| Case Report | ||
Open Vet. J.. 2026; 16(2): 1361-1368
Open Veterinary Journal, (2026), Vol. 16(2): 1361-1368 Case Report First report of Coxiella burnetii infection (Q Fever) in Libyan sheep: A case studyAbdulwahab Kammon1,2*, Abdulhamid Giweli1,3, Laila Gewili1, Esra Al-ahrash1 and Seham Kalefa11National Research Center for Tropical and Transboundary Diseases, Alzintan, Libya 2Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 3Faculty of Science, Alzintan University, Alzintan, Libya *Corresponding Author: Abdulwahab Kammon. National Research Center for Tropical and Transboundary Diseases, Alzintan, Libya. Email: a.kammon [at] nrcttd.ly Submitted: 11/10/2025 Revised: 10/01/2026 Accepted: 19/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
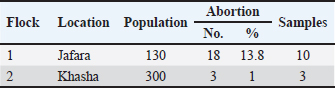
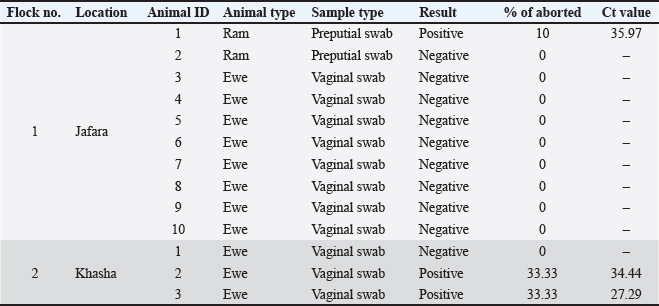
ABSTRACTBackground: Coxiella burnetii (C. burnetii) is one of the most important bacterial agents that cause abortion (Q fever) in ruminants and is a zoonotic disease. The current study reported the first confirmed detection of C. burnetii in sheep in Libya using real-time polymerase chain reaction coupled with high-resolution melt analysis. Case Description: Two flocks of sheep suffered abortion, with the aborted fetuses varying in age from early to late term. Vaginal swabs were collected, and DNA was extracted. DNA samples were analyzed using real-time PCR coupled with high-resolution melting (HRM) analysis, and the results showed infection with the zoonotic Q fever caused by C. burnetii. The animals responded to treatment with the antibiotic enrofloxacin, and the abortion was stopped after treatment. Conclusion: This study is the first documented evidence of Q fever in sheep in Libya and highlights the need for increased surveillance, further molecular and genomic studies, and the application of One Health approaches. The fact that the bacteria can persist in the environment and potentially cause zoonosis means that, despite being few in number, finding infected flocks could pose significant public health issues. Keywords: Coxiella burnetii, Q fever, Sheep, Real time PCR, High resolution melt, Zintan, Libya. IntroductionAbortion in ruminants, which can be referred to as premature death and expulsion of the fetuses before term, is a significant cause of reproductive loss in cows, camels, sheep, and goats and has substantial economic, welfare, and public health impacts. Although non-infectious causes (nutritional deficiencies, toxins, congenital anomalies, stress, and iatrogenic factors) can play important roles, infectious etiologies are largely responsible for the number of cases in domestic ruminants worldwide (Semango and Buza, 2024). Infectious abortifacients comprise several bacteria, protozoa, viruses, and fungi. Common bacterial agents include Brucella spp., Chlamydia abortus, C. burnetii (Q-fever), Listeria monocytogenes, Campylobacter, and Leptospira spp. The most important protozoan abortifacient agents are Toxoplasma gondii and N. caninum. Pestiviruses (e.g., bovine viral diarrhea virus), bluetongue virus, and Rift Valley fever virus are the most important viral causes. However, fungi (e.g., Aspergillus and zygomycetes) may cause mycotic placentitis and sporadic abortion. The relative importance of