| Research Article | ||
Open Vet. J.. 2026; 16(2): 1297-1314 Open Veterinary Journal, (2026), Vol. 16(2): 1297-1314 Research Article Computed tomography in the vivo anatomy of rabbit kidneys (Oryctolagus cuniculus)Avche Dineva1, Kamelia Stamatova-Yovcheva1*, Rosen Dimitrov1, Diyana Vladova1, David Yovchev1, Ömer Gurkan Dilek2, Hristo Hristov1, Vladi Nedev3, Nikolay Goranov3 and Tihomir Dinev11Department of Veterinary Anatomy, Histology and Embryology Faculty of Veterinary Medicine, Trakia University, Stara Zagora, Bulgaria 2Department of Anatomy, Faculty of Veterinary Medicine, Mehmet Akif Ersoy University, Burdur, Turkey 3Department of Veterinary Surgery, Faculty of Veterinary Medicine, Trakia University, Stara Zagora, Bulgaria *Corresponding Author: Kamelia Stamatova-Yovcheva. Department of Veterinary Anatomy, Histology and Embryology, Faculty of Veterinary Medicine, Trakia University, Stara Zagora, Bulgaria. Email:kameliastamatovayovcheva [at] gmail.com Submitted: 17/11/2025 Revised: 14/01/2026 Accepted: 29/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
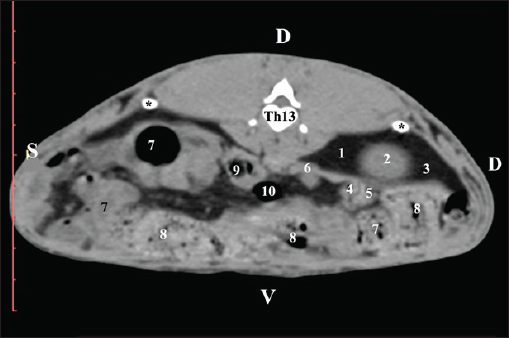
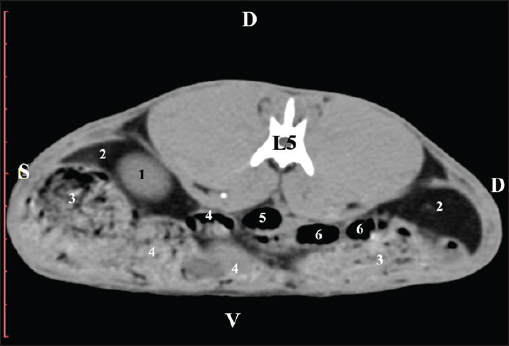
AbstractBackground: The rabbit kidney is a unipapillary organ. According to other researchers, rabbit kidneys have 6–8 pyramids. The crista renalis is well differentiated. Aim: This study aimed to describe the computed tomography (CT) anatomy and dimensions of rabbit kidneys. Methods: Ten sexually mature, clinically healthy rabbits were divided into two groups (five males and five females). The animals were positioned in the sternal position. A SOMATOM helical computed tomograph was used in the study with consecutive Dicom Viewer reconstructions of the data. Results: On transverse precontrast CT scans, the right kidney was observed between Th13 (L1) and L2–L3, and the left kidney appeared between L2 (L3) and L4 (L5). The CT value of the right kidney was 40.28 ± 6.6 HU, and that of the left kidney was 43.36 ± 5.2 HU. The CT method was precise and found an excellent positive correlation between both kidney length (Y variable) and L2 length (X variable). The Pearson correlation coefficient was 0.957 for the right kidney and 0.961 for the left kidney at 95% confidence interval. The measured length of both kidneys on CT scans demonstrated a very strong positive correlation with L2 length. Retrospective reconstruction revealed both kidneys as multipapillary organs. The cortex renis and medulla renis were sharply distinct, and the boundary between the capsula fibrosa and capsula adiposa was defined. Conclusion: The obtained in vivo results are precise and serve as an anatomical model for the use of CT in rabbits and other animal species, including humans. Keywords: Anatomy, CT, Kidney, Rabbit. IntroductionThe kidneys of rabbits are paired organs located just below the dorsal abdominal wall muscles. The position of the right kidney is asymmetrical due to the compression of adjacent organs during the embryonic period. Thus, the right kidney is placed cranially to the left one. The organs are brownish, with a specific bean-like shape, surrounded by a fibrous capsule, medially connected with the ureter, and lie retroperitoneally. The kidneys have a single papilla, and the renal pelvis is not separated. Every kidney possesses an adipose capsule, known as the capsula adiposа. After incision, the renal pelvis may be identified by the funnel-shaped expansion of the ureter’s end corresponding to the borders of the renal sinus. The renal cortex is a distinct, narrow peripheral zone. The renal medulla, including the renal papilla, forms the central part of the kidney. The renal collumnae radiate through the core (Bensley, 1948; Farhana and Wibowo, 2019). The kidneys of rabbits can be easily identified by palpation. They are highly motile. The right kidney is an intrathoracic abdominal organ (Harcourt-Brown, 2002 ). According to Brewer (2006); Vella and Donnelly (2012) and Dyce et al. (2010) the structure of the rabbit kidney is primitive compared with that of other domestic mammals. It comprises a well-defined capsule, cortex, and medulla. It is smooth and unipapillary, with only a single calyx opening directly into the ureter. The right kidney is located cranially to the left kidney. Some