| Original Article | ||
Open Vet J. 2022; 12(3): 360-369 Open Veterinary Journal, (2022), Vol. 12(3): 360–369 Original Research Serum digoxin concentrations in dogs with atrial fibrillation under long term digoxin treatmentAntonella Colella1, Serena Digiaro1, Marco Fiorentino2, Beatrice Greco1, Mariateresa Sasanelli1 and Paola Paradies1*1University of Bari, Department of Emergency and Organ Transplantation, Veterinary Clinics and Animal Production Section, Valenzano (BA), Italy 2University of Bari, Department of Emergency and Organ Transplantation, Nephrology, Dialysis and Transplantation Unit, Bari (BA), Italy *Corresponding Author: Prof. Paola Paradies. University of Bari, Department of Emergency and Organ Transplantation, Veterinary Clinics and Animal Production Section, Valenzano (BA), Italy. Email: paola.paradies [at] uniba.it Submitted: 28/02/2022 Accepted: 16/05/2022 Published: 03/06/2022 © 2022 Open Veterinary Journal
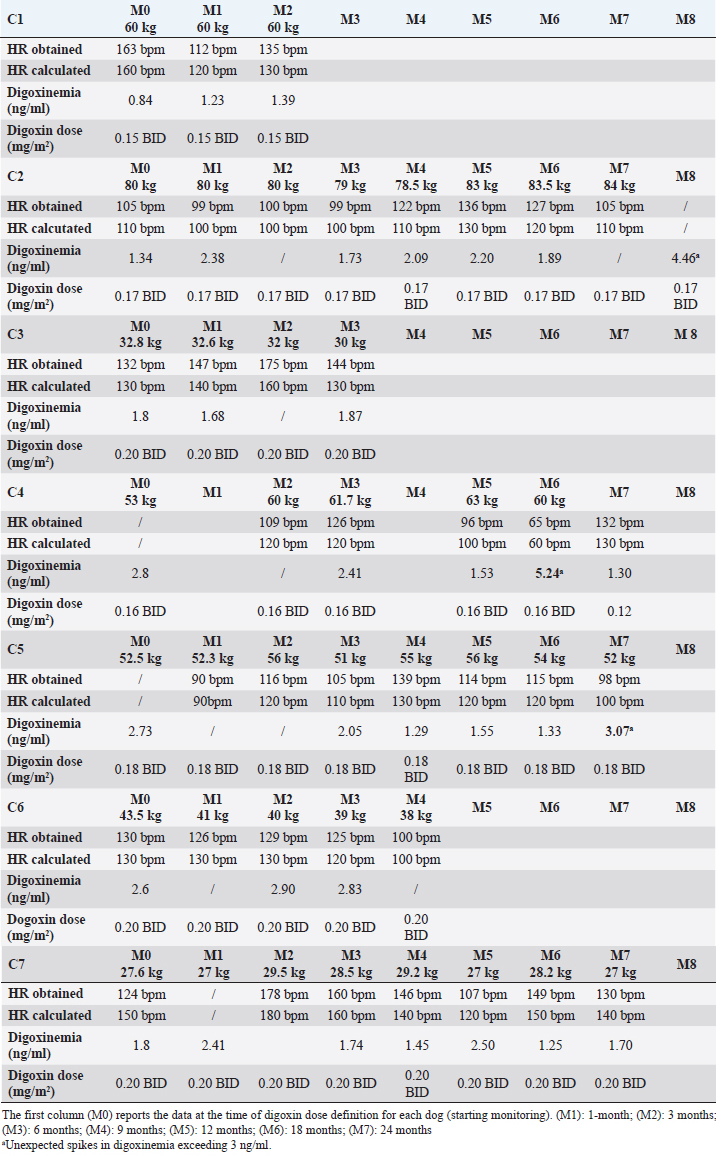
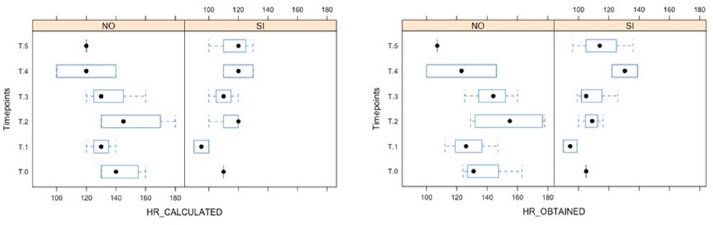
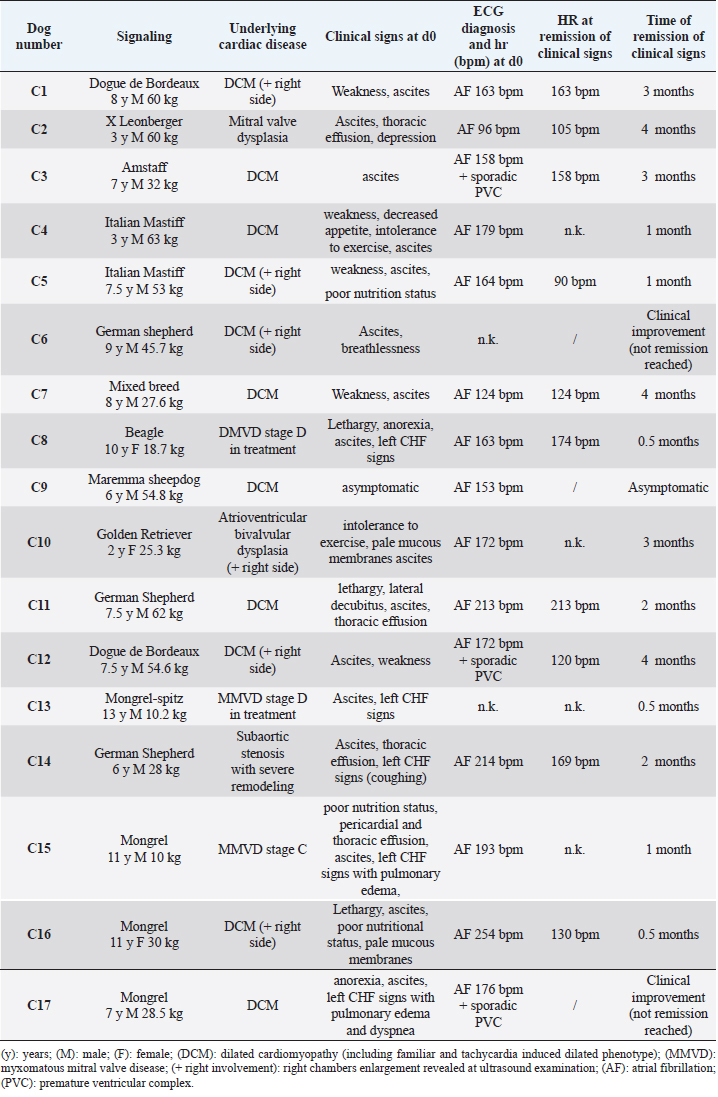
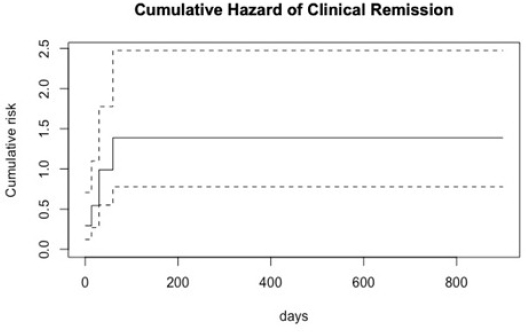
AbstractBackground: Atrial fibrillation (AF) is the most common arrhythmia in dogs, most frequently diagnosed as chronic AF associated with a structural heart disease. The therapeutic strategy, in these cases, is based on the heart rate control and digoxin is one of the most used drugs. Aim: The aim of this work was to study the serum digoxin concentration changes in dogs with AF under long-term treatment with digoxin. Furthermore, the remission of clinical signs and the correlation between digoxinemia and other clinical and laboratory variables were retrospectively evaluated. Methods: The prospective study was conducted on seven large breed dogs from the time of reaching the definitive digoxin dosage. Digoxinemia was determined at month: 1, 3, 6, 9, 12, then twice a year. A post hoc statistical analysis investigated the influence of selected clinical and laboratory variables on the risk to develop spikes in digoxinemia. Clinical data, heart rate, digoxin dosage (mg/m2), and digoxinemia (ng/ml) at all available follow-ups were retrospectively evaluated from the medical records of 17 further dogs and a linear regression analysis was performed on the whole data set. The relation between the time of remission of AF clinical signs and variables was also investigated. Results: An unexpected increase in digoxin serum concentration was recorded in three dogs after one year monitoring, in absence of digoxin dosage changes. No statistical significance of all the studied variables on the risk to develop spikes of digoxinemia was registered. Two dogs, reaching digoxinemia 4.46 and 5.24 ng/ml, showed symptoms that reversed after digoxin withdrawal. From retrospective data, 88% of dogs reached complete reverse of AF clinical signs in 2.1 months from digoxin treatment starting, regardless of digoxin initial dosage, digoxinemia, and heart rate. Conclusion: Digoxin in monotherapy remain a good option to treat AF in dogs, anyway digoxin toxicity could emerge during long-term therapy, similarly to what happen in human medicine. Life-threatening spikes of digoxinemia could occur, especially after 1-year treatment with digoxin. It is very important that practitioners be aware of this possibility and encourage the owners to monitor digoxinemia during long-term treatment to avoid dangerous and toxic effects. Keywords: Atrial fibrillation, Digoxin, Digoxin serum concentration, Dogs. IntroductionAtrial fibrillation (AF) is the most common arrhythmia in dogs with a recently reported incidence of 33.7% of all arrhythmias found on electrocardiographic examination (ECG) (Noszczyk-Nowak et al., 2017). It is related to a high mortality rate if left untreated (Bonagura, 1986; Stokhof and van den Brom, 1990; Jung et al., 2016; Pedro et al., 2018). This arrhythmia is characterized by a rapid, chaotic and disorganized atrial electrical activity, which results in the loss of an effective contraction of the atria, functional to ventricular diastolic filling, and high ventricular rate (Pedro et al., 2020a). The main hemodynamic consequences are a reduction in cardiac output and increased atrial filling pressures