each agent varies according to host species, management system, diagnostic capacity, and regional ecology (Ramo et al., 2022). In North Africa and the Eastern Mediterranean, several zoonotic abortifacients—notably Brucella spp. and C. burnetii—are present at non-trivial prevalence in domestic ruminants, posing both production and public-health risks (Ahmadinezhad et al., 2022). Previous and recent serologic and molecular surveys in Libya have confirmed ongoing exposure to Brucella spp. in sheep, goats, cattle, and camels (Ahmed et al., 2015; Al-Griw et al., 2017; Altalhy and Shukri, 2024; Ahmed, 2025; Hamad et al., 2025), and regional reviews have documented variable but often substantial seroprevalence of C. burnetii across North Africa. However, information on the presence of C. burnetii in ruminants in Libya is lacking (Vanderburg et al., 2014 ). These pathogens are frequently implicated in the development of abortions or weak neonates, and they require coordinated animal and human health surveillance (Alshekh et al., 2024 ). Emerging and epidemic-prone pathogens remain an ever-present threat (Hecker et al., 2023). This highlights the value of robust, well-resourced diagnostic capacity (culture, serology, and molecular testing) and systematic abortion investigation to identify etiologies, implement control measures (vaccination, movement restrictions, and biosecurity), and reduce zoonotic spillover to humans (Lankester et al., 2024). The wide range of potential diagnoses and the frequent occurrence of co-infections complicate the etiologic diagnosis of abortion in ruminants. Improving diagnostic network capabilities in the region and tailoring control programs in Libya and the wider North African region against important zoonoses such as brucellosis and Q fever would seem to be essential (Semango and Buza, 2024). The current study reported the first confirmed detection of C. burnetii (Q fever) in sheep in Libya using real-time polymerase chain reaction (PCR) coupled with high-resolution melt (HRM) analysis. Case DetailsThis observation demonstrated infection in Libyan sheep flocks in Zintan due to Q fever abortions. The first flock of 130 sheep was raised in the Jfara area. During September 2025, the flock had 18 (13.8%) abortions. The flock was generally healthy. The age of the aborted fetuses varied from early to late stages. Oxytetracycline was administered intramuscularly, but it did not produce any results. The owner also reported that the aborted ewes showed a desire to mate with rams immediately after the abortion. The second flock, consisting of 300 healthy sheep raised in the Khasha area, had 3 aborted sheep (1%) during October 2025. Aborted fetuses were in the late stage. Oxytetracycline, along with vitamin E and selenium, was also administered. Abortions stopped spontaneously. Samples were collected based on the complaints of owners about abortions occurring in their flocks. Vaginal swabs containing 2 ml of normal saline were collected from the aborted animals (Sánchez-Rodríguez et al., 2024). According to the owner, aborted ewes accept rams for mating after the abortion; therefore, preputial swabs were also collected from rams. The samples were submitted to the Molecular Biology Laboratory of the National Research Center for Tropical and Transboundary Diseases. Ten samples were collected from flock 1, including 8 samples from ewes and 2 samples from rams (prepuce swabs), and three samples were collected from flock 2 (Table 1). Table 1. History and sampling of Coxiella burnetii infection in sheep flocks in Zintan.