authors (Dimitrov and Chaprazov, 2012a,b) have described the bean-shaped and dorsoventrally flattened kidneys of rabbits. The kidneys have a well-developed cortex, medulla, renal papilla, and renal pelvis. They are bipyramidal, with a renal papilla projecting onto the hilus renalis. The rabbit kidney is similar to that of small ruminants and dogs. It comprises the cortex renis and medulla renis. The hilus renalis is strongly narrowed and adjacent to the extremitas cranialis. As in small ruminants and dogs, both kidneys have 6–8 pyramids, whose margins are poorly distinct. The cista renalis are well defined. The extremitas cranialis of the right kidney attains the 13th rib, and the extremitas caudalis attains L3. The extremitas cranialis of the left kidney is positioned within L3–L4, and the extremitas caudalis reaches L5 (Barone, 2020). A case of smooth-surface multipyramidal kidneys in a rabbit has been described (Stamatova-Yovcheva et al., 2022). The cortex and medulla were constructed using pyramid-shaped lobes. The apex of the lobes formed papillae, which protruded into the renal sinus calyces. The renal pelvis was concave. Other literature sources on the topography of rabbit kidneys (Barone, 2001; Hristov et al., 2006) describe them in the retroperitoneal space—spatium retroperitoneale; the right one being between the 11th/12th intercostal space and L2. The cranial pole of the organ is in anatomical contact with the liver, just beneath the last intercostal space. The right kidney’s caudal pole extends up to the duodenal pars descendens. The left kidney is placed ventrocaudally in relation to the right one, between L2 and L4. The jejunal loops are located ventrally to the left kidney, whereas the descending part of the sigmoid colon and corpus pancreatis are located ventrally (Barone, 2001; Hristov et al., 2006). The right kidney of rabbits is cranially located in relation to the left one, opposite to humans, where the left kidney is dorsally positioned in relation to the right one. This is due to the specific position of the liver, which occupies a considerable space to the left of the median plane and above the left kidney (Farhana and Wibowo, 2019). The right rabbit kidney lies between Th13 and L2, whereas the right kidney is located caudally and is located between L3 and L5. Margo lateralis does not contact the abdominal wall. The cranial pole of the right kidney extends up to the beginning of Th13 and contacts the liver’s lobus caudatus. The caudal pole of the right kidney reaches the end of L2; the organ is observed in the middle of the vertebral column–right abdominal wall distance. The left kidney is located laterally to the middle of the distance between the vertebral column and the left abdominal wall (Dimitrov and Chaprazov, 2012a,b). The rabbit is an experimental model of kidney transplantation in humans and for monitoring the effect of immunosuppressive drugs during the post-transplantation period (Wu et al., 2003; Saeed et al., 2020). Rabbits are prone to pyelonephritis and renal infarctions accompanied by hemorrhages. The urine is alkaline, dark yellow to brown, and contains substantial amounts of calcium carbonate and triple phosphate crystals (ammonium magnesium phosphate and calcium carbonate monohydrate) (Percy and Barthold, 2007; Harcourt-Brown, 2013). Computed tomography (CT) is a contemporary, intravital, prompt, and accurate imaging method for visualizing the kidneys and renal blood supply in mini-pigs and rabbits. The average volume of the right kidney in mini-pigs is greater than that of the contralateral kidney (Yoon et al., 2010; El-Ghazali et al., 2021). Pre-contrast CT examination of abdominal organs is a convenient method for obtaining anatomical information in small domestic mammals. The contrast examination is mainly used to define the morphology of specific parenchymal structures and vessels (Kleiter et al., 1999; Gielen et al., 2003). The pre-contrast CT examination is a reliable and precise modern method for visualizing the renal parenchyma, renal pelvis, adipose capsule, and peripelvic adipose tissue, as well as for detailed visualization of kidney calculi (Yewon et al., 2023). Compared with other imaging methods, CT is the method of choice for the exploration