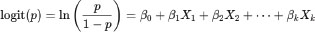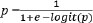| Research Article | ||
Open Vet. J.. 2026; 16(3): 1565-1577 Open Veterinary Journal, (2026), Vol. 16(3): 1565-1577 Research Article Prevalence of subclinical mastitis and associated biosecurity practices in peri-urban dairy farmsSutiastuti Wahyuwardani1, Sri Suryatmiati Prihandani1, Raphaella Widiastuti1, Prima Mei Widiyanti1, Yessi Anastasia1, Eny Martindah1, Romsyah Maryam1, Susan Maphilindawati Noor1, Rini Damayanti1, Yulvian Sani1, Aswin Rafif Khairullah1, Eni Siti Rohaeni2* and Priyono Priyono31Research Center for Veterinary Science, National Research and Innovation Agency (BRIN), Bogor, Indonesia 2Research Center for Animal Husbandry, National Research and Innovation Agency (BRIN), Bogor, Indonesia 3Research Center for Behavioral and Circular Economics, National Research and Innovation Agency (BRIN), Kuningan, Indonesia *Corresponding Author: Eni Siti Rohaeni. Research Center for Animal Husbandry, National Research and Innovation Agency (BRIN), Bogor, Indonesia. Email: eni_najib [at] yahoo.co.id Submitted: 30/09/2025 Revised: 22/01/2026 Accepted: 06/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractBackground: Subclinical mastitis (SCM) is a major constraint in peri-urban smallholder dairy production. Inconsistent biosecurity and milking hygiene may increase infection risk and productivity losses. Aim: This study aimed to assess the association between biosecurity practices (external, internal, and procedural) and SCM prevalence and severity in peri-urban dairy farms in Greater Jakarta (Jakarta, Bogor Regency, Bogor City, and Depok), Indonesia. Methods: A cross-sectional survey was conducted in 34 eligible smallholder dairy farms from April to June 2025 using structured interviews, direct observations of biosecurity practices, and California Mastitis Test (CMT). Farm-level SCM severity was defined as moderate (1) if ≥1 tested cow had CMT 2–3 in any quarter, and mild (0) otherwise. Associations were assessed using chi-square tests and binary logistic regression, and model performance was evaluated using receiver operating characteristic analysis (SPSS v26). Results: The prevalence of cow-level SCM was 72.9% (105/144; 95% CI: 65.7%–80.2%), ranging from 57.1% in Depok to 90.2% in Bogor City. For farm-level analyses (n=34), 16 of 34 farms (47.1%) were classified as moderate and 18 of 34 (52.9%) as mild. Procedural biosecurity was significantly associated with SCM severity (mild vs. moderate; χ²=6.959, p=0.008), whereas external and internal biosecurity were not (p > 0.05). In the parsimonious logistic regression model, a higher farmer education category was associated with higher odds of moderate SCM [odds ratio (OR)=1.369, p=0.027], whereas procedural biosecurity was protective (OR=0.449, p=0.011). The final model demonstrated good discrimination (area under the curve=0.845). Conclusion: Procedural biosecurity (milking and hygiene-related practices) was the only biosecurity dimension significantly associated with SCM severity and was protective against moderate SCM in peri-urban smallholder dairy farms in Indonesia. Strengthening cost-effective milking hygiene and targeted farmer training may help reduce the SCM burden in comparable settings. Keywords: Biosecurity, Subclinical mastitis, Dairy cattle, Peri-urban farming, California Mastitis Test. IntroductionMastitis remains one of the most devastating diseases in the dairy cattle industry worldwide (Ruegg, 2017). Infection is commonly caused by bacteria such as Staphylococcus aureus, Streptococcus agalactiae, Streptococcus uberis, and Escherichia coli, but non-traditional pathogens, such as coagulase-negative staphylococci, are increasingly reported in both clinical and subclinical cases (Becker et al., 2022). Factors influencing the role of pathogens include microbial virulence, environmental conditions, and host immune response (Park and Ronholm, 2021). Subclinical mastitis (SCM) is of particular concern because it is characterized by an increase in somatic cell count (SCC) without visible udder or milk changes, leading to decreased production, increased treatment costs, and higher culling rates (Chen et al., 2022). Even when clinical signs are absent or appear to resolve, subclinical infections may persist, allowing infected quarters to continue transmitting pathogens within the herd. This underscores the importance of early detection through SCC or California Mastitis Test (CMT) and the need for a more comprehensive preventive approach. Mastitis control requires an integrated One Health strategy that encompasses biosecurity, nutritional optimization, judicious antimicrobial use, and precision monitoring technologies (Silva et al., 2021a; Zanon et al., 2024). Another challenge stems from the genetic diversity and high levels of antimicrobial resistance in mastitis pathogens, which complicates empirical therapy and emphasizes the need for locally tailored control measures (Moawad et al., 2023). Practices such as dry cow therapy can also worsen resistance, making selective dry cow therapy (SDCT) a recommended approach. However, SDCT implementation requires SCC monitoring, consistent record-keeping, and veterinary supervision—conditions that are difficult to achieve on small and peri-urban farms (Tarigan et al., 2025). The limited availability of antimicrobial stewardship programs highlights the need for innovative strategies, including digital monitoring (Neculai-Valeanu et al., 2024). Mastitis control approaches increasingly integrate animal welfare, public health, and environmental sustainability (Garcia et al., 2019; Ferronato et al., 2024). Modern biosecurity also encompasses community sustainability, particularly in peri-urban farms operating in densely populated areas with shared resources such as water, labor, and wastewater disposal systems. Preventive measures include nutritional management, adequate housing, stress reduction, and biosecurity protocols to minimize pathogen exposure (Toson et al., 2024; Zanon et al., 2024). Precision technologies, such as milk conductivity sensors and real-time SCC monitoring, can improve the early detection and rapid response of udder health disorders (Silva et al., 2021a). Peri-urban dairy farming is rapidly expanding in response to the increasing demand for fresh milk in urban areas (Moje et al., 2023). However, this system faces unique challenges, such as limited land, high herd density, and management variations, which increase the risk of pathogen transmission (Duguma, 2022). SCM often goes undetected, yet causes economic losses and reduced milk quality (Ferreira et al., 2022; Stanek et al., 2024). Prompt prevention relies heavily on milking hygiene, equipment sanitation, isolation of infected animals, and proper waste management in addition to early detection (Ferreira et al., 2024; Richardet et al., 2023). Implementing these practices is crucial given the peri-urban dairy farmers’ varying levels of knowledge and resources. In Indonesia, most policies remain reactive and focus on milk quality standards, whereas a structured biosecurity framework for peri-urban smallholder farms is lacking (Kustiningsih et al., 2023). Most national studies focus on rural or large-scale farms (Nuraini et al., 2023), resulting in limited epidemiological data on SCM, risk factors, and biosecurity for smallholder farms in peri-urban areas—such as Jakarta, Bogor Regency, Bogor City, and Depok. This data gap limits the development of intervention strategies tailored to the specific needs of peri-urban production systems. Although global studies have evaluated mastitis risk factors, the role of external, internal, and procedural biosecurity dimensions in controlling SCM on smallholder peri-urban dairy farms has not been systematically examined in Indonesia (Ruegg, 2017; Khasanah et al., 2021). This is despite unique characteristics such as limited space, high herd density, and management heterogeneity, which influence disease dynamics and biosecurity compliance. Previous studies have primarily described structural challenges without assessing how these factors influence infection dynamics and biosecurity implementation in the field (Auplish et al., 2025). Despite extensive global research on mastitis, evidence disentangling the roles of external, internal, and procedural biosecurity in SCM among peri-urban smallholder dairy farms in Indonesia remains limited, given heterogeneous management practices and pronounced space and resource constraints. This study addresses this gap by (i) estimating the prevalence of cow-level SCM using standardized quarter-level CMT scoring and (ii) assessing which biosecurity dimension is most strongly associated with SCM severity under real-world peri-urban conditions, providing a practical basis for targeted, low-cost interventions. Therefore, this study aimed to examine the associations between farm biosecurity practices (external, internal, and procedural) and the prevalence of cow-level SCM and to identify which biosecurity dimension was most strongly associated with SCM severity in peri-urban smallholder dairy farms in Indonesia. Materials and MethodsStudy area and designThis cross-sectional study was conducted between April and June 2025 in peri-urban dairy farming areas of the Greater Jakarta region, Indonesia, including Bogor Regency, Bogor City, and Depok, (Fig. 1). Locations were selected based on high dairy cattle density and diverse management practices typical of peri-urban production systems (Al Zahra et al., 2024). This study used a cross-sectional design to estimate the prevalence of SCM at the cow level and identify farm-level biosecurity practices associated with SCM severity. As an inherent limitation, cross-sectional studies allow only the identification of associations rather than causal relationships because exposure and outcome are measured at a single time point. The cross-sectional design was chosen to provide rapid baseline epidemiological information and to generate preliminary evidence to guide future longitudinal or intervention studies.