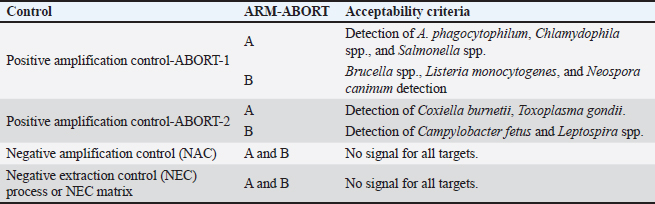
DNA was extracted from vaginal and prepuce swabs using alphaPrep™ DNA/RNA extraction kit (AlphaGene, Korea). The samples were prepared according to the manufacturer’s instructions, and extraction was completed using an automated NC-15 PLUS instrument (HanwoolTPC, Korea). A multiplex real-time PCR kit (IDVet, Innovative Diagnostics, 310, rue Louis Pasteur, Grabels, France) coupled with HRM technology was used to target 10 pathogens involved in ruminant abortions. Group A included C. burnetii, Anaplasma phagocytophilum, Chlamydophila spp., Salmonella spp., and Toxoplasma gondii, whereas group B included Brucella spp., Campylobacter fetus spp., Leptospira spp., Listeria monocytogenes, and Neospora caninum. The kit contains two reaction mixes, A and B. Each reaction mix contained Taq polymerase, primers, hydrolysis probes, nucleotides, and a fluorescent intercalant for the amplification and detection of pathogens in groups A and B. Two positive controls (PC1 and PC2) were used for the test validity. Each of these positive controls was used for every mix reaction (A and B). Two negative controls (DNAse/RNAse-free water) were also used, one for each reaction mix (NCA and NCB). For each sample, 10 μl of reaction mix A and reaction mix B were suspended in separate 0.1 ml PCR tubes. 5 µl of the sample extract (DNA) was added to each tube (for reaction mixes A and B). 5 µl of PC1 (A and B) and PC2 (A and B), as well as NCA and NCB, were added in separate tubes. The final volume of each tube was 15 µl. All tubes were loaded into the mic PCR instrument (Biomolecular Systems, Unit 5/3 Northward Upper Coomera QLD 4209, Australia). PCR conditions were as follows: polymerase activation at 95 °C for 2 min, followed by 40 cycles of denaturation at 95 °C for 10 s and elongation at 64 °C for 30 s, followed by 1 HRM cycle consisting of 1 minutes at 95 °C, 1 minutes at 40 °C, and 65 °C in continuous acquisition (Temp. steps 0.25 °C/s). The assay was validated, and the results was done via the DISoft™ software. The test is validated according to the following criteria: if one of the criteria is not accepted, the analysis cannot be reliably interpreted (Table 2). Table 2. The validation criteria used by the DISoft™ software.
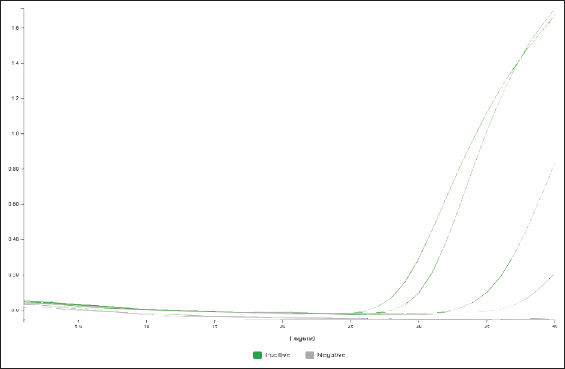
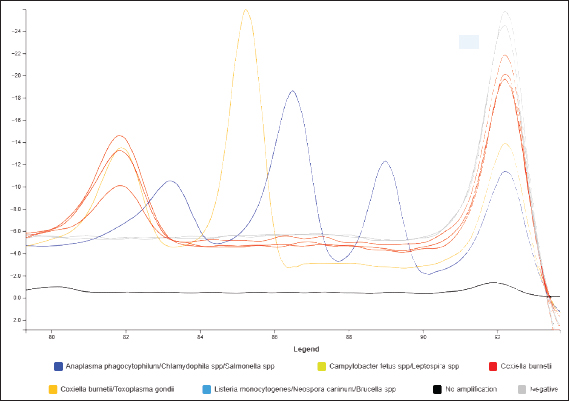
The results showed that one positive sample (1/10, 10%) for C. burnetii from flock 1 belongs to a ram. Two positive samples (2 of 3 [66.6%]) were found in flock 2 (Table 3 and Figs. 1–4). All tested samples were negative for the other nine abortifacient pathogens screened using this technique. After the owner failed to administer oxytetracycline, a course of 10% enrofloxacin was prescribed for the aborted and all pregnant ewes. Abortion was effectively stopped after treatment.
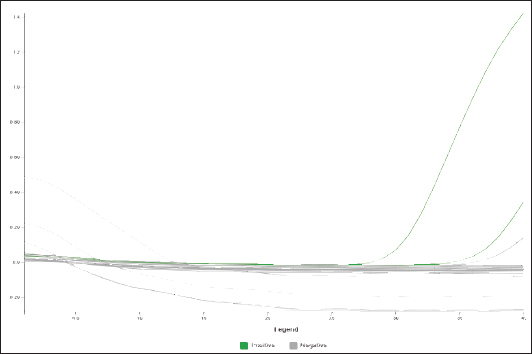
Fig. 1. Amplification curve of a positive C. burnetii sample in flock 1 and the positive and negative controls.
