| Research Article | ||
Open Vet. J.. 2026; 16(3): 1823-1836 Open Veterinary Journal, (2026), Vol. 16(3): 1823-1836 Research Article PUFA-rich oil supplement improves hair condition of cats and dogsXuan Cai1*, Chunhong Xiong2, Lihui Zhu1, Yanning Zhang3, Hui Mao1, Qidong Sun4 and Yonghong Lu11Institute of Animal Science and Veterinary Medicine, Shanghai Academy of Agricultural Sciences, Shanghai, China 2HAID Group Co, Ltd., Weihai, China 3Beijing MZZ Tech. Co. Ltd, Beijing, China 4Zhejiang Small Animal Protection Association, Hangzhou, China *Corresponding Author: Xuan Cai. Institute of Animal Science and Veterinary Medicine, Shanghai Academy of Agricultural Sciences, China. Email: caixuan1985911 [at] 163.com Submitted: 01/09/2025 Revised: 12/02/2026 Accepted: 22/02/2026 Published: 31/03/2026 © 2025 Open Veterinary Journal
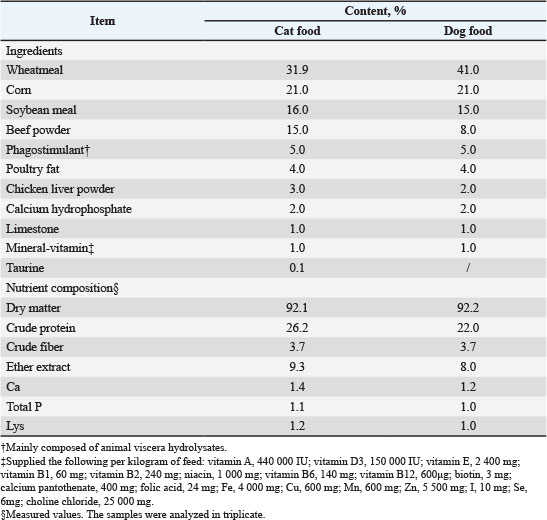
AbstractBackground: People are increasingly attentive to the quality of the hair of pets. Supplementation with polyunsaturated fatty acid (PUFA) may influence pet hair condition. Aim: This study focuses on examining the effect of PUFA-rich oil supplementation on hair improvement and overall health in cats and dogs. Methods: We investigated the effects of PUFA supplementation on hair improvement using a mixture of Hoki oil, flax seed oil, lecithin, mussel oil, and sunflower seed oil. The composition of fatty acids in the oil was determined by the capillary gas chromatography method. The ω-3 to ω-6 ratio in oil for dogs is 1:4.7, and 1:0.9 for cats. A total of 10 healthy adult cats and 12 healthy adult dogs were enrolled in this 168-day (84 days each) cross-over design trial. At the end of each period, serum was obtained from the animals via anterior limb vein blood collection for testing of blood lipids, antioxidant capacity, and inflammation-related factors. Body weight, fecal scoring, and hair condition were also recorded in this test. Results: Dog food (0.70 ml/kg BW) and cat food (0.75 ml/kg BW) high PUFA oil supplementation improved flexibility, brightness, and skin condition scores in both cats and dogs (p < 0.05). Additionally, the breaking force of hair increased by 5.79% in cats and 8.68% in dogs, respectively. The images acquired by scanning electron microscopy (SEM) show that the mechanism may be related to alterations in the microstructure of hair. This study also showed that moderate PUFA supplementation did not result in weight gain or changes in fecal scoring (p > 0.05); however, triglyceride levels increased by 13.16% in cats and by 33.33% in dogs (p < 0.05), while the levels of tumor necrosis factor-alpha/TNF-ɑ significantly decreased (p < 0.05). Conclusion: PUFA supplementation effectively enhances hair quality in companion animals but may elevate serum triglyceride levels. Thus, dosage and fatty acid composition in dietary formulations should be carefully considered. Keywords: Cat, Dog, Hair, Lipid, Polyunsaturated fatty acid. IntroductionPets, commonly referred to as companion animals, primarily serve as sources for human emotional fulfillment through companionship. Consequently, attributes such as 'beauty' associated with pets garner significant attention from society at large. To align with consumers’ preferences, several dog or cat food products claim to possess “beauty properties”. Hair represents one aspect that owners most frequently observe regarding their pets' external appearance; thus in recent years, there have been numerous nutritional supplements specifically featuring the improvement of hair quality (Guo et al., 2022; Yuan et al., 2023). Due to cats and dogs inability to effectively synthesize long-chain omega-3 fatty acids (Dominguez et al., 2021), many pet foods fail to achieve optimal levels primarily because of factors such as cost-effectiveness or preservation issues (Dominguez et al., 2021). As a result, fish oils or other ingredients rich in long-chain omega-3 content have become common nutritional supplements intended for enhancing pet fur quality (Wyrostek et al., 2023; Zhang et al., 2025). Unfortunately, even though such commercial practices are prevalent, the relationship between food nutrition and hair quality of dogs and cats is still finite. Moreover, lipids are important energy sources in pet feed, and additional lipid supplementation can increase feed energy, considering that 26% of dogs and 40 % of cats are classified as overweight or obese (Montoya et al., 2025). Feeding management plays a crucial role in weight control among cats and dogs (Spence, 2022). Some studies have indicated that elevated dietary fats may induce weight gain, particularly among cats (Cline et al., 2021; Montoya et al., 2025); thus, questions have been raised regarding the necessity and benefits of oil supplementation in commercial dog and cat foods. On the other hand, palatability is also a crucial factor in the selection of pet nutritional supplements. Pet food manufacturers have reported that the palatability of fish oil for dogs and cats is inferior to that of other animal lipids, which is consistent with the findings of our previous trial (data not shown). However, some studies have reported conflicting results (Li et al., 2020). Therefore, adopting a blended lipid approach to enhance palatability represents a novel strategy when selecting long-chain omega-3 supplements for dogs and cats. Several studies have demonstrated that diets supplemented with polyunsaturated fatty acids (PUFAs) not only enhance the coat condition of companion animals (Richards et al., 2023; Yuan et al., 2025) but also correlate with eicosapentaenoic acid concentrations in plasma, erythrocyte membranes, and semen (Pellegrino et al., 2023). Additionally, such supplementation can modulate gut microbiota composition (Ephraim et al., 2022), improve metabolic profiles—particularly hepatic function (Jamikorn and Kongpanna, 2025), and boost cognitive performance (Rodrigues et al., 2023). However, few studies have simultaneously addressed both hair quality and pet obesity. Therefore, this study focuses on testing the hair improvement effect of oil-rich PUFA supplementation in cats and dogs. The changes in body weight and blood lipid levels are also recorded in this test to evaluate whether appropriate supplementation of high omega-3 long-chain fatty acids in puffed pet food contributes to the well-being of pets regarding their coat condition and overall health. Materials and MethodsOil and dietThe basic diet of cats and dogs comprises wheatmeal, corn, beef powder, chicken fat, and so on (Table 1). This constituted a conventional expanded diet processed by HAID Group Co. Ltd. (Weihai, China). Both diets fulfilled the nutritional requirements established for complete feeds of both cats and dogs (FEDIAF, 2025). Table 1. Composition and nutrient content of the as-fed basal diet.
