| Research Article | ||
Open Vet. J.. 2025; 15(10): 5221-5229 Open Veterinary Journal, (2025), Vol. 15(10): 5221-5229 Research Article Effectiveness effects of Ulva lactuca ethanol extract on estradiol levels, estrus cycle, and endometrial thickness in MSG-induced female ratsLuh Ari Arini* and Luh Citrarasmi Dara MestikaMidwifery Program, Faculty of Medicine, Ganesha University of Education, Singaraja Bali, Indonesia *Corresponding Author: Luh Ari Arini. Midwifery Program, Faculty of Medicine, Ganesha University of Education, Singaraja Bali, Indonesia. Email: arini.ari [at] undiksha.ac.id Submitted: 27/03/2025 Revised: 24/08/2025 Accepted: 22/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
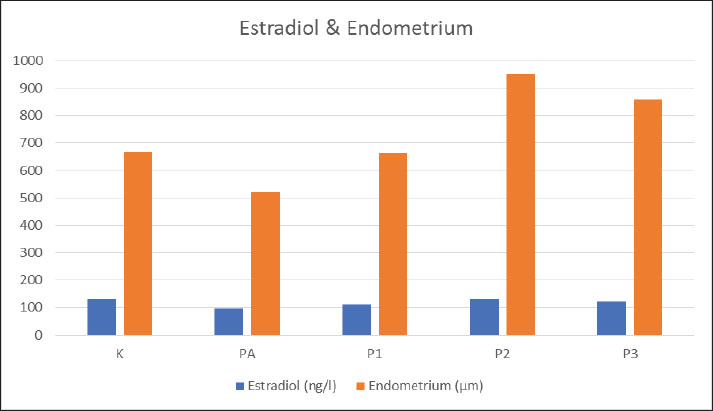
AbstractBackground: Excessive consumption of monosodium glutamate (MSG), a common food flavor enhancer, has been reported to adversely affect the female reproductive system. Therefore, to overcome the negative effects of consuming MSG, which causes an oxidant effect, this can be overcome by consuming foods that contain sources of antioxidants. Aim: This study aimed to analyse the effect of Ulva lactuca ethanol extract on the estradiol hormone levels, estrus cycle, and endometrial thickness in female rats exposed to MSG. Methods: Thirty adult female Wistar rats were divided into five groups: normal control, MSG-induced control, and three U. lactuca treatment groups (100, 200, and 400 mg/kgBW). Estradiol levels, endometrial thickness, and estrus cycle phases were assessed. Results: Administration of U. lactuca extract significantly improved estradiol levels (p=0.011), endometrial thickness (p=0.016), and estrus cycle regularity (p=0.005) compared to the MSG-induced group. Conclusion: Ulva lactuca ethanol extract showed potential in mitigating MSG-induced toxicity, supporting its use in preserving female reproductive health. Keywords: Endometrial, Estradiol hormone, Estrus cycle, Monosodium glutamate, Ulva Lactuca. IntroductionMonosodium glutamate is an addictive ingredient; thus, it can cause an additive effect if you use it in your daily diet (Vorhees, 2018; Nnadozie et al., 2019; Banerjee et al., 2021). Excessive consumption is known to cause toxic or negative effects on the body and public health, including organs and the reproductive system (Ataseven et al., 2016; Mondal et al., 2016; Niaz et al., 2018). The results of previous studies on experimental animals revealed various pathological effects due to the influence of the consumption of foods containing monosodium glutamate (MSG), such as disruption of the hormonal system, reproductive toxicity, neurotoxicity, nephrotoxicity, hepatotoxicity, and obesity (Marshall et al., 2019; Zanifirescu et al., 2019; Hajihasani et al., 2020; Wang et al., 2021). MSG has been known to damage the arcuate nucleus cells of the hypothalamus, which results in a decrease in the secretion of gonadotrophin-releasing hormone (GnRH) (Marshall et al., 2019; Wang et al., 2020). GnRH stimulates the anterior pituitary gland to secrete Follicle-stimulating hormone (FSH) and Luteinizing hormone (LH). FSH and LH stimulate follicular growth, preovulation, and secretion of estrogen and progesterone, which have a feedback mechanism to the hypothalamus (Zheng et al., 2017; Schäfer-Somi et al., 2018). If the hypothalamic arcuate nucleus is damaged, it will result in a decrease in the release of the FSH and LH, which directly or indirectly will affect the secretion of estrogen and progesterone (Kleine and Rossmanith, 2016; Prior, 2020). The estrogen functions to stimulate the development of the endometrium and other female reproductive organs (Yu et al., 2022). The progesterone stimulates the development of the uterine glands, promotes secretion, and makes the endometrium ready for embryo implantation (Bu et al., 2019). Thus, if the secretion of the hormones estrogen and progesterone is disturbed, it will affect the female reproductive organs, including the uterus, resulting in disruption of the endometrial glands and the thickness of the endometrial layer. Endometrial organs play a role in the female reproductive cycle both during pregnancy and menstruation (estrus in animals) in the absence of pregnancy (Critchley et al., 2020), which work in synergy with the ovaries, and are controlled by the hypothalamus–pituitary system through the endocrine system for maintenance of hormonal steroid (Kleine and Rossmanith. 2016). This condition cannot be avoided, but can be reduced by giving therapy in the form of herbal therapy using natural ingredients such as marine algae plants with Ulva lactuca species. Improving the morphology of female reproductive organs, including the uterus is the role of the use of probiotics and herbal extracts (Agustono et al., 2025). Ulva lactuca is a type of plant algae division Chlorophyta (green algae), have the bioactive compounds are polyphenols, sterols, carbohydrates, and vitamins (Galasso et al., 2019; Nome et al., 2019). The antioxidant compounds that are present in algae make them valuable for pharmaceutical and other industries (Munir et al., 2013; Mavrommatis, 2023). Sterols are a subgroup of steroids that are naturally present in plants, animals, and fungi, in the form of cholesterol and are an important group of organic molecules (Kopylov et al., 2021). Sterols play an important role in cellular functions and is a substrate/precursor for fat-soluble vitamins and steroid hormones (Batth et al., 2020). The sex steroid hormone plays an important role in the regulation of the female reproductive system in the reproductive cycle and the menstrual cycle/estrus (Belfiore, 2018; Schiffer et al., 2019). The results of research by Helal et al. (2021) green algae cause the recovery of damage to testicular and prostate tissue and Research by Muklisani (2018) administration of