| Research Article | ||
Open Vet. J.. 2026; 16(2): 1112-1123
Open Veterinary Journal, (2026), Vol. 16(2): 1112-1123 Research Article Experimental esophageal reconstruction in dogs using autologous platelet-rich plasma-augmented acellular amniotic membrane scaffoldBassim Kadhim Khashjoori1, Heidar Rasheed Alrafas2, Mukhalad Abdulkareem3, Murtadha Abbas4* and Ammar Maatoq Hashim11University of Basrah, College of Veterinary Medicine, Department of Surgery and Obstetrics, Basra, Iraq 2Department of Microbiology, University of Basrah, College of Veterinary Medicine, Basra, Iraq 3Department of Pathology and Forensic Medicine, University of Misan, College of Medicine, Maysan, Iraq 4Department of Public Health, Faculty of Veterinary Medicine, University of Kufa, Kufa, Iraq *Corresponding Author: Murtadha Abbas. Department of Public Health, Faculty of Veterinary Medicine,University of Kufa, Kufa, Iraq. Email: murtadhaa.alghazali [at] uokufa.edu.iq Submitted: 20/09/2025 Revised: 09/01/2026 Accepted: 25/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
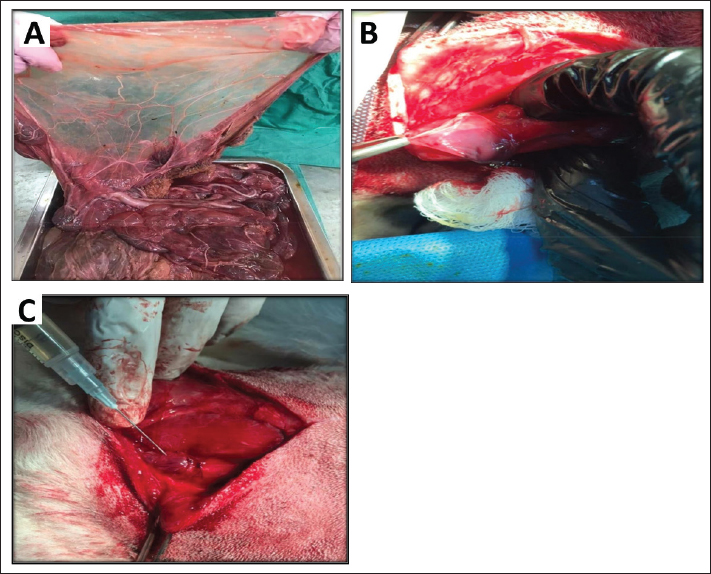
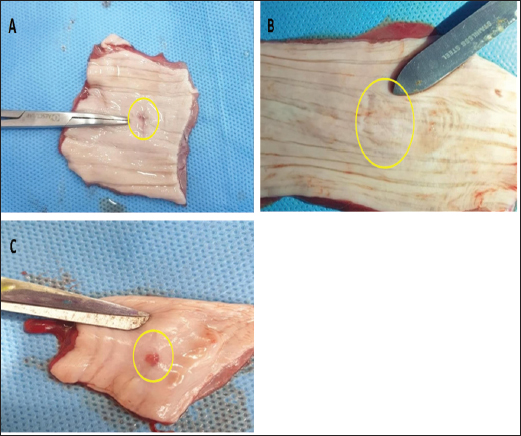
AbstractBackground: Surgical treatment of esophageal diseases in pets is associated with high morbidity and postoperative complications. Depending on the degree of tissue loss, the excision may be replaced with a patch or segmental esophagoplasty. Autografts are commonly used for esophageal repair. Autografts from vital organs expose the animal to unnecessary laparotomies or surgery on healthy organs. This has led to the development of several biological materials for esophageal repair. Aim: This study investigated the use of acellular amniotic membrane as a support structure for esophageal reconstruction in dogs supplemented with platelet-rich plasma. Methods: This experimental study was conducted with 15 mongrel dogs. The patients were divided into three equal groups: the first group was treated with acellular amniotic membrane accompanied by autologous platelet-rich plasma, the second group was treated with acellular amniotic membrane only, and the third group utilized a pedicled flap of the sternohyoideus muscle alone for closing a circumferential esophageal defect. Results: The dogs were evaluated 30 days after treatment, and the esophageal defects in the first group healed better than those in the other groups. Conclusion: The use of acellular amniotic membrane as a bioscaffold, combined with platelet-rich plasma, may be a viable option for esophageal reconstruction given the encouraging results. Keywords: Acellular amniotic membrane, Esophagus, Platelet-rich plasma, Tissue healing. IntroductionSurgery is necessary to restore continuity when the esophagus has lost several centimeters of length, especially when primary anastomosis is not feasible due to expected stress. Depending on the extent of tissue damage, patch or segmental esophoplasty is suitable for replacing resections caused by tumors, strictures, diverticula, perforation, and nonviable segments of the esophagus following pressure necrosis. The maximum length of the esophagus that can be removed is one restriction on esophageal resection. The esophagus is attached distally to the stomach and diaphragm and proximally to the pharynx; thus, its range of motion is restricted (Hashem et al., 2023). The esophagus is affected by numerous conditions and diseases. Esophageal surgery is required for these esophageal diseases, which are frequently linked to high rates of complications, including tightness, wound dehiscence, and leaks. The esophagus, unlike other digestive organs, lacks serosa and omentum; longitudinal tension in the esophageal tissue is weak; the esophagus has a segmental blood supply; and the suture site is subject to continuous motion. These factors are considered predisposing factors for poor healing. Normal esophageal healing is multifactorial, and it fails when any phase of the healing process is disrupted (Al-Ajeliet al., 2022). The amniotic membrane is now used as a biological dressing and graft in different fields, including orthopedics, ophthalmology, dermatology, and plastic surgery. In addition to containing a wealth of growth factors, anti-inflammatory cytokines, and extracellular matrix components, it acts as a natural scaffold (Parmar et al., 2025). In addition, it improves epithelialization by encouraging epithelial cell migration, strengthening basal epithelial cell adhesion, and encouraging epithelial differentiation to preserve the epithelial phenotype (Krysik et al., 2020). It also controls blood vessel ingrowth, acts as an anti-scarring agent, and changes the immune response to promote functional tissue remodeling rather than scarring. It has low immunogenicity, which means that the host’s immune system rejects the graft very little or not at all (Maljaar et al., 2022). Tissue engineering has advanced significantly and offers a novel therapeutic strategy for the restoration and repair of esophageal abnormalities (Cao et al., 2025). In addition, AM possesses antibacterial qualities and suppresses neovascularization. It acts as a natural scaffold that not only covers wounds but also actively encourages tissue recovery because it contains a wealth of growth factors, anti-inflammatory cytokines, and extracellular matrix components (Parmar et al., 2025). The amniotic membrane (AM), the innermost layer of a fetal membrane, has numerous biological features that are crucial for wound surgery. It synthesizes a range of biological substances, such as growth factors, cytokines, and neurotrophins. AM improves epithelialization by encouraging epithelial cell migration, strengthening basal epithelial cell adhesion, encouraging epithelial differentiation to preserve the epithelial phenotype, and regulating the proliferation of normal corneal, conjunctival, and limbal fibroblasts. In addition, AM possesses antibacterial properties, suppresses neovascularization, inflammation, and scarring, and stops apoptosis (Krysik et al., 2020). The AM layers are made up of