Open Veterinary Journal, (2026), Vol. 16(3): 1704-1715
Research Article
10.5455/OVJ.2026.v16.i3.28
Enzymes from mollusk-associated bacteria in Ambon waters of Wallacea: Prospective cosmetic applications
Sunelsya Surya1, Delianis Pringgenies2* , Yashwant Pathak3 , Sri Sedjati2 ,
Tatas Hardo Panintingjati Brotosudarmo4 , Dafit Ariyanto5 and Meigy Nelce Mailoa6
1Doctoral Program in Marine Science, Faculty of Fisheries and Marine Science,
Universitas Diponegoro, Semarang, Indonesia
2Department of Marine Science, Faculty of Fisheries and Marine Science,
Universitas Diponegoro, Semarang, Indonesia
3Taneja College of Pharmacy, University of South Florida, Tampa, FL 33612-4749, USA
4Food Technology Study Program, Faculty of Industrial Technology, Petra Christian University, Surabaya, Indonesia
5Research Center for Ecology, National Research and Innovation Agency (BRIN), Bogor, Indonesia
6Fishery Products Technology Study Program, University of Pattimura, Ambon, Indonesia
*Corresponding Author: Delianis Pringgenies. Department of Marine Science, Faculty of Fisheries and Marine Science, Universitas Diponegoro, Semarang, Indonesia. Email: delianispringgenies [at] lecturer.undip.ac.id
Submitted: 19/09/2025 Revised: 20/01/2026 Accepted: 05/02/2026 Published: 31/03/2026
© 2026 Open Veterinary Journal
This is an Open Access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited, and is not altered, transformed, or built upon in any way.
ABSTRACT
Background: Marine mollusks serve as hosts for symbiotic bacteria capable of producing secondary metabolites with significant biotechnological potential, including those used in the cosmetic industry. The rich aquatic biodiversity of Ambon, located within the Wallacea region, presents a unique opportunity to explore marine microbes with distinctive metabolite profiles. However, research exploring mollusk symbiont bacteria from Ambon for cosmetic-related bioactive metabolites remains scarce. The valuable metabolite-producing potential of these compounds highlights the importance of further investigating their biological activities and metabolite profiles for prospective cosmetic applications.
Aim: This study aimed to evaluate the mollusk-associated bacteria for antibacterial and enzymatic activities, identify the bacterial strains, and analyze their secondary metabolites using GC-MS to support the development of active cosmetic ingredients.
Methods: As many as 6 mollusks were selectively collected from Sopapey waters, Ambon region, and bacterial isolates were obtained using standard culturing techniques. Antibacterial activity was evaluated against Cutibacterium acnes and Staphylococcus epidermidis, enzymatic activities were tested using 1% skim milk on agar medium, and molecular identification was conducted using 16S rRNA sequencing. GC-MS analysis was employed to profile secondary metabolites from selected isolates. Statistical analysis was performed using one-way analysis of variance in RStudio to determine significant differences among the isolates and controls.
Results: Three isolates exhibited inhibitory activity, with SPG241 showing the strongest inhibition zone, 4.80 ± 0.57 mm (24 hours) and 2.75 ± 0.49 mm (48 hours) against C. acnes and 8.43 ± 1.26 mm (24 hours) and 7.04 ± 0.97 mm (48 hours) against S. epidermidis. Protease assays demonstrated that eight isolates possessed proteolytic activity, with hydrolysis zones of 11.65–13.85 mm in 48 hours. Molecular identification revealed that the potential isolates belonged to Pseudoalteromonas citrea, Pseudoalteromonas peptidolytica, Vibrio xuii, and Vibrio tubiashii. These symbionts produced bioactive metabolites, including esters (e.g., methyl palmitate and trans-13-octadecenoic acid) and functional molecules, such as 2,3-butanediol and 2-piperidinone. Isolates with the highest enzymatic activity exhibited promising cosmetic properties, including emollient, humectant, antioxidant, anti-inflammatory, and antibacterial effects.
Conclusion: The findings confirm that mollusk symbiont bacteria from Sopapey water, particularly within the Wallacea region, are a promising source of secondary metabolites for natural-based cosmetic products, reinforcing the region’s importance as a promising frontier for the discovery of innovative marine bioactive compounds.
Keywords: Cosmetics, GC-MS, Mollusks, Secondary metabolites, Symbiont bacteria, Wallacea.
Introduction
The cosmetic industry is currently experiencing rapid growth, driven by technological advances and research in biology and chemistry. Consumers are increasingly opting for cosmetic products that use natural ingredients, which are generally isolated from plants, animals, and insects, and are considered safer and more environmentally friendly than synthetic chemicals (Mohamed et al., 2021; Liu, 2022). In this context, enzymes derived from marine organisms, such as mollusks, have emerged as a promising area of innovative cosmetic research (Alnuqaydan, 2024). Mollusks inhabit diverse habitats, including the waters of Ambon, which are rich in biodiversity. Symbiotic microorganisms live and interact with their hosts within their bodies, providing ecological benefits while offering potential for various applications (Siahaan et al., 2022; Nirmal et al., 2024). One promising application is in the cosmetics industry, where the antibacterial compounds and protease enzymes produced by mollusk symbiont bacteria can be used for the development of skin care products. These symbiotic bacteria can produce secondary metabolites and enzymes with biological activities that are suitable for bioindustry applications (Pringgenies et al., 2023). Another study identified bacterial isolates from mollusks with enzymatic activity, including Alcaligenes faecalis and Alcaligenes aquatilis (Setyati et al., 2023).
Protease enzymes produced by mollusk symbiotic bacteria are of particular interest because of their ability to accelerate skin rejuvenation, enhance hydration, and protect against free radicals (Trevisol et al., 2022). Protease enzyme can remove the keratin layer on the skin, acting as an exfoliating agent and smoothening the skin surface. This can help reduce the signs of skin aging (Kuo et al., 2024). The combination of enzymatic and antibacterial properties produced by symbiotic bacteria makes them suitable candidates for anti-aging, anti-acne, exfoliating, and other skin care products. Protease enzymes and antibacterial compounds from mollusk symbiotic bacteria can work together to enhance antimicrobial activity, thereby improving the preventive and therapeutic effects against various skin problems. This combination allows cosmetic products to disrupt biofilm formation by pathogenic skin bacteria through the hydrolysis of exopolysaccharides (Efremenko et al., 2023). Furthermore, marine-derived protease enzymes and antibacterial compounds are known for their adaptability to extreme environmental conditions, which may provide greater stability in cosmetic formulations (Rathinam et al., 2024). However, research specifically investigating mollusk symbiotic bacteria from Ambon as a source of cosmetic-related bioactive ingredients remains limited. It highlights the urgency of conducting more in-depth examinations of their biological activities and metabolite profiles to support future cosmetic applications while emphasizing environmental sustainability and supporting the blue economy.