with the development or worsening of clinical signs of congestive heart failure (CHF) (Saunders et al., 2009). AF is most frequently diagnosed in association with underlying structural heart diseases, and it is often associated with CHF (Pariaut, 2017; Ward et al., 2019). Atrial enlargement, fibrous remodeling, elevated adrenergic tone, and activation of the renin–angiotensin–aldosterone system represent irreversible changes that promote the onset and maintenance of AF (Pariaut, 2017). Once established, AF contributes to further deterioration of cardiac function mainly due to the rapid and irregular ventricular response rate (VRR) which is the result of high adrenergic tone associated with a reduced ability of the atrioventricular node to block atrial impulses (Pariaut, 2017). In canine clinical practice, AF is a well-known and frequent arrhythmia. Scientific papers on dogs have been recently published documenting and discussing the association between AF and clinical signs of right CHF, the association between a post-treatment low heart rate (HR) and a longer survival time (Pedro et al., 2018; Ward et al., 2019). The role of AF as a prognostic factor in dogs with myxomatous mitral valve disease (MMVD) and dilated cardiomyopathy (DCM) in Dobermans has been investigated (Friederich et al., 2020; Guglielmini et al., 2020). Up until now, the primary therapeutic aim to treat AF in veterinary medicine is the maximization of cardiac output through two treatment strategies: the HR control or the conversion of AF to sinus rhythm (Saunders et al., 2009; Pariaut, 2017; Pedro et al., 2020a,b). The latter is reserved for a few cases (i.e., idiopathic AF), while in most cases (chronic AF associated with structural heart disease) the treatment strategy involves the use of drugs aimed to decrease VRR. Digoxin alone or in combination with other medications is one of the most commonly used drug to treat AF in dogs (Pariaut, 2017). However, prospective clinical trials including long-term safety of digoxin are missing as reported in a recent review from Pedro and collaborators (2020b). It is known from human medicine that digoxin toxicity emerges mostly during long-term therapy and that clinical features of toxicity are often nonspecific with lethargy, gastrointestinal signs, neurological symptoms, or arrhythmias that could be fatal (Tatlisu et al., 2015; Pincus, 2016). The purpose of the present work was to study the changes of serum digoxin concentration in dogs with DCM and AF under long-term treatment with digoxin. Furthermore, data from other dogs under digoxin treatment were retrospectively studied to evaluate the reversal of clinical signs and the correlations of digoxinemia with clinical, laboratory and instrumental variables. Materials and MethodsProspective studyIn dogs with a diagnosis of chronic AF, having the owners’ availability for serial and constant follow-ups, a prospective long-term serial monitoring was performed with attention to clinical and ECGs evaluation and serum digoxin concentration values. Seven dogs were included in the study (2 mixed breed, 1 German Shepherd, 1 Newfoundland, 1 Dogue de Bordeaux, 1 Italian Mastiff, 1 American Staffordshire) aging from 3 to 9 years (mean 6.5 years) with diagnosis of AF and a DCM phenotype at echocardiography and monitored under treatment with digoxin as the sole antiarrhythmic agent. Informed consent was obtained from the dog owners and contemporary standard of care was provided to each animal. The starting point for serial monitoring (M0) was considered the time at which the digoxin dose was fixed for each single dog after required adjustments. Monitoring was performed as follow: at 1 month (M=1), then each three months till 1 year (M2, M3, M4, and M5) and then each 6 months (M6 and over) till the end of the study corresponding to the patient’s death. At each follow-up, the animals underwent a complete clinical examination, including clinical evaluation of HR, blood sampling for haemato-biochemical exams, and digoxin serum concentration value and an ECG. A post hoc statistical analysis was performed to investigate the influence of the trend of clinical and laboratory variables during the first-year of follow up (clinical