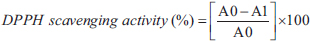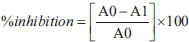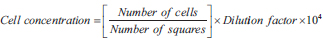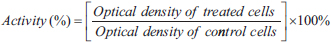Open Veterinary Journal, (2026), Vol. 16(3): -1609
Research Article
10.5455/OVJ.2026.v16.i3.17
It takes two to tango: Combined ethanolic leaf extracts of Punica granatum and Orthosiphon stamineus strongly inhibited HepG-2 and A375 cell line proliferation in vitro
Mahmood Hasan Mahmood1*, Reemy Marwan Mohammed Salih1, Dhuha Waleed Salih1,
Abdulrahman Jihad Mansoor1, Milad Adnan Mezhir2 and Mohammed Hasbi Abdulraheem3
1College of Nursing, Tikrit University, Tikrit, Iraq
2Department of Biology, Faculty of Education for Pure Sciences, Tikrit University, Tikrit, Iraq
3Faculty of Industrial Science and Technology, UMPSA, Pekan, Malaysia
*Corresponding Author: Mahmood Hasan Mahmood. College of Nursing,
Tikrit University, Tikrit, Iraq. Email: Mahmood.h.m90 [at] tu.edu.iq
Submitted: 19/08/2025 Revised: 15/01/2026 Accepted: 02/02/2026 Published: 31/03/2026
© 2026 Open Veterinary Journal
This is an Open Access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited, and is not altered, transformed, or built upon in any way.
Abstract
Background: Five plant extracts (Orthosiphon stamineus, Morus nigra, Averrhoa bilimbi, Punica granatum, and Eurycoma longifolia) were studied in this work for antioxidant activities using ABTS and DPPH; the two most potent extracts in terms of antioxidant activity were further studied for antiproliferative activity against cancer cell lines.
Aim: To study the antiproliferative activity of two potent extracts at different combination ratios on two cancer cell lines (HepG2 and A375) using the MTT assay.
Methods: About 20 g of the dried and pulverized plant materials were extracted with 200 mL of 80 % ethanol in an ultrasonic homogenizer for 15 minutes (using 15-second pulse-on and 5-second pulse-off period). The resulting extract was filtered and concentrated in a rotary evaporator to 20 mL before freeze drying to eliminate alcohol content. The phytochemical tests were done using several chemical methods. Antioxidant activity of the extracts was assessed using ABTS and DPPH methods. Two plant extracts (O. stamineus and P. granatum extracts) with the highest antioxidant activities out of the screened five extracts were used for the phytochemical identification test and MTT assays using HepG2 and A375 cell lines.
Results: The antiproliferative activity study showed that the calculated IC50 values for the individual O. stamineus and P. granatum extracts against the HepG2 cell line were (144.46 µg/mL and 174.16 µg/mL), respectively, and against the A375 cell line, the IC50 values were determined to be (136.19 and 152.77 µg/mL), respectively. For the combined O. stamineus and P. granatum extracts against the HepG2 cell line, the calculated IC50 values were (138.4 ± 0.86 µg/mL, 124.52 ± 1.62 µg/mL, and 157.88 ± 1.31 µg/mL) against HepG2 at the combination ratios of 1:1, 2:1, and 1:2, respectively, and (106.33 ± 1.14 µg/mL, 99.62 ± 0.64 µg/mL, and 118.32 ± 1.06 µg/mL) for the A375 cell line at the combination ratios of 1:1, 2:1 and 1:2, respectively.
Conclusion: This study showed that it actually takes two to tango since the antiproliferative activities of the combined extracts against the studied cell lines were stronger than their individual activities. Therefore, it may be suggested that the phytochemicals responsible for the observed activity in this study can be isolated and characterized for improved exploitation.
Keywords: Averrhoa bilimbi, Eurycoma longifolia, Morus nigra, Orthosiphon stamineus, Punica granatum.
Introduction
Cancer is the unregulated proliferation of body cells due to defects in regulatory genes. According to Sudhakar (2009) and Fouad and Aanei (2017), genotypic mutations cause transformations in normal cells into cancer cells, leading to unregulated growth, re-localization, and spread. Uncontrolled and excessive cell division results in cancerous tissue growth, which spreads to other areas and eventually becomes metastases. One of the most important public health issues of the 21st century is cancer prevention, as the global cancer burden increases. According to reports from World Cancer Research Fund International, there were 18.1 million cancer diagnoses worldwide at 2020, with 8.8 million cases women affecting and 9.3 million in men. Among all cancer types, liver cancer is 6th most prevalent globally, with a reported global incidence of 905,677 in 2020 (World Cancer Research Fund International 2020). Skin melanoma is the 17th most prevalent cancer globally, with a reported 324,635 new cases in 2020 (WCRFI, 2020).
The World Health Organization is pushing for innovative alternative cancer management techniques because of the growing incidence of cancer cases worldwide and the unfavorable impacts of the current cancer management strategies (chemotherapy and radiation). Chemotherapy and irradiation are the most common forms of cancer treatment, but they have been proven to pose several adverse effects on body of human, like bleeding, infection, fatigue, and hair loss. (Saxena et al., 2012; Kadori et al., 2026). The WHO, sourcing alternatives from natural-based products, is expedient.
