| Research Article | ||
Open Vet. J.. 2026; 16(2): 1082-1089 Open Veterinary Journal, (2026), Vol. 16(2): 1082-1089 Research Article Physiological, molecular, and histological evaluation of the effects of cobalamin on cardiac function in castrated rabbitsMohammed Hayder Asker1, Wassan Mhammed Husain2, Hadeel Kamil Khaleel3 and Mustafa Riyadh Abdullah4*1Basic Science Department, College of Dentistry, Mustansiriyah University, Baghdad, Iraq 2Medical College, Ibn Sina University of Medical and Pharmaceutical Sciences, Baghdad, Iraq 3Department of Basic Science, College of Dentistry, Al-Iraqia University, Baghdad, Iraq 4Department of Pharmacology and Toxicology, College of Pharmacy, Mustansiriya University, Baghdad, Iraq *Corresponding Author: Mustafa Riyadh Abdullah. Department of Pharmacology and Toxicology, College of Pharmacy, Mustansiriya University, Baghdad, Iraq. Email: mustafa.r.a [at] uomustansiriyah.edu.iq Submitted: 23/09/2025 Revised: 04/01/2026 Accepted: 21/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
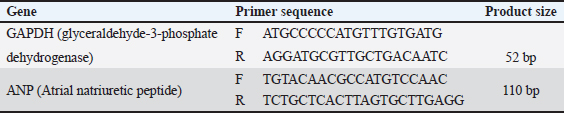
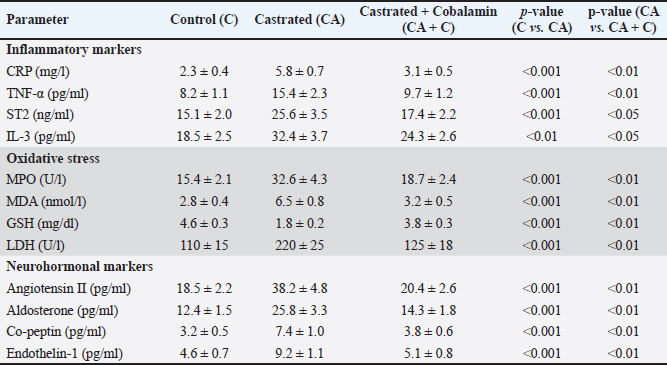
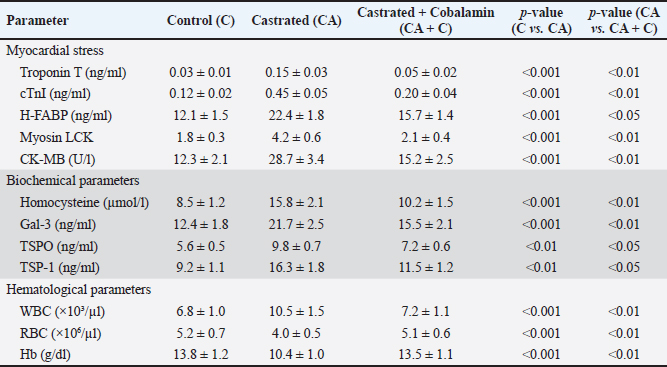
AbstractBackground: Testosterone deficiency is associated with adverse cardiac remodeling, oxidative stress, and inflammation. Aim: To investigate the physiological, biochemical, and histological effects of cobalamin (vitamin B12) supplementation on cardiac physiology in castrated rabbits. Methods: A total of 24 male rabbits were divided into three groups: control (C), castrated (CA), and castrated with cobalamin supplementation (CA + C). Biochemical markers, including cardiac troponin I (cTnI), heart-type fatty acid-binding protein (H-FABP), homocysteine, glutathione (GSH), interleukin-3 (IL-3), galectin-3 (Gal-3), translocator protein (TSPO), and thrombospondin-1 (TSP-1), were measured using Enzyme-Linked Immunosorbent Assay. Myocardial tissue was histologically evaluated alongside Glyceraldehyde-3-Phosphate Dehydrogenase (GAPDH) and Atrial Natriuretic Peptide (ANP) gene expression analysis using quantitative real-time reverse-transcription PCR. Statistical significance was set at p value < 0.05. Results: Castration significantly increased the levels of cardiac markers, such as cTnI, H-FABP, homocysteine, IL-3, and Gal-3, while reducing GSH levels (p < 0.001). Cobalamin supplementation significantly restored these biochemical parameters, with notable reductions in cTnI, H-FABP, homocysteine, IL-3, Gal-3, and GSH levels (p < 0.01). Histological analysis revealed myocardial contraction failure, fibrosis, and inflammation in castrated rabbits, which were mitigated by cobalamin supplementation. Gene expression analysis showed GAPDH and ANP upregulation in the castrated group, which was partially restored after cobalamin treatment. Additionally, cobalamin supplementation significantly improved the expression of other markers, such as TSPO and TSP-1, and its potential to modulate inflammatory and oxidative pathways in the heart. Conclusion: Cobalamin supplementation mitigates the harmful effects of testosterone deficiency on the heart by improving oxidative stress, inflammation, and cardiac remodeling. These findings support the potential of this drug for correcting cardiovascular dysfunction associated with hormonal deficiencies. Keywords: Cardiac physiology, Cobalamin, Gene expression, Inflammation Testosterone deficiency, Oxidative stress. IntroductionCardiovascular diseases remain a major global health burden, causing substantial mortality and morbidity (Ho, 2018). Such conditions are linked to biomarkers that characterize myocardial damage or stress, for example, cardiac troponin I (cTnI), Heart-Type Fatty Acid Binding Protein (H-FABP), and homocysteine (Ponikowska et al., 