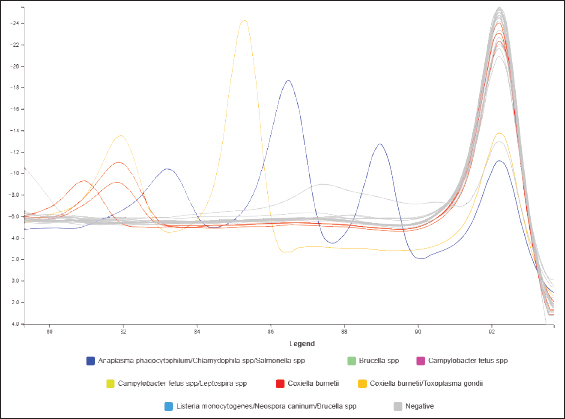
Fig. 2. Melt curve of a positive sample for C. burnetii in flock 1 (red) and the positive and negative controls.
Fig. 3. Amplification curve of a positive C. burnetii sample in flock 2 and the positive and negative controls.
Fig. 4. Melt curve of a positive sample for C. burnetii in flock 2 (red) and the positive and negative controls. Table 3. Result of Coxiella burnetii infection in sheep flocks in Zintan diagnosed by real-time PCR coupled with high-resolution methylation.
DiscussionThis study cited the first record of C. burnetii (the causative agent of Q fever) in sheep in Libya, which was identified using real-time polymerase chain reaction (PCR) coupled with high-resolution melt (HRM) analysis. The detection of C. burnetii DNA in ovine samples has significant implications for animal health, public health, and disease surveillance in Libya, where coxiellosis data remain scarce. Q fever is a well-recognized, globally distributed zoonosis in which sheep, goats, and cattle are the principal domestic reservoirs. Infected small ruminants can shed large quantities of the pathogen during parturition, contaminating the environment and creating a risk of human infection through the inhalation of contaminated aerosols (WOAH, 2018). In the present study, one positive sample (1/10, 10%) for C. burnetii from flock 1 belongs to a ram. Two positive samples (2 of 3 [66.6%]) were found in flock 2. The only positive sample of flock 1 was a preputial swab from an apparently healthy ram. According to the owner, aborted ewes accept rams for mating after the abortion, and this was confirmed by laboratory test results, which showed the presence of bacteria in preputial swabs. This observation suggests that rams (without infection) may act as mechanical carriers of the pathogen, contributing to its dissemination within the flock. Salmonella abortus ovis was found to be transmitted from infected rams to healthy ewes mostly through sexual contact (Vodas and Marinov, 1986). The low positivity rate in the current study could be attributed to the suboptimal sample type. Using the same technique (real-time PCR + HRM), we detected Brucella in cow cotyledons but not in vaginal swabs (unpublished data). However, collecting tissues (placenta) from aborted sheep is usually not possible in an extensive sheep farming system. In contrast, vaginal swabs provide practical and sensitive material for pathogen detection (Lankester et al., 2024). However, the timing of vaginal swab sampling is crucial. Molecular confirmation using real-time polymerase chain reaction (PCR) provides rapid and specific evidence of infection, proving particularly valuable in acute or subclinical cases where serological tests may yield negative results. Real-time polymerase chain reaction (PCR) assays targeting single- or multi-copy genes, such as the insertion sequences (IS1111 transposase element) of C. burnetii, are widely employed due to their enhanced analytical sensitivity and ability to quantify bacterial DNA in clinical and environmental samples. When combined with HRM analysis, this approach offers an efficient workflow: real-time PCR enables sensitive detection, while HRM facilitates rapid differentiation of sequence variants (genotypes) without the need for sequencing in many cases. Therefore, HRM analysis can aid in preliminary strain differentiation, detection of mixed infections, and validation of amplicon identity, thereby strengthening molecular diagnostic confidence (Erali et al., 2008; Modise et al., 2023). The detection of C. burnetii DNA in Libyan