| Research Article | ||
Open Vet J. 2023; 13(6): 772-781 Open Veterinary Journal, (2023), Vol. 13(6): 772-781 Original Research Influence of spirulina supplementation on growth performance, puberty traits, blood metabolites, testosterone concentrations, and semen quality in Barki male lambsMohamed M. Mansour1,2* and Moustafa M. Zeitoun31Department of Biology, College of Sciences and Arts in Unayzah, Qassim University, Buraydah, Kingdom of Saudi Arabia 2Department of Animal and Poultry Production, Faculty of Agriculture, Damanhour University, Damanhur, Egypt 3Department of Animal and Fish Production, Faculty of Agriculture (El-Shatby), Alexandria University, Alexandria, Egypt *Corresponding Author: Mohamed M. Mansour. Department of Biology, College of Sciences and Arts in Unayzah, Qassim University, Buraydah, Kingdom of Saudi Arabia. Email: m.mnasor [at] qu.edu.sa; mohamedmohsen79 [at] yahoo.com Submitted: 18/02/2023 Accepted: 28/05/2023 Published: 19/06/2023 © 2023 Open Veterinary Journal
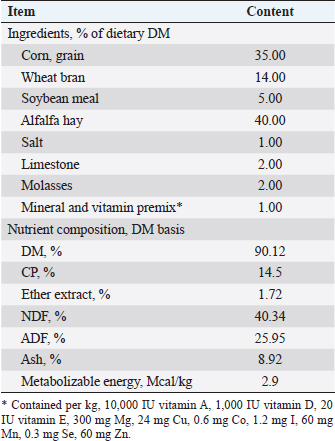
AbstractBackground: The fertility and genetic value of the flock can be enhanced by selecting lambs with highly developed early puberty characteristics. Spirulina (SP) has been evaluated as a natural product supplement to boost lamb growth, immunity, and productivity. Aim: Study growth performance, blood metabolites, puberty development traits, semen quality, and seminal plasma biochemical concentrations of growing Barki lambs when supplemented with SP at different levels. Methods: in a 24 weeks study, 30 Barki male lambs weighing an average of 21.78 ± 2.56 kg, with a body condition score of 3.20 ± 0.55 and an age of about 16 ± 0.24 weeks were used. The lambs were randomly assigned to three groups (10 lambs each) of daily SP supplementation levels per lamb of 0 ml (control), 50 ml (SP1), and 100 ml (SP2). The SP powder was made into a water suspension using SP to water ratio of 1 g:10 ml. The growth characteristics, as well as the development of puberty, blood metabolites, and semen quality analysis of every lamb, were measured. Results: The growth performance was greater (p < 0.05) in SP2 lambs compared with other lambs. While daily dry matter intake was not affected by SP treatment, feed efficiency had significantly improved in SP2 groups. Furthermore, the SP2 lambs have attained puberty at early ages than the control lambs. The testes volume of SP2 lambs was bigger (p < 0.05) than other groups throughout the pre-pubertal up to the puberty stage. The addition of SP had no effects on the total protein, glucose, and triglycerides concentrations. Meanwhile, the cholesterol concentration was lowest (p < 0.05) in the SP2 lambs. The blood and seminal plasma levels of alanine aminotransferase and aspartate aminotransferase decreased (p < 0.05) in the SP lambs more than their control counterparts. The levels of superoxide dismutase reduced glutathione, and total antioxidants had increased (p < 0.05) in the treated lambs compared with the control group. Further, the malondialdehyde levels decreased (p < 0.05) in the SP-treated lambs. Additionally, the SP2 lambs produced better semen quality than the control lambs. Conclusion: SP supplementation (100 ml/head/day) enhanced growth performance, feed efficiency, and antioxidative status, exerting a positive influence on the physiological parameters and sexual behavioral patterns at puberty in Barki lambs. Keywords: Spirulina, Growth performance, Puberty, Semen quality, Barki lambs. IntroductionIn Egypt, three major sheep breeds contribute approximately 6.4% of the total red meat production (Galal et al., 2005). Approximately 8.5% of all Egyptian sheep are Barki sheep (FAO, 2017). Besides being capable of adapting to harsh desert conditions, they also produce substantial amounts of meat, wool, and milk under those conditions (El-Wakil et al., 2008; Abousoliman et al., 2020). The Barki sheep is also a source of income for Egyptian farmers and a source of meat for the locals (Mansour, 2021). Therefore, there is a great deal of interest in understanding how these animals grow and what traits they possess to improve the quality of the meat they produce. It has also been shown that breeding programs that focus on selecting male lambs with advanced puberty characteristics enhance the flock’s fertility and genetic value (Ibarra et al., 2000, Mansour, 2021). As an alga, spirulina (SP) contains large amounts of amino acids, fatty acids, antioxidants, and carotenoids (Holman and Malau-Aduli, 2013; Ranjith et al., 2013). Additionally, SP has antiviral, antimicrobial, and anti-inflammatory properties besides immune function (Wang et al., 2007; Wu et al., 2016; Liang et al., 2020). Further, it does not harm kidneys, livers, or reproductive systems (Gutiérrez-Rebolledo et al., 2015). SP is now used in livestock systems for a variety of purposes (Shields and Lupatsch 2012). Some reports have