| Research Article | ||
Open Vet. J.. 2026; 16(2): 1151-1161
Open Veterinary Journal, (2026), Vol. 16(2): 1151-1161 Research Article Effect of phytobiotics on humoral immune response and survival rate against avian Avulavirus challenge in broiler chickensHayder Abd Al-Emier Almremdhy* and Dheyae Ali AljuhashiDepartment of Pathology and Poultry Diseases, College of Veterinary Medicine, Al-Qasim Green University, Babylon, Iraq *Corresponding Author: Hayder Abd Al-Emier Almremdhy. Department of Pathology and Poultry Diseases, College of Veterinary Medicine, Al-Qasim Green University, Iraq. Email:hiederalmremdhy [at] vet.uoqasim.edu.iq Submitted: 25/08/2025 Revised: 30/12/2025 Accepted: 16/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
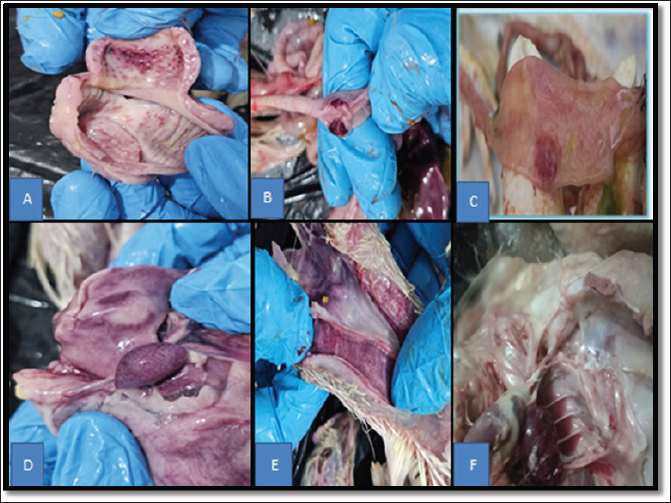
AbstractBackground: Phytobiotics are natural plant-derived products that have gained recognition as superior alternatives to antibiotics that provide benefits without the risks of antibiotic resistance and residues. Aim: This study investigated the efficacy of phytobiotics supplemented in drinking water on the immune response and survival rate of broiler chickens against challenge from the Avian Avulavirus 1. Method: 200 one-day-old broiler chickens were selected. At first day, 20 chickens were sacrificed, and their blood sample was collected to estimate the maternal antibody titer against Avian Avulavirus 1 (Newcastle disease virus) using the indirect enzyme-linked immunosorbent assay (ELISA). The remaining 180 chicks were allocated into a completely randomized design with three replicates (n=15) of four groups (n=45). Chickens in the first group were vaccinated twice against Newcastle disease virus (NDV) by live attenuated vaccine on days 9 and 18 only, without any additives. Chickens in the second and third groups were vaccinated twice against NDV on days 9 and 18, respectively, in addition to 0.3 ml/ l of coriander oil and thyme oil added to their drinking water, from day 1 to the last day of the experiment. The chickens of the fourth group were left unvaccinated and without supplements as a control group. Ten blood samples were taken from each group on day 23 to measure the serum antibody titer against NDV using an ELISA test. On day 25, all chickens in all groups were challenged with a virulent Newcastle disease virus. Chickens in all groups were monitored for 10 days after the challenge to record clinical signs, postmortem lesions, and mortality rate. Results: There was a significant difference (p ≤ 0.05) between the treatment groups (G1, G2, and G3) and the control group (G4) in the mean antibody titer against NDV at day 23 of the chicks life. In contrast, there was a significant difference (p ≤ 0.05) between the groups that were vaccinated against NDV and supplemented with phytobiots in drinking water (G2 and G3) and the G1 group that was vaccinated against NDV only without any supplement. In addition, a higher survival rate after NDV challenge was recorded in the phytobiotic supplemented groups along with fewer of clinical signs, gross lesions, and mortality rates. Conclusion: The study concluded that adding a phytobiotic (coriander or thyme oil) to the drinking water had a positive effect on the humoral immune response and survival rate of vaccinated chickens against NDV infection. This study recommends adding coriander oil or thyme oil to the drinking water of broiler chickens due to their positive effects on the immunity and health of the birds. Keywords: Phytobiotic, Avulavirus 1, Humoral immune response, Challenge, Broiler. IntroductionModern broiler hybrids are characterized by rapid growth, high production, and low feed conversion ratio if optimal health, environmental, and nutritional conditions are met (Hafez and Attia, 2020; Maharjan et al., 2021). This has had a positive impact on the poultry industry, which has become one of the largest and fastest-growing industries worldwide (Khalifa et al., 2013; Dixon, 2020). At the same time, the poultry industry is exposed to many threats related to various infectious diseases that lead to significant economic losses due to reduced bird performance and growth retardation, as well as deterioration of egg quality, and culling (Dixon, 2020; Hafez and Attia, 2020). Newcastle disease is an acute, highly contagious, and highly infectious viral disease that causes significant health and economic problems for the global poultry industry (Irshad et al., 2020; Turan et al., 2020). Newcastle disease virus (NDV) can infect more than 240 species of domestic and wild birds, regardless of sex and age; however, chickens, turkeys, ducks, and pigeons are the most commonly affected species (Suarez et al., 2020; Amoia et al., 2021). NDV can spread through several routes, primarily through direct contact between uninfected and infected birds. It can be also be transmitted through the droppings and nose, mouth, and eye secretions of infected birds. In addition, it can be transmitted by contaminated airborne, water, feed, and transport (Irshad et al., 2020). Due to its potential to spread rapidly to naïve birds in the vicinity, ND is included on the list of avian diseases that must be reported to the World Organization for Animal Health immediately upon detection (Bello et al., 2018). The causative agent of ND is a virulent strain of Paramyxovirus serotype -1 (APMV-1) also known as Avian orthoavulavirus-1, which belongs to the order Mononegavirales, family Paramyxoviridae, APMV-1 is a single-strand non segmented negative sense RNA virus (International Committee on Taxonomy of Viruses, 2023). Based on clinical signs and mortality rate in infected chickens, APMV-1 is divided into lentogenic, mesogenic, and velogenic strains. The Velogenic strains are subdivided into neurotropic velogenic NDV and viscerotropic velogenic NDV (Mansour et al., 2021; Shan et al., 2021). Broiler chickens with Newcastle disease can present with a range of clinical signs, including loss of appetite, listlessness, abnormal thirst, weakness, and conjunctivitis. Respiratory signs such as nasal discharge, sneezing, gasping, and coughing, and greenish watery diarrhea are clear intestinal signs. In addition to the nervous symptoms, which included twisting of the head and neck and paralysis of the wings and/or legs (Wajid