Open Veterinary Journal, (2026), Vol. 16(3): 1716-1724
Research Article
10.5455/OVJ.2026.v16.i3.29
Comparative analysis of delayed ovulation and associated ovarian patterns in the Ongole crossbred cattle
Herdis Herdis1, Langgeng Priyanto2, Santoso Santoso1, Fitra Aji Pamungkas1,
Rahma Isartina Anwar1, Desiana Ade Mahari1, Florentina Bety Indah Lupitasari1, Nurul Azizah1,
Anita Hafid1, Reski Amalia3, Ligaya Ita Tumbelaka4, Amrozi Amrozi4, and Pradita Iustitia Sitaresmi1*
1Research Center for Animal Husbandry, National Research and Innovation Agency,
Cibinong Science Center, Bogor, Indonesia
2Department of Animal Science, Faculty of Agriculture, Sriwijaya University, South Sumatra, Indonesia
3Department of Physiology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia
4Department of Veterinary Clinic, Reproduction and Pathology, School of Veterinary Medicine and Biomedical Science, IPB University, Bogor, Indonesia
*Corresponding Author: Pradita Iustitia Sitaresmi. Research Center for Animal Husbandry, National Research and Innovation Agency, Cibinong Science Center, Bogor, Indonesia. Email: pradita.iustitia.sitaresmi [at] brin.go.id
Submitted: 23/10/2025 Revised: 10/02/2026 Accepted: 20/02/2026 Published: 31/03/2026
© 2026 Open Veterinary Journal
This is an Open Access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited, and is not altered, transformed, or built upon in any way.
ABSTRACT
Background: Delayed ovulation can have harmful effects on cattle reproduction, especially in countries that rely on artificial insemination, resulting in significant economic losses, as evidenced by Indonesia.
Aim: This study provides novel insights into hormone dynamics and ovarian environment comparisons in delayed ovulation (DO) and Normal cows (NO) cows.
Methods: Eight Ongole-crossbred cows with DO or NO over four estrous cycles were observed. Daily ultrasonography was used to monitor dominant follicle size and corpus luteum (CL), while estrus behavior and blood sampling were used to record peripheral reproductive hormone patterns from 2 days before to 6 days after estrus onset. Statistical comparisons between the DO and NO groups were performed using the SPSS 26 software, with significance set at p < 0.05.
Results: The results showed that DO cows had similar cycle durations but ovulated later than NO cows. Furthermore, DO cows had significantly smaller follicles (1.14 ± 0.2 cm) in the pre-estrus phase, larger follicles at estrus onset, and a larger CL during pre-estrus that should have regressed. Elevated progesterone levels (2.09 ± 0.34 ng/ml) before and after estrous treatment inhibited luteinizing hormone (LH) release, thereby delaying ovulation. DO cows exhibited lower LH levels and LH surges 2–4 days after the onset of estrus, altering the ovarian environment and hindering oocyte maturation.
Conclusion: Delayed ovulation in cows is associated with hormonal and ovarian changes that reduce fertility, highlighting the need for further research on progesterone and hypothalamic-pituitary regulation to develop targeted fertility improvement strategies.
Keywords: Crossbred Ongole cows, Hormonal pattern, Prolonged estrus, Suprabasal progesterone.
Introduction
The rising demand for beef products contributes to an increase in cattle profitability. Furthermore, milk production success relies on proficient reproductive management. Reproductive failure can cost up to 25% of the economic loss due to reduced calf production and lactating cow numbers (Boyer et al., 2020). Delayed ovulation patterns may often result in anovulation or fertilization failure, leading to repeated breeding attempts in cows (Pérez-Marín and Quintela, 2023). Normally, ovulation occurs 26–28 hours after the onset of estrus in cows (Klabnik and Horn, 2023). However, in delayed ovulation (DO) cows, the sustained release of PGF2α causes follicle retention and leads to delayed ovulation (Al-Timimi et al., 2021). DO cows require 36–48 hours post-onset estrus before ovulation (Al-Timimi et al., 2021). Interestingly, despite delayed ovulation time, DO cows maintained a normal estrous cycle with a clinically normal reproductive tract. Accurate detection of mating and ovulation in cows is crucial in tropical countries, such as Indonesia, where artificial insemination (AI) is used for reproductive management. Although rare, delayed ovulation is becoming increasingly common in crossbred beef cattle, which may increase recurrent breeding in national AI projects (Klabnik and Horn, 2023), resulting in delayed gestation 2–5 months later.
Understanding hormonal patterns and follicle or corpus luteum (CL) anatomy is necessary for controlling delayed ovulation. A previous study has shown that ovulation can be delayed if there is an increase in basal progesterone levels during the follicular phase (>2 ng/ml in blood) or if the large CL takes longer to regress (>2.5 days). This condition causes abnormal progesterone release (Pérez-Marín and Quintela, 2023). High blood progesterone levels inhibit or downregulate luteinizing hormone (LH) receptors, thereby preventing ovulation (Dorfman et al., 2013). Ovarian dysfunction, characterized by repeated follicular waves that inhibit the development of large follicles (> 1 cm) or ovarian cysts, can result in delayed ovulation. Tropical cattle are more likely to have delayed ovulation due to crossbreeding between Bos taurus and local cattle, heat stress, and inadequate feed intake (Deka et al., 2021).
