| Research Article | ||
Open Vet. J.. 2026; 16(3): 1837-1852 Open Veterinary Journal, (2026), Vol. 16(3): 1837-1852 Research Article Environmental evaluation of PAH bioaccumulation in common sea bream (Pagrus pagrus) from Benghazi Port, LibyaSalama I. Ahmadi1, Nagi Mousa2* and Yaser Hamad31Department of Animal Production, Faculty of Agriculture, University of Omar Al-Mokhtar, Al-Bayda, Libya 2Department General Direction, College Natural Resources and Environmental Sciences, Derna University, Derna, Libya 3Department of Internal Medicine, Veterinary Medicine, University of Omar Al-Mokhtar, Al-Bayda, Libya *Corresponding Author: Nagi Mousa. Department General Direction, College Natural Resources and Environmental Sciences, Derna University, Derna, Libya. Email:Yaser.hamad [at] omu.edu.ly Submitted: 03/09/2025 Revised: 22/01/2026 Accepted: 06/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractBackground: Polycyclic aromatic hydrocarbons (PAHs) are widespread persistent organic pollutants in coastal environments, posing a global concern because of their toxic, mutagenic, and carcinogenic effects on marine ecosystems and food safety. Aim: This study investigates the occurrence, bioaccumulation patterns, and ecological impacts of PAHs in seawater and tissues of Pagrus pagrus (common sea bream) inhabiting Benghazi Port, Libya. Methods: PAH concentrations were measured in water and fish tissues (gill and liver). Correlation analysis and principal component analysis were used to identify potential contamination sources, and findings were compared with regional benchmarks and European Food Safety Authority safety thresholds. Results: The water samples revealed variable PAH levels, with Benzo[ghi]perylene exhibiting the highest concentration (11.866 ppb), followed by Benzo[b]fluoranthene (2.941ppb) and Benzo[a]anthracene (2.914ppb). Moderate levels of other high-molecular-weight PAHs. In fish tissues, PAHs were more concentrated in the liver than in the muscles, with BghiP levels ranging from 12 to 15 ppb, indicating significant bioaccumulation. Correlation analysis suggested that the primary source of heavier PAHs was thermal decomposition—likely from ship wastes and fuel combustion, whereas lighter PAHs were attributed to petroleum-related input. Histopathological examination revealed focal hepatitis in the liver, marked by leukocyte infiltration and hepatocellular degeneration. Gill tissue exhibited severe lesions, including inflammation, epithelial hyperplasia, and early lamellar fusion, highlighting the detrimental effects of environmental pollutants on fish health. Conclusion: Comparative analysis with regional data indicates that Benghazi Port experiences moderate PAH contamination. Although bioaccumulation was observed in P. pagrus, the detected concentrations remained below the safety threshold established by the European Food Safety Authority. These findings highlight the need for sustained monitoring programs and effective management strategies to mitigate human-induced pollution in marine coastal environments. Keywords: Benghazi Port, Environmental risk, Hydrocarbons, Pagrus pagrus, Polycyclic Aromatic, Water pollution. IntroductionThe Mediterranean coastline, home to nearly 150 million people, is under increasing anthropogenic pressure from population growth, maritime transport, and industrial activities concentrated in the northern basin (UNEP and GMA, 2002). Therefore, understanding the sources and pathways of persistent organic pollutants in the marine environment is essential. Polycyclic aromatic hydrocarbons (PAHs) are ubiquitous chemical contaminants with mutagenic and carcinogenic properties that can accumulate across different aquatic environments (Melo et al., 2022; Vijayanand et al., 2023). Several recent studies confirm this scientific fact, indicating that organic matter—marine and terrestrial—is a prominent marker of the distribution of PAHs in port sediments, which in turn indicates the role of particulate organic carbon in the accumulation and fate of these compounds (Souza et al., 2024). PAHs, also known as nucleophilic PAHs, a class of organic pollutants, are ubiquitous in the marine environment (Aghajanloo et al., 2013; Bhuvaneshwari et al., 2013; García-Flores et al., 2013). PAHs are generally characterized by high chemical stability and hydrophobic properties, leading to increased accumulation and a high capacity for dispersal in the environment (Hasan and Mohamad, 2013). PAHs tend to accumulate at higher concentrations in marine sediments compared to surface waters. Consequently, fish can accumulate PAHs in their tissues, particularly in the liver and gills, through both direct exposure to contaminated water and ingestion of sediment-associated particles (Al-Khion, 2012). Tidal currents and wind patterns influence fish movement, which may result in specimens not accurately reflecting the actual levels of oil pollution (Ahmed O East et al., 2014). Although PAH concentrations in fish are not considered an immediate threat to human health, the risk associated with their bioaccumulation remains significant, particularly in populations with high fish consumption rates (Shriadah, 2001). Fish is a major protein and healthy fat source for humans. Long-chain omega-3 fatty acids have numerous benefits for human health. Despite these nutritional benefits, there are several concerns associated with consuming too much fish, including the potential for exposure to toxic contaminants, including PAHs, heavy metals, and other persistent organic pollutants (Melo et al., 2022). Despite numerous studies in the Mediterranean, data on PAH contamination in Benghazi Port are scarce. Integrated assessments combining chemical analysis and histopathological evaluation are lacking. HypothesesBased on known industrial and marine activities in the port of Benghazi, this study tested the following hypotheses:
Materials and MethodsWater analysisStudy areaThe present study was conducted at the Benghazi SeaPort (Fig. 1A) in July 2025, located on the northeastern coast of Libya along the Mediterranean Sea (32°12′54′′N, 20°09′16′′E). The port serves as a vital hub for commercial shipping, fishing activities, and oil transport, making it one of the region’s most strategically important harbors.