The use of protease enzymes and antibacterial compounds from marine biological sources, such as symbiotic bacteria from mollusks, also supports the sustainability of the cosmetics industry. Unlike synthetic chemicals, which can pollute the environment, biological enzymes are eco-friendly, with minimal production waste and reduced carbon emissions (Abu-Tahon et al., 2025). Furthermore, with the growing global awareness of environmentally responsible practices, this innovation has become increasingly relevant and commercially valuable (Kuddus et al., 2024).
In addition to ecological and practical benefits, the development of bioactive-based cosmetic products from mollusk symbiotic bacteria can also be economically beneficial for local communities in the Ambon region through sample collection and bacterial isolation, aligning with the principles of the blue economy concept. This research emphasizes the sustainable management of marine resources while providing economic benefits to local and global communities.
Overall, this research contributes to marine biotechnology and natural marine product innovation, particularly in the field of cosmetics (Fonseca et al., 2023). This research has the potential to yield innovative and high-quality cosmetic products and strengthen scientific and technological development. Ambon waters, located in the Wallacea biogeographic region, represent a unique and biodiverse environment, offering opportunities to discover novel compounds and specific protease enzymes with properties not found elsewhere. By systematically identifying and characterizing these compounds, this study aims to generate validated insights for sustainable and innovative cosmetic applications.
Materials and Methods
Sample collection
Mollusk samples were collected using a random sampling method in the coastal waters of Sopapey Beach, Ambon Region (3°37'27"S 128°18'00"Е) using snorkeling equipment. A total of 6 species were collected from the location. Each specimen was carefully placed in sterile plastic bags, maintained at 4°C, and transported to the laboratory for further analysis. After identifying the collected gastropod samples to the species level (Dharma, 1988), the symbiotic bacteria were isolated from the whole body of the gastropods under aseptic conditions for further characterization (Pringgenies et al., 2024).
Bacterial isolation
The dissected tissues were washed with sterile seawater, and 1 g of tissue was homogenized with sterile seawater in a test tube. Serial dilutions of seawater tissue were then prepared up to 10−5. Bacterial inoculation was performed using the pour plate method by transferring 1,000 µl of the diluted sample onto sterile MNA (Nutrient Agar, Sigma-Aldrich, USA, dissolved in seawater). All inoculation procedures were performed aseptically. Plates were incubated at 28 °C for 48 hours to allow bacterial growth (Masi et al., 2021; Surya et al., 2025).
Antibacterial assay
Pure bacterial isolates were screened for antibacterial activity against selected pathogenic bacteria, namely Cutibacterium acnes and Staphylococcus epidermidis, which are common bacteria on human skin. The antibacterial activity was evaluated using Kirby–Baurer method. Pure cultures of the pathogenic bacteria were grown in NB (Nutrient Broth, Merck, Germany), whereas the symbiotic bacterial isolates were cultured in MNB (Nutrient Broth, Merck, Germany, dissolved in seawater). All cultures were shaken at 150 rpm for 24 hours at 28°C. The turbidity of all bacterial suspensions (isolates and pathogens) was standardized to 0.5 McFarland. Then, the pathogen culture was evenly spread onto MHA (Mueller-Hinton Agar, Merck, Germany). A volume of 20 µl of each gastropod isolate was dispensed onto a sterile paper disk, and a positive control consisting of 1% amoxicillin was included (Pringgenies et al., 2023). All disks were placed on MHA plates inoculated with pathogenic bacteria. Plates were incubated at 28°C, and inhibition zone measurements were performed for 2 × 24 hours (Hanif et al., 2025; Mohamed et al., 2025).
Enzymatic test
Bacterial isolates underwent further enzymatic screening. The tests focused on protease enzymes that are commonly used in industrial and cosmetic applications. Enzyme activity was assessed using the diffusion method on MNA enriched with 1% skim milk. A total of 20 µl of bacterial suspension with a turbidity of 0.5 McFarland was inoculated onto a paper disk and then placed on the agar medium. The plates were incubated at 28°C, and the observations were carried out for 4 × 12 hours. Positive enzymatic activity was indicated by the appearance of clear hydrolysis zones surrounding the colonies (Setyati et al., 2023; Surya et al., 2025).
Identification of bacteria using molecular methods (DNA analysis)
The most promising bacterial isolates were identified using molecular biology techniques. This molecular approach aimed to accurately identify symbiotic bacteria isolated from mollusks in Ambon waters, thereby providing valuable insights into their potential application in the development of cosmetics. Bacterial DNA was extracted from pure bacterial cultures using the Quick-DNA Fungal/Bacterial Miniprep Kit (Zymo Research, USA), and the 16S rRNA gene was amplified using PCR with universal bacterial primers. The amplified PCR products were purified and sequenced, and the resulting sequences were compared with reference sequences in the National Center for Biotechnology Information GenBank database using the BLAST algorithm to determine species-level identification (Pringgenies et al., 2023; Surya et al., 2025).
Gas chromatography-mass spectrometry analysis
Identified bacteria were subjected to a large-scale culture using MNB for 7 days. The culture was extracted using 96% ethanol and evaporated using a rotary evaporator. The resulting crude extract was then analyzed by GC-MS using an Agilent 5977B GC-MS instrument (HP-5MS UI column, 30 m total length, 0.25 µm inner diameter, 0.25 µm film thickness), programmed from 60°C to 280°C with a front inlet flow of 1.00 ml/minute. Helium UHP (He) was used as the carrier gas. The injected compounds were detected as they passed through the column starting at 60°C with a 2 minutes hold time, followed by heating to 280°C at a rate of 10°C/minute with an 8 minutes hold time. The total number of compounds, peak areas, percent area (% area), and compound predictions were interpreted based on the GC-MS database in the system, and the highlighted compounds were drawn using ChemDraw Professional 16.0 software (Pringgenies et al., 2023; Mohamed et al., 2025).
Statistical analysis
Statistical analysis was performed using Rstudio 4.3.2 software. Data were analyzed using one-way analysis of variance (ANOVA) to test whether there were significant differences between the isolates and the control. ANOVA indicated a significant result (p < 0.05), and Tukey’s honest significant difference tests were performed using the agricolae package to identify differences between isolates (Ariyanto and Pringgenies, 2024; Khnissi et al., 2025). Data visualization, including bar plots and boxplots, was created using the ggplot2 package (Nordmann et al., 2022).
Ethical approval
Not needed for this study.
Results
Identification of gastropods and isolation of symbiotic bacteria
Six gastropod species were successfully identified from Ambon waters and used as hosts for the isolation of symbiotic bacteria (Fig. 1), including Cypraea tigris (SPG1), Mauritia arabica (SPG2), Cypraea vitellus (SPG3), Conus querqinus (SPG4), Conus miles (SPG5), and Nebularia eremitarum (SPG6). The mollusk species collected are widely distributed across the Indo-Pacific region, including the Wallacea region, indicating that the sampled gastropods are representative of the tropical marine mollusk communities. The laboratory codes for the isolates were assigned sequentially, with the first part of the code (SPG1-SPG6) indicating each gastropod species, and the number following each code reflects the specific isolate derived from the total set of isolates. The laboratory codes for the isolates were assigned sequentially, with the first part of the code (SPG1-SPG6) indicating each gastropod species, and the number following each code reflects the specific isolate derived from the total set of isolates. Each gastropod species yielded distinct symbiotic bacterial isolates, providing a crucial basis for further enzymatic potential analysis.