HR, ECG obtained and calculated HR, digoxinemia ng/ml, digoxin dosage mg/m2, weight kg, number of premature ventricular complexes (PVCs) in 3ʹ, hemoglobin g/dl, hematocrit %, white blood cells ×1,000/µl, platelets 1,000/µl, urea mg/dl, creatinine mg/dl, phosphorus mg/dl, potassium mEq/l, albumins g/dl, total proteins g/dl) on the risk to develop spikes in digoxinemia after the first year. Two groups were created based on dogs showing spikes compared to dogs having no spikes. The comparison between the variables and the two groups was performed using linear mixed models fit by REML. All analyses were performed using the R Package (version 1.2.1093), and a p-value <0.05 was considered to be statistically significant. Retrospective studyThe medical records of dogs affected by AF and treated with digoxin in monotherapy were revised. Dogs ≥ 10 kg with a complete medical record, an ECG and an echocardiographic examination at the time of diagnosis (D0), and who had at least two further follow-ups were included in the study. The study period started from D0, included the follow ups for digoxin dose adjustment and continued till the last available follow up for each dog over time. Clinical signs at presentation and the time of remission after treatment starting were reported for each dog. Ascites with or without other cavitary effusions and weakness were considered AF clinical signs. Remission of AF clinical signs was considered if all effusions resolves (quantified by repeated ultrasound scans) and weakness clearly improve (increased activity reported by the owner). Data available at all follow ups for each dog were registered including clinical HR, ECG HRs (obtained and calculated HR), digoxin dosage (mg/m2), and digoxinemia (ng/ml). A linear regression analysis was performed to determine correlations between variables considering the whole data set (data of all available follow ups of all dogs) using the Bravais–Pearson test (data reported as r coefficient and confidence intervals). Furthermore, a Cox regression analysis was performed to investigate the relation between study variables (i.e., HR at D0, ECG HR at D0, digoxin dosage at D0 and after adjustment, and digoxinemia at the first control 1 or 2 weeks after therapy starting) and the time of remission: data are expressed as coefficient, 95% confidence interval and p-value. Electrocardiographic examinationThe ECG was performed in standard right decubitus for at least 5 minutes monitoring by applying the four main electrodes to the four limbs, precisely at the level of elbows and knees after application of alcohol or gel. An Esaote electrocardiograph (Biosound Esaote P8000) was used. A printed ECG was also registered at each follow up in each dog. Dogs were not sedated, gently handled and kept in a darkened room to reduce the effects of stress. The ECG was evaluated to confirm the AF status, assess the presence of any concomitant rhythm disturbances and assess the VRR. To derive the VRR from the ECG two different methods were used based on the value determined by the instrument or applying the 10–20 method and defined respectively as “obtained HR” and “calculated HR.” For the determination of the obtained HR, the average HR determined by the instrument and reported on printed paper was considered. In particular, three HR values reported by the electrocardiograph on 70 cm of printer paper at a speed of 50 mm/s (14 sececonds) were chosen (the highest HR value, the lowest HR value, and the intermediate value), and the arithmetic mean calculated. To obtain calculated HR, the 10 or 20 method was used. In brief, the number of QRS complexes were counted in 15 cm of printed ECG corresponding to 3 seconds when registered at 50 mm/sec or 6 seconds when registered at 25 mm/sec and, respectively, multiplied by 20 or by 10. Serum digoxin concentrationA blood sample was taken between 8 and 10 hours after the drug administration. The serum was sent to the laboratory for the determination of serum digoxin concentration values. After therapy starting and in case of dose adjustment, a blood sample was taken after 1 or 2 weeks to determine serum digoxin concentration values. Other drugsThe administration of other antiarrhythmic