| Research Article | ||
Open Vet. J.. 2026; 16(3): 1771-1780 Open Veterinary Journal, (2026), Vol. 16(3): 1771-1780 Research Article Evaluation of homemade biodesinfectant solution based on Lactobacillus (Lactobacillus casei, Lactobacillus paracasei) and baker’s yeast applied to broiler litterRoostita L. Balia1*, Daniswara Danindra Darmesti1, Lilis Suryaningsih2, Wendry Setiyadi Putranto2, Tuti Widjastuti2, Ratu Safitri3, Eka Wulandari1, Gemilang Lara Utama4 and Muhammad Farid Rizal61AVeterinary Study Program, Faculty of Medicine, Universitas Padjadjaran, Sumedang, Indonesia 2Faculty of Animal Husbandry, Universitas Padjadjaran, Sumedang, Indonesia 3Faculty of Mathematical and Natural Science, Universitas Padjadjaran, Sumedang, Indonesia 4Faculty of Agroindustrial Technology, Universitas Padjadjaran, Sumedang, Indonesia 5Doctorate Student of Primatology, Primatology program, School of postgraduate, Bogor Agricultural Institute, Bogor, Indonesia 6Urban Animal Pet Care Clinic, Bandung, Indonesia *Corresponding Author: Roostita L. Balia. Veterinary Study Program, Faculty of Medicine, Universitas Padjadjaran, Sumedang, Indonesia. Email:roostita [at] gmail.com Submitted: 25/07/2025 Revised: 02/02/2026 Accepted: 20/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractBackground: Poultry farming is a rapidly expanding sector that faces numerous challenges related to biosecurity and production efficiency. One major concern is the prevalence of pathogens, such as Escherichia coli, in broiler farm litter, which can lead to increased morbidity and mortality in flocks, ultimately affecting farm productivity. Aim: This study aimed to evaluate the efficacy of a farmer’s homemade biodisinfectant solution containing Lactobacillus casei, Lactobacillus paracasei, Saccharomyces cerevisiae, and Eco Enzyme applied to broiler farm litter for reducing Escherichia coli (E. coli) populations and their subsequent effects on broiler production performance. Methods: A biodesinfectant was applied to the litter of a commercial broiler farm over a period of 30 days. One-day-old Ross broilers (900 samples) were fed pre-starter, starter, and finisher diets as controls. The farm consisted of 3 broiler grow-out pens that were used for 3 treatment groups. As the maximum stocking density was restricted to 18 kg/m², the house space is 600 m². The biodisinfectant was applied at two concentrations in the treatment groups: TA (1 ml/100 ml) and TB (1 ml/250 ml) at 2-day intervals. Performance metrics, including FCR, slaughter body weight (BW), mortality rate, and overall health, were monitored. The control group (CON) had the lowest BW (p < 0.05) and the highest mortality rate (p < 0.05) compared with the other treatments. Results: Significant E. coli reduction at both treatments (p < 0.05) compared with the control, with no significant adverse effects on broiler performance metrics in every treatment group. Conclusion: These findings suggest that biodisinfectants offer a promising, eco-friendly alternative for enhancing both biosecurity and production performance efficiency in broiler poultry farms. Keywords: Biodesinfectant, Lactobacillus casei, Lactobacillus paracasei, Saccharomyces cerevisiae, Broiler farm. IntroductionBroiler meat production is one of Indonesia’s protein source commodities with a high potential for significant growth, with a substantial increase from 1.5 million tons in 2013 to 2.8 million tons by the end of 2024 (CIPS, 2024). Poultry farming is a rapidly expanding sector that faces numerous challenges related to biosecurity and production efficiency. One major concern is the prevalence of pathogens, such as Escherichia coli, in broiler farm litter, which can lead to increased morbidity and mortality in flocks, ultimately affecting farm productivity. Escherichia coli, a member of the Enterobacteriaceae family, is commonly found in the warm-blooded animals’ intestinal tract and their surrounding environments. Escherichia coli predominantly colonizes the lower gastrointestinal tract in poultry, serving as an indicator of environmental quality and functioning as an essential commensal microorganism. Although most E. coli strains are non-pathogenic, certain strains exhibit virulence, potentially causing economic losses in the poultry industry. Pathogenic E. coli strains in poultry, known as avian pathogenic E. coli (APEC) (FDA, 2012), cause colibacillosis, a systemic disease in broilers. The severity of APEC-induced disease is influenced by host health status, E. coli strain virulence, and predisposing factors such as stress. In the United States, subclinical colibacillosis affects approximately 30% of broiler flocks (FDA, 2013). The poultry house air can contain high concentrations of microorganisms, including