various types of collagen. AM is abundant in HA, which promotes cell motility and has anti-inflammatory and immunosuppressive qualities. It also contains more than 226 distinct growth factors, cytokines, protease inhibitors, and other bioactive compounds. The special qualities of AM allow it to promote migration, differentiation, and proliferation; control blood vessel ingrowth; operate as an anti-scarring and anti-microbial agent; and change the immune response to promote functional tissue remodeling rather than scarring. AM is special because of its low immunogenicity, which means that the host’s immune system rejects the graft very little or at all. In summary, AM holds a lot of promise as a scaffold for tissue regeneration in reconstructive surgery (Maljaar et al., 2022). Tissue engineering has advanced significantly and offers a novel therapeutic strategy for the restoration and repair of esophageal abnormalities. The body’s ability to rebuild and heal the damaged esophagus is limited, and the entire tissue cannot be renewed (Cao et al., 2025). Platelet-rich plasma (PRP) therapy has emerged as a viable regenerative medicine intervention, providing a multifaceted method for tissue repair and healing across several medical fields. PRP is a concentrated plasma variant filled with platelets, growth factors, cytokines, and other bioactive substances vital for stimulating tissue regeneration and enhancing wound healing. It provides a high concentration of growth factors and cytokines directly to the damaged site, thereby expediting the healing process, diminishing inflammation, increasing tissue repair, and restoring function. The restorative effects of PRP boost collagen synthesis and vascularity. The autologous characteristic is crucial in PRP generation, as it mitigates the danger of immunological rejection or transmission of infectious pathogens, hence improving its safety profile relative to allogeneic or xenogeneic therapy (DiMitri et al., 2025). The growth factors and cytokines in PRP promote local angiogenesis, regulate cellular activity, enhance stem cell homing, differentiation, and proliferation, and contribute to matrix protein deposition, thereby facilitating tissue regeneration (Bharti et al., 2024). PRP is crucial in tissue restoration processes, such as angiogenesis, fibroblast proliferation, and extracellular matrix formation, rendering it relevant in several regenerative medicine therapies. Notwithstanding encouraging outcomes in various tissue injuries, obstacles such as brief half-life and swift protease inactivation remain. Biomaterial-based delivery scaffolds, including amniotic membranes, have been employed in the current study to tackle these issues. PRP-loaded scaffolds address these concerns by the prolonged release of growth factors (Khandan-Nasab et al., 2025). This study provides an overview of the developments in tissue engineering for full-thickness esophagus replacement. Along with discussing difficulties in clinical practice, it also provides an overview of the material used in the design of esophageal transplants. Finally, it provides information about upcoming advancements in biotechnological methods toward this objective. This study aimed to investigate the use of the acellular amniotic membrane as a bioscaffold and platelet-rich plasma for repairing esophageal defects in dogs. Materials and MethodsFifteen healthy mongrel dogs aged 1–1.5 years (weight range, 23–25 kg) were included in the study. Before the experiment, all dogs were acclimated for at least 2 weeks and screened for underlying diseases based on the results of CBC. During the experiment, the dogs were housed separately in cages and fed dry food and water. The patients were divided into three equal groups: the first group was treated with acellular amniotic membrane accompanied by autologous platelet-rich plasma, the second group was treated with acellular amniotic membrane only, and the third group utilized a pedicle flap of the sternohyoideus muscle alone for closing a circumferential esophageal defect. The amniotic membrane preparationThe placenta was cleaned thrice in sterile physiological saline solution to remove clots and then stored in sterile physiological saline solution. The AM was promptly removed from the chorion. In a sterile container filled with a phosphate-buffered saline (PBS) solution containing antibiotics (penicillin, streptomycin, and amphotericin B), samples were collected and transported at 4°C. The samples were then divided into 10 cm2 sections and washed with cold phosphate-buffered saline. Every AM was subjected to three cycles of freezing and thawing: 22 hours in liquid nitrogen (−196 °C) and 10 minutes in a water bath at 37°C. Following a 4-hours treatment with 0.1% sodium dodecyl sulfate (SDS), the BAM membrane was soaked for 1 hour in 0.1 M NaOH, peracetic acid (PAA), and ethanol. Following a final wash with 70% ethanol for 1 hour to remove any remaining phospholipids and nucleic acids from the tissue, PBS was used as the buffer for 2 hours. To guarantee a uniform wash and no harm to the tissue ultrastructure, the membranes were physically agitated using an orbital shaker during the procedure. As a result, they were submerged once more in NaOH for 1 hour and once more in PAA for 1 hour. To remove tissue fragments and spent materials, AM was rinsed with deionized water for 30 minutes in a shaker following each decellularization phase. Lastly, PBS was used to wash each membrane four times for 30 minutes (Fig. 1) (Ballesteros et al., 2020). Preparation of the autologous PRPWhole blood (10 ml) from each dog was placed in biochemistry tubes containing citrate-phosphate dextrose solution and centrifuged for 5 minutes at 1,000 rpm. After centrifugation, three layers were obtained. The plasma layer at the top was collected in another centrifugation tube and subjected to a second centrifugation for 10 minutes at 800 rpm, after which two layers were obtained; the upper layer was PPP, and the lower layer was PRP. This lower form of liquid PRP (2 