drugs was considered an exclusion criterion. In the prospective study, all dogs were also treated with pimobendan (0.25 mg/kg/q12h), benazepril (0.25 mg/kg/q12h), and furosemide (0.5 mg/kg/ q12h). Variations in dosages if needed had to be approved and recorded. In the retrospective study, other drugs besides digoxin were administered to all animals based on the underlying cardiac disease and clinical status in a different combination and dosages for each dog (furosemide, pimobendan, benazepril, and spironolactone). Ethical approvalThe Research Ethics Committee of the Department of Emergency and Organs Transplantation, University of Bari, Italy, has approved the study (n. 227 III/13 on 07 February 2018). ResultsProspective studyTable 1 reports the HRs and the digoxinemia values from the moment of digoxin dose final adjustment M0 (obtained at least in 2 months in all dogs) until the end of monitoring. At M0 the digoxin dose ranged from 0.15 to 0.22 mg/m2 (with an average of 0.18 mg/m2) and the digoxinemia ranged from 0.84 to 2.8 ng/ml (with an average of 1.98 ng/ml). Original clinical signs of presentation (i.e., ascites, weakness) were reverted at this time. Mild digoxinemia fluctuations were recorded in the serial follow-ups in the first year of monitoring in all dogs, the clinical conditions were judged good and stable with neither relapse of AF clinical signs nor signs referable to digoxin intoxication. Three dogs died before M5 (1 year), the others were monitored for at least 2 years. In three out of the four dogs with longer follow-ups an unexpected increase in digoxinemia, exceeding 3 ng/ml, was documented in at least one of the monitoring times after M5. In particular, in two dogs (C2 and C4) serum digoxin concentration values had a sudden abnormal increase (reaching 4.46 and 5.24 ng/ml at M8 and M6, respectively) associated with signs of systemic disease (dysorexia, depression, and weakness) and severe bradyarrhythmia, which were attributed to digoxin-induced intoxication, also considering the reversibility of these signs after therapy withdrawal and reduction. No hemato-biochemical alterations were registered in these dogs. No statistical significance of all the studied variables trend (including the clinical and hemato-biochemical tests) in the first year of follow up on the risk to develop spikes in digoxinemia were reported using linear mixed models, with the exception for “obtained” HR (estimate coefficient −26.8, 95% CI: −41.06 to −12.46, p=0.01) and “calculated” HR (estimate coefficient −24.6, 95% CI: −39.7 to −9.16, p=0.02) (Fig. 1). In brief, dogs showing an HR reduction trend during the first year of monitoring had a reduced risk to develop spikes in digoxinemia in the further monitoring. Retrospective studySeventeen dogs with AF were included in the retrospective study. Of these dogs, 14 were males and 3 were females, aged between 2 and 13 years (an average of 7.4 years). Clinical information regarding the signaling, the clinical signs at D0, the diagnosis of underlying heart disease obtained through echocardiographic examination and collateral examinations, the presence of signs referable to concomitant left CHF are reported in Table 2. All dogs, except for one (C9), showed clinical signs referable to AF at D0, with ascites as a constant finding. All dogs had general improvement (increased activity referred by the owner, reduction of effusion quantified by body weight) 2 weeks after therapy starting, regardless the digoxin starting dosage. All dogs, except for two, reached complete remission of clinical signs within 4 months from D0 and precisely between 0.5 and 4 months with an average of 2.1 months (Fig. 2). Only two dogs (C6 and C17) reached clinical improvement but not complete remission of clinical signs. The remission of clinical signs was apparently independent from HR at time of remission, which ranged between 90 and 213 bpm. At the Cox regression analysis, no associations between the clinical remission and the studied variables (digoxin dosage, digoxinemia, and HR) were revealed. Table 1. Long-term follow-up of dogs included in the prospective study (n=7). ECG HR, digoxinemia values and digoxin dosages for each follow up are reported.