brown algae did not affect the scrotal circumference of Balinese cattle. From the description above, it shows that research related to the effect of green algae on reproductive organs is still very limited, especially in women. There is not enough scientific evidence to be able to utilize green algae as an alternative or complementary therapy for female fertility disorders, and research related to the effect of algae on reproductive organs are still pro and con. However, there are not many studies that specifically evaluate the role of U. lactuca in improving hormonal disorders and endometrial structure due to MSG exposure. Therefore, the study aimed to detect and analyze the effect of MSG induction and the therapeutic effect of U. lactuca in relation to the female reproductive system, specifically on endometrial thickness and estrus cycle. Materials and MethodsStudy period and locationThe research was conducted from January to April 2022 in several laboratories, Pharmacy Faculty testing service unit, the experimental animal laboratory Pharmacology Laboratory Faculty of Medicine, Laboratory Faculty of Veterinary Medicine and the Institute for Tropical Diseases Universitas Airlangga, Surabaya, Indonesia. AnimalsAll samples used in this study were female Wistar rats (Rattus norvegicus), aged 18–20 weeks, with a body weight of 150–200 g as much as 30 rats were selected by random sampling. The rats were maintained in the Pharmacology Laboratory of the Airlangga University Medical Faculty. Experimental designThe Green algae plants obtained from Gerogak Beach through BBPPBL Gondol Singaraja Bali, then the genus and species were determined in the Biology Laboratory of the Faculty of Mathematics and Natural Sciences, Udayana University, Bali, then a certificate was obtained for the determination of the taxonomy of the green algae division (Chlorophyta) species U. lactuca. Green algae extraction method using the maceration method with 96% ethanol solvent and the evaporation method using rotary evaporator. After extraction, phytochemical test was carried out using thin layer chromatography method to obtain bioactive compounds of flavonoids, terpenoids, and steroids. Wistar rats were then subjected to vaginal swabs to ensure that the rats were in the proestrus-estrus phase, and then divided into 2 groups. The control group (K) had 6 animals and the treatment group 29 animals (MSG induction). Induction with MSG was carried out for + 40 days at a dose of 400 mg/kgBW administration using a gastric tube/oral gastric tube. The MSG induction group rats were divided into 4 groups randomly, each obtained 6 rats for 4 groups. The post-induction group (PA), the algae extract 100 mg/kgBW (P1), the algae extract 200 mg/kgBW (P2) and the algae extract 400 mg/kgBW (P3) were given algae extract for + 40 days. Sample collectionAfter all treatments ended, 5 ml of blood serum was taken intracardially in the heart, previously anesthetized using 10% ketamine at a dose of 50 mg/kgBW injected intramuscularly into the thigh of the rat. The levels of estradiol and testosterone hormones were examined after treatment (posttest). Blood estradiol and testosterone hormone levels were examined using the indirect ELISA method. After that, the rats were euthanized and terminated by directly cutting the aorta. Histopathological examination of endometrial tissue was carried out by surgically taking the uterine organs of experimental mice, and then soaking them in 10% neutral buffered formalin solution, then making endometrial organ preparations with a thickness of 4–5µm and stained with Hematoxylin Eosin. Endometrial thickness measurement with histological analysis was performed using a digital microscope and image analysis software image is obtained by drawing a perpendicular line starting from the mucosal epithelium to the muscularis mucosa with a magnification of 40–100×, the measurement results are then divided into 5 parts, which are then averaged as the final value. Statistical analysisThe data in each sample group for estradiol hormone levels and endometrial thickness were normally distributed and homogeneous, while the data for the estrus cycle were not normally distributed, but homogeneous that the statistical test used was a parametric test for endometrial thickness using Anova and a nonparametric test for estrus cycle with Kruskal–Wallis with a significance level (α=0.05). Ethical approvalThis work has received approval for research ethics from Animal Care and Use Committee (ACUC) of Faculty of Veterinary Medicine Airlangga University, Surabaya (Approval No. 2.KE.145.12.2021), Dated: 29 Desember 2021. ResultsThe results of the analysis of the effect of giving U. lactuca therapy were tested based on the average thickness of the endometrium and the phase of the estrus cycle experienced between the control group, exposed to MSG, and treatment with U. lactuca. The average estradiol hormone level in the control group was 129.497 + 12.24 ng/l, which then decreased in the post-induction MSG group, which had the lowest average compared to the other groups, namely 96.105 + 18.30 ng/l. The average level of the hormone estradiol began to increase again in the treatment group with U. lactuca algae extract, with various increases at various doses tested. The results of the analysis with the Anova test showed the value of F=4.093 with p=0.011 at the level of confidence (p < 0.05) (the data are shown in Table 1). Based on this, it was concluded that the average estradiol hormone levels in each sample group had a significant difference (Table 1). Table 1. Estradiol hormone levels between sample groups. Hormone examination was carried out using the ELISA test method with statistical data analysis. used was parametric test for endometrial thickness using Anova test with a significance level (α=0.05).