Nevertheless, there are significant obstacles to overcome before using natural products as substitute cancer treatments; these obstacles include poor bioactivity (large doses are needed to produce beneficial effects), few sources (the available sources are still scarce), and increasing demand (natural product demand is currently greater than supply). These restrictions might be overcome by either finding new sources of naturally occurring extracts that are biologically active to supplement existing ones or by figuring out how to increase the bioactivity of existing ones, such as combining them rather than using them alone.
This study, antiproliferative activity extracts of five different plants, namely Morus nigra, Eurycoma longifolia, Averrhoa bilimbi, Orthosiphon stamineus, and Punica granatum, against two cancer cell lines (A375 cell lines, that is, malignant skin melanoma cell lines, and HepG-2 cell lines, that is, hepatocellular carcinoma cell lines) was studied using different experimental designs. The study involved extracts from the leaves of M. nigra, O. stamineus, and A. bilimbi, the peel of P. granatum, and the root of E. longifolia.
Studies have shown that O. stamineus extract exhibits antipyretic, diuretic, hypouricemic, antioxidant, and hepatoprotective (Nisa et al., 2020; Jumaa et al., 2024); hence, it has been used to manage kidney problems, diabetes, fever, gout, and stomach pain (Nisa et al., 2020). A. bilimbi extract exhibits antibacterial, antidiabetic, wound healing, hypolipidemic, antihypertensive, antithrombotic, and antioxidant properties (Alhassan and Ahmed, 2016; Essa et al., 2025) and, as such, has been used to manage a range of infectious and non-infectious illnesses in the traditional setting (Ali et al., 2013; Alhassan and Ahmed, 2016). A. bilimbi extracts have been used to treat diabetes, rectum irritation, and fever, as well as rheumatism, syphilis, mumps, itching, and skin eruptions. Morus nigra is a deciduous plant (Lim and Choi, 2019), and its fruits are rich in phenolic compounds and phytochemicals. Anthocyanins, which are abundant in M. nigra peels, have been thought to prevent liver cancer cells from proliferating within the human body (Lim and Choi, 2019).
Punica granatum is an edible fruit that grows naturally throughout the Mediterranean (Fernández-López and Pérez-Álvarez, 2010). Different parts of P. granatum contain various bioactive chemicals, including the fruits, which have an inedible peel that is rich by bioactive substances such as anthocyanins, tannins, polyphenols, and flavonoids. Most of the anthocyanin content of this plant is found in the fruit peels.
Eurycoma longifolia, also known as Tongkat Ali (Bhat and Karim, 2010), is a medicinal plant in Simaroubaceae family and is presence in several Southeast Asian nations. It is traditionally used in herbal medicine to treat cough, indigestion, hypertension, fever, ulcers, and high blood pressure. Its bark has been used as a vermifuge, and its fruits can cure diarrhea or dysentery (Rehman and Yoo, 2016). The roots are the most valuable part of E. longifolia because of their abundance of bioactive chemicals. They are frequently used to treat constipation, glandular swelling, sexual inadequacy, dysentery, fever, and malaria (Bhat and Karim, 2010; Rehman et al., 2016).
The main aim of the present study is to determine of individual antiproliferative activity of these plant extracts, as well as their combined activity against the cell lines under study, in a laboratory-scale study.
Materials and Methods
Experiments
Sample preparation, extraction, and phytochemical screening
The plant materials were collected from accredited vendors in Kuantan, Malaysia, and approximately 20 g of the dried and pulverized plant materials was extracted with 200 ml of 80 % ethanol in an ultrasonic bath to 15 minutes (using a 15-second pulse-on and 5-second pulse-off period). The resulting extract was filtered and concentrated to 20 ml in evaporator rotary (Mahmood et al., 2019). The resulting concentrate was freeze-dried to remove the residual ethanol and prepare the extracts for analysis.
The phytochemical tests were performed using several chemical methods; for instance.
Test for terpenoids
Terpenoids react with concentrated sulfuric acid, producing red, brown, or golden yellow coloration due to dehydration and conjugated double bond formation. We placed in test tube 2 ml from extract and mixed with 2 ml of chloroform, then by 2 ml of concentrated sulfuric acid down side of test tube to form a layer. Color change at interface was observed, where a reddish-brown interface indicates presence of terpenoids and a golden yellow/brown interface indicates the presence of triterpenoids (Neelima et al., 2011; Khanam et al., 2015).
Test for phenolic compounds/tannins
Extracts (1 mg/ml) dissolve in water, and then add of FeCl3+, when appear green color signifies to presence compounds of phenolic, or blue-black/green-black colour indicates tannins. The assay was performed in triplicate, and reported results the positive (+) for presence of phenolic compounds/tannins or negative (-ـ) for their absence (Rufai, 2016).
Test for Saponin
Dissolve of extracts (15 mg) in water (15 ml) and shaken. The foundof saponin was indicating by stable foam. The assay was performed in triplicate, and positive (+) or negative (-ـ) results indicate the presence or absence of saponins (Rufai, 2016).
Test for alkaloids
Mayer's reagent (prepared by dissolving 1.36 g of mercuric chloride and 5 g of potassium iodide in 100 ml of distilled water) was added in a few drops to the aqueous extracts(1 mg/ml). The development of a milky precipitate signified that alkaloid was present. All assays were performed in triplicate and were interpreted qualitatively of the positive (+) or negative (−) for alkaloid detection (Khanam et al., 2015).