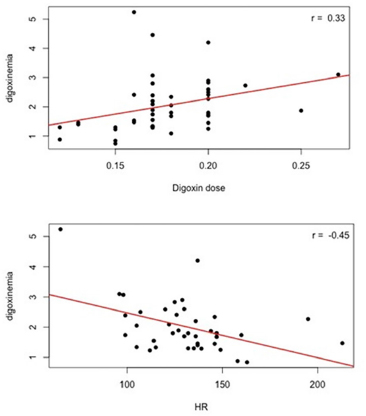
Fig. 1. Post hoc analysis. Linear mixed model fit by REML showing that in dogs of group NO, obtained and calculated HR tend to decrease during the first-year monitoring (p=0.01 and 0.02, respectively), while they tend to increase in the SI group. NO=group of dogs that do not show spikes in digoxinemia, SI=group of dogs showing spikes. The linear regression analysis on the whole data set (from 91 follow up on 17 dogs), documents a mild positive correlation between digoxinemia and digoxin dose (n=46, r=0.33, 95% CI: −0.670, −0.166, p-value=0.023) and a discrete negative correlation between digoxinemia and ECG obtained HR (n=40, r=−0.45, 95% CI: −0.559, −0.020, p-value=0.003) (Fig. 3). DiscussionIn the prospective study, all included dogs were large breed dogs with a DCM phenotype. Two purebred dogs had a documented familial predisposition for DCM (Newfoundland and German Shepherd), in the other cases the dilated hypokinetic picture may be the consequence of the arrhythmia rather than the predisposing factor, as suggested from experimental pacing models (Armstrong et al., 1986; Wilson et al., 1987). All dogs had shown AF clinical signs before treatment starting, with ascites as a constant finding. Serial monitoring began from the time the final digoxin dosage was reached after the required adjustments and AF showed clinical remission. It is known that the suggested starting dosage of digoxin is 0.22 mg/m2 or 0.005 mg/kg/BID, depending on dog’s size, but due to the different individual pharmacokinetics and the reduced safety margin, it may require adjustments based on the serum digoxin concentration. A target steady-state plasma concentration (8 hours post pill) of 0.8–1.5 ng/ml and 0.6–1.0 ng/ml has been recently suggested (Keene et al., 2019; Pedro et al., 2020b), in particular to avoid the pro-arrhythmic effect due to the increase in intracellular calcium levels. It has been further suggested that digoxin dosing should be tailored to a tolerable serum level (without signs of toxicity), not by effect on HR (Keene et al., 2019; Pedro et al., 2020b). In the past higher values till 2.5 ng/ml were reported as reference range (Pedersoli, 1978; Fox et al., 1999; Couto and Nelson, 2015). Anyway, on authors’ knowledge, no scientific data are available in the literature to define a univocal reference range for digoxinemia in dogs, in particular during long term monitoring. Once defined, the digoxin dose was no more changed until the end of the study. In all dogs and in all follow-ups, HRs remained consistently below 150 bpm in the evaluation obtained from the ECGs and no clinical relapses where registered. It has been reported that an HR lower than 140 bpm for 60% of the daytime in Holter monitoring or an average HR of 150 bpm on the ECG are considered indicators of AF adequate control (Pariaut, 2017). No posology changes in the other scheduled treatments were required. Table 2. Signaling, underlying cardiac disease, clinical signs and electrocardiographic diagnosis at D0 in the 17 dogs included in the retrospective study.
Fig. 2. Cumulative Hazard Curve showing that clinical remission was reached in all dogs at the beginning of monitoring (maximum 120 days) and was maintained throughout the study. Digoxinemia serial monitoring showed only slight fluctuations in the first-year monitoring. Differently, unexpected spikes in digoxinemia were recorded in three dogs (C2, C4, and C5) after 1 year monitoring despite treatment posology remained constant. Since only mild weight variations were registered in dogs during the monitoring, it is difficult to attribute the increase of digoxinemia to weight changes. Furthermore, the haemato-biochemical parameters did not show significative alterations to consider the altered metabolism of digoxin due to organ failure as the cause of the abnormal increase in serum digoxin. In fact, the elimination of digoxin is mainly by renal clearance, transport by P-glycoprotein also contributes to elimination. Consequently, a higher serum digoxin concentration for a given dose occurs in patients with renal impairment, weight loss and in those taking drugs (amiodarone, verapamil, macrolides, azole antifungals, and cyclosporin), which inhibit P-glycoprotein transport, which is not the case of our dogs. Other conditions could increase sensitivity to digoxin including electrolytic and acid-base disturbances, myocardial ischemia and hypoxemia (Pincus, 2016). The spikes in digoxinemia registered in dogs in this study were apparently unwarranted. The reasons inducing the spikes remain unknown and need further investigation. In fact, the post hoc statistical analysis results showed no influence of all the studied clinical and haemato-biochemical variables on the risk to develop spikes in digoxinemia during a long-term follow up, except for the ECG HR. It could be argued that dogs having a stronger HR reduction trend in the first-year monitoring had a less severe progression of the underling structural disease influencing the HR response and reducing hypoxemia. Further studies need to address the point.
