| Research Article | ||
Open Vet. J.. 2026; 16(2): 1048-1059
Open Veterinary Journal, (2026), Vol. 16(2): 1048-1059 Research Article Optimization of the best extender for liquid semen of Muscovy duck (Cairina moschata) with the addition of different concentrations of vitamin C and glutathioneBetri Wahyu Agustin, Tinda Afriani*, Tertia Delia Nova and Ananda AnandaDepartment of Animal Production Technology, Faculty of Animal Science, Universitas Andalas, Padang, Indonesia *Corresponding Author: Tinda Afriani. Department of Animal Production Technology, Faculty of Animal Science, Universitas Andalas, Padang, Indonesia. Email: tindaafriani [at] ansci.unand.ac.id Submitted: 08/08/2025 Revised: 10/01/2026 Accepted: 26/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
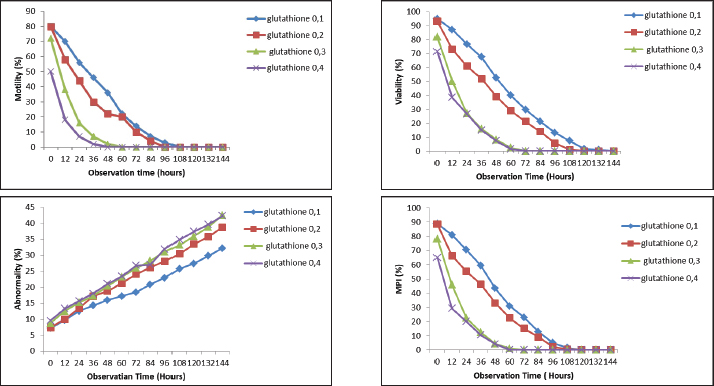
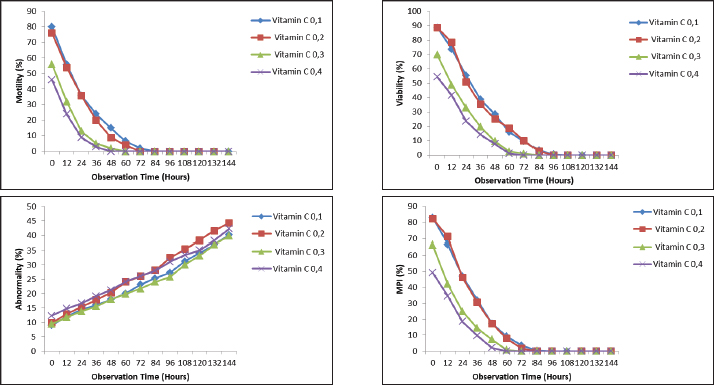
AbstractBackground: Semen quality during storage is a critical determinant of the success of artificial insemination in poultry. Muscovy duck (Cairina moschata), an important indigenous genetic resource in Indonesia, requires effective semen preservation methods to support breeding programs. Aim: This study investigated the effects of different glutathione (GSH) and vitamin C concentrations on the quality and longevity of Muscovy duck semen during cold storage. The novelty of this research lies in its focus on Muscovy ducks, the evaluation of multiple antioxidant concentrations, and detailed observation intervals to optimize semen preservation. Methods: Fresh semen was collected from adult Muscovy ducks and diluted with Ringer’s lactate extender supplemented with GSH (0.1, 0.2, 0.3, and 0.4 mM) or vitamin C (0.568, 1.136, 1.704, and 2.272 mM). The diluted semen was stored at 5°C and evaluated every 12 hours for motility, viability, abnormalities, and membrane plasma integrity. Longevity was defined as the duration during which motility and viability remained >50%. Results: Both antioxidants significantly influenced all semen quality parameters during storage (p < 0.01). Treatment with 0.1 mM GSH and 0.568 mM vitamin C consistently preserved higher motility, viability, and membrane integrity while minimizing abnormalities. These concentrations also yielded the highest semen longevity. Conclusion: Appropriate GSH and vitamin C concentrations effectively maintained the quality and longevity of Muscovy duck semen during cold storage. These antioxidants show potential for improving AI outcomes in poultry. Keywords: Cold storage, GSH, Muscovy duck, Semen extender, Vitamin C. IntroductionIndonesia possesses vast potential for livestock development due to its rich diversity of genetic resources for domesticated animals. One of these valuable indigenous genetic resources is the Muscovy duck (Cairina moschata), a poultry species that plays an important role in supplying local communities with animal protein. Adult Muscovy ducks have body weights ranging from 3 to 6 kg, making them an efficient source of meat (Djaya, 2003). Despite its potential, Muscovy duck farming in Indonesia is still largely managed through traditional methods. Males and females are typically housed together without selective breeding, which increases the risk of inbreeding. Inbreeding can compromise genetic quality, reducing reproductive efficiency and productivity. Modern reproductive technologies, such as artificial insemination (AI), have been recommended to overcome these limitations. AI enables the widespread use of genetically superior males and facilitates genetic improvement within a relatively short time (Kusumawati and Leondro, 2014). However, one of the persistent challenges in the implementation of AI in avian species is the decline in semen quality during storage. Cryopreservation and cold storage are commonly used to extend semen usability; however, these processes often cause structural damage. According to Toelihere (1985), freezing may result in intracellular ice crystal formation, which disrupts membrane integrity and leads to sperm cell death during thawing. Additionally, cold shock and the accumulation of reactive oxygen species (ROS) during storage further damage sperm cells, impairing motility and viability (Wiyanti et al., 2013). Various extenders have been developed to minimize sperm damage during storage. In a preliminary comparative study, Ringer’s lactate (RL) was found to be the most effective extender for the treatment of Muscovy duck semen. RL helps maintain electrolyte balance, osmotic pressure, and pH, all of which are critical for preserving sperm cell structure and function. Hence, RL was selected as the base extender in this study. Antioxidant additives in semen extenders have been proposed as a strategy to improve sperm quality during storage. Glutathione (GSH), a tripeptide and endogenous antioxidant, plays a key role in neutralizing ROS and protecting cellular structures from oxidative stress. GSH supplementation in poultry semen can enhance motility, viability, and DNA integrity (Zhandi et al., 2020; Zuha et al., 2024). Similarly, vitamin C (ascorbic acid) is a well-established antioxidant that reduces lipid peroxidation and protects sperm cells from oxidative injury (Fouda et al., 2021). Vitamin C supplementation in turkey semen extenders has been shown to lower malondialdehyde levels, an indicator of oxidative damage (Echekwu et al., 2021). Given the susceptibility of avian spermatozoa to oxidative stress, optimizing the composition of semen extenders is essential for preserving sperm structure and function during storage. Different antioxidant concentrations may produce varying effects on sperm quality parameters; therefore, identifying the optimal antioxidant level that maintains sperm motility and viability without inducing oxidative imbalance is necessary. Accordingly, this study was designed to evaluate the effect of different concentrations of GSH and vitamin C in Ringer’s lactate extender on the quality of Muscovy duck semen during cold storage. The objective of this research is to determine the optimal antioxidant concentration that maintains sperm quality for a longer period under storage conditions, thereby contributing to improved semen preservation techniques and supporting AI and genetic improvement programs for Indonesia’s indigenous poultry. Materials and MethodsAnimal husbandryFive 15-month-old male Muscovy ducks (C. moschata) weighing 3–5 kg were used. The ducks were housed in floor pens, provided with ad libitum access to drinking water, and