Test for flavonoids
The dissolve of extract (1 mg/ml) by methanol, and then addition four magnesium ribbons. Concentrated hydrochloric acid was later adding in drops into samples and observes to reddish coloration, indicating found of flavonoids. The assay was performed in triplicate, and the results were reported as positive (+) for a reddish coloration or negative (-) for the absence of a reddish coloration (Mir et al., 2016).
The extract concentration was determined by determining the total solids in the extract (mass of residue after solvent removal) and expressed as mg/ml the tests were conducted in triplicate.
Antioxidant activities
ABTS-based antioxidant activity assay was performed using methodology of Re et al. (1999), whereas the DPPH assay was performed using method described by (Salwa et al., 2020). Multiple biochemical assays can be used to evaluate antioxidants; however, the combined use of ABTS [2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)] and DPPH [2,2-diphenyl-1-picrylhydrazyl] radical scavenging methods is widely accepted as sufficient for primary antioxidant screening. Their simultaneous use provides reliable, reproducible, and comprehensive information regarding the radical-scavenging capacity of plant extracts, natural products, biological fluids, and purified compounds. Furthermore, antioxidants act mainly via transfers single-electron and hydrogen atom. DPPH predominantly measures single-electron transfer, while ABTS measures both transfers single-electron and hydrogen atom. Therefore, the use of both assays provides a balanced evaluation of antioxidants that work through different pathways.
DPPH radical scavenging
The determination of DPPH free radical scavenging activity of the extracts was performed using the method reported by (Noreen et al., 2017). DPPH solution (126 µM) was prepared in methanol absolute. The extracts were re-dissolved in distilled water to achieve five different concentrations (0.2, 0.4, 0.6, 0.8, and 1.0 mg/ml). One ml aliquots of each concentration were added to 2 ml of the DPPH solution in glass tubes, vortex mixed for 30 seconds and allowed to stand in the dark at ambient temperature for 30 minutes. A control sample was prepared in the same way by replacing the extract with 1 ml deionized water. Absorbance of DPPH solution was determined at wavelength of 517 nm by spectrophotometer. In the same concentration range, a reference antioxidant (ascorbic acid) was prepared. Measurements were performed in triplicate and expressed as mean ± standard deviation (SD). The equation used to calculate the radical scavenging activity (RS) was as follows: 3.2 and were expressed as percentage inhibition of DPPH radicals and IC50 values (the concentration needed to inhibit 50% of this population of DPPH radicals).
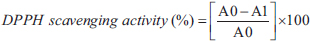
Where A0 is the absorbance of the control mixture (2 ml of DPPH solution and 1 ml of deionized water), and A1 is the absorbance of reaction mixtures (2 ml of DPPH solution and 1 ml of diluted extracts or standard solutions at different concentrations).
ABTS radical scavenging
ABTS+ solution was prepared and adjusted to a suitable absorbance of ~0.7 at 734 nm. Then, 1 ml of the ABTS solution and 1 mg/ml of the extracts were mixed and incubated in the dark for 30 minutes at room temperature. The absorbance was measured at 734 nm using a spectrophotometer, and the percentage inhibition was calculated as follows:
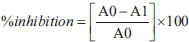
Where A0 is the absorbance of the control mixture containing 1 ml of ABTS solution and 1 ml of deionized water; A1 is the absorbance of the reaction mixtures containing 1 ml of ABTS solution and 1 ml of the diluted extracts or standard solutions with varying concentrations.
On the basis of individual screening results, the antioxidant activity of two strongest extracts was investigated using mixtures prepared at two different ratios, and the capacity of antioxidant in the mixtures was determined, as described above.
Tentative identification of phytochemicals using gas chromatography–mass spectrometry analysis
A GC-MS system (7890A, Agilent Technologies) was used to tentatively identify the phytochemicals in the plant extract. The system was fitted with an Agilent 19091J-433: 4294.60089 equipped with a capillary HP-5 5% phenyl methyl siloxane non-polar column (30 m length, 0.25 µm film thickness, and 250 µm inner diameters). Helium gas was used as transporter gas in 1 ml/minute flow rate. The oven was initially set to operate at 50°C for 0 minute, then 10°C every minute to reach 300°C, where it was maintained isothermally for 25 minutes. For the analysis, 1 µl of the material was injected. Component analysis was performed using the GC/MSD Chem Station software, and mass spectra and relative retention times were generated in comparison to the NIST 11 library.
Antiproliferative assay of plant extracts
Cell lines studied
A375-CRL-1619 and HepG2-HB-8065
Cryopreserved human malignant melanoma cell lines (A375-CRL-1619) and human hepatocellular carcinoma cell lines (HepG2-HB-8065) were sourced from the American Type Culture Collection and revived before being used in the study. A375 was established in 1973 by Giard et al. (1973) as a cell line that contains a BRAF V600E mutation, a hallmark driver mutation in melanoma. This mutation causes constitutive activation of the MAPK/ERK pathway, resulting in increased cell proliferation. The line is commonly used to study melanoma biology, metastasis, apoptosis pathways, and targeted therapies (e.g., BRAF inhibitors such as vemurafenib).
HepG2 was established in 1975 at the Children’s Hospital of the University of Alabama as a highly differentiated hepatocyte cell line that retains many normal liver functions, such as albumin synthesis, clotting factor synthesis, and active cytochrome P450 enzymes.