2022). Oxidative stress and inflammation are factors at the root of cardiovascular diseases; hence, the need to find new treatment approaches is growing rapidly (Aimo et al., 2019). Cobalamin, also known as vitamin B12, is a water-soluble vitamin that plays a prognostic role in the metabolism of homocysteine (Mshiemish, 2011; Combs and McClung, 2016). Abnormal homocysteinemia is linked to vitamin B12 deficiency because high homocysteine levels are an established cause of cardiovascular diseases due to their inflammatory and clot-inducing properties (Gospodarczyk et al., 2022). Furthermore, cobalamin has antihomocysteinemic effects and antioxidant potential due to its ability to increase the levels of glutathione (GSH), which is an important intracellular antioxidant (Van De Lagemaat et al., 2019). In addition to its involvement in homocysteine metabolism and free radical scavenging activity, cobalamin has been shown to affect inflammatory mediators. Gal-3 and thrombospondin-1(TSP-1) have been shown to play roles in cardiac remodeling, fibrosis, and inflammation. Translocator protein (TSPO) can predict and alter myocardial injury and may thus be helpful in the development of therapeutic agents for cardiac disease (Song et al., 2022). As previously stated, castration, which results in a substantial reduction in the levels of sex hormones, including testosterone, is reported to cause cardiovascular pathologies, increased oxidative stress, and impaired myocardial function Atrial Natriuretic Peptide (Ayaz and Howlett, 2015;Diaconu et al., 2021; Abdullah and Alabassi, 2024). The lipid metabolism and myocardial injury response of chemically castrated rabbits are quite similar to those of humans, and they have been extensively used for cardiovascular research (Yanni, 2004). To the best of our knowledge, this study is the first to comprehensively assess biochemical, oxidative, inflammatory, histological, and molecular (ANP gene) changes in a testosterone-deficient rabbit model. To the best of our knowledge, no previous research has evaluated such a broad panel of cardiac markers alongside structural and gene-level remodeling in response to cobalamin supplementation. Therefore, this study aimed to assess the physiological, biochemical, and histological changes within the cardiac tissue of castrated rabbits following cobalamin intervention. We hypothesized that cobalamin supplementation would attenuate oxidative stress, inflammation, molecular remodeling, and histopathological damage induced by testosterone deficiency. Materials and MethodsAnimalsTwenty-four adult male New Zealand White rabbits (average weight: 2.5–3.5 kg, age: 4–6 months) were included. Rabbits were housed in individual cages in a temperature-controlled facility (22°C ± 2°C) with a 12-hour light/dark cycle and 50%–60% humidity. All animals received standard pelleted feed and ad libitum access to water. Inclusion criteriaRabbits with normal cardiac physiology confirmed by initial veterinary screening. Exclusion criteriaRabbits with signs of pre-existing diseases, abnormalities, or significant weight loss during acclimatization were excluded. Experimental groupsThe rabbits were randomly divided into the following three experimental groups (n=8 per group): Control group (C): non-castrated rabbits, no treatment. Castrated group (CA): Castrated rabbits without cobalamin treatment. Castrated + Cobalamin Group (CA + C): Castrated rabbits treated with Co. Castration procedureThe rabbits were fasted for 12 hours preoperatively, with free access to water. The surgical sites were shaved and sterilized using povidone-iodine and 70% ethanol. General anesthesia was induced intramuscularly using ketamine (40 mg/kg) and xylazine (5 mg/kg). A local anesthetic (2% lidocaine) was