| Research Article | ||
Open Vet. J.. 2026; 16(3): 1695-1703 Open Veterinary Journal, (2026), Vol. 16(3): 1695-1703 Research Article Pollen preferences of honey bees: How do floral and pollen characteristics influence foraging strategies?Marwan M. Keshlaf1*, Farid Mitsho1 and Robert Spooner-Hart21Department of Plant Protection, Faculty of Agriculture, University of Tripoli, Tripoli, Libya 2Schools of Science and Health, University of Western Sydney, Sydney, Australia *Corresponding Author: Marwan Keshlaf. Department of Plant Protection, Faculty of Agriculture, University of Tripoli, Tripoli, Libya. Email: m.keshlaf [at] UOT.edu.ly Submitted: 12/10/2025 Revised: 22/01/2026 Accepted: 06/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
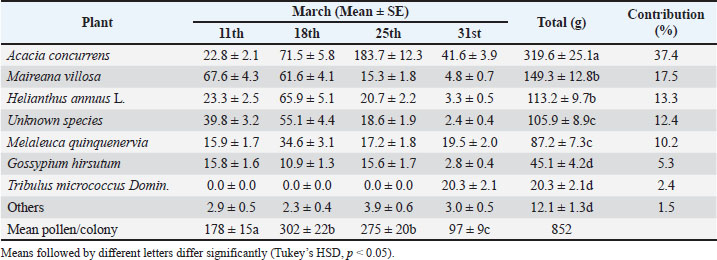
AbstractBackground: Understanding honey bee pollen foraging behavior is crucial for optimizing pollination services in agricultural systems. Floral and pollen characteristics significantly influence pollinator preferences and foraging efficiency. Aim: This study investigated the influence of floral characteristics and pollen traits on the foraging preferences of the honey bee (Apis mellifera ligustica) in Australian cotton agroecosystems. Methods: In summer 2007, pollen traps were installed on eight honey bee colonies adjacent to cotton fields. Collected pollen pellets were analyzed for their weight, color, and botanical origin. Scanning Electron Microscopy was used to examine pollen morphology and size. Statistical analyses included Pearson’s correlation and analysis of variance with Tukey’s HSD test. Results: Honey bees showed significant preferences for specific pollen sources (F₇,₂₈=84.593, p < 0.02). Acacia concurrens was the most collected species (37.4%), followed by Maireana villosa (17.5%) and Helianthus annuus (13.3%). Cotton (Gossypium hirsutum) contributed only 5.3% of the total pollen. A strong negative correlation was found between pollen grain size and pellet weight (r=−0.89, p < 0.05). Smaller, smooth-textured grains (e.g., M. villosa, 31 μm) produced heavier pellets (6.42 mg), whereas larger grains (e.g., G. hirsutum, 101 μm) produced lighter pellets (2.45 mg). Brightly colored flowers (yellow and orange) were strongly preferred over creamy white blossoms. Conclusion: Honey bees optimize foraging by selecting flowers with favorable morphology, vibrant colors, and collectible pollen traits. The low preference for cotton pollen despite its abundance highlights the importance of alternative floral resources for maintaining honey bee colonies in agricultural landscapes. These findings provide a foundational understanding of the ecology of pollen foraging, the principles of which remain relevant for informing current strategies to support pollinator health and crop pollination services in agricultural landscapes. Keywords: Apis mellifera, Foraging ecology, Pollen morphology, Scanning electron microscopy, Australia. IntroductionHoneybees require a balanced intake of carbohydrates, proteins, fats, minerals, vitamins, and water to rear their brood (Loper and Berdel, 1980). Carbohydrates are primarily obtained from nectar, while pollen is the main source of proteins, fats, minerals, and vitamins. Honeybees need to collect approximately 23–32 kg of pollen annually to meet the nutritional demands of a colony (Todd and Bishop, 1940). The optimal protein content of pollen is typically in the range of 20%–30%, is a critical indicator of its nutritional value for honeybees (Pernal and Currie, 2002; Keller et al., 2005; Ghosh and Chung-Chung, 2017; Vaudo et al., 2020). Consequently, both the quality and quantity of pollen play a crucial role in shaping foraging behavior, along with other factors such as flower characteristics, including color, shape, morphology, display area, and scent (Ruedenauer et al., 2019). Studies have demonstrated significant differences between the foraging behaviors of nectar-collecting and pollen-collecting honeybee workers, particularly in their decision-making patterns and the factors influencing their preferences (Seeley, 1986). Nectar-gatherers demonstrate a strong preference for floral sources with high sugar content, which enhances their sensitivity to nectar concentration and floral abundance in the environment (Brunet et al., 2015). In contrast, pollen gatherers prioritize pollen that is rich in essential nutrients required for brood development. Therefore, plant type and pollen quality influence their foraging behavior more strongly than nectar availability (Keller et al., 2005). This distinction underscores the specialized roles of these worker groups in maintaining colony health and efficiency. Nectar gatherers tend to move more rapidly between flowers, maximizing energy intake to support the colony’s metabolic demands and maintain food reserves (Pattrick et al., 2013). Conversely, pollen gatherers exhibit more targeted behavior, as their role is focused on fulfilling the colony’s protein requirements, resulting in less frequent but more selective visits to flowers (Leonhardt and Blüthgen, 2012). Flower shape and color are critical factors in attracting pollinators, as they often serve as visual cues for nectar or pollen availability (Chittka and Raine, 2006). Different pollinator species exhibit distinct preferences based on foraging requirements and physical adaptations. Honeybees display clear distinctions in their floral preferences based on their foraging objectives. Nectar gatherers favored tubular or radially symmetrical flowers that provided easier access to deeply stored nectar (Chittka and Thomson, 2012). Pollen gatherers, on the other hand, showed greater adaptability to flower morphology, selecting structures that facilitate efficient pollen collection (Harder, 1990). Flower color also plays a key role in attracting nectar gatherers, with blue and violet hues being particularly appealing due to their alignment with the spectral sensitivity of bees (Kevan and Backhaus, 1998). Furthermore, flower constancy in honey bees is strongly influenced by color learning and memory (Chittka et al., 1999). However, pollen gatherers are less influenced by flower color and instead prioritize pollen quality and abundance (Roulston and Goodell, 2011). Additionally, flower size affects nectar gatherers, who prefer larger flowers with abundant nectar, whereas pollen gatherers focus primarily on pollen-rich flowers, regardless