E. coli (Tabler and Wells, 2017). E. coli originating from manure initially accumulates in poultry litter and subsequently becomes aerosolized due to bird activity. Various factors, including farm type, flock size, and transmission distance, influence the likelihood of airborne transmission, with pathogen survivability playing a crucial role in transmission modeling accuracy. Moreover, E. coli can persist on surfaces within poultry production environments for extended periods, facilitating its spread over large areas through environmental vectors. Although effective, chemical disinfectants pose environmental and health concerns, necessitating the exploration of eco-friendly alternative antimicrobial agents. Extended exposure to sublethal levels of chemical disinfectants can promote the selection of tolerant microorganisms, leading to a concurrent increase in the prevalence of AMR genes and antibiotic-resistant bacteria. Chemicals used for disinfection have been associated with the emergence and spread of AMR (Nordholt et al., 2021). Probiotics, such as Lactobacillus sp. and Saccharomyces cerevisiae, have gained attention for their potential to improve animal health through microbial competition and the inhibition of harmful pathogens. Saccharomyces cerevisiae promotes environmentally sustainable farming methods (Ballet et al., 2023), whereas Lactobacillus sp. are “generally recognized as safe” by the Food and Drug Administration in the United States and recognized to possess a “qualified presumption of safety” status by the European Food Safety Authority (ESFA). Additionally, their ability to combat bacteria against broad-spectrum pathogens and potential antibiotic susceptibility patterns demonstrated their gut colonization potential and competitiveness, suggesting that Lactobacillus strains are suitable for use in poultry diets (Khan et al., 2024). These probiotics may offer a promising eco-friendly biodisinfectant solution for poultry farms when combined with eco enzyme as a fermentative substrate. This study aimed to evaluate the effects of a homemade biodisinfectant based on probiotics and eco enzymes used by poultry farmers in reducing E. coli colonization in broiler litter and to assess the impact on broiler performance, including growth, FCR, and mortality rate. The biodisinfectant was tested at two concentrations: 1 ml/250 ml and 1 ml/100 ml to determine the optimal dose for achieving both biosecurity and production efficiency. Materials and MethodsBiodisinfectant preparationProbiotics, prebiotics, herbs, and plant extracts have been studied as alternatives to AGPs, with probiotics emerging as the most prevalent choice due to their demonstrated safety and efficacy. As a result, probiotics have become crucial to current chicken production methods, providing the combined advantages of increasing productivity and addressing the need for antibiotic-free, safer poultry meat products (Rashid et al., 2023). Probiotics, particularly lactic acid bacteria (LAB), have emerged as viable AGP substitutes due to their abilities to improve gastrointestinal health, immunological function, and overall poultry production performance. LAB includes various taxa, such as Lactobacillus, Lactococcus, Streptococcus, Carnobacterium, and Enterococcus. Lacticaseibacillus paracasei (L. paracasei), previously identified as L. paracasei, has been widely studied for its probiotic advantages, which include the capacity to colonize the gastrointestinal tract and durability under acidic environments. Furthermore, L. paracasei has proven capacities in modifying immunological responses, improving digestion, and inhibiting inflammatory processes. L. paracasei supplementation considerably improved the growth performance, antioxidant capacity, and lipid metabolism of E. coli-infected broilers. In addition, the probiotic treatment resulted in a significantly lower FCR, increased BW, and improved feed efficiency (Lokapirnasari et al., 2025). Lactobacillus casei can maintain microbial balance, enhancing the digestive and absorptive functions of broilers and modulating the immune response of broilers (Zhang et al., 2021). A previous study demonstrated the robustness of BWG broilers despite high E. coli counts due to the potential immunomodulatory effects of L. casei. Lactobacillus strains, which are known to improve the function of the intestinal barrier and promote anti-inflammatory cytokines, may lessen the impact of infections on growth performance (Vasquez et al., 2022). This immune modulation may result in a more robust physiological state that promotes better growth even in the face of pathogenic stress (Jha et al., 2020). The preparation process of the homemade biodisinfectant is shown in Figure 1.
