| Case Report | ||
Open Vet J. 2022; 12(3): 323-328 Open Veterinary Journal, (2022), Vol. 12(3): 323-328 Case Report Endolymphatic sac tumor in an 8-month-old catMatteo Baccolini1*, Marco Rosati2, Federica Tirrito3, Francesca Cozzi1 and Rocco Lombardo11Clinica Neurologica Veterinaria NVA, Milan, Italy 2Section of Clinical and Comparative Neuropathology, Centre for Clinical Veterinary Medicine, Ludwig-Maximilians-Universität München, Munich, Germany 3AniCura Istituto Veterinario Novara, Organization, Granozzo con Monticello, Italy *Corresponding Author: Matteo Baccolini. Clinica Neurologica Veterinaria NVA, Milan, Italy.Email: matteo.baccolini [at] nvamilano.it Submitted: 02/02/2022 Accepted: 04/05/2022 Published: 16/05/2022 © 2022 Open Veterinary Journal
AbstractBackground: The endolymphatic sac is an organ devoid of sensory receptors. It is connected with the endolymphatic compartment and contains endolymph. Endolymphatic sac tumor (ELST) is a rare neoplasm involving the middle and inner ear described in humans and dogs that does not show cellular characteristics of malignancy, but can be locally invasive and involve destruction of the temporal bone and adjacent structures. Case Description: An 8-month-old female cat was referred because of sudden onset of vestibular signs starting 3 days prior to referral. On clinical examination, the patient showed depression, right head tilt, left-sided facial paralysis, and horizontal nystagmus with fast phase to the left. The magnetic resonance images showed a voluminous extra-axial lesion, with irregular morphology and well-defined margins, with intracranial extension in the region of the pons, rostral medulla oblongata, cerebellar vermis, floccule, and left cerebellar hemisphere. Due to progressive clinical deterioration, the cat was euthanized 2 weeks later. A necropsy was then performed and histological samples were taken. The necropsy revealed the presence of a voluminous dark red irregular mass extending from the tympanic bulla to the posterior cranial fossa following the left glossopharyngeal nerve. The histopathological exam of the extra-axial lesion featured a nonencapsulated, moderately cellular, rather loose, proliferation of cuboidal to columnar epithelium breaching through chunks of an otherwise normal appearing dura mater and invading some cranial nerves. Sections of the cerebellum and brainstem revealed moderate, focal, impingement of the parenchyma with a very mild extension of the proliferating cells into the ventral left side of the medulla oblongata. Based on these histological characteristics, the lesion was defined as ELST, a rare neoplasm described in human beings and with two reports in dogs. Conclusion: To our knowledge, this is the first report describing an ELST in a cat. Keywords: Cat, Endolymphatic sac, Inner ear, Labyrinth diseases, Neoplasia. IntroductionEndolymphatic sac tumor (ELST) is a rare neoplasm of the middle and inner ear described in human medicine as a slow-growing papillary tumor with histological features of low malignancy, but an aggressive biological behavior (Hassard et al., 1984). In human medicine, it is associated with the Von Hippel-Lindau syndrome, a rare hereditary multiple neoplasia syndrome that predispose to the development of hemangioblastomas of the central nervous system (CNS), retinal hemangioblastomas, renal cysts, and carcinomas, pheochromocytomas, pancreatic cysts, and epididymal cystadenomas (Choo et al., 2004). ELST originates from the endolymphatic sacs or ducts of the inner ear and subsequently extends to the middle and outer ear, to the labyrinth. Generally, it also affects the cerebellopontine angle and can also involve the medulla oblongata and the jugular veins. Despite having a low metastatic index, it exhibits very invasive behavior resulting in the destruction of the adjacent temporal bone (Heffner, 1989). Often these tumors are misdiagnosed as choroid plexus papilloma (CPP), ceruminous adenoma, paraganglioma, and adenomatous middle ear tumor. From a radiological point of view, they have features that can lead to the misdiagnosis of glomus jugular tumors, meningiomas, cysts, or bone tumors (Du et al., 2015). In veterinary