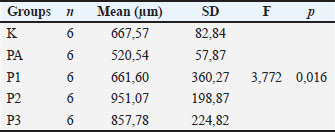
The mean endometrial thickness in the K (control group) was 667.57 + 82.84 µm, the PA (Post Induction group) was 520.24 + 57.87µm, the P1 (algae treatment 1), P2 (algae treatment 2), and P3 (algae treatment 3), respectively, are 661.60 + 360.27 µm, 951.07 + 198.87 µm, and 857.78 + 224.82 µm. The results of the analysis of significance with the one-way Anova test, showed the value of F=3.772 and the value of p=0.016. Based on this, it means that the mean endometrial thickness in all groups was significantly different (p < 0.05). Endometrial thickness in the treatment group with U. lactuca algae extract therapy was greater than the post-induction MSG group and the healthy control group (Table 2). Table 2. Endometrial thickness between sample groups. Examination of endometrial thickness using the Hematoxylin Eosin (HE) staining method, with data analysis using parametric statistical tests with the Anova test with a significance level (α=0.05).
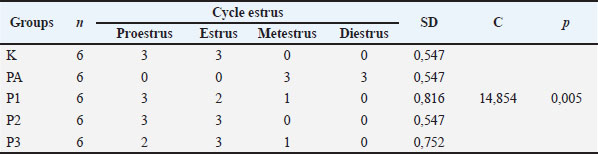
The estrus cycle in group K was in the proestrus and estrus phase with + 0.547, the PA group was in the metestrus and diestrus phase + 0.547, the P1 group was in the proestrus, estrus, and metestrus phase + 0.816, the P2 group was in the estrus phase. Proestrus and estrus + 0.547, group P3 was in the proestrus phase, estrus and metestrus + 0.752, and the MSG group was in the metestrus and diestrus phase + 0.547. The results of the analysis of significance with the Kruskal–Wallis test, showed the value of C=14,854 and the value of p=0.005 (the data is shown in Table 3). Based on this, it means that the estrus cycle in all groups was significantly different (p < 0.05). The estrus cycle in the treatment group with U. lactuca algae extract therapy resembled the healthy control group and was better than the post-induction MSG group (Table 3).
The averaga of Estradiol hormon levels & Endometrial thickness. Table 3. Estrus cycles between sample groups. Examination of the estrus cycle using the vaginal swab method and statistical analysis using Kruskall Wallis test with a significance level (α=0.05).
DiscussionThe increase in estradiol hormone levels occurred significantly in the treatment group that received the U. lactuca algae extract significantly with a p-value of 0.011. The increase occurred through various doses of slow administration in the first and second doses and then slightly decreased at the high dose/third dose, compared to the control. Prior to this increase, levels of the hormone estradiol were known to be lower in the post-induction MSG group when compared to the control group and the algae treatment group. This shows that estradiol levels may increase after administration of U. lactuca, which initially decreases due to negative effects after receiving MSG exposure. The increase occurred in the first and second dose treatment groups slowly. The first dose had not yet reached the control estradiol level, while the second dose was similar to the control, and the third dose decreased to an even lower concentration than the control and second dose. This may be due to the inability of the body to receive high doses of algae Ulva lactuca, causing a negative feedback effect on the hormone estradiol in the body and causing a decrease in estradiol levels. This condition can eventually lead to overdose problems if consumed continuously in high concentrations, and can interfere with hormonal regulation that occurs naturally in the body. Thus, the optimal dose that can be given based on this research is at a dose of 100 mg/kgBW and 200 mg/kgBW. Based on the results of previous studies, it is stated that there are certain conditions non-monotonic dose response curves (NMDRCs) that occur in cells, tissues, animals, and certain populations in response to nutrients, vitamins, pharmacological compounds, hormones, and endocrine disrupting chemicals. NMDRCs are mathematically defined as the change in sign (positive/negative) of the slope of the dose-response relationship over the tested dose range. A number of studies have recognized the occurrence of NMDRC in response to nutrients, vitamins, pharmacological compounds, and other small, molecular compounds that interact with receptors including hormones. Thus, these conditions can cause differences in body responses that occur when receiving doses that are considered irrelevant so that they can affect the regulatory process (Hill et al., 2018). The latest study related to administration of Ulva Lactuca methanolic extract (MEUL) in adult male albino rats, shown that there was no visible symptoms of toxicity or mortality among rats administered with different dose as in dose 100, 200, 250, and 300 mg/kg. No abnormal behavior was observed the four-experimental group of male rats (Bouhalit and Brahmi, 2021). Another study related to the administration of U. lactuca algae in male rats with varying doses of 100 mg/kgBW and 400 mg/kgBW orally after exposure to subchronic agents. It is known that a dose of 400 mg/kgBW can reduce oxidative stress conditions seen through a decrease in oxidative damage to cells in the liver and has a concentration of antioxidant enzymes found to be higher than at lower doses and close to control, in other words, the administration of high doses of algae can provide better outcome than other groups and has a chemoprotective effect. This does to be the best in causing the inhibitory effect of carcinogenic bioactivation from subchronic agents (Roche et al., 2019). This condition is different from research results, which show the opposite, that at high doses it causes a decrease in sex steroid hormones, is estradiol levels and histology of reproductive organ tissue. However, significant differences were seen in the sex of experimental animals and the object of