of the rabbit’s abdominal cavity. Both transverse and dorsal CT scans of rabbit kidneys depict the bilateral position of the organs in the regio abdominis lateralis dextra et sinistra. On consecutive transverse scans, the right kidney appears cranial to the left one. The right kidney and lobus caudatus possess comparable soft tissue density characteristics, with the hypoattenuated adipose capsule between. On the gray scale, the attenuation of the organ is medium, with gray shades. There is no difference between the density of the cortex renis and medulla renis (El-Ghazali et al., 2021). The pre-contrast CT examination of canine kidneys depicts the organs as normoattenuated homogeneous soft tissue findings. On pre-contrast scans, the crista renalis is a hyperattenuated structure compared with the relatively hypoattenuated medulla renis (Serra-Gomez de la Serna et al., 2024). An intravital CT examination of the kidneys of dogs and cats was performed to provide numerical values about the density of the cortex renis and medulla renis (Oliveira et al., 2013). The cortex of both kidneys had a greater density than the medulla. The volumes of both kidneys obtained from intravital CT imaging in dogs are proportional to the length and body weight of the studied organs. This correlation is essential for normal renal function, and deviations are considered an indication of several renal illnesses (Yewon et al., 2022). The proximity between the right kidney and the liver is clearly visible on sagittal computed tomography scans. Impressio renalis is an anatomical contact marker between the two organs, which is identified on transverse CT scans as a soft tissue hypoattenuated finding at the 13th thoracic vertebra (T13) level (Stamatova-Yovcheva et al., 2012; Stamatova-Yovcheva et al., 2013). Anatomical contact between the right kidney and proc. caudatus in chinchillas is observed at the Th13 level. The right kidney is visualized as a hyperattenuated soft tissue finding against the hypoattenuated liver (Dilek et al., 2024). Relationships exist between the length of both kidneys in dogs and the length of the second lumbar vertebra (L2); according to the findings, the body weight of biological subjects had no effect on this correlation (Hoey et al., 2016). Some authors (Atalar et al., 2017) have provided information on the macrodimensions (length and width) of the kidneys in Kangal dogs. The right kidney demonstrated greater values than the left kidney. The 3D reconstructions proved to be a reliable method for investigating the anatomical features of the kidneys, depending on the canine breed. The use of the rabbit kidney as an anatomical model in medical research, the susceptibility of the organ to species-specific diseases, and the contradictory anatomical theories about the lobulation of the organ were the main motivations for the present research. Materials and MethodsTen clinically healthy, sexually mature, 7-month-old New Zealand White rabbits weighing 2.5–3.0 kg (5 male and 5 female) were studied. A helical computed tomograph (SOMATOM) (go SN 168190 (Siemens Healthineers, Forchheim, Germany) was used in the study. The animals were in sternal recumbency. Anesthesia was applied at 15 mg/kg Zoletil® 50 IM (tiletamine hydrochloride 125 mg and zolazepam hydrochloride 125 mg in 5 ml of solution) (Virbac, Carros-Cedex, France). It was administered intramuscularly to the gluteal muscles. The animals were studied in the Radiology Unit of Trakia University, Faculty of Veterinary Medicine hospital. The abdominal cavity was investigated from the regio abdominis cranialis to the appertura pelvis cranialis. Twenty slices were obtained from each rabbit. The scanning parameters were as follows: the thickness of the slices was 0.7 mm, the electric current intensity was 30 mA, the collimation thickness was 16 × 0.75 mm, the X-ray tube current was 250 mA, the scanning time was from 0.8 to 1.0 seconds for each slice, the CT dose index volume was 55.10, and mGy was 10. The matrix was 1024 pixel matrix high-resolution CT, the rotation time was up to 0.5 seconds, the rotational speed was 360° in 0.8, 1, 2, 3, and 4 seconds; the pitch was 6, the anode tension was 130 kV, the zoom was 145%, the window level was 50, the window width was 350, the