fed a commercial ration (N544, PT Charoen Pokphand, Indonesia) at 160 g/duck/day. Only males with semen motility ≥70% in preliminary evaluations were included in the study. Chemicals and reagentsRinger’s lactate solution was used as the base extender. Treatments consisted of RL supplemented with GSH (0.1, 0.2, 0.3, and 0.4 mM) or vitamin C (0.568, 1.136, 1.704, and 2.272 mM). The control group (RL only) was included. The antioxidants were L(+)-ascorbic acid (Vitamin C, EMSURE®, Supelco/Merck, Cat. No. 1.00468.0100) and reduced L-GSH (BioReagent, Sigma-Aldrich, Cat. No. G6013-5G) suitable for cell culture. Additional reagents used for semen evaluation included 2% eosin, 3% NaCl, fructose, sodium citrate, penicillin, and distilled water. Semen collection and processingSemen was collected during the breeding season (December 2024 to April 2025) using the abdominal massage technique assisted by a female teaser. The ejaculates were collected in sterile 1.5 ml microtubes and immediately evaluated. The semen was then diluted to a concentration of 200 × 10⁶ sperm/ml using the respective extenders. Aliquots were stored at 5°C and assessed at 12-hours intervals until motility and viability reached 0%. Semen evaluationMacroscopic parameters included volume (ml), color (milky white, cream, or yellowish), consistency (watery, medium, or thick), and pH (measured using pH paper). Microscopic evaluations included – Mass movement, assessed at 100 × magnification and graded (+++ very good, ++ good, + poor, 0 none). –Motility, evaluated by mixing semen with saline and observing at 400 × magnification; essed as the percentage of progressively motile sperm. –Concentration, determined using a hemocytometer after dilution with 3% NaCl, following Arifiantini (2012). –Viability, assessed using 2% eosin stain, with unstained live sperm and red dead sperm. At least 200 cells were counted in 10 fields. –Abnormalities, determined from stained smears by counting ≥200 sperm; expressed as percentage. –Membrane plasma integrity was evaluated using the hypo-osmotic swelling test. The solution was prepared with 0.735 g sodium citrate and 1.351 g fructose in 100 ml distilled water (100 mOsm/kg). At least 200 sperm were examined, and membrane-intact cells with coiled tails were considered. Longevity assessmentLongevity was defined as the time (h) until sperm motility and viability declined to 0%. One drop of semen was placed on a clean slide, covered with a coverslip, and observed under 100 ×–400 × magnification at 12-hour intervals during storage. Semen remained viable in the control group (RL only) for up to 72 hours. Experimental design and analysis of dataThe experiment was arranged using a split-plot design within a CRD. • Main plots: antioxidant type and concentration of antioxidants (GSH or vitamin C). • Subplots: storage duration (0, 12, 24, 36, 48, 60, 72, 84, 96, 108, 120, 132, and 144 hours). Each treatment was replicated 5 times (biological replicates) using semen collected from different sessions. Data were analyzed using Tukey’s test in SPSS version 25 (IBM Corp., Armonk). Statistical significance was set at p < 0.05, and highly significant differences at p < 0.01. Observation parametersThe changes in sperm motility, viability, abnormalities, and membrane plasma integrity (MPI) of Muscovy duck semen during cold storage with different concentrations of GSH are illustrated in Figure 1, while the corresponding effects of vitamin C supplementation are presented in Figure 2. The diluent treatment showing the best overall performance was selected for further analysis in the subsequent phase of this study. Time and locationThis study was conducted from December 2024 to April 2025 at the Biotechnology Laboratory, Faculty of Animal Husbandry, Universitas Andalas, Indonesia. Ethical approvalThis study was approved by the Research Ethics Committee, Faculty of Medicine, Universitas Andalas, Padang, Indonesia (Approval No: 477/UN.16.2/KEP-FK/2024, dated September 6, 2024). All experimental procedures complied with the institutional guidelines for the care and welfare of animals. ResultsQuality of fresh semen from Muscovy ducksFresh semen from Muscovy ducks exhibited normal physical and microscopic characteristics. The average semen volume was 0.63 ± 0.19 ml, with a milky white color, typical odor, and thick consistency. The pH value (7.27 ± 0.46) indicated a neutral to slightly alkaline condition favorable for sperm survival. Microscopically, semen showed a mass movement score of 2.33 ± 0.49, individual motility of 83.33% ± 4.88%, viability of 94.53% ± 2.51%, and MPI of 91.2% ± 2.79%, with an abnormality rate of 5.70% ± 2.05%. The sperm concentration reached 1993 ± 533.80 million/ml, confirming that the ejaculates were of high quality and suitable for preservation, as shown in Table 1. Comparative effects of GSH and vitamin C on semen quality during storageMotilitySupplementation with both GSH and vitamin C significantly influenced sperm motility of Muscovy duck semen during cold storage (p < 0.01). In all treatments, motility progressively declined over time; however, the rate of decline was slower in the groups receiving lower antioxidant concentrations. For GSH, the 0.1 and 0.2 mM groups recorded the highest initial motility (80%), whereas the 0.4 mM group began at only 50%. The 0.1 mM treatment maintained motility above 50% for up to 48 hours, whereas the 0.4 mM treatment decreased motility to 0% by 36 hours. A similar trend was observed in vitamin C treatments, where the initial motility ranged from 80% in the 0.568 and 1.1356 mM groups to 70% in the 2.2712 mM group. The 0.568-mM vitamin C group maintained motility above 50% until 48 hours, whereas the 2.2712 mM group dropped below 50% as early as 24 hours. Statistical analysis using a split-plot design in a randomized block setup confirmed that antioxidant concentration, storage duration, and their interaction significantly affected sperm motility (p < 0.01). Overall, both 0.1 mM GSH and 0.568 mM vitamin C were the most effective in preserving motility for an extended period, while higher concentrations showed faster declines, likely due to oxidative or osmotic imbalance caused by excessive antioxidant presence. These results indicate that both antioxidants exert a dose-dependent effect on sperm motility, where optimal preservation occurs at lower concentrations, and excessive doses may induce oxidative stress, leading to reduced motility during storage, as shown in Table 2.