study, which may be related to the response of target organ cells to the presence of steroid hormones, it was the effect on sex steroid regulation in female which tends to decrease which may be related to the response of the hypothalamic–pitutary and gonadal steroids due to the administration of U. lactuca high doses. Therefore, the increase that occurs after administration of green algae extract U. lactuca is likely influenced by the compounds contained in U. lactuca, such as flavonoids as a source of antioxidants and steroids/sterols. The results of this study are in line with the statement that exogenous administration of steroid hormones such as estrogen and progesterone in the normal function ovaries of female (contraceptive users), cause negative feedback to the hypothalamus then inhibits the secretion of GnRH, preventing an increase in gonadotropin secretion by the pituitary gland which will stimulate ovulation in the middle of the cycle (Regidor, 2018; Porcu et al., 2019). Thus, exogenous steroids will interfere with and inhibit the function of the HPG axis, including the production of sex steroid hormones by the ovaries, eventually causing follicle development to be inhibited and have an effect on anovulatory. In addition, other reproductive organs that have an effect are the endometrial walls, which become thinner (Porcu et al., 2019; Critchley et al., 2020). Based on this, it shows that the administration of high doses of U. lactuca algae due to the presence of excess steroids will indirectly cause suppression of natural steroid hormone regulation in the body (endogenous), which will cause down-regulation conditions. A significant increase in endometrial thickness occurred after administration of Ulva lactuca therapy with a p-value of 0.016, which was previously experienced as a decrease after MSG exposure, as evidenced in the post-MSG induction group, and compared to the control group. The increase in endometrial thickness occurred synergistically from the dose of U. lactuca algae extract at the lowest to the highest concentrations (first and second doses). At the first dose, the increase was almost similar to the control, while at the third dose, there was a decrease compared to the second dose group. This can be caused because the organs are not able to accept excessive doses, and cause saturation, that the opposite effect occurs. However, endometrial thickness continued to increase after administration of U. lactuca compared to control and post-induction of MSG at the second and third doses. This shows that U. lactuca algae has a good effect on endometrial proliferation, which is indicated by its thickness, after exposure to MSG, which is thought to inhibit endometrial proliferation. In mice given MSG, it showed that uterine endothelial cells were hyperplastic with vacuolated cytoplasm and a few cells were necrotic, the endometrial glands were relatively reduced in number, and the ovaries were atrophic (El-Fattah et al., 2016; Mondal et al., 2017). This increase in endometrial thickness is almost in line with what occurs with increasing levels of the hormone estradiol, especially from post-induction, which decreases and then increases slowly with algal administration in various doses compared to controls. This shows that the administration of U. lactuca will initially increase levels of the hormone estradiol and then affect the thickened endometrial tissue in response to increased hormones. A microscopic picture of the endometrial mucosa, shows that the tissue structure in the control group does not tight and is still hollow, in the P1 group, the gaps seen in the cross section of the tissue begin to decrease, as well as in P2 and P3, which are getting less. However, in the post-induction group, the gaps or cavities were quite numerous and irregular, appearing in various fields. More and more gaps in the cross section of endometrial tissue indicate a small level of endometrial thickness, and vice versa. Based on the estrus cycle, it was shown that the majority of therapy with U. lactuca algae could cause proestrus and estrus phases, compared to post-exposure to MSG with a significant difference p value of 0.005. Treatment with U. lactuca algae extract had a similar estrus cycle phase to the control. Determination of the estrus cycle was carried out by vaginal smear and microscopic examination of vaginal mucus with 400X magnification. The estrus cycle in experimental rats consisted of 4 phases, namely proestrus, estrus, metestrus, and diestrus. The proestrus-estrus phase is the follicular phase or the fertile phase of experimental animals. In proestrus, there is the influence of FSH to stimulate the growth of follicles in the ovaries that the follicles develop and begin to secrete estrogen. In the proestrus phase, there is also an increase in vaginal vascularization and cornification due to higher estrogen levels (Mclean et al., 2012; Ajayi and Akhigbe, 2020). Based on the results of the analysis, the mean endometrial thickness in the algae treatment groups 1, 2, and 3 was greater than the mean endometrial thickness in the control group and the post-MSG induction group, which had the smallest mean endometrial thickness. Likewise, between the algae treatment groups, the thickness of the endometrium increased from the lowest to the highest dose. In the algae treatment group at doses 1, 2, and 3, the thickness of the endometrium was 661.60 µm, 857.78 µm and 951.07 µm, with the majority of estrus cycles in the proestrus-estrus phase. In the post-exposed group, MSG had the smallest endometrial thickness, which was 637.21µm, and experienced metestrus and diestrus phases. This shows that the thickness of the endometrium increases during the proestrus and estrus phases after algae treatment, which previously decreased after exposure to