Timely ovulation requires specific hormonal patterns and their interactions. LH and follicle-stimulating hormone (FSH) initiate ovulation via pulsatile and surging processes, respectively. However, estrogen, progesterone, and inhibin have both positive and negative effects on ovulation (Pérez-Marín and Quintela, 2023). Estrogen typically exerts a positive feedback effect by promoting the LH surge necessary for ovulation, but it can also inhibit gonadotropin release through negative feedback. Similarly, progesterone and inhibin generally suppress gonadotropin secretion to regulate ovulation timing, although their effects vary depending on the cycle phase. This study aimed to compare reproductive hormone patterns, follicular development, and CL activity in Indonesian native crossbred dairy cows with normal and delayed ovulation. This study aims to provide information that can be used in intervention methods in handling delayed ovulation cases and improving fertility and reproductive management in small-scale farming systems.
Materials and Methods
Identification of animal cases
Four of the eight local crossbred cows included in this study had DO based on medical records for 3–5 cycles. The researchers used a case study survey, including the Assisted Reproductive Techniques report and cattle breeder interviews, to determine whether the cows had repeated breeding incidents and obtained records (estrous cycle length, estrous duration, ovulation time, service per conception, and pregnancy rates). All cows aged 4–8 years and parity 2–4 underwent clinical verification to ensure good health regardless of their reproductive history. Their BCS were 3–4 on a scale of 1–5. Three consecutive cycles of DO in cows showed clinical symptoms lasting up to 4 days (>36 hours). Daily ultrasonographic examination of the ovary confirmed this diagnosis. These cows had ongoing estrus symptoms with normal cycles (20–22 days) and reproductive tract abnormalities (Honparkhe et al., 2010). Each cow was monitored during two reproductive cycles over 60 days. Daily observations tracked the cycle from 2 days before to 6 days after the onset of estrus. This study intentionally omitted synchronization to maintain data integrity and reduce reproductive hormone induction bias.
Identification of estrus and ovulation
Visual observation was used to record estrus signs from the onset to discharge, and the onset characteristics were classified every 6 hours. Ultrasound simultaneously detects ovulation (Tiro et al., 2020). Estrus onset is defined as the first observable expression of estrous behavior (heat signs) recorded by visual observation at 6-hour intervals from the initial detection of standing heat or other estrous indicators. Estrus end is defined as the cessation or discharge of estrous behavioral signs, as determined by continuous visual monitoring until the last occurrence of heat expression. Daily transrectal ultrasonography is used to confirm ovulation, which detects the disappearance or collapse of the dominant follicle previously identified on the ovary. Ultrasonographic examinations are conducted once daily, starting 2 days before estrus onset and continuing until 6 days after estrus onset, allowing precise identification of the timing of follicle rupture indicative of ovulation. The criteria for ovulation confirmation include the disappearance or collapse of the dominant follicle on ultrasound images. Ovarian scanning is performed daily using a 1–10 MHz rectal probe, with follicle and CL diameters taken each day from 2 days before to 6 days after estrus onset. The main method to determine estrus was to monitor the expression of heat. This method was chosen to improve data relevance and reliability.
Observation of follicular dynamics and measurement of corpus luteum size
Daily observations were made using a 3,000 WD ultrasound (USG, China) with a rectal probe of 1–10 Mhz (5 MHz). This approach, modified from previous research, uses repeated ovarian surface examinations to image the CL and follicles. The number and diameter of antral follicles and CL diameter in cow ovaries were measured using ultrasound (BMV Technology, China). Follicle and CL diameters were the distances (in cm) between two axis points along the longest axis of the follicle or CL. Small follicles (0.3–0.5 cm) were assigned to class I, medium-sized follicles (0.6–0.9 cm) to class II, and large follicles (> 1 cm) to class III. This categorization assisted in monitoring their development over a 21-day estrous cycle (Lucy et al., 2001).
Blood sampling and preparation of serum
Blood was drawn from the jugular vein with an EDTA tube (OneMed, Indonesia) every day, 2 days before and 6 days after onset (Kim et al., 2023). To obtain serum, the blood was left at 4°C for 30–60 minute. Following serum collection, 2 ml of serum was transferred into microtubes (Axigen, USA) and subsequently stored at −20°C until further analysis (Sitaresmi et al., 2020).
Hormone analysis
Commercial enzyme-linked immunosorbent assay (ELISA) kits (Bovine) forFSH, LH, and progesterone (DRG, Germany) were used to determine hormonal concentrations. Microplate analysis was performed using a Microplate MP96 UV® ELISA reader (Safas, Monaco, Monaco). For the hormone assays, 0.3 ml of serum was used. Following the addition of serum to the ELISA plate, a base solution, antibody, and substrate liquid were sequentially introduced into the wells. The wells were then covered with a stop solution and allowed to stand during the assay. An enzyme-linked immunosorbent assay reader was used to analyze the samples (Sitaresmi et al., 2020). Data tabulation and curve analysis of the ELISA results were conducted using the PRISM 9 software.
