| Original Article | ||
Open Vet J. 2022; 12(6): 815-821 Open Veterinary Journal, (2022), Vol. 12(6): 815–821 Original Research Comparative study of healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and combined treatment with medical therapy and a nictitating membrane flap: A retrospective studyKanokpich Imcharoon1, Natawadee Pinyosnit1, Pimpika Srilert1, Tanakanok Ngampongsai1, Vachira Hunprasit2, Nalinee Tuntivanich3 and Orapun Jaturakan3*1Faculty of Veterinary Science, Chulalongkorn University, Bangkok, Thailand 2Department of Veterinary Medicine, Faculty of Veterinary Science, Chulalongkorn University, Bangkok, Thailand 3Department of Veterinary Surgery, Faculty of Veterinary Science, Chulalongkorn University, Bangkok, Thailand Submitted: 26/05/2022 Accepted: 11/10/2022 Published: 09/11/2022 *Corresponding Author: Orapun Jaturakan. Department of Veterinary Surgery, Faculty of Veterinary Science, Chulalongkorn University, Bangkok, Thailand. Email: Orapun.j [at] chula.ac.th © 2022 Open Veterinary Journal
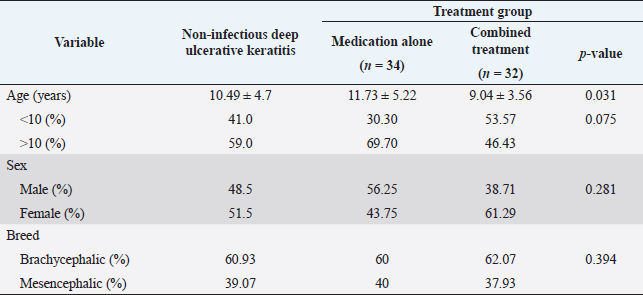
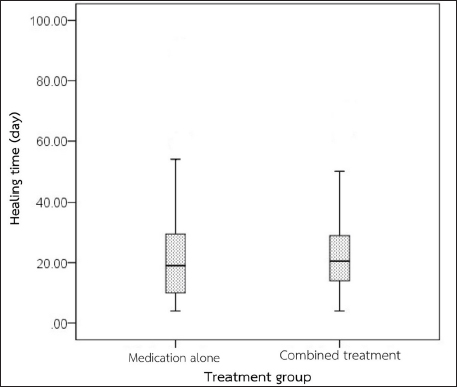
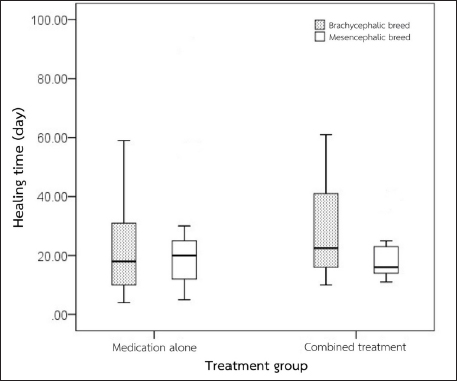
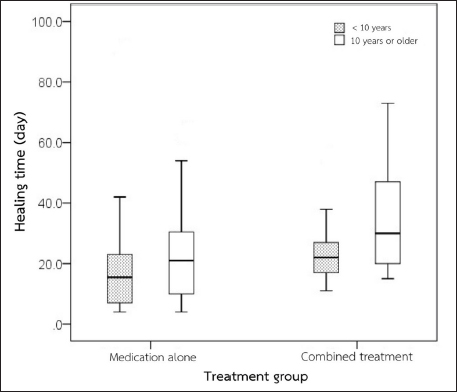
AbstractBackground: Canine non-infectious deep ulcerative keratitis is considered a severe ocular disorder that possibly can progress to perforation. Immediate treatment should be directed to stimulate corneal wound healing, control infection, and minimize self-trauma while eliminating the underlying causes. Aim: This retrospective study was aimed to compare the difference in non-infectious deep corneal wound healing time between cases treated with medical therapy alone and those treated with medical therapy combined with a nictitating membrane flap. Methods: The medical records at the Ophthalmology Clinic, Small Animal Teaching Hospital, Faculty of Veterinary Science, Chulalongkorn University between January 2018 and March 2020 were retrospectively reviewed. Sixty-six eyes (from 65 dogs) diagnosed with non-infectious deep ulcerative keratitis from the medical treatment group (n=34) and the combined treatment group (n=32) were included. The combined treatment group was prescribed the same conservative medical administrations plus a surgical nictitating membrane flap for 14 days. Results: Healing time was defined as the duration of time from the day that the dog had been diagnosed with deep ulcerative keratitis by a fluorescein staining test to the day that the corneal fluorescein stain was negative. Overall, the mean age of dogs with deep ulcerative keratitis was 10.49 ± 4.7 years. The disease was commonly evident in females more than males. Shih Tzu was the most prevalent dog breed. The corneal healing time between dogs receiving medical therapy alone and those receiving combined treatment was not statistically significant (p=0.386). Healing times were not significantly different between sex and breed (p=0.41). The median corneal healing time for dogs older than 10 years in the combined treatments group (29.5 days; ranging from 20 to 46 days) was longer than for those receiving medical therapy alone (21 days; ranging from 9.5 to 30.5 days). Conclusion: Supportive therapy including a nictitating membrane flap is suggested in dogs prone to deep corneal ulcers not involving infection. Even though the healing time is not statistically significant, a nictitating membrane flap acts as a tissue bandage to reduce friction over the cornea, and it also alleviates the healing process by moistening the ocular surface. Keywords: Corneal healing, Deep corneal ulcer, Dog, Nictitating membrane flap, Non-infectious ulcerative keratitis. IntroductionUlcerative keratitis is one of the most common ocular diseases in veterinary ophthalmology. It has been reported in many countries: 60 out of 4,500 cases from the Veterinary Clinical Complex in India (Kumar et al., 2018) and 834 