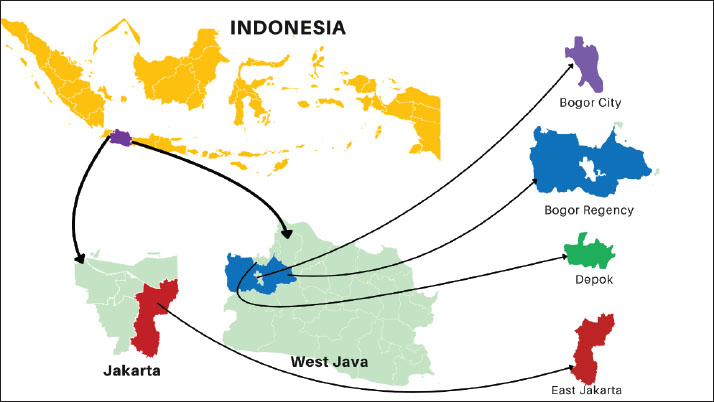
Fig. 1. Study site in peri-urban dairy farming areas of Indonesia (Source: Authors’ own elaboration). Sampling and participantsFarms were approached through local dairy networks/cooperatives in each district. Eligibility criteria were as follows: (i) peri-urban smallholder dairy farm, (ii) availability of lactating cows at the visit, and (iii) willingness to participate in the interview, observation, and CMT testing. Of the 40 farms contacted, 34 met the eligibility criteria and provided complete data; 6 were excluded (2 declined to participate, and 4 had insufficient lactating cows available for CMT testing). All lactating cows present at the visit were tested (n=144). While purposive sampling allows targeting farms relevant to the study objectives, it may introduce selection bias because participating farms might systematically differ from non-participating farms in terms of management practices or in the adoption of biosecurity measures. We cross-verified farm characteristics (herd size and location) with regional agricultural census data to mitigate potential bias and found no significant differences (p > 0.05, chi-square test). The relatively small sample size and the non-random selection of farms may lead to potential sampling errors, which may limit the precision of prevalence estimates and the generalizability of the findings. Results may not fully represent all smallholder peri-urban dairy farms in the region, particularly those less willing to participate or with distinct management practices. Despite these limitations, the sampling strategy was considered appropriate for generating preliminary insights and identifying associations between biosecurity practices and SCM in resource-limited peri-urban settings (Das et al., 2018). Herd size classificationFarms were classified into three herd-size categories based on the number of lactating cows present at the visit: small-scale (<5), medium-scale (6–20), and large-scale (>20). In this study, 4 farms were small-scale, 20 medium-scale, and 10 large-scale. These thresholds reflect the typical structure of smallholder dairy production in Indonesia. In West Java, most dairy farms are small-scale, with an average of 3 lactating cows per household (Fadillah et al., 2023). Consistent with the more intensive peri-urban context of Greater Jakarta, the sample included a higher proportion of medium- and large-scale farms. Data collectionData confidentiality was maintained through anonymization. Data were collected through three complementary approaches: (a) structured interviews captured farmer demographics (age, education, and farming experience) and farm characteristics (herd size and location); (b) on-farm observations verified the implementation of biosecurity measures and cross-checked interview responses; and (c) milk sampling was performed during the same visit to minimize disruption to routine farm activities. The questionnaire focused exclusively on farm management and biosecurity practices. Interview responses were cross-checked with on-farm observations to minimize reporting bias. Nevertheless, some practices (e.g., udder antiseptic hygiene use and handling of a cow with mastitis) may not be directly observable during a single visit and, therefore, remain susceptible to recall or social desirability bias. Biosecurity assessmentBiosecurity practices were assessed using a validated questionnaire adapted from livestock health and hygiene guidelines (Pedersen et al., 2023). The instrument comprised three dimensions: external biosecurity (9 items; e.g., controlled farm entry, visitor/vehicle hygiene, quarantine practices, and feed/water protection), internal biosecurity (6 items; e.g., separation of clean/dirty areas, waste management, and availability of hygiene facilities), and procedural biosecurity (9 items; routine operational measures related to milking and hygiene, including udder hygiene, cleaning/disinfection routines, and handling of cows suspected of mastitis). Each item was scored dichotomously (1=implemented, 0=not implemented). Subscores were calculated for external (0–9), internal (0–6), and procedural (0–9) biosecurity and summed to obtain a total biosecurity score (0–24). Based on score distribution and prior validation (Pedersen et al., 2023), farms were classified as having adequate biosecurity (score ≥12; ≥50% compliance) or inadequate biosecurity (score <12). The cutoff was supported by pilot testing (n=5 farms) and expert consultation. Milk sampling and CMT analysisCMT scores were interpreted as follows: 0=negative, 1=trace/mild SCM, 2=moderate SCM; and 3=severe SCM (National Mastitis Council, 2017). CMT was performed on all four quarters of each lactating cow using the 0–T–1–2–3 scoring scale. The highest quarter score was used to classify the SCM status of each cow. For consistency, all tests were performed by two trained assistants under veterinarian supervision, with joint calibration and repeat scoring of ambiguous reactions. Cow-level SCM prevalence was calculated as the proportion of cows with CMT ≥1 within each farm. For farm-level inferential analyses (chi-square and logistic regression), each