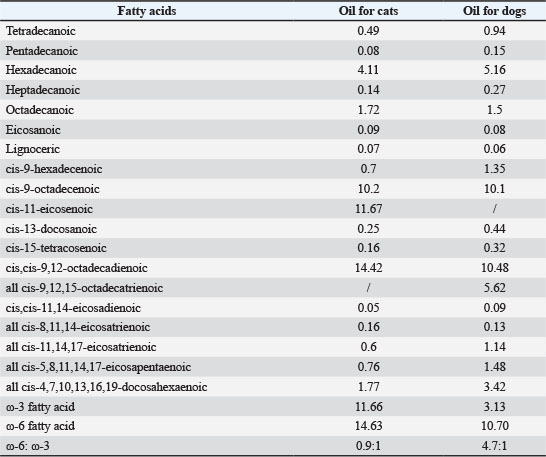
Provided by Beijing MZZ Tech. Co. Ltd., the oil was composed of Hoki oil, flax seed oil, lecithin, mussel oil, and sunflower seed oil. The composition of fatty acids in the oil was determined by capillary gas chromatography (ISO 5508/5509). Experimental design, animal control, and managementTwelve healthy dogs (6 males and 6 females) aged approximately 3.0 ± 0.2 years with a body weight (5.18 ± 0.41) kg were selected from Shanghai Academy of Agricultural Sciences for this study. The information on the dogs used in this study is shown in Supplementary Materials 1. All dogs were housed individually in 6 m2 indoor kennels with concrete floors, and the temperature within the kennels was maintained between 17°C and 27°C during the test. During 8:00–16:00, they had access to outdoor kennels, depending on the weather conditions, or they walked individually. The kennels were cleaned daily. All institutional and national guidelines regarding the care and use of animals were strictly followed. Ten healthy cats (5 males and 5 females), aged 1.2 ± 0.1 with a body weight of (3.37 ± 0.23) kg, were selected from Shanghai Academy of Agricultural Sciences. Detailed information on the cats used in this study is provided in Supplementary Materials 1. From 16:00 to 08:00 (a total of 16 consecutive hours), the cats were housed individually in cages measuring (0.75 m × 0.75 m × 1.60 m) equipped with plush cushions and litter boxes, and had access to both food and water. Between 08:00 and 12:00 or 12:00 and 16:00, the cats resided in a collective cattery measuring (5.0 m × 5.0 m × 4.5 m) that included cat scratchers and climbers for exercise and social interaction. In this setting, the cats had access to water but not food (for 4 consecutive hours for every 5 cats). The ambient temperature within the kennels was kept at (25.0 ± 3.0)°C throughout the test, and cat litters were replaced every 3 days. All procedures were performed in accordance with the institutional and national guidelines for the care and use of animals. Experimental designThe experiment spanned 28 weeks, during which dogs/cats were randomly assigned to 2 groups according to a crossover design. Prior to the commencement of the experiment, the animals underwent a 1-week adaptive feeding period in their respective kennels/cages. All animals were fed twice daily at designated times (9:00 and 16:00, respectively). The dogs were fed a 200–g basal diet, whereas the cats were fed a 50–g basal diet in the morning (9:00). The dogs and cats were fed a 300 g basal diet and a 30–g basal diet in the afternoon (16:00). If any food remained in the cats’ bowls from the morning feeding, no additional food was replenished in the afternoon, and the remaining morning food was removed. The oil was sprayed onto the food surface based on the body weight of the animals at 9:00 a.m. In particular, 0.70 ml/kg body weight (BW) oil was incorporated into dog food, whereas 0.75 ml/kg BW oil was incorporated into cat food. In most cases, dogs/cats are capable of consuming greasy food provided in the morning prior to afternoon feeding. Any remaining food was discarded by the breeder. During the initial 12 weeks of the study, half of the dogs/cats were fed a diet supplemented with oil, whereas the others received a basal diet. On day 84, the dietary regimens of the 2 groups were crossed over in accordance with the crossover trial design. Weighing measurements were conducted on three occasions, i.e., the first (Day 1), midpoint (Day 84), and final (Day 168) of the experiment. Sample collectionBlood and hair samples were collected at each assessment stage (Days 84 and 168) of the experiment. At these stages, 2 ml of blood was drawn via the jugular vein at 8:30 prior to feeding. The blood sample was immediately divided into two aliquots. One was kept in a tube containing Ethylene Diamine Tetraacetic Acid for routine hematological analysis within 1 hour; the other was stored at 4℃ for 8 hours, centrifuged at 1,500 g for 15 minutes at 4℃, and stored at 20℃ until further analysis. Hair samples were collected by using an electric shaver to shave a 1 cm × 3 cm area on one side of the midscapular region. Fecal scoringIn this study, fecal samples were scored to assess changes in stool quality before and at the end of the oil-supplemented diet. The subjective quality parameters of fresh feces were assessed according to “The Waltham® Feces Scoring System”: 1=hard, dry, and crumbly; 2=well formed, does not leave a mark when picked up; 3=moist at the beginning and then losing its form, leaving a definite mark when picked up; 4=the majority, if not all forms are lost; 5=watery diarrhea. Blood analysesBlood samples without anticoagulant were kept at 4°C overnight and subsequently centrifuged at 3,000 g at room temperature for 10 minutes to obtain serum. The serum levels of total cholesterol and triglyceride were analyzed using a clinical chemistry analyzer (Catalyst 1, IDEXX Laboratories, USA). Antioxidant indexes, including superoxide