out of 104,233 cases from Primary Veterinary Care in England (O’Neill et al., 2017). Statistical relationships between the incidence of ulcerative keratitis in dogs and other factors (breed, age, and sex) were reported (O’Neill et al., 2017; Kumar et al., 2018). There are several aetiologies of canine ulcerative keratitis (Martin, 2005; Gilger et al., 2007), of which the most common cause is exogenous trauma (Crispin, 2002). Transparency of the cornea is critical for proper vision. Its significant refractive function allows the transmission of light to be focused onto the retina. The cornea acts as a good physical barrier protecting intraocular structures from external insults; infectious and non-infectious. It is therefore important to address any primary causes of ulcerative keratitis promptly prior to the progression to aggressive corneal damage. Not only deep ulcerative keratitis has a higher risk of infection, but also a greater loss of transparency leading to severe ocular pain, a greater risk of intraocular damage, visual impairment, and blindness (Ledbetter et al., 2013). Numerous treatments have been described to treat deep ulcerative keratitis. Medical treatment should promptly be initiated as the first line of treatment. The route of administration depends on the depth, size, and location of the lesion. Extensive topical antibiotics should be given for the prevention of secondary bacterial infection, which could lead to complicated corneal ulcers (Pirdankar et al., 2020). Cycloplegics and mydriatics should be prescribed to reduce ciliary spasms and produce mydriasis. They help to relieve ocular pain and prevent the formation of synechia. Additional administration of a topical anti-collagenase agent is very useful to stimulate corneal cell proliferation (Maggs et al., 2013). Systemic administration of anti-inflammatory agents is recommended for deep corneal lesions to prevent rapid loss of vision from anterior uveitis. Superficial ulcerative keratitis wounds treated with medication alone had a recovery time of 2 weeks shorter than the recovery time for deep corneal wounds. Moreover, some deep ulcerative keratitis cases did not heal completely with medical treatment alone (Kim et al., 2009). Irritation (Maggs et al., 2013) and infection (Wang et al., 1998) were major complications during the healing period of canine deep ulcerative keratitis. Therefore, surgical intervention is recommended for deep, severe, or progressive ulcerative keratitis, in addition to medical treatment. For deep ulcerative keratitis with the loss of more than 50% of the corneal thickness, biological graft, and biomaterials were recommended (Balland et al., 2016; Jaksz et al., 2021; Santillo et al., 2021). Moreover, supportive therapy to the ocular surface, such as a nictitating membrane flap and a bandage contact lens, is widely used to protect the cornea from irritation due to blinking or exogenous stimuli (Slatter, 2001; Gilger et al., 2007). A nictitating membrane flap is one of the supportive therapies used to avoid movement of the third eyelid (Maggs et al., 2013) and support the weakened cornea (Giuliano, 2015) by covering and moisturizing it with the precorneal tear film. The procedure can easily be achieved with standard ophthalmic surgical instruments. It is reported that a 3-year-old horse with a deep corneal ulcer and 4 days of vision loss had a return of menace response and pupillary light reflex after the 90th post-operative day with a nictitating membrane flap (Jhala et al., 2011). Therefore, a nictitating membrane flap might be a good alternative treatment in senile or fragile patients with deep stromal ulcers. Due to the fact that corneal wound healing is a complex process, delayed healing time can result in incomplete/non-healed wound repair, corneal complications (Ljubimov and Saghizadeh, 2015), and loss of vision. The main