exposure time was 1981 s, CTDIvol was 1.67 mGy, the converting filter was standard, the tilt was 0.5, the level was 35, and the Field of view (FOV) was 50. Transversal slices were obtained between the planes from the Th13 thoracic vertebra to the L5 lumbar vertebra. Images were visualized using the RadiAnt Dicom Viewer (2025.1 BETA 64-bit). Retrospective reconstructions were obtained using the Angio–Right and Left, MIP B/W, Bones and Skin 1, and Bones and Skin 3 algorithms through the RadiAnt Dicom Viewer software. Soft tissue density data were statistically processed. A radiological specialist regularly calibrated the system. A gating system was used to obtain high-resolution slices. The pre-contrast CT study was conducted to study the topography of the kidneys and their adjacencies with the other abdominal organs in detail. The scans were obtained at the beginning and end of the corresponding vertebra. The CT values were measured in the images with a detailed visualization of the rabbit kidneys. The data were statistically processed using Statistica 8–StatSoftDELL (version 13.3.721), as the mean values were presented. The Region of interest (ROI) was 20% of the FOV. The CT values included all structures of the kidneys’ parenchyma and were measured 10 times in all of the studied rabbits. Measurements were performed using DicomViewer. The post-contrast study was conducted as a positive contrast agent Iopamiro 370 mg/ml (EWOPHARMA, Patheon Italia S.p. A., Bracco Imaging S.p. A., Italy) was administered through the external jugular vein at a dose of 1.8 ml/kg. The time interval between contrast agent administration and post-contrast scan was 3 minutes. The kinetics of the clearance were as follows: the first slices appeared with hyperattenuated kidneys at the fourth minute, and the renal medulla and renal pelvis were visible as structures with high attenuation at the seventh minute. The contrast enhanced the ureters at the 10th minute. Scans from Th13 to L5 were used for reconstruction. Post-contrast CT scans were performed four times: 3, 4, 7, and 10 minutes after contrast medium administration. The reconstructions were angio–right and left, bone and skin 1, and bone and skin 3. They were involved in DicomViewer. Angio–Right and Left was used to measure the macrodimensions. craniocaudal dimension (CCD), craniocaudal dimension (length), and mediolateral dimension (MLD), mediolateral dimension (width), were the studied parameters. CCD was measured using the section measurements tools in DicomViewer. The length was evaluated from the cranial to the caudal pole of the kidney. The length was measured using MPR. MLD (width) was measured from the hilus renalis to the lateral border of the kidney. The results were presented in centimeters, and a descriptive analysis was performed using Statistica 8, version 13.3.721. Soft tissue density data were statistically processed. The correlation between the length of the kidneys and the body length of L2 was determined using Pearson’s correlation coefficient and 95% confidence interval (CI) (Atalar et al., 2017). The CT kidney anatomy was interpreted in accordance with Nomina Anatomica Veterinaria (2017). To obtain objective anatomical data, the CT anatomical protocol was selected, with the direction of the slices aligned with that of the examiner (Dilek et al., 2024). Ethical approvalThe Committee of Ethics of the Ministry of Agriculture and Food, Bulgarian Animal Safety Agency, with opinion No. 293 of 29.02.2024, Sofia, Bulgaria, has been involved in the investigation by permit No. 377, in compliance with the provisions of the Animal Protection Act in Bulgaria (promulgated in State Gazette No 13/8 February 2008) and the European Convention for the Protection of Vertebrate Animals Used for Experimental Purposes. All animal experiments were conducted in accordance with the ARRIVE guidelines. ResultsPre-contrast computed tomography investigationPre-contrast CT at the Th13 level revealed the right kidney’s extremitas cranialis as a normoattenuated homogeneous soft tissue against the hypoattenuated margins of the lobus hepatis dexter and lobus caudatus (proc. caudatus). Ventrally, the right kidney was in contact with the duodenal par descendens, ileum, and cecum (Fig. 1).