medicine, there have been cases of neoplasia with characteristics similar to ELST reported in dogs (Barnes et al., 2017), but never in cats. Case DetailsAn 8-month-old female British Shorthair cat was examined for acute onset of head tilt, ataxia, and lack of appetite 3 days prior to referral. General physical examination did not show significant findings. Neurological examination revealed depression, right head tilt, right vestibular ataxia, positional horizontal/rotational nystagmus with rapid phase to the left, bilateral protrusion of the third eyelid, deficit of the palpebral reflex on the left eye, and reduction of facial motility on the left side. The examination led to neurolocalization to the right vestibular system with involvement of the left VII cranial nerve or its nucleus. The main differential diagnoses were inflammatory/infectious diseases of the middle/inner ear, or pathologies affecting the brain due to inflammatory/infectious or neoplastic diseases. Blood tests, cardiological examination, and echocardiography did not show any abnormality; enzyme-linked immunosorbent assay (ELISA) test for feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV) carried out a few weeks earlier had given negative results. A magnetic resonance study of the skull was then performed (Hitachi Airis II, 0.3T). T2-weighted and T1-weighted scans were performed on the transverse and sagittal planes, sequence of a multiecho gradient recalled echo (GET2*), short tau inversion recovery (STIR), and 3DT1 on the dorsal plane. T1-weighted scans were then acquired after the administration of paramagnetic contrast medium (Gadoteridol, Prohance 279.3 mg/ml, 0.2 ml/kg). The images showed a voluminous extra-axial lesion, with irregular morphology and defined margins, affecting the region of the left metencephalon, with intracranial extension in the region of the pons, in the rostral portion of the medulla oblongata, in the cerebellar vermis, flocculus, and left cerebellar hemisphere (Fig. 1). Erosion of the petrous portion of the left temporal bone was observed with extracranial extension and slight dorsal compression of the left tympanic bulla, which appeared slightly deformed. In addition, there was invasion/compression of the left longus colli muscle and digastric muscles. The lesion signal appeared hyperintense to the brain parenchyma and surrounding musculature and heterogeneous in the T2-weighted, fluid-attenuated inversion recovery (FLAIR), STIR, and GET2* sequences, and isointense in the T1-weighted sequences. Contrast uptake was marked and heterogeneous. The lesion resulted in compression and right dislocation of the pons, medulla oblongata, cerebellar vermis, and left cerebral hemisphere. It compressed and displaced to the right the mesencephalic aqueduct and rostrolaterally to the left, the left lateral recess of the fourth ventricle, which appeared slightly distended. There was a slight symmetrical dilation of the entire ventricular system, including the cavities of the olfactory bulbs. Marked perilesional edema was present. The extracranial portion of the lesion caused mass effect and lateral dislocation of the left hemiportion of the hyoid bone. The images obtained were strongly suggestive of an extra-axial neoplasia with intra- and extracranial involvement. Lumbar cerebrospinal fluid analysis had a total protein content of 241.1 mg/dl (normal value: <45 mg/dl), 5 nucleated cells/µl (normal value: 2–5/µl), and red blood cells 23,040/µl. Cytological examination was suggestive of blood contamination and occasional small lymphocytes. Following diagnostic investigations, the patient was hospitalized and a palliative therapy aimed to reduce perilesional edema and any bacterial infection due to compromised middle ear was started. Dexamethasone (0.1 mg/kg once daily), clindamycin (12.5 mg/kg twice daily), and maropitant (1 mg/kg once daily) were administered. After an initial improvement of vestibular ataxia, head tilt, and nystagmus, the clinical conditions worsened and the owner, considering the poor prognosis, opted for euthanasia. Permission was granted to carry out a postmortem examination and a histological examination of the neoformation.