Cell line revival
Cells were revived in 10% fetal bovine serum-supplement Dulbecco’s Modified Eagle’s Medium (DMEM). After media preparation, a cryovial of each cell type was thawed in water bath in 37°C and transfer into 25 cm2 cell culture flasks contain 4.5 ml of the prepared DMEM. The flasks were incubating at 37oC with CO2 5% in 37°C to 24 hours cells proliferation was observed every 24 hours under an inverted microscope. The successfully revived cells were allowed to confluence to approximately 80% by allowing them to grow further in the supplemented media. When confluent, the cells were subcultured on fresh media to remove dead cells and metabolic wastes, which could hamper the normal cellular metabolic processes.
Cell sub-culturing
The cells were sub-cultured on fresh DMEM after a series of washing and detachment processes. The culture media in the flasks were discarded by pipetting, after which the cells were washed three times using phosphate-buffered saline (PBS) to clear dead cells. The attached cells were dislodged using 1 ml of TrypLE, followed by incubated in 37oC with CO2 5% for 5 minutes. The cells were harvested by adding 5 ml of DMEM to the flask, followed by collection into 15 ml Falcon tubes and subsequent for 5 minutes at 4oC. The media were discarded after centrifugation, and the cells were resuspended in DMEM for culturing. Cells (0.5 ml) were cultured in 14.5 ml of DMEM supplemented with 10% FBS and 1% P-S in new sets of 75 cm2 culture flasks. The culture flasks were incubated in 37oC with CO2 5% for 24 hours, with intermittent observation of cell proliferation every 24 hours. The cells were allowed to confluence at least 80% before being used in MTT-based antiproliferative studies.
Cell dilution and seeding for the MTT assay
After the cells had confluence to approximately 80%, the cells were harvested as previously described and resuspended in the working media. The number of viable cells in the suspension was counted using an improved Neubauer counting chamber before seeding the cells. Cells were diluted with 0.4% Trypan blue (1 in 5) and counted in the chamber. The cells were counted in the 4 large corner squares of the chamber, and the number of viable cells in the suspension was estimated using the following equation:
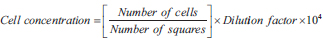
The cell concentration was diluted to 5 × 103 cells/ml in the medium and cultivates into plates of 96-well, and incubate at 37°C to 24 hours with CO2 5%. After incubation period, aspirated the media in the wells, and washed the cells twice with before PBS proceeding to cytotoxicity studies.
MTT cytotoxicity assay
For the MTT assay, a solution of individual plant extract and their combination was prepared in complete growth media at concentrations of 31.25, 62.5, 125, 250, 500, and 1,000 µg/ml. The cells were allowed to reach 80% confluence before the tests. Plates were incubated at 37oC with 5% CO2 for 24 hours. After the incubation period, 10 µl of MTT reagent was added directly to the wells and gently mixed. The plates were wrapped in aluminum foil and incubated for 4 hours at 37oC with CO2 5%. After incubated period, the MTT-containing media were carefully aspirated and replaced with 110 µl of dimethylsulfoxide to dissolve the formazan crystals formed by the viable cells’ mitochondrial dehydrogenase. After this stage, the plates were shaken and incubated for 10 minutes before reading the absorbance of the dissolved formazan dye at 490 nm using a microplate reader. The antiproliferative activity of the extracts was expressed as a percentage of the cell viability against the control as follows:
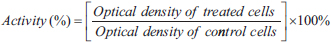
The concentration of the agents needed to achieve 50% cell death was determined from the concentration-activity plot for each extract.
Statistical analysis
Each test was run 3 times, and reported results as mean ± S.E. of the mean. Graph Pad Prism 7 software was used to calculate the IC50 values. Microsoft Excel 2017 was used for the analysis of inhibitory zone measurements. The t-test was used to compute p-values, and values < 0.050 were considered significant (Chuah et al., 2014).
Ethical approval
Not needed for this study. However, this study was prepared based on ethical instructions of the College of Nursing, Tikrit University, Iraq.
Results
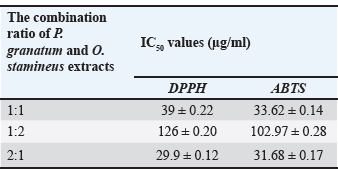
The results of phytochemical tests on the extracts are shown in Table 1. Eurycoma longifolia root extract tested positive for alkaloids, terpenoids, triterpenoids, and flavonoids. Additionally, the presence of all the evaluated phytochemical groups in O. stamineus and P. granatum extracts, the phytochemical contents of both plant materials were significantly higher than those of the rest of the plants, and the adopted extraction method (UAE method using 80% ethanol as solvent) was efficient in achieving the desired aim. The results of antioxidant activity on scavenge ABTS and DPPH radicals, The capacity of five plant extracts to scavenge ABTS and DPPH radicals was contrasted with that of ascorbic acid, a common antioxidant. The IC50 values were calculated for all extracts and are presented in Table 2. The O. stamineus and P. granatum leaf extracts had the lowest IC50 values, measuring 47.54 and 189.4 µg/ml for the DPPH test and 35.92 and 275.5 µg/ml for the ABTS test, respectively. However, the antioxidant activity of the combined extracts at different ratios showed significant differences; in comparison to O. stamineus and P. granatum, with mixing ratios of 1:2 and 1:1, P. granatum and O. stamineus at a 2:1 mixing ratio had higher antioxidant activity for both ABTS and DPPH assays (Table 3). This could be an indication of a higher concentration of phytochemicals with stronger antioxidant activity in the leaf extracts of P. granatum compared to O. stamineus extract (the higher the quantities of P. granatum extract in the combination, the stronger the antioxidant activity of the combined extract). This is the first attempt at determining the antioxidant activity of combined leaf extracts of O. stamineus and P. granatum at varying mixing ratios.