of size (Corbet et al., 1995; Pacini and Hesse, 2005). This complex interplay of factors can lead to floral competition, where more attractive species divert foragers from target crops, which is further influenced by landscape-scale resource distribution and climatic conditions (Bailey et al., 2022). This study investigates the foraging preferences of pollen-collecting honey bees in terms of flower shape and color, as well as their associations with specific flowering species in Narrabri, Australia. Given the widespread cultivation of cotton in the region and the common practice of renting honey bee colonies to enhance crop yields, the study initially hypothesized that honey bees would preferentially collect pollen from cotton flowers over neighboring plants, based on the assumption of proximity and resource abundance. However, this premise is re-evaluated in light of the actual foraging data collected. Notably, no published research currently exists on the floral associations of pollinators in this region, highlighting the significance of this study. Previous research by the authors in similar agroecosystems has documented honey bee foraging patterns and pollination efficacy in Australian Bt cotton (Keshlaf et al., 2012). Although the data were collected in 2007, this research provides a valuable historical baseline on the preferences of pollen foraging in a major cotton-growing region. The fundamental principles of how floral and pollen traits influence bee behavior are timeless, and this dataset offers a critical reference point for assessing changes in foraging ecology over time, especially in the context of landscape alteration and climate change (Goulson et al., 2015). Materials and MethodsThis study was conducted during summer 2007 on a 2-hectare commercially irrigated cotton crop at the Australian Cotton Research Institute in Narrabri (30.30°S, 149.8°E), one of New South Wales’ major cotton-growing regions. The site is characterized by a semi-arid climate with hot summers. The surrounding landscape comprises a mosaic of other cotton fields, native vegetation, and fallow land. No insecticides were applied during the study period, and standard irrigation practices were followed. The eight study hives were placed at the cotton field edge, within the typical range of several kilometers foraging range of honey bees, ensuring access to both the cotton crop and the surrounding flora. Pollen collection and analysisEight colonies of Apis mellifera ligustica were used to monitor pollen collection, each equipped with front-mounted grid-type pollen traps. The stated efficiency rate of 30% was based on standard calibrations for this common trap design as reported in the literature (Taber, 1977), though a specific validation study for these particular traps was not conducted. Over a 25-day period, pollen samples were collected weekly from the trap trays and taken to the laboratory for detailed analysis. Wet pollen pellets were weighed using a digital balance (PB403-S, Mettler Toledo, Switzerland) to assess the quantity of pollen collected. The pollen analysis involved sorting a 50 ml sample of pollen from each weekly collection based on morphological features such as color and size. The representative samples from each group were then analyzed microscopically following the protocol of (Rashad and Parker 1958). The homogeneous pollen pellets were weighed to estimate the relative contribution of each plant species to the total pollen collection. Additionally, pollen samples were collected from cotton flowers and surrounding vegetation to create a reference collection. Despite exhaustive efforts, one pollen type contributing 12.4% of the total could not be identified to the species level and remains classified as unknown. SEMScanning electron microscopy was used to analyze individual pollen grains from primary sources, allowing for a detailed examination of their morphology and size. The pollen samples were dried in desiccators containing silica gel for 24 hours, then mounted on stubs and coated with a thin layer of gold to enhance the conductivity (Erdtman, 1969). Analysis was conducted using a Philips XL30 scanning electron microscope set to operate at 10 kV. Statistical analysisStatistical analyses were conducted to investigate the relationship between pollen grain size and pollen pellet weight, using Pearson’s correlation coefficient to assess linear associations and Spearman’s rank correlation coefficient to evaluate rank-based relationships. Additionally, statistical comparisons were performed to examine variations in the mean pollen quantities collected by honey bee colonies across different weeks and plant species. To identify significant differences among plant species, a one-way analysis of variance (ANOVA) was conducted, followed by Tukey’s honestly significant difference test at a significance level of 0.05. The ANOVA model treated plant species as the independent factor and pollen weight as the dependent variable, with colonies and weeks as replicates within the model, leading to the reported degrees of freedom (F7,28). All statistical analyses were performed using SPSS version 16.0, with statistical significance set at p < 0.05. The assertion of a correlation between surface texture and pellet weight was re-examined. As texture is a categorical variable, the original Pearson’s correlation coefficient was removed. Instead, differences in pellet weight among the three texture groups (smooth, reticulated, and spiny) were analyzed using one-way analysis of variance. Ethical approvalNot needed for this study. ResultsPollen collection dynamicsThis study assessed pollen collection by honey bee colonies over a 25-day period, focusing on the weight of trapped pollen. Colonies collected between 660.5 and 980.1 g pollen, with an average of 852.7 g per colony. With a trap efficiency of 30%, the estimated total collection per colony was 2.84 kg. Weekly data (Table 1) revealed fluctuations in pollen collection, with mean weekly weights of 178.1, 301.9, 275.0, and 97.7 g per colony on March 11th, 18th, 25th, and 30th, respectively. The highest pollen yield occurred between March 18th–25th (F3,28=49.744, p < 0.001), coinciding with the peak blooming period of the dominant local plant species. The initial hypothesis that bees would prefer cotton pollen was not supported. Table 1. Weekly amount of trapped pollen (g) collected by honey bees.