ml) was used in the surgical sites of our experiment on the dogs (Saqlain et al., 2023). Surgical procedureAs mentioned by Hashem et al. (2023), Following a 12-hour fast, each dog received prophylactic antibiotics cefazolin (cefazolin 1 gm; Afasan company, Holland) at a dosage of 22 mg/kg, intravenously 30 minutes before surgery, subsequently undergoing anesthesia with xylazine hydrochloride ( Xylazine 2%; Alfasan company, Holland) at a dosage of 5 mg/kg body weight and ketamine hydrochloride (ketamine 10%; Alfasan company, Holand) at a dosage of 10 mg/kg body weight. The ventral cervical skin was shaved, sterilized with a povidone-iodine scrub and alcohol, and draped for aseptic surgery. A ventral midline incision was made, and the esophagus was exposed with blunt dissection. Partial circumferential resection of the esophagus was performed. For the patch abnormalities, the excised segment of the esophagus typically measured 3 cm in width and 5 cm in length. The patch resections included approximately 30%–40% of the esophageal circumference. The faulty site was rectified using a cellular amniotic membrane graft material. Grafts were affixed to adjacent native esophagus tissue using 3-0 polydioxanone in a simple, continuous stitching technique. Then, 2 ml of PRP solution was applied to the acellular amniotic membrane at the defect site and injected around the defect. The solution was kept for 2 minutes, allowing the platelets to adhere to the tissues. Each dog received intramuscular postoperative antibiotics for 3 days. The dogs had no oral food for 4 days post-surgery, and hydration was sustained with IV fluid infusion (normal). Ringer solution at a rate of 60 mL/kg/day. 7 mL/kg/h. The animals consumed soft food for 7 days, after which they resumed their normal diet until euthanasia on day thirty (Fig. 1A–C).
Fig. 1. (A) Placentaobtaine d form pregnant caw after natural parturition. (B) Covering the defect site with acellularized amniotic membrance graft material. (C) Reveal application of PRP solution on the acellular amniotic membrane at the site of detect. Clinical evaluationAll experimental animals were clinically observed daily for any postoperative complications. The animals were weighed on day 0 and then once a week until day 28. Histopathological evaluationsImmediately after euthanasia on day 30 post-surgery, the cervical esophagus was excised. The excised tissue encompassed the scaffold implantation area and adjacent native esophageal tissue, both proximal and distal to the graft location. The removed esophagus was incised longitudinally from distal to proximal. The exposed mucosal surface was examined. All biopsy samples from two groups were preserved in 10% neutral buffered formalin solution. The tissue specimens were processed in a tissue processor using the paraffin procedure. Tissue sections were sliced to a thickness of 5–6 μm and subsequently stained with Hematoxylin and Eosin (Hashem et al., 2023). Ethical approvalThe animal ethics committee of the veterinary medicine college at the University of Basrah approved this study (approval no. 63/2024). Dogs were clinically examined, and their blood was tested for systemic diseases. The animals were housed under standard conditions. Water and food were also easy to access. ResultsClinical signs and macroscopic examinationThe surgical procedure was completed in all dogs. Clinical symptoms were monitored in all experimental dogs. At the end of the first week, all groups experienced salivation, dysphagia, and a high fever; these symptoms gradually returned to normal in the first and second groups, while the third group continued to experience these symptoms along with a high fever, salivation, and swallowing difficulty. All groups also experienced a decrease in weight, which gradually returned to normal, while the third group continued to experience these symptoms until the 4th week (Figs. 2–6). Both the first group, which received treatment with amniotic membrane and PRP together, and the second group, which received amniotic membrane alone, displayed a significant healing process in the gross inspection by the formation of granulation tissue. The third group, which received treatment with a pedicle flap of the sternohyoidus muscle, showed an augmented inflammatory lesion in which granuloma developed in the grafting site (Fig. 7A–C). Endoscopic examinationsIt was performed immediately before euthanasia to assess the appearance and stricture of the esophageal mucosa. Gross lesions that were noticed on the esophageal lumen of all dogs were inflammation in the esophageal mucosa of all dogs at the first and second week postoperatively, while endoscopic examination of the dogs in the first and second groups showed mild inflammation, and there were no strictures, while the third group revealed acute inflammation after 1 week postoperatively. The endoscopic examination of dogs in the first group showed the rapid formation of a normal-appearing mucosal surface by approximately the second and third week postoperatively, while the second group revealed progressive stricture and inflammation. No evidence for severe inflammation, erosion, or necrosis was seen by endoscopic examination of the esophagus of animals from the first group (Fig. 8) Microscopic Examination: This study revealed variable degrees of healing among the different treatment groups. i. Group 1 (treated with AM and PRP) exhibited marked regeneration of the esophageal epithelium (Fig. 9A) and substantial development of granulation tissue (Fig. 9B). This was characterized by a regression of inflammation with marked angiogenesis (Fig. 9C) and significant collagen deposition at the transplantation site (Fig. 9D). ii. Group 2 (treated with AM only) demonstrated well-defined epithelial regeneration (Fig. 10A); however, a thin epithelial layer indicated that the epithelium had not achieved full thickness. The operative site revealed a granuloma (Fig. 10B) containing perivascular inflammation and edema foci, along with notable collagen deposition (Fig. 10C).