Fig. 1. Effect of GSH with a contrast of 0.1, 0.2, 0.3, and 0.4 mM as an antioxidant in Ringer’s lactate buffer on the quality parameters of Muscovy duck (C. moschata) spermatozoa during liquid storage at 5°C. (a) Motility, (b) viability, (c) abnormalities, and (d) intact plasma membrane at different observation times (0–144 hours).
Fig. 2. Effect of vitamin C with a contrast of 0.1, 0.2, 0.3, and 0.4 g as an antioxidant in Ringer’s lactate buffer on the quality parameters of Muscovy duck (C. moschata) spermatozoa during liquid storage at 5°C. (a) Motility, (b) viability, (c) abnormalities, and (d) intact plasma membrane at different observation times (0–144 hours). Table 1. Fresh semen quality of Muscovy duck (Cairina moschata).
ViabilitySupplementation with both GSH and vitamin C significantly affected the sperm viability of Muscovy duck semen during cold storage (p < 0.01). In all treatments, the viability decreased progressively with increasing storage duration, although the rate of decline varied depending on the antioxidant concentration. For GSH, the initial viability at 0 hours exceeded 85% in all groups, with the highest value recorded in the 0.1 mM treatment (90%). The 0.1 mM group maintained viability above 50% up to 84 hours, whereas the 0.4 mM group dropped below 50% after 48 hours. The 0.3 and 0.2 mM groups fell below this threshold at 60 and 72 hours, respectively. Similarly, in the vitamin C treatments, initial viability values were above 85% across all concentrations, with the 0568 mM group maintaining viability above 50% until 72 hours. In contrast, the 2.2712 mM group dropped below 50% after 36 hours, whereas the 1.1356 mM and 1.7034 Mm groups fell below 50% at 60 and 48 hours, respectively. Statistical analysis confirmed the significant effects of antioxidant concentration, storage duration, and their interaction (p < 0.01). Among all treatments, 0.1 mM GSH and 0.568 mM vitamin C consistently maintained higher viability percentages throughout storage, indicating superior protection at these lower doses. The sharp decline observed at higher concentrations of both antioxidants suggests that excessive supplementation may induce oxidative or osmotic imbalance, thereby accelerating the loss of sperm viability. These results demonstrate that both GSH and vitamin C exert a dose-dependent influence on sperm viability, with lower concentrations effectively extending cell survival during cold storage, as shown in Table 3. Table 2. Comparison of sperm motility between GSH and vitamin C treatments.
Table 3. Comparison of sperm viability between GSH and vitamin C treatments.
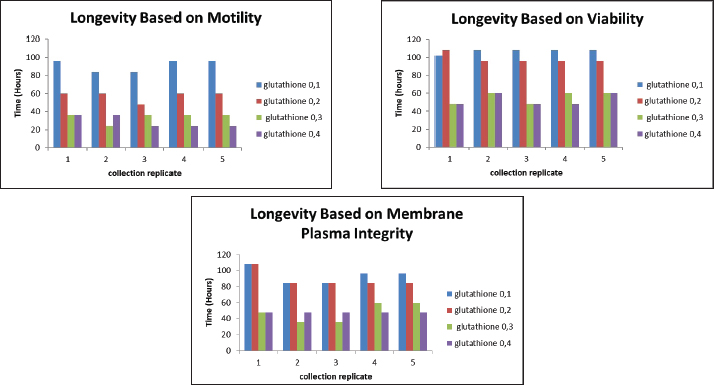
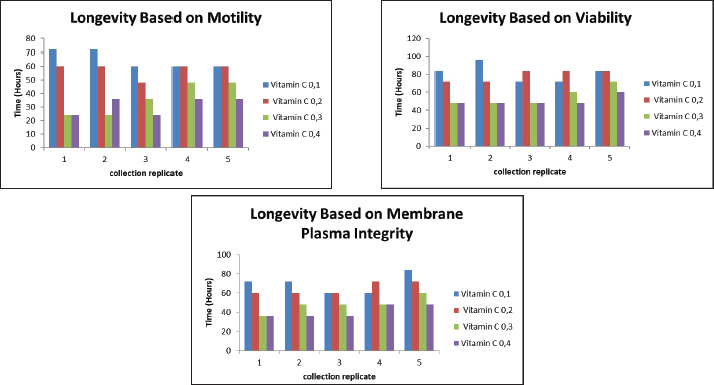
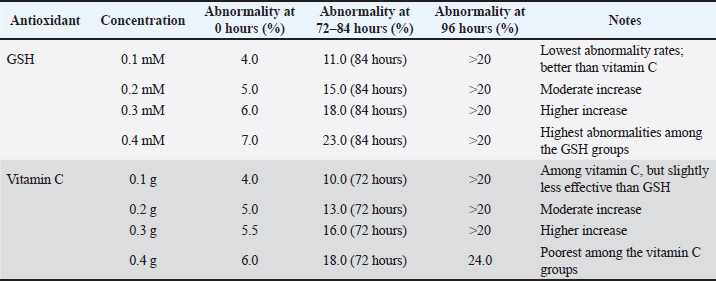
AbnormalitySperm abnormalities progressively increased during cold storage in both GSH- and vitamin C–supplemented semen; however, the lowest abnormality percentages in both treatments consistently occurred at the lowest concentrations (0.1 mM GSH and 0.568 mM vitamin C). This pattern indicates that low doses of both antioxidants effectively protect sperm morphology by reducing oxidative stress, in agreement with Sarangi et al. (2017), Pinto et al. (2020), Echekwu et al. (2021), and Fouda et al. (2021) who similarly reported improvements in morphological integrity with moderate antioxidant supplementation. The longevity of semen based on motility, viability, and membrane plasma integrity in the GSH-supplemented groups is shown in Figure 3, whereas the longevity patterns observed in vitamin C–supplemented semen are summarized in Figure 4. In contrast, higher concentrations (0.3–0.4 mM GSH; 0.568–2.2712 Mm vitamin C) resulted in increased abnormalities, suggesting that excessive antioxidant levels may become counterproductive. This aligns with the findings of Zou et al. (2021) who noted that high doses of GSH may disrupt osmotic balance, cellular signaling, or induce reductive stress, as well as the hypothesis of Lecewicz et al. (2018) that excessive vitamin C may act as a pro-oxidant in the presence of transition metals. The dose-dependent response observed in both antioxidants supports, who emphasized the importance of optimizing antioxidant concentrations to prevent oxidative or structural damage, particularly in avian spermatozoa with membranes rich in polyunsaturated fatty acids. Collectively, these findings show that low GSH and vitamin C doses provide protective effects, whereas higher doses increase the risk of morphological deterioration during storage, as shown in in Table 4.