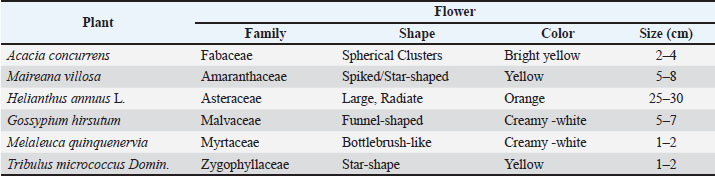
Pollen source preferencesHoney bees collected pollen from a variety of plant species (Table 2), demonstrating significant preferences (F7,28=84.593, p < 0.02) based on floral morphology and pollen characteristics. The most collected pollen was from Black Wattle (Acacia concurrens), which contributed 37.4% (319.6 g) of the total. Second, silver bluebush (Maireana villosa) and sunflower (Helianthus annuus) were significant sources, providing 17.5% (149.3 g) and 13.3% (113.2 g), respectively. An unidentified species ranked third, contributing 12.4% (105.9 g), while Paperbark Tea Tree (Melaleuca quinquenervia) accounted for 10.2% (87.2 g). Cotton (Gossypium hirsutum) and Small Caltrop (Tribulus micrococcus) made smaller contributions of 5.3% and 2.4%, respectively. Minor plant sources collectively accounted for only 1.5% (12.1 g), indicating a limited reliance on less common pollen sources during the study period. Table 2. The major sources of trapped pollen collected by honey bees and flower features.
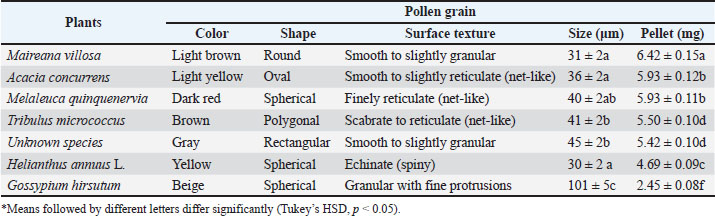
Pollen morphology and physical characteristicsPollen grains from different plant species exhibited distinct morphological characteristics (Table 3). The seven pollen types were classified into three groups based on surface texture (Fig. 1). Table 3. The major sources of trapped pollen collected by honey bees, pollen morphology, and average pollen pellet weight.
Fig. 1. Scanning electron microscopy images showing the morphology of the major trapped pollen types collected from the experimental colonies, Narrabri, 2005. (1: Acacia concurrens Pedley, 2: Maireana villosa (Lindl.) Paul G. Wilson, 3: Helianthus annuus L., 4: Unknown species, 5: Melaleuca quinquenervia (Cav.) S.F. Blake, 6: Tribulus micro-coccus Domin). Smooth to slightly granular textureThis group included M. villosa, which has oval-shaped grains with the smallest size (31 µm) and the highest pellet weight (6.42 mg). An unidentified species also fell into this category, featuring medium-sized grains (45 µm) with a regular rectangular appearance and a pellet weight of 5.42 mg. Acacia concurrens also belongs to this group, characterized by spherical grains divided into multiple polygonal sections resembling a honeycomb or quilted structure, with a size of 36 µm and an average pellet weight of 5.93 mg. Reticulate (Net-like) textureThis category includes M. quinquenervia, which has spherical to subprolate medium-sized grains (40 µm) and a pollen pellet weight of 5.93 mg. Tribulus micrococcus also belongs to this group, exhibiting polygonal to slightly oblate medium-sized grains (41 µm) with a scabrate to reticulate surface and a pellet weight of 5.50 mg. Spiny (echinate) textureThis group included H. annuus, which had small spherical to elliptical grains (30 µm) covered in spines and a pellet weight of 4.69 mg. Gossypium hirsutum had the largest grains (101 µm) with a granular texture and fine protrusions (Fig. 2), but had the smallest pellet weight (2.45 mg).