sheep aligns with findings from neighboring North African and Mediterranean countries, where coxiellosis has been reported in sheep, goats, and cattle. In North Africa, Q fever was reported by serology and/or molecular biology in ruminants in Morrocco (Benkirane et al., 1990), Egypt (Nahed and Khaled, 2012), Algeria (Menadi et al., 2022), and Tunisia (Guesmi et al., 2023). In sub-Saharan and other African countries, Q fever has been reported in ruminants in Cameroon (Maurice et al., 1968), Niger (Julvez et al., 1997), Chad (Schelling et al., 2003), Sudan (Hussien et al., 2012), Ghana (Adu-Addai et al., 2012 ), and Nigeria (Muhammad et al., 2023). Although earlier serological and limited molecular studies have indicated the circulation of C. burnetii in the region, systematic and up-to-date surveillance efforts in Libya have been lacking. This molecular confirmation in sheep represents a significant addition to the epidemiological record and underscores the need to reassess the national risk profile for Q fever (Devaux et al., 2020). C. burnetii infection in small ruminants is frequently subclinical but can cause reproductive losses, including abortion, stillbirth, and weak offspring, leading to decreased flock productivity. Infected animals may intermittently shed bacteria in milk, feces, urine, and especially in birth products, even in the absence of clinical signs. Therefore, the presence of C. burnetii within flocks has both economic and management implications. Farmers should be informed about appropriate biosecurity practices during lambing and kidding, placental and aborted material disposal, and hygiene measures to minimize environmental contamination and onward transmission (WOAH, 2018). In the current study, the animals in flock 1 responded positively to treatment with the antibiotic enrofloxacin, and the abortion was stopped after treatment. According to Plummer et al. (2018) there is a lack of scientific evidence of the efficacy of antimicrobial drugs when used in attempts to control abortion or shedding of C. burnetii. Thus, more research is needed to isolate the bacteria and conduct antibiotic sensitivity tests in vitro, taking into consideration BSL III-required conditions. However, vaccination using a phase I killed C burnetii vaccine (Coxevac, Ceva Sante Animale) is recommended for use in goats to reduce the risk of abortion and shedding of C. burnetii in vaginal fluids, feces, and milk, and in cattle to reduce shedding (Plummer et al., 2018). From a public health and One Health standpoint, the detection of C. burnetii DNA in sheep raises clear concerns. Human Q fever is primarily acquired through the inhalation of contaminated aerosols, with increased risk among farmers, abattoir workers, veterinarians, and residents of nearby communities. Because human infections are often underdiagnosed—manifesting as nonspecific febrile illness, atypical pneumonia, or chronic endocarditis—raising clinical awareness and integrating animal and human surveillance systems are of urgent importance. Even a small number of infected animals can precipitate human outbreaks in regions where small ruminant infections are present, but surveillance is limited, particularly during lambing seasons when environmental contamination peaks (Kimeli et al., 2025). Therefore, it is important to ensure that the animal and human health sectors have an effective One Health data-sharing mechanism where appropriate early warning and rapid response actions can occur through intersectoral cooperation when faced with threats such as Q fever. Conclusion: The molecular