dismutase/SOD activities and malondialdehyde/MDA content, were assessed using commercial kits from Nanjing Jiancheng Bioengineering Institute, Nanjing, China, according to the manufacturer’s protocols. Inflammation-related indicators, such as tumor necrosis factor-alpha/TNF-α, Interleukin-1/IL-1, were analyzed using commercially available enzyme-linked immunosorbent assay methods (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). Hair analysesHair condition scoreThe cats and dogs were washed with warm water (without any cleaner) and then dried using a pet drier. The hair and skin condition of cats and dogs at the end of each stage was scored by 5 professional breeders who were blinded to the study protocol—these scorers were not involved in daily feeding procedures and had no knowledge of the group allocation (Guo et al., 2022). All samples were scored in 3 increments using the following scale: Hair brightness: 1=dull, 2=medium reflective, and 3=bright Hair softness: 1=coarse, 2=medium-soft, and 3=very soft. Skin conditions: 1=dry, 2=medium greasy, and 3=very greasy. Physio-mechanical analysis of the hairThe diameter and physio-mechanical parameters of the hair, including the breaking force and maximum tensile rate, were determined. The hair diameter was assessed using an ordinary optical microscope equipped with a scale. The breaking force and maximum tensile rate of 10 randomly selected hairs were measured using a fiber strength tester (Laizhou, Shandong, China), with each test conducted on a 5 mm hair. Hair finenessAccording to the method of Zhang et al. (2025), hair fineness was measured using light microscopy. Ten randomly selected hairs from each animal were affixed to a glass slide using transparent adhesive tape. Then, the fineness of the hair was measured using length measurement software accompanied by a microscope. The final results were averaged and reported in micrometers (μm). SEMHistological evaluation was conducted using an FEI Quanta 250 scanning microscope (FEI Company, Czech Republic). The samples were pretreated according to the protocols of Roman et al. (2022). On the basis of the SEM images, various parameters were determined, such as the arrangement of scales in relation to the longitudinal axis of hair, the type of cuticle, the structure of the cuticle edges, the distance between the cuticle edges, and the structure of the cuticle edges. Statistical analysesAll data are expressed as mean ± standard error of the mean and analyzed using the Statistical Package for the Social Sciences version 17.0 software. Shapiro–Wilk tests were used to verify whether the samples followed a normal distribution. If confirmed, the statistical significance for regression coefficients was evaluated via paired two-tailed t-tests at a significance level set at 0.05. Ethical approvalThe authors confirm adherence to the ethical policies outlined on the author guidelines page of the journal and receipt of appropriate approval from an ethical review committee for this study. The authors affirm compliance with Chinese standards for protecting animals used for scientific purposes and feed legislation. The Ethics Committee of Shanghai Academy of Agricultural Sciences approved this study (authorization number: SAASPZ0522044). ResultsFatty acid profile of oilThe composition of the fatty acids of oil is presented in Table 2. Both oils used in this study exhibited a high content of unsaturated fatty acids: the ratio of unsaturated fatty acids in oil for dogs is 40.74%, and that for cats is 34.57%. The ω-3 fatty acid content in oil for dogs is 3.13%, compared with 11.66% for cats. Conversely, the ω-6 fatty content in oil for dogs is 14.63%, and that for cats is 10.70%. The ω-3 to ω-6 ratio in oil for dogs is 1:4.7, and 1:0.9 for cats. Table 2. Composition of oil fatty acids in this study Unit: %.
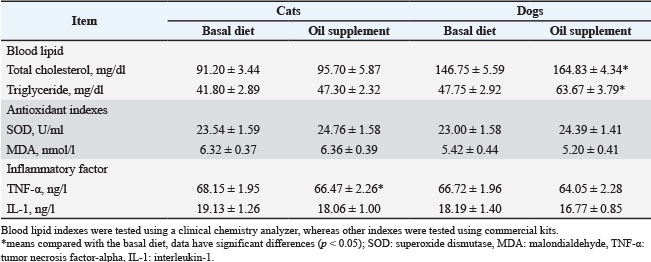
Oil supplementation alters the biochemical parameters of the serum of petsAs illustrated in Table 3, the study found no significant change in the total cholesterol levels among cats, whereas an increase was observed in dogs. Similarly, the triglyceride levels in cats did not change significantly, whereas those in dogs increased (p < 0.05). The total cholesterol and trigly ceride levels in the serum of all animals fed either a basal diet or an 84-day oil supplemented diet, were within normal range, except for one cat (data not shown in Table 3). Oil supplement resulted in the trend that increased serum superoxide dismutase (SOD) activity in both cats and dogs, although this effect did not reach statistical significance (0.05 < p < 0.1), while no effect was noted on MDA content in serum (p > 0.1). Tumor necrosis factor-alpha (TNF-α) and Interleukin (IL-1) are both proinflammatory cytokines. Table 3 showed that oil supplement significantly decreased TNF-α concentrations in the serum of both cats and dogs (p < 0.05), and no effect on IL-1 concentrations (p > 0.1). Table 3. Influence of oil supplementation on serum biochemical parameters in cats and dogs.