Fig. 2. SEM images comparing cotton pollen (Gossypium hirsutum) with M. quinquenervia (left) and Helianthus annuus L. (right), highlighting differences in grain size and texture. Statistical relationshipsTukey’s honestly significant difference test revealed significant differences in pellet weights among plant species (p < 0.05). Maireana villosa had the highest pellet weight (6.42 mg), while G. hirsutum had the lowest (2.45 mg). A one-way ANOVA revealed a significant effect of the pollen surface texture group (smooth, reticulate, and spiny) on pellet weight (Pearson’s correlation coefficient (r)=0.746, p < 0.05), with the smooth group yielding the heaviest pellets. A strong negative correlation was found between pollen grain size and pellet weight, with Pearson’s correlation coefficient (r)=−0.89, indicating that larger pollen grains were associated with lower pellet weights. This correlation was statistically significant (p < 0.05). However, Spearman’s correlation coefficient was not statistically significant (r=0.455, p > 0.05), suggesting that Pearson’s analysis may have been influenced by outliers, particularly G. hirsutum, which exhibited the largest pollen grains (101 µm) and the lowest pellet weight (2.45 mg). DiscussionThe findings of this 2007 study provide a foundational understanding of pollen foraging preferences, the core principles of which continue to inform apiculture and pollination management. While agricultural landscapes evolve, the fundamental relationships between bee behavior and floral traits documented here remain critical for designing strategies to support pollinators. The highest pollen yield (2.84 kg per colony) in this study occurred between March 18 and 25, coinciding with the peak blooming period of the dominant local flora. This temporal alignment highlights the critical role of environmental factors, such as climate and flowering phenology, in shaping the foraging patterns of honey bees. These findings are consistent with those of previous studies by Dimou and Thrasyvoulou (2007) and Olsen et al. (1979), which emphasized the influence of seasonal floral availability on pollen collection, a relationship that remains a cornerstone of pollinator ecology (Forrest and Thomson, 2019). The observed yield aligned with those of earlier studies. Synge (1947) reported recorded peak-season pollen collection ranging from 1.3 to 1.4 kg per colony, whereas Eckert (1942) documented annual yields reaching up to 55.4 kg under optimal conditions. Vaissière (1991) further contextualized these patterns by showing that daily pollen influxes varied from 3.6 to 143.5 g per colony depending on floral availability. In this study, the average daily influx of 109.0 g indicates favorable foraging conditions, likely driven by abundant floral resources during the peak bloom period. The diversity in flower shape and size among the observed floral sources complicates the assessment of their specific impact on bee preferences. Although flower colors fell within the bees’ preferred spectrum (e.g., yellow, white), morphological variations influenced their foraging behavior. For instance, A. concurrens exhibited clustered, medium-sized flowers (2–4 cm), which offered concentrated resources that improved foraging efficiency. This pattern was consistent with Goodwin (1986) observations and further supported by Johnson and Ackerman (1989), who emphasized that clustered arrangements reduce energy expenditure for pollinators, a concept now integral to planting schemes for pollinator support (Goulson et al., 2015). In contrast, H. annuus’s large, open flowers (25–30 cm) provided easy nectar access, aligning with Waser’s (1986) findings on how floral architecture enhances pollinator attraction. Similarly, Chittka and Waser (1997) reported that larger floral displays improved nectar accessibility. Conversely, smaller flowers, such as cup-shaped T. micrococcus (1–2 cm) and bottlebrush-shaped M. quinquenervia (1–2 cm), optimized feeding efficiency through compact structures, as described by Kearns and Inouye (1993) and corroborated by Goulson et al. (2008), who highlighted the role of flower miniaturization in rapid nectar collection. It is important to note that the large, composite inflorescence of H. annuus presents a fundamentally different visual and structural cue compared to the single flower of cotton, which may confound direct comparisons based on color alone. Brightly colored flowers, such as yellow (A. concurrens, M. villosa) and orange (H. annuus) flowers, strongly stimulated the foraging of honey bees and promoted repeat visits. This is consistent with Buchmann and Nabhan’s (1996) assertion that vivid hues serve as critical visual cues for pollinators. In contrast, creamy-white flowers (G. hirsutum, M. quinquenervia) attracted fewer bees, a pattern consistent with Thomson (2004) observation that muted colors reduce pollinator visitation. Notably, darker blooms did not directly drive foraging preference but likely enhanced floral visibility through landscape contrast, as proposed by Buchmann and Shipman (1990) and Free (1993), who argued that dark hues function as