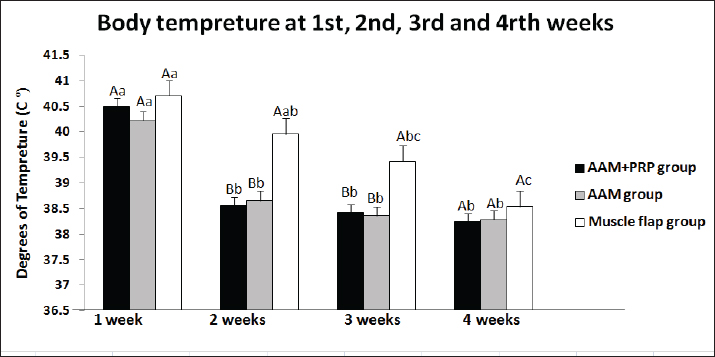
Fig. 2. Animal temperature assessment for the acellular amniotic membrane impregnated with autologous platelet-rich plasma group (AAM+PRP group), acellular amniotic membrane group (AAM group), and muscle pedicle flap group at the end of 1–4 weeks postoperatively. ABDifferent letters among groups indicate significant differences (p < 0.05). abcDifferent letters within a group indicate significant differences (p < 0.05).
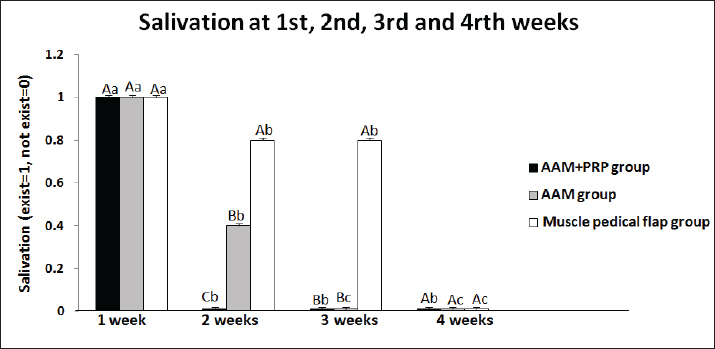
Fig. 3. The clinical study showed the presence of salivation in the acellular amniotic membrane impregnated with autologous platelet-rich plasma (AAM+PRP group), acellular amniotic membrane (AAM group), and muscle pedicle flap (MPF) groups at the end of 1–4 weeks postoperatively. ABDifferent letters among groups indicate significant differences (p < 0.05). abcDifferent letters within-group indicates significant differences (p < 0.05).
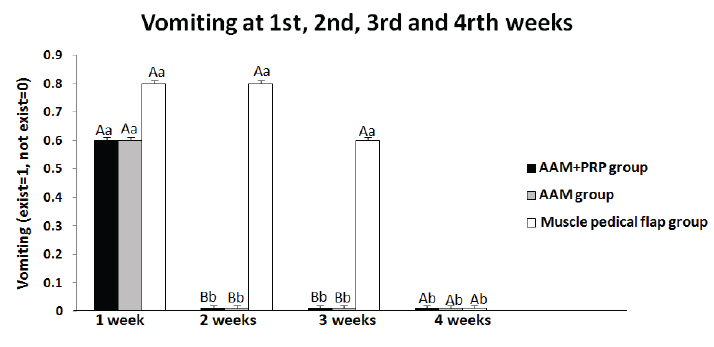
Fig. 4. The clinical study showed the presence of vomiting in the acellular amniotic membrane impregnated with autologous platelet-rich plasma group (AAM+PRP group), acellular amniotic membrane group (AAM group), and muscle pedicle flap group at the end of 1–4 weeks post-surgery. ABDifferent letters among groups indicate significant differences (p < 0.05). Different letters within a group indicate significant differences (p < 0.05).
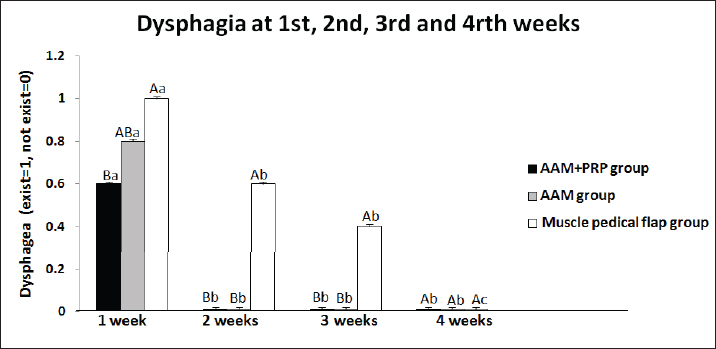
Fig. 5. Clinical study showed the presence of dysphagia in the acellular amniotic membrane impregnated with autologous platelet-rich plasma (AAM+PRP group), acellular amniotic membrane (AAM group), and muscle pedicle flap (MPF) groups at the end of 1–4 weeks postoperatively. ABDifferent letters among groups indicate significant differences (p < 0.05). abcDifferent letters within a group indicate significant differences (p < 0.05).
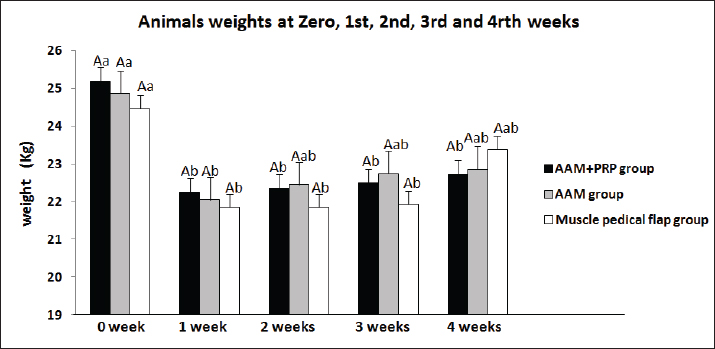
Fig. 6. Animals’ weights were assessed in the acellular amniotic membrane accompanied with autologous platelets-rich plasma (AAM+PRP) group, acellular amniotic membrane (AAM) group, and muscle pedicle flap (MPF) group before surgery and 1, 2, 3, and 4 weeks post-surgery. ABDifferent letters among groups indicate significant differences (p < 0.05). Different letters within a group indicate significant differences (p < 0.05).