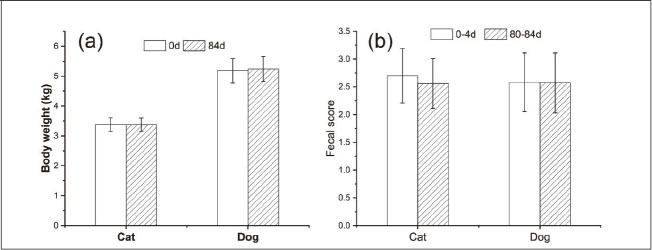
Oil supplementation does not alter body weight and fecal quality but increases blood lipid levels in petsAs depicted in Fig. 1a, there was no significant increase in body weight among animals within the 84-day oil supplementation period (p > 0.05). The fecal quality also did not show any alterations when the animals were provided with food supplemented with oil (Fig. 1b, p > 0.05). The fecal scores of cats and dogs fed either a basal diet or an oil supplement diet ranged from 2.5 to 3.0, which was considered to be an ideal stool firmness level.
Fig. 1. Effect of oil supplementation on (a) body weight and (b) fecal quality of cats and dogs. Oil supplementation enhances the sensory evaluation of cat and dog hairAs illustrated in Figure. 2, the 84-day oil supplementation significantly enhanced the hair flexibility, brightness, and skin condition of both cats and dogs (p < 0.01). Photos of cats and dogs are presented in Supplementary Materials 2.
Fig. 2. Hair and skin scores of cats (a) and dogs (b). *Significant difference compared with the basal diet (p < 0.05). Physio-mechanical properties of cat and dog hairFig. 3 shows the breaking force and maximum tensile rate of the hair from cats and dogs. As depicted in Fig. 3, the hair breaking force of cats (Fig. 3a) and dogs (Fig. 3b) exhibited a significant increase after 84 days of oil supplementation (p < 0.05). Specifically, the hair breaking force of cats increased by 5.79%, while that of dogs increased by 8.68%. respectively. However, no significant changes were observed in the maximum tensile rate (Fig. 3c and 3d) after the 84-day oil supplementation (p> 0.05).
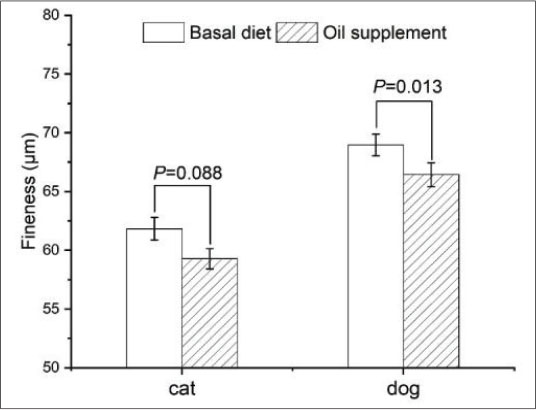
Oil supplementation decreases fineness of hair in cats and dogsHair fineness is associated with its smoothness; typically, finer hair tends to feel softer. As shown in Figure. 4, with oil supplementation, the hair fineness decreased by 4.12% in cats (p=0.088) and by 3.52% in dogs (p=0.013).
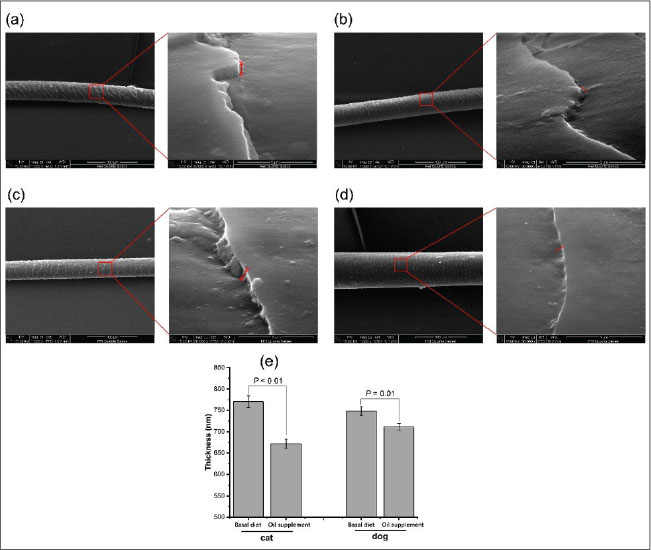
Fig. 4. Fineness of the hair of cats and dogs. Oil supplementation decreases the scale thickness of the hair of both cats and dogsAs shown in Fig. 5, at the end of the study, the scale thickness was reduced in cats (Fig. 5b) and dogs (Fig. 5d) compared with those fed with the basal diet only (Fig. 5a and 5c). Statistical analysis revealed a significant decrease in the scale thickness (Fig. 5e, p < 0.05). In addition, a reduction in scale height was noted for the hair of cats and dogs during the period when those animals were fed with oil-supplemented diets. However, data collection on low-magnification images was insufficient.