MSG. This is in line with the theory which states that the proestrus and estrus phases are characterized by an increase in estrogen secretion, thus the vaginal epithelium is confirmed to have cells proliferate. An increase in estrogen will stimulate LH and maturation of the ovum from the enlarged follicle, resulting in ovulation (Belfiore, 2018; Agbadua et al., 2020). The effect of giving algae U. lactuca causes an increase in the hormone estradiol, causing most of the rats to experience the proestrus and estrus phases, as happened in the control and before MSG induction. In the metestrus phase, there is a decrease in estrogen and an increase in progesterone produced by the ovaries. This phase is possible due to the inhibition of the estrus phase which the follicle degenerates. The diestrus phase is the final phase of the estrus cycle, which is characterized by the absence of pregnancy because there is sexual activity and estrogen levels are at their lowest concentrations. Low of estrogen levels can cause the estrus cycle to be longer or can reduce fertility (Ajayi and Akhibe, 2020). Post-induction of MSG caused a decrease in the hormone estradiol, according to past studies that abnormal levels of progesterone and estrogen were noted in animals treated with MSG and ovarian hormone abnormality, which increase on progesterone levels (Mondal et al., 2017; Agbadua et al., 2020). The disturbance in progesterone and estrogen levels in female rats caused by MSG may lead to an abnormality in functions of reproductive organ (Abdulghani, 2021). This is showed by the results of the study majority of rats being in the metestrus-diestrus phase compared to controls and before exposure to MSG. In the diestrus phase, the endometrium is in the early stages of cycle preparation that the endometrium is still thinner than in other phases. Disorders of the sex steroid hormonal system cause disturbances in the estrus cycle, which begins with histological changes of uterus and relative lumen area showed smaller than the control group (Abdulghani, 2021). Recent studies have shown that MSG administration is known to exert an effect on the uterus, showing a comparative reduction in endometrial thickness and loss of endometrial glands associated with cellular hyperplasia and reduced vacuole cells. The endometrium in mammals is important for the maintenance of a stable pregnancy, whereas in women the endometrium rapidly grows and sheds throughout the menstrual cycle to maintain optimal function (Sugiura et al., 2018). Thus, the occurrence of disturbances in the endometrium can also cause problems with its function in the reproductive system and fertility in females. The estrus cycle is actually similar to the menstrual cycle in that ovulation occurs at a time in the cycle after the endometrium has begun to thicken and drain heavily, as it prepares the uterus for possible implantation of an embryo. One difference between the two types of cycles involves the lining of the uterus if pregnancy does not occur. During the menstrual cycle, the endometrium sheds from the uterus through the cervix and vagina in bleeding known as menstruation (Kirkwood et al., 2022). The estrus cycle lasts 4–5 days, and if there is no fertilization happened in this cycle, after an initial thickening, the endometrium is reabsorbed by the uterus, and there is no profuse bleeding, and then it will be a new cycle (Catalini and Fedder, 2020). During diestrus, functional regression of the corpus luteum occurs. The vaginal mucosa is thin, and leukocytes migrate across it, providing vaginal smear preparations consisting almost exclusively of these cells. The uterus becomes very small, anemic, and only slightly contractile during diestrus, suggesting that while the corpus luteum remains, it secretes progesterone only briefly during the reproductive cycle (Gossler et al., 2017; Agbadua et al., 2020; Abdulghani, 2021). The results of the first vaginal smear in the study before MSG was not induced, showed that most had a percentage of 62% estrus phase, 28% proestrus phase, 2% metestrus, and 8% diestrus. This condition indicates that the majority of the group are in the follicular phase, where levels of the hormone estrogen are high, and also serves as a determinant that the experimental animals are in a fertile condition. Rats in a fertile condition were determined through the ongoing follicular/proestrus phase for the estrus/ovulation phase to occur, and infertile rats based on the occurrence of disturbances in the proestrus to estrus phase that no mature ovum could be ovulated by the ovaries, or it could be shown by vaginal smear, namely mice are in the luteal/diestrus phase (Ekambaram et al., 2017; Ningtyas, 2017). The estrus cycle is a cycle that is mainly regulated by the hormone estrogen, which normally lasts about 4–5 days (Auta and Hassan, 2016; Ajayi and Akhigbe, 2020). Thus, in conditions of estrogen deficiency, it causes the estrus cycle to be disrupted, in other words, the rats do not experience the peak phase of estrus/ovulation, which is a sign of a disturbance in the reproductive cycle of female rats. The results of the second vaginal smear after MSG induction showed that the majority had cycles of metestrus is 46% and diestrus phases is 54%. This shows that changes caused by exposure to MSG will slowly cause disturbances in the proestrus-estrus phase. The administration of Ulva lactuca can improve this condition, through vaginal smears after administration of U. lactuca at various doses, the results showed that the majority of the group experienced the proestrus-estrus phase, namely 50% experienced the proestrus phase, 39% the estrus phase, 11% the metestrus phase, and 0% the estrus phase diestrus. This shows that the estrus cycle began to improve, like the initial conditions before exposure to