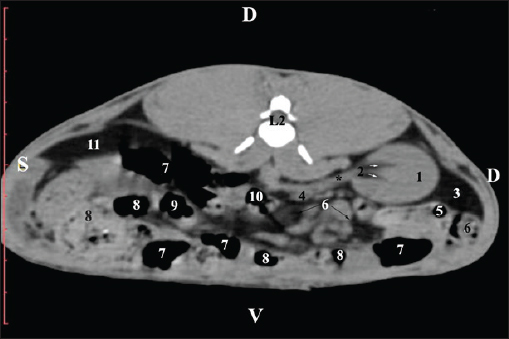
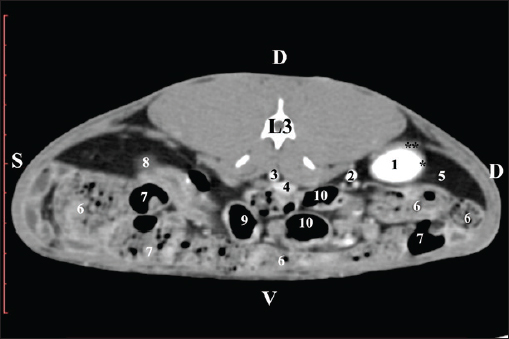
Fig. 1. Pre-contrast computed tomography (CT) anatomical scan of the regio abdominis cranialis at the Th13 level. (1) Lobus hepatis dexter; (2) ren dexter; (3) lobus caudatus (proc. caudatus) with visceral adipose tissue; (4) pars descendens of the duodenum; (5) ileum; (6) gl. adrenalis dextra; (7) colon ascendens; (8) cecum; (9) colon transversum; (10) colon descendens; (*) 13th rib. At the next scan level (L1), the normoattenuated image of the right kidney was more detailed and in contact with the lobus hepatis dexter, lobus caudatus, duodenal pars descendens, and ileum. The cortex renis and medulla renis were not clearly differentiated. Pelvis renalis appeared as a linear hypoattenuated finding against the relatively normoattenuated kidney cortex and medulla. The proximal part of the ureter was seen medial to the right kidney (Figs. 2 and 3).
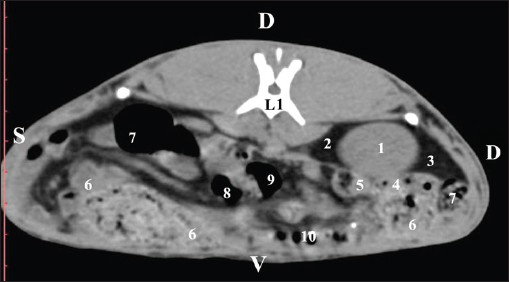
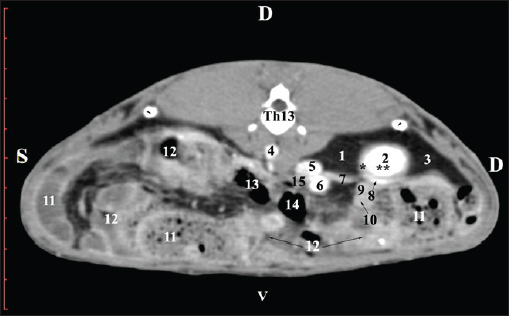
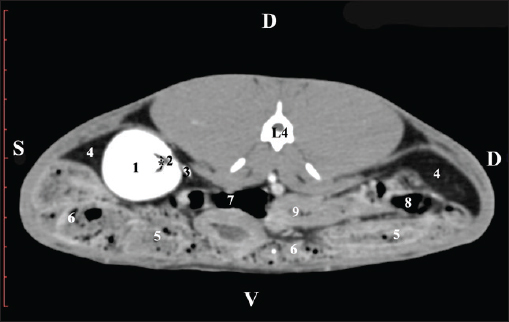
Fig. 2. Pre-contrast computed tomography (CT) anatomical scan of the regio abdominis cranialis at the L1 level. (1) Ren dexter; (2) lobus hepatis dexter; (3) lobus caudatus (proc. caudatus) with visceral adipose tissue; (4) pars descendens of the duodenum; (5) ileum; (6) cecum; (7) colon ascendens; (8) colon transversum; (9) colon descendens; (10) jejunum.