also found that SP increases sheep productivity (Bezerra et al., 2010; EL-Sabagh et al., 2014; Kashani et al., 2015; Alghonaim et al., 2022). According to Bezerra et al. (2010), lambs fed SP grew faster than lambs not fed SP. SP could also serve as an antioxidant, immune stimulant, and growth promoter in fattening lambs’ diets (EL-Sabagh et al., 2014). Furthermore, using SP as a water suspension in dual-purpose sheep increased polyunsaturated fatty acid levels in lamb (Kashani et al., 2015). Alghonaim et al. 2022 also reported that SP supplementation boosted weight gain, nutrient digestibility, and nitrogen utilization in growing Najdi lambs. Subsequently, it was hypothesized that SP could increase the lamb’s productive performance and subsequently improve puberty characteristics. Thus, the aim of this study was to evaluate the influence of different levels of SP supplementation on growth performance, blood metabolites, puberty development traits, semen quality, and seminal plasma biochemical concentrations in growing Barki lambs. Materials and MethodsAnimal management and experimental designThe present study was performed at the Agriculture Experimental Station, Faculty of Agriculture, Damanhour University, Al-Behera governorate, Egypt. A total of 30 Barki male lambs weighing an average of 21.78 ± 2.56 kg, with a body condition score of 3.20 ± 0.55 and an age of about 16 ± 0.24 weeks were used. All lambs were kept under the same environmental, hygienic, and managerial conditions. All lambs were kept under similar conditions of accommodation systems in shaded areas. Lambs were fed the same diet [2.5% of initial body weight (BW)] according to the National Research Council (2007) recommendations twice daily at 7 AM and 5 PM. The ingredients and chemical composition of the diet are stated in Table 1. The freshwater was available continuously to all lambs during the experimental period. Before starting the experiment, lambs were treated for internal and external parasites, as well as vaccinated against the most common infectious diseases. The lambs were randomly assigned into 3 groups (10 lambs each) according to the treatment types as control and SP groups. The SP powder (TAAU, Darwin, Northern Territory, Australia) was made into a water suspension using SP to water ratio of 1 g:10 ml. This was daily given to the lambs using a sheep drench to directly deliver each lamb assigned SP level of supplementations as control (0 ml), SP1 (50 ml), and SP2 (100 ml). Supplementations were administered directly to each lamb before the morning daily diet. The doses of SP were prepared according to the recommendations by Kashani et al. (2015). The duration of the experiment was 22 weeks, plus an acclimatization period of 2 weeks for adaptation. Table 1. The ingredients and chemical composition of the diet used in the experiment.
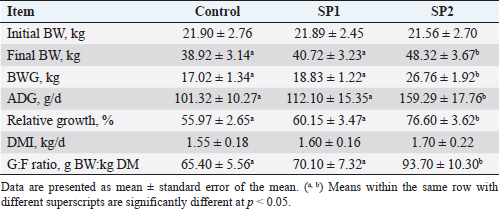
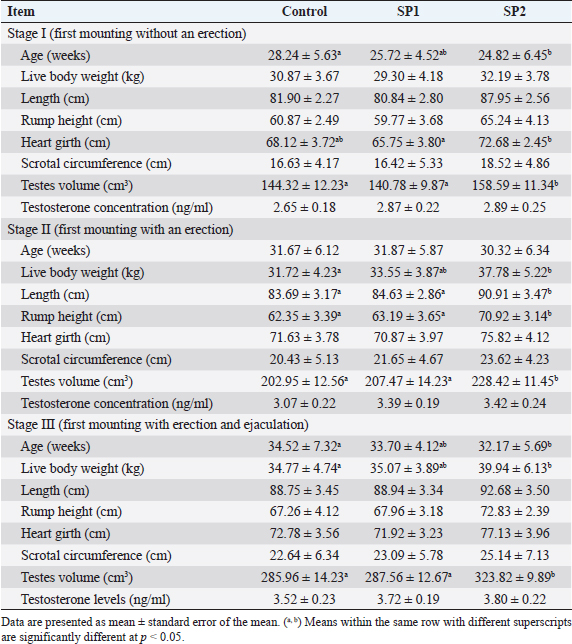
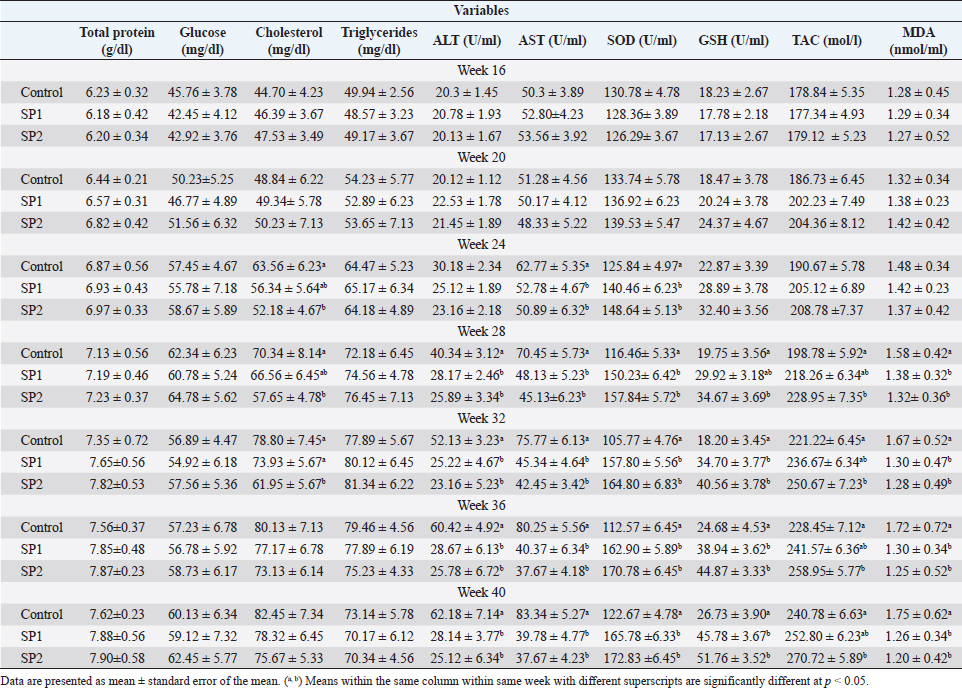
Ethical approvalThe experimental procedures were approved by the University Ethics Monitoring Committee by following the guidelines of the Animal Welfare Committee. Evaluation of the growth performance and feed efficiencyDuring the experimental period, each lamb’s live BW was measured every 15 days before morning feeding by using an electronic lamb scale. The height of the hips, the body length, and the heart circumference were also measured during puberty. The distance between the point of the shoulder and the pin bone was used to measure the length of the body (Afolayan et al., 2006). Using Ibrahim’s (2015) formula, the heart girth was measured by calculating the circumference behind the legs. While the height of the rump was measured according to Afolayan et al. (2006) from the hips to the hoof wall. Daily feed intake in dry matter (DM) was calculated by recording the weight of offered feed and feed refusals for each lamb. DM content was assessed by drying samples in a 100°C oven for 24 hours, while ash content was obtained by incinerating samples in a muffle furnace at 550°C for 3 hours. The AOAC technique (1990) was used to determine the amount of crude protein (CP), neutral detergent fiber (NDF), and acid detergent fiber (ADF), respectively. A BW body weight gain (BWG) per kilogram of DM intake was determined for each lamb’s increase in feed ratio. In addition, the relative growth was calculated according to the following equation: the relative growth=(final BW-initial BW)/initial BW × 100. Assays for pubertyAll animals were witnessed twice weekly to detect puberty improvement from the initiation of the study until the observation of puberty. Sexual behavior for each lamb was noticed as stated by El-Khalifa et al. (2013) technique. In brief, male lambs were watched within 25 minutes using estrus female, considering the following conditions: mounting without an erection (stage I), mounting with first