administered at the surgical site. Bilateral orchiectomy was performed using a midline scrotal incision. Spermatic cords were ligated using absorbable sutures, and the testes were excised. The incision was closed using absorbable sutures. Antibiotics (enrofloxacin, 10 mg/kg) were intramuscularly administered for 3 days post-surgery. Analgesics (meloxicam, 0.2 mg/kg) were administered for pain management. Cobalamin administrationRabbits in the CA + C group received intramuscular injections of cobalamin (500 µg/kg) once weekly for 8 weeks (Hagar, 2002). Biochemical analysisBlood samples were collected aseptically from cardiac puncture at the end of the study (week 8) after anesthetizing the animals using ketamine and xylazine only. The serum was then isolated through centrifugation of the sample at 3,000 × g for 15 minutes and aliquoted into culture tubes before storing at −20°C until analysis. The following parameters were measured using Enzyme-Linked Immunosorbent Assay (ELISA) kits (MyBioSource) according to the manufacturer’s protocols: Cardiac troponin I (cTnI): Determined by MyBioSource ELISA kit (#MBS729403). Homocysteine: Determined by MyBioSource ELISA kit (catalog number: MBS7252797). Heart-Type Fatty Acid Binding Protein: Evaluated using the MyBioSource ELISA kit (#MBS9302016). Co-peptin: A highly significant difference in Co-peptin concentrations was assessed using the ELISA kit (# MBS7254024). IL-3: Analyzed using MyBioSource ELISA kit # MBS282976. Glutathione: Determined with MyBioSource ELISA kit ($ #MBS9718983). Galectin-3 (Gal-3): Quantified using MyBioSource ELISA kit, MBS203603. Translocator protein: This was assessed using the MyBioSource ELISA kit (catalog number: #MBS540519). TSP-1: Determined using the MyBioSource ELISA kit (catalog number: MBS712207). Tumor necrosis factor-alpha (TNF-α): DEET receptor expression was quantified using an ELISA kit obtained from MyBioSource with catalog number MBS 702825. Suppression of tumorigenicity 2 (ST2): assessed using the commercially available MyBioSource ELISA kit MBS721869. Myeloperoxidase (MPO): Assessed by NIH 3T3 MyBioSource ELISA kit (# MBS9718983). Malondialdehyde (MDA): Quantified using the MyBioSource ELISA kit with ID of MBS9718990. Angiotensin II: Standard procedure was performed using the MyBioSource ELISA kit (#MBS700246). Aldosterone: The MyBioSource ELISA kit (catalog number: MBS700089) was used. Endothelin-1: Determined by MyBioSource enzyme-linked immunosorbent assay kit (#MBS700074). Cardiac troponin T: Determined using MyBioSource ELISA kit (MBS700057). Myosin light chain kinase: Determined using a MyBioSource ELISA kit (catalog number #MBS700018). This enzyme acts in muscle contraction and has been linked with heart and skeletal muscle function. Creatine kinase-MB (CK-MB): Determined by MyBioSource ELISA test kit (catalog number: MBS700025). All ELISA assays were performed as instructed in the manufacturer’s protocol. The samples were then analyzed on a microplate reader (BMG-Labtech) at the given absorbance values. Hematological parameterswhite blood cells (WBC), red blood cells (RBC), and hemogram were estimated using a fully automated hematology analyzer (MSLAB03). Histopathological evaluationHeart tissues were collected, rinsed in saline, and fixed in 10% buffered formalin for 24 hours. Tissues were then dehydrated, embedded in paraffin, and sectioned at 5 µm thickness. Hematoxylin and eosin staining was used to examine the general morphology. A pathologist performed the microscopic examination, and blinded investigators conducted the histopathological evaluation. The scoring system parameters areEach parameter was scored on a scale of 0 to 3, as follows: 0: No detectable abnormality 1: Mild changes (e.g., scattered