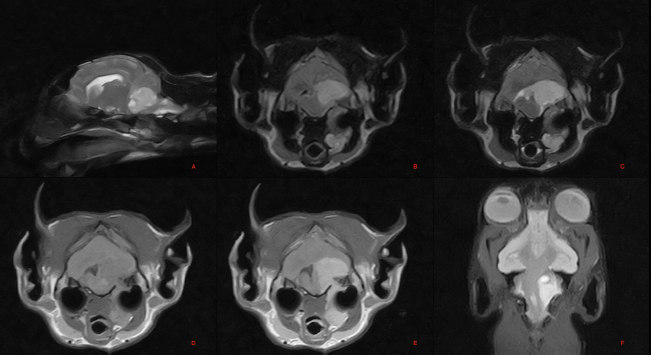
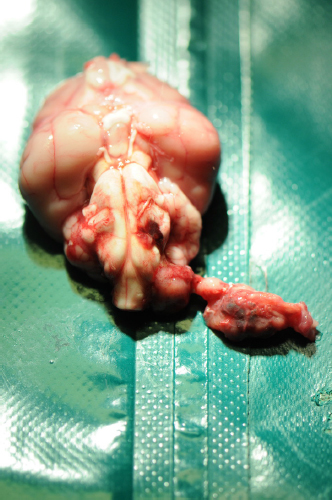
Fig. 1. Magnetic resonance imaging (MRI) of the skull: T2-weighted images of the brain, parasagittal plane (A), STIR image, transverse plane at the level of the pons (B), T2 weighted image, transverse plane at the level of the pons (C), T1 weighted image, transverse plane at the level of the pons (D), T1 weighted post contrast image, transverse plane at the level of the pons (E). Dorsal Gradient Echo T2* weighted image of the brain, dorsal plane (F). Voluminous extra axial lesion affecting the region of the left metencephalon. The necropsy revealed the presence of a voluminous dark red irregular mass which from the tympanic bulla invaded the posterior cranial fossa following the left glossopharyngeal nerve through the jugular foramen and the tympanic-occipital fissure (Fig. 2). Histological examination described a nonencapsulated, moderately cellular, rather loose, proliferation of cuboidal to columnar epithelium breaching through chunks of an otherwise normal appearing dura mater and invading some cranial nerves. Cells were arranged in papillae, large cystic lacunae with low amount of faintly eosinophilic amorphous material, or glandular architecture, depending on the region considered (Fig. 3).
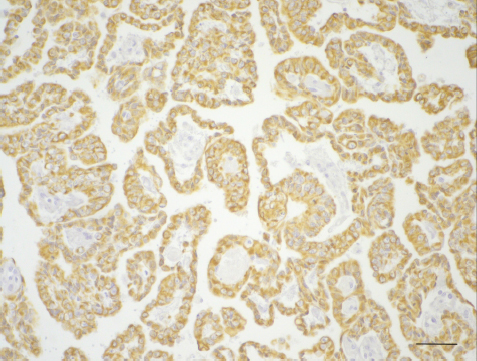
Fig. 2. A voluminous dark red irregular mass following the left glossopharyngeal nerve through the jugular foramen and the tympanic-occipital fissure. These cells were supported by a moderate amount of moderately vascularized stroma. Cells presented with moderate anisocytosis and mild anisokaryosis with variations in between cuboidal and columnar ciliated epithelium, mildly increased nucleus/cytoplasm ratio, and nonpolarized nuclei. There were 15 mitoses in 5 high-power fields. Immunohistochemistry for pancytokeratin displayed diffuse immunoreactivity of the proliferating cells (Fig. 4), also showing high ki67 positivity, while neuron-specific enolase and glial fibrillary acidic protein were negative. Sections of the cerebellum and brainstem revealed moderate, focal, impingement of the parenchyma with very mild extension of the proliferating cells into the ventral left side of the medulla oblongata. Very mild perilesional edema, mild multifocal microglial activation, and very mild diffuse astrocytosis and astrogliosis were detected at this level. Eosinophilic neuronal necrosis was also occasionally observed. Similar reactive changes of the neuroparenchyma were encountered along the ventral paramedian lobule and paraflocculus with no clear evidence of invasion by proliferating cells. Location, morphologic features, and immunohistochemical labeling were consistent with a diagnosis of ELST . Ethical approvalThis work involved the use of nonexperimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognized high standards (“best practice”) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not necessarily required. DiscussionIn human medicine, diagnosis of ELST is based on the mixture of several factors including radiological, histopathological, and clinical characteristics. The main differential diagnosis, with which these tumors can be confused, is that of CPP. However, a recent review (Du et al., 2015) identified several radiological, histopathological, and clinical characteristics to allow an easier distinction between the two neoplasms: from a radiological point of view, ELSTs are characterized by strong invasiveness with destruction of the bone component of the skull base, slow growth, and a very low