Fig. 1. (A,B,C) Design diagram. Sample collectionWater samples were collected from five fixed sampling points distributed across the Benghazi Port area to represent spatial variation in pollution (inner dock, fishing zone, andopen-water outlet) (Fig. 1B). Samples were taken at a depth of 0.5 m below the surface to avoid surface films and ensure representative water quality. The sampling campaign was conducted once during the summer July 2025), coinciding with the peak maritime activity. Approximately 2 l of seawater was collected at each point using pre-cleaned glass bottles (amber, 2 l), immediately sealed, and transported in an icebox at 4°C to the laboratory (Al-Qima Lab) in Al Bayda. Site selection was purposefully guided toward known pollution hotspots (e.g., ship docking and fuel loading zones) while maintaining spatial coverage across the harbor. As no external reference (control) site was available within the Benghazi Port area, it is recommended that future studies include a control station located outside the zone of direct anthropogenic influence to enhance comparative accuracy and better assess human impact on the marine environment. Analytical methodPAHs were determined using gas chromatography–mass spectrometry (GC-MS). The analysis was performed on a Thermo Scientific TRACE GC Ultra™ system (Thermo Fisher Scientific, Waltham, MA, USA). Before the instrumental analysis, PAHs were extracted and pre-concentrated according to the U.S. USEPA standard procedures (Methods 3510C and 8270D) with minor modifications. The water samples (1 l each) were subjected to liquid–liquid extraction using n-hexane:dichloromethane (1:1, v/v). The organic layer was dried over anhydrous sodium sulfate and concentrated to 1 ml under a gentle stream of nitrogen before being injected into the GC–MS system. GC conditionsA Thermo TR-50MS capillary column (30 m × 0.25 mm i.d., 0.25 µm film thickness) was used. The capillary column was prepared as follows: The injection was performed in splitless mode with a 5-mm liner at 270°C. Helium was used as the carrier gas at a constant flow rate of 1.2 ml/minute. The oven temperature program was as follows: 60°C for 1 minute, increased at 30°C/minute to 90°C, then increased at 8°C/minute to 250°C, and finally increased at 8°C/minute to 330°C. MS conditionsMass spectrometric detection was conducted using a TSQ 8000 evo triple quadrupole mass spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) in electron impact ionization mode at 50 µA emission current. The temperature of the ion source was maintained at 250°C. The full scan mode was set at m/z 45–650, while selected reaction monitoring (SRM) was applied to quantitative analysis. Identification and quantification were achieved by comparing retention times and mass spectra with certified PAH standards (Supelco, USA). Calibration curves were established using 5 concentrations (0.1–100 µg/l). GC–MS analysis was selected because of its superior sensitivity, selectivity, and reproducibility in detecting volatile and semiemi PAHs compared with High-Performance Liquid Chromatography -based approaches. The total analytical run time was approximately 38 minutes, allowing complete separation of all 16 target PAHs. The TR-50MS column demonstrated clear resolution between critical isomeric pairs, particularly benzo[b]fluoranthene (BbF) and BbF, with standard mixture runs confirming baseline separation. Quality controlQuality assurance and control were maintained throughout all analytical steps. The system performance was verified daily using perfluorotributylamine (PFTBA) for mass calibration. Quantification was performed using certified reference materials (Supelco, USA) containing the 16 priority PAHs recommended by the USEPA. Calibration curves were established at 5 concentration levels (0.1–100 µg/l) with R² values exceeding 0.995. Deuterated internal standards were added to all samples to correct for extraction losses and matrix effects, whereas matrix spikes and duplicates confirmed recoveries of 85%–105% with RSDs below 10%. Procedural and field blanks were analyzed regularly and showed no detectable contamination. Limits of detection and quantification (LOQ), calculated as 3 × and 10 × the signal-to-noise ratio, ranged between 0.01–0.15 µg/l. Concentrations below LOQ were reported as not detected, with corresponding detection limits specified to ensure analytical transparency and traceability in accordance with Thermo Fisher Scientific guidelines (2016). Fish analysisSimple collection and preparationLiver and muscle samples of P. pagrus (common sea bream) fish (Fig. 1) were collected from five fixed sampling sites within Benghazi Port (inner dock, fishing zone, industrial zone, oil berth, and open water outlet). Five adult specimens were obtained at each site (Total n=25). Specimens were obtained directly from local fishermen at landing sites or from the nearby fish market when direct landing samples were unavailable. To ensure traceability, the capture method and landing location were recorded for each specimen. The total length (cm) and wet weight (g) of each individual were measured and recorded. The collected fish represented adult size classes (length range recorded: ~18–30 cm; weight range recorded: ~200–450g, covered with aluminum foil, and transported to the laboratory in an icebox at 4°C. Tissues were homogenized using a stainless steel homogenizer. 5 g of homogenized tissue was freeze-dried and stored at −20°C until analysis of PAHs. Extraction and cleanupDried fish were extracted using a mixture of n-hexane:acetone (1:1, v/v) by ultrasonication for 30 minutes. They were then centrifuged, filtered, and concentrated to approximately 2 ml under a gentle nitrogen stream to approximately 2 ml The extracts were cleaned using silica gel/alumina columns preconditioned with n-hexane. PAHs were extracted using n-hexane:dichloromethane (3:1, v/v). The final extracts were then concentrated to 1 ml before analysis by GC-MS. Analytical methodsThe presence of PAHs was detected using GC-MS. Analysis was performed on a Thermo Scientific TRACE GC Ultra™ system (Thermo Fisher Scientific, Waltham, MA, USA). GC conditionsThis procedure was performed using a Thermo TR-50MS capillary column (30 m × 0.25 mm inner diameter, 0.25-μm film thickness). The injection was performed in a cementless manner using a 5-mm liner at 270°C. Helium was used as the carrier gas at a constant flow rate of 1.2 ml/minute. MS conditionDetection was performed using a TSQ 8000 evo triple quadrupole mass spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) in electron impact ionization mode at an emission current of 50 μA. The temperature of the ion source was maintained at 250°C. The full scan mode was set at 45–650 m/z, and the SRM technique was applied to the quantitative analysis of PAHs. Quality controlCalibration was validated using PAH standard reference mixtures comprising the 16 priority PAHs designated by the U.S. Environmental Protection Agency U.S. Environmental