inflammatory cells or slight disarray) 2: Moderate changes (e.g., focal necrosis or moderate fibrosis) 3: Severe changes (e.g., extensive necrosis or widespread fibrosis) The cumulative score for each sample was calculated by summing the individual scores for all parameters, providing a comprehensive assessment of myocardial damage severity (Sachdeva et al., 2014). Gene expression analysisTotal Ribonucleic Acid (RNA) was extracted from the cardiac tissue using TRIzol reagent (Thermo Fisher Scientific). RNA integrity was verified using agarose gel electrophoresis, and RNA concentration was measured using a NanoDrop spectrophotometer. Investigators blinded to the treatment groups performed the gene expression analysis. Gene expression of Glyceraldehyde-3-Phosphate Dehydrogenase (GAPDH) and ANP was analyzed using specific primers. Primers were designed using information from the National Center for Biotechnology Information-GenBank and the Primer3 plus program, and all primers were provided by Bioneer (Korea) (Table 1). Reaction conditions: 96°C for 5 minutes (initial denaturation), 45 cycles of 96°C for 20 seconds and 62°C for 45 seconds. The Livak method was used to determine gene expression levels. Folding=2−ΔΔCT ΔCT=C.T. gene C.T. Housekeeping gene ΔΔCT=ΔCT Treated or Control − ΔCT Control. Statistical analysisData were statistically analyzed using SPSS version 25. Statistical analysis was expressed as mean ± SD, and data were analyzed on the basis of (one-way Analysis of variance with Tukey’s post hoc test for group comparisons). Significance Threshold: p < 0.05 was considered statistically significant. Ethical approvalThe Institutional Animal Care and Use Committee (IACUC) approved all experimental procedures involving animals under protocol number [45] on [22/12/2024]. This study was conducted in accordance with the ARRIVE guidelines and adhered to the principles of laboratory animal care. ResultsBiochemical parametersThe study evaluated various inflammatory, oxidative stress, neurohormonal, myocardial stress, biochemical, and hematological markers across three groups: control (C), castrated (CA), and castrated with cobalamin treatment (CA + C). CRP, TNF-, ST2, and IL-3 levels were significantly different in the CA group compared with those in the control group and were reduced in the CA + C group (p < 0.001 to p < 0.05). There were significant changes in oxidative stress markers, among which MPO, MDA, and GSH were significantly altered (p < 0.001 to p < 0.01); in the CA group, MPO and MDA levels increased, and GSH levels decreased. Neurohormonal analogs, such as angiotensin II, aldosterone, copeptin, and endothelin-1, were not an exception as the CA group increased significantly and the corresponding treatment with cobalamin decreased them (p < 0.001 to p < 0.01). The CA group demonstrated significant levels of myocardial stress markers (troponin T, cTnI, H-FABP, myosin light chain kinase, and CK-MB) and, partially, the CA + C control group (p < 0.001 to p < 0.05). Significant changes were also observed in the homocysteine, Gal-3, TSPO, and TSP-1 parameters, in which the CA preparedness was high, and that of the CA + C group decreased (0.001–0.05). The hematological parameters demonstrated significant changes with increasing WBC counts and decreasing RBC counts and hemoglobin levels in the CA group and partially normalizing in the CA + C group (p < 0.001 to p < 0.01). These results emphasize the high effect of castration on different physiological parameters and the protective effects of cobalamin treatment (Tables 2 and 3). Table 1. Primer sequences for qRT–PCR.
Table 2. Inflammatory, oxidative stress, and neurohormonal markers.
Table 3. Myocardial stress, biochemical parameters, and hematological parameters.