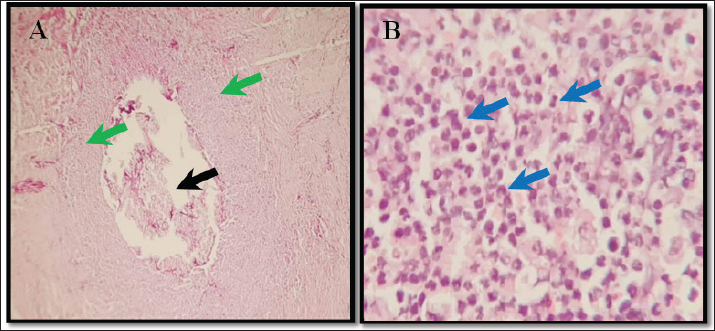
Fig. 7. (A–C) Gross appearance of the esophagus, both (A) which treated with amniotic membrane and PRP together and (B) which with amniotic membrane only shows marked healing process in the gross inspection by formation of granulation tissue while (C) which treated with sterno-hyoidus muscle reveals the augmented inflammatory lesion in which it develops granuloma in the graftingsite.
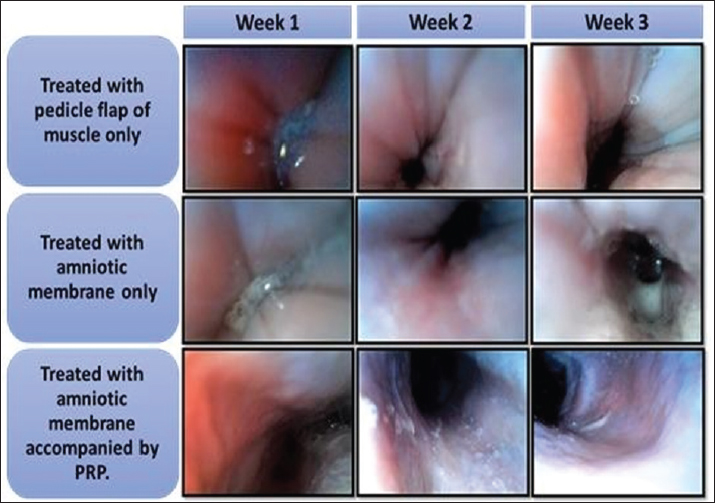
Fig. 8. Reveal the pathological signs of the esophagus of all groups after endoscopic examination. iii. Group 3 (control group) showed severe acute inflammation in the operative area surrounding the surgical sutures (Fig. 11A and B). The group treated with both AM and PRP demonstrated superior results compared with the positive control group and the group treated with AM alone. This is indicated by the development of ongoing inflammation at the healing site, suggesting delayed healing. The third group exhibited the poorest outcomes, with a granuloma at the grafting site. DiscussionEsophageal regeneration remains challenging because of its intricate anatomical structure and low inherent regenerative capacity. Congenital or acquired long-gap esophageal abnormalities are associated with high rates of morbidity and mortality. Conventional replacement procedures, which are frequently associated with significant complications, may be replaced with tissue-engineered grafts (Kunisaki and Coran, 2017). Decellularized biological grafts have garnered interest among the many scaffolds studied because they preserve the native extracellular matrix architecture, which may promote cellular adhesion and tissue integration while lowering inflammatory responses (Adly et al., 2024). The use of a decellularized amniotic membrane (AM) scaffold was associated with improved esophageal healing in the treated group in the current exploratory study. These results are consistent with those of earlier studies, which suggested that the amniotic membrane can provide a conducive milieu for tissue healing. According to Barrett et al. (2017), AM acts as a biological scaffold that promotes the migration and proliferation of epithelial cells and secretes epithelial growth factors, such as epidermal growth factor (EGF), kinase-like growth factor (KGF), and human growth factor (HGF). Similar functions of AM in promoting cell differentiation and preventing apoptosis were reported by Shanbhag et al. (2019) and Paris Fdos et al. (2013). These mechanisms might help explain the observed healing patterns, even if they were not directly evaluated in the current investigation. The lack of esophageal constriction at the defect site may indicate an anti-fibrotic effect in dogs treated with AM. According to an earlier research, AM can restrict excessive collagen deposition and scar formation by suppressing TGF-β signaling in fibroblasts (Parmar et al., 2025). According to Munoz-Torres et al. (2023), AM helps maintain tissue shape and function by preventing pathogenic fibrotic tissue development. However, these conclusions should be viewed as hypothesis-generating because the current study lacks a direct molecular investigation.