Fig. 1. Preparation of biodesinfectan. Organic waste products formed by fermenting waste products, such as vegetables and fruit peels, can produce lactic acid, which is considered to act as an alternative acidifier and aid digestion (Palupi et al., 2024). The preparation of homemade biodisinfectants begins with the collection of fruit and vegetable waste from household sources, which is then cut into small pieces. A clean, non-transparent 15-l container is prepared by washing it with soap, thoroughly rinsing to remove any residue, and filling it with clean water up to 60% of its volume. Chopped fruit and vegetable waste is then added at a ratio of 30% of the water’s weight. Molasses, comprising 10% of the water’s weight, is incorporated into the mixture and stirred thoroughly. A microbial inoculum comprising 44 g of baker’s yeast (Saccharomyces cerevisiae) and Lactobacillus species (L. casei and L. paracasei) starter was then introduced. The opening and lid of the container are sanitized before being tightly sealed. During the first 2 weeks, the lid is briefly opened to release the fermentation gases. After this period, the container is permanently sealed, allowing fermentation to proceed. The biodisinfectant is ready for harvest after completion of the fermentation process is completed. The mixture undergoes an additional 72-hour fermentation before application to broiler litter. Experimental designThis study was conducted on a commercial broiler farm in Ciamis, West Java, Indonesia, from July to September 2024, using a completely randomized design with three treatment groups. The farm consisted of three identical broiler grow-out pens, with each pen representing one experimental unit. A total of 900 1-day-old Ross 308 chicks were randomly allocated to three pens (300 birds per pen). The treatment groups were as follows:
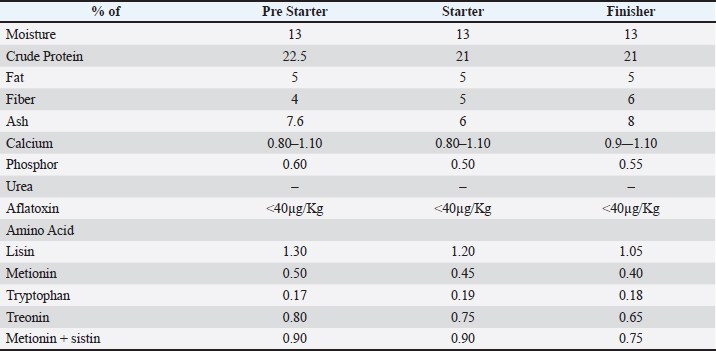
All pens were managed under identical environmental conditions, including housing design, ventilation, lighting program, stocking density (maximum 18 kg/m²), feed formulation, and water access. Rice hulls were used as bedding material in all pens. The pen was used as the experimental unit for performance parameters (body weight [BW], feed intake, FCR, and mortality). For microbiological analysis, each litter swab sample was considered an observational unit, whereas the pen remained the treatment unit. To minimize management bias, the personnel responsible for daily management and data collection followed the same standardized procedures across all pens. With standard commercial feed and water provided. Rice hulls were used as litter bedding material. Animals and their managementA total of 900 day-old Ross 308 chicks (Silga Perkasa, Sukabumi, Indonesia) were used in this study. All day-old chicks obtained from the same parent stock with >95% uniformity were housed simultaneously for each group and cycle within the trial and raised for 32 d. Commercial feed (Table 1) and water were provided ad libitum. The protein levels of feed ranged between 22.0% and 20.5% CP until slaughter. All other management aspects were performed in accordance with the Ross broiler pocket guide (Aviagen, 2020). Table 1. Feed composition percentage.
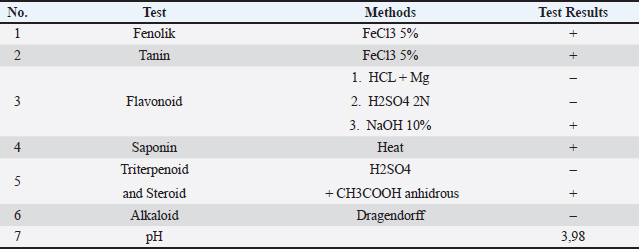
Litter treatmentRice hulls were used as the bedding material in all groups. The acceptable depth of the bedding materials should be averaged at 2-5 cm to ensure proper ammonia absorption, effective moisture absorption, and support overall broiler health and management (Haque and Chowdhury, 1994). The bedding material was dispersed once before housing the birds, whereby 5 cm depth bedding material per m² was dispersed using a spreader. Litter is turned over every day and sprayed with biodisinfectant starting at the age of 7 days. Application of the biodisinfectantThe bioproduct can be used inside farms during the growth cycle without putting broilers at health risk or requiring significant infrastructure changes (Burguet-Fernández et al., 2018). The biodisinfectant was applied directly to the litter in each treatment pen every 2 days from the age of 9 years over a 32-day grow-out period. The litter was evenly sprayed with the disinfectant solution to ensure uniform coverage. Secondary metabolite testSecondary metabolites are chemical compounds produced by organisms during secondary metabolism. Unlike primary metabolites, secondary metabolites are not directly necessary for basic processes (Kolackova et al., 2023). The secondary metabolites to be tested in this study are phenolics using the FeCl3 5% method, tannins using the FeCL3 method, and flavonoids using the following methods: (1) HCl + Mg, (2) H2S042N, and (3) 10% NaOH. Saponins were tested using the heat method, triterpenoids and steroids using the H2SO4+ anhydrous CH3COOH method, alkaloids using the Drafendorff method, and pH test. Data collectionMicrobial analysisLitter samples were collected from each group after 23 days of application from the age of 9 days at the end of the study (32 days of age) to assess E. coli levels. To ensure adequate statistical power, 25 l swab samples were collected from three different areas of each broiler house: around water lines, feeder lines, and the free area (modified from Tasistro et al., 2004). Samples were sent to the Laboratory of Animal Health and Veterinary Public Health of West Java, Indonesia. Samples were cultured in EMBA with standard microbial culture techniques according to SNI 19-2897: 1993, and E. coli counts were expressed as CFU per gram of litter. Broiler performanceThe following production performance metrics were monitored throughout the experiment:
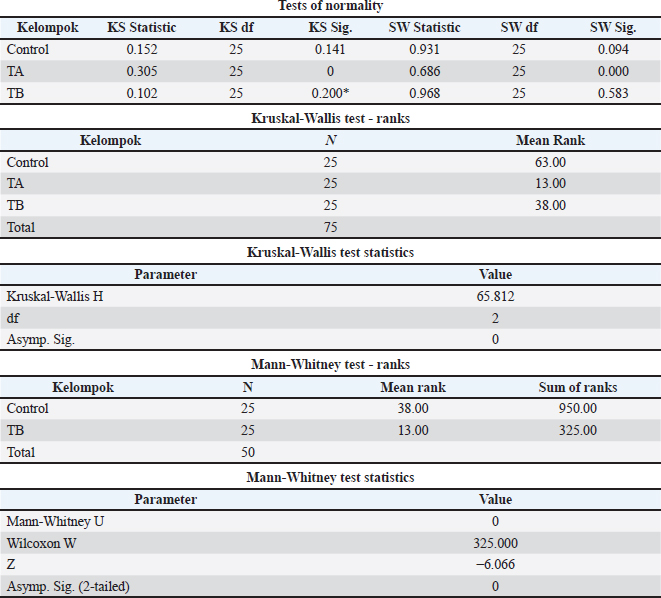
Statistical analysisAll statistical analyses were performed using IBM SPSS Statistics version 25 for Windows. The Shapiro–Wilk test was used to assess data normality. Because most variables were not normally distributed, non-parametric analysis was performed. Differences in litter E. coli counts and final average BW among treatment groups (CON, TA, and TB) were analyzed using the Kruskal–Wallis test at a significance level of p < 0.05. When a significant overall effect was detected, pairwise comparisons between groups were performed using Dunn’s post hoc test with Bonferroni adjustment to control for multiple comparisons. Final BW data are presented as mean ± SD. FCR and mortality rate were calculated at the pen level using cumulative feed intake and the total number of dead birds during the entire production cycle. FCR and mortality rate are presented as mean SD. Because each treatment was applied to a single pen, within-group replication was not available; therefore, measures of variance (SD) could not be estimated for these parameters; accordingly, FCR and mortality data are presented descriptively. The pen was used as the experimental unit for performance parameters, whereas individual litter samples were considered as observational units for microbiological analysis. Ethical approvalThe study was approved by the Research Ethics Committee Universitas Padjadjaran Bandung (Approval No. 596/UN6.KEP/EC/X/2022). ResultsThe physicochemical characterization of the biodisinfectant demonstrated an acidic profile, with a measured pH of 3.98, and the presence of several bioactive secondary metabolites, including phenolics, tannins, saponins, and steroid compounds (Table 2). Qualitative screening indicated positive reactions for phenolics, tannins, saponins, and steroids, whereas flavonoids were partially detected, and alkaloids were not observed. These findings confirm that the fermented formulation contains multiple compounds with potential antimicrobial activity. Table 2. pH measurement and identification of secondary metabolites of bio-disinfectants.
Application of the biodisinfectant to broiler litter resulted in a significant reduction of E. coli populations compared with the untreated control group (p < 0.05). Both treatment concentrations (1:100 and 1:250) showed comparable efficacy in suppressing bacterial counts, indicating that even the lower concentration was sufficient to achieve microbial control under field conditions. Statistical analysis using the Kruskal–Wallis test confirmed a significant overall treatment effect on E. coli concentration (Table 3). Table 3. Quantification of E. coli in Broiler Litter Samples from Control and Treated Groups (CFU/g) Over 32 Days (cfu/gr).
Broiler production performance was evaluated based on final BW, feed conversion ratio (FCR), and mortality rate. Birds from both treatment groups exhibited slightly higher final BW compared with the control group, and the difference was statistically significant (p < 0.05). However, the magnitude of BW improvement was relatively small and may not translate into substantial economic benefit. Feed efficiency, expressed as FCR, showed a numerically lower value in the treatment groups compared with the control, suggesting a trend toward improved feed utilization, although this parameter was analyzed descriptively due to the absence of pen-level replication (Table 4). Table 4. Broiler performance parameter.
Mortality rate was lower in both biodisinfectant-treated groups compared with the control group (Table 5). This reduction indicates a potential improvement in overall flock health associated with the application of the probiotic-based biodisinfectant. Non-parametric analysis further confirmed significant differences among treatments for mortality rate and BW parameters. Table 5. Results of the non-parametric Kruskal-Wallis test.