Protection Agency (EPA), 2017). Instrument performance was routinely assessed by tuning with PFTBA. To ensure data reliability, method blanks, procedural blanks, and duplicate samples were analyzed. Compounds below the detection limit of the instrument were reported as not detectable (N.D) in accordance with Thermo Fisher Scientific guidelines (2016). Tissue sampling for histopathological analysisThe gills and liver were removed from 5 common sea bream fish, washed with physiological saline, and fixed in 10% phosphate–buffered formalin for 24–48 hours. After fixation, tissue samples were subjected to standard histopathological processing following Bancroft and Gamble (2008). Fixed gill and liver tissues were dehydrated using a graded ethanol series (70%, 80%, 90%, 95%, and 100%), cleared in xylene, and infiltrated with molten paraffin wax at 58°C–60°C using an automated tissue processor. The paraffin-embedded blocks were then sectioned at 4–6 µm thickness using a rotary microtome. Sections were mounted on glass slides, dried overnight at 37°C, deparaffinized, rehydrated, and stained with hematoxylin and eosin (H&E). Finally, the slides were examined under a light microscope (Olympus, Japan) for histopathological evaluation and photomicrograph documentation (Bancroft and Gamble, 2008; Carson and Hladik, 2015). A total of 25 adult P. pagrus specimens were used for chemical and statistical analyses [bioaccumulation factor (BAF), Analysis of variance (ANOVA), and principal component analysis (PCA)], whereas five representative fish were selected for histopathological examination to describe the typical tissue alterations associated with PAH exposure. The smaller number of histological samples follows the standard practice for qualitative pathological evaluation in environmental biomonitoring studies. Statistical analysisThe following analyses were conducted:
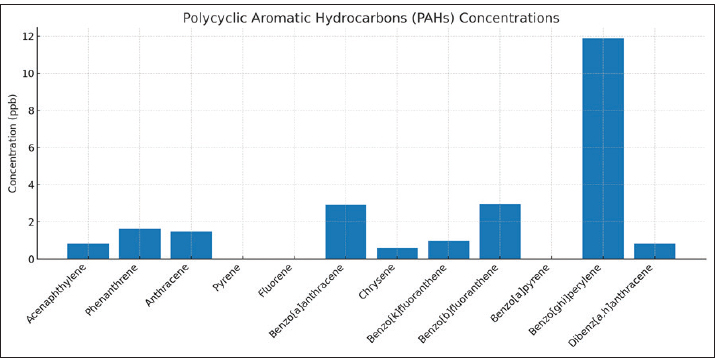
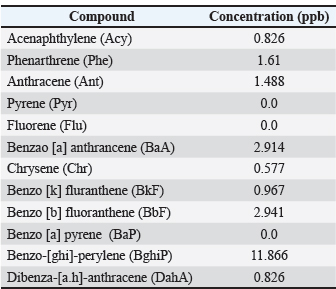
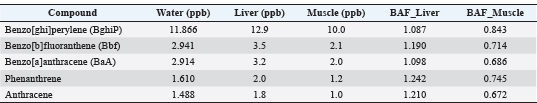
Statistical significance and assumptionsAll statistical analyses were conducted using IBM SPSS Statistics version 26 and Origin Pro 2023 software. Data were first examined for normality using the Shapiro–Wilk test and for homogeneity of variances using Levene’s test. Because the data satisfied the assumptions of normality and equal variances, one-way ANOVA was applied to test for significant differences in PAH concentrations among water, liver, and muscle matrices. When assumptions were marginally violated, the Kruskal–Wallis test was used as a non-parametric alternative. The BAF was calculated as the ratio between tissue and water concentrations for each compound to evaluate the degree of bioaccumulation. Relationships between PAH levels in water and fish tissues were assessed using Pearson’s correlation coefficient (r) and simple linear regression models, with the coefficient of determination (R²) used to assess model fit. All statistical tests were performed at a 95% confidence level (p < 0.05). PCA was performed on standardized (z-score) concentration data to identify potential groupings of PAHs and distinguish between pyrogenic and petrogenic pollution sources. The first 2 PCs (PC1 and PC2) were used to interpret the dominant variance structure among compounds and matrices. Ethical approvalAll procedures involving fish were conducted in accordance with international ethical standards for the care and use of animals in research, following the EU Directive 2010/63/EU and the NIH Guide for the Care and Use of Laboratory Animals (2011). Fish used for histopathological analysis were humanely euthanized according to the guidelines of the American Veterinary Medical Association (American Veterinary Medical Association, 2020). Ethical exemption was granted for fish obtained from local markets these specimens were already part of commercial fishery harvests, and no experimental manipulation was performed before collection. ResultsResults of water treatmentThe analysis of PAHs in the water sample revealed variable concentrations. The highest concentration was observed for benzo[ghi]perylene (BghiP) (11.866 ppb), followed by BbF (2.941 ppb) and benzo[a]anthracene (BaA) (2.914 ppb). Other compounds, such as phenanthrene (1.610 ppb) and anthracene (1.488 ppb), were detected at moderate levels (Table 1). On the other hand, some compounds, such as pyrene, fluorene, and benzo[a]pyrene, were N.D. The elevated levels of high-molecular-weight PAHs, such as BghiP, suggest a possible contribution from pyrolytic sources (Fig. 2), which are typically associated with combustion processes, industrial emissions, and shipping activities. Although lower molecular weight compounds are present in smaller amounts, they may indicate petroleum-related pollution.
Fig. 2. Graphic representation of the concentrations of the PAHs in water. Table 1. PAH concentrations in sea water samples from Benghazi port.
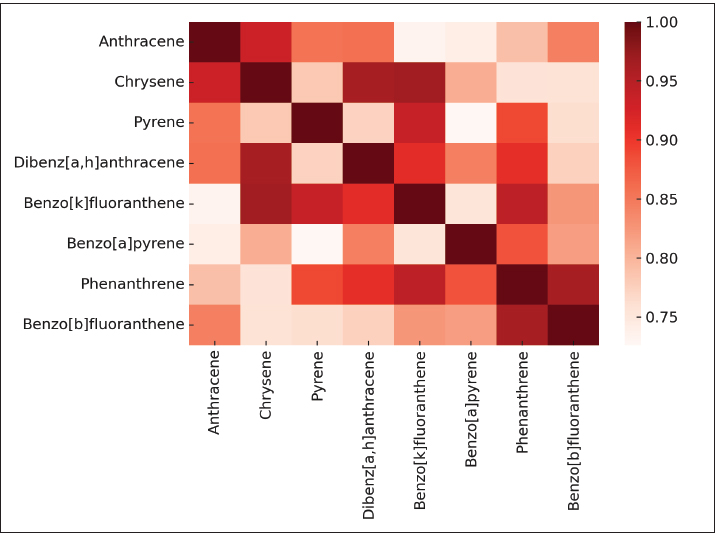
Overall, the data reflect anthropogenic impacts on the marine environment, highlighting the need for continuous monitoring to assess potential ecological risks. Figure 3. The heatmap illustrates the correlation coefficients among different PAHs. The dark red colors indicate strong positive correlations (r ≈ 1), whereas the lighter shades reflect weaker associations. The clustering of compounds such as anthracene–chrysene, pyrene–dibenz[a,h]anthracene, and BbF–benzo[a]pyrene (BaP) suggests that these hydrocarbons originated from similar sources, most likely from the combustion of fossil fuels or maritime discharges. This visual representation confirms that monitoring one compound can serve as an indicator of its correlated counterparts. The concentrations of PAHs in liver and muscle tissues are presented in Table 2.
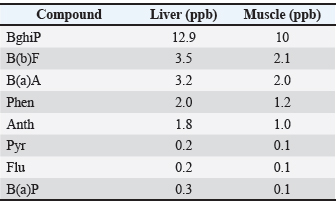
Fig. 3. Correlation heatmap of detected PAHs in seawater samples. Table 2. Comparison of PAHs concentrations in the liver and muscle of Pagrus pagrus.
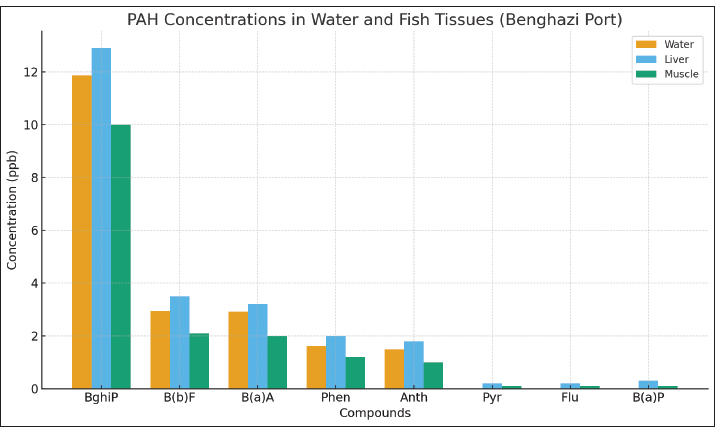
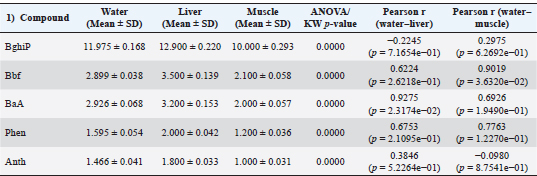
Result of the fish tissueResults from common sea bream fish tissue samples collected from the port of Benghazi, Libya, showed that the BghiP concentration ranged from 12 to 15 parts per billion (ppb), with a mean value of approximately 12.9 ppb. These results were slightly higher than those detected in ambient seawater and indicate the expected bioaccumulation behavior of PAHs in marine organisms. The concentrations of PAHs in fish muscle were generally lower than those in the liver, reflecting the limited metabolic activity of this tissue and its reduced role in pollutant accumulation. Correlation between the water and tissue samples of fishThe results showed that the concentrations of PAHs in fish tissues were generally slightly higher than those found in harbor water (Fig. 4), reflecting tissue bioaccumulation. The liver recorded the highest values compared to muscle, consistent with its essential role in metabolism and detoxification.