purpose of the study was to compare the corneal healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and medical therapy combined with a nictitating membrane flap. Materials and MethodsSample populationsThe medical records of dogs visiting the Ophthalmology Clinic at the Small Animal Teaching Hospital, Chulalongkorn University, Bangkok, Thailand between January 2018 and March 2020 were retrospectively reviewed. All dogs received complete physical and ophthalmic examinations. The inclusion criteria of the study consisted of a diagnosis of non-infectious deep ulcerative keratitis regarding a history taking, no previous topical steroid administration, no related signs of ocular surface infection, and a fluorescein staining test on the medical records. Stromal corneal ulcers involved not exceeding 50% thickness of the cornea. Records were completely and correctly filled from the first day of diagnosis until the ulcers were completely healed, which was confirmed by a negative fluorescein stain. Sixty-six eyes diagnosed with deep ulcerative keratitis were divided into two groups: the medical treatment group (n=34) and the combined treatment group (n=32). In the medical treatment group, a single topical antibiotic such as tobramycin, ofloxacin, moxifloxacin, or levofloxacin was applied every 2 hours for 2 weeks. Amoxycillin–clavulanic acid (12.5–25 mg/kg, bid), clindamycin (15 mg/kg, bid), doxycycline (10–15 mg/kg, bid), or enrofloxacin (5 mg/kg, sid) was selectively used as a systemic antibiotic. Atropine sulfate (twice daily) was given in all cases. Systemic administration of anti-inflammatory agents was considered in some cases with anterior uveitis, such as prednisolone (0.5–1 mg/kg; sid–bid), firocoxib (5 mg/kg; sid), or carprofen (2.2 mg/kg; bid). Some cases received topical anti-collagenase agents, such as autologous fresh serum or acetylcysteine four times daily. The combined treatment group received a surgical nictitating membrane flap in addition to medication. The temporal nictitating membrane flap to the upper bulbar conjunctiva remained for 14 days. Demographic variables including age, sex, and breed as well as a therapeutic method were reviewed. The corneal healing time was defined as the duration of time from the day that the dog had been diagnosed with positive fluorescein staining to the day that the corneal fluorescein staining test was negative. Statistical analysisDemographic variables were statistically assessed. The normality assumption of age was evaluated using the Kolmogorov–Smirnov test. Due to the normality assumption, age was presented as the mean and standard deviation. Breed and sex were expressed as percentages. Dogs were further classified into two treatment groups and the demographic variables of each group were analyzed. To compare the difference in age between the two groups, the Student’s t-test was performed. The difference between sex among the two treatment groups was evaluated using the Chi-square test. The normality assumption of healing time was assessed using the Kolmogorov–Smirnov test. The difference in healing time among the two treatment groups was analyzed using either the Student’s t-test or the Mann–Whitney U test depending on the normality assumption of the data. Furthermore, age was categorized based on the quantile method into younger than 10 years and 10 years or older while breed was categorized into either brachycephalic or mesencephalic based on skull conformation, then compared between the treatment groups using the Chi-square test. The healing times of each treatment group based on age and breed were compared descriptively. The statistical analyses were performed using Statistical Package for the Social Sciences (IBM SPSS Statistics for Windows, Version 25.0, IBM Corp., Armonk, NY). The statistical significance was considered when p < 0.05. Ethical approvalThis study was a