et al., 2018; Suarez et al., 2020). Al-Habeeb et al., (2013) reported that the severity of these clinical signs is dependent on many factors, such as the virus strain, host species, age of the host, secondary infection, environmental stress, and immune status. In Iraq, Newcastle disease has been causing huge economic losses in the poultry industry since, it was first isolated from chickens in 1968, and this isolate was named AG/68, and since that date, Iraq has become endemic for Newcastle disease (Allan et al., 1978). As with many other countries, Iraq has recognized the importance of combating the disease. Control methods for this disease depend on the application of strict biosecurity measures and intensive vaccination (Ahmed and Odisho, 2018 ; Getabalew Alemneh et al., 2019; Chung et al., 2021). The primary purpose of all poultry vaccinations is to induce a protective immune response that could prevent or reduce the economic loss caused by viral infection and diseases (Getabalew Alemneh et al., 2019). Despite the use of various vaccines and the implementation of various vaccination programs on a large scale in poultry farms to prevent and control Newcastle disease infection, the annual outbreak of Newcastle disease continues in vaccinated birds (Wang et al., 2015). Although the use of immunostimulants is not a prerequisite for improving the immunity of broiler chickens, they are a widely used and effective tool in the poultry industry to enhance immune responses and reduce disease susceptibility (Ali et al., 2019). Herbal modulators can be administered along with the vaccine to elicit a faster and stronger immune response (Deng et al., 2014). Many researchers have used various feed additives, such as antibiotics, probiotics, prebiotics, and herbal plants, in animal diets to improve the immune responses, decrease the negative influence of environmental stressors, and improve growth performance (Lin et al., 2015; Lai et al., 2015; Lin et al., 2016; Teng et al., 2017; Lin et al., 2017; Lin et al., 2018). Phytobiotics are natural plant-derived products that have gained recognition as a superior alternative to antibiotics that provide benefits without the risks of antibiotic resistance and residues, which can jeopardize consumer safety and animal welfare (Seidavi et al., 2021). These include essential oils, plant extracts, herbs, and spices (Suresh et al., 2018). Obianwuna et al. (2023) indicated that phytobiotics can be used as feed additives in the poultry industry to improve gut health, providing a safer and more sustainable option for broiler production. Gut health is essential for maintaining overall health and performance in broiler chickens, as well as enhancing intestinal antioxidant capacity, immune function, epithelial barrier integrity, gut microbiota composition, and intestinal villus morphology. Previous studies have confirmed that phytoiotics have natural antimicrobial, anti-inflammatory, and antioxidant properties (Crespo et al., 2019; Gessner et al., 2017; Gilani et al., 2021; Obianwuna et al., 2022). Almremdhy and Al-khafaji (2020a,b) Almremdhy (2020) and Almremdhy and Al-khafaji (2020a,b) pointed to the ability of phytoiotic to stimulate the immune response and improve productive performance in broiler chickens. Coriander (Coriandrum sativum) belongs to the Apiaceae family, and thyme (Thymus vulgaris) belongs to the mint family Lamiaceae, both herbs, are traditionally used in medicine due to their antimicrobial, antioxidant, and anti-inflammatory properties (Miraj and Kiani, 2016; Naderi-Farsani et al., 2019). They are also used in the poultry industry as natural growth promoters and alternatives to antibiotics, enhancing broiler performance and the immune system of birds (Khoobani et al. 2019; Khoobani et al. 2020; Alireza et al., 2021). This study aimed to investigate the effect of adding of two types of phytobiotics, coriander (Coriander sativum) and thyme (Thymus vulgaris) oils to broiler drinking water on humoral immune response and protective rate against Newcastle disease. Materials and MethodsThis experiment was conducted at the Department of Pathology and Poultry Diseases, College of Veterinary Medicine, Al-Qasim Green University. A total of 200 one-day-old broiler chicks of Ross 308 hybrid were brought from a local hatchery in Babylon province for this experiment. On the first day of the experiment, 20 chicks were randomly selected to be sacrificed to collect blood samples for serum separation to estimate the maternal antibody titer against NDV using the indirect enzyme-linked immunosorbent assay (ELISA) ProFLOK® ND ELISA Kit (Synbiotics–USA), according to the manufacturer ҆s instructions. The remaining 180 chicks were randomly divided into four groups with 45 chicks (1, 2, 3, 4), and three replicates (n=15) for each group. These chicks were reared on sawdust litter in separate pens after being equipped with all broiler breeding equipment for 35 days in good hygienic conditions. The chicks were fed ad libitum on a standard balanced nutrient ration commensurate with their age, in addition to clean drinking water throughout the experiment. The birds were fed a starter diet from 1 to 20 days and a finisher diet from day 21 to day 35, as required by national research council requirement (NRC, 1994). Chickens in the first group were vaccinated twice against NDV on days 9 and 18 only, without any water additions. Chickens in the second and third groups were vaccinated twice against NDV on days 9 and 18, respectively, in addition to 0.3 ml/l of coriander oil or thyme oil added to their drinking water, from day 1 to the last day of the experiment. The chickens of the fourth group were left unvaccinated and without supplements as a control group. The Nobilis ND live attenuated vaccine clone 30, MSD (Intervet International B.V. Boxmeer-Holland) was used for vaccination of chickens in groups 1, 2, and 3 via intraocular and intranasal routes. On day 23 of the chicks' life, blood (2 ml) was drawn from the jugular vein of the chicks in the experimental groups to determine the humoral immune response against Newcastle disease virus using the ProFLOK® ND ELISA indirect ELISA kit (Synbiotics-USA), according to the manufacturer ҆s instructions. All chicks in all experimental groups were challenged