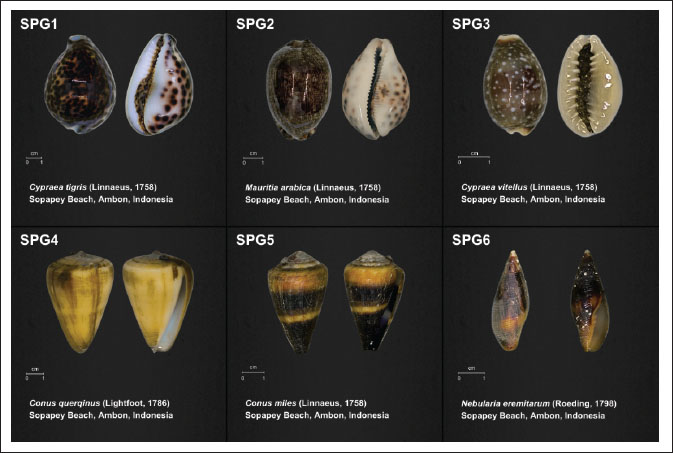
Fig. 1. Host species of Ambon marine gastropod symbiont bacteria.
Antibacterial activity of bacteria
Antibacterial screening was initially performed using the pathogenic bacteria C. acnes and S. epidermidis. Of the 11 isolates tested, two (SPG241 and SPG241.2) showed decent antibacterial activity, while the third (SPG341) exhibited non-significant inhibition (Fig. 2). The assay revealed variation in the inhibitory effects of the isolates against both pathogens. The positive control, 1% amoxicillin, produced consistently high inhibition zones against C. acnes (24.40 ± 0.42 mm at 24 hours and 21.80 ± 0.14 mm at 24 and 48 hours, respectively) and S. epidermidis (21.40 ± 0.57 mm at 24 hours and 21.60 ± 0.99 mm at 24 and 48 hours, respectively). The isolate SPG241 exhibited inhibitory activity against C. acnes of 4.80 ± 0.57 mm at 24 hours and 2.75 ± 0.49 mm at 48 hours. Its activity against S. epidermidis was higher, with inhibition zones of 8.43 ± 1.26 mm in 24 hours and 7.04 ± 0.97 mm in 48 hours. Isolate SPG241.2 showed no activity against C. acnes (0 mm) but inhibited S. epidermidis growth, producing inhibition zones of 8.40 ± 1.27 mm at 24 hours and 4.92 ± 0.79 mm at 48 hours. The difference in letters indicates significant values (p < 0.05).
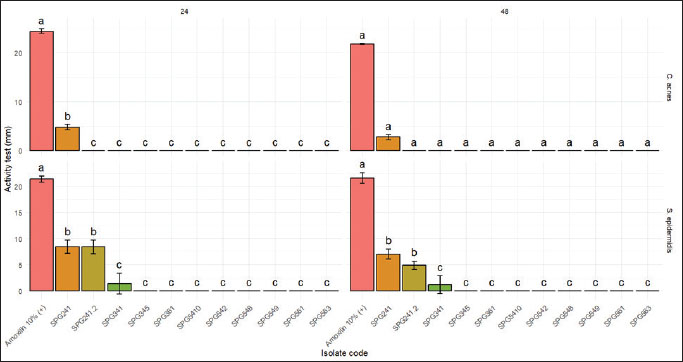
Fig. 2. The antibacterial test of symbiotic bacterial isolates from the Ambon gastropods.
Isolate SPG341 did not exhibit significant antibacterial activity against either pathogen, showing only very small inhibition zones against S. epidermidis (1.40 ± 1.98 mm at 24 hours and 1.19 ± 1.68 mm at 48 hours) and no inhibition against C. acnes. The bar chart results show that SPG241 and SPG241.2 demonstrated moderate inhibition against S. epidermidis, which was significantly different from that of the other isolates. On the other hand, SPG241 showed weak but significant activity against C. acnes at 24 hours, which decreased and became non-significant at 48 hours. In contrast, SPG341 showed minimal inhibition, with no significant differences from the inactive group. These results indicate that only SPG241 and SPG241.2 had antibacterial effects against both pathogens.
Overall, these results indicate that isolates SPG241 and SPG241.2 possess more promising antibacterial activity, particularly against S. epidermidis, with potential for further development as candidate sources of antibacterial agents.
Bacterial proteolytic enzymatic activity
Protease hydrolysis assays were conducted on all symbiotic bacterial isolates from Ambon gastropods to determine their ability to produce proteolytic enzymes. The results revealed variation in hydrolytic activity among isolates at different incubation times (12–48 hours). As shown in Fig. 3, the negative control exhibited no hydrolytic activity throughout the observation period. The SPG241 isolate demonstrated consistently increasing protease activity, with hydrolysis zones measuring 2.25 ± 0.07 mm at 12 hours, rising to 6.15 ± 0.49 mm at 24 hours and 10.40 ± 0.14 mm at 36 hours, and reaching 13.65 ± 0.92 mm at 48 hours. Similar patterns were observed in isolates SPG548, SPG549, and SPG5410, which produced substantial hydrolysis zones, with final values of 12.70 ± 0.85 mm, 13.65 ± 0.07 mm, and 13.85 ± 1.63 mm at 48 hours, respectively.
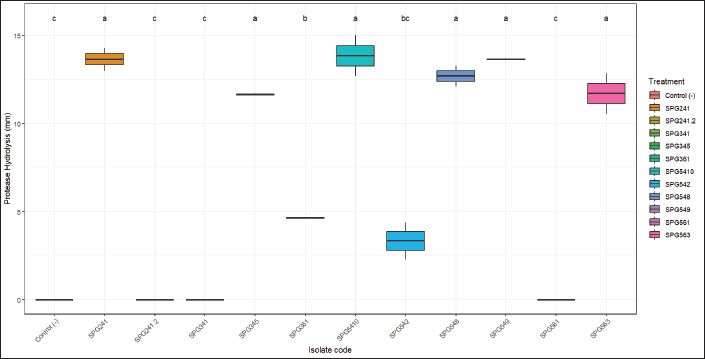
Fig. 3. Proteolytic enzymatic activity of Ambon gastropod symbiotic bacterial isolates.