protrusion of the penile (stage II), and puberty as age at the first collection of motile sperm (stage III). Moreover, testicular traits (scrotal circumference, and testes volume) were measured at each stage of puberty. An elastic tape was used to measure the circumference of the scrotum at the point of maximum circumference of the paired testes. Previously described methods were used to assess testes volume (El-Zelaky et al., 2011; Mansour, 2021). Blood samplingThroughout the study (from weeks 16 to 40), blood samples were collected from the jugular vein in a heparinized tube once a week. As soon as blood was collected, plasma was removed from it by centrifuging at 1,500× g for 15 minutes at 4°C, aliquoting, and storing it at −−20°C until it was needed for the examination. Blood biochemical and enzymatic variables assayAt week 16 of the study and every 4 weeks thereafter until the end of the study blood plasma was used to analyze the biochemical traits (total protein, glucose, cholesterol, and triglycerides), and enzymes activities [alanine aminotransferase (ALT), aspartate aminotransferase (AST), Superoxide dismutase (SOD), Reduced glutathione (GSH), total antioxidant capacity (“TAC”), malondialdehyde (“MDA”)]. All the previous traits were analyzed by using commercial kits (Diagnostic Product Company, Los Angeles, CA), and a microplate reader according to the manufacturer’s instructions. Testosterone examinationPlasma testosterone concentrations were determined by Radioimmunoassay (RIA) procedure using a commercial kite (Diagnostic Product Company, Los Angeles, CA). Assay sensitivity was 0.2 ng/ml with a coefficient of variation of <10%. Semen and seminal plasma collecting and evaluationLambs were ejaculated twice weekly for 2 weeks to collect semen samples. Each lamb was sampled after reaching stage III of puberty. Immediately after being transported to the laboratory, the semen ejaculates were immersed in a 37°C water bath to evaluate the quality of the semen. Sperm cell counts, volume, initial motility, live spermatozoa, and abnormal sperm percentage were examined in semen samples. Seminal plasma was collected by centrifugation at 1,500 × ɡ for 15 minutes at 4°C and stored at −20°C until analysis. Seminal plasma metabolites including total protein, fructose, ALT, AST, TAC, MDA, GSH, and SOD were measured using commercial kits (Diagnostic Product Company, Los Angeles, CA), and a microplate reader according to the manufacturer’s instructions. Statistical analysisData were analyzed with two-way ANOVA, using the general linear model procedure adapted of Stat View 5.0 (SAS Institute Inc., Cary, NC) and JMP 6 (SAS Institute Inc., Cary, NC). All values are presented as means ± SEM and differences among means were tested according to the Tukey test. The differences were considered to be statistically significant at p < 0.05. ResultsGrowth performance and feed efficiencyThe final BW, BWG, average daily gain (ADG), and relative growth were greater (p < 0.05) in SP2 lambs compared with the control and SP1 lambs (Table 2). While daily dry matter intake (DMI) was not affected by SP treatment. In contrast, feed efficiency (G: F ratio) had significantly improved in SP2 treatment as compared to SP1 and control groups. Puberty developmentThe progress in the reproductive performance of the lambs prepubertal and up to puberty is illustrated in Table 3. The SP2 lambs had reached stage I of puberty (first mounting without an erection) at early ages (p < 0.05) compared to control lambs. Furthermore, the SP2 lambs have attained stage III of puberty (first mounting with erection and ejaculation) at more early ages (p < 0.05) than control lambs. However, the lambs’ ages at stage II of puberty (first mounting with an erection) were not different. While, at stage II of puberty, the SP2 lambs had the tallest (p < 0.05) body length and rump height compared to SP1 and control lambs, respectively. In addition, the heart girth of SP2 lambs was bigger than SP1 lambs at stage I of puberty (p < 0.05, Table 3). Moreover, the lambs on SP2 treatment gained more weight (p < 0.05) than the control groups at stages II and III of puberty. Likewise, the testes volume of the SP2 groups was bigger (p < 0.05) than the control and SP1 lambs through the prepubertal up to the puberty stage (Table 3). At all stages of puberty, lambs’ scrotal circumferences did not differ significantly. Blood biochemical and enzymatic variablesThe blood plasma levels of biochemical and enzymatic variables measured in the study are shown in Table 4. Within the treated lambs, no differences were found in the total protein, glucose, and triglyceride concentrations between the groups during the study. Meanwhile, the cholesterol concentration was lowest (p < 0.05) in the SP2 lambs compared with control and SP1 lambs during the period from week 24 until week 32 of the experiment. Concerning the liver enzymes, there