with a field isolate of NDV registered in GenBank (paramyxovirus 1 strain AAvV1-NVMoh11/Iraq/2016) on day 25 of life. Each chick was inoculated intracrop with a titer of 105 EID50/0.5 ml of the virulent NDV EID50 /Chick according to a previous study (Muhammed and Rabee, 2024). This isolate was obtained from the Department of Pathology and Poultry Diseases at the College of Veterinary Medicine/University of Karbala. After being its propagated in 9–11-day-old embryonated chicken eggs by allantoic cavity inoculation according to a previous study (Suarez et al., 2020). The chicks were monitored for 10 days post challenge to record clinical signs, morbidity, postmortem lesions, mortality rates, and survival rate. The morbidity rate was calculated by dividing the total number of sick chickens by the total population of chickens in each group post challenge. The mortality rate was calculated by dividing the number of dead chickens by the number of chickens in each group post-challenge. Statistical analysisThe trial data were analyzed using a completely randomized design, and differences between the means of the antibody titers, mortality rate, and survival rate, plus or minus the standard error, among groups were tested using analysis of variance (ANOVA), and the least significant difference test was used to detect significant differences at the 0.05 probability level. The data obtained during this study were analyzed using the one-way ANOVA test. Ethical approvalAll procedures were performed in accordance with the laws governing animal care and the guidelines on animal welfare of the Pathology Department, College of Veterinary Medicine, Al-Qasim Green University, Babylon, Iraq. The ethical criteria established by the local Animal Welfare Committee (No.2650 on 21/10/2024) were followed in the conduct of this investigation. ResultsThe results revealed that the titer of maternal antibody against NDV at 1 day of age of broiler chickens, which was measured by indirect ELISA, was 5647. The mean of antibody titer against NDV at day 23 of chick life among the experimental groups (G1, G2, G3, and G4) is summarized in Figure 1. The most pronounced and severe clinical signs were observed on chickens in Group 4, which started at 3 days post-challenge. The morbidity rate was 100%, and all chickens in this group exhibited clear symptoms, including a significant reduction in feed intake, severe depression, ruffled feathers, coughing, gasping, sneezing, and foamy mouth discharge. Conjunctivitis, swelling of the infraorbital sinuses, and watery green diarrhea were also evident. In addition to nervous signs (tremors, paralysis, and head and neck twisting). In contrast, chickens within the treatment groups (G1, G2, and G3) showed only mild clinical signs, including conjunctivitis, nasal discharge, and depression. These signs were first observed in chickens in these groups on day 5 in G1 and on day 6 in both G2 and G3 post challenge. The morbidity rate in chickens in G1 was 33% (n=15), while that in G2 and G3 was 22% (n=10) and 26% (n=12), respectively. The mortality rate among the experimental groups varied from (n=9) 20% in G1 and (n=6) 13.3% in G2 to (n=7) 15.5% in G3, and finally was (n=36) 80% in G4 as explained in Table 1. The measurement of the survival rate against challenge NDV was determined by the absence of clinical signs and mortality post-challenge, and the results are summarized in Table 2. At necropsy, the gross lesions were observed in the dead chickens in all experimental groups, but the most severe gross lesions were observed in the G4 chicks, which exhibited typical visceral lesions, similar to those observed in natural field cases from day 3 post challenge. In this group, chickens showed hemorrhagic lesions in the proventriculus (Fig. 2A), cecal tonsils (Fig. 2B), Peyer’s patches of the intestines (Fig. 2C), moderate splenomegaly with hemorrhagic spots (Fig. 2D), edematous, hemorrhagic, and congested trachea (Fig. 2E), congestion, hemorrhage, and focal areas of consolidation in the lung (Fig. 2F).
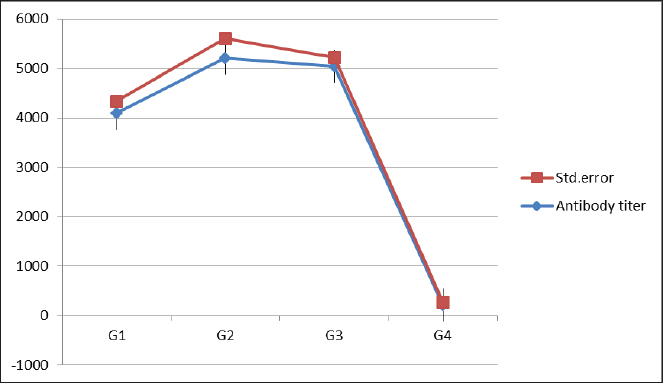
Fig. 1. Effect of phytobiotics added to broiler drinking water on ELISA antibody titer against NDV at day 23 of life among experimental groups. *G 1=The chicks were vaccinated against NDV twice on days 9 and 18 without any supplement. G 2=The chicks were vaccinated against NDV twice on days 9 and 18 with Coriander oil (0.3 ml/l) supplement in the drinking water from the first day until the end of the experiment. G 3=The chicks were vaccinated against NDV twice on days 9 and 18 with supplement Thyme oil (0.3 ml/l) in the drinking water from the first day until the end of the experiment G 4=The chicks were not vaccinated against NDV and were provided with tap water. Table 1. Effect of Phytobiotics added to broiler drinking water on mortality rate (%) among experimental groups post challenge with virulent NDV on day 25 of chicken life.
DiscussionNewcastle disease is an acute, highly contagious viral disease that causes significant health and economic problems in the poultry industry worldwide (Irshad et al., 2020; Turan et al., 2020). It could be controlled by applying strict biosecurity measure and administrated of effective vaccines (Ravikumar et al., 2022). Although numerous types of live and inactivated vaccines are used to protect poultry against ND in Iraq, disease outbreaks still occur in vaccinated broiler (Ahmed and Odisho, 2018). In the poultry industry, improving immunity is very important to prevent infectious diseases. Immunodeficiency can be induced by vaccination failure, infection by immune-suppressive diseases, and abuse of antibiotics. One solution applied in the poultry sector to improve immunity and decrease susceptibility to infectious disease is the use of immune stimulators. Deng et al. (2014) reported that herbal modulators can be administered along with a vaccine to elicit a faster and stronger immune response. Therefore, this study aimed to test the ability of two herbal (Coriander oil and thymus) as immunostimulator in inducing an immune response against NDV in broiler chicken. Table 2. Effect of phytobiotics added to broiler drinking water on survival rate (%) among experimental groups post challenge with virulent NDV on day 25 of chicken life.