metastatic index (Bell et al., 2011), while CPPs tend to invade the brain parenchyma without concomitant destruction of dura mater or bone. From a histopathological point of view, ELSTs are generally characterized by a papillary-cystic glandular architecture and by the presence of a thyroid-like pattern, in which the glandular-cystic structure expands and presents a colloidal content within it (Megerian et al., 1995). Similar features also characterize the CPP, with which ELSTs have in some cases been confused. However, CPPs are mainly associated with the ventricular system, are rarely located in the cerebellopontine angle, and moreover never cause bone destruction. Additional histopathological distinguishing parameters include greater vascularization of ELSTs than CPPs (Du et al., 2015); furthermore, the nuclei of ELST are often positioned in the middle or near the apical surface of the cells of the cavity or the papillae, while the nuclei of CPP are mainly polarized and located near the basal membrane (Du et al., 2015). ELST shows various amounts of two cell types, clear cells and epithelioid cells, with eosinophilic cytoplasm; vacuoles in the cytoplasm are detectable on light microscopy in some of the human cases, while electron microscopy reveals abundance of small cytoplasmic vesicles, similar to those expected in normal endolymphatic sac epithelium (Du et al., 2015). On the contrary, tumor cells of CPP usually have pale eosinophilic cytoplasm; clear cells are scarce and cytoplasmic vacuoles are not present. Psammoma bodies often appeared in CPP (7/8 cases of CPP and 2/3 cases of atypical CPP had psammoma bodies), but are not reported for ELST. Instead, a thyroid-like structure, more predominant in ELST, seldom appeared in CPP (Du et al., 2015).
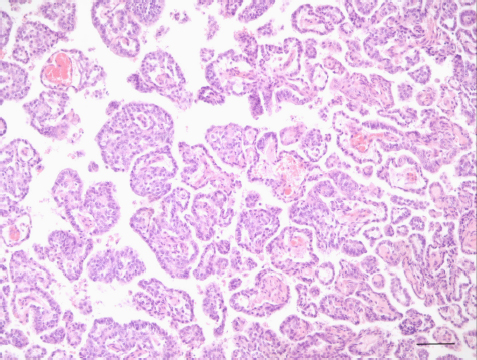
Fig. 3. ELST. Neoplastic cells grow forming papillary projections lined by one to two layers of polygonal cells and supported by a delicate fibrovascular stroma (H&E stain; scale bar=100 µm).
Fig. 4. Immunohistochemistry for pancytokeratin displaying diffuse immunoreactivity of polygonal cells revealing the epithelial origin of the neoplastic cells (3,3′-Diaminobenzidine stain; scale bar=100 µm). Finally, ELSTs presented with bone destruction and are epidural located, while CPP are mainly located within the brain and do not show bone invasion (Du et al., 2015). Clinically, ELSTs have very variable age of onset (4–71 years) and some studies suggest a slight female predisposition (Heffner, 1989; Schipper et al., 2006). A recent review confirmed previously published data indicating that in juvenile forms the disease appears to have a female predilection (2:1 ratio) and may be associated with more aggressive clinical behavior (Bausch et al., 2016; Talukdar et al., 2022). While the most frequently reported clinical symptoms in CPPs are related to the presence of increased intracranial pressure (headaches, vomiting, and nausea), symptoms related to vertigo, tinnitus, and hearing loss prevail in ELSTs (Du et al., 2015). In the two canine reports, there was no evidence of invasion of the cranial cavity, both cases were successfully treated surgically (total ear canal ablation and bullae osteotomy), and no relapses were reported at 9 and 12 months after surgery (Barnes et al., 2017). Similar to canine cases, the histology of the neoplastic mass in this patient revealed a proliferation of ciliated epithelial cells arranged in anastomosing papillae and lining cystic cavities resembling endolymphatic sac epithelium. Cystic lacunae with a low