was a decrease (p < 0.05) in the ALT levels of the treated groups compared with the control lambs starting from week 28 until the end of the trial (week 40). Likewise, from week 24 onwards, SP lambs also had lower AST levels (p < 0.05) than their control counterparts (Table 4). Regarding the antioxidant status, the SOD concentrations were increased (p < 0.05) in the treated lambs with SP compared with the control group starting from week 24 until the end of the trial. Additionally, GSH levels increased (p < 0.05) from week 28 onward when compared with the control lamb. Also, SP2 lambs had higher TAC levels (p < 0.05) than control lambs during the period from weeks 28 to 40 of the study. Further, the MDA levels in the SP-treated lambs decreased from week 28 onward, compared with the control group (Table 4). Table 2. BW, BWG, ADG, DMI, and feed efficiency (G:F ratio) of Barki lambs supplemented with SP levels of 0 ml (control), 50 ml (SP1), and 100 ml (SP2).
Table 3. Puberty characteristics of Barki lambs supplemented with SP levels of 0 ml (control), 50 ml (SP1), and 100 ml (SP2).
Table 4. Blood plasma concentrations of biochemical and enzymatic variables of Barki lambs supplemented with SP levels of 0 ml (control), 50 ml (SP1), and 100 ml (SP2)
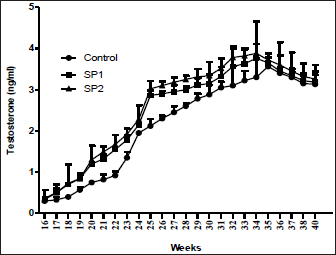
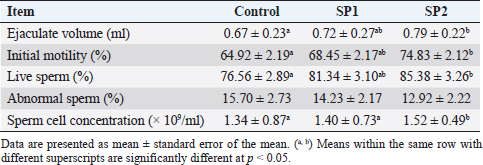
Testosterone concentrationsThe plasma concentrations of testosterone during the experimental period are presented in Figure 1. An increase in linear terms was observed in plasma testosterone levels toward advanced ages in all lambs throughout the pre-pubertal up to the puberty stage. However, testosterone levels were not affected by SP treatment. Furthermore, at all puberty stages, there were no differences in the plasma testosterone concentrations between the lambs’ group (Table 3). Semen propertiesAs stated in Table 5, seminal characteristics (volume, initial motility, live spermatozoa, and sperm cell concentration) at puberty were better (p < 0.05) in SP2 lambs than in control lambs. However, there were no differences between SP1 lambs and their control counterparts regarding the previous seminal properties. In contrast, the sperm cell concentration of SP lambs was higher (p < 0.05) compared with that of control lambs (Table 5).
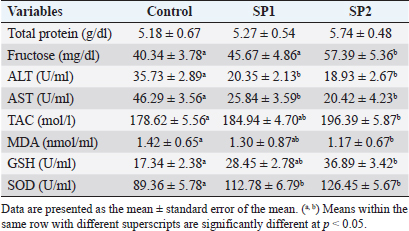
Fig. 1. Plasma testosterone (mean ± standard) concentration (ng/ml) of Barki lambs supplemented with SP levels of 0 ml (control), 50 ml (SP1), and 100 ml (SP2). Seminal plasma biochemical and enzymatic variablesAs shown in Table 6, there was no difference in the total protein concentrations in seminal plasma between the lambs’ groups. In contrast, the fructose level was higher (p < 0.05) in the seminal plasma of SP2 lambs than those of SP1, and control lambs. Furthermore, SP lambs had lower ALT, and AST concentrations in the seminal plasma (p < 0.05) than their control counterparts (Table 6). Moreover, there was an increase (p < 0.05) in the seminal plasma TAC, SOD, and GSH concentration in the treated groups, in particular, SP2 lambs compared with the control lambs. Besides, the seminal plasma level of MDA was lowest (p < 0.05) in the SP2 lambs compared with their counterparts (Table 6). DiscussionIt will be possible to significantly increase lamb production income by selecting lambs that grow faster. In this sense, studies have been conducted to examine SP as a natural product supplement for boosting growth, immunity, and productivity in lambs (Bezerra et al., 2010; EL-Sabagh et al., 2014; Kashani et al., 2015; Alghonaim et al., 2022). Similarly, the current study showed that feed efficiency and growth performance of Barki lambs were improved by SP addition (100 ml/head/day). SP supplementation also significantly improved blood liver enzymes and antioxidant status in the present study. The findings are similar to previous reports that show supplementing growing lambs with SP increases growth, BWG, DMI, and feed conversion ratio (Holman et al., 2012; EL-Sabagh et al., 2014; Alghonaim et al., 2022). Supplementing lambs with SP may lead to superior growth performance because of its high nutritional density and the stimulation of extracellular enzyme secretion by the gut microbiota (Lamminen et al., 2019). A further benefit of SP may be that it contains highly concentrated levels of linoleic and -linolenic acids, which are essential constituents in the synthesis of animal metabolism (Liu et al., 2019). Other factors contributing to the improved performance of growing lambs could be amino acids, polysaccharides, docosahexaenoic acid, and other growth-promoting compounds in SP (Liu et al., 2019). Furthermore, the current study found that SP addition (100 ml/head/day) increased the final lamb’s BW, higher average daily growth rate, and relative growth rate, possibly related to the higher digestibility of DM. It agrees with previous research by Alghonaim