Fig. 2. Explain the necropsy finding of chickens in G4 which unvaccinated and non-treated with any addition post challenge with NDV as follows: A. hemorrhagic lesions in the proventriculus. B. hemorrhagic lesions in the cecal tonsils. C. hemorrhage in Peyer’s patches of the intestines. D. moderate splenomegaly with hemorrhagic spots. E. hemorrhagic and congested trachea. F. congestion, hemorrhage, and focal areas of consolidation lung. In this study, ELISA was employed for the detection of antibodies against NDV due to ELISA is more accurate, sensitive, and rapid than other serological tests, as confirmed by (Al Sultani et al., 2025). The result indicates that the ELISA maternal derived antibody(MDA) titer in the serum of chicks at day one of age was 5647. MDA plays an important role in protecting chicks, especially during the first few weeks of age when their immune system is not yet fully functional (Hamal et al., 2006). Previous studies have found that the IgY level in the blood of chicks is high in the first days of life and then gradually decreases with age until it reaches an undetectable level on the 21st day of life (Marangons and Busani, 2006; Al-Shahery et al., 2008; Banu et al., 2009). The results of this study corresponded with our results, where the MDA titer in G4, which was unvaccinated against NDV, declined gradually from 1 day old to reach an undetectable titer on day 23 of age, based on this result, the challenge date was set at the 25th day of the life of the chicken. In this study, there were significant differences in antibody titers at 23 days of age between the treatment groups (G1, G2, and G3) and the control group (G4), as shown in Figure 1. The reason beyond this result may be that the chickens in the treatment groups were vaccinated twice at days 9 and 18 against NDV leading to the induction of humoral immune response detected by ELISA. This finding is in agreement with that (Kapczynski et al., 2013), who reported that neutralizing antibodies can be detected already 6–10 days after vaccination with live vaccines against NDV. In addition, there were significant difference in antibody titer at day 23 among G1, G2, and G3, although all chicks in these groups were vaccinated twice against NDV at the same time and same routes of vaccination. This difference may be attributed to the fact that the chicks in G2 were treated with coriander oil and those in G3 were treated with thymus oil. Both herbal oils are rich in active compounds, such as flavonoids, which extend the activity of vitamin C and, act as antioxidants to enhance immune function. On other the hand, thymol and carvacrol have been reported to have antibacterial, antiviral, and antioxidant activities, and an increase in the immune responses of chicks through the improvement mononuclear phagocyte system, cellular, and humoral immunity is expected (Botsoglou et al., 2002; Acamovic and Brooker, 2005). This explanation corresponded with Abou-Elkhair and Ahmed Selim (2014); Almremdhy (2020); Almremdhy and Al-khafaji (2020); Talazadeh and Mayahi (2017); Almremdhy and Al-Khafaji (2020a,b); Almremdhy and Al-khafaji (2020a,b) who reported the ability of coriander and thyme extract in improving the specific antibody response against various vaccines virus. In this study, the mortality in chickens exposed to challenge started from day 3 post challenge and continue for 10 days post challenge. This finding is in accordance with the finding of Hamid et al. (1991) who reported that incubation period of NDV was 2–16 days and majority of birds died between 4 and 12 days post challenge. Tables 1 and 2 show difference in the mortality rate % and survive rate % between the treatment (G1, G2, and G3) and the control group (G4). This may be attributed to the fact that vaccination of chickens with live attenuated vaccines provided adequate protection against challenge, lead to a decrease mortality rate in G1. this concurred with finding of Al-Baroodi et al., (2008); Cornax et al. (2012); Kapczynski et al. (2013); Miller et al. (2013) who referred to the control of ND based on strict biosecurity together with a proper vaccination program, which varies from country to country. In addition, we agree with Mushtaq Al-Zuhariy (2023) and Geletu and Robi (2024) who reported that eye drop multivaccination of chickens against NDV induces a greater antibody response and improved defense against the NDV challenge. In G2 and G3, may be attributed to the effect of vaccine in addition to coriander oil or thyme oil in G2 and G3, respectively. Both phytogenic are rich in thymol (5-methyl-1-2-isopropyl phenol) and carvacrol (5-isopropyl-2-methyl phenol), which possess potent antioxidant, hypolipidemic, hypocholesterolemic, and anticonvulsant properties, antibacterial activity, antiviral, antifungal and anticoccidial, and no cytotoxic effects that lead to an elevated immune response of broilers ultimately, improving general health (Crespo et al., 2019; Cross et al., 2007; Delaquis, 2002; Hagmüller et al., 2006). In the current study, the incubation period among the experimental groups ranged 3–6 days post-challenge, which this may be due to differences in immunity levels among groups. Alexander and Senne, (2008) confirmed that incubation period and clinical signs of ND depend on multiple factors, such as the virulence and dose of the virus, host species, host age, and host immunity. In the present study, the clinical signs observed in chickens post-challenge ranged from mild to severe among the experimental group. Severe clinical signs were observed in chickens in G4 (unvaccinated-challenged group) from day 3 post-challenge. All birds in this group displayed decreased feed intake, depression, ruffled feathers, sneezing, gasping, and foamy discharge in the mouth cavity, conjunctivitis, swollen infraorbital sinuses, and greenish diarrhea from day 3 post-challenge. These symptoms may be attributed to the replication of NDV and poor immunity of chickens. This result is in agreement with Sarcheshmei et al. (2016) and Ismail (2017) who found that chickens