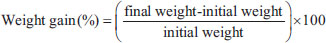| Research Article | ||
Open Vet. J.. 2026; 16(3): 1757-1770 Open Veterinary Journal, (2026), Vol. 16(3): 1757-1770 Research Article Integrative use of murine and zebrafish models for acute toxicity observation of ethanolic extract of Ocimum sanctum LinnMade Bagus Auriva Mataram1, Ulayatul Kustiati2, Golda Rani Saragih3, Dinda Aliffia3, Wilda Bunga Tina Sanjaya3, Anisa Putri Aviana3, Siti Isrina Oktavia Salasia4, Bondan Ardiningtyas5, Dwi Aris Agung Nugrahaningsih6, Yusril Yusuf7, Ika Dewi Ana8, Hevi Wihadmadyatami3, and Dwi Liliek Kusindarta3*1Laboratory of Clinical Pathology, Faculty of Veterinary Medicine, Brawijaya University, Malang, Indonesia 2Department of Pharmacology, Faculty of Veterinary Medicine, Brawijaya University, Malang, Indonesia 3Department of Anatomy, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogakarta, Indonesia 4Department of Clinical Pathology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia 5Department Pharmaceutics, Faculty of Pharmacy, Universitas Gadjah Mada, Yogyakarta, Indonesia 6Department of Pharmacology and Therapy, Faculty of Medicine, Public Health, and Nursing, Universitas Gadjah Mada. Indonesia 7Deparment of Physis, Faculty of Mathematics and Science, Universitas Gadjah Mada, Yogyakarta, Indonesia 8Department of Dental Biomedical Sciences, Faculty of Dentistry, Universitas Gadjah Mada, Yogyakarta, Indonesia *Corresponding Author: Dwi Liliek Kusindarta. Department of Anatomy, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, 55281, Indonesia. Email: indarta [at] ugm.ac.id Submitted: 24/06/2025 Revised: 15/01/2026 Accepted: 02/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
ABSTRACTBackground: Ocimum sanctum is used as a natural treatment for a variety of conditions and has been proven to possess a diverse array of therapeutic functions supported by pharmacological evidence. Aim: This study investigated the acute toxicity and determined the safe doses of O. sanctum Linn. ethanolic extracts, commonly used in traditional herbal medicine, using zebrafish embryos and murine models. Methods: Acute oral toxicity was used on 25 adult male Deutschland, Denken, and Yoken albino mice weighing 35–45 g, which were divided into five groups based on Organization for Economic Cooperation and Development 423. The O. sanctum Linn. Ethanolic extracts were given once and followed by observation for 15 days, after which the mice were sacrificed for hematological, biochemical, macroscopic, and histopathological organ analysis. The acute toxicity test for zebrafish embryos used five treatment concentrations, where one concentration group used 20 embryos placed on a plate and observed every 24–96 hours. Observations include hatchability, life or death of zebrafish embryos based on the occurrence of embryo coagulation, somite formation, tail release, and observations of abnormalities, including the presence or absence of heart rate. Results: The analysis results show the O. sanctum Linn. ethanolic extract proposes no toxicity according to acute oral toxicity in mice model and is considered relatively safe with an LC50 classification of category V (100 mg/L< LC50) in the zebrafish embryo acute toxicity test. Conclusion: These studies conducted by a combination of mice and zebrafish models have collectively demonstrated that the ethanolic extract of O. sanctum Linn. is safe for use. Keywords: Acute toxicity, Ethanolic extracts, Holy basil; Ocimum sanctum Linn; Zebrafish. IntroductionThroughout our daily existence, we encounter an abundance of flora. Plants present in homes, workplaces, and gardens can come into contact with humans and animals, whether they are used in food and drink, as herbal remedies, or simply for decorative purposes. Nevertheless, the absence of awareness regarding the possible hazards presented by these plants has consequences for human well-being, the welfare of animals, and the environment. Ocimum sanctum belongs to the Lamiaceae family. It is located in tropical Asia and the broader Indomalay regions. Additionally, it is present in India, Pakistan, Malaysia, Indonesia, Taiwan, and East Polynesia. The National Health Policy of India includes O. santum Linn, a native aromatic plant widely distributed throughout the Indian sub-continent, among the therapeutic plants regularly utilized in indigenous Indian medical systems (Valsan et al., 2022; Amarapalli et al., 2023). Ocimum sanctum, commonly referred to as holy basil, is a medicinal herb extensively employed in traditional practices to alleviate numerous disorders, including respiratory infections, febrile illnesses, dysentery, ocular complications such as glaucoma and cataracts, as well as gastrointestinal and hepatic disturbances (Prakash and Gupta, 2005; Pattanayak et al., 2010). Empirical evidence has further demonstrated its broad spectrum of pharmacological potentials encompassing analgesic, immunomodulatory, anti-ulcerogenic, anti-asthmatic, anticonvulsant, anticancer, antidiabetic, antistress, and antihyperlipidemic effects, together with cognitive-enhancing and neuroprotective activities (Kusindarta et al., 2016; Kusindarta et al., 2018; Raditya et al., 2020; Vithalkar and Patel, 2022). This plant has bioactive components, including flavonoids, tannins, phenols, saponins, and alkaloids with concentrations of 47.23% w/b, 21.68% w/b, 12.14% w/b, 1.28% w/b, and 0.22% w/b, respectively (Kustiati et al., 2022). However, thorough research is needed to determine the toxicity of this plant. Currently, limited evidence is available on assessing the toxicity of ethanol extract derived from basil leaves (Ocimum sanctum). The potential harmful effects of consuming the ethanolic extract of Ocimum sanctum Linn are currently unknown. Evaluating the toxicity of the ethanol extract derived from basil leaves is crucial, as it addresses safety concerns and identifies potential toxicological impacts (Ugbogu et al., 2021; Oliveira et al., 2024). A wide range of studies have proven that several herbal remedies, including mistletoe, germander, chamomile, comfrey, skullcap, and margosa oil, have been proven to be hepatotoxic. Regrettably, the precise frequency at which herbal treatments induce toxicity remains uncertain. The factors contributing to this uncertainty include patient overuse, remedy change, and medication misidentification (Modarresi Chahardehi et al., 2020; Veeren et al., 2020; Yang et al., 2020; Nguyen et al., 2021; Zhang et al., 2022). Therefore, investigations on the adverse effects and establishing safe doses of O. sanctum as a traditional medicine candidate were conducted in this study using mice and zebrafish embryo models. Thus, the purpose of this investigation was to ascertain the toxic effects and determine the safe doses of O. sanctum ethanolic extracts, commonly used in traditional herbal medicine, using murine models and zebrafish embryos. Materials and MethodsStudy period and locationThe research was carried out from May to October 2023 at the Integrated Laboratory for Research and Testing, Universitas Gadjah Mada, and the Drug, Food, and Cosmetic Testing Laboratory of Universitas Islam, Yogyakarta, Indonesia. Preparation of ethanolic extract of O. sanctum Linn leavesThe primary component utilized to prepare the extract was 1 kg of O. sanctum dried leaf powder (Merapi Farma Herbal, Yogyakarta, Indonesia). Using the maceration method, an ethanolic extract of O. sanctum was produced. For 48 hours, O. sanctum powder was soaked in 96% ethanol (Merck, Darmstadt, Germany), with occasional stirrings. Ethanol was soaked in till the yield of filtering dropped. To achieve a concentrated extract in the form of a paste with a weight of 10% of the main component, the filtrate was collected and evaporated using a vacuum rotary evaporator (Buchi, Flawil, Switzerland) on a waterbath Memmert, Schwabach, Germany) at a temperature of 60°C. The ethanolic extract was concentrated using a rotary evaporator and dried to a constant weight to ensure complete removal of the solvent. The resulting dried extract was reconstituted in 0.5% Na-CMC (sodium carboxymethyl cellulose) for oral administration; therefore, the animals were not exposed to ethanol during dosing. Mice oral acute toxicity test (OECD 423)All experimental procedures involving mice were performed in accordance with institutional ethical guidelines and approved by the Animal Ethics Committee, Faculty of Veterinary Medicine at Universitas Gadjah Mada, Indonesia, with registration number No.062/EC-FKH/Eks./2023. Male adult Mus musculus Deutschland, Denken, and Yoken (DDY strain) albino mice, 8 weeks old and weighing 35–45 g, were obtained from Universitas Gadjah Mada, Yogyakarta’s Integrated Laboratory for Research and Testing. Mice were housed in groups of five in a Tecniplast Plastic Reusable Cage (2154F00SU), Hohenpeißenberg, Germany, under standard environmental conditions prior to treatment. These included a regulated temperature of 23°C ± 3°C, a humidity of 75% ± 4%, and a 12-hour light/dark cycle every 7 days. Food and water were available at all times (ad libitum). The methodology for the oral acute toxicity study of the ethanolic O. sanctum Linn. extract was carried out in compliance with the national guidelines for non-clinical toxicity tests in vivo, adopted by the Organization for Economic Cooperation and Development (OECD) number 423, and has been issued by the National Drug and Food Control, Indonesia (2022). Every 5 individuals were grouped in one group, and in this study, five doses were used, namely the dose groups of 5 mg / kg, 50 mg / kg, 300 mg / kg, and 2,000 mg / kg. The control group was given 0.5% Na-CMC. The observations were made the first half-hour after the dosing and every 4 hours for 15 days. Every 3 days, the weight of the mice was noted. The following formula was used to determine the animal’s weight growth (%) (Ugwah-Oguejiofor et al., 2019):