retrospective study. The medical records of dogs visiting the Ophthalmology Clinic at the Small Animal Teaching Hospital, Chulalongkorn University, Bangkok, Thailand between January 2018 and March 2020 were retrospectively reviewed without using animal experiments. The authors confirm that the ethical policies of the journal have been followed and that no ethical approval was required for this study. ResultsThere were 65 dogs (66 eyes) included in this study. When categorized into two treatment groups, the mean age of the dogs in the medical treatment group (11.73 ± 5.22 years) was older than that of the combined treatment group (9.04 ± 3.56 years) (p=0.031). With regard to sex, the proportion of male dogs in the medical treatment group (56.25%) was more than in the combined treatment group (38.71%). Meanwhile, the proportion of female dogs in the medical treatment group (43.75%) was less than in the other group (61.29%). Sex was not significantly different between the two treatment groups (p=0.281) (Table 1). In this study, 11 dog breeds were identified and presented in Table 2. Of all dogs, 39 cases (60.93%) were brachycephalic breeds while the rest were mesencephalic breeds. Non-infectious deep ulcerative keratitis was found in female dogs (51.5%) more than in male dogs (48.5%). The average age of the dogs diagnosed with non-infectious deep ulcerative keratitis was 10.49 ± 4 years, ranging from 2 to 20 years. Corneal healing time was not significantly different between the medical treatment group (median 19.0 days; range 10.0–30.0 days) and the combined treatment group (median 20.5 days; range 14.0–29.5 days) (p=0.386) (Fig. 1). The result showed that sex (median males 19.0 days; range 13.0–30.5 days, median females 20 days; range 11–30 days) and breed (median brachycephalic breeds 21 days; range 11–38 days, median mesencephalic breeds 18 days; range 14.0–24.5 days) were not significantly different (Fig. 2) between the two groups. The median healing time of dogs older than 10 years receiving combined treatment (29.5 days; range 20.0–46.0 days) was longer than for those receiving medical therapy alone (21.0 days; range 9.5–30.5 days) (Fig . 3). DiscussionThe result from this study showed that the healing time of non-infectious deep ulcerative keratitis involving not exceeding 50% of corneal thickness was not significantly different between the medical treatment group and the combined treatment group. According to the healing time of combined treatment, Vongsakul et al. (2009) revealed that the average healing time for eyes receiving a nictitating membrane flap was 9.17 ± 0.31 days, which was shorter than that in our study. The duration for the covering flap reported by Vongsakul et al. (2009) was 7 days, but it was 14 days which was longer in this study. The true healing time could not be assessed while corneal ulcers were being entirely covered by the nictitating membrane flap. It could therefore be speculated that, in some cases, ulcers may have completely healed prior to flap removal. Moreover, healing time could be influenced by the severity of the corneal ulcer (Spiess et al., 2014), individual variation (Williams and Burg, 2017; Iwashita et al., 2020), ocular surface conditions (Ollivier et al., 2007), etc. Corneal stroma involves the majority of the entire corneal thickness, and healing time ranges between days and weeks (Gelatt and Plummer, 2022). The healing time of canine fungal keratitis was from 2 to 6 weeks (median time 30 days, range 21–43 days) (Ledbetter et al., 2016) while the healing time of canine bacterial keratitis was from 2 to 4 weeks (Farghali et al., 2021). Table 1. Demographic information of dogs with non-infectious deep ulcerative keratitis categorized into two treatment groups.
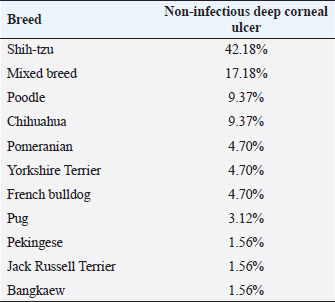
Table 2. Breed distribution of dogs with non-infectious deep ulcerative keratitis.