infected with virulent NDV suffer from severe clinical signs, including reduced feed intake, severe depression, ruffled feathers, and greenish diarrhea, conjunctivitis, and respiratory signs, starting from day 3 post-challenge. The clinical signs observed in chickens of the treatment groups (G1, G2, and G3) post- challenge were mild. This may be attributed to the effect of vaccination only in G1, which elicited a productive level of humoral immunity and cellular immunity to protect chickens in this group against challenge, or attributed to vaccination and phytobiotic materials in G2 and G3. This result is in agreement with Sarcheshmei et al. (2016) who showed significantly mild clinical signs in the vaccinated challenge group compared with the unvaccinated challenged group. This result concurred with Cornax et al. (2012) and Dortmans et al. (2012) who said that the lentogenic vaccine strains can provide protection against vNDV if the vaccination is properly applied. The use of phytobiotic to improve the welfare and performance of poultry has attracted increasing attention. Phytobiotics offer a range of benefits, including antimicrobial, antioxidant, immune-modulating, and growth-promoting effects leading to improvements in gut health (Nandagopal and Kumari, 2007). They enhance gut morphology, support nutrient absorption, and maintain a balanced population of beneficial bacteria while suppressing harmful pathogens (Gilani et al., 2021; Obianwuna et al., 2022; Abd El-Hack et al., 2022; Ayalew et al., 2022). The results of gross lesions in this study revealed that typical gross lesions of ND were observed in dead chickens in the control and treatment groups post-challenge. All these lesions were focused in the visceral, that indicated that the isolate, used in challenge was a viscerotropic NDV strain. However, the most severe gross lesion was observed in chickens in G4 from days 5 to 8 post challenge. The reason for this, may be that a virulent NDV replicated in the mucosal epithelium of the proventriculus. The virus then, spread via blood to the spleen and other lymphoid tissues in the intestine tract, leading to a severe inflammatory response and hyperplasia of lymphocytes in these organs, resulting in splenomegaly and swelling and haemorrhages in caecal tonsils. This interpretation is in agreement with the work of Washburn and Schirrmacher, (2002); Rue et al., (2011) who stated that vNDV induced a robust innate immune response against vNDV, leading to severe pathological damage. The same gross lesions observed in this study were revealed by (Sarcheshmei et al., 2016) who observed hemorrhagic lesions in the proventriculus, cecal tonsils, and Peyer’s patches beside edematous, hemorrhagic, and congested trachea. ConclusionFrom the results of this study, we can conclude that repeated vaccinations with live attenuated vaccines against NDV stimulate humoral immunity but do not provide complete protection against the challenge with field strains of NDV. We can also conclude that adding phytobiotics to the drinking water of broiler chickens has positive effects on raising the level of humoral immunity, increasing the level of protection against the challenge and reducing disease severity and mortality. This study recommends adding coriander oil or thyme oil to the drinking water of broiler chickens due to their positive effects on immunity and the health of the birds. AcknowledgmentsThe researchers extend their sincere thanks to the Department of Pathology and Poultry Diseases, College of Veterinary Medicine, Al-Qasim Green University, for allowing us to use the poultry house and scientific laboratories. Conflict of interestThe authors declare no conflict of interest. FundingThis research did not receive any external funding. Authors’ contributionsConceptualization and design: HA and WH; Practical work: HA, WH, and DA; formal analysis and interpretation of data: HA and DA; writing-original draft preparation: HA and WH; all authors revised and approved the final manuscript for publication. Data availabilityAll data are available. ReferencesAl-Baroodi, S.Y., Z. Al-zubeady, A., Shahery, S. and N. Al, M. 2008. Evaluation of cell-mediated immune response in chickens vaccinated with Newcastle disease virus. Iraqi. J. Vet. Sci. 22(1), 21–24. Abd El-hack, M.E., El-Saadony, M.T., Salem, H.M., El-Tahan, A.M., Soliman, M.M., Youssef, G.B.A., Taha, A.E., Soliman, S.M., Ahmed, A.E., El-Kott, A.F., Al Syaad, K.M. and Swelum, A.A. 2022. Alternatives to antibiotics for organic poultry production: types, modes of action and impacts on bird’s health and production. Poult. Sci. 101(4), 101696; doi:10.1016/j.psj.2022.101696 Abou-Elkhair, R. and Ahmed, S. Selim. 2014. Effects of Black Pepper (Piper Nigrum), Turmeric Powder (Curcuma Longa) and Coriander Seeds (Coriandrum Sativum) and Their Combinations as Feed Additives on Growth Performance, Carcass Traits, Some Blood Parameters and Humoral Immune. Asian-Australas. J. Anim. Sci. 27(6), 847–854. Acamovic, T. and Brooker, J.D. 2005. Biochemistry of plant secondary metabolites and their effects in animals. Proc. Nutr. Soc. 64, 403–412. Ahmed, A.I. and Odishosh, M. 2018. Isolation Identification and pathotyping of Newcastle Disease Viruses from Naturally Infected Chickens in Iraqi Kurdistan Region. Iraqi J. Agricult. Sci. 1(1), 132–141. Al Sultani , S.W., Al-Doori, A.A. and Samawi, F.T. 2025. Prevalence and Zoonotic Potential of Chlamydia Psittaci in Domestic Pigeons from Baghdad: eLISA and Rapid Test Insights. DJMS 1(3), 69–76; doi:10.65204/DJMS-PZPCP Alexander, D.J. and Senne, D.A. 2008. Newcastle disease, other avian paramyxoviruses, and pneumovirus infections. In Diseases of poultry. Saif, Y.M., Fadly, A.M., Glisson, J.R., McDougald, L.R., Nolan, L.K. and Swayne, D.E. 12th. Ames, IA: Blackwell Publishing Professional, pp: 75–100. Al-Habeeb, M., Mohamed, M. and Sharawi, S. 2013. Detection and characterization of Newcastle disease virus in clinical samples using real time RT-PCR and melting curve analysis based on matrix and fusion genes amplification. Vet. World. 