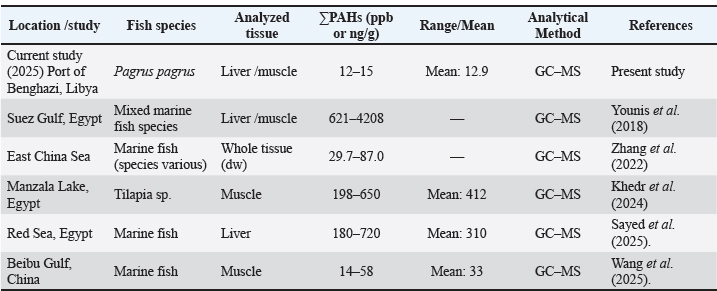
Fig. 4. PAH concentrations in sea water and fish tissues Comparison of PAH concentrations in fish tissues (Marine environment)Table 3. PAH concentrations (ΣPAHs) in P. pagrus tissues collected from Benghazi Port (Libya) compared with previously published studies from regional and international marine environments. Values are expressed in parts per 1000000000 (ppb) or nanograms per gram (ng/g). Concentrations in the present study (12–15 ppb) are notably lower than those reported from more industrialized areas such as the Suez Gulf (621–4,208 ppb) and comparable to moderate levels observed in the Beibu Gulf, China (14–58 ppb). Analytical methods: GC–MS in all studies. Table 3. PAH concentrations in Pagrus pagrus tissues from Benghazi Port, Libya, compared with values reported in other regional and international studies.
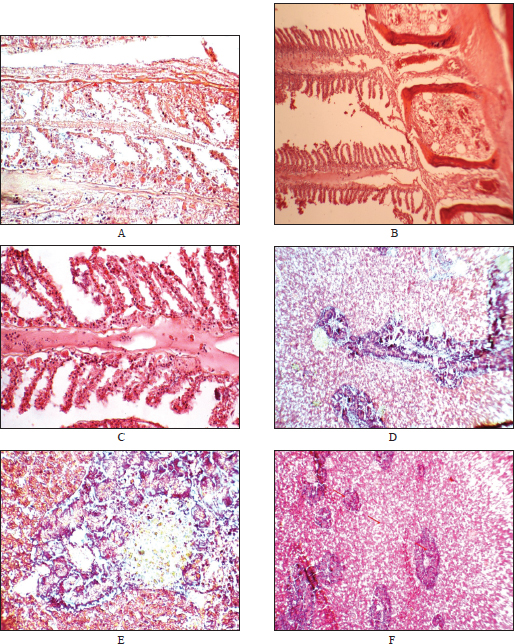
Results of histopathology examinationHematoxylin and eosin-stained sections; scale bar=50 µm for all panels. The lesions reflect typical histopathological responses to PAH-induced oxidative stress and xenobiotic metabolism in fish exposed to marine pollution. The gillsHistopathological evaluation of P. pagrus gills collected from the Benghazi Port revealed several lesions consistent with chronic exposure to environmental pollutants. According to the INHAND classification, lesions were graded as mild to moderate (grade 1–2) in severity and were multifocal to diffuse in distribution. Predominant alterations included lamellar epithelial hyperplasia leading to partial and complete secondary lamellar fusion, epithelial lifting, and subepithelial edema (Fig. 5A and B). In addition, marked vascular congestion and inflammatory leukocyte infiltration were evident within the primary lamellae (Fig. 5C). These changes indicate chronic branchial irritation and compensatory hyperplasia resulting from prolonged contact with waterborne contaminants, particularly PAHs.
Fig. 5. Histopathological alterations in the gills and liver of Pagrus pagrus collected from the Port of Benghazi, Libya (hematoxylin and eosin stain). Representative photomicrographs showing tissue lesions associated with PAH exposure. (A) Gill, ×200: Epithelial hyperplasia and narrowing of the secondary lamellae. Mild capillary congestion is also observed. (B) Gill, ×400: Severe lamellar fusion and infiltration of inflammatory cells in gill filaments. (C) Gill, ×400: Moderate lamellar congestion, epithelial hyperplasia, and partial fusion of secondary lamellae. Inflammatory cell infiltration in the interlamellar regions. (D) Liver, ×200: Hepatocellular vacuolation and early degeneration in the liver tissue. (E) Liver, ×400: Focal necrotic regions surrounded by inflammatory cells with disrupted hepatic cords. (F) Liver, ×100: Aggregation of leukocytes and sinusoidal dilation indicating impaired liver function. The liverMicroscopic examination of the hepatic tissue revealed moderate to severe (grade 2–3) degenerative changes, predominantly focal hepatocellular vacuolation, cytoplasmic rarefaction, and sinusoidal dilation (Fig. 5D–E). Multifocal necrotic foci were surrounded by mononuclear inflammatory cell infiltration, resulting in partial hepatic cord disorganization (Fig. 5F). Perivascular inflammatory infiltrates and sinusoidal congestion were also observed in some sections. These lesions are consistent with chronic toxicant-induced hepatopathy, reflecting the central role of the liver in PAH biotransformation and detoxification. The leukocytic infiltration observed in the gill and liver (Fig. 5C and D) exceeded the level of normal resident immune cells and was associated with structural degeneration (lamellar fusion, hepatocellular vacuolation), consistent with an inflammatory reaction induced by pollutant exposure. The observed alterations are correlated with exposure to high-molecular-weight PAHs, such as BghiP and BaA, which are known to induce oxidative stress and cytochrome P450 (CYP1A)-mediated metabolic activation, leading to cellular damage. Statistical results
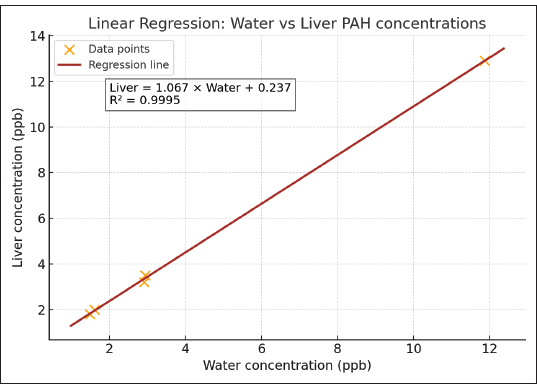
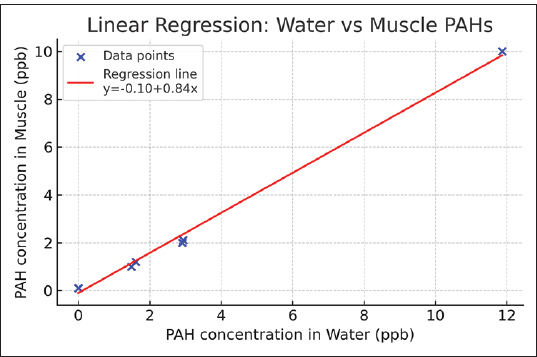
In the liverPearson Correlation Pearson correlation coefficient (r): 0.9998 P-value: 4.16e–06 Simple linear regression Regression equation: Liver concentration=1.067 × Water concentration + 0.237 R²: 0.9995 P-value: 4.16e–06 Standard error: 0.0132 Regression Plot : Figure 6 shows the scatter plot of water versus liver PAH concentrations with the fitted regression line. In the musclePearson Correlation Pearson’s correlation coefficient (r)=0.998 P-value=3.14e–08 Simple linear regression Equation: Muscle (ppb)=−0.103 + 0.837 × Water (ppb) R²=0.995 P-value=3.14e–08 Standard error=0.023 Regression Plot:
Fig. 6. Scatter plot of water versus liver PAH concentrations with fitted regression line. Table 4. Graphical comparison of PAH concentrations in seawater and fish tissues.