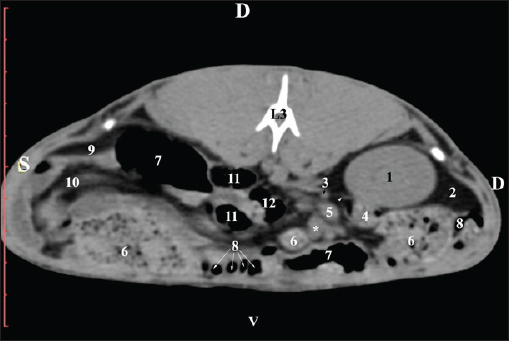
Fig. 3. Pre-contrast computed tomography (CT) anatomical scan of regio abdominis cranialis at the L1 level. (1) Medulla renis; (2) cortex renis; (3) pelvis renalis; (4) lobus caudatus (proc. caudatus) with visceral adipose tissue; (5) ureter; (6) pars descendens of the duodenum; (7) jejunum; (8) cecum; (9) colon ascendens; (10) lien; (11) colon descendens; (12) colon transversum. At the L2 level, the right kidney was in contact with the liver’s proc. caudatus, pars descendens of the duodenum, and the ileum. Pelvis renalis was a distinct hypoattenuated funnel-shaped soft tissue finding, whose narrow end pointed at the renal hilum and passed into the ureter. Recessus renalis was a clearly visible hypoattenuated widening of the central part of the renal pelvis in the dorsal and ventral directions (Figs. 4 and 5).
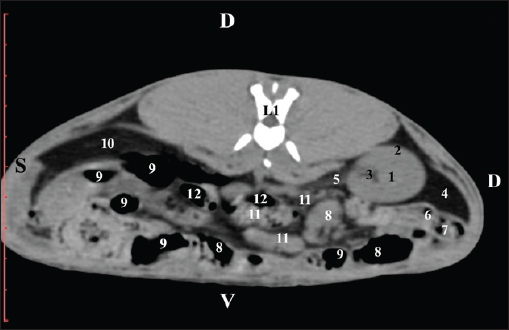
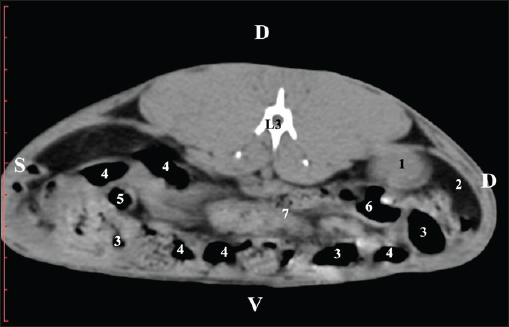
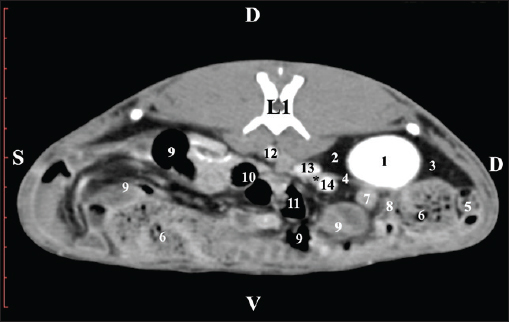
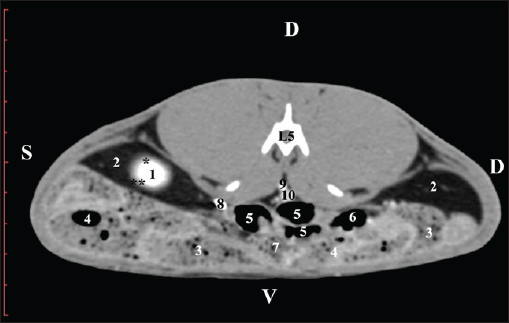
Fig. 4. Pre-contrast CT anatomical scan of the regio-abdominis media at the cranial L2 edge level. (1) Ren dexter; (2) pelvis renalis; (3) lobus caudatus (proc. caudatus) with visceral adipose tissue; (4) pars descendens of the duodenum; (5) ileum; (6) jejunum; (7) colon ascendens; (8) cecum; (9) colon transversum; (10) colon descendens; (11) lien; (12) pars transversa of the duodenum ; (*) ureter; (white arrow) recessus renalis.