farm was classified as moderate (code 1) if at least one tested cow had the highest quarter CMT score of 2–3 and mild (code 0) otherwise. This pragmatic dichotomization distinguished farms with evidence of higher subclinical inflammation from those with only trace/mild reactions under field conditions. Statistical analysisAll data analyses were performed using the Statistical Package for the Social Sciences version 26. The descriptive statistics included frequencies, means, medians, standard deviations, and 95% confidence intervals. All inferential analyses were conducted at the farm level (n=34), consistent with farm-level biosecurity assessment. Bivariate associations between each biosecurity dimension (external, internal, and procedural) and farm-level SCM severity (0=mild, 1=moderate) were examined using chi-square tests. Subsequently, binary logistic regression was used to assess associations between farmer/farm characteristics and biosecurity dimensions with farm-level SCM severity. A complete model (Model 1, full model) initially included all the predictors. Then, backward stepwise elimination was applied to remove non-significant variables and obtain a parsimonious model (Model 2). Model performance was evaluated using the Omnibus test, Nagelkerke R², and the Hosmer–Lemeshow goodness-of-fit statistic. Model discrimination was assessed using receiver operating characteristic (ROC) curves and the area under the curve (AUC), together with the corresponding sensitivity, specificity, and overall accuracy. The predicted probability (p) for moderate farm-level SCM was derived from the fitted logistic regression model, expressed as follows:
as
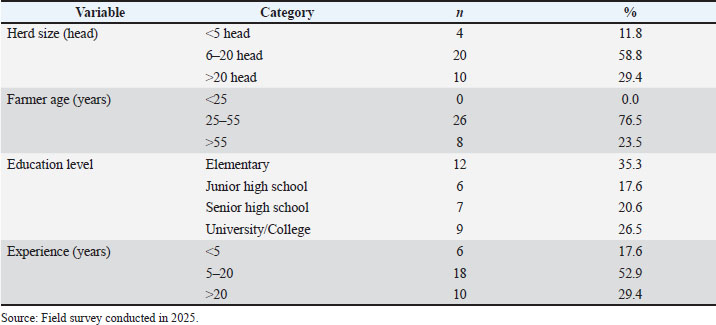
where p is the predicted probability that a farm was classified as moderate (1) rather than mild (0), statistical significance was set at p < 0.05. Ethical approvalNot needed for this study. The study involved non-invasive procedures (questionnaire-based interviews and on-farm observation) and excluded biological sampling beyond routine farm practice. All participants provided verbal informed consent before data collection. The ethical review requirement was determined in accordance with the applicable institutional/national guidelines. ResultsFarm characteristics and characteristicsA total of 34 smallholder dairy farmers were included in the analysis. Table 1 summarizes the characteristics of the participating farms. Most farms (58.8%) maintained 6–20 lactating cows, 29.4% had more than 20, and only 11.8% had 5 or fewer. Most farmers were in the productive age group of 41–55 years (72.4%), with a predominance of low educational attainment; nearly half (48%) had only completed elementary school, and only 7% had tertiary education. Farming experience varied, with most farmers having 5–20 years (52.9%), followed by those with more than 20 years (29.4%), while only 17.6% had less than 5 years. This predominance of moderately experienced farmers is consistent with previous findings in Bogor, where experience significantly influenced farmers’ knowledge, attitudes, and practices in livestock disease control programs (Kustiningsih et al., 2023). Table 1. The characteristics of participating farms and farmers are summarized.
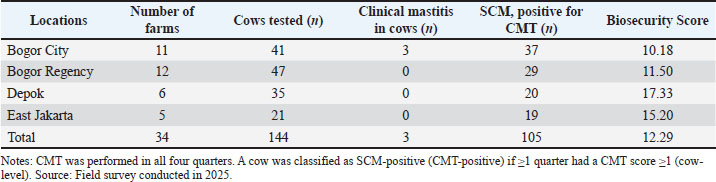
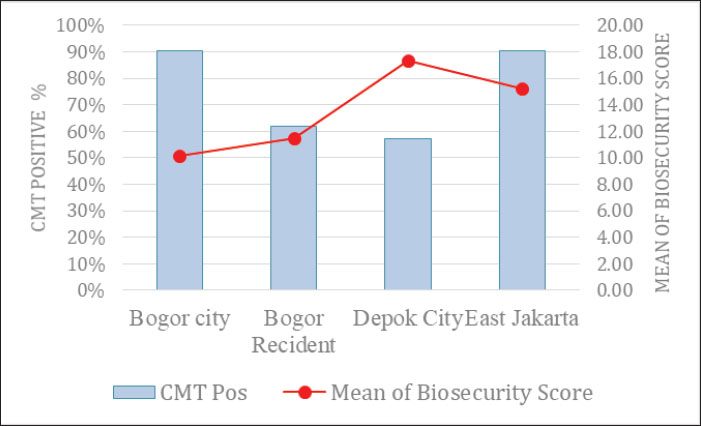
Prevalence of subclinical mastitisThe prevalence of cow-level SCM was 72.9% (105/144; 95% CI: 65.7%–80.2%). At the farm level (n=34), 16 farms (47.1%) were classified as moderate (≥1 tested cow with the highest quarter CMT score of 2–3), and 18 farms (52.9%) were classified as mild (all tested cows had the highest quarter CMT score of 0–1). The district-level cow prevalence ranged from 57.1% (Depok) to 90.2% (Bogor City). Consistent with the inferential analyses conducted at the farm level, the proportion of moderate farms varied across districts (Table 2; Fig. 2). Table 2. Distribution of mastitis status (clinical and subclinical) and biosecurity scores by location (n=10).
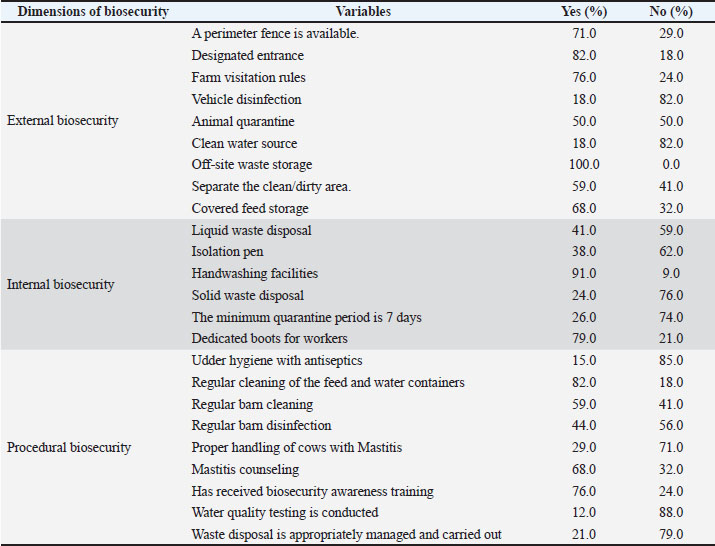
Fig. 2. CMT positivity percentage and mean biosecurity score by location. (Source: Authors’ own data analysis, 2025). Biosecurity implementationExternal biosecurity was implemented in a variety of ways: off-site waste storage was universal (100%), and most farms reported a designated entrance (82%) and visitor rules (76%). Covered feed storage was reported by 68%, whereas vehicle disinfection and access to a clean water source were rare (both 18%) (Table 3). Basic facilities dominated internal biosecurity, with handwashing facilities (91%) and dedicated boots for workers (79%) being the most common, while isolation pens (38%) and a minimum 7-day quarantine period (24%) were less frequently reported (Table 3). Procedural biosecurity practices were generally higher for routine cleaning and training, regular cleaning of feed/water containers (82%) and training (76%), but critical practices such as water quality testing (12%), udder hygiene with antiseptic (15%), and proper handling of mastitis cows (29%) remained limited (Table 3). Table 3. Parameters for external, internal, and procedural biosecurity assessment in dairy cattle farms.