In comparing the group receiving medical treatment and that receiving combined treatment, we herein report a longer corneal healing time in the combined treatment group. In canine refractory superficial keratitis receiving topical medications only, healing times with antibiotics alone, anti-inflammatory drug alone, and proteinase inhibitor alone were 31.99 ± 21.96, 41.55 ± 35.17, and 30.95 ± 19.56 days, respectively (Hvenegaard et al., 2011). Although we did not analyze healing time by type of medication, the range of healing time for medication alone in our study was from 10 to 30 days. It should be considered that various types of cornea damage also play a role in healing time (Kern, 1990). With the nictitating membrane flap procedure, the corneal healing rate may indirectly be enhanced by inhibiting the movement of the membrane, while moisturizing the injured cornea with precorneal tear film (Magg et al., 2013; Giuliano, 2015). Furthermore, a nictitating membrane flap may be a selective choice in cases with limited health conditions to receive general anesthesia, poor economic status, or prevention of progressive corneal damage. However, in a progressive deep stromal ulcer (involving more than 50% of stromal thickness), infectious ulcerative keratitis, or uncontrolled animal, surgical intervention such as conjunctival, corneal, amniotic membrane, or biomaterial grafts is recommended (Gelatt and Plummer, 2022), in association with primary cause elimination.
Fig. 1. Comparison of the healing time between two treatment groups.
Fig. 2. Comparison of the healing time between treatment groups and breeds.
Fig. 3. Comparison of the healing time between treatment groups and ages. The brachycephalic breed is a major risk factor for canine ulcerative keratitis and keratomalacia (Tsvetanova et al., 2021). In this study, the majority of dogs are brachycephalic. Shih Tzu is the most common breed and it is known for a predisposition to keratoconjunctivitis sicca that possibly leads to ulcerative keratitis (Kim et al., 2009; Costa et al., 2021). When proteinase activity is uncontrolled during corneal wound healing, keratomalacia usually occurs and is in association with deep ulcerative keratitis. The healing rate of melting keratitis was only 50% when receiving intensive medical treatment (Guyonnet et al., 2020). Additional supportive therapy such as a nictitating membrane flap may be required to enhance the healing rate of deep corneal ulcers by preventing self-trauma and providing corneal moisture. The proportion of dogs older than 10 years receiving combined treatment was lower than the proportion of dogs that received medication only. This could imply that age is one of the important factors when selecting appropriate treatment. We also observed that the healing time in dogs older than 10 years receiving either medication only or combined treatment was longer than that of dogs younger than 10 years. The prolonged healing time in older animals may be due to the lack of healing growth factors in these animals. In humans, the corneal healing process is delayed in elderly patients. This is caused by an accumulation of caveolin-1, one of the integral proteins, which is found to be age-dependent and hypo responsive to growth factors in corneal epithelial cells (Rhim et al., 2010). Moreover, malfunctions of age-related ocular surface homeostasis had been reported in many previous studies; which are lower lacrimation rate, meibomian gland malfunction, lower keratocyte, and corneal nerve density, decrease corneal endothelial cell, lower systemic immunological function (Berlau et al., 2002; Hartley et al., 2006; Weiskopf et al., 2009; Knop et al., 2011; Gipson, 2013; Dees et al., 2017; Iwashita et al., 2020), all of which exacerbated corneal ulcerative keratitis severity. It would be interesting to further investigate corneal growth factors in