Overall, the results demonstrate that the probiotic-based biodisinfectant effectively reduced E. coli contamination in broiler litter without adversely affecting growth performance, while showing potential benefits in mortality reduction and feed efficiency. DiscussionTable 3 shows a significant decrease in E. coli colonization in the treatment group compared to the control group (p < 0.05). This result indicates that the probiotic-based biodisinfectant is effective in eliminating E. coli from litter. Broiler chicken litter is a major reservoir of microorganisms and pathogens (Carpenter, 1986). Approximately 70% to 80% of litter mass is the dry matter content, which can contain abundant organisms and compounds that can affect the quality of the poultry housing environment (Schulz et al., 2016). Broiler litter is often heavily contaminated with E. coli due to the nature of chicken excrement, which can readily colonize and multiply within the litter material. Acidic pH and secondary metabolites of biodisinfectant also contribute to E. coli reduction, pH measurement, and identification of biodisinfectant secondary metabolites (Table 3). The toxicity of saponins against bacteria generally occurs by damaging the bacterial cell membrane proteins and reducing their permeability. Damage to the membrane surface results in bacterial lysis and death (Mahyuni and Trirakhma, 2018). Saponins with surface-active properties may insert into the lipid bilayer, bind to cholesterol, form domains enriched with cholesterol-saponin complexes, and eventually lyse cells. Saponin extracts increase the number of early apoptotic cells in a dose-dependent manner (Arabski et al., 2012). Flavonoids are among the most abundant naturally occurring bioactive compounds produced by plants. These secondary metabolites have a variety of activities against various cells and systems. Undoubtedly, one of the most intriguing is the antimicrobial, which is activated via various molecular mechanisms. In fact, flavonoids are effective not only in directly damaging the envelopes of gram-negative and gram-positive bacteria but also in acting on specific molecular targets required for the survival of these microorganisms (Donadio et al., 2021). The flavonoid content inhibits the growth of fungi by disrupting the permeability of cell membranes, causing a change in the components of nutrient transport and causing toxic effects against fungi (Komala et al., 2020). Tannin targets cell wall polypeptides, which cause damage to the cell wall and inhibit nucleic acid production (Rahayu et al., 2021). The mechanism of action of phenol is the denaturation of bacterial cell proteins, which disturbs the metabolism of the bacterial cell (Ramadani et al., 2022). Based on the results (Table 4), information was obtained about BW performance in the three groups. The average BW in the control group reached 1970.6 g with a standard deviation of 3.021, and the minimum and maximum values were 1965 g and 1975 g, respectively. In the TA 1:100 group, the average BW was recorded at 1973.4 g with a standard deviation of 2.834 g, with the lowest and highest scores of 1966 g and 1977 g, respectively. Meanwhile, the TB group of 1:250 showed an average BW of 1974.71 g with a standard deviation of 3.962, as well as minimum and maximum values ranging between 1967 and 1980 g. Overall, the results of this study suggested a possible cumulative effect on BW for TA and TB. The effects of these treatments were significantly greater than CON (p < 0.05) by the end of the growth-out period (32 days). The result of BW measurements showed that there is significance in statistical tests, but the BW differences between the CON and treatment groups showed no economic benefit. Table 4 shows a significant difference in the mortality rate between both treatment groups compared with the control group (p < 0.05). This result shows a possible correlation between the reduction of E. coli colonization in litter and overall poultry health. E. coli is a widespread commensal microorganism present in human and animal intestinal flora and can easily be excreted into the environment. Poultry litter, which contains a combination of feces, feed, water, and bedding materials that accumulate throughout the farming process, may be a potential source of infectious agents. E. coli is commonly used as an indicator to assess the presence of infectious agents (McCrea et al., 2008). Lower ammonia level from weekly monitoring in TA and TB indicates a correlation with lower mortality rate. Elevated ammonia levels can lead to upper respiratory tract deciliation (Oyetunde et al., 1978). In broiler housing, inhalable dust concentrations have been recorded at 8.29 mg/m³, while respirable dust levels reach 1.419 mg/m³ (Ellen et al., 1999). Dust sources include not only litter but also feed, down feathers, excrement, microorganisms, and mold (Aarnink et al., 1999). Additionally, E. coli can be detected in the trachea, with its relative abundance declining as broilers gain BW over time (Johnson et al., 2018). Acidic pH