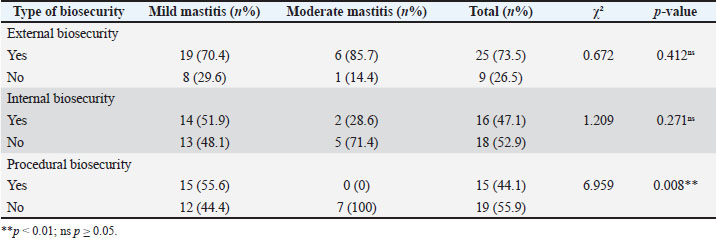
Association between biosecurity and severity of SCMChi-square analysis showed no significant association between external or internal biosecurity and SCM severity (external: χ²=0.672, p=0.412; internal: χ²=1.209, p=0.271). Procedural biosecurity was significantly associated with SCM severity (χ²=6.959, p=0.008) (Table 4). Given the presence of a zero cell, the association was also checked using an exact test, yielding consistent significance (p < 0.05). Notably, all moderate SCM cases occurred in farms without procedural biosecurity (7/7; 100%), whereas none were observed in farms with procedural biosecurity (0/15; 0%). Table 4. Chi-square results between biosecurity and SCM severity level (mild vs. moderate).
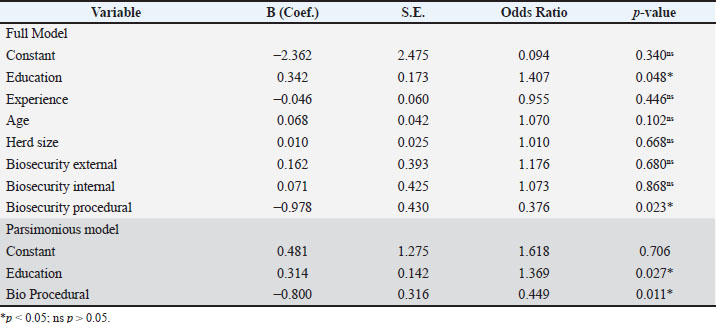
Binary logistic regression of biosecurity predictors of mastitisBinary logistic regression was performed to identify farmer- and farm-level SCM severity predictors (0=mild; 1=moderate). In the full model (Model 1), education level was positively associated with the odds of moderate SCM [odds ratio (OR)=1.407, 95% confidence interval (CI): 1.003–1.975; p=0.048], whereas adequate procedural biosecurity was associated with lower odds of moderate SCM (OR=0.376, 95% CI: 0.162–0.874; p=0.023). Other covariates (experience, age, flock size, and external and internal biosecurity) were not herd size different (p > 0.05). The parsimonious model (Model 2), including only education and procedural biosecurity, produced consistent estimates (education: OR=1.369, 95% CI: 1.036–1.809; p=0.027; procedural biosecurity: OR=0.449, 95% CI: 0.242–0.835; p=0.011). The detailed regression results are presented in Table 5. Table 5. Binary logistic regression analysis of factors associated with SCM.
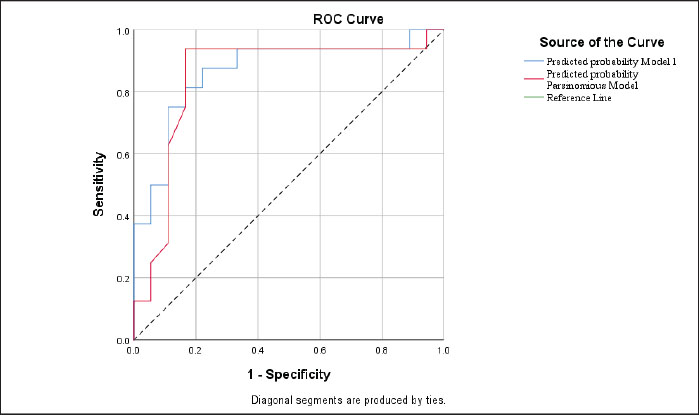
ROC curve analysisThe ROC curve analysis showed good discriminative performance for both models in distinguishing mild from moderate SCM. The full model achieved an AUC of 0.865, whereas the parsimonious model achieved an AUC of 0.845 (Fig. 3). A note of tied predicted probabilities was reported in the ROC output, which may slightly influence the AUC estimate precision. These findings indicate that both models can discriminate between mild and moderate SCM (Fig. 3).