secondary visual markers rather than primary attractants. This is consistent with Dafni (1992) hypothesis that darker pigmentation improves floral detectability under variable light conditions, offering an adaptive advantage in visual signaling systems. Scent, a factor not measured in this study, also plays a crucial role in attraction and could interact with color cues (Raguso, 2008). Recent research has shown that bees evaluate both sugar concentration and viscosity when assessing nectar quality, demonstrating the complexity of foraging decisions (Pattrick et al., 2024). The foraging behavior observed in this study aligns with broader research on the ecology of honey bees in agricultural systems. Sustainable beekeeping practices emphasize the importance of diverse floral resources for maintaining colony health (Keshlaf, 2017). Understanding these foraging preferences is crucial for developing effective pollination strategies in cotton cultivation. The color of pollen pellets varied significantly among plant species (Table 2). However, its influence on flower preference remains ambiguous. For example, A. concurrens, the most frequently collected pollen source, produced light brown pellets, whereas H. annuus, with its bright yellow pellets, accounted for only 13.3% of the total pollen collected. In contrast, despite having dark brown pellets, M. villosa contributed a substantial proportion (17.5%) of the pollen, indicating that factors such as pellet shape and resource availability may play a more critical role than color alone in shaping bee foraging behavior. Cook et al. (2003) reported that while the color of pollen can provide information about plant species and their nutrient profile, it does not consistently determine bee preference. This aligns with the findings of Pernal and Currie (2002), who demonstrated that bees prioritize pollen quality, such as nutritional value, over visual characteristics, such as color. More recent research has confirmed that nutritional quality, particularly protein and lipid content, is a primary driver of pollen selection (Vaudo et al., 2020; Ruedenauer et al., 2021; Lawson et al., 2022). Together, these observations suggest that pollen color may serve as a secondary cue; other factors—including morphology, chemical composition, and resource abundance—were likely more influential in determining bee foraging decisions (Vaudo et al., 2023; Ruedenauer et al., 2024). The shape of the pollen grains varied widely (Fig. 1), including round, oval, spherical, irregular, and polygonal forms. Acacia concurrens (36 µm) and M. villosa (31 µm) had high pellet weights (5.93 and 6.42 mg, respectively), suggesting that their smooth surface texture and compact grains facilitated efficient packing into pollen baskets. In contrast, irregular grains (e.g., an unidentified species, 45 µm) and polygonal grains (T. micrococcus, 41 µm) were less efficiently collected due to handling challenges, consistent with the findings of Roulston and Cane (2000a,b). Spiny grains, similar to those of G. hirsutum, hindered adhesion and compacting, limiting collection (5.4% of total pollen), as also noted by Roulston and Cane (2000a,b). This demonstrates a clear link between pollen physical traits and foraging efficiency. Cotton pollination involves complex interactions among multiple pollinator species. Pollen beetles (Carpophilus aterrimus) also play a role in cotton pollination and may interact with honey bee foraging behavior (Keshlaf et al., 2025). These complex ecological interactions highlight the need for integrated approaches to pollination management in agricultural systems. An inverse relationship exists between pollen grain size and pollen pellet weight. Smaller grains, such as those from M. villosa (31 µm) and H. annuus (30 µm), significantly contributed to the total pollen collection. Their compact size likely enhanced their transport and packing efficiency. Conversely, larger grains, such as those of G. hirsutum (101 µm) (Fig. 2), were associated with lower pellet weights (2.45 mg), suggesting handling challenges or reduced nutritional appeal. While this correlation is clear, the evolutionary interpretations regarding plant strategy and co-evolution are speculative and extend beyond the direct evidence provided by this study, which focused on bee foraging outcomes rather than plant reproductive biology. Acacia concurrens emerged as the most significant pollen source, consistent with Goodwin and Perry’s (1992) findings, who noted the attractiveness of clustered flowers due to their accessibility and concentrated resources. This preference is supported by earlier studies, such as those by Rashad and Parker (1958) and Percival (1955), which demonstrated that plants offering high pollen availability and moderate grain size tend to dominate honey bee diets. The moderate grain size (36 µm) and substantial pellet weight (5.93 mg) of A. concurrens reinforce its status as a primary and highly favored honeybee resource. A key finding is the clear rejection of the initial hypothesis; honey bees showed a strong preference for pollen collection from alternative flora