6(5), 239–243; doi:10.5455/vetworld.2013.239-243 Al-Shahery, M.N., Al-Zubeady, A.Z. and Al-Barood, S.Y. 2008. Evaluation of cell-mediated immune response in chickens vaccinated with Newcastle disease virus. Iraqi. J. Vet. Sci. 22(1), 21–24. Ali, M.F., Abdel Hafez, M.S. and Mohamed, M.A. 2019. The ameliorating effect of some immune-stimulants on bursal and tracheal lesions of broiler chickens challenged with ND virus. JoBAZ 80(4), 2–10; doi:10.1186/s41936-019-0073-7 Alireza, S., Masomeh, T. and Marina, S. 2021. The use of some plant-derived products as effective alternatives to antibiotic growth promoters in organic poultry production: areview. Environ. Sci. Pollut. Res; doi: 10.1007/s11356-021-15460-7. Allan, W.H., Lancaster, J.E. and Toth, B. 1978. Newcastle disease vaccines-Their production and use. FAO Animal Production and Health Series No. 10. Rome, Italy: FAO. Almremdhy, H.A. 2020. The Effect Of Coriander Oil In Drinking Water Of Broiler Chickens In Growth Performance And Immune Response. Plant Arch. 20, 1999–2002. Almremdhy, H.A. and Al-khafaji, M.A. 2020. Drinking Thyme Extract on Growth Performance and Immune Response of Broiler Chickens. Indian Vet. J. 97(04), 32–35. Amoia, C.F.A.N., Nnadi, P.A., Ezema, C. and Couacy-Hymann, E. 2021. Epidemiology of Newcastle disease in Africa with emphasis on Côte d'Ivoire: a review. Vet. World. 14(7), 1727. Ayalew, H., Zhang, H., Wang, J., Wu, S., Qiu, K., Qi, G., Tekeste, A., Wassie, T. and Chanie, D. 2022. Potential feed additives as antibiotic alternatives in broiler production. Front. Vet. Sci. 9, 916473. Banu, N.A., Islam, M.S., Chowdhury, M.M.H. and Islam, M.A. 2009. Determination of immune response of Newcastle disease virus vaccines in layer chickens. J. Bangladesh. Agril. Univ. 7(2), 329–334. Bello, M.B., Yusoff, K., Ideris, A., Hair-Bejo, M., Peeters, B.P.H. and Omar, A.R. 2018. Diagnostic and vaccination approaches for Newcastle disease virus in poultry: the current and emerging perspectives. BioMed Res. Int. 2018, 7278459. Botsoglou, N.A., Florou-Paneri, P., Christaki, E., Fletouris, D.J. and Spais, A.B. 2002. Effect of dietary oregano essential oil on performance of chickens and on iron-induced lipid oxidation of breast, thigh and abdominal fat tissues. Br. Poult. Sci. 43(2), 223–230; doi:10.1080/00071660120121436 Chung, E.L.T., Algirani. and Kamalludin. 2021. Do different vaccination regimes affect the growth performance, immunestatus, carcass characteristics and meat quality of broilers?. Br. Poult. Sci. 62, 32–37. Cornax, I., Miller, P.J. and Afonso, C.L. 2012. Characterization of live LaSota vaccine strain-induced protection in chickens upon early challenge with a virulent Newcastle disease virus of heterologous genotype. Avian Dis. 56(3), 464–470; doi:10.1637/10043-122011-Reg.1 Crespo, Y.A., Bravo Sánchez, L.R., Quintana, Y.G., Cabrera, A.S.T., Bermúdez Del Sol, A. and Mayancha, D.M.G. 2019. Evaluation of the synergistic effects of antioxidant activity on mixtures of the essential oil from Apium graveolens L., Thymus vulgaris L. and Coriandrum sativum L. using simplex-lattice design. Heliyon 5(6), 1942. Cross, D.E., Mcdevitt, R.M., Hillman, K. and Acamovic, T. 2007. The effect of herbs and their associated essential oils on performance, dietary digestibility and gut microflora in chickens from 7 to 28 days of age. Br. Poult. Sci. 48(4), 496–506; doi:10.1080/00071660701463221 Delaquis, P. 2002. Antimicrobial activity of individual and mixed fractions of dill, cilantro, coriander and eucalyptus essential oils. Int. J. Food Microbiol. 74(1-2), 101–109; doi:10.1016/s0168-1605(01)00734-6 Deng, Y.Y., Yi, Y., Zhang, L.F., Zhang, R.F., Zhang, Y., Wei, Z.C., Tang, X.J. and Zhang, M.W. 2014. Immunomodulatory activity and partial characterisation of polysaccharides from Momordica charantia. Molecules 19(9), 13432–13447; doi:10.3390/molecules190913432 Dixon, L.M. 2020. Slow and steady wins the race: the behaviour and welfare of commercial faster growing broiler breeds compared to a commercial slower growing breed. PLos One. 15(4), e0231006; doi:10.1371/journal.pone.0231006 Dortmans, J.C.F.M., Peeters, B.P.H. and Koch, G. 2012. Newcastle disease virus outbreaks: vaccine mismatch or inadequate application?. Vet. Microbiol. 160(1-2), 17–22; doi:10.1016/j.vetmic.2012.05.003 Geletu, A.S. and Robi, D.T. 2024. Evaluation of the immune response of layer chickens to Newcastle disease virus vaccines using the new vaccination regimens. Vet. Med. Sci. 10(3), 1428; doi:10.1002/vms3.1428 Gessner, D.K., Ringseis, R. and Eder, K. 2017. Potential of plant polyphenols to combat oxidative stress and inflammatory processes in farm animals. J. Anim. Physiol. Anim. Nutr. (Berl). 101(4), 605–628; doi:10.1111/jpn.12579 Getabalew M Alemneh., Akeberegn, T., Getahun, D. and Zewdie, D. 2019. Epidemiology, Diagnosis & Prevention of Newcastle disease in poultry. Am. J. Biomed. Sci. Res. 16, 50–59. Gilani, S.M.H., Rashid, Z., Galani, S., Ilyas, S., Sahar, S., Zahoor-Ul-Hassan., Al-Ghanim, K., Zehra, S., Azhar, A., Al-Misned, F., Ahmed, Z., Al-Mulham, N. and Mahboob, S. 2021. Growth performance, intestinal histomorphology, gut microflora and ghrelin gene expression analysis of broiler by supplementing natural growth promoters: a nutrigenomics approach. Saudi J. Biol. Sci. 28(6), 3438–3447; doi:10.1016/j.sjbs.2021.03.008 Hafez, H.M. and Attia, Y.A. 2020. Challenges to the Poultry Industry: current Perspectives and Strategic Future After the COVID-19 Outbreak. Front. Vet. Sci. 7, 516; doi:10.3389/fvets.2020.00516 Hagmüller, W., Jugl-Chizzola, M., Zitterl-Eglseer, K., Gabler, C., Spergser, J., Chizzola, R. and Franz, C. 2006. The use of Thymi Herba as feed additive (0.1%, 0.5%, 1.0%) in weanling piglets with assessment of the shedding of haemolysing E. coli and the detection of thymol in the blood plasma. Berl. Munch. Tierarztl. Wochenschr. 119(1-2), 50–54. Hamal, K.R., Burgess, S.C., Pevzner, I.Y. and Erf, G.F. 2006. Maternal antibody transfer from dams to their egg yolks, egg whites, and chicks in meat lines of chickens. Poult. Sci. 85(8), 1364–1372; doi:10.1093/ps/85.8.1364 Hamid, H., Campbell, R.S.F. and Parede, L. 1991. Studies of the pathology of velogenic Newcastle disease: virus infection in non-immune and immune birds. Avian. Pathol. 