After the experiment was completed, the samples were collected. Collected organ samples were weighed in order to observe the gross anatomy and histopathological examinations. Furthermore, the blood samples were run for hematology and bioassay analysis (OECD, 2002; BPOM, 2022). Toxicology analysis in miceToxicity in the acute oral test annotated with 50% Lethal Dose (LD50) in mice was determined by evaluating the global harmonized system (GHS) levels according to the categories specified in OECD 423, which include category 1 (less than 5 mg/kg), category 2 (between 5 mg/kg and 50 mg/kg), category 3 (between 50 mg/kg and 300 mg/kg), category 4 (between 300 mg/kg and 2,000 mg/kg), and category 5 (between 2,000 mg/kg and 5,000 mg/kg). Gross anatomy analysisThe 15th day brought the sacrifice of every mouse. The method of euthanasia involved intraperitoneal injections of xylazine (Interchemie, Metaalweg, Netherlands) at 2% dosages and ketamine (Kepro, Maagdenburgstraat, Netherlands) at 10% doses of 100 mg/kg BB. This approach facilitated swift and painless euthanasia, adhering to OECD 423. The organ samples, i.e., brain, lung, kidney, liver, spleen, and testes, were collected. The samples were rinsed with NaCl 0.9%. (Merck, Darmstadt, Germany). Filter paper was used to dry the organ samples. The gross pathological features, such as color, irregularities, and consistency, were evaluated. The organ sample mass was weighed, and in relation to body weight, the relative organ weights were computed and documented using the following formula:
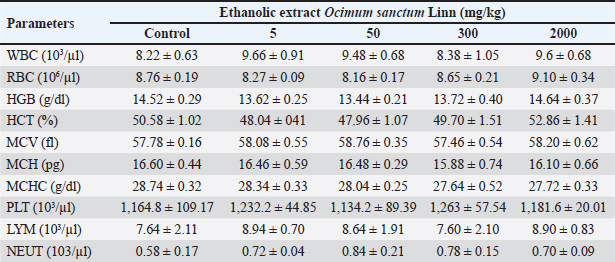
After the relative organ weight procedure, the organs were then collected into 10% paraformaldehyde and used for histology slide preparation. Histopathological analysisFor histopathological analysis, the fixed tissue was dehydrated with an ethanol series (Merck, Darmstadt, Germany), cleared with xylol (Merck, Darmstadt, Germany), and embedded with paraffin wax (Leica, Melbourne, Australia). Embedded tissues were cut using Rotary Microtome Shandon Finesse 325 from Thermo Fisher Scientific (Watertown, USA) in serial sections with 5 µm thickness. After being put on slides, the tissues were heated to ambient temperature and then cooled. For hematoxylin and eosin staining (Leica, Wetzlar, Germany), the sections underwent xylene deparaffinization (Merck, Darmstadt, Germany) and hydrated with descending ethanol (Merck, Darmstadt, Germany). The slides were stained with hematoxylin and eosin and inspected using a light microscope (Olympus BX51, Tokyo, Japan). The images were taken using Optilab software (Optilab, Yogyakarta, Indonesia). Hematology analysisThe blood samples were collected using microhematocrit and were preserved in EDTA-coated tubes (Vaculab, Surabaya, Indonesia). Hematology was performed using a Sysmex KX-21 hematology analyzer (Pelican Medical Systems, Kochi, India). Hemoglobin (HGB), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular volume (MCV), lymphocyte (LYM), red blood cells (RBC), hematocrit (HCT), white blood cells (WBC), neutrophil (NEUT), platelet count (PLT), and mean corpuscular hemoglobin (MCH) were among the hematological parameters examined. Biochemical analysisThe blood samples were collected using microhematocrit and were preserved in plain tubes (Vaculab, Surabaya, Indonesia). Serum was obtained by centrifuging blood at 4,000 rpm for 10 minutes. Serum samples were analyzed to determine the concentrations of albumin, glucose, creatinine, blood urea nitrogen (BUN), alanine aminotransferase (ALT), aspartate aminotransferase (AST), and cholesterol. Zebrafish embryo acute toxicity (OECD 236)All zebrafish experimental protocols were conducted following the OECD Guideline No. 236 and approved by the Drug, Food, and Cosmetic Testing Laboratory of Universitas Islam Indonesia, Indonesia, with registration number No.004/LPOMK/VII/2023. Fertilized zebrafish (Danio rerio) embryos at 2–4 hours post-fertilization were selected. Only viable and morphologically normal embryos were used, while unfertilized or coagulated ones were excluded. The Embryos of zebrafish that have passed the selection stage are then transferred into the well. Each concentration tested used 20 embryos (one embryo per well), which were included in each plate. Embryos were transferred using a 100 µl micropipette. A negative control/solvent using distilled water and a positive control (3,4-Dichloroaniline) were added at each concentration. Plates containing zebrafish embryos were placed at room temperature (±26ºC) and observed every 24–96 hours. The zebrafish embryos were exposed to the extract at concentrations of 6.25, 12.5, 25, 50, and 70 µg/ml, selected in accordance with OECD Guideline No. 236 (Fish Embryo Acute Toxicity Test). The chosen concentrations were within the solubility limit of the extract and below the threshold for inducing physical stress. Observations include hatchability, life or death of zebrafish embryos based on the occurrence of embryo coagulation, somite formation, tail release less than 24 hours after exposure, and the ability to hatch out of the chorion less than 48 hours after exposure, and observations of abnormalities, or abnormalities including the presence or absence of heart rate (OECD, 2013). Toxicology analysis in fish embryo testToxicity values from the fish embryo test on zebrafish (Danio rerio) annotated with 50% Lethal concentration (LC50) were classified into the following categories: class I (less than 0.1 mg/l), class II (between 0.1 mg/l1 and mg/l), class III (between 1 mg/l and 10 mg/l), class IV (between 10 mg/l and 100 mg/l), and class V (above 100 mg/l) (Xiong et al., 2022). Statistical analysisThe experimental data were examined by GraphPad Prism version 9 (La Jolla, CA, USA). Data were expressed as mean ± standard error of the mean (SEM). Prior to analysis, the assumptions of normality and homogeneity of variance were verified using the Shapiro–Wilk and Levene’s tests, respectively. One-way Analysis of variance was conducted to assess differences among groups, followed by Tukey’s post-hoc test for pairwise comparisons when applicable. Statistical significance was accepted at p < 0.05. Ethical approvalThe study was granted approval by the Drug, Food, and Cosmetic Testing Laboratory of Universitas Islam Indonesia, Indonesia, with registration number No.004/LPOMK/VII/2023 for zebrafish embryo acute toxicity, and the Ethics Committee of the Faculty of Veterinary Medicine at Universitas Gadjah Mada, Indonesia, with registration number No.062/EC-FKH/Eks./2023 for mice oral acute toxicity. ResultsAcute oral toxicity on mice modelClinical and behavioral analysisMice were watched for any harmful signs or symptoms at various time intervals: 0 minutes; 1, 2, 3, 4, 8, 12, 16, 20, 24 hours; and then every day for a duration of 15 days. There were no clinical symptoms or potentially dangerous indicators shown by the observations (Table 1). No mortality was observed in mice administered a dose of 2,000 mg/kg, indicating that the median lethal dose (LD50) was not achieved. Accordingly, this herbal formulation can be considered safe at the highest tested dose level. The herbal product falls under category 5 or is not categorized, which indicates that, according to the global harmonization system standards for acute toxicity, it is essentially non-toxic (2,000 mg/kg BW < LD50). The mice’s body weights rose after the herbal preparation was administered, albeit there were differences in each mouse’s dosage and individual results (Fig. 1; Table 2). Furthermore, there was no statistically significant difference (p > 0.05) seen in the average organ weight and relative organ weight index across all groups (Fig. 2; Tables 4 and 5).