aging dogs. There are limitations in this retrospective study. First, the evaluation of the exact depth, width, and location of the ulcer is limited. Various depths of the corneal ulcer may lead to different healing times. Measurement of the depth and width of corneal ulcers with the use of image evaluation programs will provide good evidence of the corneal wound healing process. Second, corneal culture was not performed to rule out ocular surface microorganisms that might prolong healing time. A prospective randomized control study should be performed to evaluate the difference in healing time between therapeutic methods. In conclusion, the healing time of the non-infectious deep ulcerative keratitis not exceeding 50% thickness of the cornea between medical treatment alone and medical therapy combined with a nictitating membrane flap is relatively comparable. While medications are definitely required, a nictitating membrane flap can be additionally considered not only to prevent complications but to support the healing process. Special attention should be concerned to senile and brachycephalic dogs. AcknowledgmentsThis study was financially supported by the Senior project funding, Faculty of Veterinary Science, Chulalongkorn University. The authors are sincerely grateful for support from the staff of the Ophthalmology Clinic, Small Animal Teaching Hospital, Faculty of Veterinary Science, Chulalongkorn University. ReferencesBalland, O., Poinsard, A.S., Famose, F., Goulle, F., Isard, P.F., Mathieson, I. and Dulaurent, T. 2016. Use of a porcine urinary bladder acellular matrix for corneal reconstruction in dogs and cats. Vet. Ophthalmol. 19, 454–463. Berlau, J., Becker, H.H., Stave, J., Oriwol, C. and Guthoff, R.F. 2002. Depth and age-dependent distribution of keratocytes in healthy human corneas: a study using scanning-slit confocal microscopy in vivo. J. Cataract Refract. Surg. 28, 611–616. Costa, J., Steinmetz, A. and Delgado, E. 2021. Clinical signs of brachycephalic ocular syndrome in 93 dogs. Irish Vet. J. 74, 1–8. Crispin, S. 2002. The cornea. BSAVA manual of small animal ophthalmology. Gloucester, UK: British Small Animal Veterinary Association. Dees, D.D., Fritz, K.J., Wagner, L., Paglia, D., Knollinger, A.M. and Madsen, R. 2017. Effect of bandage contact lens wear and postoperative medical therapies on corneal healing rate after diamond burr debridement in dogs. Vet. Ophthalmol. 20, 382–389. Farghali, H.A., Naglaa A. AbdElKader, N.A., AbuBakr, H.O., Ramadan, E.S., Khattab, M.S., Salem, N.Y. and Emam, I.A. 2021. Corneal ulcer in dogs and cats: novel clinical application of regenerative therapy using subconjunctival injection of autologous platelet-rich plasma. Front. Vet. Sci. 8, 1–17. Gelatt, K.N. and Plummer, C.E. 2022. Essentials of veterinary ophthalmology. Ames, IA: Wiley Blackwell. Gilger, B.C., Ollivier, F.J. and Bentley, E. 2007. Diseases and surgery of the canine cornea and sclera. In Veterinary ophthalmology, 4th ed. Ed., Gellatt, K.N. Ames, IA: Wiley-Blackwell, pp: 690–752. Gipson, I.K. 2013. Age-related changes and diseases of the ocular surface and cornea. Invest. Ophthalmol. Vis. Sci. 54, 48–53. Giuliano, E.A. The third eyelid: time to get into a flap. In BSAVA Congress Proceedings, BSAVA Library, Birmingham, UK, 2015, pp: 47–48. Guyonnet, A., Desquilbet, L., Faure, J., Bourguet, A., Donzel, E. and Chahory, S. 2020. Outcome of medical therapy for keratomalacia in dogs. J. Small Anim. Pract. 61, 253–258. Hartley, C., Williams, D.L. and Adams, V.J. 2006. Effect of age, gender, weight, and time of day on tear production in normal dogs. Vet. Ophthalmol. 9, 53–57. Hvenegaard, A.P, Vieira, J.E., Leandro, D.C., Góes, A.C., Safatle, A. and Barros, P.S. 2011. Retrospective study on clinical management of indolent ulcers in Boxer dogs. Pesq. Vet. Bras. 31, 910–915. Iwashita, H., Wakaiki, S., Kazama, Y. and Saita, A. 2020. Breed prevalence of canine ulcerative keratitis according to depth of corneal involvement. Vet. Ophthalmol. 