Statistical analysis
The data in this study were classified as before (−2 to 0 days) onset of estrus and after onset estrus 1–6 days after estrus. This study used statistical analysis to compare the parameter values in DO and NO cows. The study compared single-factor data using an independent-sample t-test. SPSS version 26.0 was used for statistical analysis. An independent t-test was used to illustrate the average values and standard deviations (± SD) for all parameters, with a significance threshold of p < 0.05.
Ethical approval
The Animal Care and Use Committee of the BRIN (Badan Riset dan Inovasi National) approved all experimental procedures with cows under protocol number 065/KE.02/SK/10/2022.
Results
Identification of estrus and ovulation
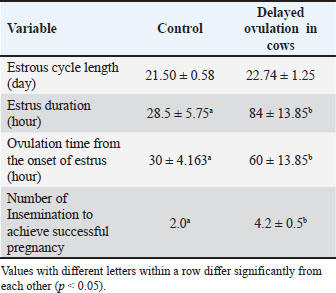
For estrus detection, cows with onset characteristics were classified every 6 hours. Both NO and DO cows had the same estrous cycle duration of 20–25 days. Variations in estrus duration, ovulation time, and insemination per gestation were observed. The data showed that DO cows had significantly higher values (p < 0.05) than controls NO for all parameters (Table 1). During pre-onset estrus, DO cows had significantly smaller follicle diameters (1.34–1.44 cm) than Normal cows (NO) cows (p < 0.05), but instead experienced enlargement after the onset of estrus, which should have undergone lysis due to the ovulatory process (Fig. 1).
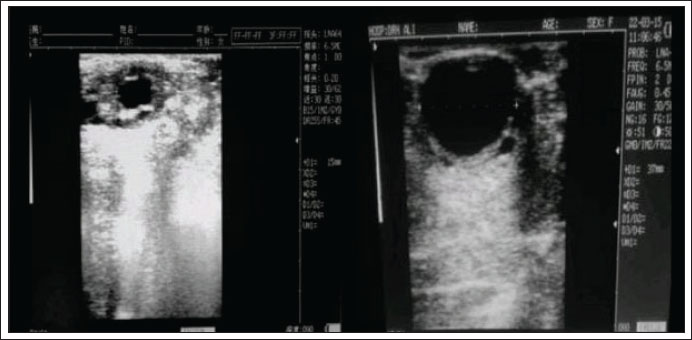
Fig. 1. Diameter size of the de Graaf follicle in DO and NO cows This figure illustrates a comparison of follicle diameters between normal cows (left) and those with D-0 estrus (right) at the onset of estrus. The Graafian follicle diameter in normal cows is 1.5 cm, which is significantly smaller than that in cows experiencing delayed ovulation, where it exceeds 3 cm.
Table 1. Comparison of the reproductive cycle monitoring of Ongole crossbreed cows with and without delayed ovulation.
FDD and CL measurements
In contrast, the CL sizes of DO cows were higher than those of NO cows, particularly in the pre-estrus. Table 2 shows no significant differences in follicle size or CL at post-onset estrus. Interestingly, DO cows maintained a follicular development pattern until days 3–4 after onset-estrous onset (Figs. 1 and 2), whereas the follicle size of NO cows shrank and even disappeared near estrous onset (day 0). Moreover, in DO cows, the CL did not directly undergo luteolysis at the onset of estrus and remained for several days, in contrast to NO cows, where the CL had been lysed a while before the onset of estrus. Furthermore, a few days from d-5 to d-6 after ovulation, both DO and NO cows had similar CL sizes (Table 2 and Fig. 2).
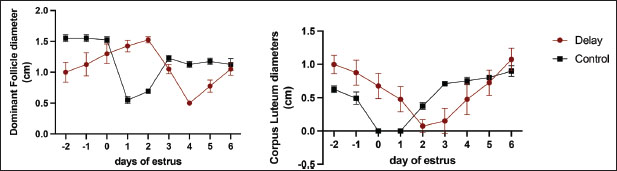
Fig. 2. Comparative analysis of ovarian size, focusing on the dimensions of follicles and the corpus luteum, in cows exhibiting ovulation delay versus normal cows. Measurements were taken at multiple reproductive stages: before estrus, at the onset of estrus, and throughout the luteal phase. This figure illustrates the differences in ovarian morphology associated with ovulation timing, highlighting potential physiological variations between the two groups across the estrous cycle phases.