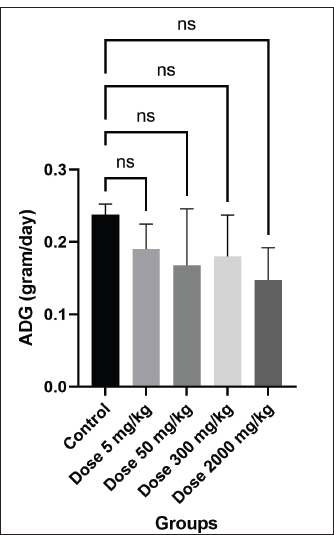
Fig. 1. The increase in weight for each treatment group and the control group (p > 0.05). ADG: Average Daily Gain.
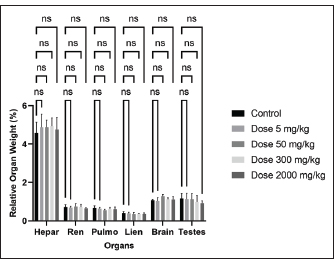
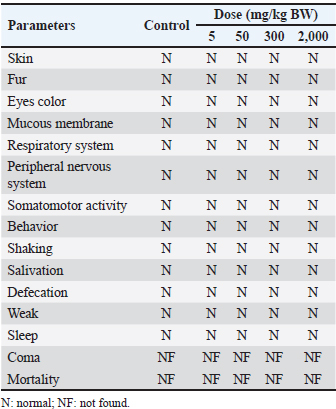
Fig. 2. The relative organ weight of all treatment groups and the control group (p > 0.05). Table 1. Clinical signs and behavioral observations of acute toxicity study of ethanolic extract Ocimum sanctum Linn. for control and treated groups.
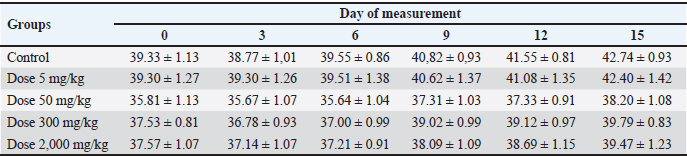
Table 2. Male mice weights (mean ± SEM) after administration of the herbal preparation during the experiment.
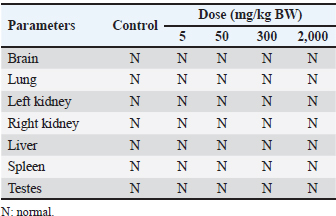
Table 3. Gross pathology of the vital organ in male mice at day-15 after treatment with ethanolic extract of Ocimum sanctum Linn.
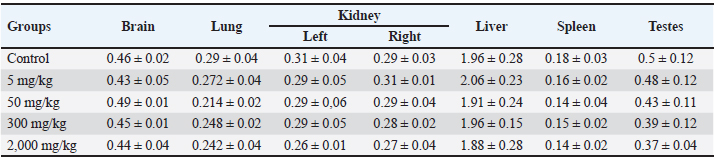
Table 4. Effect of oral administration of ethanolic extract of Ocimum sanctum Linn on average organ weight (g) of male mice.
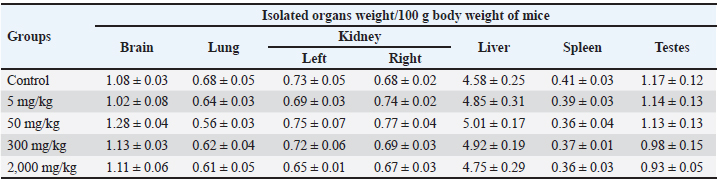
Table 5. Effect of oral administration of ethanolic extract of Ocimum sanctum Linn on relative organ weight (g) of male mice.
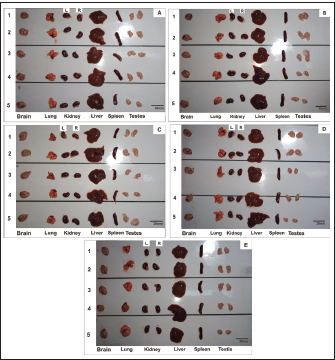
Macroscopic examinationsVital organs, including the brain, lungs, kidneys, liver, spleen, and testes of both the control group of mice and the animals receiving the herbal mixture at the highest dosage (2,000 mg/kg BW), are examined microscopically. The macroscopic alterations in the treatment groups and the control group did not significantly vary from one another (Fig. 3; Table 3). There is no discernible difference in any of the groups’ organ-to-body weight indices (p > 0.05).
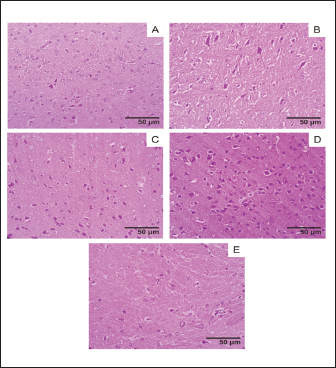
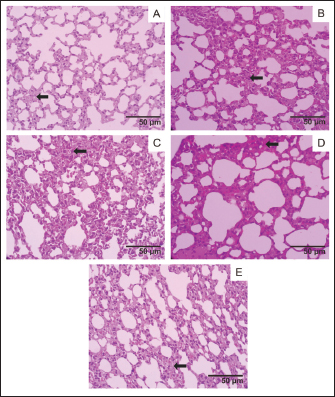
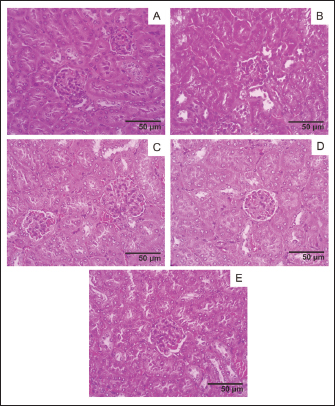
Fig. 3. Gross Macroscopy of the Vital Organ (Brain, Lung, Kidney, Liver, Spleen, and Testes) after administration of Ethanolic Extract Ocimum sanctum Linn. (A) Control Group; (B) Dose 5 mg/kg; (C) Dose 50 mg/kg; (D) Dose 300 mg/kg; (E) 2,000 mg/kg. L: left; R: right. Histopathological examinationsHistological analysis of major organs showed normal architecture in all groups, with no dose-related lesions. While the control group and the treatment groups both had lung symptoms consistent with interstitial pneumonia (Figs. 4–9), indicating that these findings were likely incidental and not attributable to the O. sanctum extract.