Table 1. Results of phytochemical tests on the plant extracts studied.
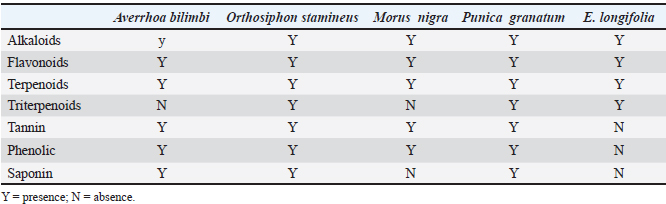
Table 2. IC50 values of plant extracts using the DPPH and ABTS assays.
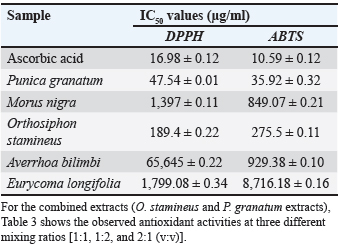
Table 3. Antioxidant activity of the combined extracts at varying ratios.
The results of GC- MS-based tentative identification of phytochemicals, in recognition of the strong antioxidant activities of the O. stamineus and P. granatum extracts were analyzed for phytochemical content using the GC-MS method. GC-MS analysis revealed 65 phytochemicals in ethanolic extracts of O. stamineus. Approximately 19 of the 65 compounds detected in the extract have antioxidant and antimicrobial properties, including octadecanoic acid (22.58%), oleic acid (0.08%), bis(2-ethylhexyl) phthalate (1.4%), eicosanoic acid (16.41%), 9,12,15-octadecatrien-1-ol, (Z, Z, Z)- (10.24%), 9,17-octadecadienal, (Z)- (13.65%), 4H-pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- (0.71%), and n-hexadecanoic acid (10.02%). In the P. granatum extract, 36 volatile compounds were identified, of which 9 compounds [oleic acid (33.48%), tetradecanoic acid (2.08%), n-hexadecanoic acid (16.321%), geranyl vinyl ether (1.00%), octadecanoic acid (4.74%), 11-trimethyl-, (Z, E)- (0.31%), 2,6,10-dodecatrien-1-ol, 3,7, 2-hexyn-1-ol (0.54%), octadec-9-enoic acid (0.67%), and bis(2-ethylhexyl) phthalate (1.30%)] have been found to exhibit antimicrobial and antioxidant properties.
The result of antiproliferative activity of the combined plant leaf extract, the different combinations of O. stamineus and P. granatum leaf extracts were tested for antiproliferative activity on liver cancer (Hep-G2) and skin melanoma (A375) cell lines use MTT assay (Figs. 1 and 2, respectively). The figures show evidence of the downregulation of proliferation of study cells lines by the combined extracts at different concentrations. The reduction in cell number was measured by measuring the absorbance of the oxidized form of the MTT dye. The calculated IC50 values for the antiproliferative activity of the combined extracts of P. granatum and O. stamineus against HepG2 cell line were (138.4 ± 0.86, 124.52 ± 1.62, and 157.88 ± 1.31 µg/ml, respectively, at the mix ratios of 1:1, 2:1, and 1:2. For the A375 cell line, the calculated IC50 values for the antiproliferative activity of the combined O. stamineus and P. granatum extracts were (106.33 ± 1.14, 99.62 ± 0.64, and 118.32 ± 1.06 µg/ml, respectively, at the mix ratios of 1:1, 2:1, and 1:2. For the individual plant extracts, the calculated IC50 values of the antiproliferative activities of P. granatum and O. stamineus against HepG2 cell line were (144.46 µg/ml and 174.16 µg/ml, respectively, and 136.19 and 152.77 µg/ml, respectively, against the A375 cell lines (complete data not provided).
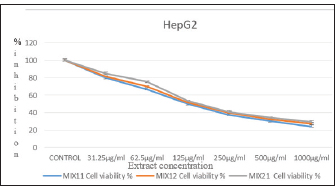
Fig. 1. Antiproliferative activity of the combined extracts against the Hep-G2 cell line after 24 hours incubation at 37oC and 5% CO2.
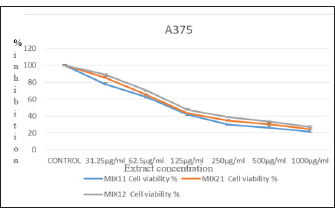
Fig. 2. Antiproliferative activity of the combined extracts against the A375 cell line after 24 hours incubation at 37oC and 5% CO2.
Evidently, the combined extracts achieved lower IC50 values against the cell lines than the individual extracts. This could be due to the synergistic interaction between the different phytochemicals found in the individual plants, which improved the response of the cell lines to the combined extracts. Furthermore, the quantity of P. granatum leaf extract in the combination seems to be the determinant of the level of antiproliferative activity of the combination; for instance, the antiproliferative activity of the combined extract was higher for both cell lines (124.52 ± 1.62 µg/ml for HepG2 and 99.62 ± 0.64 µg/ml for A375 cell line) at the O. stamineus and P. granatum combination ratio of 2:1 compared to the ratios of 1:1 (138.4 ± 0.86 µg/ml for HepG2 cell line and 106.33 ± 1.14µg/ml for A375 cell line) and 1:2 (157.88 ± 1.31 µg/ml for HepG2 cell line and 118.32 ± 1.06 µg/ml for A375 cell line).