20(4), 561–575; doi:10.1080/03079459108418796 International Committee on Taxonomy of Viruses (ICTV). 2023. Current ICTV taxonomy release. Available online: https://ictv.global/taxonomy (accessed on 16 May 2023). Irshad, I., Aslam, A., Tipu, M.Y., Ashraf, K., Zahid, B. and Wajid, A. 2020. Pathotyping and genetic characterization of Avian Avulavirus-1 and low pathogenicity H9N2 avian influenza viruses isolated from Punjab, Pakistan. Pak. J. Zool. 52(1), 1–5; doi:10.17582/journal.pjz/2020.52.1.1.5 Ismail, H.T.H. 2017. Biochemical and Hematological Studies on the Effect of Neem (Azadirachta Indica) Leaves Aqueous Extract on Newcastle Disease Vaccine and Infection in Broiler Chickens. Int. J. Recent Sci. Res. 8(3), 15876–15884; doi:10.24327/ijrsr.2017.0803.0002 Kapczynski, D.R., Afonso, C.L. and Miller, P.J. 2013. Immune responses of poultry to Newcastle disease virus. Dev. Comp. Immunol. 41(3), 447–453; doi:10.1016/j.dci.2013.04.012 Khalifa, K.A., Abedlrahim, E.S., Elnasri, I.M. and Ahmed, S.O. 2013. A retrospective study (2000-2005) of poultry diseases diagnosed at department of avian diseases and diagnosis, veterinary research institute (VRI)-Khartoum. Sudanese. J. Anim. Sci. 9(7), 42–45. Khoobani M, Hasheminezhad SH, Javandel F, Nosrati M, Seidavi A, Kadim IT, Laudadio V and Tufarelli V. 2019. Effects of dietary chicory (Chicorium intybusL.) and probiotic blend as natural feed additives on performance traits, blood biochemistry, and gut microbiota of broiler chickens. Antibiotics (Basel). 9(1), 5; doi: 10.3390/antibiotics9010005 Lai, L.P., Lee, M.T., Chen, C.S., Yu, B. and Lee, T.T. 2015. Effects of co-fermented Pleurotus eryngii stalk residues and soybean hulls by Aureobasidium pullulans on performance and intestinal morphology in broiler chickens. Poult. Sci. 94(12), 2959–2969; doi:10.3382/ps/pev302 Lin, M.J., Chang, S.C., Jea, Y.S., Chen, W.S. and Lee, T.T. 2015. Effects of dietary garlic scape meal on the growth and meat characteristics of geese. Br. Poult. Sci. 56(6), 716–722; doi:10.1080/00071668.2015.1096012 Lin, M.J., Chang, S.C., Jea, Y.S., Liao, J.W., Fan, Y.K. and Lee, T.T. 2016. In vitro antioxidant capability and performance assessment of White Roman goose supplemented with dried ToonaSinensis. J. Appl. Anim. Res. 44(1), 359–402; doi:10.1080/09712119.2015.1091342 Lin, W.C., Lee, M.T., Chang, S.C., Chang, Y.L., Shih, C.H., Yu, B. and Lee, T.T. 2017. Effects of mulberry leaves on production performance and the potential modulation of antioxidative status in laying hens. Poult. Sci. 96(5), 1191–1203; doi:10.3382/ps/pew350 Lin, W.C., Lee, M.T., Lo, C.T., Chang, S.C. and Lee, T.T. 2018. Effects of dietary supplementation of Trichoderma pseudokoningii fermented enzyme powder on growth performance, intestinal morphology, microflora and serum antioxidantive status in broiler chickens. Ital. J. Anim. Sci. 17(1), 153–164; doi:10.1080/1828051X.2017.1355273 Maharjan P., Martinez D.A., Weil J., Suesuttajit N., Umberson, C., Mullenix, G., Hilton, K.M., Beitia A. and HYPERLINK "https://www.sciencedirect.com/author/7006410045/craig-nelson-coon" Coon, C.N. 2021. Physiological growth trend of current meat broilers and dietary protein and energy management approaches for sustainable broiler production. Animal 15(3), 100284; doi:10.1016/j. animal.2021.100284 Mansour, S.M.G., Elbakrey, R.M., Mohamed, F.F., Hamouda, E.E., Abdallah, M.S., Elbestawy, A.R., Ismail, M.M., Abdien, H.M.F. and Eid, A.A.M. 2021. Avian Paramyxovirus Type 1 in Egypt: epidemiology, Evolutionary Perspective, and Vaccine Approach. Front. Vet. Sci. 8, 647462; doi:10.3389/fvets.2021.647462 Marangon, S. and Busani, L. 2006. The use of vaccination in poultry production. Rev. Sci. Tech. 26(1), 265–274. Miller, P.J. and Koch, G. 2013. Newcastle disease. In: Swayne, D.E., Glisson, J.R., McDougald, L.R., Nolan, L.K., Suarez, D.L. and Nair, V.L. editors. Diseases of poultry. 13th edition. Ames, IA:Wiley-Blackwell in partnership with the American Association of Avian Pathologists, pp: 89 -107. Miraj, S. and Kiani, S. 2016. Study of pharmacological effect of Thymus vulgaris: a review. Pharm. Lett. 8(9), 315–320. Muhammed, Q.O. and Rabee, R.H.S. 2024. Assessment of Oil Vaccination Protection to Newcastle Disease Virus Challenges in Layer. J. Anim. Health Prod. 12(Spl.1), 351–356; doi:10.17582/journal.jahp/2024/12.s1.351.356 Mushtaq T. B. Al-Zuhariy. 2023. Evalution of the best vaccinal routes against Newcastle in the production stage of laying hens. Iraqi. J. Agric. Sci. 54(3), 748–754; doi:10.36103/ijas.v54i3.1757 Naderi Farsani, M., Hoseinifar, S.H., Rashidian, G., Ghafari Farsani, H., Ashouri, G. and Van Doan, H. 2019. Dietary effects of Coriandrum sativum extract on growth performance, physiological and innate immune responses and resistance of rainbow trout (Oncorhynchus mykiss) against Yersinia ruckeri. Fish. Shellfish. Immunol. 91, 233–240; doi:10.1016/j.fsi.2019.05.031 Nandagopal, S. and Kumari, B.R. 2007. Phytochemical and antibacterial studies of Chicory (Cichorium intybus L.)-a multipurpose medicinal plant. Adv. Biol. Res. 1(1-2), 17–21. Nrc. 1994. Nutrient Requirements of Poultry. Obianwuna, U.E., Agbai Kalu, N., Wang, J., Zhang, H., Qi, G., Qiu, K. and Wu, S. 2023. Recent Trends on Mitigative Effect of Probiotics on Oxidative-Stress-Induced Gut Dysfunction in Broilers under Necrotic Enteritis Challenge: a Review. Antioxidants 12(4), 911; doi:10.3390/antiox12040911 Obianwuna, U.E., Oleforuh-Okoleh, V.U., Wang, J., Zhang, H.J., Qi, G.H., Qiu, K. and Wu, S.G. 2022. Potential Implications of Natural Antioxidants of Plant Origin on Oxidative Stability of Chicken Albumen during Storage: a Review. Antioxidants (Basel). 11(4), 630; doi:10.3390/antiox11040630 Ravikumar, R., Chan, J. and Prabakaran, M. 2022. Vaccines against Major Poultry Viral Diseases: strategies to Improve the Breadth and Protective Efficacy. Viruses 31(6), 1195; doi:10.3390/v14061195 Rue, C.A., Susta, L., Cornax, I., Brown, C.C., Kapczynski, D.R., Suarez, D.L., King, D.J., Miller, P.J. and Afonso, C.L., 2011. Virulent Newcastle disease virus elicits a strong innate immune response in chickens. J. Gen. Virol. 