Fig. 4. Brain histology acute oral toxicity test of mice on day 15 of single dose administration. (A) Control Group; (B) Dose 5 mg/kg; (C) Dose 50 mg/kg; (D) Dose 300 mg/kg; (E) 2,000 mg/kg. (400×, H&E stained).
Fig. 5. Lung histology, acute oral toxicity test of mice on day 15 of single dose administration. (A) Control Group; (B) Dose 5 mg/kg; (C) Dose 50 mg/kg; (D) Dose 300 mg/kg; (E) 2,000 mg/kg. (400×, H&E stained).
Fig. 6. Kidney histology, acute oral toxicity test of mice on day 15 of single dose administration. (A) Control Group; (B) Dose 5 mg/kg; (C) Dose 50 mg/kg; (D) Dose 300 mg/kg; (E) 2,000 mg/kg. (400×, H&E stained).
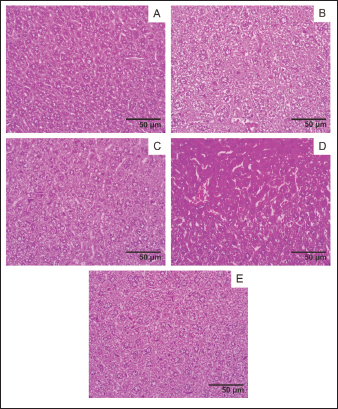
Fig. 7. Liver histology, acute oral toxicity test of mice on day 15 of single dose administration. (A) Control Group; (B) Dose 5 mg/kg; (C) Dose 50 mg/kg; (D) Dose 300 mg/kg; (E) 2,000 mg/kg. (400×, H&E stained).
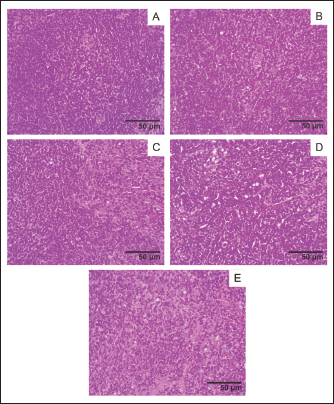
Fig. 8. Spleen histology, acute oral toxicity test of mice on day 15 of single dose administration. (A) Control Group; (B) Dose 5 mg/kg; (C) Dose 50 mg/kg; (D) Dose 300 mg/kg; (E) 2,000 mg/kg. (400×, H&E stained).
Fig. 9. Testes histology, acute oral toxicity test of mice on day 15 of single dose administration. (A) Control Group; (B) Dose 5 mg/kg; (C) Dose 50 mg/kg; (D) Dose 300 mg/kg; (E) 2,000 mg/kg. (400×, H&E stained). Blood hematology and serum biochemistry analysisThe effects of acute oral toxicity on hematological tests are summarized in Table 6. Hematological parameters such as PLT, LYM, NEUT, MCV, MCH, HCT, WBC, RBC, HGB, and HCT show no significant differences (p > 0.05) between treated mice and control, and the same results also provide in serum biochemistry analysis all the parameters including ALT, AST, BUN, total protein, creatinine, glucose, albumin, cholesterol are presents in normal range (p > 0.05) (Table 7). Table 6. Effect of acute oral toxicity of ethanolic extract of Ocimum sanctum Linn administration on hematological parameters in male mice (mean ± SEM).
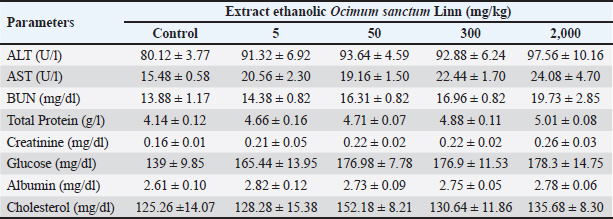
Table 7. Effect of acute oral toxicity of ethanolic extract of Ocimum sanctum Linn administration on blood biochemistry parameters in male mice.
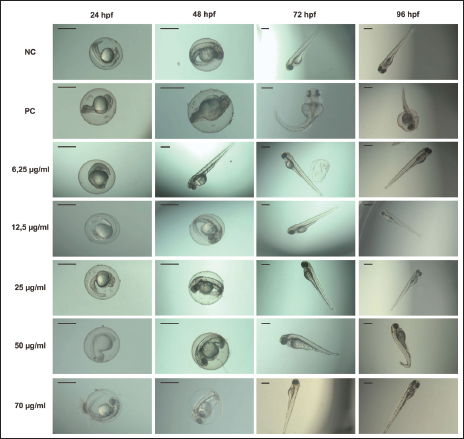
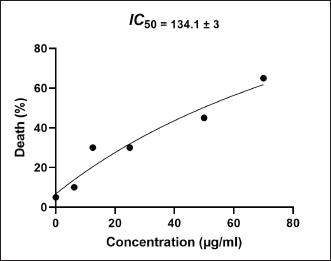
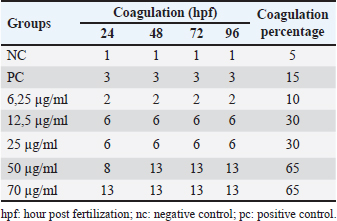
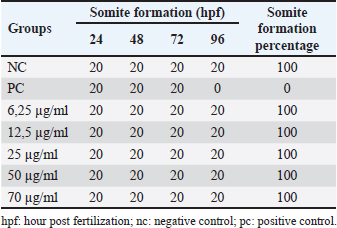
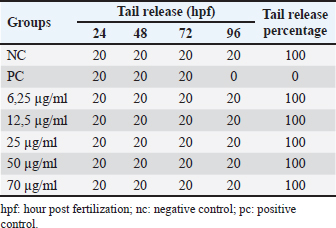
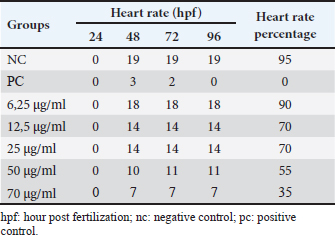
Fish embryo acute toxicity on the zebrafish (Danio rerio)The goal of the fish embryo acute toxicity (FET) test, which is outlined in OECD test guideline (TG) 236, is to assess a substance’s toxicity using zebrafish (Danio rerio) embryos. The goal of the FET test’s invention was to match the acute fish test’s sensitivity level. The method makes use of four basic morphological endpoints: the embryo’s coagulation, the absence of somite development, the absence of a heartbeat, and the tail’s non-detachment. Based on the zebrafish embryo toxicity analysis, coagulation was observed at a heart rate reduction of approximately 35% when exposed to concentrations up to 70 μg/ml of Ocimum sanctum ethanolic extract (Fig. 10; Tables 8 and 11). Moreover, all tail buds are released within 96 hours (Fig. 10; Table 10), and somite development occurs in 100% of the embryos. The herbal product is classified as category V according to the system criteria for acute toxicity (Fig. 11).
Fig. 10. The representative toxicological effects of Ethanolic Extract Ocimum sanctum Linn of zebrafish embryo toxicity tests observed under the stereomicroscope (scale bar 500 µm). hpf: hours post fertilization.
Fig. 11. Linear regression of Ethanolic Extract Ocimum sanctum Linn of zebrafish embryo log LC50. Table 8. Coagulation in zebrafish (Danio rerio) embryo after 96 hours of exposure.