over cotton. Although nectar-gathering bees contribute to cotton pollination, several studies have indicated that they tend to reject cotton pollen. Bees actively remove cotton pollen if it sticks to their bodies before returning to their hives, as noted by Keshlaf (2009) and other studies (Moffett et al., 1975; Danka, 2005; Liu et al., 2005; Muhammad et al., 2020). Keshlaf (2009) reported that this behavior results from the chemical composition or texture of cotton pollen, which makes it less attractive to bees. This highlights a critical behavioral dichotomy in which cotton is a nectar source but not a preferred pollen source, a distinction that must be considered in pollination management plans. The low collection of cotton pollen could be due to genuine disinterest (e.g., poor nutritional quality, difficult morphology) or could be influenced by the high relative abundance and proximity of more attractive species in the surrounding landscape, a factor not quantified in this study. This finding is consistent with earlier observations in Australian cotton agroecosystems where honey bees showed limited pollen collection from cotton despite its availability (Keshlaf, 2009). Management and policy implicationsThe insights from this study have direct applications in sustainable agriculture. To enhance cotton pollination and support the health of honey bees. Floral resource planningFarmers and land managers should maintain, or plant preferred alternative pollen sources (e.g., native shrubs like Acacia) near cotton fields. This provides bees with essential nutrition, which can strengthen colonies and potentially improve their overall pollination activity. Colony placement and timingBeekeepers contracting hives for cotton pollination should ensure that colonies are strong and placed in landscapes with diverse floral resources to meet their nutritional needs, as cotton alone is insufficient. Coordinating hive placement with the peak flowering of cotton and key alternative pollen sources could maximize foraging efficiency. Habitat conservationAgricultural policy should encourage the conservation of natural vegetation patches within farming landscapes to sustain pollinator populations throughout the season, thereby reducing the reliance on a single, nutritionally inadequate crop resource. ConclusionThese findings underscore the complexity of the foraging behavior of honey bees, which is shaped by both floral and pollen characteristics. A key observation is floral competition, where more attractive alternative flowers divert bees from crops like cotton for pollen collection, although cotton remains a nectar source. Managing floral landscapes by conserving native flora or strategically planting bee-preferred species could optimize pollination efficiency and support colony health. This study establishes that a combination of attractive floral displays (color, morphology) and collectible pollen traits (small size, smooth texture) drives honey bee pollen foraging in Australian cotton systems, with cotton being a low-preference pollen source. By applying these timeless ecological principles—ensuring access to diverse pollen resources—farmers and beekeepers can work together to sustain healthy honey bee populations for effective crop pollination. This approach is consistent with sustainable beekeeping practices that emphasize the importance of maintaining diverse floral resources for colony health and productivity (Keshlaf, 2017). The data from 2007 serve as a valuable baseline for understanding these fundamental relationships. AcknowledgmentsThe authors would like to thank Dr. Robert Mensah for his invaluable comments and suggestions on this article. The authors are deeply grateful to the staff at the University of Western Sydney (UWS), particularly Oleg Nicetic for his guidance and statistical analysis, and Liz Kabanoff for her assistance in providing SEM images of the pollen. Special thanks to the Australian Cotton Research Institute (ACRI) for granting access to their cotton fields and research facilities, which were essential for this study. Thanks to Mr. Warren Jones, who generously provided all the beehives and equipment required during the research period. Conflict of interestThe authors declare no conflict of interest. FundingThis study received no external funding. Authors' contributionsThe authors have contributed to this study. All authors have read and approved the final version of the manuscript. Data availabilityAll data are provided in the revised manuscript. ReferencesBailey, S., Requier, F., Nusillard, B., Roberts, S.P., Potts, S.G. and Bouget, C. 2022. Distance from forest edge affects bee pollinators in oilseed rape fields. Ecol. Evol. 12, 8625. Brunet, J., Zhao, Y. and Clayton, M.K. 2015. Linking pollinator visitation rate and pollen receipt: the importance of plant and floral traits. Am. J. Bot. 102(3), 385–395; doi:10.1016/j.ajb.2013.03.010 Buchmann, S.L. and Nabhan, G.P. 1996. The forgotten pollinators. Washington, D.C: Island Press. Buchmann, S.L. and Shipman, K. 1990. Flower color and its role in pollinator behavior. Annu. Rev. Ecol. Syst. 21(1), 207–233. Chittka, L. and Raine, N.E. 2006. Recognition of flowers by pollinators. Curr. Opin. Plant Biol. 9(4), 428–435. Chittka, L. and Waser. 