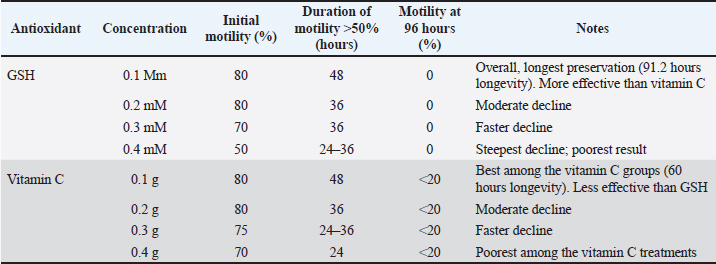
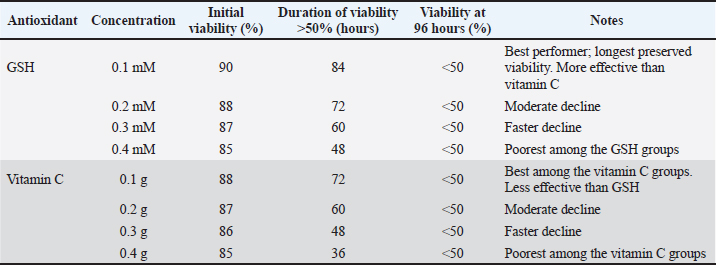
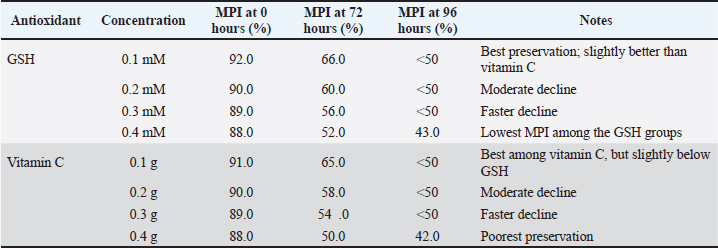
Fig. 3. The effect of GSH with a contrast of 0.1, 0.2, 0.3, and 0.4 mM as an antioxidant in Ringer’s lactate buffer on the longevity of Muscovy duck (C. moschata) spermatozoa during liquid storage at 5°C. (a) Longevity based on motility, (b) longevity based on viability, and (c) longevity based on intact plasma membrane at different observation times (0–144 hours).
Fig. 4. Effect of vitamin C with a contrast of 0.1, 0.2, 0.3, and 0.4 g as an antioxidant in Ringer’s lactate buffer on the longevity of Muscovy duck (C. moschata) spermatozoa during liquid storage at 5°C. (a) Longevity based on motility, (b) longevity based on viability, and (c) longevity based on intact plasma membrane at different observation times (0–144 hours). Membrane plasma integrityThe MPI of Muscovy duck spermatozoa progressively declined during cold storage in all treatment groups, with both GSH and vitamin C showing significant effects (p < 0.01). The initial MPI values at 0 hours ranged between 88.00% and 92.00% across all antioxidant concentrations. Among GSH treatments, the 0.1 mM group consistently maintained higher MPI values throughout storage, retaining 66.00% at 72 hours, while the 0.4 mM group dropped to 52.00% at the same time and reached 43.00% by 96 hours. Similarly, vitamin C treatments showed a comparable trend, where the 0.568 mM group maintained an MPI value of 65.00% at 72 hours, whereas the 2.2712 mM group decreased to 50.00%, and by 96 hours, all groups had fallen below 50% by 96 hours, with the lowest value (42.00%) observed in the 2.2712 mM treatment. Statistical analysis confirmed the significant effects of antioxidant concentration, storage duration, and their interaction (p < 0.01). In both antioxidants, higher concentrations led to a faster decline in MPI, whereas lower doses (0.1 mM GSH and 0.568 mM vitamin C) effectively preserved membrane integrity for a longer period during storage. These findings demonstrate that both GSH and vitamin C help stabilize sperm plasma membranes, but at lower concentrations that minimize oxidative or osmotic stress, optimal protection is achieved, as shown in Table 5. Semen longevitySemen longevity, defined as the duration during which sperm motility and viability remained above 50% during cold storage, was significantly affected by both GSH and vitamin C supplementation (p < 0.01). In the GSH treatments, the 0.1 mM group exhibited the longest preservation period, maintaining motility above 50% for up to 91.2 hours and viability for 84 hours. The 0.2 mM group sustained motility and viability above 50% for 60 and 72 hours, respectively, whereas the 0.3 and 0.40 mM groups dropped below these thresholds after 48–60 and 36–48 hours, respectively. Similarly, in the vitamin C treatments, the 0.568 mM group maintained motility above 50% for 60 hours and viability for 72 hours, followed by the 1.1356 mM group at 48 and 60 hours, and the 1.7034 mM group at 36 and 48 hours. The shortest preservation was recorded in the 2.2712 mM group, which fell below 50% for motility and viability after only 24 and 36 hours, respectively. Statistical analysis confirmed that antioxidant concentration, storage time, and their interaction significantly influenced semen longevity (p < 0.01). Overall, both GSH and vitamin C demonstrated dose-dependent effects, with lower concentrations (0.1 and 0.568 mM) providing optimal protection and prolonging semen longevity, while higher concentrations accelerated quality deterioration, likely due to oxidative or osmotic imbalance during storage. Control group (Ringer’s lactate only)In the RL-only control group, sperm quality declined rapidly during cold storage. Motility decreased from 80% at 0 hours to 0% at 72 hours (80, 60, 40, 24, 12, 4, and 0). Viability followed a similar pattern, dropping from 86.4% at 0 hours to 0% at 72 hours (86.4, 69.4, 45.5, 28.9, 14.6, 7.2, and 0, respectively). The abnormalities progressively increased from 8.1% at 0 hours to 29.4% at 72 hours (8.1, 11.8, 15.5, 18.9, 24.6, 27.7, and 29.4). Semen longevity in the control group, defined as the time during which motility and viability remained above 50%, averaged 50.4 hours across five replicates (60, 48, 60, 36, and 48 hours). These results confirmed that without antioxidant supplementation, Ringer’s Lactate could maintain semen quality for a maximum of 72 hours under cold storage. Table 4. Comparison of sperm abnormality between GSH and vitamin C treatments.