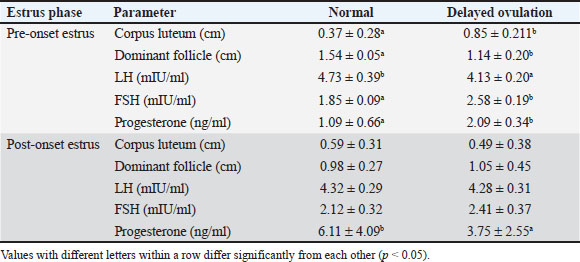
Table 2. Comparison of the diameter of the dominant follicle and corpus luteum and reproductive hormone concentration of Ongole crossbreed cows with and without delayed ovulation.
Hormone analysis
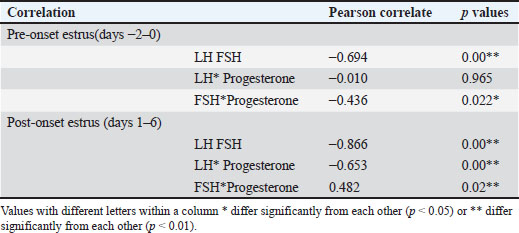
Hormonal analysis revealed that DO cows exhibited significantly lower LH levels during the pre-onset estrus phase compared with NO cows (4.13 ± 0.20 vs. 4.73 ± 0.39 mIU/ml, p < 0.05) (Table 2). This finding supports the role of elevated SBP in suppressing LH pulsatility and delaying the LH surge critical for ovulation. In contrast, FSH levels were higher in DO cows during pre-estrus (2.58 ± 0.19 vs. 1.85 ± 0.09 mIU/ml, p < 0.05), indicating that progesterone does not directly inhibit FSH secretion. Instead, the elevated FSH likely reflects a compensatory response to reduced negative feedback from estradiol due to smaller dominant follicles in DO cows. LH concentrations did not differ significantly between groups, while progesterone remained elevated longer in DO cows, consistent with delayed luteolysis and follicular dynamics.
Correlation analysis further demonstrated a strong negative relationship between LH and FSH levels during pre-onset estrus (r=−0.694, p=0.00), underscoring the differential regulation of these gonadotropins during DOO (Table 3). Progesterone was negatively correlated with FSH pre-estrus (r=−0.436, p=0.022), suggesting complex feedback interactions modulating FSH levels indirectly through estrogen rather than direct progesterone inhibition. These hormonal patterns align with ultrasound observations of smaller dominant follicles and larger retained corpora lutea in DO cows during pre-estrus, supporting the endocrine environment characterized by suprabasal progesterone suppressing LH release timing and amplitude, while FSH rises as a compensatory mechanism to altered estrogen feedback.
Table 3. Correlation of the reproductive hormonal pattern of Ongole crossbreed cows with and without delayed ovulation.
Discussion
Delayed ovulation is defined as ovulation occurring considerably later than the initiation of estrus in livestock (López-Gatius et al., 2005). The study highlighted that the postponement of LH surges, which is linked to heightened levels of suprabasal progesterone, leads to delayed ovulation. Notably, this condition occurs without clinical abnormalities, such as ovarian or CL cysts. The data from the current study support these explanations. Over three cycles, DO cows had a cycle length of 21–23 days, similar to that of normal cows (Table 1). Similar findings have been reported in previous studies (Das et al., 2009). This study also found that DO cows had a longer estrous duration and delayed ovulation time (p < 0.05) than normal cows, but still had the same estrous cycle, which is in accordance with previous research on Simmental cattle (Singh et al., 2012) and crossbred cows (Das et al., 2009). Cows with delayed ovulation had a longer estrous duration (52–79 hours) than normal cows (<36 hours). The causes of ovulation delay remain uncertain. A conceivable explanation is that suprabasal progesterone secretion (> 1 ng/ml) approached the estrous time, which provided negative feedback on the LH surge stimulation of the cow’s ovary (Medeiros et al., 2021). However, this explanation must be compared with comparable ovulation studies to determine whether similar results have been obtained. Delaying ovulation reduces reproductive efficiency by 35% and causes repeat breeding in beef cattle (Cardoso Consentini et al., 2021). These findings are corroborated by the AI records within this study, indicating that S/C DO cows require 4–5 insemination. This requirement arises from the delayed ovulation observed in post-heat estrus cows (>36 hours), which can lead to inaccurate determination of the optimal timing for AI and ovulation. Consequently, the inseminated sperm is rendered nonviable (as sperm in the reproductive tract only survive for 12–24 hours) before ovulation in DO cows, resulting in fertilization failure. This necessitates re-insemination and contributes to a high service per conception (S/C) (SPC) rate.
