with Inflammatory Cytokines, Atherosclerotic Parameters, and Lifestyle Factors in the Setting of Heart Failure: A 12-Month Follow-Up Study. Int. J. Mol. Sci. 20, 5811. Silva, M.C. and Furlanetto, T.W. 2018. Intestinal absorption of vitamin D: a systematic review. Nutr. Rev. 76, 60–76. Taniguchi, F., Wibisono, H., Khine, Y.M. and Harada, T. 2021. Animal models for research on endometriosis. Front. Biosci. 13, 37. Xia, Y., Chen, H., Xiao, H., Yang, J., Li, Z., Wang, Y., Yang, T. and Wang, B. 2019. Immune regulation mechanism of vitamin D level and IL-17/IL-17R pathway in Crohn’s disease. Exp. Ther. Med. 17(5), 3423–3428. Yildirim, B., Guler, T., Akbulut, M., Oztekin, O. and Sariiz, G. 2014. 1–Alpha, 25–Dihydroxyvitamin D3 regresses endometriotic implants in rats by inhibiting neovascularization and altering regulation of matrix metalloproteinase. Postgrad. Med. 126, 104–110. Zondervan, K.T., Becker, C.M. and Missmer, S.A. 2020. Endometriosis. N. Engl. J. Med. 382, 1244–1256. | ||
| How to Cite this Article |
| Pubmed Style Burjiah AR, Sa'adi A, Widjiati W, . Vitamin D inhibited endometriosis development in mice model through interleukin 17 modulation. Open Vet J. 2022; 12(6): 956-964. doi:10.5455/OVJ.2022.v12.i6.23 Web Style Burjiah AR, Sa'adi A, Widjiati W, . Vitamin D inhibited endometriosis development in mice model through interleukin 17 modulation. https://www.openveterinaryjournal.com/?mno=115890 [Access: September 01, 2024]. doi:10.5455/OVJ.2022.v12.i6.23 AMA (American Medical Association) Style Burjiah AR, Sa'adi A, Widjiati W, . Vitamin D inhibited endometriosis development in mice model through interleukin 17 modulation. Open Vet J. 2022; 12(6): 956-964. doi:10.5455/OVJ.2022.v12.i6.23 Vancouver/ICMJE Style Burjiah AR, Sa'adi A, Widjiati W, . Vitamin D inhibited endometriosis development in mice model through interleukin 17 modulation. Open Vet J. (2022), [cited September 01, 2024]; 12(6): 956-964. doi:10.5455/OVJ.2022.v12.i6.23 Harvard Style Burjiah, A. R., Sa'adi, A., Widjiati, W. & (2022) Vitamin D inhibited endometriosis development in mice model through interleukin 17 modulation. Open Vet J, 12 (6), 956-964. doi:10.5455/OVJ.2022.v12.i6.23 Turabian Style Burjiah, Alfi Ruham, Ashon Sa'adi, Widjiati Widjiati, and . 2022. Vitamin D inhibited endometriosis development in mice model through interleukin 17 modulation. Open Veterinary Journal, 12 (6), 956-964. doi:10.5455/OVJ.2022.v12.i6.23 Chicago Style Burjiah, Alfi Ruham, Ashon Sa'adi, Widjiati Widjiati, and . "Vitamin D inhibited endometriosis development in mice model through interleukin 17 modulation." Open Veterinary Journal 12 (2022), 956-964. doi:10.5455/OVJ.2022.v12.i6.23 MLA (The Modern Language Association) Style Burjiah, Alfi Ruham, Ashon Sa'adi, Widjiati Widjiati, and . "Vitamin D inhibited endometriosis development in mice model through interleukin 17 modulation." Open Veterinary Journal 12.6 (2022), 956-964. Print. doi:10.5455/OVJ.2022.v12.i6.23 APA (American Psychological Association) Style Burjiah, A. R., Sa'adi, A., Widjiati, W. & (2022) Vitamin D inhibited endometriosis development in mice model through interleukin 17 modulation. Open Veterinary Journal, 12 (6), 956-964. doi:10.5455/OVJ.2022.v12.i6.23 |