Discussion
The results of phytochemical tests on the extracts are shown in Table 1. were similarity with previously reported by (Mahmood et al., 2019). The leaf extract of Morus nigra tested positive for tannins, terpenoids, flavonoids, alkaloids, and phenolics, as reported by (Aulifa et al., 2017). Alkaloids, flavonoids, saponin, phenolics, and tannins were detected in Averrhoa bilimbi leaf extract (Aziz et al., 2014), whereas saponin, tannins, phenolics, flavonoids, terpenoids, and alkaloids were detected in peel extracts of P. granatum, as reported by (Sajjad et al., 2015; Priyanka et al., 2019). The presence of triterpenoids was also detected in the P. granatum extracts, as reported by (Priyanka et al., 2019). As reported by Malahubban et al. (2013) O. stamineus leaf extract tested positive for flavonoids, terpenoids, tannins, saponins, and alkaloids. The higher capability of O. stamineus and P. granatum extracts to scavenge the radicals in both assays is indicative of the presence of higher concentrations of phenolic compounds in these plant materials (Becker et al., 2019; Nasr et al., 2023a,b). The combined extracts recorded better antioxidant activity than their individual counterparts, likely due to the synergy in the activity of the phytochemicals contained in the respective extracts (Elbakry et al., 2023a,b). GC-MS analysis revealed 65 phytochemicals in the ethanolic extract of O. stamineus. Approximately 19 of the 65 compounds detected in the extract have antioxidant and antimicrobial properties. This result of present study agreement with results (Cushnie et al., 2014) who reported the natural, semisynthetic and synthetic alkaloids of all classes are considered, looking first at those with direct antibacterial activity and those with antibiotic-enhancing activity. Also, in the P. granatum extract, 36 volatile compounds were identified, of which 9 compounds have been found to exhibit antimicrobial and antioxidant properties, this result agreement with (Shah et al., 2018; Muruthi, 2023) they reported the extracts showed effect on bacteria growth inhibition in concentrations dependent manner. All extracts were significantly inhibition on bacterial species of gram positive and lower inhibition on bacterial species of gram negative. The result of antiproliferative activity of the combined plant leaf extract O. stamineus and P. granatum activity on liver cancer (Hep-G2) and skin melanoma (A375) cell lines use the MTT assay, the figures show evidence of the downregulation of proliferation of study cell lines by the combined extracts at different concentrations. This result was similarity with (Nasr et al., 2023a,b; Sameh et al., 2025) they reported both pomegranate seed and peel extracts showed cytotoxic effect on liver cancer cell line. Cell proliferation inhibitory concentration was much effective in case of seed extract tan in peel extract. According to Patel and Goyal (2012) antiproliferative agents promote reactive oxygen species, angiogenesis, mitotic kinase, topoisomerase, and apoptosis. The groups of hydroxyl in phytochemical engage with mitochondrial cytochrome P450 enzyme’s polar receptor region to cause metabolic disturbance in cells, which is a straightforward mechanism for the extracts’ reported antiproliferative effects (Zerrouki et al., 2022). The pathway of intrinsic apoptotic, characterized by high permeability of the cell membrane and swelling of the mitochondria, rupture of the outer mitochondrial membrane, and release of pro-apoptotic factors from the intermembrane space, may result from the attachment of these OH groups to the P450 enzyme (Forbes-Hernández et al., 2014; Elbakry et al., 2023a,b). Hence, it may be logical to conclude that ‘it actually takes two to tango” with respect to the observed higher antiproliferative potentials of the combined extracts against the studied cell lines compared with those of the individual extracts.
Conclusion
The biological activity of natural products can be considerably improved using different approaches, but the combination of two or more natural extracts/products could be the most feasible and biologically accepted method to maximize their potential benefits. This study demonstrated a significantly higher antiproliferative activity of two plant extracts (ethanolic leaf extracts of O. stamineus and P. granatum at different combination ratios) against skin melanoma (A375) and hepatic cancer (Hep-G2) cell lines. The results showed that the combined extracts achieved lower IC50 values against the cell lines than the individual extracts, possibly due to the synergistic interaction between the different phytochemicals found in the individual plants, which improved the response of the cell lines to the combined extracts. Therefore, future studies might focus on the isolation and characterization of the phytochemicals responsible for the observed activity in these extracts for more pharmacological studies.
Acknowledgment
This study was supported by the College of Nursing, Tikrit University. Iraq.
Conflict of interest
The authors have no conflicts of interest to disclose.
Funding
No grant was received for this study, and it has been self-funded.
Authors’ contributions
Reemy M. M. was in charge of the five plant extracts, while Mahmood H. M. served as the article’s corresponding author. A. M. Abdulraham, A. M. Milad, and H. A. Mohd were in charge of the research management and study observations. Each author contributed equally to the writing of the article.
Data availability
All data are provided in the revised manuscript.
References
Alhassan, A.M. and Ahmed, Q.U. 2016. Averrhoa bilimbi Linn: a review of its Ethnomedicinal Uses, Phytochemistry and Pharmacology. J. Pharm. Bioal. Sci. 8(4), 265.
Ali, M.R., Hossain, M., Runa, J.F. and Hasanuzzaman, M. 2013. Preliminary Cytotoxic Activity of Different Extracts of Averrhoa bilimbi (Fruits). Internat. Curr. Pharm. J. 2(3), 83–84.