detection of Coxiella burnetii in Libyan sheep using real-time PCR with HRM analysis represents an important finding in the epidemiological search for knowledge about the bacterium in the region. Being the first documented evidence of its existence in Libya, this study highlights the need for increased surveillance, further molecular and genomic studies, and the application of One Health approaches. The fact that the bacteria can persist in the environment and potentially cause zoonosis means that, despite being few in number, finding infected flocks could pose significant public health issues. AcknowledgmentsThe authors highly value the support provided by the Zintan Municipality. FundingThis study received no specific funding. Authors’ contributionsThe authors contributed equally to the success of this scientific paper. Conflict of interestThe authors have no conflicts of interest to declare. Data availabilityAll data are included in this manuscript. ReferencesAdu-Addai, B., Koney, E.B., Addo, P., Kaneene, J., Mackenzie, C. and Agnew, D.W. 2012. Importance of infectious bovine reproductive diseases: an example from Ghana. Vet. Rec. 171(2), 47. Ahmadinezhad, M., Mounesan, L., Doosti-Irani, A. and Behzadi, M.Y. 2022. The prevalence of Q fever in the Eastern Mediterranean region: a systematic review and meta-analysis. Epidemiol. Health 44, e2022097; doi:10.4178/epih.e2022097 Ahmed, M.O. 2025. Brucellosis in Libya: past and Present Challenges. Libyan. JVet. Med. Sci. 6(2), 2025. Ahmed, M.O., Abouzeed, Y.M., Bennour, E.M. and Van Velkinburgh, J.C. 2015. Brucellosis update in Libya and regional prospective. Pathog. Glob. Health. 109(1), 39–40; doi:10.1179/2047773214Y.0000000170 Ahmed, M.O., Alshekh, K.A., Shahlol, A.M., Mostafa, K.K.B., Othman, A.A., Hiblu, M.A., Abouzeed, Y.M. and Daw, M.A. 2024. Seroprevalence of brucellosis in sheep and goats from Al Jufrah district in Libya. Pan Afr. Med. J. 28(48), 23; doi: 10.11604/pamj.2024.48.23.38566 Al-Griw, H.H., Kraim, E.S., Farhat, M.E., Perrett, L.L. and Whatmore, A.M. 2017. Evidence of ongoing brucellosis in livestock animals in North West Libya. J. Epidemiol. Glob. Health. 7(4), 285–288; doi: 10.1016/j.jegh.2017.09.001 Altalhy, A. and Shukri, K. 2024. Molecular Diagnosis and Antibodies Tests for Brucellosis Detected in Aborted and Apparently Healthy Sheep and Goats in and around Al Bayda City, Libya. AJMAS 1002, 997–1002; doi:10.54361/ajmas.247414 Alshekh, K.A., Shahlol, A.M., Mostafa, K.K.B., Othman, A.A., Hiblu, M.A., Abouzeed, Y.M., Daw, M.A. and Ahmed, M.O. 2024. Seroprevalence of brucellosis in sheep and goats from Al Jufrah district in Libya. The Pan African Med. J. 48, 23; doi: 10.11604/pamj.2024.48.23.38566 Benkirane, A., Jabli, N. and Rodolakis, A. 1990. Frequency of abortion and seroprevalence of the principal diseases causing ovine infectious abortion in the area of Rabat (Morocco). Annales De Recherches Veterinaires. Ann. Vet. Res. 21(4), 267–273. Devaux, C.A., Osman, I.O., Million, M. and Raoult, D. 2020. Coxiella burnetii in Dromedary Camels (Camelus dromedarius): a Possible Threat for Humans and Livestock in North Africa and the Near and Middle East?. Front. Vet. Sci. 7, 558481; doi:10.3389/fvets.2020.558481 Erali, M., Voelkerding, K.V. and Wittwer, C.T. 2008. High resolution melting applications for clinical laboratory medicine. Exp. Mol. Pathol. 