Table 5. Comparison of sperm membrane plasma integrity (MPI) between GSH and vitamin C treatments.
DiscussionFresh semenMuscovy duck semen showed normal physical and microscopic characteristics, indicating good fertility potential and suitability for dilution and storage. The average volume (0.63 ± 0.19 ml) and pH (7.27 ± 0.46) were within the normal physiological range, with a milky-white color, thick consistency, and typical odor. Microscopically, sperm motility (83.33 ± 4.88%), viability (94.53% ± 2.51%), and membrane plasma integrity (91.2% ± 2.79%) were high, whereas abnormality remained low (5.70% ± 2.05%), and sperm concentration reached 1.993 ± 533.80 × 10⁶/ml. These results confirm that the semen collected was of high quality and comparable to values previously reported in poultry and waterfowl species. The favorable characteristics observed in the fresh semen reflect the natural antioxidative stability of Muscovy duck spermatozoa, which plays a critical role in maintaining motility and viability before storage (Lecewicz et al., 2018; Fouda et al., 2021; Echekwu et al., 2021). MotilityThe motility of Muscovy duck sperm progressively declined during cold storage in all treatment groups. Supplementation with antioxidants, however, significantly slowed this reduction compared to the control. Among all concentrations, 0.1 mM GSH and 0.568 mM vitamin C maintained the highest motility rates throughout the observation period, indicating optimal antioxidant protection at these concentrations. GSH supplementation effectively preserved sperm motility, with 0.1 mM showing the best results, in agreement with previous studies reporting that low GSH levels reduce oxidative stress and stabilize mitochondrial activity (Pinto et al., 2020; Zhandi et al., 2020; Syafitri et al., 2022). In contrast, higher concentrations (0.3–0.4 mM) caused a faster motility decline, likely due to osmotic imbalance and reductive stress (Zou et al., 2021). Vitamin C treatment showed a concentration-dependent effect. The 0.568 mM dose significantly preserved motility, whereas higher concentrations reduced it more rapidly. This pattern aligns with previous findings in poultry, suggesting that low-dose vitamin C enhances motility by maintaining mitochondrial and membrane stability (Fouda et al., 2021; Echekwu et al., 2021). However, excessive vitamin C may act as a pro-oxidant, generating hydrogen peroxide and accelerating oxidative stress (Lecewicz et al., 2018). Overall, both antioxidants improved sperm motility at low doses, but GSH (0.1 mM) and vitamin C (0.568 mM) provided the most effective preservation. These findings indicate that optimal antioxidant concentration plays a crucial role in maintaining sperm motility during cold storage. ViabilityAntioxidant supplementation significantly affected the viability of Muscovy duck sperm during cold storage. Both GSH and vitamin C showed a concentration-dependent response, with the lowest doses producing the best results. GSH supplementation at 0.1 mM maintained sperm viability for a longer period than higher concentrations. This result indicates that low-dose GSH effectively protects cells from oxidative damage during preservation. GSH neutralizes ROS that cause lipid peroxidation and disrupt membrane integrity (Pinto et al., 2020). The improved viability observed at 0.1 mM supports the findings of Syafitri et al. (2022), who reported enhanced sperm survival in poultry when low GSH levels were used. However, elevated concentrations (0.3–0.4 mM) decreased viability, possibly due to osmotic or oxidative imbalance. Zou et al. (2021) noted similar inhibitory effects of excessive GSH in goat semen, emphasizing that oversupplementation can trigger reductive stress and impair cell stability. Vitamin C treatment showed a similar trend. The concentration of 0.568 Mm preserved sperm viability most effectively, consistent with earlier studies demonstrating that moderate vitamin C enhances sperm survival by reducing oxidative damage and maintaining membrane stability (Surai, 2016; Fouda et al., 2021; Echekwu et al., 2021). In contrast, higher doses (1.1356–2.2712 mM) reduced viability, likely due to the pro-oxidant potential of vitamin C at excessive concentrations. Lecewicz et al. (2018) reported that surplus ascorbic acid may generate hydrogen peroxide in the presence of metal ions, aggravating oxidative stress. This dose-dependent behavior supports the observation, who emphasized the importance of maintaining optimal antioxidant balance for sperm preservation. Overall, both antioxidants enhanced sperm viability at low concentrations, but excessive doses diminished their benefits. GSH (0.1 mM) and vitamin C (0.568 mM) were identified as the most effective levels for prolonging sperm viability during cold storage, highlighting the importance of precise antioxidant optimization in Muscovy duck semen preservation. Membrane plasma integrityThe results of this study demonstrated that both GSH and vitamin C supplementation significantly influenced the preservation of MPI in Muscovy duck sperm during cold storage, with the lowest concentrations (0.1 mM GSH and 0.568 mM vitamin C) consistently maintaining the highest MPI values throughout the storage period. As an intracellular antioxidant, GSH stabilizes membrane phospholipids and prevents lipid peroxidation, thereby minimizing cold-induced damage (Sarangi et al., 2017; Pinto et al., 2020). Similarly, vitamin C, a potent water-soluble antioxidant, protects the sperm plasma membrane by neutralizing ROS and preventing polyunsaturated fatty acid oxidative degradation (Surai, 2016). These findings are consistent with previous studies in poultry semen showing that low doses of GSH and vitamin C enhance membrane integrity and post-storage sperm functionality (Zhandi et al., 2020; Echekwu et al., 2021; Fouda et al., 2021; Syafitri et al., 2022). Conversely, higher concentrations (0.3–0.4 mM GSH and 1.7034–2.2712 Mm