et al. (2022), which reported that SP dietary supplementation of 8ppm enhanced weight gain and nutrient digestibility of growing Najdi lambs. Furthermore, previous research on SP has shown that it can enhance rumen microbial CP production by decreasing protein degradation and changing bacterial community composition in steers (Panjaitan et al., 2010). Moreover, Furbeyre et al. (2017) reported higher jejunum villus heights because of higher DM digestibility for pigs that received SP. Besides, the unchanged DMI alongside improvement in ADG can partly explain the enhancement in feed efficiency of lambs fed SP diets. The improvement in growth performance is associated with an increase in nutrient utilization of diet and an improvement in the microbial nitrogen supply and the body nitrogen retention (Abdel-Wahed et al., 2022) Table 5. Semen characteristics of Barki lambs supplemented with SP levels of 0 ml (control), 50 ml (SP1), and 100 ml (SP2).
Table 6. Seminal plasma concentrations of biochemical and enzymatic variables of Barki lambs supplemented with SP levels of 0 ml (control), 50 ml (SP1), and 100 ml (SP2).
The results of the present study show that SP lambs attained puberty earlier than control (without SP) ones by about (2.52 and 3.42 weeks, for SP1 and SP2, respectively). Early puberty occurrence in SP lambs might be related to SP richness in major substances, such as essential amino acids, fatty acids, and carotenoids (Holman and Malau-Aduli, 2013; Ranjith et al., 2013). It has been proven that puberty achievement is linked to nutritional concentrations, in particular the protein percentage, which has a significant influence on the achievement of BW, sexual maturity, and the onset of puberty in males (Foruie et al., 2004). Achieving adequate body mass is also necessary for puberty to begin (Nieto et al., 2013). At puberty, SP2 lambs’ live BW was greater than those of control lambs, according to the current study. It is therefore reasonable to conclude that SP2 lambs’ body amounts improved due to improved growth, feed efficiency, and nutrient digestibility. In this study, the supplement of different levels of SP did not affect the total protein, glucose, and triglycerides concentrations. Besides, both supplemented and control animals fell within the normal range. The results are consistent with previous reports on sheep (EL-Sabagh et al., 2014; Malau-Aduli and Holman, 2015; Lamminen et al., 2019; Alghonaim et al., 2022). Moreover, the present study showed that SP addition (100 ml/head/day) decreased blood cholesterol levels, which might be due to its antioxidant characteristics (Liang et al., 2020). Besides, the present improvement of blood liver enzymes, and antioxidant status in SP lambs, suggested that SP addition could play a vital role in the achievement of BW, sexual maturity, and onset of puberty. Furthermore, SP supplementation is not harmful to the health and production of lambs. The present tendency of plasma testosterone concentrations to rise throughout the pre-pubertal up to puberty stage is harmonious with our previous observation (Mansour, 2021), which indicated that testosterone concentrations were little at the initial stages of puberty and progressively increased with advanced age. Furthermore, it had been demonstrated that testis parameters increased with age until puberty (Mansour, 2021). Likewise, the SP2 lambs had the biggest testes volume during the pre-pubertal up to the puberty stage. Too, age, and BW had an important impact on testicular length, and testicular diameter (Karakus, 2010). However, there were no differences in the plasma testosterone concentrations between the lambs’ group in the current study. Whereas, SP has not been studied enough to provide any evidence regarding its effect on testosterone in growing lambs. In rabbit bucks, blood plasma testosterone increased when received daily drinking water supplemented with 150, 300 mg SP/l for one month before mating of semen collection of artificial insemination (El-Ratel and Gabr, 2020). However, it is important to note that the exact mechanism(s) that account for the potential effects of SP on blood testosterone levels of growing lambs has yet to be resolved. In the present study, the SP2 lambs produced better quality and quantity of semen than the control lambs. Semen characteristics improved with improved performance and increased testis growth, suggesting that the number of spermatozoa per ejaculate has a direct correlation with testis size and testis mass (Bernardini et al., 2011). Furthermore, there is evidence that this effect is associated with the size of the testicle since the seminiferous epithelium and the seminiferous tubules are expanding (El-Khalifa et al., 2013). In addition, SP contains a high protein content of about 60%–70% by dry weight (Hosseini et al., 2013). Likewise, protein supplementation increased performance and enhanced