85(1), 50–58; doi:10.1016/j.yexmp.2008.03.012 Guesmi, K., Kalthoum, S., Mamlouk, A., Baccar, M.N., Belhajmohamed, B., Hajlaoui, H., Toumi, A., Cherni, J., Seghaier, C. and Messadi, L. 2023. Seroprevalence of zoonotic abortive diseases and their associated risk factors in Tunisian sheep. BMC. Vet. Res. 19, 50; doi:10.1186/s12917-022-03541-9 Hamad, Y., Meriz, O., Zafir, Z. and El-khodery, S. 2025. Prevalence and zoonotic implication of animal brucellosis in Libya: a systematic meta-analysis. J. Adv. Vet. Res. 15(3), 370–374. Hecker, Y.P., González-Ortega, S., Cano, S., Ortega-Mora, L.M. and Horcajo, P. 2023. Bovine infectious abortion: a systematic review and meta-analysis. Front. Vet. Sci. 10, 1249410; doi: 10.3389/fvets.2023.1249410 Hussien, M.O. ElFahal, AM., Enan, KA., Taha, KM., Mohammed, MS., Salih, DA., Mohammadain, SI., Saeed, AA. and El-Hussein, AM. 2012. Seroprevalence of q fever in goats in the Sudan. Vet. World 5(7), 394 -397. Julvez, J., Michault, A. and Kerdelhue, C. 1997. Etude sérologique des rickettsioses à Niamey, Niger Serological study of rickettsia infections in Niamey, Niger. Medecine. Tropicale. Revue. Du. Corps. De. Sante. Colonial. 57(2), 153–156. Kimeli, P., Mwacalimba, K., Tiernan, R., Mijten, E., Miroshnychenko, T. and Poulsen Nautrup, B. 2025. Important Diseases of Small Ruminants in Sub-Saharan Africa: a Review with a Focus on Current Strategies for Treatment and Control in Smallholder Systems. Animals. (Basel). 15(5), 706; doi:10.3390/ani15050706 Lankester, F., Kibona, T.J., Allan, K.J., De Glanville, W., Buza, J.J., Katzer, F., Halliday, J.E., Mmbaga, B.T., Wheelhouse, N., Innes, E.A., Thomas, K.M., Nyasebwa, O.M., Swai, E., Claxton, J.R. and Cleaveland, S. 2024. Livestock abortion surveillance in Tanzania reveals disease priorities and importance of timely collection of vaginal swab samples for attribution. Elife 13, RP95296; doi:10.7554/eLife.95296 Maurice, Y., Fernagut, R. and Gerome, R. 1968. Rickettsial diseases of North Cameroon; epidemiological study. Revue. D’elevage. Et. De. Medecine. Veterinaire. Des. Pays. Tropicaux. 21(3), 341–349. Menadi, S.E., Chisu, V., Santucciu, C., Di Domenico, M., Curini, V. and Masala, G. 2022. Serological, Molecular Prevalence and Genotyping of Coxiella burnetii in Dairy Cattle Herds in Northeastern Algeria. Vet. Sci. 9(2), 40; doi:10.3390/vetsci9020040 Modise, B.M., Mpoloka, S.W., Settypalli, T.B.K., Hyera, J., Natale, A., Ceglie, L., Gcebe, N., Marobela-Raborokgwe, C., Viljoen, G.J., Cattoli, G. and Lamien, C.E. 2023. A novel multiplex qPCR HRM assay for the simultaneous detection of four abortive zoonotic agents in cattle, sheep, and goats. Sci. Rep. 13(1), 12282; doi:10.1038/s41598-023-39447-1 Muhammad, K.A., Gadzama, U.N. and Onyiche, T.E. 2023. Distribution and Prevalence of Coxiella burnetii in Animals, Humans, and Ticks in Nigeria: a Systematic Review. Infect. Dis. Rep. 15(5), 576–588; doi:10.3390/idr15050056 Nahed, H.G. and Khaled, A.A.M. 2012. Seroprevalence of Coxiella burnetii antibodies among farm animals and human contacts in Egypt. J. Am. Sci. 8, 619–621. WOAH. 2018. Q Fever. In Manual of diagnostic tests and vaccines for terrestrial animals. Paris, France: World Organisation for Animal Health. Available from: http://www.woah.org/en/disease/q-fever/ Plummer, P.J., Mcclure, J.T., Menzies, P., Morley, P.S., Van Den Brom, R. and Van Metre, D.C. 2018. Management of Coxiella burnetii infection in livestock populations and the associated zoonotic risk: a consensus statement. J. Vet. Intern. Med. 32, 1481–1494; doi:10.1111/jvim.15229 Ramo, M.D.L.A., Benito, A.A., Quílez, J., Monteagudo, L.V., Baselga, C. and Tejedor, M.T. 2022. Coxiella burnetii and Co-Infections with Other Major Pathogens Causing Abortion in Small Ruminant Flocks in the Iberian Peninsula. Animals 12(24), 3454; doi:10.3390/ani12243454 Sánchez-Rodríguez, O., Palomares-Resendiz, E.G., Díaz-Aparicio, E., Arellano-Reynoso, B., Martínez-Chavarría, L.C., Martínez-Serrano, G. and Hernández-Castro, R. 2024. Molecular Detection of Coxiella burnetii in Vaginal Swab Samples from Sheep That Aborted. Vector Borne Zoonotic Dis. (Larchmont. NY). 