Fig. 3. ROC curves for predicting mild versus moderate severity of subclinical mastitis using the full and parsimonious logistic regression models (Source: Authors’ analysis, 2025). DiscussionBiosecurity practices and prevalence of subclinical mastitisFarmer characteristics influence the implementation of biosecurity practices (Nyokabi et al., 2023). In our study, adoption patterns were heterogeneous: some farmers implemented simple, low-cost measures such as handwashing facilities and basic waste disposal, whereas others were reluctant to invest in more resource-intensive practices (e.g., vehicle disinfection or quarantine pens), largely due to financial constraints (Postma et al., 2016; Buckel et al., 2024). Education and farming experience may also shape compliance, with better-informed farmers tending to adopt additional precautionary measures (Ritter et al., 2017; Otieno et al., 2023). Risk perception further contributes to variability: in the absence of recent outbreaks, several farmers perceived disease threats as minimal and relaxed routine preventive behaviors (Loeillot et al., 2025). Local norms may also affect adherence, as farmers may mimic neighboring farms rather than follow formal guidelines (Buckel et al., 2024). This heterogeneity is also consistent with evidence that resources and behavioral determinants shape biosecurity compliance (Alavedra et al., 2025). Perceived costs remain a commonly reported barrier in smallholder settings (Lestari et al., 2022). Similar cost-related constraints have also been reported in Ethiopia, where high compliance costs for milk safety practices hinder adoption and may affect food and nutrition security (Feyisa et al., 2023). Time burden is another important constraint; 20.87% of farmers in Indonesia reported that time limitations reduced the uptake of biosecurity measures (Lestari et al., 2022). Risk perception is another key factor: when farmers underestimate the likelihood of disease, motivation to implement preventive routines declines, as reported in dairy systems where individual perceptions of biosecurity strongly influenced compliance (Power et al., 2024). Social influences further affect adoption. Trusted advisors, particularly veterinarians, can increase uptake; for example, 76% of farmers indicated that they would follow veterinarians’ recommendations in one European context (Toson et al., 2024). Peer comparison and perceived social norms may also reinforce implementation, with evidence that greater perceived importance of biosecurity correlates with higher adoption (Renault et al., 2018). Finally, knowledge gaps remain critical: 64.35% of farmers in Indonesia cited insufficient knowledge as a primary barrier to biosecurity adoption (Lestari et al., 2022), supporting the need for targeted training and coaching approaches; structured training interventions have been shown to improve biosecurity-related practices in other settings (Khan et al., 2024). Although financial limitations can restrict the uptake of infrastructure- or capital-intensive measures, low-cost procedural routines, such as udder cleaning, equipment sanitation, and worker hygiene, remain feasible and should be prioritized regardless of herd size or capital availability. Peer norms can influence compliance, but strengthening procedural routines offers a practical entry point for behavior change without major investment (Kalverkamp et al., 2025). These gaps in routine biosecurity have clear implications for udder health, particularly given the widespread occurrence of SCM. SCM was notably more prevalent than clinical mastitis, consistent with its largely asymptomatic nature and limited routine testing in many smallholder systems (Ranasinghe et al., 2021). A meta-analysis from African dairy farms reported an average SCM prevalence of 48% (Khasapane et al., 2023), and similar patterns have been linked to poor udder hygiene and suboptimal milking routines (El Daous et al., 2025). Our findings are consistent with this global pattern. SCM prevalence varied across locations in the study area and was closely linked to procedural biosecurity indicators. Bogor City had the highest proportion of CMT-positive samples (90.2%) and the lowest procedural biosecurity scores, particularly in udder hygiene and water management (Silva et al., 2021b; Zanon et al., 2024). Depok exhibited better biosecurity and correspondingly lower SCM prevalence, whereas Bogor Regency fell between these extremes, possibly reflecting greater farming experience and engagement in extension programs. The situation in East Jakarta indicates that access to clean water alone does not guarantee lower SCM levels; consistent milking hygiene, proper equipment sanitation, and adequate separation of affected cows remain essential. Although SCM prevalence differed across locations, our inferential analysis focused on SCM severity (mild vs. moderate). External and internal biosecurity measures were not significantly associated with SCM severity across locations, whereas procedural biosecurity emerged as the only significant predictor, supporting the view that daily operational routines may have the most immediate impact on pathogen transmission (Haldar et al., 2022). The difficulty of implementing SDCT on smallholder farms further underscores the broader challenges identified in this study. SDCT requires reliable diagnostic information, skilled labor, and structured treatment protocols, which are often unavailable or inconsistently applied in small-scale operations (Saleh et al., 2022). These constraints highlight the need for strengthened extension services and practical, low-cost support tools to guide evidence-based udder health management in resource-limited contexts. Procedural measures, such as udder hygiene, cleaning of milking equipment, proper handling of mastitis-affected cows, and worker hygiene, directly influence bacterial exposure at the teat end. External biosecurity (e.g., fencing, visitor control) and internal measures (e.g., waste management, isolation pens) primarily affect pathogen introduction or environmental load, which may not immediately translate into changes in udder health. Thus, procedural routine deficiencies may have a more pronounced association with SCM severity in smallholder peri-urban systems. Determinants of mastitis risk and implications of the studyOur chi-square and logistic regression analyses indicate that adherence to procedural biosecurity routines was consistently associated with more favorable SCM outcomes, supporting the prioritization of routine hygiene and milking practices. The positive association between education level and moderate SCM should be interpreted with caution. Although education was a significant predictor, this relationship may reflect unmeasured management and labor-related factors (e.g., herd intensification, milking task delegation to hired labor, supervision quality, and hygiene routine variability) rather than a direct causal effect of education (Chen et al., 2023). Behavioral and managerial attributes, such as employee supervision, routine adherence, and task delegation, may exert stronger influences on udder health than formal education alone (Lind et al., 2023). Similar findings from other contexts indicate that higher awareness does not necessarily translate into preventive behavior, particularly for mastitis control (Zhang et al., 2022). Training, coaching, and peer-supported