vitamin C) resulted in a faster decline of MPI, likely due to osmotic or redox imbalance and potential pro-oxidant effects leading to increased oxidative stress (Lecewicz et al., 2018; Zou et al., 2021). These results confirm the dose-dependent nature of antioxidant effects, where optimal levels maintain sperm membrane stability and functional longevity, while excessive supplementation disrupts redox homeostasis and accelerates membrane deterioration. LongevityThe results of this study demonstrated that both vitamin C and GSH supplementation significantly influenced the longevity of Muscovy duck semen during cold storage, as indicated by the duration during which sperm motility and viability remained above 50%. Optimal preservation was observed at low concentrations (0.568 Mm vitamin C and 0.1 mM GSH), whereas higher concentrations (0.3–0.4) led to a faster decline in semen quality. GSH plays a central role in intracellular antioxidant defense by scavenging ROS and maintaining redox homeostasis within sperm cells. During semen storage, oxidative stress is a major contributor to motility loss, membrane disruption, and apoptosis (Sarangi et al., 2017). The improved longevity in the 0.1 mM group supports the findings of Syafitri et al. (2022) and Zou et al. (2021), indicating that GSH supplementation stabilizes mitochondria and membrane structure, delaying oxidative damage. However, excessive GSH (0.3–0.4 mM) may cause reductive stress or osmotic imbalance, resulting in premature sperm inactivation (Zou et al., 2021). Vitamin C (ascorbic acid) acts as a potent extracellular antioxidant that neutralizes ROS and prevents lipid peroxidation in sperm membranes. The 0.568 mM concentration extended the functional lifespan of sperm more effectively than higher doses, consistent with findings in turkeys and ducks (Echekwu et al., 2021; Fouda et al., 2021). In contrast, higher concentrations (1.7034–2.2712 mM) exhibited a pro-oxidant effect, possibly due to hydrogen peroxide formation in the presence of metal ions (Lecewicz et al., 2018). When compared directly, both antioxidants exhibited a similar dose-dependent pattern—optimal at low concentrations but harmful when excessive. However, their modes of action differ: GSH primarily acts within the cell to maintain enzymatic redox systems, whereas vitamin C scavenges free radicals in the extracellular environment. This complementary mechanism suggests that both could be beneficial when properly balanced within the extender formulations. The findings align with those of who emphasized that antioxidant efficacy is dose- and species-dependent. In conclusion, GSH (0.1 mM) and vitamin C (0.568 mM) effectively extend the longevity of Muscovy duck semen during cold storage by minimizing oxidative stress and preserving sperm function, whereas higher concentrations compromise preservation efficiency. Control group (Ringer’s lactate only)The present study demonstrated that RL was the most effective extender for preserving Muscovy duck semen during cold storage, particularly in maintaining motility and viability up to 48–60 hours. This superior performance is attributed to its balanced electrolyte composition and lactate content, which act as a natural buffer to stabilize pH and as an energy source to support sperm metabolism (Ananda et al., 2023; Jaswandi et al., 2023; Agustin et al., 2025). Compared with phosphate buffered saline, Beltsville poultry semen extender, and NaCl, RL consistently provided higher motility and viability values and a slower increase in abnormalities, confirming its stronger protective capacity. These findings are consistent with those of Khaeruddin et al. (2016) and Agustin et al. (2025), who observed >70% motility in chicken spermatozoa stored in RL for up to 60 hours at 5°C, and Wajo et al. (2023), who reported better results with RL than with NaCl or other diluents. The protective effect of RL is likely related to its ability to maintain osmotic balance and reduce oxidative stress-induced membrane damage (Mateu‐Sanz et al., 2020; Pitaloka et al., 2023). Other studies (Pearlin et al., 2020; Burilo and Kashoma, 2023) have also emphasized that extenders containing balanced electrolytes, antioxidants, and energy sources can prolong sperm survival by preventing oxidative stress and preserving membrane integrity. ConclusionThis study demonstrated that Ringer’s lactate supplemented with GSH or vitamin C effectively preserved the quality of Muscovy duck semen during cold storage. Among the treatments, 0.1 mM GSH and 0.568 mM vitamin C maintained higher motility, viability, and membrane plasma integrity while reducing abnormalities compared to higher concentrations. The semen quality of the control group (RL only) was sustained for up to 72 hours, whereas the addition of antioxidants extended longevity to 91.2 hours (GSH) and 72 hours (vitamin C). These findings highlight the importance of optimal antioxidant supplementation in semen extenders to improve artificial insemination outcomes in Muscovy ducks. AcknowledgmentsThe authors express their appreciation to Dr. Masrizal for his valuable assistance and guidance in experimental design and data analysis, which greatly contributed to the completion of this research. Conflict of interestThe authors declare no conflict of interest. FundingThe authors express their sincere gratitude to the Universitas Andalas Institute for Research and Community Service (LPPM) for funding this research under Contract Number: 184/UN.16.19/PT.01.03/PTM/2025 dated September 6, 2024. Author’s contributionsBWA was responsible for data collection and manuscript drafting. TA reviewed the manuscript and acted as the corresponding author. A validated the data, reviewed the manuscript, and supervised the laboratory activities. TDN supervised the field activities and reviewed the manuscript. All authors have read and approved the final version of the manuscript. Data availabilityAll data are available in the revised manuscript. ReferencesAgung, D., Marawali, A., Uly, K. and Telupere, F. 2023. Pengaruh penambahan beberapa level glutathione dalam pengencer air kelapa kuning telur terhadap kualitas semen sapi Angus. J. Nukleus. Peternakan. 