Aulifa, D.L., Fitriansyah, S.N., Ardiansyah, S.A., Wibowo, D.P., Julata, Y.A. and Christy, D.S. 2017. Phytochemical Screening, Antibacterial Activity, and Mode of Action on Morus nigra. Pharm. J. 10, 167–171.
Aziz, M., Rahman, S., Islam, M. and Begum, A. 2014. A comparative study on antibacterial activities and cytotoxic properties of various leaves extracts of Averrhoa bilimbi. Internat. J. Pharm. Sci. Res. 5, 913–918.
Becker, M.M., Nunes, G.S., Ribeiro, D.B., Silva, F.E., Catanante, G. and Marty, J.L. 2019. Determination of the antioxidant capacity of red fruits by miniaturized spectrophotometry assays. J. Braz. Chem. Soc. 30, 1108–1114.
Bhat, R. and Karim, A.A. 2010. Tongkat Ali (Eurycoma longifolia Jack): a review on its Ethnobotany and Pharmacological Importance. Fitoterapia 81(7), 669–679.
BMC Complementary Medicine and Therapies, 23, 118
Chuah, E., Zakaria, Z.A., Suhaili, Z., Abu Bakar, S. and Mohd Desa, M.N. 2014. Antimicrobial Activities of Plant Extracts against Methicillin-Susceptible and Methicillin-Resistant Staphylococcus Aureus. J. Microbiol. Res. 4(6), 6–10.
Cushnie, T.P.T., Cushnie, B. and Lamb, A.J. 2014. Alkaloids: an overview of their antibacterial, antibiotic-enhancing and antivirulence activities. Int. J. Antimicrob. Agents. 44(5), 377–386.
Elbakry, A.A., Nasr, F.A., Hassan, R.A. and Mohamed, H.S. 2023. Phenolic profiling and antioxidant potential of medicinal plant extracts using DPPH and ABTS assays. J. Herbal Med. 39, 100662.
Elbakry, M.M.M., Elbakary, N.M., Hagag, S.A. and Hemida, E.H.A. 2023. Pomegranate Peel Extract Sensitizes Hepatocellular Carcinoma Cells to Ionizing Radiation, Induces Apoptosis and Inhibits MAPK, JAK/STAT3, β-Catenin/NOTCH, and SOCS3 Signaling. Integr. Cancer Therapies 22, 15347354221151021.
Essa, M., Kadori, A., Suleiman, J.M., Jumma, Q. and Aziz, A. 2025. Evaluation of the quality of fish storage, isolation and identification of the bacteria Escherichia coli and Aeromonas hydrophila. Egypt. J. Aquatic Biol. Fisheries 29(5), 455–468.
Forbes-Hernández, T.Y., Giampieri, F., Gasparrini, M., Mazzoni, L., Quiles, J.L., Alvarez-Suarez, J.M. and Battino, M. 2014. The effects of bioactive compounds from plant foods on mitochondrial function: a focus on apoptotic mechanisms. Food. Chem. Toxicol. 68, 154–182.
Fouad, Y.A. and Aanei, C. 2017. Revisiting the Hallmarks of Cancer. Am. J. Cancer Res. 7(5), 1016–1036.
Giard, D.J., Aaronson, S.A., Todaro, G.J., Arnstein, P., Kersey, J.H., Dosik, H. and Parks, W.P. 1973. In vitro cultivation of human tumors: establishment of cell lines derived From a Series of Solid Tumors. Natl. Cancer Inst. 51(5), 1417–1423.
Jumaa, Q.S., Al-Bayati, H.H.K., Abdullah, S.A., Shihab, T.J. and Sultan, M. 2024. Immunogenicity of culture filtrated proteins and whole-cell killed formalin of Listeria monocytogenes to induced cellular immune response in vivo. Open Vet. J. 14(12), 3581–3598.
Kadori, A., Dakheel, M., Ahmed, S., Abd, A. and Jumma, Q. 2026. Detection and characterization of Salmonella species in fresh fish meat in Tikrit markets. Egypt. J. Aquatic. Biol. Fisheries. 30(1), 855–864.
Khanam, Z., Wen, C.S. and Bhat, I.H. 2015. Phytochemical Screening and Antimicrobial Activity of Root and Stem Extracts of Wild Eurycoma longifolia Jack (Tongkat Ali). J. King Saud Univ. Sci. 27(1), 23–30.
Lim, S.H. and Choi, C.I. 2019. Pharmacological properties of Morus nigra L. (Black Mulberry) as a promising nutraceutical resource. Nutrients, 11(2), 437.
Mahmood, M.H., Osama, A.K., Makky, E.A., Rahim, M.H., Ali, N.H.M. and Hazrudin, N.D. 2019. Phytochemical Screening, Antimicrobial and Antioxidant Efficacy of Some Plant Extracts and Their Mixtures. In IOP Conference Series: Earth Environ. Sci. 346, 012003.
Malahubban, M., Alimon, A.R., Sazili, A.Q., Fakurazi, S. and Zakry, F.A. 2013. Phytochemical Analysis of Andrographis Paniculata and Orthosiphon stamineus Leaf extracts for their antibacterial and antioxidant potential. Trop. Biomed. 30(3), 467–480.
Mir, M.A., Parihar, K., Tabasum, U. and Kumari, E. 2016. Estimation of Alkaloid, Saponin and Flavonoid, Content in Various Extracts of Crocus Sativa. J. Med. Plants. Stud. 4(5), 171–174.