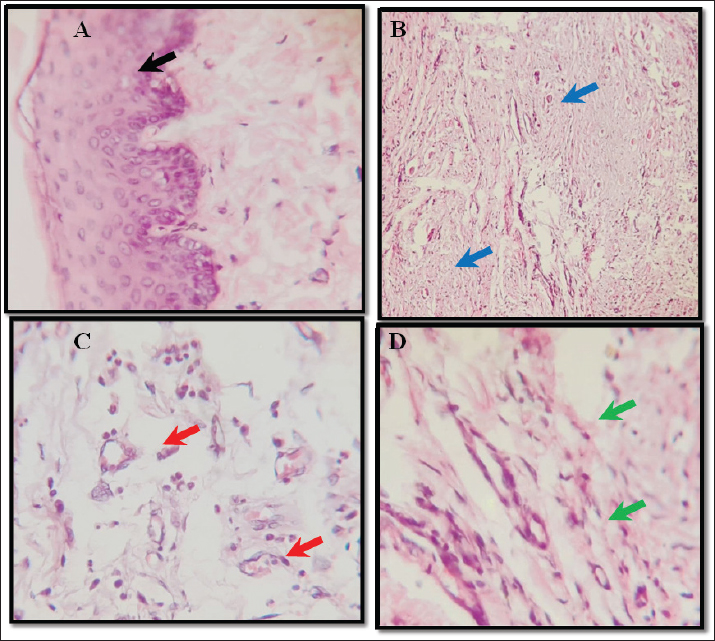
Fig. 9. Group of amniotic membrane and PRP treated group shows (A) marked regeneration of the esophageal eithelium (black arrow) 40X. (B) granulation tissue development in the site if intervention (blue arrow) 10X. (C) marked angiogenesis in the site of granulation tissue (red arrow) 40X (D) marked deposition of collagen fibers in the site of granulation tissue (green arrow) 40X. H&E Further improvement in esophageal healing was observed with the administration of platelet-rich plasma in conjunction with the amniotic membrane, a synergistic effect. Growth factors that contribute to tissue repair processes, such as PDGF, FGF, VEGF, EGF, TGF-β1, IGF, CTGF, and HGF, are enriched in PRP (Gentile and Garcovich, 2020). These elements may promote wound healing by promoting angiogenesis, cellular proliferation, and chemotaxis. The results of this study may be partially explained by the increased neovascularization and decreased local inflammation after PRP treatment, as reported by Santos et al. (2021) and Xu et al. (2020). In addition, platelets exhibit immunomodulatory and antibacterial properties. According to Oral et al. (2020), platelet concentration at injury sites may accelerate granulation tissue formation and re-epithelialization. Although the direct evaluation of antimicrobial activity or immunological modulation was outside the purview of this study, the combined administration of PRP and AM was associated with enhanced clinical healing. Therefore, these results should be considered preliminary. Animals receiving the AM graft showed no indications of immunological rejection. This finding is consistent with those of earlier studies reporting low immunogenicity of amniotic membrane tissue, especially after decellularization (Brouki Milan et al., 2020). According to Adamowicz et al. (2019), the resulting immunological response is clinically insignificant even when AM expresses HLA class I and II molecules because T-cell activation and cytokine release are inhibited. Although the study did not directly assess the immunological characteristics, the biocompatibility of the scaffold is supported by the absence of overt rejection.
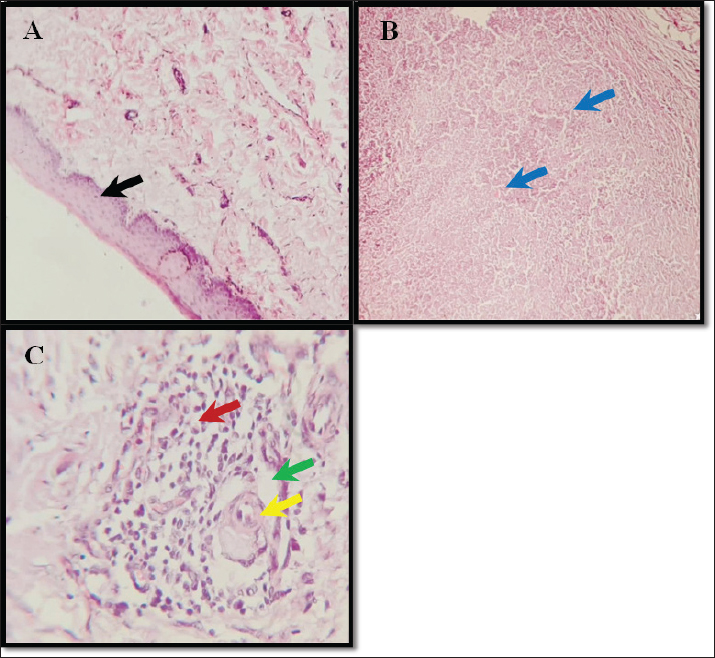
Fig. 10. shows the amniotic membrane-treated group. (A) Marked regeneration of the esophageal epithelium (black arrow) 40X. (B) Granulomatous inflammation at the operation site (blue arrow) 10X. C) Focus of intensive perivascular inflammation (red arrow), edema (green arrow), and newly generated blood vessels (yellow arrow) 40X. H&E All animals experienced postoperative clinical symptoms, such as fever, dysphagia, vomiting, hypersalivation, and weight loss, which eventually resolved. Animals treated with AM and PRP exhibited a more pronounced decrease in symptom severity, although observer variability may affect subjective clinical evaluations. PRP-derived secretomes control angiogenesis, extracellular matrix remodeling, macrophage recruitment, and inflammatory cascades (Verma et al., 2023). Furthermore, Thu (2022) reported that the production of hepatocyte growth factor and other modulatory cytokines mediated the analgesic and anti-inflammatory properties of PRP. By suppressing pro-inflammatory cytokines such as IL-1β, TNF-α, and IL-6 and promoting anti-inflammatory mediators such as IL-10, the amniotic membrane may also help manage inflammation (Munoz-Torres et al., 2023). AM has also been shown to decrease lymphocyte proliferation and alter immune cell polarization. Although direct mechanistic confirmation was not performed, these features may have contributed to the observed clinical improvement. According to Svobodova et al. (2023), AM can reduce nociception by protecting exposed nerve endings, maintaining wound hydration, and releasing bioactive lipid mediators such as palmitoylethanolamide. These protective and analgesic qualities may also be related to the pain reduction and swallowing function restoration observed in the treated animals. However, this trial could not objectively measure pain.