The SPG345 isolate also displayed protease activity, with hydrolysis zones increasing from 1.90 ± 0.71 mm at 12 hours to 11.65 ± 0.07 mm at 48 hours. Conversely, SPG563 produced relatively stable hydrolysis zones, reaching a maximum of 11.70 ± 1.63 mm at 48 hours. Some isolates displayed low or negligible activity. For instance, SPG361 reached only 4.65 ± 0.07 mm at 48 hours, whereas SPG542 showed a small hydrolysis zone of 3.35 ± 1.49 mm at 48 hours. SPG561 exhibited no proteolytic activity throughout the observation period. Generally, isolates SPG241, SPG345, SPG548, SPG549, SPG5410, and SPG563 can be categorized as having relatively high and stable protease activity compared to the other isolates, indicating the potential of certain symbiotic bacteria from Ambon gastropods as sources of proteolytic enzymes relevant to cosmetic applications, particularly in skin care formulations requiring protease activity.
Among the six isolates, strain SPG241 was particularly noteworthy because it had previously demonstrated significant antibacterial activity against S. epidermidis in addition to exhibiting relatively high protease activity. Its superior multi-activity profile (antibacterial and proteolytic) led to its selection for GC-MS analysis as a candidate for the development of cosmetic ingredients.
Molecular identification of the selected bacteria
The 16S rRNA gene sequence analysis revealed that the symbiotic bacterial isolates from Ambon gastropods exhibited high similarity to bacterial species registered in the GenBank database (Table 1). Isolate SPG241 was identified as Pseudoalteromonas citrea with 100% query coverage and 100% identity (Accession Number NR_037073). Isolate SPG345 showed high homology with Pseudoalteromonas peptidolytica, with 99.86% identity (NR_113971).
Table 1. BLAST homology analysis of symbiotic bacteria from marine gastropods.
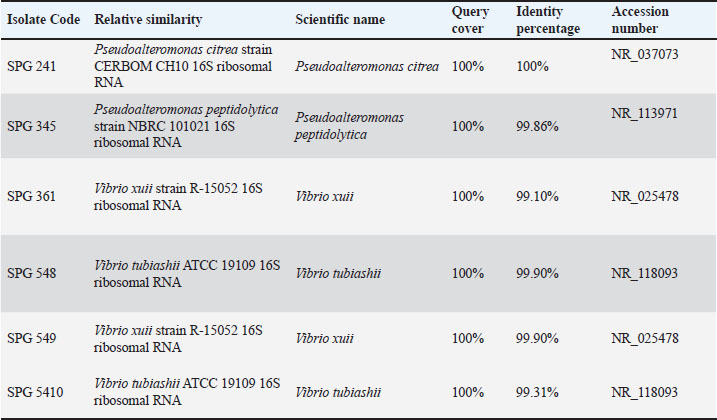
Isolates SPG361 and SPG549 were both identified as Vibrio xuii, with identities of 99.10% and 99.90%, respectively (NR_025478). Isolates SPG548 and SPG5410 were identified as Vibrio tubiashii, with identities of 99.90% and 99.31%, respectively (NR_118093).
These results confirm that the symbiotic bacteria isolated from Ambon gastropods were predominantly from Pseudoalteromonas and Vibrio genera. Both genera are renowned for producing bioactive metabolites, including enzymes with proteolytic and antimicrobial activities, relevant for the development of active ingredients in cosmetic applications.
Chromatographic analysis of the symbiont metabolites
GC-MS analysis of extracts from symbiotic bacterial isolates of Ambon gastropods revealed 33 compounds with varying similarity indices. As shown in Figure 4, several major compounds dominating the analysis were represented by peaks with relatively high areas. The dominant compounds identified included isopropyl alcohol, with a relative area of 28.53% (tR 4.36 minutes), and 9-hexadecenoic acid, methyl ester, (Z)-, with a relative area of 26.78% (tR 17.30 minutes). Additionally, (Z)-methyl hexadec-11-enoate was detected in a substantial proportion of 18.51% (tR 17.44 minutes). Other compounds present at lower proportions included ethanol, 2-nitro- (7.94%), 10-octadecenoic acid, methyl ester (4.76%), and methyl tetradecanoate (3.09%). Several compounds, namely methyl palmitate, 2,3-butanediol, and 2-piperidinone, can be highlighted for their ability as bioactive ingredients for cosmetic products (Fig. 5).
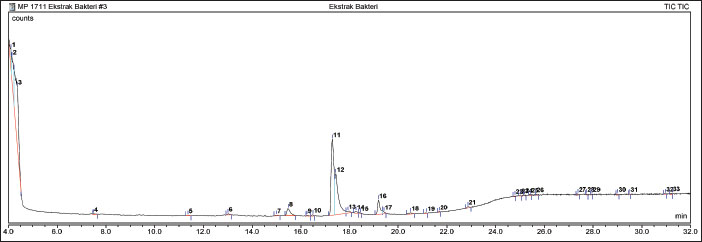
Fig. 4. Chromatographic analysis of the symbiont strain SPG241.
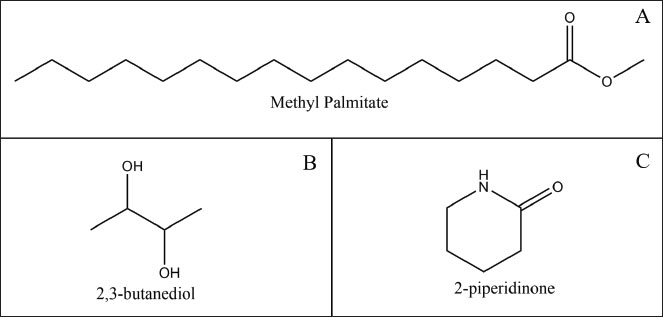
Fig. 5. Chemical structures of methyl palmitate (A), 2,3-butanediol (B), and 2-piperidinone (C).
Furthermore, several siloxane derivatives (hexasiloxane and octasiloxane) and steroid derivatives, such as ethyl iso-allocholate, were detected, although their relative areas were minor (<1%). These components may originate from bacterial secondary metabolites or represent contaminants introduced during the extraction process.
Proteolytic activity significantly differed among the symbiotic bacterial isolates (p < 0.05), as shown in Fig. 3. No hydrolysis was detected in the negative control and three isolates (SPG341.2, SPG641, and SPG631), indicating the absence of detectable protease production. In contrast, SPG345 and SPG6410 exhibited the highest protease activity, with hydrolysis zones exceeding 15 mm, and were statistically distinct from the low-activity isolates (group c). Several isolates, including SPG241, SPG241.2, SPG341, SPG361, SPG648, SPG649, and SPG653, clustered within the same significance group (group a), with hydrolysis zones ranging from approximately 12 to 16 mm. SPG642 and SPG361 formed an intermediate group (group bc), exhibiting moderate activity of 5–12 mm. These results indicate that multiple isolates possess strong protease activity, with SPG345 and SPG6410 demonstrating the greatest enzymatic potential.
Discussion
Six gastropod species were successfully identified from Ambon waters, which were used as hosts for symbiotic bacterial isolation. Ambon is in the Wallacea region, a biogeographic zone delineated by the Wallace Line, which is a major faunal boundary between Asia and Australia, reflecting the complexity and uniqueness of its marine ecosystems (Ali and Heaney, 2023). This makes the Wallacea waters, including Ambon, a marine biodiversity hotspot with high potential for exploring symbiotic microbes with unique characteristics.