Under normal conditions, the dominant follicle is >0.8 cm and produces a large number of liquor folliculi, especially during the proestrus phase (day −2 to day 0 before the onset of estrus) (Pfeifer et al., 2012). Estrous expression begins with a massive increase in estrogen levels. This study showed normal estrogen hormone levels, indicating that both groups could express estrus. Cows with delayed ovulation before the onset of estrus have smaller dominant follicles (<1 cm) but still have a larger CL, leading to poor reproductive performance. Previous research has indicated that cows with follicle sizes <12 mm in the proestrus have a 50% lower fertilization potential than those with follicle sizes of 13–15 mm (de Lima et al., 2020), which was also apparent in the current study (S/C >4). Smaller follicles led to reduced CL size and PH secretion. This can lead to unsuccessful embryo implantation and fertilization within the uterus. This is in line with the findings of the current study, which found that DO cows had slightly lower CL size and progesterone hormone concentration than normal cows in post-onset estrus (day 1–6), Progesterone level was significantly different but CL size was not. The lower level of progesterone in DO could have been due to the regressing Old CL that was retained, whereas the higher progesterone level in NO could be due to the newly formed CL after ovulation. Additionally, smaller dominant follicles may have lower LH receptor activity, leading to a delay in ovulation because the LH surge takes longer (Xie et al., 2023). This correlation was strengthened and clarified in the subsequent hormone data analysis (Table 3).
DO cows had unusually extended dominant follicles on day −2 to +6 during the estrous cycle, as observed by ultrasound. This dominant follicle shrinks near the onset of estrus, followed by ovulation 9–10 hours later (Miyamoto and Shirasuna, 2009). In this study, the dominant follicle remained stable in DO cows and increased shortly after the onset of estrus until the second day. Moreover, one out of four cows with delayed ovulation had a follicle size of up to 3.7 cm after estrus (Fig. 3), which shrank on days 3–4 after estrus, indicating that ovulation occurred 3–4 days later. In a previous study, the CL exhibited a discernible pattern throughout the proestrus phase. The CL diminishes in size during the progression toward the estrous phase, propelled by prostaglandin hormones, approximately 17–18 days after estrus, which concludes the diestrus phase (Trevisol et al., 2020). Remarkably, the CL of cows with delayed ovulation was larger than that of normal cows. In earlier studies on ovulation delay, the CL released suprabasal progesterone during the follicular phase (Friis Wang et al., 2019). This hormone decreases FSH and LH secretion, resulting in smaller dominant follicles and delayed ovulation (Wiltbank et al., 2018). Vascular endothelial growth factor A (VEGFA) activity in the CL may influence this phenomenon (Fraser et al., 2000), which supports the findings of the present study (Table 2). Insufficient prostaglandin pulses or less sensitive CL receptors may also cause this delay (Pate and Hughes, 2023). A simpler explanation is that there may be a problem with the natural shrinkage of the CL in delayed estrus cows. Whether this is due to a low pulse of prostaglandin or insensitive receptors could help explain this delay. Another potential explanation is the insensitivity of luteolysis accelerators, including endothein-1, angiotensin II, oxytocin, and luteolysis-related genes such as Steroidogenic Acute Regulatory protein (STAR), VEGFA, Luteinizing Hormone/Choriogonadotropin Receptor (LHCGR), and Fas Ligand (FAS-L), found in the CL (Pfeifer et al., 2012). However, this explanation should be validated in future studies. A normal dominant follicle has a diameter of >0.8 cm and accumulates follicular fluid, especially during the proestrus phase (day 2 to day 0 before estrus) (Pfeifer et al., 2012). This significantly increases estrogen hormone levels and initiates estrous. This study predicted normal estrogen hormone levels, indicating that both groups could express estrus.
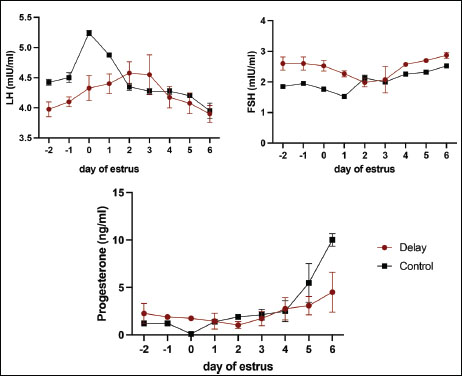
Fig. 3. Hormonal profiles in cows with ovulation delay (DO) compared with normal ovulating cows (NO) measured before estrus, at the onset of estrus, and throughout the luteal phase. This figure depicts the temporal changes in key reproductive hormones, such as progesterone and estradiol, highlighting the distinctive hormonal dynamics associated with the timing of ovulation. Differences between the DO and NO groups indicate altered endocrine regulation in delayed ovulation, which may explain the observed variations in ovarian function and follicular development across the estrous cycle stages.
In this study, the reduced and prolonged LH pulse in cows with delayed ovulation was comparable to previous findings in high-productivity cows with delayed ovulation. This hormonal pattern showed that the pre-ovulatory LH amplitude remained the same for many days following the expression of estrus. Three days later, the cows were spiked with a much lower average LH pulse than that of the normal cows. Delay ovulation causes cows to release less estrogen because of fewer follicles before estrous expression. Initiating an LH surge at the right time and increasing levels becomes difficult. Multiple studies have demonstrated that insufficient energy intake can be attributed to the inability or delay of LH surge. Hypothalamic Insulin growth factor-1 (IGF-1) receptor insensitivity also stops ovulation by inhibiting LH surge (Endo, 2022). IGF-binding proteins (IGFBP-2, IGFBP-3, and IGFBP-5) in the pituitary gland fluctuate with cow diet and affect LH secretion. Further studies are required to confirm this association. LH and FSH levels were negatively correlated (p−0.694) throughout the follicular phase. Higher FSH levels were expected in ovulation-delayed cows before estrus to counteract the negative feedback of the hypothalamus by estrogen from the dominant follicle. Small follicles in cows with delayed ovulation indicate that estrogen does not trigger a feedback loop. Follicle development continues past estrus due to the inability of the ovary to generate negative feedback (Borş and Borş, 2020). This was observed in elevated FSH levels near estrus, similar to the results of this study (Fig. 2).