10(1), 27–37. Agustin, B.W., Afriyani, T., Nova, T.D. and Ananda. 2025. Evaluation of different extenders on the longevity and quality of Muscovy duck (C. moschata) semen during storage. Int. J. Vet. Sci. 14(6), 1221–1228; doi:10.47278/journal.ijvs/2025.080 Amini, M.R., Kohram, H., Shahneh, A.Z. and Zare-Shahaneh, A. 2015. Effects of vitamin C and E supplementation during semen freezing on post-thawed sperm quality in rams. Iran. J. Appl. Anim. Sci. 5(1), 147–152. Ananda, A., Jaswandi, J., Rusfidra, R. and Gusdinal, H. 2023. Sperm longevity and motility in ringer’s lactate solution with addition of egg yolk among five phenotypes of kokok balenggek chicken. Buletin Peternakan 47(3), 127. doi: 10.21059/buletinpeternak.v47i3.83647 Arifiantini, R.I., 2012. Teknik Koleksi Dan Evaluasi Semen Pada Ternak. IPB PresS, Bogor. Azura, I., Diah, L.M., Wurlina, W. and Wahyuningsih, W. 2020. The role of antioxidants in semen extender to reduce oxidative stress. J. Ilm. Peternakan Terpadu 8(1), 10–15. Burilo, A. and Kashoma, I. 2023. Effect of semen extenders, dilution rates and storage periods on spermatozoa quality of horasi chicken ecotype. Appl. Vet. Res. 2(4), 2023020; doi:10.31893/avr.2023020 Çelik, E.S., Köse, M., Türk, G. and Yüce, A. 2020. Role of oxidative stress and antioxidants in male fertility. J. Anim. Sci. Adv. 10(3), 45–52. Djaya, A.S. 2003. Penggemukan Itik Jantan Potong. Penerbit Penebar Swadaya, Jakarta. Echekwu, C.A., Uzochukwu, I.C. and Ugwu, S. 2021. Vitamin C supplementation improves semen quality in indigenous Nigerian turkeys. J. Vet. Sci. Anim. Husbandry. 9(1), 18–25. Eslami, M., Ghasemzadeh-Nava, H., Kohram, H. and Zhandi, M. 2018. Effects of antioxidant supplementation in semen extender on rooster sperm quality. Anim. Reprod. 15(2), 145–152. Fan, H., Liu, Z., Zhan, P. and Jia, G. 2022. Pericentric inversion of chromosome 6 and male fertility problems. Open Med. 17(1), 191–196. Fouda, S.F., El-Darawany, A.A. and Rania, A.A. 2021. Effect of vitamin C supplementation on semen quality in poultry. J. Anim. Reprod. 45(3), 214–222. Hamidian, F., Aghaz, F. and Khani, F. 2020. High-dose vitamin C induces oxidative stress in rooster sperm: implications for antioxidant balance. Theriogenology. Insight. 10(1), 1–9. Han, X., Zhou, N., Cui, Z., Ma, M., Li, L., Cai, M. and Cao, J. 2011. Association between urinary PAHs and sperm DNA damage. Environ. Health Perspect. 119(5), 652–657. Harissatria, H., Hendri, J., Elinda, F., Jaswandi, J., Hendri, H., Zumarni, Z. and Afrini, D. 2023. Evaluation of sperm motility and viability in poultry semen with antioxidant supplementation. J. Trop. Vet. Sci. 7(1), 33–40. Jaswandi, J., Ananda, A., Rusfidra, R., Subekti, K., Gusdinal, H., Sri Wahyuni,, R. and Ariansyah Caniago, F. 2023. Fertility rate, fertility period, and DOC sex ratio of Kokok Balenggek chicken after Artificial Insemination. Anim. Vet. Sci. 11(5), 795–801; doi:10.17582/journal.aavs/2023/11.5.795.801 Khaeruddin, K., Arifiantini, R.I., Sumantri, C. and Darwati, S. 2016. Kualitas spermatozoa ayam peranakan sentul dalam pengencer ringer laktat kuning telur dengan berbagai monosakarida (quality of sentul crossbreed chicken spermatozoa in ringer lactate-egg yolk diluents supplemented with various monosaccharide). Jurnal. Kedokteran. Hewan. - Indonesian J. Vet. Sci. 10(2), 166–169. Khoirunnisa, L., Nur, Z. and Astuti, P. 2020. Protective role of vitamin C and E in improving post-thawed semen quality. J. Reprod. Sci. 4(2), 90–95. Kusumawati, E.D. and Leondro, H. 2014. Inseminasi Buatan. Fakultas Peternakan dan Kedokteran Hewan, Malang. Lecewicz, A., Strzeżek, R., Fraser, L. and Ząbek, T. 2018. The effect of vitamin C on sperm chromatin integrity. Theriogenology 118, 112–117. Mahmood, S. and Naoman, M.H. 2023. Comparative effect of glutathione and melatonin on sperm antioxidant capacity in poultry. Int. J. Vet. Sci. 12(1), 49–56. Mateu‐Sanz, M., Tornín, J., Brulin, B., Khlyustova, A., Ginebra, M., Layrolle, P. and Canal, C. 2020. Cold plasma-treated ringer’s saline: a weapon to target osteosarcoma. Cancers 12(1), 227; doi:10.3390/cancers12010227 Maulana, H., Nurcahyo, W. and Prastiya, E. 2016. Abnormal morphology in duck spermatozoa during cold storage. J. Peternakan. Nusantara. 21(1), 41–48. Mustafa, M., Dasrul, D., Yaman, M., Wahyuni, S. and Sabri, M. 2017. Pengaruh pakan fermentasi dan vitamin E terhadap kualitas semen ayam Arab. J. Agripet. 17(1), 43–52. Oliveira, R., Viu, M. and Gambarini, M. 2015. Glutathione supplementation in goat semen extenders. Anim. Reprod. 12(2), 230–236. Pearlin, V.B., Mohan, J., Tyagi, J.S., Gopi, M., Kolluri, G., Prabakar, G. and Shanmathy, M. 2020. Efficiency of different diluents and dilution rates on the fertilization potential of chicken spermatozoa. Indian. J. Anim. Res. 55, 139–144 Pinto, S., Almeida, d., Alves, M., Florez-Rodriguez, S., Júnior, G., Alves, N. and Souza, F. 2020. Supplementation of vitamin C and glutathione in semen extender. Arq. Bras. Med. Vet. Zootec. 72(1), 9–17. Pitaloka, D.G., Mas’Ud Hariadi., Suherni Susilowati., Budi Utomo., Tatik Hernawati., Eka Pramyrtha Hestianah. and Samuel Inioluwa Akeju. 2023. The effect of addition of melon (Cucumis melo L.) flesh juice into lactated ringer’s-egg yolk extender on spermatozoa plasma membrane integrity and spermatozoa morphological abnormalities of semen of native rooster. Ovozoa J. Anim. Reprod. 12(1), 49–58. Pratiwi, D., Nugroho, T. and Susilawati, T. 2019. Antioxidant supplementation in semen extender. J. Ilm. Peternak. 