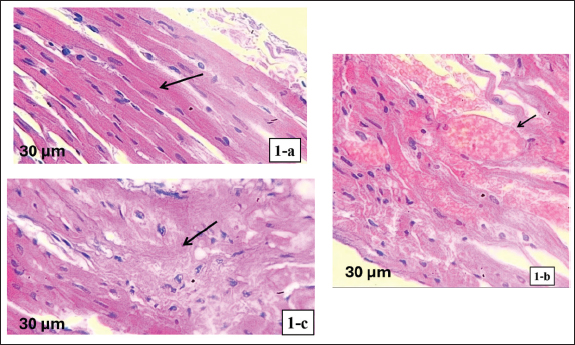
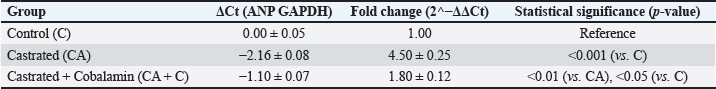
Histopathological findingsThe control group had normal myocardial tissue architecture as shown by myocardial tissue histological studies. The cardiomyocytes were well-organized and had clear striations, had intact intercalated discs, and did not have any signs of pathological alteration, including fibrosis, inflammation, or cell disarray (Fig. 1a). The myocardial tissue of the castrated group exhibited a high level of pathological changes. Disarranged myocardial fibers, widespread interstitial fibrosis, and severe inflammatory cell infiltration were observed. Cardiomyocyte hypertrophic alterations were also observed, which denoted structural damage by testosterone deficiency (Fig. 1b). Lastly, histological examination of the cobalamin-treated group revealed significant structural changes in the myocardium compared with the castrated group. Fibrosis and inflammatory cell infiltration were much less prominent, and the myocardial fibers were more organized. Although some abnormalities remained, the general structure of the tissue was more similar to that of the control group, indicating the restorative action of cobalamin supplementation (Fig. 1c). Gene expression analysisGAPDH expression was uniform in all groups, confirming that it can be used as a quality reference gene to normalize the analysis. In contrast, ANP expression in the CA group was significantly increased (p < 0.001) compared with that in the control group, indicating cardiac stress and pathological remodeling. Castrated + Cobalamin Group (CA + C): ANP levels were significantly lower in the CA + C group (1.8-fold increase vs. C; 0.01 vs. CA), although the effect did not completely restore ANP levels to baseline (Table 4). DiscussionCastration-induced testosterone deficiency generated definite biochemical, structural, and molecular signs of heart damage. These results are consistent with the literature that testosterone depletion is associated with oxidative stress, inflammatory conditions, and myocardial remodeling (Piquereau et al., 2016; Jones and Kelly, 2018; Lima et al., 2023). The current study has shown that cobalamin supplementation has a positive effect and reduces many of these adverse effects in numerous complementary ways.
Fig. 1. Histological appearance of myocardial tissue (hematoxylin and eosin stain, 400x). (a) Control group: normal myocardial architecture with well-organized cardiomyocytes, intact intercalated discs, and absence of fibrosis or inflammation. (b) Castrated group: disorganized myocardial fibers, interstitial fibrosis, inflammatory cell infiltration, and hypertrophic cardiomyocytes, indicating structural damage due to testosterone deficiency. (c) Castrated + cobalamin group: improved myocardial structure with reduced fibrosis and inflammation, myocardial fibers appear more organized, approaching normal architecture. Table 4. Relative ANP expression across study groups.
Antioxidant effects of cobalaminUsing castrated rabbits, it was found that GSH was reduced significantly and that the oxidative stress markers (MDA and MPO) increased significantly, which is a strong oxidative injury. Cobalamin treatment was important in enhancing GSH levels and minimizing oxidative mediators, which conform to its reported antioxidative property in inhibiting redox-related tissue damage (Chatthanawaree, 2010; Van De Lagemaat et al., 2019). These effects were probably the causes of the reduced myocardial stress markers in the treated group. Anti-inflammatory effectsThe castration was accompanied by a significant increase in the levels of inflammatory mediators, such as IL-3, TNF-a, CRP, and ST2. In addition, both Galectin-3 and TSPO, which participate in inflammation-based cardiac remodeling, were elevated in the CA group. Cobalamin treatment had a significant effect on these markers, which showed a wide anti-inflammatory action. This agrees with the findings that cobalamin regulates the effect of cytokines and minimizes inflammation in tissues (Herrmann and Herrmann, 2022; Ahmed et al., 2023). Anti-fibrotic and anti-remodeling effectsMyocardial