MSG, even better. During the previous normal estrus cycle, there has been an increase in endometrial thickness, a decrease in progesterone (P4), and an increase in estradiol (E2), which is a balance of response between ovarian steroid hormones and the capacity of the endometrium to respond to these hormones (Xu et al., 2011; Sugiura et al., 2018). Estradiol is responsible for the proliferation of endometrial epithelial cells, causing an increase in tissue thickness. E2 also stimulates the production of VEGF, which triggers the growth of new blood vessels in the endometrium, increasing blood flow and nutrients to the uterine tissue (Sugiura et al., 2018). Endometrial thickness increases throughout the estrus cycle due to hyperemia and edematous development throughout the follicular phase and decreases after ovulation. The role of steroid hormones is crucial in the regulation of uterine function, including morphological changes in the endometrium throughout the estrus cycle. Changes in endometrial thickness and uterine blood flow patterns are closely related to ovarian hormones during the estrus cycle. Blood flow to the uterus is relatively stable during diestrus and increases during pro-estrus and the oestrus phase. Estrogen is generally considered to be the most important hormone responsible for uterine changes during the estrus cycle (Singh et al., 2017). Due to the influence of ovarian hormone levels as estradiol and progesterone, the endometrium undergoes morphological and biochemical changes, such as proliferation, apoptosis, and cellular differentiation during the estrus cycle (Xu et al., 2011; Liu et al., 2020). The endometrial glands will increase during proestrus, due to edema and endometrial hyperplasia, then will decrease slightly during the estrus phase (Annie et al., 2018). The increase in the number of epithelial cells that proliferate in endometrial cell cultures is due to high levels of estradiol, which correlates with increased expression of estrogen receptors (Kotlarczyk et al., 2021). Estradiol-regulated early signaling mediates cross-talk with estrogen receptor-α to elicit a late uterine growth response (Gao et al., 2011; Hamilton et al., 2017). ConclusionBased on the results of the study showed that the effect of giving U. lactuca therapy after MSG induction caused an increase in estradiol hormone levels, an increase in endometrial thickness, and changes in the estrus cycle, which were observed based on the phase in the cycle that resembled the control and conditions before being given treatment. Ulva lactuca ethanol extract showed potential in mitigating MSG-induced toxicity, supporting its use in preserving female reproductive health. For further research, development can be carried out towards the toxicity of herbal supplement ingredients, and testing can be carried out directly on humans. AcknowledgmentsThe authors are grateful to the Faculty of Medicine, Ganesha of Education University and for the support in completing my research and Faculty of Medicine, Airlangga University for the providing animal enclosures for this study. The authors did not receive any funds for this study. Conflict of interestThe authors confirm that the content of this article has no conflict of interest and that they have no competing interests. FundingNone. Author contributionAA: conceptualization, data collection, and writing of the original manuscript draft. DM: sharing in the writing and editing of manuscript drafts. All authors have approved the final manuscript for publication. Data availabilityAll data were provided in the manuscript. ReferencesAbdulghani, M.A. 2021. Short-term effect of monosodium glutamate on serum hormonal level and histological changes of uterus during estrus phase in rat. Research square. 1-13 Agbadua, O.G., Idusogie, L.E. and Chukwuebuka, A.S. 2020. Evaluating the Protective and Ameliorative Potential of Unripe Palm Kernel Seeds on Monosodium Glutamate-Induced Uterine Fibroids. Libr. J. 7, 1–11. Agustono, B., Yunita, M.N., Lokapirnasari, W.P., Warsito, S.H., Marbun, T.D. and Windria, S. 2025. Dietary supplementation of microbiota inoculum and single clove garlic extract on growth performance, egg quality, reproductive organ, and hematological trait in laying quail. Open Vet. J. 15(2), 690–699. Ajayi, A.F. and Akhigbe, R.E. 2020. Staging of the estrous cycle and induction of estrus in experimental redents: an update. Fertility Res. Pract. 6(5), 1–15. Annie, L., Gurusubramanian, G. and Roy, V.K. 2018. Estrogen and progesterone dependent expression of visfatin/NAMPT regulates proliferation and apoptosis in mice uterus during estrous cycle. J. Steroid Biochem. Mol. Boil. 185, 225–236. Ataseven, N., Yüzbaşıoğlu, D., Keskin, A.C. and Ünal, F. 2016. Genotoxicity of monosodium glutamate. Food Chem. Toxicol. 91, 8–18. Auta, T. and Hassan, A.T. 2016. Alteration in oestrus cycle and implantation in Mus musculus administered aqueous wood ash extract of Azadirachta indica (neem). Asian Pacific J. Reprod. 5(3), 188–192. Banerjee, A., Mukherjee, S. and Maji, B.K. 2021. Worldwide flavor enhancer monosodium glutamate combined with high lipid diet provokes metabolic alterations and systemic anomalies: an overview. Toxicol. Rep. 8, 938–961. Batth, R., Nicolle, C., Cuciurean, I.S. and Simonsen, H.T. 2020. Biosynthesis and industrial production of androsteroids. Plants 9, 1–11. Belfiore. 2018. Principles of Endocrinology and Hormone Action. Cham, Switzerland: Springer. Bouhalit, S and Brahmi,N. 2021. Ameliorating role of Ulva lactuca extract on neurobehavioral and neurochemical abnormalities caused by Lithium in rats. J. Biores. Manag. 8(4), 121–134. Bu, Z., Yang, X., Song, L., Kang, B. and Sun, Y. 2019. The impact of endometrial thickness change after progesterone administration on pregnancy outcome in patients transferred with single frozen-thawed blastocyst. Reprod. Biol. Endocrinol. 