23, 849–855. Jaksz, M., Fischer, M.C., Fenollosa-Romero, E. and Busse, C. 2021. Autologous corneal graft for the treatment of deep corneal defects in dogs: 15 cases (2014-2017). J. Small Anim. Pract. 62, 123–130. Jhala, S.K., Joy, N., Mehraj, D.D., Tiwari, D.K., Patil, D.B. and Parikh, P.V. 2011. Management of corneal ulcer using third eyelid flap technique in a horse. Indian J. Vet. Surg. 33, 73. Kern, T. 1990. Ulcerative keratitis. Vet. Clin. North Am. Small Anim. Pract. 20, 643–666. Kim, J.Y., Won, H.J. and Jeong, S. 2009. A retrospective study of ulcerative keratitis in 32 dogs. Intern. J. Appl. Res. Vet. Med. 7, 27–31. Knop, E., Knop, N., Millar, T., Obata, H. and Sullivan, D.A. 2011. The International Workshop on Meibomian Gland Dysfunction: report of the Subcommittee on anatomy, physiology, and pathophysiology of the meibomian gland. Invest. Ophthalmol. Vis. Sci. 52, 1938–1978. Kumar, T., Punia, M., Agnihotri, D., Sindhu, N. and Jain, V.K. 2018. Incidence of ophthalmic affections in dog—a short study. Int. J. Curr. Microbiol. App. Sci. 7, 1560–1565. Ledbetter, E.C. and Gilger, B.C. 2013. Diseases and surgery of the canine cornea and sclera. In Veterinary ophthalmology, 5th ed. Eds., Gellatt, K.N., Gilger, B.C. and Kern, T.J. Ames, IA: John Wiley & Sons, pp: 976–1059. Ledbetter, E.C., Norman, M.L. and Starr, J.K. 2016. In vivo confocal microscopy for the detection of canine fungal keratitis and monitoring of therapeutic response. Vet. Ophthalmol. 19, 220–229. Ljubimov, A.V. and Saghizadeh, M. 2015. Progress in corneal wound healing. Prog. Retin. Eye Res. 49, 17–45. Maggs, D.J., Miller, P.E. and Ofri, R. 2013. Cornea and sclera. Slatter’s fundamentals of veterinary ophthalmology, 5th ed. St. Louis, MO: Elsevier, pp: 184–219. Martin, C.L. 2005. Cornea and sclera. Ophthalmic disease in veterinary medicine. London, UK: Manson Publishing, pp: 241–297. Ollivier, F.J., Gilger, B.C., Barrie, K.P., Kallberg, M.E., Plummer, C.E., O’Reilly, S., Gelatt, K.N. and Brooks, D.E. 2007. Proteinases of the cornea and preocular tear film. Vet. Ophthalmol. 10, 199–206. O’Neill, D.G., Lee, M.M., Brodbelt, D.C., Church, D.B. and Sanchez, R.F. 2017. Corneal ulcerative disease in dogs under primary veterinary care in England: epidemiology and clinical management. Canine Genet. Epidemiol. 4, 1–12. Pirdankar, O.H., Kenia, R.V. and Kenia, V.P. 2020. Diagnosis and management protocol of acute corneal ulcer. Int. J. Health Sci. 10, 69–78. Rhim, J.H., Kim, J.H., Yeo, E.J., Kim, J.C. and Park, S.C. 2010. Caveolin-1 as a novel indicator of wound healing capacity in aged human corneal epithelium. Mol. Med. 16, 527–534. Santillo, D., Mathieson, I., Corsi, F., Göllner, R. and Guandalini, A. 2021. The use of acellular porcine corneal stroma xenograft (BioCorneaVet™) for the treatment of deep stromal and full thickness corneal defects: a retrospective study of 40 cases (2019–2021). Vet. Ophthalmol. 24, 469–483. Slatter, D. 2001. Principles of ophthalmic surgery. Fundamentals of veterinary ophthalmology. St. Louis, MO: W.B. Saunders Company. Spiess, B.M., Pot, S.A., Florin, M. and Hafezi, F., 2014. Corneal collagen cross-linking (CXL) for the treatment of melting keratitis in cats and dogs: a pilot study. Vet. Ophthalmol. 17, 1–11. Tsvetanova, A., Powell, R.M., Tsvetanova, K.A., Smith, K.M. and Gould, D.J. 2021. Melting corneal ulcers (keratomalacia) in dogs: a 5-year clinical and microbiological study (2014-2018). Vet. Ophthalmol. 24, 265–278.ª Vongsakul, S., Tuntivanich, P., Sirivaidyapong, S. and Kalpravidh, M. 2009. Canine amniotic membrane transplantation for ocular surface reconstruction of created deep corneal ulcers in dogs. Thai. J. Vet. Med. 39, 135–144. Wang, A.G., Wu, C.C. and Liu, J.H. 1998. Bacterial corneal ulcer: a multivariate study. Ophthalmologica 212, 126–132. Weiskopf, D., Weinberger, B. and Grubeck-Loebenstein, B. 2009. The aging of the immune system. Transpl. Int. 22, 1041–1050. Williams, D.L. and Burg, P. 2017. Tear production and intraocular pressure in canine eyes with corneal ulceration. Open Vet. J. 7, 117–125. | ||