disarrangement of the fibers, interstitial fibrosis, inflammation infiltrates, and cardiomyocyte hypertrophy were the signs of adverse cardiac remodeling in castrated rabbits. The structural abnormalities were significantly reduced in the group treated with cobalamin. The enhancement is consistent with the downregulation of Gal-3 and TSP1, which are major mediators of fibrosis (Sygitowicz et al., 2021). The same study has been conducted in the past, with results indicating that cobalamin decreases tissue remodeling in cardiac stress models (Santoso et al., 2019; Asker et al., 2025). Homocysteine-lowering mechanismCastration had a major effect on increasing the level of homocysteine, which was probably caused by disrupted methylation pathways under the impact of testosterone deficiency (Corona et al., 2018). This increase was reversed by cobalamin supplementation, which reinstated the coenzyme activity of B12 in the metabolism of homocysteine, which was strengthened by other studies attributing B12 to reduced cardiovascular risk (Bajic et al., 2022). Reduction of oxidative and inflammatory cardiac injury by lower homocysteine levels may also be indirect. Molecular adaptations: ANP gene expressionANP expression was increased more than fourfold in castrated rabbits, reflecting heightened cardiac stress and remodeling. Cobalamin significantly reduced ANP expression, although not fully to baseline, indicating partial molecular recovery. Similar partial normalization has been observed in other models where structural cardiac changes persist despite biochemical improvement (Brasil et al., 2015; Kuloğlu et al., 2024). Cobalamin improves myocardial stress signaling but does not fully reverse long-standing remodeling without combination therapy or a longer treatment duration. ConclusionCobalamin supplementation reduces the negative physiological effects of testosterone deficiency on the biochemical, histological, and molecular levels, so that cobalamin can be useful for treating the cardiotoxic effects of testosterone deficiency. Limitations of the studyA limitation of this study is the limited number of parameters analyzed. Limitations include the small sample size, single gene marker analysis, dose-response model absence, and lack of long-term follow-up. Future studies should incorporate additional remodeling and oxidative stress genes and compare different cobalamin formulations. AcknowledgmentsThe authors would like to thank Mustansiriyah University and Al-Iraqia University for providing the necessary facilities to conduct this research. Conflict of interestThe authors declare no conflict of interest. FundingThis research received no specific grant. Authors’ contributionsMohammed Hayder Asker: Conceptualization, methodology, animal handling and surgeries, biochemical assays, data curation, and original manuscript drafting. Wassan Mhammed Husain: Methodology, histopathological processing and evaluation, validation, investigation, and critical review of the manuscript were performed. Hadeel Kamil Khaleel: Molecular experiments (RNA extraction and quantitative reverse transcription polymerase chain reaction), formal analysis, data interpretation, and manuscript review and editing. Mustafa Riyadh Abdullah: Conceptualization, supervision, project administration, interpretation of pharmacological/toxicological aspects, and final critical revision of the manuscript; serves as the guarantor of the work and corresponding author. All authors have read and approved the final version of the manuscript. Data availabilityThe datasets generated and analyzed during the current study are available upon reasonable request from the corresponding author. ReferencesAbdullah, M.R. and Alabassi, H.M. 2024. The potential influence of immune modulatory molecules (TGF-βIII and CTLA-4) in pathogenicity of PCOS. Karbala. Int. J. Mod. Sci. 10(3), 2; doi: 10.33640/2405-609X.3361 Ahmed, T.H., Mshimesh, B.A.R. and Raoof, I.B. 2023. Effect of nicorandil on endothelial markers and tissue remodeling in pulmonary arterial hypertension model of male rats. Al-Rafidain J. Med. Sci. 5(1S), S87–S93; doi:10.54133/ajms.v5i1S.334 Aimo, A., Castiglione, V., Borrelli, C., Saccaro, L.F., Franzini, M., Masi, S., Emdin, M. and Giannoni, A. 2019. Oxidative stress and inflammation in the evolution of heart failure: from pathophysiology to therapeutic strategies. Eur. J. Prev. Cardiol. 