from biodisinfectant (3,98), in accordance with a laboratory study to simulate open-lot feedyards, Shi et al. (2001) showed that lower pH adjustment for controlling NH3 emissions with amendments of aluminum sulfate resulted in decreased pH and NH3 volatilization reduction (98%). The poultry excreta generally build up litter with an alkaline pH of approximately 8.0 to 9.0, and NH3 is created through the breakdown of UA from manure in poultry litter. Irritation from ammonia exposure levels on the respiratory epithelium, such as inflammation caused by acute lung injury, can alter the local immune environment and increase susceptibility to respiratory diseases in broilers and disrupt the normal respiratory microbiota, promoting the proliferation of opportunistic pathogens such as E. coli (Poroyko et al., 2015). This result showed no adverse effect of probiotics-based biodisinfectant applications on broiler litter to broiler performance and can improve broiler health status based on mortality rate parameter. Limitation of the studyThe trial was conducted within a single grow-out cycle under specific environmental conditions. Variations in seasonal climate, humidity, and different types of initial litter materials may influence the performance of the probiotic-based solution. The microbiological assessment of the litter was conducted shortly after the final application. While this captures the immediate efficacy of the bio-disinfectant, it does not provide data on the long-term residual effects or the microbial rebound rate over a longer duration between applications. The phytochemical screening of the homemade bio-disinfectant was primarily qualitative. Quantitative analysis of specific metabolites (such as precise concentrations of organic acids or bacteriocins) would provide a deeper understanding of the exact antimicrobial mechanisms at play. ConclusionThis study shows that a probiotic-based biodisinfectant effectively reduces E. coli in broiler litter, as indicated by a significant decrease in bacterial counts (p < 0.05). The biodisinfectant acidic pH and bioactive compounds, such as saponins, flavonoids, tannins, and phenolics, disrupt bacterial cell structures and contribute to its antibacterial action. Treatment had a positive impact on health performance. The mortality rates were significantly lower in the treatment groups than in the control group. This suggests a link between the reduction of E. coli levels in litter and the improvement of poultry health status. Overall, probiotic-based biodisinfectants offer a promising, eco-friendly alternative to chemical disinfectants for improving poultry farming hygiene and productivity. AcknowledgmentsThe authors express their deepest gratitude and appreciation for the assistance and support received throughout the completion of this research paper. The authors want to take this opportunity to acknowledge the contributions of Agus Hari Wardana, Poultry Production Manager. FundingThis study received financial and facility support from Academic Leadership Grand Universitas Padjadjaran. Authors' contributionsRoostita L Balia was responsible for the overall research design and execution. Daniswara Danindra Darmesti was responsible for data collection and analysis. The manuscript was written and all necessary revisions were made by all authors. Roostita L. Balia and Lilis Suryaningsih conceptualized the study and contributed to the experimental design. Ratu Safitri, Eka Wulandari, and Gemilang Lara Utama helped with data analysis and interpretation. Tuti Widjiastuti and Wendry Setiyadi Putranto are responsible as supervisors. Muhammad Farid Rizal contributed to the literature review, interpretation of results, and final manuscript editing. All authors have read and approved the final version of the manuscript before publication in the journal. Conflict of interestThe authors have not declared any conflict of interest. Data availabilityAll data are available in the article ReferencesAarnink, A.J.A., Roelofs, P.F.M.M., Ellen, H. and Gunnink, H. 1999. Dust sources in animal houses. In Proceedings of the International Symposium on Dust Control in Animal Production Facilities (pp. 34–40). Scandinavian Congress Center, Aarhus, Denmark. Arabski, M., Węgierek-Ciuk, A., Czerwonka, G., Lankoff, A. and Kaca, W. 2012. Effects of saponins against clinical E. coli strains and eukaryotic cell line. J. Biomed. Biotechnol. 2012, 286216; doi:10.1155/2012/286216 Aviagen. 2020. Ross broiler pocket guide: The pocket guide. Aviagen Co. Ballet, N., Renaud, S., Roume, H., George, F., Vandekerckove, P., Boyer, M. and Durand-Dubief, M. 2023. Saccharomyces cerevisiae: multifaceted applications in One Health and the achievement of Sustainable Development Goals. Encyclopedia 3, 602–613; doi:10.3390/encyclopedia3020043 Burguet-Fernández, G., Paredes-Sánchez, J.P. and Xiberta-Bernat, J. 2018. Bioenergy valuation of poultry litter by applying an enzyme product for environmental purposes: a new applied technology. Proceedings 2(23), 1480; doi:10.3390/proceedings2231480 Carpenter, G.A. 1986. Dust in livestock buildings—Review of some aspects. J. Agricult. Eng. Res. 33(3), 227–241; doi:10.1016/S0021-8634(86)80038-5 Center for Indonesian Policy Studies (CIPS). 