testis growth as well as increased the quality and quantity of semen (Fernández et al., 2004). Further, in the present study, SP addition improved the antioxidant enzymes in the seminal plasma, which may result in improved testicular growth, increased sperm production, and decreased abnormal spermatozoa. Besides, enhanced sperm quality among the SP groups may be due to antioxidant components of SP that activate the antioxidant defenses in sperm cells, preventing cell damage. Likewise, the same trends were also demonstrated in rabbits (Fouda and Ismail, 2017, El-Ratel and Gabr, 2020). Furthermore, it was found that buck rabbits who were given drinking water containing SP had reduced AST and ALT aminotransferase activities in the seminal plasma (El-Ratel and Gabr, 2020), leading to higher semen quality. Besides, the decrease in AST and ALT activity was associated with a reduction in sperm viability and cell concentration (Al-Daraji et al., 2001). Furthermore, supplementation of semen extender with SP had a beneficial effect on sperm motility, speed parameters, and antioxidant activity of Arabian stallion spermatozoa, particularly after cryopreservation in a dose-dependent manner (Zeitoun et al., 2022). These positive effects seemed to be due to the improvement of the antioxidant activities and the diminishing rates of lipid peroxidation of the cryopreserved spermatozoa (Zeitoun et al., 2022). Moreover, in the present study, SP supplementation increased the seminal plasma fructose concentration, which is agreeable with El-Ratel and Gabr (2020), suggesting that fructose levels in seminal plasma could have a positive correlation with most sperm characteristics. Besides, the beneficial outcomes of SP can be attributed to its content of significant mixtures such as protein and essential amino acids (Farag et al., 2016), essential fatty acids, alpha-linolenic acid, gamma-linolenic acid, and sub- Oleic acid (Mendes et al., 2003), nutrient pigments (Keservani et al., 2015), vitamins such as thiamine, niacinamide, riboflavin, folic acid, pyridoxine, vitamins A, D and E (Hosseini et al., 2013) and minerals such as calcium, potassium, chromium, copper, manganese, iron, phosphorus, magnesium, sodium, zinc, and selenium (Babadzhanov et al., 2004). ConclusionSP addition (100 ml/head/day) enhanced the growth performance and feed efficiency of growing Barki lambs. Moreover, SP supplementation at 100 ml/head/day improved puberty development by improving the accomplishment of BW, blood antioxidant components, sexual maturity, and the commencement of puberty. In addition, supplementing lambs with SP at the previous levels had improved the testicular characteristics, seminal plasma antioxidant status, and increased semen quality at puberty. ReferencesAbdel-Wahed, A.M., Khattab, I.M., Zaher, M., Khattab, A.S., El-Damrawy, S.Z., El Keredy, A. and Soltan, Y.A. 2022. Growth performance, nutrient utilization, rumen fermentation, blood biochemistry, and carcass traits of lambs fed Atriplex nummularia L. hay-based diet supplemented with yeast or bacterial direct- fed microbial. Anim. Biotechnol. 33(22), 1–9. Abousoliman, I., Reyer, H., Oster, M., Muráni, E., Mourad, M., Rashed, M.A., Mohamed, I. and Wimmers, K. 2020. Analysis of Candidate Genes for Growth and Milk Performance Traits in the Egyptian Barki Sheep. Animals (Basel). 10 (2), 197. Afolayan, R.A, Adeyinka, I.A. and Lakpini, C.A.M. 2006. The estimation of live weight from body measurements in Yankasa sheep. Czech J. Anim. Sci. 51(8), 343–348. Al-Daraji, H.J., Al-Hassani, D.H., Al-Tikriti, B.T.O. and Abd-Alabaas, M.H. 2001. The influence of breed and season on semen quality of cocks. IPA. J. Agri. Res. 11, 152–162. Alghonaim, A.A., Alqahtani, M.F., Al-Garadi, M.A., Suliman, G.M., Al-Baadani, H.H., Al-Badwi, M.A., Abdelrahman, M.M., Alowaimer, A.N., Khan, R.U. and Alhidary, I. A. 2022. Effects of different levels of spirulina (Arthrospira platensis) supplementation on productive performance, nutrient digestibility, blood metabolites, and meat quality of growing Najdi lambs. Trop. Anim. Health Prod. 54(2), 124. AOAC. 1990. Association of Official Agriculture Chemists, official method of analysis. 