Isolation was performed from the entire tissues of the gastropods, including the gills, mantle, and digestive glands. This approach provides a high probability of obtaining symbiotic bacteria with various biological activities, as different internal microhabitats in mollusks can support diverse microbial communities. The genera Pseudoalteromonas and Vibrio are commonly found as symbionts in marine organisms, including mollusks and nudibranchs in the Maluku region and its surroundings, with the ability to produce bioactive metabolites (Ng et al., 2022; Amran et al., 2025). The biogeographic context of Wallacea further underscores the significance of these findings, as this region remains underexplored compared to other areas within the Coral Triangle. The combination of geographic factors, geological history, and complex ocean currents has facilitated the formation of distinctive marine communities, including microbial symbionts (Ngandjui et al., 2024). Therefore, the isolation of bacteria from Ambon gastropods not only provides a scientific basis for biotechnological studies but also contributes to a broader understanding of the Wallace Line’s unique biodiversity.
Antibacterial assays revealed that only three isolates exhibited inhibitory activity against C. acnes and S. epidermidis. SPG241 and SPG241.2 demonstrated the most pronounced activity, particularly against S. epidermidis (Fig. 2). This is consistent with previous studies reporting that marine symbiotic bacteria tend to produce bioactive metabolites with a narrow spectrum of activity, yet they can exhibit high efficacy against specific targets (Srinivasan et al., 2021).
The inhibition zones produced by SPG241 against S. epidermidis (8.43 mm at 24 hours and 7.04 mm at 48 hours) and SPG241.2 (8.40 mm at 24 hours and 4.92 mm at 48 hours) indicate moderate antibacterial activity that remained stable up to 48 hours. The slight decrease in antibacterial activity between 24 and 48 hours may be attributed to the isolate exhibiting bacteriostatic action, in which growth inhibition of the pathogen is reflected by a reduction in viable bacterial cells (Saleem et al., 2022). Staphylococcus epidermidis is an important component of the human skin microbiota and can function as an opportunistic pathogen under certain conditions, particularly in biofilm formation, which triggers inflammation and exacerbates skin conditions (Severn and Horswill, 2023).
The activity against C. acnes was relatively low but remains significant, as this bacterium is a primary causative agent of acne (Fournière et al., 2020). The positive control, 1% amoxicillin, produced much larger inhibition zones than the symbiont isolates, confirming that marine bacterial metabolites are not equivalent to synthetic antibiotics in terms of inhibitory potency. However, in the context of cosmetics, moderate activity is actually preferable because it does not drastically disrupt the skin microbiota balance but can function as a skin microbiome balancer (De Lucas et al., 2024).
These findings further highlight Ambon waters as a potential source of symbiotic bacteria with unique metabolite profiles. Only a small fraction of isolates (2.4% of the total) exhibited activity, indicating high selectivity in the production of bioactive metabolites. This selectivity may be influenced by the unique ecological conditions of Wallacea, including variable ocean currents and the complex habitats of gastropods, which can promote the formation of microbial communities with specialized metabolic traits (Cui et al., 2024). Therefore, SPG241 and SPG241.2 isolates can be considered potential candidates for development as natural antibacterial agents in cosmetic products, particularly acne-care formulations, which require both skin-friendly and environmentally sustainable active ingredients.
The enzymatic assays demonstrated variation in proteolytic hydrolysis capabilities among the symbiotic bacterial isolates from Ambon gastropods, which showed significant hydrolysis zones over the incubation period, reaching final values greater than 11 mm at 48 hours. This consistent increase indicates active and sustained protease production (Fig. 3). These findings are consistent with previous reports indicating that marine symbiotic bacteria, particularly from the genera Vibrio and Pseudoalteromonas, are well-known producers of proteolytic enzymes (Cheng et al., 2020). Proteases derived from marine microbes exhibit high stability and are often functional under extreme conditions, such as high pH or salinity, making them highly attractive for industrial and cosmetic applications (Rao et al., 2022).
In the context of cosmetics, proteases play a crucial role in skin care formulations, particularly as exfoliating agents to remove dead skin cells, accelerate epidermal regeneration, and enhance the absorption of other active ingredients. Enzymes in cosmetic products promote exfoliation of the epidermal surface layer, thereby increasing the skin’s ability to absorb air and other ingredients (Ioannou and Labrou, 2022; Kuo et al., 2024). Enzymes derived from marine bacteria tend to exhibit higher stability and greater attractiveness for industrial and cosmetic applications due to their ability to adapt to fluctuating environmental conditions, such as variations in pH, salinity, temperature, nutrient availability, and other extreme stresses (Rao et al., 2022). The stable protease activity observed up to 48 hours, as demonstrated by isolates SPG241 and SPG5410, indicates significant potential for long-term topical applications, such as in anti-aging creams or skin-brightening products. These results underscore the importance of exploring symbiotic bacteria in the Wallacea region. Ambon waters, located in this biogeographic zone, are known for their high biodiversity and unique environmental conditions influenced by the Wallace Line (White et al., 2021). These ecological factors may promote the formation of microbial communities with distinctive metabolic and enzymatic pathways, including proteases with properties that differ from those of conventional sources.
Therefore, SPG241, SPG345, SPG548, SPG549, SPG5410, and SPG563 isolates can be considered as potential sources of natural proteolytic enzymes. This opens opportunities for the development of novel, environmentally friendly active ingredients in alignment with global trends toward sustainable and natural-based cosmetic products (Matos et al., 2024).
16S rRNA gene sequence analysis provided clear taxonomic information regarding the symbiotic bacterial isolates from Ambon gastropods. SPG241 exhibited 100% homology with P. citrea, while SPG345 showed close similarity to P. peptidolytica (99.86%). Additionally, SPG361 and SPG549 were identified as V. xuii, and SPG548 and SPG5410 were identified as V. tubiashii, with sequence identities exceeding 99%.
These findings are significant because Pseudoalteromonas spp. and Vibrio spp. have long been recognized as dominant symbionts in marine invertebrates, including mollusks, sponges, and tunicates (Pringgenies et al., 2024). The genus Pseudoalteromonas is widely known for producing bioactive metabolites, such as pigments, antibacterial and antifungal compounds, and enzymatic substances with high biotechnological potential (Chau et al., 2021). Several Vibrio species are capable of producing proteases, lipases, and other industrially valuable enzymes, although they are sometimes associated with pathogenicity in marine organisms (Mamangkey et al., 2021).
The identification of P. citrea as one of the primary isolates aligns with the results of the antibacterial assay, in which SPG241 exhibited significant activity against S. epidermidis. Pseudoalteromonas species produce antimicrobial compounds, such as tropodithietic acid and yellow pigments, which function as symbiont defense agents (Setiyono et al., 2020). The close similarity of isolate SPG345 to P. peptidolytica reinforces the potential of this genus as a producer of proteolytic enzymes, consistent with the high hydrolysis zones observed in enzymatic assays.