routines have been shown to improve procedural compliance, reduce somatic cell counts, and enhance udder health outcomes (Amalraj et al., 2024; Keba et al., 2025). Strengthening preventive strategies remains more effective than relying on antimicrobial treatment (Demil et al., 2022). Our results provide insights from peri-urban Indonesia, an underrepresented context in mastitis research, and highlight the need for practical, context-specific interventions. Priorities should focus on low-cost, high-impact procedural measures, including udder hygiene, equipment sanitation, and water management. Farmer cooperatives can serve as platforms for routine training, peer learning, and shared monitoring (e.g., periodic udder health scoring or CMT screening) (Okello et al., 2023). Extension officers and dairy advisors may support this process through hands-on coaching, checklists, and structured milking protocols to reinforce consistent routines. These cooperative-based and behavior-focused strategies are feasible within existing Indonesian dairy systems and can help reduce SCM, improve milk quality, and enhance the sustainability of peri-urban dairy production (Dobrut et al., 2024). Study limitationsSeveral limitations should be acknowledged. The findings describe associations between biosecurity practices and SCM outcomes but cannot establish causality because the study used a cross-sectional design; longitudinal or intervention-based studies would be more appropriate for evaluating causal pathways (Kumar et al., 2023). In addition, although direct observations were conducted, some biosecurity practices were self-reported, which may introduce social desirability or recall bias and potentially overestimate compliance, particularly for procedural biosecurity (Alhussien and Dang, 2020). The sample size was relatively small (34 farms and 144 lactating cows), and all the sites were located in the peri-urban areas of Greater Jakarta. Therefore, the findings may not be fully generalizable to rural dairy systems or larger commercial operations. Farm-level characteristics such as access to veterinary services, labor availability, economic constraints, and cow genetics were not captured, limiting the ability to account for all relevant factors influencing udder health (Gilbert et al., 2021). Information bias may also be present. Although structured interviews were complemented by on-farm observations, some practices (e.g., udder hygiene, milking routines, and mastitis-handling procedures) may not have been observable during a single visit and may be influenced by recall or social desirability bias (Kappes et al., 2023). CMT scoring relies on a subjective visual assessment and may result in minor misclassification of SCM severity; future studies should validate CMT findings using laboratory-based SCC measurements or bacteriological cultures (National Mastitis Council, 2017). The consistent association observed for procedural biosecurity suggests that routine, low-cost hygiene practices remain a plausible and actionable leverage point for SCM control in similar peri-urban settings, although causal inference cannot be established from this cross-sectional design. Despite these limitations, the study provides valuable preliminary evidence to inform mastitis prevention strategies in peri-urban dairy systems and supports follow-up research with larger samples, repeated observations, and a broader range of behavioral and managerial variables. ConclusionThis study provides evidence to inform strategies for strengthening dairy health programs in peri-urban Indonesia. The findings suggest that improved procedural biosecurity, particularly routine udder hygiene, equipment sanitation, and water management, may serve as a feasible and cost-effective leverage point for mitigating SCM burden in resource-constrained settings. Extension programs should prioritize hygiene-focused training modules and support peer-learning networks, cooperative-based mentoring, and accessible veterinary advisory services to strengthen routine milking practices. Such approaches can improve milk quality, reduce avoidable production losses, and support efforts to optimize antimicrobial use, thereby contributing to sustainable dairy development and the One Health agenda. Overall, education level and procedural biosecurity were identified as key determinants of SCM severity in peri-urban dairy farms. Farms with better adherence to procedural routines tended to have less severe SCM. These results support the development of low-cost, targeted interventions for smallholder systems. Future studies using larger samples and longitudinal or intervention designs are warranted to confirm effectiveness and inform scalable policy implementation. AcknowledgmentsThe authors would like to thank the National Research and Innovation Agency and PT Kuntum Hijau Lestari (Kuntum Farmfield) for accuracy their animals to be used as research samples. Conflict of interestThe authors declare no conflict of interest. FundingThis study is part of a research project funded by the Research Organization for Health, the National Research and Innovation Agency of Indonesia. Author’s contributionsSW and SSP: Conceived, designed, and coordinated the study. RW, PP, and PMW: Designed data collection tools, supervised the field sample and data collection, and performed laboratory work and data entry. YA and EM: Validation, supervision, and formal analysis of data. ARK, RM, and SMN: contributed analysis tools. RD, YS, and ESR: Carried out the statistical analysis and interpretation and participated in the preparation of the manuscript. All authors have read, reviewed, and approved the final version of the manuscript. Data availabilityAll data are available in the revised manuscript. ReferencesAl Zahra, W., Van Middelaar, C.E., Oosting, S.J. and De Boer, I.J.M. 2024. Nutrient imbalances of smallholder dairy farming systems in Indonesia: the relevance of manure management. Agric. Syst. 218(1), 103961. Alavedra, M., Moura, D., Cenci-Goga, B., Saraiva, S., Silva, F., Pires, I., Saraiva, C., Coelho, A.C. and García-Díez, J. 2025. Biosecurity practices in Portuguese small ruminant farms: current status and future directions. Vet. Sci. 12(4), 334. Alhussien, M.N. and Dang, A.K. 2020. Sensitive and rapid lateral-flow assay for early detection of subclinical mammary infection in dairy cows. Sci. Rep. 10(1), 11161. Amalraj, A., Van Meirhaeghe, H., Chantziaras, I. and Dewulf, J. 2024. Assessing the impact of on-farm biosecurity coaching on farmer perception and farm biosecurity status in Belgian poultry production. Animals. 