15th ed. Arlington, VA: AOAC, pp: 69–88. Bernardini, A., Hozbor, F., Sanchez, E., Forne´ s, M.W., Alberio, R.H. and Cesari, A. 2011. Conserved ram seminal plasma proteins bind to the sperm membrane and repair cryopreservation damage. Theriogenology 76 (3), 436–447. Bezerra, L.R., Silva, A.M.A., Azevedo, S.A., Mendes, R.S., Mangueira, J.M. and Gomes, A.K.A. 2010. Performance of Santa Inès lambs submitted to the use of artificial milk enriched with Spirulina platensis. Ciencia Anim Brasileira, 11(2), 258–263. EI-Khalifa, M.E., Ahmed, Y.H., Hafez, O.A., El-Zolaky, K.M., Bahera and Abido, A.A. 2013. Age at puberty and fertility of Rahmani sheep fed on biological inoculated corn silage. Ann. Agric. Sci. 58(2), 163–172. El-Ratel, I.T. and Gabr, A.A. 2020. Potential impact of Spirulina Alga as an antioxidant on improving semen production and oxidative stress in blood and seminal plasma of rabbit bucks. Egypt. Poult. Sci. J. 40(1), 209–224. EL-Sabagh, M.R., Abd Eldaim, M.A., Mahboub, D.H. and Abdel- Daim, M. 2014. Effects of Spirulina platensis algae on growth performance, antioxidative status and blood metabolites in fattening lambs. J. Agri. Sci. 6(3), 92. El-Wakil, S.I., Shemeis, A.R., Ahmed, A.M. and Abdallah, O.Y. 2008. Genetic and phenotypic relationships involving body weight, degree of maturity and measurer of gain rate of Barki sheep without having recourse to fitting growth curves. J. Agric. Sci. Mansoura Univ. 33, 4835–4848. El-Zelaky, O.A., Khalifa, E.I., Mohamed, A.H., Bahera, Mohamed, K. and Hussein, A.M. 2011. Productive and reproductive performance of Rahmani male lambs fed rations containing jatropha cake. Egypt. J. Sheep Goat Sci. 6(2), 15–24. FAO. 2017. Food and agriculture organization of the United Nations. FAOSTAT online statistical service, FAO. Available via http://Foastat.Fao.org. Farag M.R., Alagawany, M., Abd El-Hac, M.E. and Dhama, K. 2016. Nutritional and healthical aspects of Spirulina (Arthrospira) for poultry, animals, and human. Int. J. Pharmacol. 12(1), 36–51. Fernandez, M., Giralde, F.J., Frutos, P., Lavin, P. and Mantecon, A.R. 2004. Effect of undegradable protein supply on testicular size, spermiogram parameters and sexual behavior of mature Assaf rams. Theriogenology 62, 299–310. Foruie, P.J., Schwalbach, L.M., Neser, F.W.C. and Van der Westhuizen, C. 2004. Scrotal, testicular and semen characteristics of young dorper rams managed under intensive and extensive conditions. Small Rumin. Res. 54, 53–59. Fouda, S.F. and Ismail, R.F.S.A. 2017. Effect of Spirulina platensis on reproductive performance of rabbit bucks. Egyptian J. Nutr. Feeds 20, 55–66. Furbeyre, H., van Milgen, J., Mener, T., Gloaguen, M. and Labussiere, E. 2017. Effects of dietary supplementation with freshwater microalgae on growth performance, nutrient digestibility and gut health in weaned piglets. Animal 11, 183–192. Galal, S., Abdel-rasoul, F., Anous, M.R. and Shaat, I.M. 2005. Onstation characterization of small ruminant breeds in Egypt. In Characterization of small ruminant breeds in West Asia and North Africa. Ed., Iniguez, L. Aleppo, SY: ICARDA, Vol. 2, pp: 141–193. Available via https://cgspace.cgiar.org/handle/10568/66656. Gutiérrez-Rebolledo, G.A., Galar-Martínez, M., García-Rodríguez, R.V., Chamorro-Cevallos, G.A., Hernández-Reyes, A.G. and Martínez-Galero, E. 2015. Antioxidant effect of spirulina (Arthrospira) maxima on chronic inflammation induced by freund’s complete adjuvant in Rats. J. Med. Food. 18(8), 865–871. Holman, B.W. and Malau-Aduli, A.E. 2013. Spirulina as a livestock supplement and animal feed. J. Anim. Physiol. Anim. Nutr. (Berl). 97(4), 615–623. Holman, B.W.B., Kashani, A. and Malau-Aduli, A.E.O. 2012. Growth and body conformation responses of genetically divergent Australian sheep to spirulina (Arthrospira platensis) supplementation. J. Exp. Agri. Int. 2(2), 160–173. Hosseini, S.M., Khosravi-Darani, K. and Mozafari, M.R. 2013. Nutritional and medical applications of spirulina microalgae. Mini Rev. Med. Chem. 13(8), 1231–1237. Ibarra, D., Laborde, D. and Van-Lier, E. 2000. Repeatability and relationship with field mating performance of a serving capacity pen test in rams. Small Rum. Res. 37, 156–169. Ibrahim A.H.M. 2015. Variability of prion protein (prp) gene and its association with productive performance in Barki lambs. J. Amer. Sci. 11(2), 89–96. Karakus, K. 2010. Appropriate growth model describing some testicular characteristics in Norduz male lambs. J. Anim. Plant Sci. 20(1), 1–4. Kashani, A., Holman B., Nichols P. and Malau-Aduli A. 2015. Effect of level of spirulina supplementation on the fatty acid compositions of adipose, muscle, heart, kidney and liver tissues in Australian dual-purpose Lambs. Ann. Anim. Sci. 15(4), 945–960. Keservani, R.K., Kesharwani, R.K., Sharma, A.K. and Jarouliya, U. 2015. Dietary supplements, nutraceutical, and functional foods in immune response (immunomodulators). In Nutraceutical and functional foods in human life and disease prevention. Boca Raton UK: CRC Press, Taylor and Francis, pp: 343–60. Lamminen, M., Halmemies-Beauchet-Filleau, A., Kokkonen, T., Jaak kola, S. and Vanhatalo, A. 2019. Different microalgae species as a substitutive protein feed for soya bean meal in grass silage based dairy cow diets. Anim. Feed Sci. Techno. 