1997. Why red flowers are not invisible to bees. Proc. R. Soc. B. 264(1388), 461–466. Chittka, L. and Thomson, J.D. 2012. Cognitive ecology of pollination. Cambridge, UK: Cambridge University Press. . Chittka, L., Shmida, J. and Menzel, R. 1999. Flower constancy in honey bees: the importance of colors in learning. Insects Soc. 46(3), 223–230. Cook, S.M., Awmack, C.S., Murray, D.A. and Williams, I.H. 2003. Are honey bees’ foraging preferences affected by pollen amino acid composition?. Ecological Entomol. 28(5), 622–627. Corbet, A., Williams, M. and Kearns, T. 1995. Pollinator visits to different types of flowers: the effect of flower size. Ecol. Lett. 8(3), 271–276. Dafni, A. 1992. Pollination ecology: a practical approach. Oxford, UK: Oxford University Press. Danka, R.G. 2005. High levels of cotton pollen collection observed for honey bees (Hymenoptera: apidae) in south-central Louisiana. J. Entomol. Sci. 40(3), 316–326. Dimou, D. and Thrasyvoulou, A. 2007. Pollen foraging preferences of honey bees in Mediterranean environments. Bee. World. 88(1), 16–20. Eckert, E. 1942. Pollen collection by honey bees. J. Econ. Entomol. 35(3), 439–443. Erdtman, G. 1969. Handbook of Palynology: morphology, Taxonomy, and Ecol. New York, NY: Hafner Publishing Co. Forrest, J.R. and Thomson, J.D. 2019. Pollination and plant phenology. In Phenology: An Integrative Environmental Science. Eds., Lipow, S.R. and Wyatt, R. Cham, Switzerland: Springer, pp: 439–456. Free, J.B. 1993. Insect pollination of crops. 2nd ed. London, UK: Academic Press. Ghosh, S. and Chung-Chung, J. 2017. Pollen quality and its impact on the development of honey bee colonies. Insect. Physiol. 104, 68–75. Goodwin, R. and Perry, J. 1992. Flower clustering and pollinator visitation: a study of honey bee foraging behavior. Ecol. Entomol. 17, 75–83. Goodwin, R.M. 1986. Bee pollination efficiency in kiwifruit orchards with different arrangement systems. N. Z. J. Bot. 24(3), 401–407. Goulson, D., Nicholls, E., Botías, C. and Rotheray, E.L. 2008. Bee declines and the consequences for pollination. Conserv 141(10), 2422–2436. Goulson, D., Nicholls, E., Botías, C. and Rotheray, E.L. 2015. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 347, 1255957. Harder, D. 1990. Pollen foraging by honey bees: quantifying worker bee foraging efficiency. Insect. Behav. 9(5), 563–571. Johnson, S.D. and Ackerman, J.D. 1989. Pollination and partitioning of floral resources among three co-occurring plant species. Oecologia 81(3), 345–350. Kearns, C.A. and Inouye, D.W. 1993. Techniques for Pollination Biology. Denver, CO: University Press of Colorado. Keller, L., Blüthgen, S. and Boehm, M. 2005. Pollen and nectar preferences of honey bees: implications for foraging decisions. Apidologie 36(3), 263–274. Keshlaf, M. 2009. Assessment of Honeybee Foraging Activity and Pollination in Australian Bt Cotton PhD dissertation, University of Western Sydney, Sydney, Australia. Keshlaf, M. 2017. The past and present status of beekeeping in Libya. J. Apicultural Res. 56(3), 190–195. Keshlaf, M., Spooner-Hart, R. and Mensah, R. Honeybee foraging activity and pollination efficacy in Australian Bt cotton. In: Proceedings of the XXIII International Congress of Entomology. Daegu, South Korea, Published by the Entomological Society of America, 2012. Keshlaf, M., Spooner-Hart, R. and Mirwan, A.H. 2025. Role of pollen beetles (Carpophilus aterrimus: nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae). Open Vet. J. 15(10), 5069–5077. Kevan, P. and Backhaus, W. 1998. Pollinator behavior and the role of flower color. Pollin. Ecol. 2, 87–93. Lawson, S.P., Nicholls, E. and Wright, G.A. 2022. Pollen nutritional content predicts foraging preferences in honey bees. Insect Physiol 140:104408. Leonhardt, S. and Blüthgen, S. 2012. Specialization in bee foraging behavior: ecological and evolutionary implications. Ecol. Lett. 15(9), 1053–1064. Liu, B., Xu, C., Yan, F. and Gong, R. 2005. Impacts of the pollen of insect-resistant transgenic cotton on honeybees. Biodivers. Conserv. 14, 3487–3496. Loper, G. and Berdel, D. 1980. The Nutritional Requirements of Honey Bee Brood: a Review. J. Apic. Res. 19(4), 212–220. Moffett, J.O., Stith, L.S., Burkhart, C.C. and Shipman, C.W. 1975. Honey bee visits to cotton flowers. Environ. Entomol. 4(2), 203–206. Muhammad, W., Ahmad, M. and Ahmad, I. 2020. Pollination behavior and management of cotton crops. In Cotton production and uses: agronomy, crop protection, and postharvest technologies. Eds., Ahmad, S. and Hasanuzzaman, M. Singapore: Springer Nature, pp: 163–175; doi: 10.1007/978-981-15-1472-2_9 Olsen, W.C., Nye, W.P. and Hepworth, G.W. 1979. Relationship of honey bee foraging to pollen availability in alfalfa. Environ. Entomol. 8(3), 278–281. Pacini, E. and Hesse, M. 2005. Pollen morphology and its relationship with pollinator preferences. Plant Biol. 7(4), 313–322. Pattrick, J.G. 2013. The role of nectar quality in bee foraging decisions. J. Exp. Biol. 216(4), 623–631; doi: 10.1242/jeb.078568. Pattrick, J.G., Doering, G.N. and Whitney, H.M. 2024. Bees evaluate nectar quality through integrated sugar concentration and viscosity assessments. Anim. Behav. 