| How to Cite this Article |
| Pubmed Style KI, Pinyosnit N, Srilert P, Ngampongsai T, Hunprasit V, Tuntivanich N, Jaturakan O. Comparative study of healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and combined treatment with medical therapy and a nictitating membrane flap: A retrospective study. Open Vet J. 2022; 12(6): 815-821. doi:10.5455/OVJ.2022.v12.i6.5 Web Style KI, Pinyosnit N, Srilert P, Ngampongsai T, Hunprasit V, Tuntivanich N, Jaturakan O. Comparative study of healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and combined treatment with medical therapy and a nictitating membrane flap: A retrospective study. https://www.openveterinaryjournal.com/?mno=48209 [Access: July 27, 2024]. doi:10.5455/OVJ.2022.v12.i6.5 AMA (American Medical Association) Style KI, Pinyosnit N, Srilert P, Ngampongsai T, Hunprasit V, Tuntivanich N, Jaturakan O. Comparative study of healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and combined treatment with medical therapy and a nictitating membrane flap: A retrospective study. Open Vet J. 2022; 12(6): 815-821. doi:10.5455/OVJ.2022.v12.i6.5 Vancouver/ICMJE Style KI, Pinyosnit N, Srilert P, Ngampongsai T, Hunprasit V, Tuntivanich N, Jaturakan O. Comparative study of healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and combined treatment with medical therapy and a nictitating membrane flap: A retrospective study. Open Vet J. (2022), [cited July 27, 2024]; 12(6): 815-821. doi:10.5455/OVJ.2022.v12.i6.5 Harvard Style , K. I., Pinyosnit, . N., Srilert, . P., Ngampongsai, . T., Hunprasit, . V., Tuntivanich, . N. & Jaturakan, . O. (2022) Comparative study of healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and combined treatment with medical therapy and a nictitating membrane flap: A retrospective study. Open Vet J, 12 (6), 815-821. doi:10.5455/OVJ.2022.v12.i6.5 Turabian Style , Kanokpich Imcharoon, Natawadee Pinyosnit, Pimpika Srilert, Tanakanok Ngampongsai, Vachira Hunprasit, Nalinee Tuntivanich, and Orapun Jaturakan. 2022. Comparative study of healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and combined treatment with medical therapy and a nictitating membrane flap: A retrospective study. Open Veterinary Journal, 12 (6), 815-821. doi:10.5455/OVJ.2022.v12.i6.5 Chicago Style , Kanokpich Imcharoon, Natawadee Pinyosnit, Pimpika Srilert, Tanakanok Ngampongsai, Vachira Hunprasit, Nalinee Tuntivanich, and Orapun Jaturakan. "Comparative study of healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and combined treatment with medical therapy and a nictitating membrane flap: A retrospective study." Open Veterinary Journal 12 (2022), 815-821. doi:10.5455/OVJ.2022.v12.i6.5 MLA (The Modern Language Association) Style , Kanokpich Imcharoon, Natawadee Pinyosnit, Pimpika Srilert, Tanakanok Ngampongsai, Vachira Hunprasit, Nalinee Tuntivanich, and Orapun Jaturakan. "Comparative study of healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and combined treatment with medical therapy and a nictitating membrane flap: A retrospective study." Open Veterinary Journal 12.6 (2022), 815-821. Print. doi:10.5455/OVJ.2022.v12.i6.5 APA (American Psychological Association) Style , K. I., Pinyosnit, . N., Srilert, . P., Ngampongsai, . T., Hunprasit, . V., Tuntivanich, . N. & Jaturakan, . O. (2022) Comparative study of healing time of canine non-infectious deep ulcerative keratitis between medical therapy alone and combined treatment with medical therapy and a nictitating membrane flap: A retrospective study. Open Veterinary Journal, 12 (6), 815-821. doi:10.5455/OVJ.2022.v12.i6.5 |