DiscussionThe beneficial effect of polyunsaturated supplementation on animal hair has been investigated in numerous studies (Kirby et al., 2009; Guo et al., 2022). The current study confirmed that oils rich in PUFA could improve the hair condition of cats and dogs, while also protecting the health of pets through antioxidation (Pacheco et al., 2018). However, no consensus has been reached regarding the optimal content of ω-3 fatty acids in canine and feline diets. The FEDIAF Nutritional Guidelines (FEDIAF, 2025) only provide the minimum recommended levels of linoleic acid (ω-6), arachidonic acid (ω-6), alpha-linolenic acid (ω-3), EPA + DHA (ω-3) for puppies, as well as linoleic acid (ω-6) for adult dogs (1.32% for 110 kcal/kg⁰·⁷⁵ diet, while 1.53% for 95 kcal/kg⁰·⁷⁵ diet); and the minimum recommended levels of linoleic acid (ω-6), arachidonic acid (ω-6), alpha-linolenic acid (ω-3), EPA + DHA (ω-3) for kittens, along with linoleic acid (ω-6) for adult cats (0.50% for 110 kcal/kg⁰·⁷⁵ diet, while 0.67% for 95 kcal/kg⁰·⁷⁵ diet), and arachidonic acid (ω-6) (60 mg/kg for 110 kcal/kg⁰·⁷⁵ diet, while 80 mg/kg for 95 kcal/kg⁰·⁷⁵ diet). No recommended levels for ω-3 fatty acids have been specified. Most studies on PUFA supplementation have focused on ω-3 fatty acid supplementation. According to previous studies, ω-6 fatty acids, particularly arachidonic acid, are also essential for pets because of their limited delta-6 desaturase activity (Lenox and Bauer, 2013). In this study, the ratio of ω-6 to ω-3 fatty acid supplements was 4.7:1 in cat oil and 0.9:1 in dog oil. Cats possess a more restricted capacity to synthesize arachidonic acid than dogs (Bauer, 2008). Additionally, some cat foods use fish meat and/or fish oil as raw materials, which also contain ω-3 fatty acids. The benefits of supplementing with long-chain ω-3 fatty acids for animals, such as improving cardiovascular health and alleviating inflammatory responses, are widely recognized ( Zárate et al., 2017; Shahidi and Ambigaipalan, 2018). However, their potential adverse effects on dogs and cats—including altered platelet function, gastrointestinal issues, lipid peroxidation, weight gain, modified immune function, and nutrient-drug interactions—are often neglected (Lenox and Bauer, 2013). A recent study by Richards et al. (2023) showed that there was no significant difference in coat quality between camelina, flaxseed, and canola oil supplementation in healthy adult dogs. This suggests that further investigation is needed regarding the effects of oil supplementation on health and hair quality in both dogs and cats. To address these uncertainties, this study evaluated various parameters, including fecal morphology, blood lipid, antioxidant indicators, inflammation-related markers, body weight, and hair beautification, after feeding fish oil-based nutritional supplements to dogs and cats. First, fecal morphology is an intuitive indicator of gastrointestinal health status (Hernot et al., 2004; Grellet et al., 2012). In this study-the supplementation of oils did not result in any significant changes in fecal scores for either species, indicating that the supplementation did not lead to severe gastrointestinal adverse effects to a certain extent. Usually, the assessment of fecal microbiota composition provides valuable insights into gastrointestinal health (Shaikh et al., 2023). Regrettably, fecal samples were not collected in this study, which meant that analyses related to the gut microbiota could not be performed. It is generally considered that ω-3 fatty acid supplementation contributes positively toward lowering blood lipid levels, although some studies report different conclusions (et al., 1996). These discrepancies may be attributed to variations in the types or concentrations of ω-3 fatty acids present within the supplements. In this study, the ω-3 fatty acid content in oil supplements for cats and dogs was 11.66% and 3.13%, respectively, which is lower than the long-chain ω-3 fatty acid levels reported in other studies (Dominguez et al., 2021; Xia et al., 2024; Zhang et al., 2025). The remaining fatty acids were absorbed by the intestines after supplementation, resulting in increased blood triglyceride levels within the normal range. Additionally, this could account for the inconsistent results across studies investigating unsaturated fatty acid supplementation and serum lipid peroxidation in animals. While high PUFA intake is typically linked to antioxidant activity and reduced lipid peroxides (Zaloga, 2021; Ravić et al., 2022), the associated potential increase in blood lipids may offset this advantage, resulting in unclear trends in serum lipid peroxidative capacity (Zaloga, 2021). Triglyceride serves as an essential energy source for mammals (Borén et al., 2022), and fluctuations within the normal blood lipid range may not negatively affect animal health. Obesity remains a significant public health concern that induces widespread anxiety relative to blood lipids. Additional oil supplementation is correlated with higher metabolic energy intake. However, recent studies have indicated that specific types of dietary fatty acids exhibit strong associations with obesity (Clavel et al., 2014). Notably, certain long-chain PUFAs have antiobesity properties (Cheng et al., 2006). Consistent with the findings