Table 9. Somite formation in zebrafish (Danio rerio) embryo after 96 hours of exposure.
Table 10. Release of the tail-bud portion of the yolk in zebrafish (Danio rerio) embryo after 96 hours of exposure.
Table 11.Heart rate in zebrafish (Danio rerio) embriyo after 96 hours exposure.
DiscussionAcute oral toxicity refers to an assessment conducted on a test substance that is administered by oral routes in order to determine the description of exposure likely to result in death, as well as identifying the character of the effects likely to occur within a set observation period. The aim of acute oral systemic toxicity studies or studies with a comparable official method, guidelines, or regulatory requirement, is to identify the nature of the ‟acute toxic class” and to apply factors which give an indication of LD50, the dose expected to cause death in the near term, so that appropriate labelling can be determined. The study is based on the limit dose, although further studies might be run using lower doses. Identification of the class of toxicity depends on the substance’s means of exposure, but for those with oral means of exposure, it is possible to classify substances in the acute toxicity class based on the study. Heavily colored test substances may interfere with the measurement of test substance concentrations in the application solution, resulting in unreliable or misleading results. Such substances need to be given careful attention. Additionally, sparingly soluble substances that are used in pre-weighed packets should be assessed for homogeneity prior to dose formulation. From all the toxic class methods that were identified, a total of six were utilized, which include the up and down procedure, acute toxic class, the OECD toxic class, the British toxic class, the US EPA, and the oral LD50 method. These methods often differentiate between the toxic class of vapors, medical, herbal, and gases, and that of solids and liquids. The OECD 423 toxic technique was used in this study due to its background (Alelign et al., 2020; Burden et al., 2020; Kharchoufa et al., 2020; Moxon et al., 2020; Mansouri et al., 2021; Bercu et al., 2021; Graham et al., 2021; Huang et al., 2021; Pistollato et al., 2021). Based on an acute oral toxicity paradigm in mice, the analytical findings indicate that the ethanolic extract of O. sanctum Linn. does not present any toxicity. Generally, O. sanctum is regarded as a medicinal plant with very low toxicity. However, variations may occur among different Ocimum species or in future studies, depending on the extract standardization, experimental methods, and plant material used. Additionally, targeted controlled studies seem to indicate that there is a need for the traditional herbal medicines to be screened for toxic substances and to be quality controlled. Such experiments need to be conducted with different methodologies. The toxicity profile observed in this study corresponds to a single species, O. sanctum. However, future investigations on other Ocimum species or different cultivation conditions may reveal variations due to environmental and genetic factors. The acute oral toxicity assessment of O. sanctum ethanolic extract showed no mortality or observable toxic symptoms at a dose of 2,000 mg/kg body weight, indicating that the LD50 exceeds this level and the extract can be considered practically non-toxic according to OECD and GHS classification. No significant (p > 0.05) changes were observed in body weight, organ weight indices, or behavioral responses, suggesting that the extract did not interfere with physiological or metabolic functions. Histopathological examination of the brain, lung, kidney, liver, spleen, and testis revealed normal tissue architecture in all treatment groups compared with the control (Figs. 4–9). The liver exhibited intact hepatic cords and central veins without evidence of necrosis or fatty degeneration. Kidney sections showed normal glomeruli and tubular structures with no cellular degeneration or interstitial inflammation. Brain tissues maintained normal neuronal morphology without signs of edema or hemorrhage. The spleen displayed preserved white and red pulp structures, while testicular sections revealed regular spermatogenic cell layers and seminiferous tubule organization. Although mild interstitial pneumonia was noted in both control and treated lungs, it appeared non-dose-dependent and likely incidental. Overall, the absence of dose-related pathological alterations supports the conclusion that O. sanctum ethanolic extract at doses up to 2,000 mg/kg did not induce histological toxicity. Hematological and biochemical evaluation revealed no significant (p > 0.05) changes following acute oral administration of O. sanctum ethanolic extract up to 2,000 mg/kg (Tables 6 and 7). Key hematological indices (WBC, RBC, HGB, HCT, MCV, MCH, MCHC, PLT) remained stable and showed no dose-dependent trends, indicating no effect on erythropoiesis, leukopoiesis, or thrombopoiesis. Liver enzymes (ALT, AST) and renal markers (BUN, creatinine) displayed only minor, non-significant (p > 0.05) fluctuations at higher doses and remained within expected physiological ranges for mice, supporting preserved hepatic and renal function. Glucose and cholesterol levels also varied slightly without statistical significance. Taken together with the absence of mortality at 2,000 mg/kg (LD50 not reached), these findings indicate that a single acute oral exposure to the extract does not produce clinically relevant hematological or biochemical toxicity under the study conditions. Overall, these findings demonstrate that O. sanctum ethanolic extract is safe in acute exposure and supports its continued use as a medicinal plant, although further studies are required to evaluate its sub-chronic and chronic safety profiles. Oral route is one such route where the chances of toxicity are less compared to the parenteral route or other routes, considering acute effects. All animals remained normal in appearance and behavior throughout the observation period. Parameters such as skin, fur, mucous membranes, respiratory and nervous system responses, as well as somatomotor activity, showed no deviations from normal. The absence of tremors, salivation, or defecation abnormalities further supports the non-toxic nature of the extract. Of note, the widespread use of the herb for various therapeutic purposes within the country underscores the importance of evaluating its safety and potential toxicity. The effect also needs to be studied in the long-term use and in subchronic and chronic effects (Kamel et al., 2023; Luke et al., 2021; Yuniarti et al., 2021; Žabka et al., 2021;). Following the acute oral toxicity evaluation in mice, which revealed no mortality or significant organ-specific toxicity up to the tested dose, the safety assessment was further supported using an aquatic model to evaluate developmental toxicity. The FET test, as outlined in OECD Test Guideline 236, provides a sensitive and ethically preferred alternative to the acute fish test for evaluating chemical or natural product toxicity (OECD, 