The dominance of the genus Vibrio among the isolates (SPG361, SPG548, SPG549, and SPG5410) also reflects the strong ecological association between gastropods and tropical marine microbial communities. Although known as a bivalve larval pathogen, V. tubiashii possesses a complex metabolic system, including protease production, which can be harnessed for industrial and cosmetic applications (Richards et al., 2021). Isolate SPG5410, exhibiting the highest protease activity, corresponds to its identification as V. tubiashii, further supporting the correlation between molecular identity and functional profile. Although these Vibrio are considered opportunistic in marine organisms, they are not opportunistic in humans, thereby retaining the potential for human-oriented applications (Fleischmann et al., 2022; Wang et al., 2022; Kim et al., 2025).
These findings were obtained from Ambon gastropods, located in the Wallacea region. Wallacea represents a unique biogeographic zone with high levels of endemism due to its position between the Sunda and Sahul shelves, separated by the Wallace Line (Trethowan, 2020). The distinct geographical and ecological conditions of this region enable the formation of microbial communities with unique metabolic traits that are rarely found elsewhere. The presence of Pseudoalteromonas and Vibrio as symbionts of Ambon gastropods not only enriches the understanding of marine biodiversity in Wallacea but also offers significant opportunities for discovering new candidates for natural-based cosmetic applications.
GC-MS analysis of the symbiotic bacterial isolate SPG241 from Ambon gastropods successfully identified several bioactive metabolites with potential cosmetic applications. It revealed a dominance of fatty acid methyl esters (FAMEs), including hexadecanoic acid, methyl palmitate, trans-13-octadecenoic acid, and methyl ester, as well as specific metabolites such as 2,3-butanediol and 2-piperidinone (Fig. 2). This profile confirms that the symbiont is a rich source of secondary metabolites conducive to the development of active cosmetic ingredients.
FAMEs, including methyl palmitate and C18 derivatives, exhibit emollient, antioxidant, and anti-inflammatory activities that are crucial for skin health. Methyl palmitate restores the epidermal lipid layer, reduces transepidermal water loss, and functions as an anti-inflammatory agent. It can also inhibit the growth of certain pathogenic bacteria, making it a potential candidate for anti-acne, moisturizing, and anti-aging formulations (Shu et al., 2023).
The presence of 2,3-butanediol, a natural humectant that enhances skin hydration through its water-binding capacity, adds functional value to the extract. This property supports the stabilization of cosmetic formulations and provides an environmentally friendly alternative to synthetic glycols commonly used in the cosmetic industry (Ding et al., 2022). Additionally, 2-piperidinone, a cyclic compound with antibacterial activity, holds potential as a natural preservative. It can inhibit the growth of opportunistic skin microbes, thereby enhancing the safety and stability of cosmetic products while providing an alternative to synthetic preservatives, whose use has been increasingly questioned (Das et al., 2025).
The variation in metabolites produced by the symbiotic isolates is consistent with previous findings that Pseudoalteromonas and Vibrio, as the main symbionts of Ambon gastropods, are significant producers of bioactive compounds. These results are further supported by antibacterial and enzymatic assays demonstrating high functional activity in certain isolates (SPG241, SPG5410). The combination of molecular and chemical metabolite data provides a comprehensive picture that Ambon gastropod symbionts are promising candidates for cosmetic bioprospecting. Combined, these results indicate that the symbiotic bacterial isolate from Ambon gastropods produces a diverse array of bioactive compounds, particularly unsaturated fatty acid esters, alcohols, and steroid derivatives, known to possess biological activities relevant to cosmetic applications, including antimicrobial, antioxidant, and emollient properties. Dominant compounds, such as 9-hexadecenoic acid, methyl ester (Z), and (Z)-methyl hexadec-11-enoate, are of particular interest because they are recognized for their roles in maintaining skin hydration, exhibiting anti-aging effects, and providing protection against free radicals.
The Wallacea biogeographic context further underscores these findings’ uniqueness. The waters surrounding Ambon, located along the Wallace Line, represent a transitional ecosystem with exceptionally high marine biodiversity (Hawkins et al., 2024). Such ecological distinctiveness enables mollusk symbionts in this region to produce secondary metabolites with chemical profiles, including rarely reported bioactive compounds, that differ from those of other areas. Consequently, these GC-MS results have implications not only for the development of natural-based active cosmetic ingredients but also for reinforcing Wallacea’s status as a hotspot for innovative marine metabolite exploration.
Conclusion
A total of 11 symbiotic bacterial mollusk isolates were obtained from the Sopapey waters, Ambon region. This study identified six potential isolates, namely SPG241, SPG345, SPG361, SPG548, SPG549, and SPG5410, which demonstrated notable variations in enzymatic activity and antibacterial activity against skin pathogenic bacteria. Molecular identification revealed that these isolates belonged to P. citrea, P. peptidolytica, V. xuii, and V. tubiashii. Among all isolates, P. citrea exhibited the highest performance and contained fatty acids, alcohols, and lactams. These findings open opportunities for the development of environmentally friendly and innovative active ingredients while emphasizing the importance of conserving marine biodiversity for future biotechnological applications.
Acknowledgments
The authors would like to express their gratitude to the Rector of Universitas Diponegoro for the support in strengthening research capacity and international collaboration through the Visiting Professor Offline World Class University Program of the Faculty of Fisheries and Marine Science, as stated in Rector Decree No. 31/UN7.A/HK/X/2025.
Conflict of interest
The authors have no conflicts of interest to declare.
Funding
The Indonesian Directorate General of Higher Education supported this research through the PMDSU program (grant number: 359-077/UN7.D2.1/PP/V/2032). We gratefully acknowledge their financial and institutional support.
Authors’ contributions
All authors equally contributed to the design, execution, analysis, and writing of this manuscript.
Data availability
All data generated or analyzed during this study are included in the manuscript.