7(1), 54–61. Quraini, M., Luthfi, H. and Hidayat, B. 2022. Sperm viability and motility in duck semen with different extenders. J. Trop. Anim. Sci. 2(3), 45–50. Rakha, B.A., Ansari, M.S. and Akhter, S. 2016. Freezing tolerance and post-thawed fertility of Muscovy duck semen. Reprod. Domest. Anim. 51(1), 56–60. Ramahtia, D., Cahyono, B. and Huda, M.N. 2024. Effects of extender type and vitamin addition on sperm characteristics in birds. J. Trop. Vet. Sci. 5(1), 33–40. Saieed, H.A., Sharafi, M. and Hosseini, S. 2019. Redox balance in poultry semen. Anim. Reprod. Sci. 205, 77–85. Sarangi, A., Singh, P., Virmani, M., Yadav, A., Sahu, S., Ajithakumar, H. and Rath, A. 2017. Effect of glutathione supplementation on semen quality. Vet. World. 10(6), 675–681. Shabani, A., Mozaffari, M. and Rahimi, S. 2022. Vitamin C toxicity in sperm preservation: a cautionary study. Int. J. Vet. Sci. 11(2), 132–139. Shaliutina-Kolešová, A., Štolc, L., Štiavnická, M. and Lukáč, N. 2013. Effect of glutathione on oxidative stress in avian semen. J. Microbiol. Biotechnol. Food. Sci. 2(Special Issue), 2162–2164. Solihati, N., Wahyuningsih, W., Wurlina, W. and Isnaini, N. 2020. Role of glutathione and vitamin C on sperm quality. J. Ilm. Peternakan. Terpadu. 8(2), 25–30. Syafitri, M., Prabowo, T., Sitaresmi, P., Yusiati, L., Bintara, S. and Widayati, D. 2022. The effect of glutathione addition in semen diluent on sperm quality. In Proc. Int. Conf. Agric. Dordrecht, The Netherlands: Atlantis Press. Toelihere, M.R. 1985. Fisiologi Reproduksi pada Ternak. Angkasa, Bandung. Uzochukwu, I.C., Ezeokeke, C.T. and Okechukwu, I.U. 2020. Antioxidant supplementation improves chilled semen quality of turkey toms. J. Adv. Vet. Anim. Res. 7(3), 539–545. Wajo, M.J., Widayati, I. and Ratlalan, B. 2023. Penambahan yolk puyuh, sari pepaya (Carica papaya L.), ringer’s serta lama penyimpanan pada suhu 50C terhadap kualitas spermatozoa ayam kub. Jurnal. Ilmu. Peternakan. Dan. Veteriner. Tropis. 13(3), 159–166. Wiyanti, D.C., Isnaini, N. and Trisunuwati, P. 2013. Pengaruh lama simpan semen dalam pengencer NaCl terhadap kualitas spermatozoa ayam kampung. J. Kedokteran. Hewan. 7(1), 53–55. Xia, W., Chen, W. and Abouelezz, K. 2020. Dietary Se and reproductive performance in laying ducks. Poult. Sci. 99(8), 3971–3978. Zhandi, M., Seifi-Ghajalo, E., Shakeri, M., Yousefi, A.R., Sharafi, M. and Seifi-Jamadi, A. 2020. Effect of glutathione supplementation to semen extender on post-thawed rooster sperm quality indices frozen after different equilibration times. Cryo. Lett. 41(2), 92–99. Zhang, X.G., Li, H., Wang, L., Hao, Y.Y., Liang, G.D., Ma, Y.H., Yang, G.S. and Hu, J.H. 2016. The effects of different levels of superoxide dismutase in modena on boar semen quality during liquid preservation at 17°C. Anim. Sci. J. 88(1), 55–62. Zou, M., Zhou, Y., Hu, J. and Zhang, Z. 2021. Glutathione protects chicken sperm during cryopreservation. Cryobiology 98, 79–85. Zuha, S., Rakha, B.A., Akhter, S., Ansari, M.S. and Waseem, K. 2024. Effect of reduced glutathione on quality, lipid peroxidation and antioxidant potential of frozen‐thawed ring‐necked pheasant semen. Reproduct. Domestic Animals 59(2), e14535; doi:10.1111/rda.14535 | ||
| How to Cite this Article |
| Pubmed Style Agustin BW, Afriani T, Nova TD, Ananda A. Optimization of the best extender for liquid semen of Muscovy duck (Cairina moschata) with the addition of different concentrations of vitamin C and glutathione. Open Vet. J.. 2026; 16(2): 1048-1059. doi:10.5455/OVJ.2026.v16.i2.25 Web Style Agustin BW, Afriani T, Nova TD, Ananda A. Optimization of the best extender for liquid semen of Muscovy duck (Cairina moschata) with the addition of different concentrations of vitamin C and glutathione. https://www.openveterinaryjournal.com/?mno=276128 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.25 AMA (American Medical Association) Style Agustin BW, Afriani T, Nova TD, Ananda A. Optimization of the best extender for liquid semen of Muscovy duck (Cairina moschata) with the addition of different concentrations of vitamin C and glutathione. Open Vet. J.. 2026; 16(2): 1048-1059. doi:10.5455/OVJ.2026.v16.i2.25 Vancouver/ICMJE Style Agustin BW, Afriani T, Nova TD, Ananda A. Optimization of the best extender for liquid semen of Muscovy duck (Cairina moschata) with the addition of different concentrations of vitamin C and glutathione. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1048-1059. doi:10.5455/OVJ.2026.v16.i2.25 Harvard Style Agustin, B. W., Afriani, . T., Nova, . T. D. & Ananda, . A. (2026) Optimization of the best extender for liquid semen of Muscovy duck (Cairina moschata) with the addition of different concentrations of vitamin C and glutathione. Open Vet. J., 16 (2), 1048-1059. doi:10.5455/OVJ.2026.v16.i2.25 Turabian Style Agustin, Betri Wahyu, Tinda Afriani, Tertia Delia Nova, and Ananda Ananda. 2026. Optimization of the best extender for liquid semen of Muscovy duck (Cairina moschata) with the addition of different concentrations of vitamin C and glutathione. Open Veterinary Journal, 16 (2), 1048-1059. doi:10.5455/OVJ.2026.v16.i2.25 Chicago Style Agustin, Betri Wahyu, Tinda Afriani, Tertia Delia Nova, and Ananda Ananda. "Optimization of the best extender for liquid semen of Muscovy duck (Cairina moschata) with the addition of different concentrations of vitamin C and glutathione." Open Veterinary Journal 16 (2026), 1048-1059. doi:10.5455/OVJ.2026.v16.i2.25 MLA (The Modern Language Association) Style Agustin, Betri Wahyu, Tinda Afriani, Tertia Delia Nova, and Ananda Ananda. "Optimization of the best extender for liquid semen of Muscovy duck (Cairina moschata) with the addition of different concentrations of vitamin C and glutathione." Open Veterinary Journal 16.2 (2026), 1048-1059. Print. doi:10.5455/OVJ.2026.v16.i2.25 APA (American Psychological Association) Style Agustin, B. W., Afriani, . T., Nova, . T. D. & Ananda, . A. (2026) Optimization of the best extender for liquid semen of Muscovy duck (Cairina moschata) with the addition of different concentrations of vitamin C and glutathione. Open Veterinary Journal, 16 (2), 1048-1059. doi:10.5455/OVJ.2026.v16.i2.25 |