of Rutherfurd-Markwick et al. (2013), no significant body weight gain was observed in either cats or dogs. Inflammation constitutes another concern. It is well established that elevated dietary intake of ω-6 PUFAs has been associated with inflammation primarily due to the role of arachidonic acid as a precursor for pro-inflammatory factors (Innes and Calder, 2018), whereas ω-3 fatty acids may mitigate inflammatory responses (Calder, 2017). The data revealed that omega-3-enriched oil supplementation significantly reduced serum TNF-α levels in both dogs and cats, indicating that omega-3-rich oils possess antiinflammatory properties. However, this study also found no statistically significant difference in IL-1 levels between the 2 animal groups. Given that immune modulation is a sophisticated biological process, substantial efforts are still required to explore the regulatory effects of omega-3-rich lipid supplementation on the immune system of dogs and cats. Quantitative and objective evaluation of hair quality is a challenging task, as it is largely influenced by emotional perception. Physio-mechanical properties serve as relatively objective indicators. This study demonstrated that oil supplementation increased the breaking force of hair in both cats and dogs. Similarly, Roman et al. (2022) indicated that the addition of PUFA increased hair breaking tension without a statistical difference (Roman et al., 2022). One contributing factor may be that PUFA supplementation improves hair sebum coverage (Roman et al., 2022). Studies have shown that lipids secreted by the sebaceous glands in the skin not only exert a protective effect by shielding the coat from damage caused by water, light, and other adverse factors but also contribute to the promotion of hair growth (Cerrato et al., 2013). Unfortunately, the chemical composition of hair and hair follicles was not further analyzed in this study. The thickness, density, shape, and arrangement structure of scales can directly affect various aspects of hair quality, such as glossiness, softness, and feel (Clavel et al., 2014). A thinner scale thickness corresponds to a lower friction coefficient and a smoother scale surface (Clavel et al., 2014). Our findings revealed that PUFA supplementation resulted in decreased scale thickness in both cats and dogs, which may account for the increase in hair brightness and flexibility. It is widely known that PUFAs are highly susceptible to autoxidation, which could shorten the stocking time of pet food (Kara, 2021). Therefore, extra tocopherol or other antioxidants are needed in the PUFA-supplemented diet. Indeed, studies have indicated that diet type, cleaning practices, and food bowl materials are key factors influencing the microbiological contamination of pet food bowls (Raspa et al., 2023). Undoubtedly, such an approach of additional oil supplementation poses new challenges for pet food bowl sanitation maintenance. Current research on nutritional interventions aimed at enhancing pet hair quality remains limited. Further investigation into evidence-based strategies to improve coat condition through dietary supplementation is clearly needed. ConclusionThis study demonstrated that dietary supplementation with a specific blend of EPA-rich oil significantly enhanced the hair quality of cats and dogs, as evidenced by improvements in flexibility, brightness, and skin condition. The underlying mechanism may be attributed to the increased mechanical strength of hair and alterations in its microstructure, such as reduced scale thickness. However, it is noteworthy that supplementation also resulted in elevated serum lipid levels in both species, particularly triglycerides and total cholesterol in dogs, although these values remained within normal ranges. These findings highlight the dual effects of PUFA supplementation: while PUFA supplementation is beneficial for coat health, careful consideration must be given to the fatty acid composition and dosage to mitigate potential adverse metabolic impacts. Future research should explore optimized formulations to maximize hair benefits while minimizing lipid-related risks, ensuring balanced nutritional strategies for companion animals. AcknowledgmentsThe authors would like to express their gratitude to Ce Liu and Gongyan Liu for their assistance with the physio-mechanical analysis of the hair. Conflicts of interestAll authors declare no conflict of interest. FundingThis study was supported by funding from SAAS Program for Excellent Research Team 2025(026). Authors' contributions“Conceptualization, C.X. Z.Y. and L.Y.; methodology, X.C. and C.X.; software, X.C. and C.X.; validation, X.X., Y.Y. and Z.Z.; formal analysis, C.X.; investigation, C.X. and X.C; resources, Z.Y. and S.Q.; data curation, C.X.; writing—original draft preparation, C.X. and M.H.; writing—review and editing, Z.L. and L.Y.; supervision, C.X. and L.Y.; project administration, L.Y.; funding acquisition, L.Y. All authors have read and approved the published version of the manuscript. Data availabilityThe datasets generated and analyzed in this study are available upon reasonable request from the corresponding author. ReferencesBauer, J.J.E. 2008. Essential fatty acid metabolism in dogs and cats. Rev. Bras. Zootec. 