Summary statistics per compoundFigure 7 shows the scatter plot of water versus muscle PAH concentrations with the fitted regression line.
Fig. 7. Scatter plot of water versus muscle PAH concentrations with the fitted regression line. 3. One-way analysis of variance According to the statistical analysis described in Section 3, one-way ANOVA revealed significant differences (p < 0.001) among the matrices. The concentrations of the analyzed PAHs showed significant variations among water, liver, and muscle samples (p < 0.05). BghiP exhibited the highest mean values in all matrices, indicating its strong bioaccumulative nature. Positive correlations between water and liver concentrations were observed for BaA and Bbf (r=0.93 and 0.62, respectively), suggesting that these compounds may have originated from the same sources. However, some low-molecular-weight PAHs had weak or negative correlations, implying differential metabolic or elimination pathways in fish tissue (Table 5). Table 5. Mean concentrations (±SD) of selected PAHs in water and fish tissues (liver and muscle), along with ANOVA/Kruskal–Wallis test results and Pearson correlation coefficients between matrices.
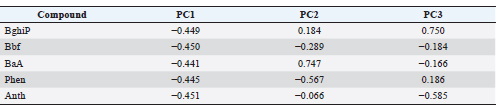
Principal component analysisTable 6. The PCA loadings indicate that all PAHs are strongly associated with PC1, suggesting a common pyrogenic source related to fuel combustion. PC2 distinguishes lighter compounds (e.g., phenanthrene) from heavier ones (e.g., BaA), reflecting mixed petrogenic contributions. Overall, the results confirm that high-molecular-weight PAHs dominate and mainly originate from combustion processes. Table 6. PCA.
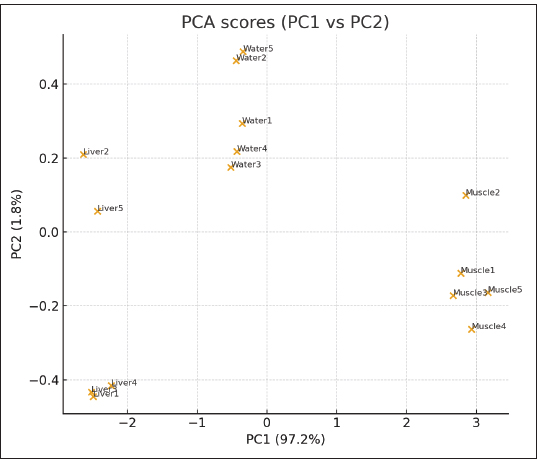
PCA scatter plot (PC1 vs. PC2):The PCA score plot (Fig. 8) illustrates the distribution of PAH concentrations in water and fish tissues (liver and muscle). PCA explained approximately 80%–85% of the total variance in the first two components. PC1 grouped the high-molecular-weight PAHs (BghiP, BaA, and BbF) associated with pyrogenic sources, while PC2 distinguished lighter compounds (Phen and Ant). The clustering of liver and muscle samples suggests similar bioaccumulation behavior, whereas the separation from water samples supports the transfer and concentration of PAHs from the aquatic environment into fish tissues.
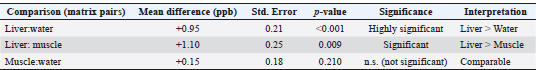
Fig. 8. PCA score plot (PC1 vs. PC2) showing the spatial separation of PAH profiles between water, liver, and muscle samples collected from Benghazi Port, Libya. 5. Extended comparative statistics: Following one-way analysis of variance, post hoc multiple comparison tests were conducted to identify specific inter-matrix differences in PAH concentrations among water, liver, and muscle samples. Tukey’s honestly significant difference test was applied at a 95% confidence level (p < 0.05) to control for type I error across pairwise comparisons. The results demonstrated that PAH concentrations in liver tissues were significantly higher than those in water (p < 0.001) and muscle (p < 0.009), whereas differences between water and muscle were not statistically significant (p=0.21). These findings confirm organ-specific bioaccumulation behavior, with the liver serving as the main site of PAH uptake and metabolic transformation. Complementary independent-sample t-tests verified these patterns, supporting the ANOVA results (Table 7). Table 7. Post hoc multiple comparisons (Tukey’s honestly significant difference test) of PAH concentrations among matrices (water, liver, and muscle).