207, 33–45. Percival, M. 1955. Pollen-foraging behavior in honey bees: the role of flowering time. J. Apic. Res. 17(4), 347–356. Pernal, S.F. and Currie, R.W. 2002. Discrimination and preferences for pollen-based cues by foraging honeybees, Apis mellifera L. Anim. Behav. 63(2), 369–390. Raguso, R.A. 2008. Wake up and smell the roses: ecology and evolution of floral scent. Annu. Rev. Ecol. Evol. Syst. 39, 549–569. Rashad, M. and Parker, G. 1958. Identification of pollen grains using light microscopy. J. Econ. Entomol. 51(5), 678–681. Roulston, T.H. and Cane, J.H. 2000a. Nutritional content and digestibility of pollen in animals. Plant. Syst. 222(1-4), 187–209. Roulston, T.H., Freeman, M. and Willis, L. 2000b. Pollen foraging by honey bees: effects of flower size and pollen availability. Insectes. Soc. 47(1), 20–28. Roulston, T.H. and Goodell, K. 2011. The influence of pollen quality and quantity on honey bee foraging behavior. Insect. Behav. 24(4), 237–245. Ruedenauer, F.A., Leonhardt, S.D. and Spaethe, J. 2024. Nutrient balancing regulates bee pollen collection strategies. Funct. Ecol. 38(2), 256–268. Ruedenauer, F.A., Spaethe, J. and Leonhardt, S.D. 2021. Bees sense nutrients in pollen: the interplay of taste, texture, and chemistry. Proc. Roy. Soc. B. 288, 1794. Ruedenauer, F.A., Spaethe, J., Van Der Kooi, C.J. and Leonhardt, S.D. 2019. Pollinator or pedigree: which factors determine pollen nutrient evolution?. Oecologia 191, 349–358. Seeley, T. 1986. The foraging behavior of honey bees: a comparative analysis. Ecology 67(5), 1164–1173. Synge, H. 1947. The pollen collection of honey bees. J. Agric. Sci. 37(2), 163–174. Taber, S. 1977. A new design for a honey bee pollen trap. Bee. J. 117, 484–485. Thomson, J.D. 2004. Flower color and pollination preferences: a meta-analysis. Annu. Rev. Ecol. Evol. Syst. 35, 89–111. Todd, A. and Bishop, R. 1940. Role of pollen in honey bee nutrition. J. Econ. Entomol. 33(5), 726–731. Vaissière, B.E. 1991. Variations in honeybee colonies’ pollen collecting activity in relation to floral resources. Apidologie 22(4), 419–430. Vaudo, A.D., Patch, H.M., Mortensen, D.A., Tooker, J.F. and Grozinger, C.M. 2020. Nutritional ecology of pollen: protein–lipid ratios drive foraging preferences of bumblebees Current Opinion in Insect Science, 38, 33–40. Vaudo, A.D., Stabler, D., Patch, H.M. and Grozinger, C.M. 2023. Mechanisms of pollen selection: integrating nutrition, floral traits, and bee physiology. Annu. Rev. Entomol. 68, 235–255. Waser, N.M. 1986. Flower constancy: definition, cause, and measurement. Am. Nat. 127(5), 593–603; doi: 10.1086/284507 | ||
| How to Cite this Article |
| Pubmed Style Keshlaf MM, Mitsho F, Spooner-hart R. Pollen preferences of honey bees: How do floral and pollen characteristics influence foraging strategies?. Open Vet. J.. 2026; 16(3): 1695-1703. doi:10.5455/OVJ.2026.v16.i3.27 Web Style Keshlaf MM, Mitsho F, Spooner-hart R. Pollen preferences of honey bees: How do floral and pollen characteristics influence foraging strategies?. https://www.openveterinaryjournal.com/?mno=289848 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.27 AMA (American Medical Association) Style Keshlaf MM, Mitsho F, Spooner-hart R. Pollen preferences of honey bees: How do floral and pollen characteristics influence foraging strategies?. Open Vet. J.. 2026; 16(3): 1695-1703. doi:10.5455/OVJ.2026.v16.i3.27 Vancouver/ICMJE Style Keshlaf MM, Mitsho F, Spooner-hart R. Pollen preferences of honey bees: How do floral and pollen characteristics influence foraging strategies?. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1695-1703. doi:10.5455/OVJ.2026.v16.i3.27 Harvard Style Keshlaf, M. M., Mitsho, . F. & Spooner-hart, . R. (2026) Pollen preferences of honey bees: How do floral and pollen characteristics influence foraging strategies?. Open Vet. J., 16 (3), 1695-1703. doi:10.5455/OVJ.2026.v16.i3.27 Turabian Style Keshlaf, Marwan M., Farid Mitsho, and Robert Spooner-hart. 2026. Pollen preferences of honey bees: How do floral and pollen characteristics influence foraging strategies?. Open Veterinary Journal, 16 (3), 1695-1703. doi:10.5455/OVJ.2026.v16.i3.27 Chicago Style Keshlaf, Marwan M., Farid Mitsho, and Robert Spooner-hart. "Pollen preferences of honey bees: How do floral and pollen characteristics influence foraging strategies?." Open Veterinary Journal 16 (2026), 1695-1703. doi:10.5455/OVJ.2026.v16.i3.27 MLA (The Modern Language Association) Style Keshlaf, Marwan M., Farid Mitsho, and Robert Spooner-hart. "Pollen preferences of honey bees: How do floral and pollen characteristics influence foraging strategies?." Open Veterinary Journal 16.3 (2026), 1695-1703. Print. doi:10.5455/OVJ.2026.v16.i3.27 APA (American Psychological Association) Style Keshlaf, M. M., Mitsho, . F. & Spooner-hart, . R. (2026) Pollen preferences of honey bees: How do floral and pollen characteristics influence foraging strategies?. Open Veterinary Journal, 16 (3), 1695-1703. doi:10.5455/OVJ.2026.v16.i3.27 |