14(17), 2498. Auplish, A., Mjuberi, K., Magwisha, H., Tago, D., Buckel, A., Ciamarra, U.P., Mclaws, M. and Heilmann, M. 2025. Using a co-created checklist to improve on-farm biosecurity: an observational pilot intervention with pig farmers and livestock field officers in Sumbawanga, Tanzania. Front. Vet. Sci. 12(1), 1567072. Becker, K., Both, A., Weißelberg, S., Heilmann, C. and Rohde, H. 2020. Emergence of coagulase-negative staphylococci. Expert. Rev. Anti. Infect. Ther. 18(4), 349–366. Buckel, A., Afakye, K., Koka, E., Price, C., Kabali, E. and Caudell, M.A. 2024. Understanding the factors influencing biosecurity adoption on smallholder poultry farms in Ghana: a qualitative analysis using the COM-B model and Theoretical Domains Framework. Front. Vet. Sci. 11(1), 1324233. Chen, S., Zhang, H., Zhai, J., Wang, H., Chen, X. and Qi, Y. 2023. Prevalence of clinical mastitis and its associated risk factors among dairy cattle in mainland China during 1982–2022: a systematic review and meta-analysis. Front. Vet. Sci. 10(1), 1185995. Chen, X., Chen, Y., Zhang, W., Chen, S., Wen, X., Ran, X., Wang, H., Zhao, J., Qi, Y. and Xue, N. 2022. Prevalence of subclinical mastitis among dairy cattle and associated risks factors in China during 2012-2021: a systematic review and meta-analysis. Res. Vet. Sci. 148(1), 65–73. Das, D., Panda, S.K., Jena, B. and Sahoo, A.K. 2018. Somatic Cell Count: a Biomarker for Early Diagnosis and Therapeutic Evaluation in Bovine Mastitis. Int. J. Curr. Microbiol. Appl. Sci. 7(03), 1459–1463. Demil, E., Teshome, L., Kerie, Y., Habtamu, A., Kumilachew, W., Andualem, T. and Mekonnen, S.A. 2022. Prevalence of subclinical mastitis, associated risk factors and antimicrobial susceptibility of the pathogens isolated from milk samples of dairy cows in Northwest Ethiopia. Prev. Vet. Med. 205(1), 105680. Dobrut, A., Siemińska, I., Sroka-Oleksiak, A., Drożdż, K., Sobońska, J., Mroczkowska, U. and Brzychczy-Włoch, M. 2024. Molecular and phenotypic identification of bacterial species isolated from cows with mastitis from three regions of Poland. BMC Vet. Res. 20(1), 193. Duguma, B. 2022. Farmers' perceptions of major challenges to smallholder dairy farming in selected towns of Jimma Zone, Oromia Regional State, Ethiopia: possible influences, impacts, coping strategies and support required. Heliyon 8(6), 9581. El Daous, H., Alm El Din, N., Nafei, E., Abdallah, M., Abd-el Hamed, A.M., Abdel-Wadoud, I.M., Elgazzar, E., Elkhayat, M. and Hafez, E. 2025. Biosecurity measures, bacterial prevalence, and economic implications of environmental mastitis and hygienic milking practices on an Egyptian dairy farm. Int. J. Vet. Sci. 14(1), 7989. Fadillah, A., Van Den Borne, B.H.P., Poetri, O.N., Hogeveen, H., Umberger, W., Hetherington, J. and Schukken, Y.H. 2023. Smallholder milk-quality awareness in Indonesian dairy farms. J. Dairy. Sci. 106(11), 7965–7973. Ferreira, J.S., Baccili, C.C., Nemoto, B.S., Vieira, F.K., Sviercoski, L.M., Ienk, T., Pagno, J.T. and Gomes, V. 2024. Biosecurity practices in the dairy farms of southern Brazil. Front. Vet. Sci. 11(1), 1326688. Ferronato, G., Simonetto, A., Gilioli, G. and Zecconi, A. 2024. Modeling mastitis risk management effects on dairy milk yield and global warming potential. Animals 15(1), 50 Feyisa, B.W., Haji, J. and Mirzabaev, A. 2023. The impact of adoption of milk safety practices on food and nutrition security: evidence from smallholder dairy farmers in Ethiopia. Res. Glob. 7(1), 100157. Garcia, S.N., Osburn, B.I. and Cullor, J.S. 2019. A one health perspective on dairy production and dairy food safety. One. Health. 7(1), 100086. Gilbert, W., Thomas, L.F., Coyne, L. and Rushton, J. 2021. Mitigating the risks posed by intensification in livestock production: the examples of antimicrobial resistance and zoonoses. Animal 15(2), 100123. Haldar, L., Raghu, H.V. and Ray, P.R. 2022. Milk and Milk Product Safety and Quality Assurance for Achieving Better Public Health Outcomes. In Agriculture, Livestock Production and Aquaculture. Eds., Kumar, A., Kumar, P., Singh, S.S., Trisasongko, B.H. and Rani, M. Springer. Kalverkamp, K., Petzl, W. and Sorge, U.S. 2025. Risk factors for intramammary infections on Bavarian dairy farms—a herd-level analysis. Animals 15(17), 2616. Kappes, A., Tozooneyi, T., Shakil, G., Railey, A.F., Mcintyre, K.M., Mayberry, D.E., Rushton, J., Pendell, D.L. and Marsh, T.L. 2023. Livestock health and disease economics: a scoping review of selected literature. Front. Vet. Sci. 10(1), 1168649. Keba, A., Tola, A., Kaylegian, K.E., Kebede, M. and Zewdu, A. 2025. Impact of hygienic milk production training on knowledge, attitudes and practices of women farmers in the central highlands of Ethiopia. Front. Sustain. Food. Syst. 8(1), 1539559. Khan, J., Ali, A., Khan, S., Khan, M., Mohsin, S. and Madsen, C. 2024. Transformative advances in veterinary laboratory practices: evaluating the impact of preliminary training in Khyber Pakhtunkhwa and Balochistan provinces of Pakistan. J. Biosaf. Biosecurity 6(4), 258–264. Khasanah, H., Setyawan, H.B., Yulianto, R. and Widianingrum, D.C. 2021. Subclinical mastitis: prevalence and risk factors in dairy cows in East Java, Indonesia. Vet. World. 14(8), 2102–2108. Khasapane, N.G., Byaruhanga, C., Thekisoe, O., Nkhebenyane, S.J. and Khumalo, Z.T.H. 2023. Prevalence of subclinical mastitis, its associated bacterial isolates and risk factors among cattle in Africa: a systematic review and meta-analysis. BMC. Vet. Res. 19(1), 123. Kumar, R., Thakur, A. and Sharma, A. 2023. Comparative prevalence assessment of subclinical mastitis in two crossbred dairy cow herds using the California mastitis test. J. Dairy Vet. Anim. Res. 12(2), 98–102. Kustiningsih, H., Sudarnika, E., Basri, C. and Sudarwanto, M. 2023. Dairy farmers’ knowledge, attitudes, and practices regarding the brucellosis surveillance and control program in Bogor, Indonesia. Vet. World 16(1), 126–133. Lestari, V.S., Rahardja, D.P. and Sirajuddin, S.N. Barriers to Adopt Biosecurity at Smallholder Farmers. In IOP Conf. Ser. Earth Environ. Sci. 2022 1012(1), p 12020. Lind, N., Hansson, H., Emanuelson, U. and Lagerkvist, C.J. 2023. Explaining farmers’ adoption of mastitis control practices using psychological constructs from the theory of planned behavior. Livest. Sci. 277(1), 105341. Loeillot, T., Gisclard, M., Trabucco, B., Charrier, F., Jori, F., Antoine-Moussiaux, N. and Delabouglise, A. 2025. Assessing the adoption of biosecurity measures among extensive livestock producers: a case study in the free-range pig sector of Corsica. BMC. Vet. Res. 21(1), 69. Moawad, A.A., Hotzel, H., Monecke, S., Tomaso, H., Neubauer, H., Ehricht, R. and El-Adawy, H. 2023. Whole genome sequence-based analysis of Staphylococcus aureus isolates from clinical bovine mastitis in Thuringia, Germany. Front. Microbiol. 14(1), 1216850. Moje, N., Waktole, H., Kassahun, R., Megersa, B., Chomen, M.T., Leta, S., Debela, M. and Amenu, K. 2023. Status of animal health biosecurity measures of dairy farms in urban and peri-urban areas of central Ethiopia. Front. Vet. Sci. 10(1), 1086702. National Mastitis Council. 2017. Laboratory handbook on bovine mastitis 3rd ed. National Mastitis Council, Inc. Neculai-Valeanu, A., -S.., Apetroaei, M., -R.., Drobota, M. and Creanga. 2024. From herd health to public health: digital tools for combating antibiotic resistance in dairy farms. Antibiotics. (Basel). 13(7), 634. Nuraini, D.M.N., Andityas, M., Sukon, P. and Phuektes, P. 2023. Prevalence of mastitis in dairy animals in Indonesia: a systematic review and meta-analysis. Vet. World. 