2024. Improving Indonesia’s Poultry Competitiveness: Exploring Broiler Chicken Meat Trade Opportunities. doi: 10.35497/585489 Donadio, G., Mensitieri, F., Santoro, V., Parisi, V., Bellone, M.L., De Tommasi, N., Izzo, V. and Dal Piaz, F. 2021. Interactions with Microbial Proteins Driving the Antibacterial Activity of Flavonoids. Pharmaceutics 13(5), 660; doi:10.3390/pharmaceutics13050660 Ellen, H.H., Bottcher, R.W., von Wachenfelt, E. and Takai, H. 2000. Dust levels and control methods in poultry houses. J. Agric. Saf. Health 6(4), 275–282; doi:10.13031/2013.1910 Food and Drug Administration (FDA). 2012. Guidance for industry #209: The judicious use of medically important antimicrobial drugs in food-producing animals. Paris: FDA. Food and Drug Administration (FDA). 2013. Guidance for industry #213: New animal drugs and new animal drug combination products administered in or on medicated feed or drinking water of food-producing animals. Paris: FDA. Haque, M.I. and Chowdhury, S.D. 1994. Use of rice husk litter at different depths for broiler chicks during summer. Br. Poultry. Sci. 35(5), 809–812; doi:10.1080/00071669408417745 Jha, R., Das, R., Oak, S. and Mishra, P. 2020. Probiotics (direct-fed microbials) in poultry nutrition and their effects on nutrient utilization, growth and laying performance, and gut health: a systematic review. Animals 10(10), 1863; doi:10.3390/ani10101863 Johnson, T.J., Youmans, B.P., Noll, S., Cardona, C., Evans, N.P., Karnezos, T.P., Ngunjiri, J.M., Abundo, M.C. and Lee, C.W. 2018. A consistent and predictable commercial broiler chicken bacterial microbiota in antibiotic-free production displays strong correlations with performance. Appl. Environ. Microbiol. 84(12), e00362-18; doi:10.1128/AEM.00362-18 Khan, U., Rahman, S.M., Khan, S., Roy, S. and Hossain, K.M. 2024. Effects of probiotics on productive performances and serum lipid profile of broiler as substitute of antibiotics. Sci. Prog. 107(3), 107; doi:10.1177/00368504241276259 Kolackova, M., Janova, A., Dobesova, M., Zvalova, M., Chaloupsky, P., Krystofova, O., Adam, V. and Huska, D. 2023. Role of secondary metabolites in distressed microalgae. Environ. Res. 224, 115392; doi:10.1016/j.envres.2023.115392 Komala, O. and Siwi, F.R. 2020. Aktivitas antijamur ekstrak etanol 50% dan etanol 96% daun pacar kuku Lawsonia inermis L terhadap Trichophyton mentagrophytes. Ekologia 19(1), 12–19; doi:10.33751/ekol.v19i1.1657 Lokapirnasari, W.P., Maslachah, L., Saputro, A.L., Rokana, E., Yulianto, A.B., Rosyada, Z.N.A., Firdaus, M.A., Silfia, H.I., Lisnanti, E.F., Baihaqi, Z.A., Marbun, T.D., Khairullah, A.R. and Shakeel, M. 2025. Potency of Lacticaseibacillus paracasei as an alternative to antibiotic growth promoter in broiler chicken challenged with avian pathogenic Escherichia coli. Vet. World 18(5), 1180–1189; doi:10.14202/vetworld.2025.1180-1189 Mahyuni, S. and Trirakhma, S. 2018. Kadar saponin dan aktivitas antibakteri ekstrak daun Fillicium decipens (Wight & Arn.) Thwaites terhadap Staphylococcus aureus, Escherichia coli, dan Candida albicans. Jurnal Ilmiah Farmasi 8(2), 1–9. Mccrea, B.A., Macklin, K.S., Norton, R.A., Hess, J.B. and Bilgili, S.F. 2008. Recovery and genetic diversity of Escherichia coli isolates from deep litter, shallow litter, and surgical shoe covers. J. Appl. Poultry. Res. 17(2), 237–242; doi:10.3382/japr.2007-00057 National Water Research Institute. 2016. Final report of an NWRI independent advisory panel: Recommended DPR general guidelines and operational requirements for New Mexico. https://www.nwri-usa.org Nordholt, N., Kanaris, O., Schmidt, S.B.I. and Schreiber, F. 2021. Persistence against benzalkonium chloride promotes rapid evolution of tolerance during periodic disinfection. Nature Commun. 12(1), 6792; doi:10.1038/s41467-021-27107-0 Oyetunde, O.O.F., Thomson, R.G. and Carlson, H.C. 1978. Aerosol exposure of ammonia, dust and Escherichia coli in broiler chickens. Can. Vet. J. 19(7), 187–193. Palupi, R., Nurdin, A.S., Lubis, F.N.L., Sahara, E., Zebua, E.S. and Panjaitan, I. 2024. The effectiveness of adding eco-enzymes in drinking water on production performance and carcass of Lohmann roosters. J. Xi’. Shiyou Univ. Natural Sci. Ed. 20(2), 216–230. Poroyko, V., Meng, F., Meliton, A., Afonyushkin, T., Ulanov, A., Semenyuk, E., Latif, O., Tesic, V., Birukova, A.A. and Birukov, K.G. 2015. Alterations of lung microbiota in a mouse model of LPS-induced lung injury. Am. J. Physiol-Lung. Cellular Mol. Physiol. 