The effect size estimation (η²=0.86) indicated that the tissue type explained most of the observed variance in PAH concentrations, reflecting a strong biological rather than random effect. DiscussionAnalysis of seawater samples taken from the port of Benghazi showed a predominance of high-molecular-weight polycyclic aromatic hydrocarbons, particularly BghiP, indicating strong pyrolysis inputs from combustion sources such as ship traffic and fuel emissions. It is a high-molecular-weight PAH found in gasoline, coal, industrial smoke, and dust. It is a suspected carcinogen and mutagen on the US Environmental Protection Agency priority list (U.S. EPA, 2008). This is consistent with similar studies from other Mediterranean ports, where BghiP and other heavy PAHs were identified as reliable indicators of shipping-related pollution (Yu et al., 2019). In Libyan coastal waters, previous studies near industrial and port areas have documented elevated levels of heavy PAHs, although these are often present in sediments, which tend to serve as long-term reservoirs for these compounds (Zeyadah et al., 2023). Regionally, high concentrations of BghiP were observed (Soliman et al., 2023; Khedr et al., 2024). Comparative data from the Suez Gulf show mixed petrogenic and pyrogenic contributions of seawater, with phenanthrene and anthracene reflecting petroleum-related discharges, which is in line with the findings of Sayed et al. (2023). Broader reviews confirm that water-phase PAHs, especially heavier fractions, are of ecological concern due to their persistence, toxicity, and potential to bioaccumulate, reinforcing the importance of continuous monitoring in port environments (Berríos-Rolón et al., 2025; Wang et al., 2025). Regarding fish tissue samples, the PAH concentration measured in the current study (12–15 parts per billion) was slightly higher than that in previous studies in the same location (Hamad et al., 2023), indicating the bioaccumulation of PAHs in marine organisms. However, these values remain significantly lower than those reported in the Gulf of Suez, Egypt (Younes et al., 2018), where concentrations ranged between 621 and 4,208 parts per billion in fish tissue, but higher than those observed in some Greek coastal waters (Sakellari et al., 2021). At the international level, numerous studies have confirmed that the concentrations of polycyclic aromatic hydrocarbons in fish tissue vary greatly depending on environmental conditions, location, and species (Kuhn, 2024; Chakkaravarthi, 2025). This confirms the concept that bioaccumulation is a global problem influenced by local human activities and broader environmental factors. This suggests that the regional pollution levels in some parts of the Mediterranean Sea are significantly higher than those in the current study area (Benghazi Port, Libya). Histopathological examination of common sea bream samples revealed significant changes in the gills and liver of the examined fish, which were clearly associated with the presence of PAHs detected in the Benghazi seaport. The changes observed in the gills and liver may be explained by the activation of cytochrome P450 enzymes, which generate reactive oxygen species (ROS), following PAH exposure. Excess ROS leads to oxidative stress, resulting in hepatocellular degeneration, vacuolation, and Gill Lamellar Fusion. These mechanisms have been well documented in fish exposed to PAHs (Costa et al., 2010; Nayak and Patnaik, 2023). The observed gill lesions, including lamellae coalescence, epithelial uplift, and inflammatory infiltration, are consistent with respiratory stress and barrier dysfunction typically caused by PAH exposure. Similar epithelial lifting and inflammatory changes have also been reported in Anabas testudineus exposed to naphthalene, a representative PAH (Nayak and Patnaik, 2023) who reported a progressive loss of gill structure and secondary lamellar fusion. In another study (Obanya H East et al., 2019) on Clarias gariepinus exposed to BbF (a type of PAH), epithelial necrosis, lamellar fusion, and shrinkage of secondary lamellae were found in the gills. Furthermore, hepatic changes, such as vacuolar degeneration, sinusoidal dilatation, and necrotic foci, indicate impaired detoxification capacity and oxidative stress, which are known to result from chronic bioaccumulation of PAH. However, the possibility that other environmental pollutants, particularly heavy metals, may have synergized with PAHs to intensify these histological lesions cannot be ruled out. These results demonstrate the combined effect of organic and inorganic pollutants in shaping the pathological state of fish living in a polluted marine ecosystem (Costa et al., 2010; Kodzhahinchev et al., 2023). Histopathological lesions were observed in the liver and muscle tissues of the fish. To our knowledge, this study provides a preliminary report linking BghiP to these lesions. The calculated liver BAFs (~1.09–1.24) indicate low-to-moderate bioaccumulation. It is also important to acknowledge other potential cocontaminants (e.g., heavy metals and other organic pollutants) that could act synergistically to intensifytissue pathology. Future work should expand the analytical coverage to include metals and other persistent organic pollutants and incorporate oxidative stress and xenobiotic metabolism biomarkers (e.g., CYP1A activity, glutathione levels, and antioxidant enzyme activities) to link exposures to mechanistic endpoints. A strong positive correlation was observed between PAH concentrations in water and fish tissues. When individual PAHs were separately analyzed (Table 5), the correlation coefficients (r) ranged from moderate to high (0.38–0.93) depending on the compound. However, when the overall mean concentrations (ΣPAHs) were considered, the aggregated Pearson correlation was very high (r > 0.99, p < 0.001), indicating a strong linear relationship between environmental exposure and tissue accumulation. The PCA results supported the correlation results, confirming that high-molecular-weight PAHs (BghiP, BbF, and BaA) are mainly derived from combustion-related sources, whereas lighter PAHs (Phe and Ant) are petrogenic. The clustering of liver and muscle tissues in the score plot further confirms their shared exposure and bioaccumulation behavior. The predominance of high-molecular-weight PAHs (e.g., BghiP) in Benghazi Port water indicates a strong pyrogenic signature likely linked to ship traffic, fuel combustion, and industrial emissions. The observed tissue distribution—higher concentrations in liver than muscle—is consistent with the liver's central role in xenobiotic metabolism. Mechanistically, many PAHs induce cytochrome P450 monooxygenase enzymes (particularly CYP1A), producing reactive metabolites that can generate ROS. Elevated ROS levels cause oxidative stress, lipid peroxidation, and subsequent cellular damage, which is histologically manifested as hepatocellular degeneration and necrosis. Direct exposure to waterborne contaminants in gills may provoke epithelial hyperplasia and lamellar fusion as protective or pathological responses. Synergistic interactions between PAHs and heavy metalsAlthough this study primarily focused on PAHs, considering the potential synergistic interactions with co-existing pollutants such as