References
Abu-Tahon, M.A., Housseiny, M.M., Aboelmagd, H.I., Daifalla, N., Khalili, M., Isichei, A.C., Ramadan, A., Abu El-saad, A.M., Seddek, N.H., Ebrahim, D., Ali, Y.H., Saeed, I.K., Rikabi, H.A. and Eltaib, L. 2025. A holistic perspective on the efficiency of microbial enzymes in bioremediation process: mechanism and challenges: A review. Int. Int. J. Biol. Macromol. 308, 142278; doi:10.1016/j.ijbiomac.2025.142278
Ali, J.R. and Heaney, L.R. 2023. Alfred R. Wallace’s enduring influence on biogeographical studies of the Indo-Australian archipelago. J. Biogeogr. 32(40), 32–40; doi:10.1111/jbi.14470
Alnuqaydan, A.M. 2024. The dark side of beauty : an in-depth analysis of the health hazards and toxicological impact of synthetic cosmetics and personal care products. Front. Public Health 12, 1439027; doi:10.3389/fpubh.2024.1439027
Amran, R.H., Jamal, M.T., Bowrji, S., Sayegh, F., Santanumurti, M.B. and Satheesh, S. 2025. Mini review : antimicrobial compounds produced by bacteria associated with marine invertebrates. Folia Microbiol. 70, 271–292; doi:10.1007/s12223-024-01209-5
Ariyanto, D. and Pringgenies, D. 2024. Nutrient Distribution in Natural and Rehabilitated Mangroves with Various Types of Mangroves, Lampung, Indonesia. Int. J. Des. Nat. Ecodyn. 19(6), 2171–2178; doi:10.18280/ijdne.190633
Chau, R., Pearson, L.A., Cain, J., Kalaitzis, J.A. and Neilan, B.A. 2021. A Pseudoalteromonas clade with remarkable biosynthetic potential. Appl. Environ. Microbiol. 87(6), 1–16; doi:10.1128/AEM.02604-20
Cheng, T.H., Ismail, N., Kamaruding, N., Saidin, J. and Danish-Daniel, M. 2020. Industrial enzymes-producing marine bacteria from marine resources. Biotechnol. Rep. 27, 482; doi:10.1016/j.btre.2020.e00482
Das, S.S., Talukder, A., Sarma, D., Porasar, P., Sahariah B.J., Majumder, M., Dua, T.K. and Dutta, K.N. 2025. In silico research in biomedicine exploring the therapeutic potential of three indigenous chillies of North East India : chemical composition, ADME properties and antibacterial activity. Silico. Res. Biomed. 1, 10005; doi:10.1016/j.insi.2025.100005
Dharma, B. 1988. Indonesian shells. PT. Denpasar, Indonesia: Sarana Graha, p: 135.
Ding, J., Wu, B. and Chen, L. 2022. Application of marine microbial natural products in cosmetics. Front. Microbiol. 13, 892505; doi:10.3389/fmicb.2022.892505
Fleischmann, S., Herrig, I., Wesp, J., Stiedl, J., Reifferscheid, G., Strauch, E., Alter, T. and Brennholt, N. 2022. Prevalence and distribution of potentially human pathogenic Vibrio spp. on German North and Baltic Sea Coasts. Front. Cell. Infect. Microbiol. 12, 1–15; doi:10.3389/fcimb.2022.846819
Fonseca, S., Amaral, M.N., Reis, C.P. and Custódio, L. 2023. Marine natural products as innovative cosmetic ingredients. Mar. Drugs. 21, 170; doi:10.3390/md21030170
Fournière, M., Latire, T., Souak, D., Feuilloley, M.G.J. and Bedoux, G. 2020. Staphylococcus epidermidis and Cutibacterium acnes: two major sentinels of skin microbiota and the influence of cosmetics. Microorganisms 8, 1752; doi:10.3390/microorganisms8111752
Hanif, M.I., Pringgenies, D. and Setyati, W.A. 2025. Pigmented gastropods-assosiate bacteria: an innovation in natural antioxidants, antibacterials, and sun protection. Hayati J. Biosci. 32(6), 1453–1465; doi:10.4308/hjb.32.6.1453-1465
Hawkins, S., Zetika, G.A., Kinaston, R., Firmando, Y.R., Sari, D.M., Suniarti, Y., Lucas, M., Roberts, P., Reepmeyer, C., Maloney, T., Kealy, S., Stirling, C., Reid, M., Barr, D., Kleffmann, T., Kumar, A., Yuwono, P., Litster, M., Husni, M., Ririmasse, M., Mahirta., Mujabuddawat, M., Harriyadi. and O’Connor, S. 2024. Earliest known funerary rites in Wallacea after the last glacial maximum. Sci. Rep. 1(18), 1–18; doi:10.1038/s41598-023-50294-y
Ioannou, E. and Labrou, N.E. 2022. Development of enzyme-based cosmeceuticals: studies on the proteolytic activity of Arthrospira platensis and its efficient incorporation in a hydrogel formulation. Cosmetics 9, 106; doi:10.3390/cosmetics9050106
Khnissi, S., Maaloul, R., Chalouati, H., Mara, L., Aouadi, D., Adouani, B., Dattena, M., Khémiri, I. and Fattouch, S. 2025. Nanotechnology-assisted cryopreservation of ovine semen: evaluation of Thymus vulgaris essential oil as a natural antioxidant. Front. Anim. Sci. 5, 1479602; doi:10.3389/fanim.2024.1479602
Kim, H.J., Park, K.I. and Kim, S.G. 2025. Genomic and pathogenic characterization of Vibrio alginolyticus CG—1 Isolated from Crassostrea gigas. Curr. Microbiol. 82, 556; doi:10.1007/s00284-025-04552-z
Kuddus, M., Roohi., Bano, N., Sheik, G.B., Joseph, B., Hamid, B., Sindhu, R. and Madhavan, A. 2024. Cold-active microbial enzymes and their biotechnological applications. Microb. Biotechnol. 17(4), 1–19; doi:10.1111/1751-7915.14467
Kuo, C.H., Wang, H.M.D. and Shieh, C.J. 2024. Enzymes in biomedical, cosmetic and food application. Catalysts 14, 162; doi:10.3390/catal14030162
Liu, J.K. 2022. Natural products in cosmetics. Nat. Prod. Bioprospect. 12(1), 40; doi:10.1007/s13659-022-00363-y
De Lucas, R., Martínez, H., Nieto, C., Ruiz, C., Rafael, A., Nerea, B., Garcia-, S., Gonzalez-, S.P., Palacios, D., Guerra, M.A. and Bou, L. 2024. New clinical approach in facial mild—moderate acne : stabilization of skin microbiota balance with a topical biotechnological phytocomplex. J. Cosmet. Dermatol. 3616(3627), 3616–3627; doi:10.1111/jocd.16452
Mamangkey, J., Suryanto, D., Munir, E., Mustopa, A.Z., Sibero, M.T., Mendes, L.W., Hartanto, A., Taniwan, S., Ek-Ramos, M.J., Harahap, A., Verma, A., Trihatmoko, E., Putranto, W.S., Pardosi, L. and Rudia, L.O.A.P. 2021. Isolation and enzyme bioprospection of bacteria associated to Bruguiera cylindrica, a mangrove plant of North Sumatra, Indonesia. Biotechnol. Rep. 30, 617; doi:10.1016/j.btre.2021.e00617
Matos, M., Custódio, L. and Reis, C.P. 2024. Marine Invasive algae’s bioactive ingredients as a sustainable pathway in cosmetics: the Azores Islands as a case study. Mar. Drugs 22, 575; doi:10.3390/md22120575