27(5), 494–510; doi:10.1177/2047487319870344 Asker, M.H., Al-Zuhairy, N.A.H.S., Husain, W.M. and Abdullah, M.R. 2025. Protective effects of allicin against stanozolol-induced cardiotoxicity: physiological and histopathological evidence in a rabbit model. Anim. Models Exp. Med. 8(7), 1196–1205; doi: 10.1002/ame2.70035 Ayaz, O. and Howlett, S.E. 2015. Testosterone modulates cardiac contraction and calcium homeostasis: cellular and molecular mechanisms. Biol. Sex Differ. 6, 1–10; doi:10.1186/s13293-015-0027-9 Bajic, Z., Sobot, T., Skrbic, R., Stojiljkovic, M.P., Ponorac, N., Matavulj, A. and Djuric, D.M. 2022. Homocysteine, vitamins B6 and folic acid in experimental models of myocardial infarction and heart failure—How strong is that link?. Biomolecules 12(4), 536; doi:10.3390/biom12040536 Brasil, G.A., Lima, E.M.D., Nascimento, A.M.D., Caliman, I.F., Medeiros, A.R.S.D., Silva, M.S.B., Abreu, G.R.D., Reis, A.M.D., Andrade, T.U.D. and Bissoli, N.S. 2015. Nandrolone decanoate induces cardiac and renal remodeling in female rats, without modification in physiological parameters: the role of ANP system. Life. Sci. 137, 65–73; doi:10.1016/j.lfs.2015.07.005 Chatthanawaree, W. 2010. Biomarkers of cobalamin (vitamin B12) deficiency and its application. J. Nutr. Health Aging 15(3), 227–231; doi:10.1007/s12603-010-0280-x Combs, G.F., Jr. and McClung, J.P. 2016. The vitamins: fundamental aspects in nutrition and health. Cambridge, MA: Academic Press; doi: 10.1016/B978-0-323-90473-5.20001-3 Corona, G., Rastrelli, G., Di Pasquale, G., Sforza, A., Mannucci, E. and Maggi, M. 2018. Testosterone and cardiovascular risk: meta-analysis of interventional studies. J. Sex. Med. 15(6), 820–838; doi:10.1016/j.jsxm.2018.04.641 Diaconu, R., Donoiu, I.S.L.T.W.C.B., Mirea, O. and Bălşeanu, T.A. 2021. Testosterone, cardiomyopathies, and heart failure. Asian. J. Androl. 23(4), 348–356; doi:10.4103/aja.aja_80_20 Gospodarczyk, A., Marczewski, K., Gospodarczyk, N., Widuch, M., Tkocz, M. and Zalejska-Fiolka, J. 2022. Homocysteine and cardiovascular disease—A current review. Wiad. Lek. 75, 2862–2866; doi:10.36740/WLek202211202 Hagar, H.H. 2002. Folic acid and vitamin B12 supplementation attenuates isoprenaline-induced myocardial infarction in experimental hyperhomocysteinemic rats. Pharmacol. Res. 46(3), 213–219; doi:10.1016/S1043-6618(02)00095-6 Herrmann, W. and Herrmann, M. 2022. The controversial role of HCY and vitamin B deficiency in cardiovascular diseases. Nutrients 14(7), 1412; doi:10.3390/nu14071412 Ho, K.J. 2018. Cardiovascular diseases. In Nutritional Aspects of Aging. Ed., Chen, L.H. Boca Raton, FL: CRC Press, pp: 75–100; doi: 10.1201/9781351075145 Jones, T. and Kelly, D. 2018. Randomized controlled trials – mechanistic studies of testosterone and the cardiovascular system. Asian. J. Androl. 20(2), 120; doi:10.4103/aja.aja_6_18 Kuloğlu, N., Karabulut, D., Kaymak, E., Akin, A.T., Ceylan, T., Yıldırım, A.B. and Yakan, B. 2024. Effect of vitamin B12 on methotrexate-induced cardiotoxicity in rats. Iran. J. Basic Med. Sci. 27(6), 733–739; doi:10.22038/ijbms.2024.74161.16120 Lima, P.R.D.V., Ronconi, K.S., Morra, E.A., Rodrigues, P.L., Ávila, R.A., Merlo, E., Graceli, J.B., Simões, M.R., Stefanon, I. and Júnior, R.F.R. 2023. Testosterone deficiency impairs cardiac interfibrillar mitochondrial function and myocardial contractility while inducing oxidative stress. Front. Endocrinol. 14, 1206387; doi:10.3389/fendo.2023.1206387 Mshiemish, B.A. 2011. Role of the clinical pharmacist in reducing preventable adverse drug events. Iraqi J. Pharm. Sci. 20(2), 85–90. Piquereau, J., Moulin, M., Zurlo, G., Mateo, P., Gressette, M., Paul, J., Lemaire, C., Ventura-Clapier, R., Veksler, V. and Garnier, A. 2016. Cobalamin and folate protect mitochondrial and contractile functions in a murine model of cardiac pressure overload. J. Mol. Cell. Cardiol. 