Fig. 3. Linear regression correlation curve showing the mild positive correlation between digoxin dose and digoxinemia (r=0.33) and the negative correlation between the ECG HR and digoxinemia (r=−0.45) on the whole data collected throughout the study. Curves using obtained and calculated ECG HR were overlapping. In two dogs (C2 and C4), spikes of digoxinemia (>4 ng/ml) were associated with bradyarrhythmia (HR 40 bpm in C2 and 65 bpm in C4) and with signs of systemic disease (weakness, depression and dysorexia). These signs and HR reverse to normal with the temporary suspension of therapy and following dose reduction. It is known that the mean elimination half-life in dogs is 73 hours; therefore, if digoxin toxicity is suspected, digoxin should be discontinued for several days, and then restarted at a lower dose (Kuhlmann et al., 1979). In our study, after the first year of treatment we scheduled follow ups twice a year. Based on our data, it could be suggested that monitoring of digoxinemia need to be scheduled more frequently to prevent intoxication. Signs of intoxication could be underestimated by the owner because often vague, non-specific and misinterpreted as a progression of underlying cardiac disease. In the retrospective study, all dogs diagnosed with AF had underlying heart disease with a prevalence of 64% of subjects affected by DCM, 18% by MMVD, and 18% by congenital pathologies, accordingly to the prevalence previously reported (Pedro et al., 2018; Ward et al., 2019). DCM and MMVD are known to be the most frequently structural disease associated with AF (Bonagura, 1986; Guglielmini et al., 2000; Van den Berg et al., 2002; Menaut et al., 2005; Pedro et al., 2018; Guglielmini et al., 2020). All dogs, except for one, were symptomatic due to AF at the time of diagnosis. Ascites was a constant clinical sign, sometimes associated with other cavity effusions and weakness. The development of signs of right CHF in dogs with AF and left-sided structural heart diseases (MMVD and DCM) is explained by the fact that AF causes an acute increase in pressure in the right atrium that is hemodynamically more significant than the increase in pressure in an already dilated left atrium (Ward et al., 2019). It is interesting to note that only six dogs out of 16 with ascites had right side remodeling at echocardiography (i.e., eccentric hypertrophy, right atrium dilation). The reversal of AF clinical signs occurred in 88.2% of dogs (15/17) and was achieved within a maximum of 4 months from therapy starting (with an average of about 2 months), regardless the initial digoxin dosage, digoxinemia and HR. The remission of clinical signs resulted independent from HR registered at ECG at time of remission. Anyway, it could be possible that HR was influenced by the emotional status of the dog in an ambulatory setting, or the chosen statistical method may not have enough power to capture the correlation of HR to clinical remission (Gelzer et al., 2015; Pariaut, 2017). The possible effect of concurrent treatments on HR reduction and clinical improvement needs also to be considered in this study, particularly in dogs with AF secondary to decompensated cardiac diseases. In fact, drugs employed for treatment of CHF can have a positive effect in reducing high ventricular rate in dogs with concurrent AF. In general, from the evaluation of HRs data throughout the study, clinical HRs differ from ECG HR values; while ECG calculated and obtained HR values were quite overlapping. Cardiac auscultation is recognized inaccurate for HR estimation in dogs with AF (Glaus et al., 2003). The method of evaluation through the 1-to-5-minute ECG in clinical practice is a reliable method but tends to overestimate the HR for increased sympathetic stimulus. Holter remains the recommended method, but it is not always practicable and can be expensive. From the whole data set analysis, digoxin serum concentration resulted only mildly correlated to the digoxin dosage probably because of the individual response as already known and negatively correlated with ECG HR, indicating that the negative chronotropic effect of the digoxin drug is influenced by the serum concentration (Teske et al., 1976; Nagashima et al., 2001). Although the superiority of the association of digoxin with diltiazem has been suggested to more effectively slow HR even during exercise, excitement, and stress, in the prospective study, digoxin monotherapy was chosen under the owner authorization to avoid the influence of other variables on digoxinemia (Gelzer et al., 2009; Jung et al., 2016; Pariaut, 2017). In the retrospective study dogs have been treated in monotherapy mostly under owner concerns to give further drugs. Results of this study show that AF signs can be controlled with digoxin alone. When owner compliance is improved by limiting the number of cardiac medications prescribed to his pet, AF treatment in monotherapy is a good option to suggest. Limitations of this study include, among the others, the small dog population both in the retrospective and in the prospective study (particularly