Fig. 11. Control group shows (A) site of surgical suture (black arrow) surrounded by intensive inflammation (green arrow) 10X. (B) intensive non-specific inflammation (blue arrow) 40X. H&E staining. ConclusionsAccording to the results of this exploratory investigation, using a decellularized amniotic membrane in conjunction with platelet-rich plasma may promote esophageal healing by fostering a biological milieu conducive to tissue repair. The results are consistent with improved epithelialization, decreased inflammation, and preserved esophageal structure. However, these findings should be considered exploratory and hypothesis-generating given the experimental methodology used in this study and the lack of precise mechanistic evaluation. Before recommending widespread clinical application, more controlled and randomized research is necessary to confirm these results and elucidate the underlying molecular pathways. AcknowledgmentsThe authors extend their sincere thanks to the Faculty of Veterinary Medicine /University of Basra for their unwavering support. We also deeply appreciate the contributions of the Veterinary Medicine Laboratory staff for their technical support and collaboration in sample collection and analysis. FundingNone. Authors’ contributionsAll authors have made significant intellectual and academic contributions to this study. Each author has read, reviewed, and approved the final manuscript for publication. Conflict of interestThere are no conflicts of interest concerning the publication of this manuscript. Data availabilityAll datasets formed or examined during this work are included in this publication. ReferenceAdamowicz, J., Breda, S.V., Tyloch, D., Pokrywczynska, M. and Drewa, T. 2019. Application of amniotic membrane in reconstructive urology: a promising biomaterial worthy of further investigation. Expert. Opinion. Biol. Therapy. 19(9), 23–24. Adly, H., El-Okby, A., Yehya, A., El-Shamy, A., Galhom, R., Hashem, M. and Ahmed, M. 2024. Circumferential esophageal reconstruction using a tissue-engineered decellularized tunica vaginalis graft. J. Pediatr. Surg. 59(8), 1486–1497. Al-Ajeli, R., Alhasan, A.M. and Al-Mahmood, S. 2022. Role of magnesium oxide nanoparticles in the healing process of cervical esophageal anastomosis in dogs: an experimental study. Egypt. J. Vet. Sci. 53(4), 547–557. Barret, M., Pratico, C.A., Camus, M., Badaoui, F., Jarraya, M., Nicco, C. and Mangialavori, L. 2012. Amniotic membrane grafts for preventing esophageal stricture after circumferential endoscopic submucosal dissection. PLos One 12(12), 189348. Bharti, D., Ajith, Y., Sharun, K., Banu, S.A., Kumar, A., Bhardwaj, A., Sidar, S.K. and Dhaleshwari. 2024. Therapeutic applications of canine platelets and their derivatives: a narrative review. Topics Companion Anim. Med. 58, 100849. Cao, T., Wu, Q., Fan, W., Liu, Z. and Zhan, J. 2025. Recent advances in biomedical scaffolds for esophageal regeneration. Bionic. Eng. 22(1), 1–17. Di Mitri, M., D’Antonio, S., Collautti, E., Di Carmine, A., Libri, M., Gargano, T. and Lima, M. 2024. Platelet-rich plasma in pediatric surgery: a comprehensive review. Children 11(8), 971. Gentile, P. and Garcovich, S. 2020. Potential implications of different platelet-rich plasma concentrations in regenerative medicine for tissue repair: a systematic review. Int. J. Mol. Sci. 21(16), 5702. Hashem, M.A., Metwally, E., Mahmoud, Y.K., Helal, I.E. and Ahmed, M.F. 2023. Reconstruction of a partial esophageal defect using tunica vaginalis and buccal mucosa autograft: an experimental study in mongrel dogs. Vet. Med. Sci. 85(3), 344–357. Khandan-Nasab, N., Torkamanzadeh, B., Abbasi, B., Mohajeri, T., Oskuee, R.K. and Sahebkar, A. 2025. Application of platelet-rich plasma-based scaffolds in regeneration of soft and hard tissues. Tissue Eng. 31(3), 177–191. Krysik, K., Dobrowolski E Wylegala. and Boron. 2020. The amniotic membrane as a main component in treatments supporting healing and patch grafts in corneal perforations. Ophthalmology. J. 5(1), 1–9. Kunisaki, S.M. and Coran, A.G. 2017. Esophageal replacement. Seminars. Pediatric. Surg. 26(2), 105–115. Maljaars, L.P., Bendaoud, S., Kastelein, A.W., Guler, Z. and Hooijmans, C.R. 2022. Application of amniotic membranes in internal organ reconstructive surgery: a systematic review and meta-analysis. Tissue Eng. Regenerative Med. 16, 1069–1090. Milan, P.B., Amini, N., Joghataei, M.T., Ebrahimi, L., Amoupour, M., Sarveazad, A., Kargozar, S. and Mozafari, M. 2020. Decellularized human amniotic membrane: from animal models to clinical trials. Methods 171(11), 11–19. Munoz-Torres, J.R., Martínez-González, S.B., Lozano-Luján, A.D., Martínez-Vázquez, M.C., Velasco-Elizondo, P., Garza-Veloz, I. and Martinez-Fierro, M.L. 2023. Biological properties and surgical applications of human amniotic membranes. Front. Bioeng. Biotechnol. 