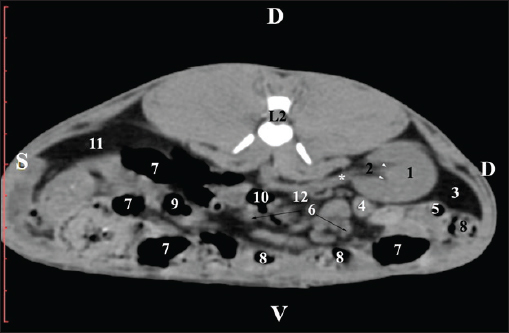
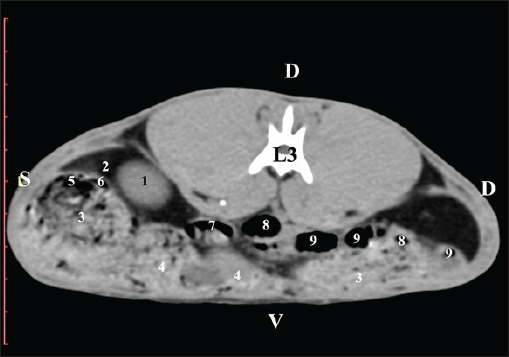
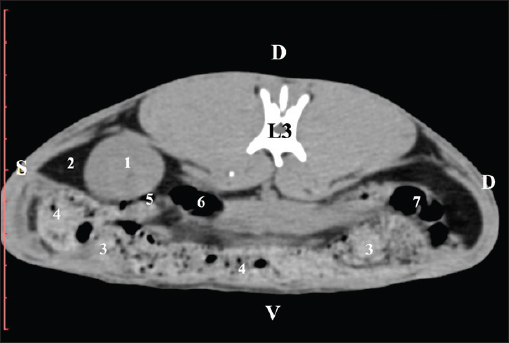
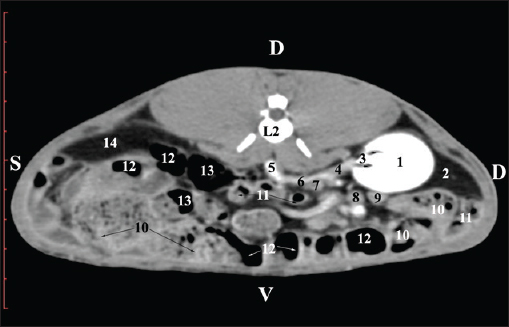
Fig. 5. Pre-contrast computed tomography (CT) anatomical scan of the regio-abdominis media at the caudal L2 edge level. (1) Ren dexter; (2) pelvis renalis; (3) lobus caudatus (proc. caudatus) with visceral adipose tissue; (4) gl. adrenalis dextra; (5) ileum; (6) jejunum; (7) colon ascendens; (8) cecum; (9) colon transversum; (10) colon descendens; (11) lien; (*) ureter; (white arrow) recessus renalis. At the L3 level, the anatomical adjacencies of the right kidney with the pars descendens of the duodenum, ileum, cecum, and colon descendens were visualized. Hypoattenuated right ureter and normoattenuated gl. adrenalis dextra were situated medially to the right kidney. Extremitas caudalis of the right kidney was a clearly visible normoattenuated finding, surrounded by hypoattenuated borders of abdominal adipose collections (Figs. 6 and 7).
Fig. 6. Pre-contrast CT anatomical scan of the regio-abdominis media at the cranial L3 edge level. (1) Ren dexter; (2) lobus caudatus (proc. caudatus) with visceral adipose tissue; (3) gl. adrenalis dextra; (4) pars descendens of the duodenum; (5) ileum; (6) cecum; (7) colon ascendens; (8) jejunum; (9) lien; (10) colon ascendens; (11) colon transversum; (12) colon descendens, (*) mesenterium.
Fig. 7. Pre-contrast computed tomography (CT) anatomical scan of the regio-abdominis media at the caudal L3 edge level. (1) Ren dexter; (2) adipose collection; (3) cecum; (4) colon ascendens; (5) colon transversum; (6) colon descendens; (7) mesenterium. The transverse pre-contrast examination of the regio abdominis media at the L3 level (through the cranial part of the vertebra) revealed the left kidney’s extremitas cranialis as an oval soft tissue with normoattenuated soft tissue characteristics. Ventrally, the kidney was adjacent to the duodenal pars ascendens, ileum, colon ascendens, and colon descendens. Hypoattenuated abdominal adipose collections were observed around it (Figs. 8 and 9).