Mohamed, H., Marusich, E., Afanasev, Y. and Leonov, S. 2022. Bacterial outer membrane permeability increase underlies the bactericidal effect of fatty acids from Hermetia illucens (black soldier fly) larvae fat against hypermucoviscous isolates of Klebsiella pneumoniae. Front. Microbiol. 13, 844811; doi: 10.3389/fmicb.2022.844811
Mohamed, H., Marusich, E., Pustovalova, M. and Leonov, S. 2025. Mechanism of bactericidal efficacy against nosocomial pathogenic Staphylococcus aureus strain caused by fatty acids from Hermetia illucens larvae fat. Sci. Rep. 15, 30305; doi:10.1038/s41598-025-15858-0
Ng, M.S., Soon, N., Chang, Y. and Wainwright, B.J. 2022. Bacterial and fungal co-occurrence in the nudibranch, Pteraeolidia semperi. Life 12(12), 1988; doi:10.3390/life12121988
Ngandjui, Y.A.T., Kereeditse, T.T., Kamika, I., Madikizela, L.M. and Msagati, T.A.M. 2024. Nutraceutical and medicinal importance of Marine Molluscs. Mar. Drugs. 22, 201; doi:10.3390/md22050201
Nirmal, N., Demir, D., Ceylan, S., Ahmad, S., Goksen, G., Koirala, P. and Bono, G. 2024. Polysaccharides from shell waste of shellfish and their applications in the cosmeceutical industry: a review. Int. J. Biol. Macromol. 265, 131119; doi:10.1016/j.ijbiomac.2024.131119
Nordmann, E., Mcaleer, P., Toivo, W., Paterson, H. and Debruine, L.M. 2022. Data visualization using R for researchers who do not use R. Adv. Meth. Pract. Psychol. Sci. 5(2), doi: 10.1177/25152459221074654
Pringgenies, D., Soenardjo, N., Setyati, W.A. and Pramesti, R. 2024. Secondary metabolites from symbiotic bacteria of seagrass and molluscs as a reference for natural food preservatives. Trends Sci. 21(5), 7404; doi:10.48048/tis.2024.7404
Pringgenies, D., Wilis, A.S., Feliatra, F. and Ariyanto, D. 2023. The antibacterial and antifungal potential of marine natural ingredients from the symbiont bacteria of mangrove. Glob. J. Environ. Sci. Manage. 9(4), 819–832; doi:10.22035/gjesm.2023.04.11
Rao, A.S., Nair, A., Nivetha, K., More, V.S., Anantharaju, K.S. and More, S.S. 2022. Molecular adaptations in proteins and enzymes produced by extremophilic microorganisms. In Extremozymes and their Industrial Applications. INC, pp: 205–30. https://doi.org/ 10.1016/B978-0-323-90274-8.00002-2
Rathinam , W.V.C.G., Bragadeeswaran, S., Kumaresan, S., Gunamathy, K., Visnu, B., Asarudeen, J.M., Sasidharan, T and Srikavibharathi, S. 2024. Marine resources: a sustainable and promising source for cosmetic industries. Multidisciplinary Applications of Marine Resources: A Step Towards Green and Sustainable Future, 103–140; doi:10.1007/978-981-97-5057-3_6
Richards, G.P., Watson, M.A., Madison, D., Soffer, N., Needleman, D.S., Soroka, D.S., Uknalis, J., Baranzoni, G.M., Church, K.M., Polson, S.W., Elston, R., Langdon, C. and Sulakvelidze, A. 2021. Bacteriophages against Vibrio coralliilyticus and Vibrio tubiashii: isolation, characterization, and remediation of larval oyster mortalities. Appl. Environ. Microbiol. 87(10), e00008; doi:10.1128/AEM.00008-21
Saleem, N., Ryckaert, F., Snow, T.A.C., Satta, G., Singer, M. and Arulkumaran, N. 2022. Mortality and clinical cure rates for pneumonia: a systematic review, meta-analysis, and trial sequential analysis of randomized control trials comparing bactericidal and bacteriostatic antibiotic treatments. Clin. Microbiol. Infect. 28, 936–945; doi:10.1016/j.cmi.2021.12.021
Setiyono, E., Adhiwibawa, M.A.S., Indrawati, R., Prihastyanti, M.N.U., Shioi, Y. and Brotosudarmo, T.H.P. 2020. An Indonesian marine bacterium, Pseudoalteromonas rubra, produces antimicrobial prodiginine pigments. ACS. Omega. 5(9), 4626–4635; doi:10.1021/acsomega.9b04322
Setyati, W.A., Pringgenies, D., Soenardjo, N. and Pramesti, R. 2023. Enzyme-producing symbiotic bacteria in gastropods and bivalves molluscs: candidates for bioindustry materials. Biodiv 24(1), 20–25; doi:10.13057/biodiv/d240103
Severn, M.M. and Horswill, A.R. 2023. Staphylococcus epidermidis and its dual lifestyle in skin health and infection. Nat. Rev. Microbiol. 21(2), 97–111; doi:10.1038/s41579-022-00780-3
Shu, P., Li, M., Zhao, N., Wang, Y., Zhang, L. and Du, Z. 2023. Efficacy and mechanism of retinyl palmitate against UVB-induced skin photoaging. Front. Pharmacol. 14, 1–13; doi:10.3389/fphar.2023.1278838
Siahaan, E.A., Agusman., Pangestuti, R., Shin, K.H. and Kim, S.K. 2022. Potential cosmetic active ingredients derived from marine by-Products. Mar. Drugs 20, 734; doi:10.3390/md20120734
Srinivasan, R., Kannappan, A., Shi, C. and Lin, X. 2021. Marine bacterial secondary metabolites: a treasure house for structurally unique and effective antimicrobial compounds. Mar. Drugs 19(10), 1–36; doi:10.3390/md19100530
Surya, S., Pringgenies, D. and Sedjati, S. 2025. Antibacterial and enzymatic activities of symbiotic bacteria from gastropods and bivalves in marine skincare applications. Int. J. Des. Nat. Ecodyn. 20(1), 83–90; doi:10.18280/ijdne.200109
Trethowan, L.A. 2020. Could the environment limit dispersal between Sunda and Sahul?. J. Veg. Sci. 1(7), 1–7; doi:10.1111/jvs.12978
Trevisol, T.C., Henriques, R.O., Souza, A.J.A. and Furigo, A. 2022. An overview of the use of proteolytic enzymes as exfoliating agents. J. Cosmet. Dermatol. 21, 3300–3307; doi:10.1111/jocd.14673
Wang, R., Liu, X., Wang, J., Huizhu., Lin, X., Sun, J., Mou, H., Zhang, T. and Ma, X. 2022. Proteomic differences between Vibrio tubiashii strains with high—or low—virulence levels isolated from diseased ivory snail Babylonia areolata. Aquac. Int. 30, 2579–2591; doi:10.1007/s10499-022-00919-2
White, A.E., Dey, K.K., Stephens, M. and Price, T.D. 2021. Dispersal syndromes drive the formation of biogeographical regions, illustrated by the case of Wallace’s Line. Glob. Ecol. Biogeogr. 30, 685–696; doi:10.1111/geb.13250