309(1), L76–L83; doi:10.1152/ajplung.00061.2014 Rahayu, M.R., Muliarta, I.N. and Situmeang, Y.P. 2021. Acceleration of production natural disinfectants from the combination of eco-enzyme domestic organic waste and frangipani flowers (Plumeria alba). SEAS 5(1), 15–21. Ramadani, A.H., Karima, R. and Ningrum, R.S. 2022. Antibacterial activity of pineapple peel (Ananas comosus) eco-enzyme against acne bacteria (Staphylococcus aureus and Propionibacterium acnes). Indonesian J. Chem. Res. 9(3), 201–207. Rashid, S., Alsayeqh, A.F., Akhtar, T., Abbas, R.Z. and Ashraf, R. 2023. Probiotics: alternative to antibiotics in poultry production. J. Appl. Anim. Res. 51(1), 123–134. Schulz, J., Ruddat, I., Hartung, J., Hamscher, G., Kemper, N. and Ewers, C. 2016. Antimicrobial-resistant Escherichia coli survived in dust samples for more than 20 years. Front. Microbiol. 7, 866; doi:10.3389/fmicb.2016.00866 Shi, Y., Parker, D.B., Cole, N.A., Auvermann, B.W. and Mehlhorn, J.E. 2001. Surface amendments to minimize ammonia emissions from beef cattle feedlots. Trans. ASAE 44(3), 677–682. Tabler, T. and Wells, J. 2017. Economic impact of Mississippi’s poultry industry (pp. 1–8). Mississippi State University Extension. Tasistro, A.S., Kissel, D.E. and Bush, P.B. 2004. Sampling broiler litter: how many samples are needed?. J. Appl. Poultry. Res. 13(2), 163–170; doi:10.1093/japr/13.2.163 Vasquez, R., Oh, J.K., Song, J.H. and Kang, D.K. 2022. Gut microbiome-produced metabolites in pigs: a review on their biological functions and the influence of probiotics. J. Anim. Sci. Technol. 64(4), 671–695; doi:10.5187/jast.2022.e58 Y. Shi., D. B. Parker., N. A. Cole., B. W. Auvermann. and J. E. Mehlhorn. 2001. Surface amendments to minimize ammonia emissions from beef cattle feedlots. Trans. ASAE. 44(3), 677–682; doi:10.13031/2013.6117 Zhang, L., Zhang, R., Jia, H., Zhu, Z., Li, H. and Ma, Y. 2021. Supplementation of probiotics in water beneficial growth performance, carcass traits, immune function, and antioxidant capacity in broiler chickens. Open. Life. Sci. 16(1), 311–322; doi:10.1515/biol-2021-0031 | ||
| How to Cite this Article |
| Pubmed Style Balia RL, Darmesti DD, Suryaningsih L, Putranto WS, Widjastuti T, Safitri R, Wulandari E, Utama GL, Rizal MF. Evaluation of homemade biodesinfectant solution based on Lactobacillus (Lactobacillus casei, Lactobacillus paracasei) and baker’s yeast applied to broiler litter. doi:10.5455/OVJ.2026.v16.i3.34 Web Style Balia RL, Darmesti DD, Suryaningsih L, Putranto WS, Widjastuti T, Safitri R, Wulandari E, Utama GL, Rizal MF. Evaluation of homemade biodesinfectant solution based on Lactobacillus (Lactobacillus casei, Lactobacillus paracasei) and baker’s yeast applied to broiler litter. https://www.openveterinaryjournal.com/?mno=273127 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.34 AMA (American Medical Association) Style Balia RL, Darmesti DD, Suryaningsih L, Putranto WS, Widjastuti T, Safitri R, Wulandari E, Utama GL, Rizal MF. Evaluation of homemade biodesinfectant solution based on Lactobacillus (Lactobacillus casei, Lactobacillus paracasei) and baker’s yeast applied to broiler litter. doi:10.5455/OVJ.2026.v16.i3.34 Vancouver/ICMJE Style Balia RL, Darmesti DD, Suryaningsih L, Putranto WS, Widjastuti T, Safitri R, Wulandari E, Utama GL, Rizal MF. Evaluation of homemade biodesinfectant solution based on Lactobacillus (Lactobacillus casei, Lactobacillus paracasei) and baker’s yeast applied to broiler litter. doi:10.5455/OVJ.2026.v16.i3.34 Harvard Style Balia, R. L., Darmesti, . D. D., Suryaningsih, . L., Putranto, . W. S., Widjastuti, . T., Safitri, . R., Wulandari, . E., Utama, . G. L. & Rizal, . M. F. (2026) Evaluation of homemade biodesinfectant solution based on Lactobacillus (Lactobacillus casei, Lactobacillus paracasei) and baker’s yeast applied to broiler litter. doi:10.5455/OVJ.2026.v16.i3.34 Turabian Style Balia, Roostita L., Daniswara Danindra Darmesti, Lilis Suryaningsih, Wendry Setiyadi Putranto, Tuti Widjastuti, Ratu Safitri, Eka Wulandari, Gemilang Lara Utama, and Muhammad Farid Rizal. 2026. Evaluation of homemade biodesinfectant solution based on Lactobacillus (Lactobacillus casei, Lactobacillus paracasei) and baker’s yeast applied to broiler litter. doi:10.5455/OVJ.2026.v16.i3.34 Chicago Style Balia, Roostita L., Daniswara Danindra Darmesti, Lilis Suryaningsih, Wendry Setiyadi Putranto, Tuti Widjastuti, Ratu Safitri, Eka Wulandari, Gemilang Lara Utama, and Muhammad Farid Rizal. "Evaluation of homemade biodesinfectant solution based on Lactobacillus (Lactobacillus casei, Lactobacillus paracasei) and baker’s yeast applied to broiler litter." doi:10.5455/OVJ.2026.v16.i3.34 MLA (The Modern Language Association) Style Balia, Roostita L., Daniswara Danindra Darmesti, Lilis Suryaningsih, Wendry Setiyadi Putranto, Tuti Widjastuti, Ratu Safitri, Eka Wulandari, Gemilang Lara Utama, and Muhammad Farid Rizal. "Evaluation of homemade biodesinfectant solution based on Lactobacillus (Lactobacillus casei, Lactobacillus paracasei) and baker’s yeast applied to broiler litter." doi:10.5455/OVJ.2026.v16.i3.34 APA (American Psychological Association) Style Balia, R. L., Darmesti, . D. D., Suryaningsih, . L., Putranto, . W. S., Widjastuti, . T., Safitri, . R., Wulandari, . E., Utama, . G. L. & Rizal, . M. F. (2026) Evaluation of homemade biodesinfectant solution based on Lactobacillus (Lactobacillus casei, Lactobacillus paracasei) and baker’s yeast applied to broiler litter. doi:10.5455/OVJ.2026.v16.i3.34 |