24(12), 838–841; doi:10.1089/vbz.2023.0153 Schelling, E., Diguimbaye, C., Daoud, S., Nicolet, J., Boerlin, P., Tanner, M. and Zinsstag, J. 2003. Brucellosis and Q-fever seroprevalences of nomadic pastoralists and their livestock in Chad. Prev. Vet. Med. 61(4), 279–293; doi:10.1016/j.prevetmed.2003.08.004 Semango, G.P. and Buza, J. 2024. Review of the Current Status on Livestock Abortigenic Diseases Surveillance in Africa and Asia. Vet. Sci. 11(9), 425; doi:10.3390/vetsci11090425 Vanderburg, S., Rubach, M.P., Halliday, J.E.B., Cleaveland, S., Reddy, E.A. and Crump, J.A. 2014. Epidemiology of Coxiella burnetii infection in Africa: a OneHealth systematic review. PLos Neglected Trop. Dis. 8(4), e2787; doi:10.1371/journal.pntd.0002787 Vodas, K. and Marinov, M.F. 1986. Roliata na kochovete v epizootologiiata na salmonelozniia abort po ovtsete Role of rams in the epizootiology of salmonellal abortion in sheep. Vet. Med. Nauki. 23(9), 61–68. | ||
| How to Cite this Article |
| Pubmed Style Kammon A, Giweli A, Gewili L, Al-ahrash E, Kalefa S. First report of Coxiella burnetii infection (Q Fever) in Libyan sheep: A case study. Open Vet. J.. 2026; 16(2): 1361-1368. doi:10.5455/OVJ.2026.v16.i2.52 Web Style Kammon A, Giweli A, Gewili L, Al-ahrash E, Kalefa S. First report of Coxiella burnetii infection (Q Fever) in Libyan sheep: A case study. https://www.openveterinaryjournal.com/?mno=289639 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.52 AMA (American Medical Association) Style Kammon A, Giweli A, Gewili L, Al-ahrash E, Kalefa S. First report of Coxiella burnetii infection (Q Fever) in Libyan sheep: A case study. Open Vet. J.. 2026; 16(2): 1361-1368. doi:10.5455/OVJ.2026.v16.i2.52 Vancouver/ICMJE Style Kammon A, Giweli A, Gewili L, Al-ahrash E, Kalefa S. First report of Coxiella burnetii infection (Q Fever) in Libyan sheep: A case study. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1361-1368. doi:10.5455/OVJ.2026.v16.i2.52 Harvard Style Kammon, A., Giweli, . A., Gewili, . L., Al-ahrash, . E. & Kalefa, . S. (2026) First report of Coxiella burnetii infection (Q Fever) in Libyan sheep: A case study. Open Vet. J., 16 (2), 1361-1368. doi:10.5455/OVJ.2026.v16.i2.52 Turabian Style Kammon, Abdulwahab, Abdulhamid Giweli, Laila Gewili, Esra Al-ahrash, and Seham Kalefa. 2026. First report of Coxiella burnetii infection (Q Fever) in Libyan sheep: A case study. Open Veterinary Journal, 16 (2), 1361-1368. doi:10.5455/OVJ.2026.v16.i2.52 Chicago Style Kammon, Abdulwahab, Abdulhamid Giweli, Laila Gewili, Esra Al-ahrash, and Seham Kalefa. "First report of Coxiella burnetii infection (Q Fever) in Libyan sheep: A case study." Open Veterinary Journal 16 (2026), 1361-1368. doi:10.5455/OVJ.2026.v16.i2.52 MLA (The Modern Language Association) Style Kammon, Abdulwahab, Abdulhamid Giweli, Laila Gewili, Esra Al-ahrash, and Seham Kalefa. "First report of Coxiella burnetii infection (Q Fever) in Libyan sheep: A case study." Open Veterinary Journal 16.2 (2026), 1361-1368. Print. doi:10.5455/OVJ.2026.v16.i2.52 APA (American Psychological Association) Style Kammon, A., Giweli, . A., Gewili, . L., Al-ahrash, . E. & Kalefa, . S. (2026) First report of Coxiella burnetii infection (Q Fever) in Libyan sheep: A case study. Open Veterinary Journal, 16 (2), 1361-1368. doi:10.5455/OVJ.2026.v16.i2.52 |