16(7), 1380–1389. Nyokabi, N.S., Berg, S., Mihret, A., Almaw, G., Worku, G.G., Lindahl, J.F., Wood, J.L.N. and Moore, H.L. 2023. Adoption of biosecurity practices in smallholder dairy farms in Ethiopia. Transbound. Emerg. Dis. 1(1), 2277409. Okello, E., Elashmawy, W.R., Williams, D.R., Lehenbauer, T.W. and Aly, S.S. 2023. Effect of dry cow therapy on antimicrobial resistance of mastitis pathogens post-calving. Front. Vet. Sci. 10(1), 1132810. Otieno, W.A., Nyikal, R.A., Mbogoh, S.G. and Rao, E.J.O. 2023. Adoption of farm biosecurity practices among smallholder poultry farmers in Kenya—An application of latent class analysis with a multinomial logistic regression. Prev. Vet. Med. 217(1), 105967. Park, S. and Ronholm, J. 2021. Staphylococcus aureus in agriculture: lessons in evolution from a multispecies pathogen. Clin. Microbiol. Rev. 34(2), 182. Pedersen, L., Houe, H., Rattenborg, E. and Nielsen, L.R. 2023. Semi-quantitative biosecurity assessment framework targeting prevention of the introduction and establishment of Salmonella Dublin in dairy cattle herds. Animals 13(16), 2649. Postma, M., Backhans, A., Collineau, L., Loesken, S., Sjölund, M., Belloc, C., Emanuelson, U., Grosse Beilage, E., Stärk, K.D.C. and Dewulf, J. 2016. The biosecurity status and its associations with production and management characteristics in farrow-to-finish pig herds. Animal 10(3), 478–489. Power, G.M., Renaud, D.L., Miltenburg, C., Spence, K.L., Hagen, B.N.M. and Winder, C.B. 2024. Ontario dairy producers' and veterinarians' perspectives: barriers to biosecurity implementation. J. Dairy. Sci. 107(8), 5738–5753. Ranasinghe, R.M.S.B.K., Deshapriya, R.M.C., Abeygunawardana, D.I., Rahularaj, R. and Dematawewa, C.M.B. 2021. Subclinical mastitis in dairy cows in major milk-producing areas of Sri Lanka: prevalence, associated risk factors, and effects on reproduction. J. Dairy Sci. 104(12), 12900–12911. Renault, V., Damiaans, B., Sarrazin, S., Humblet, M.F., Dewulf, J. and Saegerman, C. 2018. Biosecurity practices in Belgian cattle farming: level of implementation, constraints and weaknesses. Transbound. Emerg. Dis. 65(5), 1246–1261. Richardet, M., Solari, H.G., Cabrera, V.E., Vissio, C., Agüero, D., Bartolomé, J.A., Bó, G.A., Bogni, C.I. and Larriestra, A.J. 2023. The economic evaluation of mastitis control strategies in Holstein-Friesian Dairy Herds. Animals 13(10), 1701. Ritter, C., Jansen, J., Roche, S., Kelton, D.F., Adams, C.L., Orsel, K., Erskine, R.J., Benedictus, G., Lam, T.J.G.M. and Barkema, H.W. 2017. Invited review: determinants of farmers' adoption of management-based strategies for infectious disease prevention and control. J. Dairy Sci. 100(5), 3329–3347. Ruegg, P.L. 2017. A 100-year review: mastitis detection, management, and prevention. J. Dairy. Sci. 100(12), 10381–10397. Saleh, N., Allam, T., Omran, A. and Mohamed, A. 2022. A review of the subclinical mastitis in cattle with special reference to the new approaches of its diagnosis and control. J. Curr. Vet. Res. 4(1), 33–46. Silva, A.C., Laven, R. and Benites, N.R. 2021a. Risk factors associated with mastitis in smallholder dairy farms in Southeast Brazil. Animals 11(7), 2089. Silva, S.R., Araujo, J.P., Guedes, C., Silva, F., Almeida, M. and Cerqueira, J.L. 2021b. Precision technologies to address dairy cattle welfare: focus on Lameness, mastitis and body condition. Animals 11(8), 2253. Stanek, P., Żółkiewski, P. and Januś, E. 2024. A review on mastitis in dairy cows research: current status and future perspectives. Agriculture 14(8), 1292. Tarigan, R., Lukman, D.W., Latif, H. and Pisestyani, H. 2025. Hematological and mineral alterations associated with subclinical mastitis in dairy cattle following the foot-and-mouth disease outbreak in West Java, Indonesia. Vet. World 18(9), 2723–2732. Toson, M., Pozza, M.D. and Ceschi, P. 2024. Farmers' biosecurity awareness in small-scale Alpine dairy farms and the crucial role of Veterinarians. Animals.. 14(14), 2032. Zanon, T., Alrhmoun, M. and Gauly, M. 2024. Assessing the impact of biosecurity practices and animal welfare in small-scale mountain dairy farming. Sci. Rep. 14(1), 13294. Zhang, T., Boonyayatra, S. and Niu, G. 2022. Association of mastitis and farm management with contamination of antibiotics in Bulk Tank Milk in Southwest, China. Animals. 12(23), 3392. | ||
| How to Cite this Article |
| Pubmed Style Wahyuwardani S, Prihandani SS, Widiastuti R, Widiyanti PM, Anastasia Y, Martindah E, Maryam R, Noor SM, Damayanti R, Sani Y, Khairullah AR, Rohaeni ES, Priyono P. Prevalence of subclinical mastitis and associated biosecurity practices in peri-urban dairy farms. Open Vet. J.. 2026; 16(3): 1565-1577. doi:10.5455/OVJ.2026.v16.i3.14 Web Style Wahyuwardani S, Prihandani SS, Widiastuti R, Widiyanti PM, Anastasia Y, Martindah E, Maryam R, Noor SM, Damayanti R, Sani Y, Khairullah AR, Rohaeni ES, Priyono P. Prevalence of subclinical mastitis and associated biosecurity practices in peri-urban dairy farms. https://www.openveterinaryjournal.com/?mno=287454 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.14 AMA (American Medical Association) Style Wahyuwardani S, Prihandani SS, Widiastuti R, Widiyanti PM, Anastasia Y, Martindah E, Maryam R, Noor SM, Damayanti R, Sani Y, Khairullah AR, Rohaeni ES, Priyono P. Prevalence of subclinical mastitis and associated biosecurity practices in peri-urban dairy farms. Open Vet. J.. 2026; 16(3): 1565-1577. doi:10.5455/OVJ.2026.v16.i3.14 Vancouver/ICMJE Style Wahyuwardani S, Prihandani SS, Widiastuti R, Widiyanti PM, Anastasia Y, Martindah E, Maryam R, Noor SM, Damayanti R, Sani Y, Khairullah AR, Rohaeni ES, Priyono P. Prevalence of subclinical mastitis and associated biosecurity practices in peri-urban dairy farms. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1565-1577. doi:10.5455/OVJ.2026.v16.i3.14 Harvard Style Wahyuwardani, S., Prihandani, . S. S., Widiastuti, . R., Widiyanti, . P. M., Anastasia, . Y., Martindah, . E., Maryam, . R., Noor, . S. M., Damayanti, . R., Sani, . Y., Khairullah, . A. R., Rohaeni, . E. S. & Priyono, . P. (2026) Prevalence of subclinical mastitis and associated biosecurity practices in peri-urban dairy farms. Open Vet. J., 16 (3), 1565-1577. doi:10.5455/OVJ.2026.v16.i3.14 Turabian Style Wahyuwardani, Sutiastuti, Sri Suryatmiati Prihandani, Raphaella Widiastuti, Prima Mei Widiyanti, Yessi Anastasia, Eny Martindah, Romsyah Maryam, Susan Maphilindawati Noor, Rini Damayanti, Yulvian Sani, Aswin Rafif Khairullah, Eni Siti Rohaeni, and Priyono Priyono. 2026. Prevalence of subclinical mastitis and associated biosecurity practices in peri-urban dairy farms. Open Veterinary Journal, 16 (3), 1565-1577. doi:10.5455/OVJ.2026.v16.i3.14 Chicago Style Wahyuwardani, Sutiastuti, Sri Suryatmiati Prihandani, Raphaella Widiastuti, Prima Mei Widiyanti, Yessi Anastasia, Eny Martindah, Romsyah Maryam, Susan Maphilindawati Noor, Rini Damayanti, Yulvian Sani, Aswin Rafif Khairullah, Eni Siti Rohaeni, and Priyono Priyono. "Prevalence of subclinical mastitis and associated biosecurity practices in peri-urban dairy farms." Open Veterinary Journal 16 (2026), 1565-1577. doi:10.5455/OVJ.2026.v16.i3.14 MLA (The Modern Language Association) Style Wahyuwardani, Sutiastuti, Sri Suryatmiati Prihandani, Raphaella Widiastuti, Prima Mei Widiyanti, Yessi Anastasia, Eny Martindah, Romsyah Maryam, Susan Maphilindawati Noor, Rini Damayanti, Yulvian Sani, Aswin Rafif Khairullah, Eni Siti Rohaeni, and Priyono Priyono. "Prevalence of subclinical mastitis and associated biosecurity practices in peri-urban dairy farms." Open Veterinary Journal 16.3 (2026), 1565-1577. Print. doi:10.5455/OVJ.2026.v16.i3.14 APA (American Psychological Association) Style Wahyuwardani, S., Prihandani, . S. S., Widiastuti, . R., Widiyanti, . P. M., Anastasia, . Y., Martindah, . E., Maryam, . R., Noor, . S. M., Damayanti, . R., Sani, . Y., Khairullah, . A. R., Rohaeni, . E. S. & Priyono, . P. (2026) Prevalence of subclinical mastitis and associated biosecurity practices in peri-urban dairy farms. Open Veterinary Journal, 16 (3), 1565-1577. doi:10.5455/OVJ.2026.v16.i3.14 |