92, 931–939. Sarcheshmei, M., Dadras, H., Mosleh, N. and Mehrabanpour, M.J. 2016. Comparative Evaluation of The Protective Efficacy of Different Vaccination Programs Against a Virulent Field Strain of the Newcastle Disease Virus in Broilers. Braz. J. Poult. Sci. 18(3), 363–370; doi:10.1590/1806-9061-2015-0128 Seidavi, A., Tavakoli, M., Slozhenkina, M., Gorlov, I., Hashem, N.M., Asroosh, F., Taha, A.E., Abd El-Hack, M.E. and Swelum, A.A. 2021. The use of some plant-derived products as effective alternatives to antibiotic growth promoters in organic poultry production: a review. Environ. Sci. Pollut. Res. Int. 28(35), 47856–47868; doi: 10.1007/s11356-021-15460-7. Shan, S., Bruce, K., Stevens, V., Wong, F., Wang, J., Johnson, D., Middleton, D., O’Riley, K., Mccullough, S., Williams, D. and Bergfeld, J. 2021. In Vitro and In Vivo Characterization of a Pigeon Paramyxovirus Type 1 Isolated from Domestic Pigeons in Victoria, Australia 2011. Viruses 13(3), 429; doi:10.3390/v13030429 Suarez, D.L., Miller, P.J., Koch, G., Mundt, E. and Rautenschlein, S. 2020. HYPERLINK "https://doi.org/10.1002/9781119371199.ch3" Newcastle disease, other avian paramyxoviruses, HYPERLINK "https://doi.org/10.1002/9781119371199.ch3" and avian metapneumovirus infections. In: Diseases of poultry. Eds., Swayne, D.E., Boulianne, M., Logue, C.M., McDougald, L.R. 14th ed. NJ: Wiley Blackwell, pp: 109–166; doi:10.1002/9781119371199.ch3 Suresh, G., Das, R.K., Kaur Brar, S., Rouissi, T., Avalos Ramirez, A., Chorfi, Y. and Godbout, S. 2018. Alternatives to antibiotics in poultry feed: molecular perspectives. Crit. Rev. Microbiol. 44(3), 318–335; doi:10.1080/1040841X.2017.1373062 Talazadeh, F. and Mayahi, M. 2017. Immune response of broiler chickens supplemented with pediatric cough syrup including thyme extract in drinking water against influenza vaccine. J. Herbmed. Pharmacol. 6(1), 33–36. Teng, P.Y., Chung, C.H., Chao, Y.P., Chiang, C.J., Chang, S.C., Yu, B. and Lee, T.T. 2017. Administration of Bacillus Amyloliquefaciens and Saccharomyces Cerevisiae as Direct-Fed Microbials Improves Intestinal Microflora and Morphology in Broiler Chickens. J. Poult. Sci. 54(2), 134–141; doi:10.2141/jpsa.0160069 Turan, N., Ozsemir, C., Yilmaz, A., Cizmecigil, U.Y., Aydin, O., Bamac, O.E., Gurel, A., Kutukcu, A., Ozsemir, K., Tali, H.E., Tali, B.H., Yilmaz, S.G., Yaramanoglu, M., Tekelioğlu, B.K., Ozsoy, S., Richt, J.A., Iqbal, M. and Yilmaz, H. 2020. Identification of Newcastle disease virus subgenotype VII.2 in wild birds in Turkey. BMC. Vet. Res. 16(1), 277; doi:10.1186/s12917-020-02503-3 Wajid, A., Basharat, A., Bibi, T. and Rehmani, S.F. 2018. Comparison of protection and viral shedding following vaccination with Newcastle disease virus strains of different genotypes used in vaccine formulation. Trop. Anim. Health Prod. 50(7), 1645–1651; doi:10.1007/s11250-018-1607-6 Wang, J., Liu, H., Liu, W., Zheng, D., Zhao, Y., Li, Y., Wang, Y., Ge, S., Lv, Y., Zuo, Y., Yu, S. and Wang, Z. 2015. Genomic characterizations of six pigeon paramyxovirus type 1 viruses isolated from live bird markets in China during 2011 to 2013. PLos One. 10(4), 124261; doi:10.1371/journal.pone.0124261 Washburn, B. and Schirrmacher, V. 2002. Human tumor cell infection by Newcastle Disease Virus leads to upregulation of HLA and cell adhesion molecules and to induction of interferons, chemokines and finally apoptosis. Int. J. Oncol. 21, 85–93. | ||
| How to Cite this Article |
| Pubmed Style Almremdhy HAA, Aljuhashi DA. Effect of phytobiotics on humoral immune response and survival rate against avian Avulavirus challenge in broiler chickens. Open Vet. J.. 2026; 16(2): 1151-1161. doi:10.5455/OVJ.2026.v16.i2.33 Web Style Almremdhy HAA, Aljuhashi DA. Effect of phytobiotics on humoral immune response and survival rate against avian Avulavirus challenge in broiler chickens. https://www.openveterinaryjournal.com/?mno=279668 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.33 AMA (American Medical Association) Style Almremdhy HAA, Aljuhashi DA. Effect of phytobiotics on humoral immune response and survival rate against avian Avulavirus challenge in broiler chickens. Open Vet. J.. 2026; 16(2): 1151-1161. doi:10.5455/OVJ.2026.v16.i2.33 Vancouver/ICMJE Style Almremdhy HAA, Aljuhashi DA. Effect of phytobiotics on humoral immune response and survival rate against avian Avulavirus challenge in broiler chickens. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1151-1161. doi:10.5455/OVJ.2026.v16.i2.33 Harvard Style Almremdhy, H. A. A. & Aljuhashi, . D. A. (2026) Effect of phytobiotics on humoral immune response and survival rate against avian Avulavirus challenge in broiler chickens. Open Vet. J., 16 (2), 1151-1161. doi:10.5455/OVJ.2026.v16.i2.33 Turabian Style Almremdhy, Hayder Abd Al-emier, and Dheyae Ali Aljuhashi. 2026. Effect of phytobiotics on humoral immune response and survival rate against avian Avulavirus challenge in broiler chickens. Open Veterinary Journal, 16 (2), 1151-1161. doi:10.5455/OVJ.2026.v16.i2.33 Chicago Style Almremdhy, Hayder Abd Al-emier, and Dheyae Ali Aljuhashi. "Effect of phytobiotics on humoral immune response and survival rate against avian Avulavirus challenge in broiler chickens." Open Veterinary Journal 16 (2026), 1151-1161. doi:10.5455/OVJ.2026.v16.i2.33 MLA (The Modern Language Association) Style Almremdhy, Hayder Abd Al-emier, and Dheyae Ali Aljuhashi. "Effect of phytobiotics on humoral immune response and survival rate against avian Avulavirus challenge in broiler chickens." Open Veterinary Journal 16.2 (2026), 1151-1161. Print. doi:10.5455/OVJ.2026.v16.i2.33 APA (American Psychological Association) Style Almremdhy, H. A. A. & Aljuhashi, . D. A. (2026) Effect of phytobiotics on humoral immune response and survival rate against avian Avulavirus challenge in broiler chickens. Open Veterinary Journal, 16 (2), 1151-1161. doi:10.5455/OVJ.2026.v16.i2.33 |