amount of eosinophilic amorphous material as seen in human cases of ELST were detected in our cat but were not described in dogs. Likewise, immunohistochemistry for pancytokeratin identified neoplastic cells as of epithelial origin. The clinical, radiological, and histopathological findings presented by this patient are therefore compatible with the diagnosis of ELST. ConclusionELST is a very rare neoplasm in veterinary medicine; to the best of our knowledge, we reported this neoplasm for the first time in a feline patient. Tumor location, extension, and histomorphology appeared typical for ELST. Extension within the cranial cavity was associated with a worst prognosis in our cat compared to that reported in dogs in which only the tympanic bulla and ear canal were involved and surgical removal proved successful. Surgical treatment of human patients provides a good prognosis, and a recent work has proposed the use of radiotherapy in those cases in which it is not possible to obtain undamaged margins after surgical removal (Mendenhall et al., 2018). Further conclusions on prognosis and efficacy of different treatment modalities in veterinary medicine require a bigger cohort of patients and multicentric studies. AcknowledgmentThe authors declare that there are no conflicts of interest. Conflict of interestThe authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. FundingThe authors did not receive any financial support for this research, authorship, and/or publication of this article. Consent to participateInformed consent (either verbal or written) was obtained from the owner or legal custodian of the animal described in this work for the procedures undertaken. For any animals or humans individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved. Authors contributionAll authors contributed to the study conception and design. The clinical case was followed up by Matteo Baccolini and Federica Tirrito. Magnetic resonance imaging was evaluated by Matteo Baccolini, Federica Tirrito, and Francesca Cozzi. Material preparation and data collection were performed by Rocco Lombardo. Macroscopical, histological, and immunohistochemical examination were performed by Marco Rosati. The first draft of the manuscript was written by Matteo Baccolini; final version was written by Matteo Baccolini; and Marco Rosati and all authors commented on previous versions of the manuscript. All authors read and approved the final version of the manuscript. ReferencesBarnes, K.J., Clear, V., Youmans, K. and Hardcastle, M.R. 2017. Endolymphatic sac tumor in two dogs. Vet. Pathol. 54(4), 683–685. Bausch, B., Wellner, U., Peyre, M., Boedeker, C.C., Hes, F.J., Anglani, M., de Campos, J.M., Kanno, H., Maher, E.R., Krauss, T., Sansó, G., Barontini, M., Letizia, C., Hader, C., Schiavi, F., Zanoletti, E., Suárez, C., Offergeld, C., Malinoc, A., Zschiedrich, S., Glasker, S., Bobin, S., Sterkers, O., Ba Huy, P.T., Giraud, S., Links, T., Eng, C., Opocher, G., Richard, S., Neumann, H.P. and International Endolymphatic Sac Tumor (ELST) Consortium. 2016. Characterization of endolymphatic sac tumors and von Hippel-Lindau disease in the International Endolymphatic Sac Tumor Registry. Head Neck. 38(Suppl. 1), E673–E679. Bell, D., Gidley, P., Levine, N. and Fuller, G.N. 2011. Endolymphatic sac tumor (aggressive papillary tumor of middle ear and temporal bone): sine qua non radiology-pathology and the University of Texas MD Anderson Cancer Center experience. Ann. Diagn. Pathol. 