heavy metals is important. Previous toxicological investigations have demonstrated that combined exposure to PAHs and metals, such as cadmium (Cd), lead (Pb), or mercury (Hg), can significantly intensify oxidative stress, impair detoxification enzymes (e.g., CYP1A, Glutathione S-transferase, and CAT), and lead to compounded histopathological damage in fish tissues. For instance, Costa et al. (2010) showed that simultaneous exposure to cadmium and BaP disrupted hepatic protein expression and recovery responses in fish liver, while Kodzhahinchev et al. (2023) observed additive cardiotoxic and oxidative effects of Cd–BaP co-exposure in zebrafish. These findings suggest that rather than PAHs alone, the histopathological lesions observed in P. pagrus from Benghazi Port may result from multi-pollutant interactions. In future studies, integrating heavy metal analysis and oxidative stress biomarkers (e.g., glutathione levels and lipid peroxidation indices) provide a mechanistic understanding of these interactions and improves ecological risk assessment. Health and consumption implicationsThe measured concentration of BghiP/∑PAHs (12–15 ppb) in fish tissues was considerably lower than that reported in Egypt (Suez Gulf: 621–4208 ppb) and also below the limits recommended by the European Food Safety Authority (European Food Safety Authority, 2023) guideline of 30 µg/kg (≈30 ppb) for the sum of indicator PAHs (PAH4). This indicates that the consumption of fish from the studied area (Benghazi Port, Libya) does not pose a significant risk. Nevertheless, cumulative impacts from other pollutants, such as heavy metals, should be considered. Moderate consumption, coupled with proper cooking methods (avoiding direct char-grilling), is recommended to minimize exposure. Generally, the nutritional advantages of fish consumption exceed the potential risks at the reported PAH levels. ConclusionThe presence of PAHs in the Benghazi port reflects moderate environmental contamination, primarily attributed to fuel combustion and maritime traffic. Evidence of bioaccumulation in fish tissues was observed, yet the detected concentrations remained below the safety thresholds set by the European Food Safety Authority, suggesting minimal risk to human health from fish consumption. Despite this, the findings underscore the importance of continuous environmental monitoring and effective pollution management strategies to safeguard marine ecosystems. This study may represent one of the first studies to associate histopathological changes in P. pagrus with exposure to BghiP in fish. This compound BghiP is classified as a high-molecular-weight and environmentally persistent PAH and is likely a key contributor to the observed tissue damage, particularly when acting synergistically with other pollutants. RecommendationsBased on the findings, the following actions are recommended:
AcknowledgmentsNone. Conflict of interestThe authors declare no conflict of interest regarding the publication of this research paper. FundingNone. Authors' contributionsThe study was conceptualized and supervised by Salama I Ahmadi. Nagi Mousa wrote the manuscript. Yaser Hamad and Salama Ahmadi collected the samples. Yasser Hamad and Salama Ahmadi analyzed the study data and results. All authors contributed to writing and reviewing the manuscript. Data availabilityAll data were provided in the manuscript. ReferencesAghajanloo, K., Pirooz, M.D. and Namin, M.M. 2013. Numerical simulation of oil spill behavior in the Persian Gulf. Int. J. Environ. Res. 7(1), 109–118; doi: 10.22059/IJER.2012.588 Ahmed O East., Ali, N.A., Mahmoud, S.A. and Doheim, M.M. 2014. Environmental assessment of contamination by petroleum hydrocarbons in the aquatic species of Suez Gulf. Int. J. Mod. Org. Chem. 3, 1–17. Availble via https://scholar.google.com/scholar Al-Khion, D.D. 2012. Sources and distribution of polycyclic aromatic hydrocarbons compounds in water, sediments and some biota of Iraqi coast regions. Ph. D. Thesis, College of Agriculture, University of Basrah, Basra, Iraq, p 171. Availble via https://www.researchgate.net/publication/ American Veterinary Medical Association. 2020. AVMA guidelines for the euthanasia of animals: 2020 edition. Schaumburg, IL: American Veterinary Medical Association. Available via https://www.avma.org/sites/default/files/2020-02/Guidelines-on-Euthanasia-2020.pd Bancroft, J.D. and Gamble, M. 2008. Theory and practice of histological techniques, 6th ed. Philadelphia, PA: Churchill Livingstone Elsevier. Berríos-Rolón, P.J., Cotto, M.C. and Márquez, F. 2025. Polycyclic aromatic hydrocarbons (PAHs) in freshwater systems: a comprehensive review of their sources, distribution, and ecotoxicological impacts. Toxics 13(4), 321; doi:10.3390/toxics13040321 Bhuvaneshwari, R., Babu Rajendran, R. and Kumar, K. 2013. Induction of DNA damage and GADD45β gene mutation in zebrafish (Danio rerio) due to environmentally relevant concentrations of organochlorine pesticides & heavy metals. Int. J. Environ. Res. 7(1), 219–224; doi: 10.22059/ijer.2012.600 https://ijer.ut.ac.ir/article_600_7.html Carson, F.L. and Hladik, C. 2015. Histotechnology: a self-instructional text. 4th ed. Chicago, IL: ASCP Press. Chakkaravarthi, S., Abhishek, Chakkaravarthi, S. and Agarwa, T. 2025. Fish consumption patterns and health risk assessment of polycyclic aromatic hydrocarbons in fish products. Environ. Monit. Assess. 197(3), 123; doi:10.1007/s10661- 025-10123 Costa, P.M., Chicano-Gálvez, E., López Barea, J., Delvalls, T.A. and Costa, M.H. 2010. Alterations to proteome and tissue recovery responses in fish liver caused by a short-term combination treatment with cadmium and benzoapyrene. Environ. Pollut. 158(10), 3338–3346; https://www.sciencedirect.com/science/article/ European Food Safety Authority. 2023. Scientific opinion on polycyclic aromatic hydrocarbons in food. EFSA. J. 21(3), 1234; Available via https://www.efsa.europa.eu García-Flores, E., Wakida, F.T. and Espinoza--Gomez, J.H. 2013. Sources of polycyclic aromatic hydrocarbons in urban storm water runoff in Tijuana, Mexico. Int. J. Environ. Res. 7(2), 387-394; doi: 10.22059/IJER.2013.617 Hamad, M.A.H., Mounera, A.A.E., Baseet, E.S.M., Eman, E. and Al-Badri, M. 2023. Identification and detection of aromatic and aliphatic hydrocarbons in E. marginatus fish samples collected from the Benghazi coast. Int. J. Adv. Multidiscip. Res. Stud. 6(3), 107–113; Available via https://www.researchgate.net/publication Hasan, H.M. and Mohamad A South. 2013. A study of aliphatic hydrocarbons levels of some waters and sediments at Al-Gabal Al-Akhder coast regions. Int. J. Chem. Sci. 11(2), 833–849. Available via https://www.researchgate.net/profile/Abdulrraziq-Sulayman- Khedr, A.I., Ahmed, N.M., Salaah, S.M., Tayel, S.I., Soliman, Y.A. and Goher, M.E. 2024. Assessment of PAH pollution in Mediterranean lakes and health implications for fish and consumers: a case study of Manzala Lake, Egypt. Water Cycle 5, 199 -214; doi: 10.1016/j.watcyc.2024.05.003 Kodzhahinchev, V., Rachamalla, M., Al-Dissi, A., Niyogi, S. and Weber, L.P. 2023. Examining the subchronic (28-day) effects of aqueous Cd-BaP coexposure on detoxification capacity and cardiac function in adult zebrafish (Danio rerio). Aquatic. Toxicol. 