247, 112–126. Liang, Y., Bao, Y., Gao, X., Deng, K., An, S., Wang, Z., Huang, X., Liu, D., Liu, Z., Wang, F. and Fan, Y. 2020. Effects of spirulina supplementation on lipid metabolism disorder, oxidative stress caused by high-energy dietary in Hu sheep. Meat Sci. 164, 108094. Liu, X., Han, Y.S. and Kim, I.H. 2019. Growth performance, nutrient digestibility, antioxidant enzyme activity, and fecal microbial flora in growing pigs fed diets containing Spirulina. Can. J. Anim. Sci. 99(4), 840–847. Malau-Aduli, A.E.O. and Holman, B.W.B. 2015. Effect of Spirulina supplementation on plasma metabolites in crossbred and purebred Australian Merino lambs. Int. J. Vet. Sci. Med. 3(1-2), 13–20. Mansour, M.M. 2021. Associations between age at puberty, growth traits, plasma IGF-I and leptin concentrations with reproductive performance of Rahmani and Barki male Lambs. Egypt. J. Sheep Goat Sci. 16(2), 1–10. Mendes R.L., Nobre, B.P., Cardoso, M.T., Pereira, A.P. and Palavra, A.F. 2003. Supercritical car- bon dioxide extraction of compounds with pharmaceutical importance from microalgae. Inorganica. Chimica. Acta. 356, 328–234. Nieto, C.A.R., Ferguson, M.B., Macleay, C.A., Briege, J.R., Martin, G.B. and Thompson, A.N. 2013. Selection for superior growth advances the onset of puberty and increases reproductive performance in ewe lambs. Animal 7(6), 990–997. Panjaitan, T., Quigley, S.P., McLennan, S.R. and Poppi, D.P. 2010. Effect of the concentration of Spirulina (Spirulina platensis) algae in the drinking water on water intake by cattle and the proportion of algae bypassing the rumen. Anim. Prod. Sci. 50, 405–409. Ranjith, L., Shukla, S.P., Vennila, A. and Purushothaman, C.S. 2013. Growth performance of spirulina (Arthrospira) platensis in a low-cost medium: an assessment. Acta Bio. Indica 2(1), 335–342. Shields, R.J. and Lupatsch, I. 2012. Algae for aquaculture and animal feeds. J. Anim. Sci. 21, 23–37. Wang, L., Pan, B., Sheng, J., Xu, J. and Hu, Q. 2007. Antioxidant activity of Spirulina platensis extracts by supercritical carbon dioxide extraction. Food Chem. 105, 36–41. Wu, Q., Liu, L., Miron, A., Klímová, B., Wan, D. and Kuča, K. 2016. The antioxidant, immunomodulatory, and anti-inflammatory activities of Spirulina: an overview. Arch. Toxicol. 90(8), 1817–1840. Zeitoun, M.M., Ateah, M.A., Almaiman, A.T. and Mansour, M.M. 2022. Spirulina supplementation to the semen extender influences the quality and antioxidant parameters of chilled or cryopreserved Arabian stallion spermatozoa. J. Equine Vet. Sci. 118, 104108. | ||
| How to Cite this Article |
| Pubmed Style Mansour MM, Zeitoun MM. Influence of spirulina supplementation on growth performance, puberty traits, blood metabolites, testosterone concentrations, and semen quality in Barki male lambs. Open Vet J. 2023; 13(6): 772-781. doi:10.5455/OVJ.2023.v13.i6.12 Web Style Mansour MM, Zeitoun MM. Influence of spirulina supplementation on growth performance, puberty traits, blood metabolites, testosterone concentrations, and semen quality in Barki male lambs. https://www.openveterinaryjournal.com/?mno=142835 [Access: July 27, 2024]. doi:10.5455/OVJ.2023.v13.i6.12 AMA (American Medical Association) Style Mansour MM, Zeitoun MM. Influence of spirulina supplementation on growth performance, puberty traits, blood metabolites, testosterone concentrations, and semen quality in Barki male lambs. Open Vet J. 2023; 13(6): 772-781. doi:10.5455/OVJ.2023.v13.i6.12 Vancouver/ICMJE Style Mansour MM, Zeitoun MM. Influence of spirulina supplementation on growth performance, puberty traits, blood metabolites, testosterone concentrations, and semen quality in Barki male lambs. Open Vet J. (2023), [cited July 27, 2024]; 13(6): 772-781. doi:10.5455/OVJ.2023.v13.i6.12 Harvard Style Mansour, M. M. & Zeitoun, . M. M. (2023) Influence of spirulina supplementation on growth performance, puberty traits, blood metabolites, testosterone concentrations, and semen quality in Barki male lambs. Open Vet J, 13 (6), 772-781. doi:10.5455/OVJ.2023.v13.i6.12 Turabian Style Mansour, Mohamed M., and Moustafa M. Zeitoun. 2023. Influence of spirulina supplementation on growth performance, puberty traits, blood metabolites, testosterone concentrations, and semen quality in Barki male lambs. Open Veterinary Journal, 13 (6), 772-781. doi:10.5455/OVJ.2023.v13.i6.12 Chicago Style Mansour, Mohamed M., and Moustafa M. Zeitoun. "Influence of spirulina supplementation on growth performance, puberty traits, blood metabolites, testosterone concentrations, and semen quality in Barki male lambs." Open Veterinary Journal 13 (2023), 772-781. doi:10.5455/OVJ.2023.v13.i6.12 MLA (The Modern Language Association) Style Mansour, Mohamed M., and Moustafa M. Zeitoun. "Influence of spirulina supplementation on growth performance, puberty traits, blood metabolites, testosterone concentrations, and semen quality in Barki male lambs." Open Veterinary Journal 13.6 (2023), 772-781. Print. doi:10.5455/OVJ.2023.v13.i6.12 APA (American Psychological Association) Style Mansour, M. M. & Zeitoun, . M. M. (2023) Influence of spirulina supplementation on growth performance, puberty traits, blood metabolites, testosterone concentrations, and semen quality in Barki male lambs. Open Veterinary Journal, 13 (6), 772-781. doi:10.5455/OVJ.2023.v13.i6.12 |