15(2), 117–123. Choo, D., Shotland, L., Mastroianni, M., Glenn, G., van Waes, C., Linehan, W.M. and Oldfield, E.H. 2004. Endolymphatic sac tumors in von Hippel-Lindau disease. J. Neurosurg. 100(3), 480–487. Du, J., Wang, J., Cui, Y., Zhang, C., Li, G., Fang, J., Yue, S. And Xu, L. 2015. Clinicopathologic study of endolymphatic sac tumor (ELST) and differential diagnosis of papillary tumors located at the cerebellopontine angle. Neuropathology. 35(5), 410–420. Hassard, A.D., Boudreau, S.F. and Cron, C.C. 1984. Adenoma of the endolymphatic sac. J. Otolaryngol. 13(4), 213–216. Heffner, D.K. 1989. Low-grade adenocarcinoma of probable endolymphatic sac origin a clinicopathologic study of 20 cases. Cancer 64(11), 2292–2302. Megerian, C.A., McKenna, M.J., Nuss, R.C., Maniglia, A.J., Ojemann, R.G., Pilch, B.Z. and Nadol, J.B. Jr. 1995. Endolymphatic sac tumors: histopathologic confirmation, clinical characterization, and implication in von Hippel-Lindau disease. Laryngoscope. 105(8 Pt 1), 801–808. Mendenhall, W.M., Suárez, C., Skálová, A., Strojan, P., Triantafyllou, A., Devaney, K.O., Williams, M.D., Rinaldo, A. and Ferlito, A. 2018. Current treatment of endolymphatic sac tumor of the temporal bone. Adv. Ther. 35(7), 887–898. Schipper, J., Maier, W., Rosahl, S.K., van Velthoven, V., Berlis, A., Boedeker, C.C., Laszig, R., Teszler, C.B. and Ridder G.J. 2006. Endolymphatic sac tumours: surgical management. J. Otolaryngol. 35(6), 387–394. Talukdar, R., Epari, S., Sahay, A., Choudhari, A., Dasgupta, A., Chatterjee, A. and Gupta, T. 2022. Endolymphatic sac tumor: single-institution series of seven cases with updated review of literature. Eur. Arch. Otorhinolaryngol. 279(5), 2591–2598. | ||
| How to Cite this Article |
| Pubmed Style MB, Rosati M, Tirrito F, Cozzi F, Lombardo R. Endolymphatic sac tumor in a 8 months old cat. Open Vet J. 2022; 12(3): 323-328. doi:10.5455/OVJ.2022.v12.i3.3 Web Style MB, Rosati M, Tirrito F, Cozzi F, Lombardo R. Endolymphatic sac tumor in a 8 months old cat. https://www.openveterinaryjournal.com/?mno=17447 [Access: July 27, 2024]. doi:10.5455/OVJ.2022.v12.i3.3 AMA (American Medical Association) Style MB, Rosati M, Tirrito F, Cozzi F, Lombardo R. Endolymphatic sac tumor in a 8 months old cat. Open Vet J. 2022; 12(3): 323-328. doi:10.5455/OVJ.2022.v12.i3.3 Vancouver/ICMJE Style MB, Rosati M, Tirrito F, Cozzi F, Lombardo R. Endolymphatic sac tumor in a 8 months old cat. Open Vet J. (2022), [cited July 27, 2024]; 12(3): 323-328. doi:10.5455/OVJ.2022.v12.i3.3 Harvard Style , M. B., Rosati, . M., Tirrito, . F., Cozzi, . F. & Lombardo, . R. (2022) Endolymphatic sac tumor in a 8 months old cat. Open Vet J, 12 (3), 323-328. doi:10.5455/OVJ.2022.v12.i3.3 Turabian Style , Matteo Baccolini, Marco Rosati, Federica Tirrito, Francesca Cozzi, and Rocco Lombardo. 2022. Endolymphatic sac tumor in a 8 months old cat. Open Veterinary Journal, 12 (3), 323-328. doi:10.5455/OVJ.2022.v12.i3.3 Chicago Style , Matteo Baccolini, Marco Rosati, Federica Tirrito, Francesca Cozzi, and Rocco Lombardo. "Endolymphatic sac tumor in a 8 months old cat." Open Veterinary Journal 12 (2022), 323-328. doi:10.5455/OVJ.2022.v12.i3.3 MLA (The Modern Language Association) Style , Matteo Baccolini, Marco Rosati, Federica Tirrito, Francesca Cozzi, and Rocco Lombardo. "Endolymphatic sac tumor in a 8 months old cat." Open Veterinary Journal 12.3 (2022), 323-328. Print. doi:10.5455/OVJ.2022.v12.i3.3 APA (American Psychological Association) Style , M. B., Rosati, . M., Tirrito, . F., Cozzi, . F. & Lombardo, . R. (2022) Endolymphatic sac tumor in a 8 months old cat. Open Veterinary Journal, 12 (3), 323-328. doi:10.5455/OVJ.2022.v12.i3.3 |