2013). The test assesses four morphological endpoints—embryo coagulation, absence of somite formation, lack of heartbeat, and non-detachment of the tail bud—within 96 hours post-fertilization (hpf). In this study, zebrafish embryos exposed to Ocimum sanctum ethanolic extract exhibited a dose-dependent increase in coagulation and a reduction in somite formation at higher concentrations. Specifically, coagulation increased up to 65% at 50 µg/ml and 70 µg/ml, while somite formation decreased to approximately 35% at 70 µg/ml, indicating moderate developmental interference at elevated doses (Tables 8 and 9). Despite these effects at the upper exposure levels, no significant (p > 0.05) heartbeat cessation or tail detachment abnormalities were observed, suggesting that embryonic development was largely maintained within nonlethal concentration ranges. Based on OECD TG 236 classification criteria, the extract can be categorized as Class V (low acute toxicity), consistent with previous findings on Ocimum species showing limited embryotoxicity and favorable safety profiles in aquatic and mammalian models (Capparucci et al., 2022). While zebrafish FET trials were thought to be generally safe, with an LC50 categorization of category V, data showed that there were occurrences of embryo death up to 96 hours. Therefore, it is advisable to administer low or moderate doses for regenerative or anti-inflammatory therapy. Overall, the zebrafish embryotoxicity model shows significant potential in evaluating the toxicity of herbal substances and serves as an excellent substitute for traditional laboratory animals like rats, mice, or rabbits. The embryotoxicity assessment in zebrafish adheres to the 3R principle of animal welfare and follows a comprehensive screening strategy of herbal medicine to arrive at clinical results. The model’s low cost, ease of handling, ability to employ small quantities of test chemicals, and high throughput screening contribute to its competitiveness and performance in toxicity investigations. Significantly, the early developmental phases of zebrafish exhibit reduced sensitivity to chemical exposure, resulting in minimal pain or discomfort. Notably, zebrafish embryos and larvae below 5 days post-fertilization (dpf) have underdeveloped nociceptive and nervous systems, rendering them less sensitive to chemical-induced distress. According to OECD and EU animal welfare guidelines, experiments conducted during these early developmental stages are categorized below the pain perception threshold and are therefore exempt from higher animal ethics restrictions. Consequently, the zebrafish model is widely recognized as a scientifically valid and ethically acceptable alternative for preliminary toxicity evaluation of herbal compounds (Jayasinghe and Jayawardena, 2019). Our findings align with existing literature demonstrating a broad safety margin for various Ocimum species. Acute and sub-chronic toxicity studies of O. basilicum have shown no mortality or behavioral alterations at oral doses up to 5 g/kg in Wistar rats (Rasekh et al., 2012). Similarly, O. tenuiflorum exhibited no significant adverse effects at doses up to 2,000 mg/kg in Wistar rats (Murugan et al., 2025). Ocimum gratissimum also demonstrated safety at doses up to 5,000 mg/kg in Wistar rats (Assih et al., 2022). Furthermore, Ocimum forskolei ethanolic extracts had an LD50 exceeding 8 g/kg (Zahran and El-Khadragy, 2019). To complement rodent toxicity data and provide insight into potential developmental effects, zebrafish embryo models have also been employed to assess the toxicity of Ocimum extracts. Zebrafish embryo models have been employed to assess the developmental toxicity of Ocimum extracts. For instance, O. basilicum essential oil caused dose-dependent effects such as delayed hatching, pericardial edema, and reduced heart rate at concentrations up to 200 µl/l (Capparucci et al., 2022). In contrast, herbal formulations containing O. tenuiflorum and O. gratissimum generally exhibited lower toxicity compared to commercial products Ashwin et al., 2025. Collectively, these findings support the conclusion that O. sanctum, like other Ocimum species, is pharmacologically active yet toxicologically benign at relevant experimental concentrations, validating its use in traditional and therapeutic applications. ConclusionStudies conducted by a combination of mice and zebrafish models have collectively demonstrated that the ethanolic extract of O. sanctum Linn. exhibits low acute toxicity. These findings suggest that the extract possesses a favorable safety margin within the tested concentrations. The demonstrated safety profile supports its use in formulating safe herbal preparations and guides dosing strategies for future pharmacological studies, including neuroprotective or combination therapy investigations. Furthermore, zebrafish embryo data provide an additional in vivo model to assess developmental toxicity, helping to streamline preclinical safety evaluation for new herbal products. However, further research is needed to better understand the potential sub-acute and chronic oral toxicities associated with this extract. Additionally, it is important to establish a definitive, safe dosage to prevent adverse effects arising from excessive consumption. We recommend conducting additional studies to include sub-acute and chronic toxicity evaluations in order to establish the extract’s long-term safety profile, determine its no-observed-adverse-effect level, and facilitate dose extrapolation to humans. AcknowledgmentThis research was supported by the Directorate General of Higher Education, Research and Technology, Ministry of Education, Culture, Research and Technology, Republic of Indonesia, through the Regular Fundamental Research in the year of 2025. Conflict of interestThe Authors declare that there is no conflict of interest. FundingThe research was funded by the Ministry of Education, Culture, Research, and Technology 2025. Authors’ contributionsMBAM, SIOS, BA, DLK, HW: Helped in the study’s conception and design. MBAM, UK, GRS: Performed sample and data collection. MBAM, DA, WBTS, UK, DLK, HW: validated data and conducted statistical analysis. MBAM, APA, UK, HW: Created the graphs and wrote the manuscript. SIOS, BA, DLK, HW: Supervised the study. Every author has read, evaluated, and given their approval to the completed work. Data availabilityAll data supporting the findings of this study are available within the manuscript. ReferencesAlelign, T., Chalchisa, D., Fekadu, N., Solomon, D., Sisay, T., Debella, A. and Petros, B. 2020. Evaluation of acute and sub-acute toxicity of selected traditional antiurolithiatic medicinal plant extracts in Wistar albino rats. Toxicol. Rep. 7, 1356–1365. Amarapalli, G., Pidigam, S., Rao, P.S., Thuraga, V., Mohammad, S.K. and Suresh, J. 2023. Holy Basil (Ocimum sanctum L.): an important Indian medicinal and aromatic plant: its properties, utilization and genetic improvement. In Medicinal and aromatic plants of india. Eds., Máthé, A and István, S. Cham, Switzherland: Springer International Publishing, pp: 13–35. Ashwin, S.P., Remmiya, M.V., Aravind, K.S. and Rajeshkumar, S. 2025. Comparative embryonic toxicology evaluation of Ocimum tenuiflorum and Ocimum gratissimum herbal formulation-based oral rinse and commercial oral rinse. Plant Sci. Today 12(1), 1–10. Assih, M., Akinmoladun, O.F. and Akinmoladun, A.F. 2022. In-vitro and in-vivo toxicological studies of hydroethanolic leaf extract of Ocimum gratissimum Linn (Lamiaceae) in Wistar rats. J. Med. Palnts. Res. 16(4), 91–98. Bercu, J., Masuda-Herrera, M.J., Trejo-Martin, A., Hasselgren, C., Lord, J., Graham, J., Schmitz, M., Milchak, L., Owens, C., Lal, S.H., Robinson, R.M., Whalley, S., Bellion, P., Vuorinen, A., Gromek, K., Hawkins, W.A., Van De Gevel, I., Vriens, K., Kemper, R., Naven, R., Ferrer, P. and Myatt, G.J. 2021. A cross-industry collaboration to assess if acute oral toxicity (Q)SAR models are fit-for-purpose for GHS classification and labelling. Regul. Toxicol. Pharmacol. 