Progesterone levels derived from the CL are integral to the regulation of the hypothalamus-pituitary-gonadal axis (Abreu et al., 2018). A significant portion of the effects of progesterone is mediated through endometrial histotroph secretions, which impact embryonic and fetal growth and developmental biology, or through gonadotropin secretion, which affects follicular function and oocyte competence (Borş and Borş, 2020). Furthermore, endometrial histotroph secretion may modify estrous patterns in animals. During delayed ovulation, progesterone hormones increase throughout the follicular phase, delaying ovulation, aging the oocyte, and affecting fertilization (Di Nisio et al., 2022).
After ovulation, the follicle anatomically becomes the CL to increase the progesterone levels during gestation and implantation. However, in delayed ovulation, cows increase progesterone (suprabasal progesterone) from proestrus to estrus and inhibit the pituitary gland, which in turn suppresses FSH and LH production, resulting in smaller follicles during proestrus. Smaller follicles, which extend the estrous phase, reduce the capability of hypothalamic LH receptors. Subsequently, this inhibition leads to a decreased release of the LH surge, ultimately causing a delayed ovulation (Duncan, 2021). This study supports all the aforementioned assertions. Previous studies have shown that cows with irregular ovulation have significantly higher suprabasal progesterone levels than normal cows during the follicular phase. Intriguingly, this study revealed that cows with delayed ovulation symptoms were still capable of undergoing ovulation, albeit with delay. According to previous research, almost 70% of repeat breeders in livestock have suprabasal progesterone levels > 1 ng/ml during the onset of estrus (Prashar et al., 2023). When the suprabasal levels of progesterone increase, follicular receptors respond by reducing GnRH secretion, which subsequently leads to decreased follicle proliferation. The dominant follicles expanded to 3.2 cm after estrus expression on day 0 in DO cows (Fig. 1). The results of Prashar et al. (2023) are consistent with the findings of this study. Progesterone alters the pulsative action of LH and slows ovulation (Santos et al., 2016). In the presence of progesterone, there were fewer receptor activities in the pituitary gland than in the hypothalamus. This suggests that the hormone directly inhibits or slows down LH pulsation. Progesterone also influences the release of hypothalamic kisspeptin upstream. The oocyte quality decreases when suprabasal progesterone levels are high. This causes them to age, proteins to become phosphorylated, and meiosis to begin again from the diplotene stage of prophase I (Santos et al., 2016). In contrast to the NO cows, the DO cows exhibited a low LH pattern and sustained mature oocytes for a longer duration than the normal cows. Oocytes of cows with delayed ovulation may be of inferior quality due to their age, thereby increasing the risk of fertilization failure. Oocytes from cows with delayed ovulation require further investigation both in vivo (AI) and in vitro In vitro fertilization (IVF).
Conclusion
This study describes the association of DO in cows with prolonged estrus duration, delayed LH surges, and altered follicular and CL dynamics, which are linked to reduced reproductive efficiency. Elevated suprabasal progesterone levels preceding estrus were observed alongside inhibited LH release timing, smaller dominant follicles, extended follicular phases, and delayed ovulation without obvious ovarian abnormalities. These hormonal and ovarian changes coincide with increased insemination attempts and lower fertility, highlighting the complex endocrine relationships during the estrous cycle. The precise molecular mechanisms underlying LH pulsatility modulation and follicular progesterone responsiveness remain unclear. Future research may investigate the interactions between progesterone, kisspeptin signaling, and hypothalamic-pituitary axis regulation, as well as assess oocyte quality in cows exhibiting delayed ovulation using advanced in vivo and in vitro reproductive technologies. Such studies could contribute to the development of targeted strategies to improve fertility outcomes in cows with delayed ovulation.
Limitations
This study has several limitations related to its small sample size (N < 10), which restricts the robustness of parametric statistical analyses. The current standards emphasize the need for explicit justification when applying parametric tests in such small samples, including conducting normality and sphericity assessments. However, these prerequisites were not fully addressed, limiting the confirmatory strength of the findings and necessitating an exploratory interpretation. Additionally, the study does not explicitly discuss potential violations of independence assumptions, a critical gap highlighted in veterinary research where only a minority of small-sample studies address this issue. Given the evidence that the hormonal data in bovine studies with tiny samples (N < 8) often violate homoscedasticity assumptions, the reliance on traditional p-values may be insufficient. However, more robust approaches, such as reporting effect sizes with confidence intervals and employing bootstrapping techniques, could provide more reliable insights. These limitations underline the need for cautious interpretation and suggest that these methodological safeguards should be incorporated in future research to strengthen validity.