102, 34–44; doi:10.1016/j.yjmcc.2016.11.010 Ponikowska, B., Iwanek, G., Zdanowicz, A., Urban, S., Zymliński, R., Ponikowski, P. and Biegus, J. 2022. Biomarkers of myocardial injury and remodeling in heart failure. J. Pers. Med. 12(5), 799; doi:10.3390/jpm12050799 Sachdeva, J., Dai, W. and Kloner, R.A. 2014. Functional and histological assessment of an experimental model of Takotsubo’s cardiomyopathy. J. Am. Heart. Assoc. 3(3), e000921; doi:10.1161/jaha.114.000921 Santoso, D.I.S., Murthi, A.K., Yolanda, S., Amani, P. and Sianipar, I.R. 2019. The impact of cobalamin deficiency on heart function: a study on abnormalities in electrocardiography patterns. Int. J. Appl. Pharm. 11(6), 33–36; doi:10.22159/ijap.2019.v11s6.33533 Song, Z., Zhong, X., Ning, Z. and Song, X. 2022. The protective effect of MIR-27-3P on ischemia-reperfusion-induced myocardial injury depends on HIF-1A and galectin-3. J. Cardiovasc. Transl. Res. 15(4), 772–785; doi:10.1007/s12265-021-10203-y Sygitowicz, G., Maciejak-Jastrzębska, A. and Sitkiewicz, D. 2021. The diagnostic and therapeutic potential of galectin-3 in cardiovascular diseases. Biomolecules 12(1), 46; doi:10.3390/biom12010046 Van De Lagemaat, E.E., De Groot, L.C.P.G.M. and Van Den Heuvel, E.G.H.M. 2019. Vitamin B12 in relation to oxidative stress: a systematic review. Nutrients 11(2), 482; doi:10.3390/nu11020482 Yanni, A.E. 2004. The laboratory rabbit: an animal model of atherosclerosis research. Lab. Anim. 38(3), 246–256; doi:10.1258/002367704323133628 | ||
| How to Cite this Article |
| Pubmed Style Asker MH, Husain WM, Khaleel HK, Abdullah MR. Physiological, molecular, and histological evaluation of the effects of cobalamin on cardiac function in castrated rabbits. Open Vet. J.. 2026; 16(2): 1082-1089. doi:10.5455/OVJ.2026.v16.i2.28 Web Style Asker MH, Husain WM, Khaleel HK, Abdullah MR. Physiological, molecular, and histological evaluation of the effects of cobalamin on cardiac function in castrated rabbits. https://www.openveterinaryjournal.com/?mno=286080 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.28 AMA (American Medical Association) Style Asker MH, Husain WM, Khaleel HK, Abdullah MR. Physiological, molecular, and histological evaluation of the effects of cobalamin on cardiac function in castrated rabbits. Open Vet. J.. 2026; 16(2): 1082-1089. doi:10.5455/OVJ.2026.v16.i2.28 Vancouver/ICMJE Style Asker MH, Husain WM, Khaleel HK, Abdullah MR. Physiological, molecular, and histological evaluation of the effects of cobalamin on cardiac function in castrated rabbits. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1082-1089. doi:10.5455/OVJ.2026.v16.i2.28 Harvard Style Asker, M. H., Husain, . W. M., Khaleel, . H. K. & Abdullah, . M. R. (2026) Physiological, molecular, and histological evaluation of the effects of cobalamin on cardiac function in castrated rabbits. Open Vet. J., 16 (2), 1082-1089. doi:10.5455/OVJ.2026.v16.i2.28 Turabian Style Asker, Mohammed Hayder, Wassan Mhammed Husain, Hadeel Kamil Khaleel, and Mustafa Riyadh Abdullah. 2026. Physiological, molecular, and histological evaluation of the effects of cobalamin on cardiac function in castrated rabbits. Open Veterinary Journal, 16 (2), 1082-1089. doi:10.5455/OVJ.2026.v16.i2.28 Chicago Style Asker, Mohammed Hayder, Wassan Mhammed Husain, Hadeel Kamil Khaleel, and Mustafa Riyadh Abdullah. "Physiological, molecular, and histological evaluation of the effects of cobalamin on cardiac function in castrated rabbits." Open Veterinary Journal 16 (2026), 1082-1089. doi:10.5455/OVJ.2026.v16.i2.28 MLA (The Modern Language Association) Style Asker, Mohammed Hayder, Wassan Mhammed Husain, Hadeel Kamil Khaleel, and Mustafa Riyadh Abdullah. "Physiological, molecular, and histological evaluation of the effects of cobalamin on cardiac function in castrated rabbits." Open Veterinary Journal 16.2 (2026), 1082-1089. Print. doi:10.5455/OVJ.2026.v16.i2.28 APA (American Psychological Association) Style Asker, M. H., Husain, . W. M., Khaleel, . H. K. & Abdullah, . M. R. (2026) Physiological, molecular, and histological evaluation of the effects of cobalamin on cardiac function in castrated rabbits. Open Veterinary Journal, 16 (2), 1082-1089. doi:10.5455/OVJ.2026.v16.i2.28 |