10, 1067482. Oral, O., Nomikos, G. and Nomikos, N. 2020. Regenerative biological effects of platelet-rich plasma therapy in orthopedic sports medicine. Am. J. Sports. Sci. 8(1), 20–22. Paris, F.D.S., Gonçalves, E.D., Campos, M.S.D.Q., Sato, E.H., Dua, H.S. and Gomes, J.A.P. 2013. Amniotic membrane transplantation versus anterior stromal puncture in bullous keratopathy: a comparative study. Br. J. Ophthalmology 97(8), 980–984. Parmar, U.P.S., Surico, P.L., Scarabosio, A., Barone, V., Singh, R.B., D’Ancona, F., Zeppieri, M., Parodi, P.C., Mori, T., Cutrupi, F., Ma, D.H.K., Di Zazzo, A. and Coassin, M. 2025. Amniotic membrane transplantation for wound healing, tissue regeneration, and immune modulation. Stem Cell Rev. Rep. 21(1), 1–16. Santos, R.G., Santos, G.S., Alkass, N., Chiesa, T.L., Azzini, G.O., Fonseca, L.F., Santos, A.F., Rodrigues, B.L. and Mosaner, T. 2021. Regenerative mechanisms of platelet-rich plasma: a review. Cytokine 144, 155560. Saqlain, N., Mazher, N., Fateen, T. and Siddique, A. 2023. Comparison of single and double centrifugation methods for platelet-rich plasma (PRP) preparation. Pak. J. Med. Sci. 39(3), 634–637. Shanbhag, S., Patel, C., Goyal, R., Donthineni, P., Singh, V. and Basu, S. 2019. Simple limbal epithelial transplantation: indications, surgical technique, mechanism, outcomes, and limitations. Indian J. Ophthalmology 67(8), 1265–1277. Svobodova, A., Vrkoslav, V., Smeringaiova, I. and Jirsova, K. 2018. Distribution of an analgesic palmitoylethanolamide and other N-acylethanolamines in human placental membranes. PLos One 18(2), 281790. Thu, A.C. 2022. Use of platelet-rich plasma in musculoskeletal pain management: a narrative review. Yeungnam. Med. Sci. 39(3), 206–215. Verma, R., Kumar, S., Garg, P. and Verma, Y. 2023. Platelet-rich plasma: a comparative and economical therapy for wound healing. Cell. Tissue. Banking. 24, 285–306. Villamil Ballesteros, A.C., Segura Puello, H.R., Lopez-Garcia, J.A., Bernal-Ballen, A., Nieto Mosquera, D.L., Muñoz Forero, D.M., Segura Charry, J.S. and Neira Bejarano, Y.A. 2020. Bovine decellularized amniotic membrane: extracellular matrix as scaffold for mammalian skin. Polymers 12(3), 664. Xu, P., Wu, Y., Zhou, L., Yang, Z., Zhang, X., Hu, X., Yang, J., Wang, M., Wang, B. and Luo, G. 2020. Platelet-rich plasma accelerates skin wound healing by promoting re-epithelialization. Burns Trauma 8, 1–13. | ||
| How to Cite this Article |
| Pubmed Style Khashjoori BK, Alrafas HR, Abdulkareem M, Khashjoori MABK, Alrafas HR, Abdulkareem M, Abbas M, Hashim AM. Experimental esophageal reconstruction in dogs using autologous plateletrich plasma-augmented acellular amniotic membrane scaffold. Open Vet. J.. 2026; 16(2): 1112-1123. doi:10.5455/OVJ.2026.v16.i2.30 Web Style Khashjoori BK, Alrafas HR, Abdulkareem M, Khashjoori MABK, Alrafas HR, Abdulkareem M, Abbas M, Hashim AM. Experimental esophageal reconstruction in dogs using autologous plateletrich plasma-augmented acellular amniotic membrane scaffold. https://www.openveterinaryjournal.com/?mno=285222 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.30 AMA (American Medical Association) Style Khashjoori BK, Alrafas HR, Abdulkareem M, Khashjoori MABK, Alrafas HR, Abdulkareem M, Abbas M, Hashim AM. Experimental esophageal reconstruction in dogs using autologous plateletrich plasma-augmented acellular amniotic membrane scaffold. Open Vet. J.. 2026; 16(2): 1112-1123. doi:10.5455/OVJ.2026.v16.i2.30 Vancouver/ICMJE Style Khashjoori BK, Alrafas HR, Abdulkareem M, Khashjoori MABK, Alrafas HR, Abdulkareem M, Abbas M, Hashim AM. Experimental esophageal reconstruction in dogs using autologous plateletrich plasma-augmented acellular amniotic membrane scaffold. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1112-1123. doi:10.5455/OVJ.2026.v16.i2.30 Harvard Style Khashjoori, B. K., Alrafas, . H. R., Abdulkareem, . M., Khashjoori, . M. A. B. K., Alrafas, . H. R., Abdulkareem, . M., Abbas, . M. & Hashim, . A. M. (2026) Experimental esophageal reconstruction in dogs using autologous plateletrich plasma-augmented acellular amniotic membrane scaffold. Open Vet. J., 16 (2), 1112-1123. doi:10.5455/OVJ.2026.v16.i2.30 Turabian Style Khashjoori, Bassim Kadhim, Heidar Rasheed Alrafas, Mukhalad Abdulkareem, Murtadha Abbas Bassim Kadhim Khashjoori, Heidar Rasheed Alrafas, Mukhalad Abdulkareem, Murtadha Abbas, and Ammar Maatoq Hashim. 2026. Experimental esophageal reconstruction in dogs using autologous plateletrich plasma-augmented acellular amniotic membrane scaffold. Open Veterinary Journal, 16 (2), 1112-1123. doi:10.5455/OVJ.2026.v16.i2.30 Chicago Style Khashjoori, Bassim Kadhim, Heidar Rasheed Alrafas, Mukhalad Abdulkareem, Murtadha Abbas Bassim Kadhim Khashjoori, Heidar Rasheed Alrafas, Mukhalad Abdulkareem, Murtadha Abbas, and Ammar Maatoq Hashim. "Experimental esophageal reconstruction in dogs using autologous plateletrich plasma-augmented acellular amniotic membrane scaffold." Open Veterinary Journal 16 (2026), 1112-1123. doi:10.5455/OVJ.2026.v16.i2.30 MLA (The Modern Language Association) Style Khashjoori, Bassim Kadhim, Heidar Rasheed Alrafas, Mukhalad Abdulkareem, Murtadha Abbas Bassim Kadhim Khashjoori, Heidar Rasheed Alrafas, Mukhalad Abdulkareem, Murtadha Abbas, and Ammar Maatoq Hashim. "Experimental esophageal reconstruction in dogs using autologous plateletrich plasma-augmented acellular amniotic membrane scaffold." Open Veterinary Journal 16.2 (2026), 1112-1123. Print. doi:10.5455/OVJ.2026.v16.i2.30 APA (American Psychological Association) Style Khashjoori, B. K., Alrafas, . H. R., Abdulkareem, . M., Khashjoori, . M. A. B. K., Alrafas, . H. R., Abdulkareem, . M., Abbas, . M. & Hashim, . A. M. (2026) Experimental esophageal reconstruction in dogs using autologous plateletrich plasma-augmented acellular amniotic membrane scaffold. Open Veterinary Journal, 16 (2), 1112-1123. doi:10.5455/OVJ.2026.v16.i2.30 |