Acknowledgments
The authors extend their sincere gratitude to the LPDP and BRIN for their funding support through the third wave of the Research and Innovation Scheme for Advanced Indonesia.
Conflict of interest
The authors declare no conflicts of interest.
Funding
LPDP and BRIN through the third wave of the Research and Innovation Scheme for Advanced Indonesia under agreement numbers B-846/II.75/FR.06/5/2023 and B861/III.11/FR.06/5/2023.
Authors’ contribution
All authors declare that they have made equal contributions. LP, PIS, S, and H: Contributed to the study conception and design. PIS, H, SS, RIA, FBIL, DAM, sample and data collection. PIS performed data validation and statistical analysis. PIS and LP drafted and prepared the manuscript. All authors supervised the study. All authors have read, reviewed, and approved the final version of the manuscript.
Data availability
All data supporting this study’s findings are available in the manuscript.
References
Abreu, F.M., Coutinho da Silva, M.A., Cruppe, L.H., Mussard, M.L., Bridges, G.A., Harstine, B.R., Smith, G.W., Geary, T.W., Day, M.L. 2018. Role of progesterone concentrations during early follicular development in beef cattle: characteristics of LH secretion and oocyte quality. Anim. Reprod. Sci. 196, 59–68.
Al-Timimi, H., Khammas, D.J. and Al-Hamedawi, T.M. 2021. Delayed ovulation in bovine: diagnosis and treatment. Iraqi J. Vet. Med. 25(1), 77–90.
Borş, S.I. and Borş, A. 2020. Ovarian cysts, an anovulatory condition in dairy cattle. J. Vet. Med. Sci. 82(10), 1515–1522.
Boyer, C.N., Griffith, A.P. and DeLong, K.L. 2020. Reproductive failure and long-term profitability of spring- and fall-calving beef cows. JARE 45(1), 78–91.
Cardoso Consentini, C.E., Wiltbank, M.C. and Sartori, R. 2021. Factors that optimize reproductive efficiency in dairy herds with an emphasis on timed artificial insemination programs. Animals 11(2), 301–310.
Das, J.M., Dutta, P., Deka, K.C., Biswas, R.K., Sarmah, B.C. and Dhali, A. 2009. Comparative study on serum macro and micro mineral profiles during oestrus in repeat breeding crossbred cattle with impaired and normal ovulation. LRRD. 21(5), 1–5.
De Lima, M.A., Morotti, F., Bayeux, B.M., De Rezende, R.G., Botigelli, R.C., De Bem, T.H.C., Fontes, P.K., Nogueira, M.F.G., Meirelles, F.V., Baruselli, P.S., Da Silveira, J.C., Perecin, F. and Seneda, M.M. 2020. Ovarian follicular dynamics, progesterone concentrations, pregnancy rates and transcriptional patterns in Bos indicus females with a high or low antral follicle count. Sci. Rep. 10(1), 19557–19567.
Deka, R.P., Magnusson, U., Grace, D., Randolph, T.F., Shome, R. and Lindahl, J.F. 2021. Estimates of the economic cost caused by five major reproductive problems in dairy animals in Assam and Bihar, India. Animals (Basel). 11(11), 3116–3126.
Di Nisio, V., Antonouli, S., Damdimopoulou, P., Salumets, A. and Cecconi, S. 2022. in vivo and in vitro postovulatory aging: when time works against oocyte quality?. J. Assist. Reprod. Genet. 39(4), 1–12.
Dorfman, V.B., Saucedo, L., Di Giorgio, N.P., Inserra, P.I., Fraunhoffer, N., Leopardo, N.P., Halperín, J., Lux-Lantos, V. and Vitullo, A.D. 2013. Variation in progesterone receptors and gnrh expression in the hypothalamus of the pregnant south american plains vizcacha, lagostomus maximus (mammalia, rodentia). Biol. Reprod. Suppl. 89(5), 1–12.
Duncan, W.C. 2021. The inadequate corpus luteum. Reprod. Fertility 2(1), C1–C7.
Endo, N. 2022. Possible causes and treatment strategies for the estrus and ovulation disorders in dairy cows. JRD 68(2), 85–89.
Fraser, H.M., Dickson, S.E., Lunn, S.F., Wulff, C., Morris, K.D., Carroll, V.A. and Bicknell, R. 2000. Suppression of luteal angiogenesis in the primate after neutralization of vascular endothelial growth factor. Endocrinology 141(3), 995–1000.
Friis Wang, N., Skouby, S.O., Humaidan, P. and Andersen, C.Y. 2019. Response to ovulation trigger is correlated to late follicular phase progesterone levels: a hypothesis explaining reduced reproductive outcomes caused by increased late follicular progesterone rise. Hum. Reprod. 34(5), 942–948.