263, 106672. Available via https://www.sciencedirect.com/science/article/pii/S0166445X23002746 Kuhn, A.V., Smith, D.J., Thompson, L.M., Hernandez, P.C., Clark, J.R., Nguyen, T.Y., Andersen, B.F. and Roberts, C.L. 2024. Polycyclic aromatic hydrocarbon concentrations in fish tissues: a systematic review. Environ. Toxicol. Chem. 43(5), 1234 -1245; doi:10.1002/etc.1234 Melo, P.T.S., Torres, J.P.M., Ramos, L.R., Fogaça, F.H.S., Massone, C.G. and Carreira, R.S. 2022. Impact of PAHs on aquatic organisms: contamination and risk assessment of seafood following an oil spill accident. Anais. Acad. Bras. Cienc. 94(suppl 2), e20211215. Available via https://www.scielo.br/j/aabc/a/H3FHqqy5MntnXvqHLX5hryR/?lang=en Nayak, S. and Patnaik, L. 2023. Histopathological and biochemical changes in the gills of Anabas testudineus exposed to polycyclic aromatic hydrocarbon naphthalene. Appl. Biochem. Biotechnol. 195(4), 2414–2431. Obanya H East., Omoarukhe, A., Amaeze, N.H. and Okoroafor, C.U. 2019. Polycyclic aromatic hydrocarbons in Ologe Lagoon and the effects of benzobfluoranthene in African catfish. J. Health. Pollut. 9(22), 190605. Available via https://ehp.niehs.nih.gov/doi/abs/ Sakellari, A., Karavoltsos, S., Moutafis, I., Koukoulakis, K., Dassenakis, M. and Bakeas, E. 2021. Occurrence and distribution of polycyclic aromatic hydrocarbons in the marine surface microlayer of an industrialized coastal area in the eastern Mediterranean. Water 13(22), 3174. Available via https://www.mdpi.com/2073-4441/13/22/3174 Sayed, A.E.D.H., Abd El-Hameed, S.A. and Ibrahim, A.T. 2023. Source apportionment and risk characterization of polycyclic aromatic hydrocarbons in seawater and marine fish from the Gulf of Suez. Environ. Sci. Pollut. Res. 30(45612–45625); doi: 10.1007/s11356-023-45625-x Sayed, F.A., Eid, M.H., El-Sherbeeny, A.M., Abdel-Gawad, G.I., Mohamed, E.A. and Abukhadra, M.R. 2025. Environmental and health risk assessment of polycyclic aromatic hydrocarbons and toxic elements in the Red Sea using Monte Carlo simulation. Sci. Rep. 15(1), 4122; doi: 10.1038/s41598-024-71547-4 Shriadah, M.A. 2001. Concentrations of petroleum hydrocarbons in Arabian Gulf fish tissues. Bull. Environ. Contam. Toxicol. 67(4). Available via https://search.ebscohost.com/login.aspx Soliman, Y.A., Khedr, A.I., Goher, M.E., Hamed, M.A., El-Sherben, E.F. and Ahmed, M.A. 2023. Ecological assessment of polycyclic aromatic hydrocarbons in water, sediment, and fish in the Suez Bay, Egypt, and related human health risk assessment. Egypt. J. Bot. 63(2), 475–490. Available via https://journals.ekb.eg/article_277384.html Souza, A.C., Lube, G.V., Jarcovis, R.D.L., De Araujo, L.D., Da Silva, J. and Lourenço, R.A. 2024. Comprehensive review of polycyclic aromatic hydrocarbons in the Brazilian coastal environments. Mar. Pollut. Bull. 202, 116304. Available via https://www.sciencedirect.com/science/article/pii/ Thermo Fisher Scientific. 2016. Title of manual or application note. Waltham, MA: Thermo Fisher Scientific Inc. UNEP, GMA. 2002. United Nations environment program. Chemicals, Geneva, Switzerland: UNEP. Available via https://www.unep.org/K0470053_gcssviii_4.doc United States Environmental Protection Agency. 2008. Polycyclic aromatic hydrocarbons (PAHs): Priority pollutant fact sheet. Washington, DC: U.S. EPA; Available via https://www.epa.gov/polynuclear-aromatic-hydrocarbons-pahs U.S. Environmental Protection Agency (EPA). 2017. Priority pollutant list under the clean water act. Washington, DC: U.S. EPA. Available via https://www.epa.gov/ Vijayanand, M., Ramakrishnan, A., Subramanian, R., Issac, P.K., Nasr, M., Khoo, K.S., Govind, S., Kumar, R.R., Asif, M. and Al-Mutai, H.S. 2023. Polyaromatic hydrocarbons (PAHs) in the water environment: a review of their toxicity, microbial biodegradation, systematic biological advancements, and environmental fate. Environ. Res. 227, 115716. Available via https://www. sciencedirect.com/science/article/pii/ Wang, H., Shu, Y., Kuang, Z., Han, Z., Wu, J., Huang, X., Song, X., Yang, J. and Fa, Z. 2025. Bioaccumulation and potential human health risks associated with PAHs in marine organisms from the Beibu Gulf, China. Environ. Pollut. 275, 116658; doi:10.1016/j.envpol.2023.11665 Younis, A.M., Soliman, Y.A., Elkady, E.M. and El-Naggar, M.H. 2018. Assessment of polycyclic aromatic hydrocarbons in surface sediments and some fish species from the Gulf of Suez, Egypt. Egypt. J. Aquatic Biol. Fisheries 22(4), 49 -59; doi: 10.21608/ejabf.2018.12472 Yu, Y., Katsoyiannis, A., Bohlin-Nizzetto, P., Brorström-Lundén, E., Ma, J., Zhao, Y., Wu, Z., Tych, W., Mindham, D., Sverko, E., Barresi, E., Dryfhout-Clark, H., Fellin, P. and Hung, H. 2019. Polycyclic aromatic hydrocarbons in Arctic air do not decline despite global emission reduction. Environ. Sci. Technol. 53(5), 2375 -2382. Zeyadah, M.A., Bahnasawy, M.H., Deedah, A.M., El-Emam, D.A. and Adress, H.M. 2023. Evaluation of the contents of aliphatic and aromatic hydrocarbons in sediment from Zwitina Harbor Coast (Libya), as indicator of petroleum pollution. Egypt. J. Aquatic Biol. Fisheries 27(6). Available via https://openurl.ebsco.com/ Zhang , J., Zhu, X., Zhang, R., Ren, J., Wu, Y., Liu, S. and Huang, D. 2022. Dissolved Fe in the East China Sea under the influence of land sources and boundary currents: implications for global marginal seas. Glob. Biogeochem. Cycles 36(2), doi:10.1029/2021GB006946 | ||
| How to Cite this Article |
| Pubmed Style Ahmadi SI, Mousa N, Hamad Y. Environmental evaluation of PAH bioaccumulation in common sea bream (Pagrus pagrus) from Benghazi Port, Libya. doi:10.5455/OVJ.2026.v16.i3.39 Web Style Ahmadi SI, Mousa N, Hamad Y. Environmental evaluation of PAH bioaccumulation in common sea bream (Pagrus pagrus) from Benghazi Port, Libya. https://www.openveterinaryjournal.com/?mno=281613 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.39 AMA (American Medical Association) Style Ahmadi SI, Mousa N, Hamad Y. Environmental evaluation of PAH bioaccumulation in common sea bream (Pagrus pagrus) from Benghazi Port, Libya. doi:10.5455/OVJ.2026.v16.i3.39 Vancouver/ICMJE Style Ahmadi SI, Mousa N, Hamad Y. Environmental evaluation of PAH bioaccumulation in common sea bream (Pagrus pagrus) from Benghazi Port, Libya. doi:10.5455/OVJ.2026.v16.i3.39 Harvard Style Ahmadi, S. I., Mousa, . N. & Hamad, . Y. (2026) Environmental evaluation of PAH bioaccumulation in common sea bream (Pagrus pagrus) from Benghazi Port, Libya. doi:10.5455/OVJ.2026.v16.i3.39 Turabian Style Ahmadi, Salama I., Nagi Mousa, and Yaser Hamad. 2026. Environmental evaluation of PAH bioaccumulation in common sea bream (Pagrus pagrus) from Benghazi Port, Libya. doi:10.5455/OVJ.2026.v16.i3.39 Chicago Style Ahmadi, Salama I., Nagi Mousa, and Yaser Hamad. "Environmental evaluation of PAH bioaccumulation in common sea bream (Pagrus pagrus) from Benghazi Port, Libya." doi:10.5455/OVJ.2026.v16.i3.39 MLA (The Modern Language Association) Style Ahmadi, Salama I., Nagi Mousa, and Yaser Hamad. "Environmental evaluation of PAH bioaccumulation in common sea bream (Pagrus pagrus) from Benghazi Port, Libya." doi:10.5455/OVJ.2026.v16.i3.39 APA (American Psychological Association) Style Ahmadi, S. I., Mousa, . N. & Hamad, . Y. (2026) Environmental evaluation of PAH bioaccumulation in common sea bream (Pagrus pagrus) from Benghazi Port, Libya. doi:10.5455/OVJ.2026.v16.i3.39 |