Muruthi, C.W. 2023. In vitro antiproliferative effects and phytochemical analysis of selected plant extracts against cancer cell lines. J. Dietary. Supplements. 20(1), 123–139.
Nasr, F.A., El-Bakry, A.A., Hassan, R.A., and Mohamed, H.S. (2023). In vitro antioxidant and cytotoxic activities of selected medicinal plant extracts. BMC Complement. Med. Ther. 23, 118.
Nasr, M., Naeem, S.A., El-Shenbaby, I., Mohamed, F.M.A., Mahmoud, S.M., Abuamara, T.M.M., Abd-Elhay, W.M., Elbayoumy, F.M.A.E., Elkot, A., Shikhon, T., Abo-Akrab, M., Doma, M.A. and Hasan, A. 2023. Pomegranate Seeds and Peel Ethanolic Extracts Anticancer Potentials and Related Genetic, Histological, Immunohistochemical, Apoptotic and Oxidative Stress Profiles: in vitro Study. J. Exp. Pharmacol. 15, 191–205.
Neelima, N., Naikwadi G Sudhakar. and Kiran, J. 2011. A Preliminary Phytochemical Investigation on the Leaves of Solanum Xanthocarpum. Internat. J. Res. 2(3), 845–850.
Nisa, U., Zulkarnain Z Fitriani. and Wijayanti, E. 2020. Pasien Laki-Laki Usia Tahun Dengan Urolithiasis Di Klinik Saintifikasi Jamu: studi Kasus. J. Sains Dan Kesehatan 2(3), 171–173.
Noreen, H., Semmar, N., Farman, M. and Mccullagh, J.S.O. 2017. Measurement of Total Phenolic Content and Antioxidant Activity of Aerial Parts of Medicinal Plant Coronopus Didymus. Asian Pacific J. Trop. Med. 10(8), 792–801.
Patel, S. and Goyal, A. 2012. Recent developments in mushrooms as anti-cancer therapeutics: a review. 3 Biotech 2, 1–15.
Priyanka, J., Packiyalakshmi, R., Padmapriya, P. and Pavithra, M.K.S. 2019. Phytochemical and Anti-Microbial Analysis of Punica granatum Peel and Rind Extract. Internat. J. Innov. Technol. Explor. Engineer. 8, 88–90.
Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M. and Rice-Evans, C. 1999. Antioxidant Activity Applying an Improved ABTS Radical Cation Decolorization Assay. Free Radical Biol. Med. 26(9), 1231–1237.
Rehman, S., Choe, K. and Yoo, H. 2016. Review on a traditional herbal medicine, Eurycoma longifolia Jack (Tongkat Ali): its traditional uses, chemistry, evidence-based pharmacology and toxicology. Molecules 21(3), 331.
Rufai, Y. 2016.Comparative phyto-constituents analysis from the root bark and root core extractives of Cassia ferruginea (Schrad D. C) Plant. Scholars J. Agricult. Vet. Sci. 3, 275–283.
Sajjad, W., Sohail, M., Ali, B., Haq, A., Din, G., Hayat, M. and Khan, S. 2015. Antibacterial Activity of Punica granatum Peel Extract. Mycopathologia 13, 105–111.
Salwa, S.A.G., Azahar, N.F. and Mokhtar, N.F. 2020. Response surface optimization of high antioxidative extraction from Curcuma Zedoaria Leaves. J. Advan. Res. Fluid. Mech. Thermal. Sci. 43(1), 90–103.
Sameh, S., Abdollah, M.R.A., Elissawy, A.M., Al-Sayed, E., Labib, R.M., Ye, L., Chang, F.R. and Singab, A.N.B. 2025. Corchorus olitorius exhibits antiproliferative potential supported by metabolic profiling and integrative biological analyses. Scientific Rep. 15, 4192.
Saxena, N., Hartman, M., Bhoo-Pathy, N., Lim, J.N.W., Aw, T.C., Iau, P., Taib, N.A., Lee, S.C., Yip, C.H. and Verkooijen, H.M. 2012. Breast Cancer in South East Asia: comparison of Presentation and Outcome between a Middle Income and a High Income Country. World. J. Surg. 36(12), 2838–2846.
Shah, S.R., Ukaegbu, C.I., Hamid, H.A. and Alara, O.R. 2018. Evaluation of Antioxidant and Antibacterial Activities of the Stems of Flammulina Velutipes and Hypsizygus Tessellatus (White and Brown Var.) Extracted with Different Solvents. Food Measure. 12, 1947–1961.
Sudhakar, A. 2009. History of Cancer, Ancient and Modern Treatment Methods. J. Cancer Sci. Ther. 1(2), 1–4.
Viuda-Martos, M., Fernández-López, J. and Pérez-Álvarez, J.A. 2010. Pomegranate and its many functional components as related to human health: a review. Compr. Rev. Food Sci. Food Saf. 9(6), 635–654.
World Cancer Research Fund International (WCRFI). 2020. Global Cancer Prevalence.
Zerrouki, S., Mezhoud, S., Yağlioğlu, A.S., Bensouici, C., Atalar, M.N., Demirtas, I., Ameddah, S. and Mekkiou, R. 2022. Antioxidant, anticancer activities, and HPLC-DAD analyses of the medicinal halophyte Limoniastrum guyonianum Dur. extracts. J. Res. Pharm. 26(3), 598–608.