120, 104843. BPOM. 2022. Peraturan Badan Pengawas Obat Dan Makanan Nomor 10 Tahun 2022 Tentang Pedoman Uji Toksisitas Praklinik Secara in vivo. Badan Pengawas Obat Dan Makanan Republik Indonesia. 1–220. Burden, N., Benstead, R., Benyon, K., Clook, M., Green, C., Handley, J., Harper, N., Maynard, S.K., Mead, C., Pearson, A., Ryder, K., Sheahan, D., Van Egmond, R., Wheeler, J.R. and Hutchinson, T.H. 2020. Key opportunities to replace, reduce, and refine regulatory fish acute toxicity tests. Environ. Toxicol. Chem. 39(10), 2076–2089. Capparucci, F., De Benedetto, G., Natale, S., Pecoraro, R., Iaria, C. and Marino, F. 2022. Evaluation of anaesthetic effect of commercial basil (Ocimum basilicum) on zebrafish (Danio rerio) embryos. Fishes 7(6), 318. Graham, J.C., Rodas, M., Hillegass, J. and Schulze, G. 2021. The performance, reliability and potential application of in silico models for predicting the acute oral toxicity of pharmaceutical compounds. Regul. Toxicol. Pharmacol. 119, 104816. Huang, T., Sun, G., Zhao, L., Zhang, N., Zhong, R. and Peng, Y. 2021. Quantitative Structure-Activity Relationship (QSAR) Studies on the Toxic Effects of Nitroaromatic Compounds (NACs): a Systematic Review. Int. J. Mol. Sci. 22(16), 8557. Jayasinghe, C.D. and Jayawardena, U.A. 2019. Toxicity Assessment of Herbal Medicine Using Zebrafish Embryos: a Systematic Review. eCAM 1, 7272808. Kamel, F.O., Karim, S., Bafail, D.A.O., Aldawsari, H.M., Kotta, S. and Ilyas, U.K. 2023. Hepatoprotective effects of bioactive compounds from traditional herb Tulsi (Ocimum sanctum Linn) against galactosamine-induced hepatotoxicity in rats. Front. Pharmacol. 14, 1213052. Kharchoufa, L., Bouhrim, M., Bencheikh, N., El Assri, S., Amirou, A., Yamani, A., Choukri, M., Mekhfi, H. and Elachouri, M. 2020. Acute and Subacute Toxicity Studies of the Aqueous Extract from Haloxylon scoparium Pomel (Hammada scoparia (Pomel)) by Oral Administration in Rodents. BioMed. Res. Int. 1, 4020647. Kusindarta, D.L., Wihadmadyatami, H. and Haryanto, A. 2016. Ocimum sanctum Linn. stimulate the expression of choline acetyltransferase on the human cerebral microvascular endothelial cells. Vet. World. 9(12), 1348–1354. Kusindarta, D.L., Wihadmadyatami, H., Jadi, A.R., Karnati, S., Lochnit, G., Hening, P., Haryanto, A., Auriva, M.B. and Purwaningrum, M. 2018. Ethanolic extract Ocimum sanctum. Enhances cognitive ability from young adulthood to middle aged mediated by increasing choline acetyl transferase activity in rat model. Res. Vet. Sci. 118, 431–438. Kustiati, U., Wihadmadyatami, H. and Kusindarta, D.L. 2022. Dataset of Phytochemical and secondary metabolite profiling of holy basil leaf (Ocimum sanctum Linn) ethanolic extract using spectrophotometry, thin layer chromatography, Fourier transform infrared spectroscopy, and nuclear magnetic resonance. DIB 40, 107774. Luke, A.M., Patnaik, R., Kuriadom, S.T., Jaber, M. and Mathew, S. 2021. An in vitro study of Ocimum sanctum as a chemotherapeutic agent on oral cancer cell-line. Saud. J. Biol. Sci. 28(1), 887–890. Mansouri, K., Karmaus, A.L., Fitzpatrick, J., Patlewicz, G., Pradeep, P., Alberga, D., Alepee, N., Allen, T.E.H., Allen, D., Alves, V.M., Andrade, C.H., Auernhammer, T.R., Ballabio, D., Bell, S., Benfenati, E., Bhattacharya, S., Bastos, J.V., Boyd, S., Brown, J.B., Capuzzi, S.J., Chushak, Y., Ciallella, H., Clark, A.M., Consonni, V., Daga, P.R., Ekins, S., Farag, S., Fedorov, M., Fourches, D., Gadaleta, D., Gao, F., Gearhart, J.M., Goh, G., Goodman, J.M., Grisoni, F., Grulke, C.M., Hartung, T., Hirn, M., Karpov, P., Korotcov, A., Lavado, G.J., Lawless, M., Li, X., Luechtefeld, T., Lunghini, F., Mangiatordi, G.F., Marcou, G., Marsh, D., Martin, T., Mauri, A., Muratov, E.N., Myatt, G.J., Nguyen, D.T., Nicolotti, O., Note, R., Pande, P., Parks, A.K., Peryea, T., Polash, A.H., Rallo, R., Roncaglioni, A., Rowlands, C., Ruiz, P., Russo, D.P., Sayed, A., Sayre, R., Sheils, T., Siegel, C., Silva, A.C., Simeonov, A., Sosnin, S., Southall, N., Strickland, J., Tang, Y., Teppen, B., Tetko, I.V., Thomas, D., Tkachenko, V., Todeschini, R., Toma, C., Tripodi, I., Trisciuzzi, D., Tropsha, A., Varnek, A., Vukovic, K., Wang, Z., Wang, L., Waters, K.M., Wedlake, A.J., Wijeyesakere, S.J., Wilson, D., Xiao, Z., Yang, H., Zahoranszky-Kohalmi, G., Zakharov, A.V., Zhang, F.F., Zhang, Z., Zhao, T., Zhu, H., Zorn, K.M., Casey, W. and Kleinstreuer, N.C. 2021. CATMoS: collaborative acute toxicity modeling suite. Environ. Health Perspect. 129(4), 47013. Modarresi Chahardehi, A., Arsad, H. and Lim, V. 2020. Zebrafish as a successful animal model for screening toxicity of medicinal plants. Plants 9(10), 1345. Moxon, M., Strupp, C., Aggarwal, M., Odum, J., Lewis, R., Zedet, S. and Mehta, J. 2020. An analysis of the setting of the acute reference dose (ARfD) for pesticides in Europe. Regul. Toxicol. Pharmacol. 113, 104638. Murugan, S.K., Bethapudi, B. and Reddy, P.S. 2025. Assessment of genotoxicity and acute oral toxicity of a standardized Ocimum tenuiflorum extract (Holixer™). J. Toxicol. Sci. 50(1), 1–10. Nguyen, P.H., Tran, V.D., Pham, D.T., Dao, T.N.P. and Dewey, R.S. 2021. Use of and attitudes towards herbal medicine during the COVID-19 pandemic: a cross-sectional study in Vietnam. Eur. J. Integr. Med. 44, 101328. OECD. 2002. Test No. 423: Acute Oral toxicity—Acute Toxic Class Method. OECD. 2013. Test No. 236: Fish Embryo Acute Toxicity (FET) Test. Oliveira, G.D.S., Mcmanus, C., Sousa, H.A.D.F., Santos, P.H.G.D.S. and Dos Santos, V.M. 2024. A mini-review of the main effects of essential oils from Citrus aurantifolia, Ocimum basilicum, and Allium sativum as safe antimicrobial activity in poultry. Animals 14(3), 382. Pattanayak, P., Behera, P., Das, D. and Panda, S. 2010. Ocimum sanctum Linn. A reservoir plant for therapeutic applications: an overview. Phcog. Rev. 4(7), 95–105. Pistollato, F., Madia, F., Corvi, R., Munn, S., Grignard, E., Paini, A., Worth, A., Bal-Price, A., Prieto, P., Casati, S., Berggren, E., Bopp, S.K. and Zuang, V. 2021. Current EU regulatory requirements for the assessment of chemicals and cosmetic products: challenges and opportunities for introducing new approach methodologies. Arch. Toxicol. 