17(1), 99. Catalini, L and Fedder J, 2020. Characteristics of the endometrium in menstruating species: lessons learned from the animal kingdom. Biol. Reprod. 2020, 102(6), 1160–1169. Critchley, H.O.D., Maybin, J.A., Armstrong, G.M. and Williams, A.R.W. 2020. Physiology of the Endometrium and Regulation of Menstruation. Physiol. Rev. 100(3), 1149–1179. Ekambaram, G., SKS, K. and Joseph, L.D. 2017. Comparative Study on the Estimation of Estrous Cycle in Mice by Visual and Vaginal Lavage Method. J. Clin. Diagnostic Res. 11(1), AC05–AC07. El-Fattah, A., Beltagy, B.M. and Elghaweet, H.A. 2016. Adverse effect of Monosodium Glutamate on The Reproductive Organs of Adult Female Albino Rats and The Possible Ameliorated Role of Carob (Ceratonia Siliqua). J. Bioscience Appl. Res. 2(3), 170–184. Galasso, C., Gentile, A., Orefice, I., Ianora, A., Bruno, A., Noonan, D.M., Sansone, C., Albini, A. and Brunet, C. 2019. Microalgal derivatives as potential nutraceutical and food supplements for human health: a focus on cancer prevention and interception. Nutrients 11, 1–8. Gao, F., Ma, X., Ostmann, A.B. and Das, S.K. 2011. GPR30 activation opposes estrogen-dependent uterine growth via inhibition of stromal ERK1/2 and estrogen receptor alpha (ERa) phosphorylation signals. Endocrinology 152(4), 1434–1447. Gossler, V.S., Santos, F.A., Azevedo, A.R., Goncalves, P.C., Rigolo, H.A., Trevisan, C., Masseno, A.P., Cardoso, P.M., Papa, P.C., Castilho, C. and Giometti, I.C. 2017. Evaluation of cell proliferation and endometrial thickness of bitches in different periods of diestrus. Ann. Braziliam. Acad. Sci. 89(3), 1719–1727. Hajihasani, M.M., Soheili, V., Zirak, M.R., Sahebkar, A. and Shakeri, A. 2020. Natural products as safeguards against monosodium glutamate induced Toxicity. Iranian J. Basic Med. Sci. 23, 416–430. Hamilton, K.J., Hewitt, S.C., Arao, Y. and Korach, K.S. 2017. Estrogen hormone biology. Curr. Top. Dev. Biol. 125, 109–146. Helal, A.M., Abdel-Latif, M.S., Abomughaid, M.M., Ghareeb, D.A. and El-sayed, M.M. 2021. Potential Therapeutic Effects of Ulva Lactuca Water Fraction on Monosodium Glutamate-Induced Testicular and Prostatic Tissue Demage in Rats. Environ. Sci. Pollut. Res. 28(23), 29629–29642. Hill, C.E., Myers, J.P. and Vandenberg, L.N. 2018. Nonmonotonic dose-response curves occur in dose ranges that are relevant to regulatory decision-making. Dose-response. Int. J. 16(3), 1–4. Kirkwood, P.M., Shaw, I.W. and Saunders, P.T.K. 2022. Mechanisms of scarless repair at time of menstruation: insights from Mouse Models. Front. Reproductive Health 3, 1–12. Kleine, B. and Rossmanith, W.G. 2016. Hormones and the Endocrine System. Springer. Springer. pp: 1–11 Kopylov, A.T., Malsagova, K.A., Stepanov, A.A. and Kaysheva, A.L. 2021. Diversity of Plant Sterols Metabolism: the Impact on Human Health, Sport, and Accumulation of Contaminating Sterols. Nutrients 13, 1–30. Kotlarczyk, A., Grzyb, M. and Korzekwa, A.J. 2021. Regulation of uterine function during estrous cycle, anestrus phase and pregnancy by steroids in red deers (Cervus elaphus L). Scientific Rep. 36–38 Liu, G., Yu, X., Li, S., Shao, W. and Zhang, N. 2020. Pengaruh suplemen mikroalga (Schizochytrium spp.) dalam pakan terhadap performa susu, parameter darah, dan komposisi asam lemak susu pada sapi perah. Czech J. Anim. Sci. 65(5), 162–171. Marshall, T.M., Dardia, G.P., Colvin, K.L., Nevin, R. and Macrellis, J. 2019. Neurotoxicity Associated with Traumatic Brain Injury, Blast, Chemical, Heavy Metal and Quinoline Drug Exposure. Altern. Ther. Health Med. 25(1), 28–34. Mavrommatis, A., Tsiplakou, E., Zerva, A., Pantiora, P.D., Georgakis, N.D., Tsintzou, G.P., Madesis, P. and Labrou, N.E. 2023. Mikroalga sebagai sumber antioksidan berkelanjutan dalam nutrisi, kesehatan, dan pengembangan ternak. Antioksidan (Basel) 12(10), 1882. Mclean, A.C., Valenzuela, N., Fai, S. and Bennett, S.A.L. 2012. Performing vaginal lavage, crystal violet staining, and vaginal cytological evaluation for mouse estrous cycle staging identification. J. Vis. Exp. 15(67), e4389. Mondal, M., Kaushik, S., Partha, P.N. and Gouta, P. 2016. Monosodium Glutamate Depresses the Function of Female Preproductive System in Rat by Promoting Oxidative Stress Induced Changes in The Structure of Uterus. Int. J. Pharma Bio Sci. 7(4), 799–804. Mondal, M., Sarkar, K., Nath, P.P. and Paul, G. 2017. Monosodium Glutamate Suppresses the Female Reproductive Function by Impairing the Functions of Ovary and Uterus in Rat. Environ. Toxicol. 33(2), 198–208. Muklisani. 2018. Efektifitas Alga Coklat (Sargassum SP.) Sebagai Pakan Konsentrat Terhadap Lingkar Scrotum Sapi Bali. Skripsi. Jurusan Ilmu Peternakan Fakultas Sains Dan Teknologi Universitas Islam Negeri Alauddin Makassar. Munir, N., Sharif, N., Naz, S. and Manzoor, F. 2013. Algae: a Potent Antioxidant Sources. Sky J. Microbiol. Res. 1(3), 22–31. Niaz, K., Zaplatic, E. and Spoor, J. 2018. Extensive use of monosodium glutamate: a threat to public health?. EXCLI. J. 17, 273–278. Ningtyas, N.S. 2017. Pengaruh Pemberian Minyak Buah Merah (Pendanus Conoidus Lam.) terhadap Histopatologi Folikel de Graaf Pada Mencit (Mus Musculus) Model Infertil. Jurnal Sangkareang Mataram 3(3). Nnadozie, J.O., Chijioke, U.O., Okafor, O.C., Olusina, D.B., Oli, A.N., Nwonu, P.C., Mbagwu, H.O. and Chijioke, C.P. 2019. Chronic toxicity of low dose monosodium glutamate in albino Wistar rats. BMC. Res. Notes 12(1), 593. Nome, W., Salosso, Y. and Eoh, C.B. 2019. Analisis Metabolit Sekunder Dan Kandungan Nutrisi Dari Mikroalga Hijau (Chlorophyceae) Diperairan Teluk Kupang. Jurnal Aquatic 2(1), 100–112. Porcu, P., Serra, M. and Concas, A. 2019. The brain as a target of hormonal contraceptives: evidence from animal studies. Front. Neuroendocrinol. 