for a post hoc analysis), the HR evaluation by ECG and not by Holter which represent the gold standard, the concomitant administration of other cardiologic drugs to control the underlying disease that could have influenced some data. ConclusionResults of this study suggest that digoxin in monotherapy still remain a good option to treat AF in dogs since signs associated with AF can be controlled and reversed. Anyway, due to its narrow therapeutic index and toxicity, digoxin should be used carefully. In this study, the importance of long-term monitoring is emphasized since digoxin toxicity could emerge during long-term therapy similarly to what happen in human medicine. Long-term monitoring of digoxin serum concentration can reveal increases that require suspension or reduction of dosage to avoid dangerous and toxic effects, especially after one year treatment. It should be scheduled not only in patients in which weight changes, organs failure (azotemia, renal failure) or concomitant diseases occur, but in all patients under treatment. In fact, spikes in digoxinemia could occur independently from clinical and biochemical changes. In clinical practice, owners of dogs with good clinical control of AF mostly continue treatment at the same dose life-long; furthermore, clinical features of toxicity in dogs are often not-specific and misinterpreted from the owner as a progression of underlying cardiac disease. Thus, it is very important that practitioners are aware of this possibility and encourage the owners to serial monitoring of digoxinemia to avoid intoxication that could be fatal. Conflict of interestThe Authors declare that there is no conflict of interest. Authors’ contributionsConceptualization and writing the original draft: Antonella Colella, Paola Paradies. Investigation: Antonella Colella, Serena Digiaro, Paola Paradies. Data curation: Antonella Colella, Mariateresa Sasanelli, Beatrice Greco. Statistical analysis: Marco Fiorentino. Writing review and editing: all authors. Supervision: Paola Paradies. ReferencesArmstrong, P.W., Stopps, T.P., Ford, S.E. and de Bold, A.J. 1986. Rapid ventricular pacing in the dog: pathophysiologic studies of heart failure. Circulation. 74(5), 1075–1084. Bonagura, J.D. 1986. Atrial fibrillation in the dog: clinical findings in 81cases. J. Am. Anim. Hosp. Assoc. 22, 111–120. Couto, C.G. and Nelson, R.W. 2015. Medicina interna del cane e del gatto, 5th ed. Milan, Italy: Edra Masson. Fox, P.R., Sisson, D.D. and Moise, N.S. 1999. Textbook of canine and feline cardiology: principles and clinical practice. Readfield, ME: Saunders Company. Friederich, J., Seuß, A.C. and Wess, G. 2020. The role of atrial fibrillation as a prognostic factor in doberman pinschers with dilated cardiomyopathy and congestive heart failure. Vet. J. 264, 105535. Gelzer, A.R., Kraus, M.S. and Rishniw, M. 2015 Evaluation of in-hospital electrocardiography versus 24-hour Holter for rate control in dogs with atrial fibrillation. J. Small Anim. Pract. 56(7), 456–462. Gelzer, A.R., Kraus, M.S., Rishniw, M., Moïse, N.S., Pariaut, R., Jesty, S.A. and Hemsley, S.A. 2009. Combination therapy with digoxin and diltiazem controls ventricular rate in chronic atrial fibrillation in dogs better than digoxin or diltiazem monotherapy: a randomized crossover study in 18 dogs. J. Vet. Intern. Med. 23(3), 499–508. Glaus, T.M., Hässig, M. and Keene, B.W. 2003. Accuracy of heart rate obtained by auscultation in atrial fibrillation. J. Am. Anim. Hosp. Assoc. 39(3), 237–239. Guglielmini, C., Chetboul, V., Pietra, M., Pouchelon, J.L., Capucci, A. and Cipone, M. 2000. Influence of left atrial enlargement and body weight on the development of atrial fibrillation: retrospective study on 205 dogs. Vet. J. 160(3), 235–241. Guglielmini, C., Goncalves, Sousa, M., Baron, Toaldo, M., Valente, C., Bentivoglio, V., Mazzoldi, C., Bergamin, I., Drigo, M. and Poser, H. 2020. Prevalence and risk factors for atrial fibrillation in dogs with myxomatous mitral valve disease. J. Vet. Intern. Med. 34(6), 2223–2231. Jung, S.W., Sun, W., Griffiths, L.G. and Kittleson, M.D. 2016. Atrial fibrillation as a prognostic indicator in medium to large-sized dogs with myxomatous mitral valvular degeneration and congestive heart failure. J. Vet. Intern. Med. 30(1), 51–57. Keene, B.W., Atkins, C.E., Bonagura, J.D., Fox, P.R., Häggström, J., Fuentes, V.L., Oyama, M.A., Rush, J.E., Stepien, R. and Uechi, M. 2019. ACVIM consensus guidelines for the diagnosis and treatment of myxomatous mitral valve disease in dogs. J. Vet. Intern. 33(3), 1127–1140. Kuhlmann, J., Rietbrock, N. and Schnieders, B. 1979. Tissue distribution and elimination of digoxin and methyldigoxin after single and multiple doses in dogs. J. Cardiovasc. Pharmacol. 1(2), 219–234. Menaut, P., Bélanger, M.C., Beauchamp, G., Ponzio, N.M. and Moïse, N.S. 2005. Atrial fibrillation in dogs with and without structural or functional cardiac disease: a retrospective study of 109 cases. J. Vet. Cardiol. 