95(6), 1867–1897. Prakash, P. and Gupta, N. 2005. Therapeutic uses of Ocimum sanctum Linn (Tulsi) with a note on eugenol and its pharmacological actions: a short review. Indian J. Physiol. Pharmacol. 49(2), 125–131. Raditya, M.N., Bagus, A.M.M., Kustiati, U., Wihadmadyatami, H. and Kusindarta, D.L. 2020. Data of the expression of Serotonin in Alzheimer’s Disease (AD) rat model under treatment of Ethanolic extract Ocimum sanctum Linn. DIB 30, 105654. Rasekh, H.R., Hosseinzadeh, L., Mehri, S., Kamli-Nejad, M., Aslani, M. and Tanbakoosazan, F. 2012. Safety assessment of Ocimum basilicum hydroalcoholic extract in Wistar rats: acute and subchronic toxicity studies. Iran. J. Basic Med. Sci. 15(5), 1030–1037. Ugbogu, O.C., Emmanuel, O., Agi, G.O., Ibe, C., Ekweogu, C.N., Ude, V.C., Uche, M.E., Nnanna, R.O. and Ugbogu, E.A. 2021. A review on the traditional uses, phytochemistry, and pharmacological activities of clove basil (Ocimum gratissimum L.). Heliyon 7(11), e08404. Ugwah-Oguejiofor, C.J., Okoli, C.O., Ugwah, M.O., Umaru, M.L., Ogbulie, C.S., Mshelia, H.E., Umar, M. and Njan, A.A. 2019. Acute and sub-acute toxicity of aqueous extract of aerial parts of Caralluma dalzielii N. E. Brown in mice and rats. Heliyon 5(1), 1179. Valsan, A., Bose, A., Kumar, A. and Saseendran, S. 2022. Preliminary phytochemical screening of indigenous medicinal plants Ocimum tenuiflorum, Ocimum basilicum, and Ocimum gratissimum. Res. Jr. Agril. Sci. 13(4), 925–930. Veeren, B., Ghaddar, B., Bringart, M., Khazaal, S., Gonthier, M.P., Meilhac, O., Diotel, N. and Bascands, J.L. 2020. Phenolic profile of herbal infusion and polyphenol-rich extract from leaves of the medicinal plant Antirhea borbonica: toxicity assay determination in Zebrafish Embryos and Larvae. Molecules 25(19), 4482. Vithalkar, A. and Patel, Y.K. 2022. Research Article Holy Basil : an Ayurvedic Gratutty. Int. J. Curr. Res. 14(12), 22986–22993. Xiong, Y., Chen, X., Li, F., Chen, Z. and Qin, Z. 2022. Zebrafish larvae acute toxicity test: a promising alternative to the fish acute toxicity test. Aquat. Toxicol. 246, 106143. Yang, R., Liu, H., Bai, C., Wang, Y., Zhang, X., Guo, R., Wu, S., Wang, J., Leung, E., Chang, H., Li, P., Liu, T. and Wang, Y. 2020. Chemical composition and pharmacological mechanism of Qingfei Paidu Decoction and Ma Xing Shi Gan Decoction against Coronavirus Disease 2019 (COVID-19): in silico and experimental study. Pharmacol. Res. 157, 104820. Yuniarti, W.M., Krismaharani, N., Ciptaningsih, P., Celia, K., Ma’Ruf, A. and Lukiswanto, B.S. 2021. The protective effect of Ocimum sanctum leaf extract against lead acetate-induced nephrotoxicity and hepatotoxicity in mice (Mus musculus). Vet. World. 14(1), 250–258. Žabka, M., Pavela, R., Kovaříková, K., Tříska, J., Vrchotová, N. and Bednář, J. 2021. Antifungal and Insecticidal Potential of the Essential Oil from Ocimum sanctum L. against Dangerous Fungal and Insect Species and Its Safety for Non-Target Useful Soil Species Eisenia fetida (Savigny, 1826). Plants 10(10), 2180. Zahran, E.M. and El-Khadragy, M.F. 2019. The antiinflammatory activity and LD50 of Ocimum forskolei Benth. J. Appl. Pharm. Sci. 9(8), 29–34. Zhang, W., Yan, Y., Wu, Y., Yang, H., Zhu, P., Yan, F., Zhao, R., Tian, P., Wang, T., Fan, Q. and Su, Z. 2022. Medicinal herbs for the treatment of anxiety: a systematic review and network meta-analysis. Pharmacol. Res. 179, 10620. | ||
| How to Cite this Article |
| Pubmed Style Mataram MBA, Kustiati U, Saragih GR, Aliffia D, Sanjaya WBT, Aviana AP, Salasia SIO, Ardiningtyas B, Nugrahaningsih DAA, Yusuf Y, Ana ID, Wihadmadyatami H, Kusindarta DL. Integrative use of murine and zebrafish models for acute toxicity observation of ethanolic extract of Ocimum sanctum Linn. doi:10.5455/OVJ.2026.v16.i3.33 Web Style Mataram MBA, Kustiati U, Saragih GR, Aliffia D, Sanjaya WBT, Aviana AP, Salasia SIO, Ardiningtyas B, Nugrahaningsih DAA, Yusuf Y, Ana ID, Wihadmadyatami H, Kusindarta DL. Integrative use of murine and zebrafish models for acute toxicity observation of ethanolic extract of Ocimum sanctum Linn. https://www.openveterinaryjournal.com/?mno=258189 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.33 AMA (American Medical Association) Style Mataram MBA, Kustiati U, Saragih GR, Aliffia D, Sanjaya WBT, Aviana AP, Salasia SIO, Ardiningtyas B, Nugrahaningsih DAA, Yusuf Y, Ana ID, Wihadmadyatami H, Kusindarta DL. Integrative use of murine and zebrafish models for acute toxicity observation of ethanolic extract of Ocimum sanctum Linn. doi:10.5455/OVJ.2026.v16.i3.33 Vancouver/ICMJE Style Mataram MBA, Kustiati U, Saragih GR, Aliffia D, Sanjaya WBT, Aviana AP, Salasia SIO, Ardiningtyas B, Nugrahaningsih DAA, Yusuf Y, Ana ID, Wihadmadyatami H, Kusindarta DL. Integrative use of murine and zebrafish models for acute toxicity observation of ethanolic extract of Ocimum sanctum Linn. doi:10.5455/OVJ.2026.v16.i3.33 Harvard Style Mataram, M. B. A., Kustiati, . U., Saragih, . G. R., Aliffia, . D., Sanjaya, . W. B. T., Aviana, . A. P., Salasia, . S. I. O., Ardiningtyas, . B., Nugrahaningsih, . D. A. A., Yusuf, . Y., Ana, . I. D., Wihadmadyatami, . H. & Kusindarta, . D. L. (2026) Integrative use of murine and zebrafish models for acute toxicity observation of ethanolic extract of Ocimum sanctum Linn. doi:10.5455/OVJ.2026.v16.i3.33 Turabian Style Mataram, Made Bagus Auriva, Ulayatul Kustiati, Golda Rani Saragih, Dinda Aliffia, Wilda Bunga Tina Sanjaya, Anisa Putri Aviana, Siti Isrina Oktavia Salasia, Bondan Ardiningtyas, Dwi Aris Agung Nugrahaningsih, Yusril Yusuf, Ika Dewi Ana, Hevi Wihadmadyatami, and Dwi Liliek Kusindarta. 2026. Integrative use of murine and zebrafish models for acute toxicity observation of ethanolic extract of Ocimum sanctum Linn. doi:10.5455/OVJ.2026.v16.i3.33 Chicago Style Mataram, Made Bagus Auriva, Ulayatul Kustiati, Golda Rani Saragih, Dinda Aliffia, Wilda Bunga Tina Sanjaya, Anisa Putri Aviana, Siti Isrina Oktavia Salasia, Bondan Ardiningtyas, Dwi Aris Agung Nugrahaningsih, Yusril Yusuf, Ika Dewi Ana, Hevi Wihadmadyatami, and Dwi Liliek Kusindarta. "Integrative use of murine and zebrafish models for acute toxicity observation of ethanolic extract of Ocimum sanctum Linn." doi:10.5455/OVJ.2026.v16.i3.33 MLA (The Modern Language Association) Style Mataram, Made Bagus Auriva, Ulayatul Kustiati, Golda Rani Saragih, Dinda Aliffia, Wilda Bunga Tina Sanjaya, Anisa Putri Aviana, Siti Isrina Oktavia Salasia, Bondan Ardiningtyas, Dwi Aris Agung Nugrahaningsih, Yusril Yusuf, Ika Dewi Ana, Hevi Wihadmadyatami, and Dwi Liliek Kusindarta. "Integrative use of murine and zebrafish models for acute toxicity observation of ethanolic extract of Ocimum sanctum Linn." doi:10.5455/OVJ.2026.v16.i3.33 APA (American Psychological Association) Style Mataram, M. B. A., Kustiati, . U., Saragih, . G. R., Aliffia, . D., Sanjaya, . W. B. T., Aviana, . A. P., Salasia, . S. I. O., Ardiningtyas, . B., Nugrahaningsih, . D. A. A., Yusuf, . Y., Ana, . I. D., Wihadmadyatami, . H. & Kusindarta, . D. L. (2026) Integrative use of murine and zebrafish models for acute toxicity observation of ethanolic extract of Ocimum sanctum Linn. doi:10.5455/OVJ.2026.v16.i3.33 |