37, 20–27. Borén, J., Taskinen, M.R., Björnson, E. and Packard, C.J. 2022. Metabolism of triglyceride-rich lipoproteins in health and dyslipidaemia. Nat. Rev. Cardiol. 19, 577–592. Calder, P.C. 2017. Omega-3 fatty acids and inflammatory processes: from molecules to man. Biochem. Soc. Trans. 45, 1105–1115. Cerrato, S., Ramió-Lluch, L., Fondevila, D., Rodes, D., Brazis, P. and Puigdemont, A. 2013. Effects of Essential Oils and Polyunsaturated Fatty Acids on Canine Skin Equivalents: skin Lipid Assessment and Morphological Evaluation. J. Vet. Med. 2013, 1–9. Cheng, C.H., Ou, B.R., Shen, T.F. and Ding, S.T. 2006. Effects of dietary algal docosahexaenoic acid oil supplementation on fatty acid deposition and gene expression in laying Tsaiya ducks. Asian-Australasian J. Anim. Sci. 19, 1047–1053. Clavel, T., Lepage, P., Charrier, C. 2014. The family coriobacteriaceae. In The Prokaryotes, Ed., Rosenberg E., Berlin: Springer, pp. 201-238. Cline, M.G., Burns, K.M., Coe, J.B., Downing, R., Durzi, T., Murphy, M. and Parker, V. 2021. AAHA Nutrition and Weight Management Guidelines for Dogs and Cats. J. Am. Anim. Hosp. Assoc. 57(4), 153–178. Dominguez, T.E., Kaur, K. and Burri, L. 2021. Enhanced omega-3 index after long- versus short-chain omega-3 fatty acid supplementation in dogs. Vet. Med. Sci. 7, 370–377. Ephraim, E., Brockman, J.A. and Jewell, D.E. 2022. A Diet Supplemented with Polyphenols, Prebiotics and Omega-3 Fatty Acids Modulates the Intestinal Microbiota and Improves the Profile of Metabolites Linked with Anxiety in Dogs. Biology 11(7), 976. FEDIAF(the European pet food industry). 2025. FEDIAF Nutritional Guidelines for Complete and Complementary Pet Food for Cats and Dogs. https://europeanpetfood.org/self-regulation/nutritional-guidelines/ Grellet, A., Feugier, A., Chastant-Maillard, S., Carrez, B., Boucraut-Baralon, C., Casseleux, G. and Grandjean, D. 2012. Validation of a fecal scoring scale in puppies during the weaning period. Prev. Vet. Med. 106, 315–323. Guo, D., Zhang, L., Zhang, L., Han, S., Yang, K., Lin, X., Wen, C., Tong, A., Zhang, M. and Yin, Y. 2022. Effect of Dietary Methylsulfonylmethane supplementation on growth performance, hair quality, fecal microbiota, and metabolome in ragdoll kittens. Front. Microbiol. 13, 838164. Hamazaki, T., Sawazaki, S., Asaoka, E., Itomura, M., Mizushima, Y., Yazawa, K., Kuwamori, T. and Kobayashi, M. 1996. Docosahexaenoic acid-rich fish oil does not affect serum lipid concentrations of normolipidemic young adults. J. Nutr. 126, 2784–2789. Hernot, D.C., Weber, M.P., Biourge, V.C., Martin, L.J., Dumon, H.J. and Nguyen, P.G. 2004. Relationship between electrolyte apparent absorption and fecal quality in adult dogs differing in body size. J. Nutr. 134, 2031S–2034S. Innes, J.K. and Calder, P.C. 2018. Omega-6 fatty acids and inflammation. Prostaglandins. Leukot. Essent. Fatty Acids 132, 41–48. Jamikorn, U. and Kongpanna, P. 2025. Safety study of polyunsaturated fatty acids extracted from New Zealand green lipped mussel and krill on hematology and blood chemistry profiles of healthy. Thai. J. Vet. Med. 55(1), 15. Kara, K. 2021. Effect of stocking conditions on fatty acid composition and oxidation capacities of different class and type dog food. Ital. J. Anim. Sci. 20, 1042–1053. Kirby, N.A., Hester, S.L., Rees, C.A., Kennis, R.A., Zoran, D.L. and Bauer, J.E. 2009. Skin surface lipids and skin and hair coat condition in dogs fed increased total fat diets containing polyunsaturated fatty acids. J. Anim. Physiol. Anim. Nutr. 93, 505–511. Lenox, C.E. and Bauer, J.E. 2013. Potential adverse effects of Omega-3 fatty acids in dogs and cats. J. Vet. Intern. Med. 27, 217–226. Li, H., Wyant, R., Aldrich, G. and Koppel, K. 2020. Preference ranking procedure: method validation with dogs. Animals 10, 710. Montoya, M., Péron, F., Hookey, T., Morrison, J., German, A.J., Gaillard, V. and Flanagan, J. 2025. Overweight and obese body condition in ~4.9 million dogs and ~1.3 million cats seen at primary practices across the USA: prevalences by life stage from early growth to senior. Prev. Vet. Med. 235, 106398. Pacheco, G.F.E., Bortolin, R.C., Chaves, P.R., Moreira, J.C.F., Kessler, A.M. and Trevizan, L. 2018. Effects of the consumption of polyunsaturated fatty acids on the oxidative status of adult dogs. J. Anim. Sci. 96, 4590–4598. Pellegrino, F., Corrada, Y., Picco, S., Relling, A. and Risso, A. 2023. Association between dietary polyunsaturated fatty acids and their concentration in blood plasma, red blood cell, and semen of dogs. Open Vet. J. 13(3), 348–351. Raspa, F., Schiavone, A., Pattono, D., Galaverna, D., Cavallini, D., Vinassa, M., Bergero, D., Dalmasso, A., Bottero, M.T. and Valle, E. 2023. Pet feeding habits and the microbiological contamination of dog food bowls: effect of feed type, cleaning method and bowl material. BMC. Vet. Res. 19, 261. Ravić, B., Debeljak-Martacić, J., Pokimica, B., Vidović, N., Ranković, S., Glibetić, M., Stepanović, P. and Popović, T. 2022. The effect of fish oil-based foods on lipid and oxidative status parameters in police dogs. Biomolecules 12, 1092. Richards, T.L., Burron, S., Ma, D.W.L., Pearson, W., Trevizan, L., Minikhiem, D., Grant, C., Patterson, K. and Shoveller, A.K. 2023. Effects of dietary camelina, flaxseed, and canola oil supplementation on inflammatory and oxidative markers, transepidermal water loss, and coat quality in healthy adult dogs. Front. Vet. Sci. 10, 1085890. Rodrigues, R.B.A., Zafalon, R.V.A., Rentas, M.F., Risolia, L.W., Macedo, H.T., Perini, M.P., Silva, A.M.G.D., Marchi, P.H., Balieiro, J.C.D.C., Mendes, W.S., Vendramini, T.H.A. and Brunetto, M.A. 2023. The Supplementation of Docosahexaenoic acid-concentrated fish oil enhances cognitive function in puppies. Animals 13(18), 2938. Roman, K., Wilk, M., Książek, P., Czyż, K. and Roman, A. 2022. The effect of the season, the maintenance system and the addition of polyunsaturated fatty acids on selected biological and physicochemical features of rabbit fur. Animals 12, 971. Rutherfurd-Markwick, K.J., Hendriks, W.H., Morel, P.C.H. and Thomas, D.G. 2013. The potential for enhancement of immunity in cats by dietary supplementation. Vet. Immunol. Immunopathol. 