Honparkhe, M., Singh, J., Dadarwal, D., Ghuman, S.P.S., Dhaliwal, G.S. and Kumar, A. 2010. Effect of midluteal phase gnrh treatment in repeat breeder cattle. Indian J. Vet. Pathol. 87(4), 351–353.
Kim, D., Kwon, W.S., Ha, J., Kim, J., Kim, D., Lee, W., Moon, J. and Yi, J. 2023. Effect of oestrus synchronisation through ovulation delay by vaccination against foot-and-mouth disease in hanwoo (Bos taurus coreanae) cows. Vet. Med. Sci. 9(2), 974–981.
Klabnik, J. and Horn, E. 2023. When the plan goes awry: how to negotiate estrus synchronization errors in beef cattle. Clin. Theriogenology. 15(9265), 1–9.
López-Gatius, F., Santolaria, P., Mundet, I. and Yániz, J.L. 2005. Walking activity at estrus and subsequent fertility in dairy cows. Theriogenology 63(5), 1419–1429.
Lucy, M.C., Billings, H.J., Butler, W.R., Ehnis, L.R., Fields, M.J., Kesler, D.J., Kinder, J.E., Mattos, R.C., Short, R.E., Thatcher, W.W., Wettemann, R.P., Yelich, J.V. and Hafs, H.D. 2001. Efficacy of an intravaginal progesterone insert and an injection of pgf2α for synchronizing estrus and shortening the interval to pregnancy in postpartum beef cows, peripubertal beef heifers, and dairy heifers. J. Anim. Sci. 79(4), 982–995.
Medeiros, S.F., Barbosa, B.B., Medeiros, M.A.S. and Yamamoto, M.M.W. 2021. Morphology and biochemistry of ovulation. Rev. Bras. Ginecol. Obstet. 43(6), 480–486.
Miyamoto, A. and Shirasuna, K. 2009. Luteolysis in the cow : a novel concept of vasoactive molecules. Anim. Reprod. /. Colegio. Brasileiro. De. Reproducao. Anim. 6(1), 47–59.
Pate, J.L. and Hughes, C.H.K. 2023. Review: luteal prostaglandins: Mechanisms regulating luteal survival and demise in ruminants. Animal 17(Suppl 1), 100739.
Pérez-Marín, C.C. and Quintela, L.A. 2023. Current insights in the repeat breeder cow syndrome. Animals 12, 2187–2197.
Pfeifer, L.F.M., de Souza, L.S. del. C.B., Schneider, A., Schmitt, E. and Correa, M.N. 2012. Effect of the ovulatory follicle diameter and progesterone concentration on the pregnancy rate of fixed-time inseminated lactating beef cows. R. Bras. Zootec. 41, 1004–1008.
Prashar, V., Arora, T., Singh, R., Sharma, A. and Parkash, J. 2023. Hypothalamic kisspeptin neurons: integral elements of the gnrh system. Reprod. Sci. 20(3), 802–822.
Santos, J.E.P., Wiltbank, M.C., Ribeiro, E.S. and Bisinotto, R.S. 2016. Aspects and mechanisms of low fertility in anovulatory dairy cows. Anim. Reprod. 13(3), 290–299.
Singh, J., Ghuman, S.P.S., Honparkhe, M., Dadarwal, D. and Dhaliwal, G.S. 2012. Risk factors for prolonged estrus in crossbred dairy cattle. Indian. J. Anim. Sci. 82(1), 20–23.
Sitaresmi, P.I., Widyobroto, B.P., Bintara, S. and Widayati, D.T. 2020. Effects of body condition score and estrus phase on blood metabolites and steroid hormones in saanen goats in the tropics. Vet. World 13(5), 833–839.
Tiro, B.M.W., Tirajoh, S., Beding, P.A. and Baliarti, D.E. 2020. Estrus cycle and reproductive hormone profile of Ongole crossbred cows compared with Simmental-Ongole crossbred. Jurnal. Pertanian. Agros. 22(2), 105–112.
Trevisol, E., Mogollón García, H.D., Ackermann, C.L., Lacerda, W., Pires, R.M.L., Laufer-Amorin, R., Carvalho, R.F., Franchi, F.F., Castilho, A.C.S., Rizzoto, G., Kastelic, J.P. and Ferreira, J.C.P. 2020. Partial luteolysis during early diestrus in cattle downregulates vegfa expression and reduces large luteal cell and corpus luteum sizes and plasma progesterone concentration. Theriogenology 158, 188–195.
Wiltbank, M.C., Mezera, M.A., Toledo, M.Z., Drum, J.N., Baez, G.M., García-Guerra, A. and Sartori, R. 2018. Physiological mechanisms involved in maintaining the corpus luteum during the first two months of pregnancy. Anim. Reprod. 3(15), 805–821.
Xie, Y., Guo, W., Shen, X., Yu, W., Kuang, Y., Chen, Q., Long, H., Lyu, Q. and Wang, L. 2023. A delayed ovulation of progestin-primed ovarian stimulation (ppos) by downregulating the lhcgr/pgr pathway. IScience 26(8), 107357.