7(2), 75–83. Nagashima, Y., Hirao, H., Furukawa, S., Hoshi, K., Akahane, M., Tanaka, R. and Yamane, Y. 2001. Plasma digoxin concentration in dogs with mitral regurgitation. J. Vet. Med. Sci. 63(11), 1199–1202. Noszczyk-Nowak, A., Michałek, M., Kałuża, E., Cepiel, A. and Paslawska, U. 2017. Prevalence of Arrhythmias in Dogs Examined between 2008 and 2014. J. Vet. Res. 61(1), 103–110. Pariaut, R. 2017. Atrial fibrillation: current therapies. Vet. Clin. North. Am. Small Anim. Pract. 47(5), 977–988. Pedersoli, W.M. 1978. Serum digoxin concentrations in healthy dogs treated without a loading dose. J. Vet. Pharmacol. Ther. 1(4), 279–284. Pedro, B., Dukes-McEwan, J., Oyama, M.A., Kraus, M.S. and Gelzer, A.R. 2018. Retrospective evaluation of the effect of heart rate on survival in dogs with atrial fibrillation. J. Vet. Intern. Med. 32(1), 86–92. Pedro, B., Fontes-Sousa, A.P. and Gelzer, A.R. 2020a. Canine atrial fibrillation: Pathophysiology, epidemiology and classification. Vet. J. 265, 105548. Pedro, B., Fontes-Sousa, A.P. and Gelzer, A.R. 2020b. Diagnosis and management of canine atrial fibrillation. Vet. J. 265, 105549. Pincus, M. 2016. Management of digoxin toxicity. Aust. Prescr. 39(1), 18–20. Saunders, A., Gordon, S. and Miller, M. 2009. Canine atrial fibrillation. Compend. Contin. Educ. Vet. 31(11), E1–E9. Stokhof, A.A. and van den Brom, W.E. 1990. Survival time in dogs with spontaneous atrial fibrillation related to scintigraphically measured cardiac performance. Vet. Q. 12(3), 161–165. Tatlisu, M.A., Ozcan, K.S., Gungor, B., Zenging, A., Karatas, M.B. and Nurkalem, Z. 2015. Inappropriate use of digoxin in patients presenting with digoxin toxicity. J. Geriatr. Cardiol. 12(2), 143–146. Teske, R.H., Bishop, S.P., Righter, H.F. and Detweiler, D.K. 1976. Subacute digoxin toxicosis in the beagle dog. Toxicol. Appl. Pharmacol. 35(2), 283–301. Van den Berg, M.P., Tjeerdsma, G., Jan de Kam, P., Boomsma, F., Crijns, H.J. and van Veldhuisen, D.J. 2002. Longstanding atrial fibrillation causes depletion of atrial natriuretic peptide in patients with advanced congestive heart failure. Eur. J. Heart Fail. 4(3), 255–262. Ward, J., Ware, W. and Viall, A. 2019. Association between atrial fibrillation and right-sided manifestations of congestive heart failure in dogs with degenerative mitral valve disease or dilated cardiomyopathy. J. Vet. Cardiol. 21, 18–27. Wilson, J.R., Douglas, P., Hickey, W.F., Lanoce, V., Ferraro, N., Muhammad, A. and Reichek, N. 1987. Experimental congestive heart failure produced by rapid ventricular pacing in the dog: cardiac effects. Circulation. 75(4), 857–867. | ||
| How to Cite this Article |
| Pubmed Style Colella A, SD, Fiorentino M, Greco B, Sasanelli M, Paradies P. Serum digoxin concentrations in dogs with atrial fibrillation under long term digoxin treatment. Open Vet J. 2022; 12(3): 360-369. doi:10.5455/OVJ.2022.v12.i3.9 Web Style Colella A, SD, Fiorentino M, Greco B, Sasanelli M, Paradies P. Serum digoxin concentrations in dogs with atrial fibrillation under long term digoxin treatment. https://www.openveterinaryjournal.com/?mno=94691 [Access: July 27, 2024]. doi:10.5455/OVJ.2022.v12.i3.9 AMA (American Medical Association) Style Colella A, SD, Fiorentino M, Greco B, Sasanelli M, Paradies P. Serum digoxin concentrations in dogs with atrial fibrillation under long term digoxin treatment. Open Vet J. 2022; 12(3): 360-369. doi:10.5455/OVJ.2022.v12.i3.9 Vancouver/ICMJE Style Colella A, SD, Fiorentino M, Greco B, Sasanelli M, Paradies P. Serum digoxin concentrations in dogs with atrial fibrillation under long term digoxin treatment. Open Vet J. (2022), [cited July 27, 2024]; 12(3): 360-369. doi:10.5455/OVJ.2022.v12.i3.9 Harvard Style Colella, A., , . S. D., Fiorentino, . M., Greco, . B., Sasanelli, . M. & Paradies, . P. (2022) Serum digoxin concentrations in dogs with atrial fibrillation under long term digoxin treatment. Open Vet J, 12 (3), 360-369. doi:10.5455/OVJ.2022.v12.i3.9 Turabian Style Colella, Antonella, Serena Digiaro, Marco Fiorentino, Beatrice Greco, Mariateresa Sasanelli, and Paola Paradies. 2022. Serum digoxin concentrations in dogs with atrial fibrillation under long term digoxin treatment. Open Veterinary Journal, 12 (3), 360-369. doi:10.5455/OVJ.2022.v12.i3.9 Chicago Style Colella, Antonella, Serena Digiaro, Marco Fiorentino, Beatrice Greco, Mariateresa Sasanelli, and Paola Paradies. "Serum digoxin concentrations in dogs with atrial fibrillation under long term digoxin treatment." Open Veterinary Journal 12 (2022), 360-369. doi:10.5455/OVJ.2022.v12.i3.9 MLA (The Modern Language Association) Style Colella, Antonella, Serena Digiaro, Marco Fiorentino, Beatrice Greco, Mariateresa Sasanelli, and Paola Paradies. "Serum digoxin concentrations in dogs with atrial fibrillation under long term digoxin treatment." Open Veterinary Journal 12.3 (2022), 360-369. Print. doi:10.5455/OVJ.2022.v12.i3.9 APA (American Psychological Association) Style Colella, A., , . S. D., Fiorentino, . M., Greco, . B., Sasanelli, . M. & Paradies, . P. (2022) Serum digoxin concentrations in dogs with atrial fibrillation under long term digoxin treatment. Open Veterinary Journal, 12 (3), 360-369. doi:10.5455/OVJ.2022.v12.i3.9 |