152, 333–340. Shahidi, F. and Ambigaipalan, P. 2018. Omega-3 polyunsaturated fatty acids and their health benefits. Annu. Rev. Food. Sci. Technol. 9, 345–381. Shaikh, S.D., Sun, N., Canakis, A., Park, W.Y. and Weber, H.C. 2023. Irritable bowel syndrome and the gut microbiome: a comprehensive review. J. Clin. Med. 12, 2558. Spence, C. 2022. Gastrophysics for pets: tackling the growing problem of overweight/obese dogs. Appl. Anim. Behaviour. Sci. 256, 105765. Wyrostek, A., Czyż, K., Sokoła-Wysoczańska, E., Patkowska-Sokoła, B. and Bielas, W. 2023. The effect of ethyl esters of linseed oil on the changes in the fatty acid profile of hair coat sebum, blood serum and erythrocyte membranes in healthy dogs. Animals 13, 2250. Xia, J., Yin, S., Yu, J., Wang, J., Jin, X., Wang, Y., Liu, H. and Sun, G. 2024. Improvement in glycolipid metabolism parameters after supplementing fish oil-derived omega-3 fatty acids is associated with gut microbiota and lipid metabolites in type 2 diabetes mellitus. Nutrients 16, 3755. Yuan, C., Ren, L., Sun, X. Yun. X.., Zang, X., Zhang, A. and Wu, M. 2023. Mannan oligosaccharides improve the fur quality of raccoon dogs by regulating the gut microbiota. Front. Microbiol. 14, 1324277. Yuan, Y., Zhu, L., Cai, X., Mao, H., Wang, T., Tao, S., Bao, J., Liu, C. and Feng, J. 2025. The effect of Japanese eel as a main ingredient on hair condition, antioxidant ability, apparent total tract digestibility and body weight gain in cat food. Front. Vet. Sci. 12, 1553320. Zaloga, G.P. 2021. Narrative review of n-3 polyunsaturated fatty acid supplementation upon immune functions, resolution molecules and lipid peroxidation. Nutrients 13, 662. Zárate, R., El Jaber‐vazdekis, N., Tejera, N., Pérez, J.A. and Rodríguez, C. 2017. Significance of long chain polyunsaturated fatty acids in human health. Clin. Transl. Med. 6, 25. Zhang, Z.X., Lin, Y.C., Lian, M., Li, Y.F., Chen, J.F., Ma, X.L., Guo, D.S., Yang, G. and Sun, X.M. 2025. Evaluation of the efficacy and safety of omega-3 fatty acid nutritional supplements from Schizochytrium sp. in dog food. Algal. Res. 89, 104072. Supplementary MaterialSupplementary Materials 1. Information of dogs and cats used in this study.
Supplementary Materials 2a. Photos of cats feed with different diets.
Supplementary Materials 2b. Photos of dogs feed with different diets. | ||
| How to Cite this Article |
| Pubmed Style Cai X, Xiong C, Zhu L, Zhang Y, Mao H, Sun Q, Lu Y. PUFA-rich oil supplement improves hair condition of cats and dogs. Open Vet. J.. 2026; 16(3): 1823-1836. doi:10.5455/OVJ.2026.v16.i3.38 Web Style Cai X, Xiong C, Zhu L, Zhang Y, Mao H, Sun Q, Lu Y. PUFA-rich oil supplement improves hair condition of cats and dogs. https://www.openveterinaryjournal.com/?mno=281163 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.38 AMA (American Medical Association) Style Cai X, Xiong C, Zhu L, Zhang Y, Mao H, Sun Q, Lu Y. PUFA-rich oil supplement improves hair condition of cats and dogs. Open Vet. J.. 2026; 16(3): 1823-1836. doi:10.5455/OVJ.2026.v16.i3.38 Vancouver/ICMJE Style Cai X, Xiong C, Zhu L, Zhang Y, Mao H, Sun Q, Lu Y. PUFA-rich oil supplement improves hair condition of cats and dogs. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1823-1836. doi:10.5455/OVJ.2026.v16.i3.38 Harvard Style Cai, X., Xiong, . C., Zhu, . L., Zhang, . Y., Mao, . H., Sun, . Q. & Lu, . Y. (2026) PUFA-rich oil supplement improves hair condition of cats and dogs. Open Vet. J., 16 (3), 1823-1836. doi:10.5455/OVJ.2026.v16.i3.38 Turabian Style Cai, Xuan, Chunhong Xiong, Lihui Zhu, Yanning Zhang, Hui Mao, Qidong Sun, and Yonghong Lu. 2026. PUFA-rich oil supplement improves hair condition of cats and dogs. Open Veterinary Journal, 16 (3), 1823-1836. doi:10.5455/OVJ.2026.v16.i3.38 Chicago Style Cai, Xuan, Chunhong Xiong, Lihui Zhu, Yanning Zhang, Hui Mao, Qidong Sun, and Yonghong Lu. "PUFA-rich oil supplement improves hair condition of cats and dogs." Open Veterinary Journal 16 (2026), 1823-1836. doi:10.5455/OVJ.2026.v16.i3.38 MLA (The Modern Language Association) Style Cai, Xuan, Chunhong Xiong, Lihui Zhu, Yanning Zhang, Hui Mao, Qidong Sun, and Yonghong Lu. "PUFA-rich oil supplement improves hair condition of cats and dogs." Open Veterinary Journal 16.3 (2026), 1823-1836. Print. doi:10.5455/OVJ.2026.v16.i3.38 APA (American Psychological Association) Style Cai, X., Xiong, . C., Zhu, . L., Zhang, . Y., Mao, . H., Sun, . Q. & Lu, . Y. (2026) PUFA-rich oil supplement improves hair condition of cats and dogs. Open Veterinary Journal, 16 (3), 1823-1836. doi:10.5455/OVJ.2026.v16.i3.38 |