55. Prior, J.C. 2020. Women’s reproductive system as balanced estradiol and progesterone actions- a revolutionary, paradigm-shifting concept in women’s health. Drug Discovery Today. Dis. Models. Women. Health 32, 31–40. Regidor, P.A. 2018. The clinical relevance of progestogens in hormonal contraception: present status and future developments. Oncotarget 2(77), 34628–34638. Roche, L.D., Rodeiro, I., Riera, M., Herrera, J.A., Venturi, I., Hernandez. and et al.. 2019. Chemoprotective Effect of Ulva lactuca (green seaweed) Aqueous-Ethanolic Extract Against Subchronic Exposure to Benzo(a)pyrene by CYP1A1 Inhibition in Mice. Phytother Res. (4),958–967. Schäfer-Somi, S., Kaya, D., Sözmen, M., Kaya, S. and Aslan, S. 2018. Pre-pubertal treatment with a GnRH agonist in bitches-Effect on the uterus and hormone receptor expression. Reprod. Domest. Anim. 53(3), 103–109. Schiffer, L., Barnard, L., Baranowski, E.S., Gilligan, L.C., Taylor, A.E., Arlt, W., Shackleton, C.H.L. and Storbeck, K.H. 2019. Human steroid biosynthesis, metabolism and excretion are differentially reflected by serum and urine steroid metabolomes: a comprehensive review. J. Steroid Biochem. Mol. Biol. 194, 1–25. Singh, D., Brar, P. and Honparkhe, M. 2017. Endometrial thickness and uterine vascularization in relation to ovarian hormones during estrous cycle in buffalo (bubalus bubalis). Indian Vet. J. 94(7), 27–29. Sugiura, T., Akiyoshi, S., Inoue, F., Yanagawa, Y., Moriyoshi, M., Tajima, M. and Katagiri, S. 2018. Relationship between bovine endometrial thickness and plasma progesterone and estradiol concentrations in natural and induced estrus. J. Reprod. Develop. 64(2), 135–143. Vorhees, C.V. 2018. A test of dietary monosodium glutamate developmental neurotoxicity in rats: a reappraisal. Ann. Nutr. Metab. 73(5), 36–42. Wang, C., Zhang, Y., Feng, Y., Liang, L., Ling, S., Chong, S., Jiang, J. and Chun-Li, Q. 2021. The Reproductive Toxicity of Monosodium Glutamate by Damaging GnRH Neurons be Relieved Spontaneously Over Time. Drug Design. Develop. Therapy. Scientific Med. Res. 15, 3499–3508. Wang, Z., Zhang, J., Wu, P., Luo, S., Li, J., Wang, Q., Huang, P., Li, Y., Ding, X., Hou, Z., Wu, D., Huang, J., Tu, Q. and Yang, H. 2020. Effects of oral monosodium glutamate administration on serum metabolomics of suckling piglets. J. Anim. Physiol. Anim. Nutr. 104(1), 269–279. Xu, J., Hu, F.F., Cui, Y.G., Luo, J., Jiang, C.Y., Gao, L., Qian, X.Q., Mao, Y.D. and Liu, J.Y. 2011. Effect of estradiol on proliferation and differentiation of side population stem/progenitor cells from murine endometrium. Reproductive Biol. Endocrinol. 9(1), 103. Yu, K., Huang, Z.Y., Xu, X.L., Li, J., Fu, X.W. and Deng, S.L. 2022. Estrogen receptor function: impact on the human endometrium. Front. Endocrinol. 13, 1–16. Zanifirescu, A., Ungurianu, A., Tsatsakis, A., Nitulescu, G., Kouretas, D., Veskoukis, A. and Tsoukalas, D. 2019. A review of the alleged health hazards of monosodium glutamate. Compr. Rev. Food Sci. Food Saf. 18(4), 1111–1134. Zheng, J., Mao, J. and Xu, H. 2017. Pulsatile GnRH therapy may restore hypothalamus-pituitary-testis axis function in patients with congenital combined pituitary hormone deficiency: a prospective, self-controlled trial. J. Clin. Endocrinol. Metab. 102(7), 2291–2300. | ||
| How to Cite this Article |
| Pubmed Style Arini LA, Mestika LCD. Effectiveness effects of Ulva lactuca ethanol extract on estradiol levels, estrus cycle, and endometrial thickness in MSG-induced female rats. Open Vet. J.. 2025; 15(10): 5221-5229. doi:10.5455/OVJ.2025.v15.i10.38 Web Style Arini LA, Mestika LCD. Effectiveness effects of Ulva lactuca ethanol extract on estradiol levels, estrus cycle, and endometrial thickness in MSG-induced female rats. https://www.openveterinaryjournal.com/?mno=249709 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.38 AMA (American Medical Association) Style Arini LA, Mestika LCD. Effectiveness effects of Ulva lactuca ethanol extract on estradiol levels, estrus cycle, and endometrial thickness in MSG-induced female rats. Open Vet. J.. 2025; 15(10): 5221-5229. doi:10.5455/OVJ.2025.v15.i10.38 Vancouver/ICMJE Style Arini LA, Mestika LCD. Effectiveness effects of Ulva lactuca ethanol extract on estradiol levels, estrus cycle, and endometrial thickness in MSG-induced female rats. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5221-5229. doi:10.5455/OVJ.2025.v15.i10.38 Harvard Style Arini, L. A. & Mestika, . L. C. D. (2025) Effectiveness effects of Ulva lactuca ethanol extract on estradiol levels, estrus cycle, and endometrial thickness in MSG-induced female rats. Open Vet. J., 15 (10), 5221-5229. doi:10.5455/OVJ.2025.v15.i10.38 Turabian Style Arini, Luh Ari, and Luh Citrarasmi Dara Mestika. 2025. Effectiveness effects of Ulva lactuca ethanol extract on estradiol levels, estrus cycle, and endometrial thickness in MSG-induced female rats. Open Veterinary Journal, 15 (10), 5221-5229. doi:10.5455/OVJ.2025.v15.i10.38 Chicago Style Arini, Luh Ari, and Luh Citrarasmi Dara Mestika. "Effectiveness effects of Ulva lactuca ethanol extract on estradiol levels, estrus cycle, and endometrial thickness in MSG-induced female rats." Open Veterinary Journal 15 (2025), 5221-5229. doi:10.5455/OVJ.2025.v15.i10.38 MLA (The Modern Language Association) Style Arini, Luh Ari, and Luh Citrarasmi Dara Mestika. "Effectiveness effects of Ulva lactuca ethanol extract on estradiol levels, estrus cycle, and endometrial thickness in MSG-induced female rats." Open Veterinary Journal 15.10 (2025), 5221-5229. Print. doi:10.5455/OVJ.2025.v15.i10.38 APA (American Psychological Association) Style Arini, L. A. & Mestika, . L. C. D. (2025) Effectiveness effects of Ulva lactuca ethanol extract on estradiol levels, estrus cycle, and endometrial thickness in MSG-induced female rats. Open Veterinary Journal, 15 (10), 5221-5229. doi:10.5455/OVJ.2025.v15.i10.38 |