Fig. 8. Pre-contrast computed tomography (CT) anatomical scan of the regio-abdominis media at the cranial L3 edge level. (1) Ren sinister; (2) adipose collections; (3) cecum; (4) colon ascendens; (5) jejunum; (6) pars ascendens of the duodenum; (7) colon descendens; (8) colon transversum; (9) colon descendens.
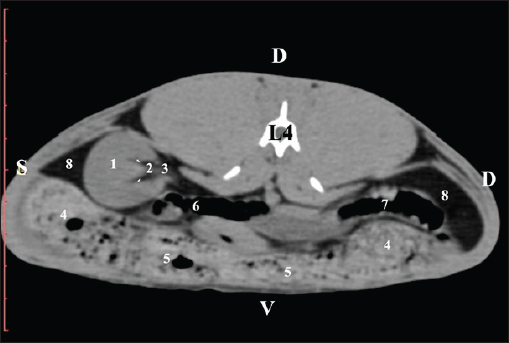
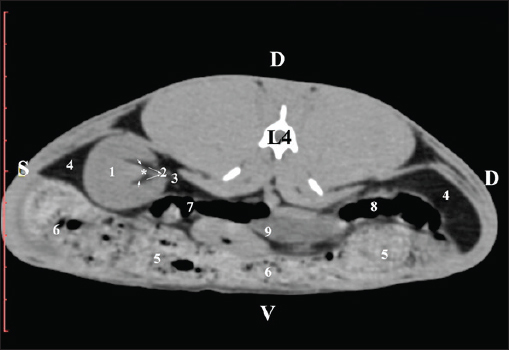
Fig. 9. Pre-contrast computed tomography (CT) anatomical scan of the regio-abdominis media at the caudal L3 edge level. (1) Ren dexter; (2) adipose collections; (3) cecum; (4) colon ascendens; (5) jejunum; (6) pars ascendens of the duodenum; (7) colon descendens. The pre-contrast anatomical scan at the L4 level provided data about the location of the renal pelvis within the renal sinus. The initial part of the pelvis renalis appeared as a distinct funnel-shaped hypoattenuated soft tissue, whose central part was widened and formed the recessus renalis. The transition of the narrowed end of the renal pelvis into the hypoattenuated left ureter was without a sharp boundary. The left kidney was in contact with the cecum, colon ascendens, and colon transversum ventrally. Crista renalis was observed as a hypoattenuated linear soft tissue finding (Figs. 10 and 11).
Fig. 10. Pre-contrast computed tomography (CT) anatomical scan of the regio-abdominis media at the cranial L4 edge level. (1) Ren sinister; (2) pelvis renalis and crista renalis; (3) ureter; (4) cecum; (5) colon ascendens; (6) colon transversum; (7) colon descendens; (8) adipose collections.
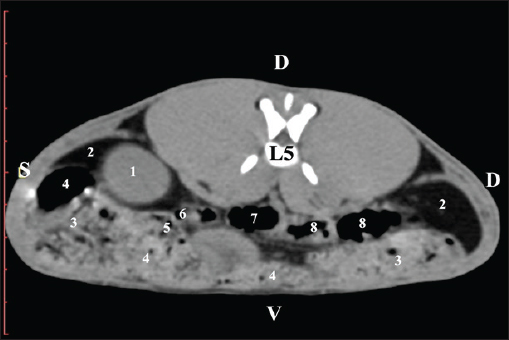
Fig. 11. Pre-contrast computed tomography (CT) anatomical scan of the regio-abdominis media at the caudal L4 edge level. (1) Ren sinister; (2) pelvis renalis; (3) ureter; (4) adipose collections; (5) cecum; (6) colon ascendens; (7) colon transversum; (8) colon descendens; (9) mesenterium; (white arrows) recessus renalis; (*) crista renalis. The L5 scan revealed the caudal end of the left kidney. It was adjacent to some small intestinal segments, including the duodenal pars ascendens, ileum, cecum, and colon ascendens (Figs. 12 and 13).
Fig. 12. Pre-contrast computed tomography (CT) anatomical scan of the regio-abdominis media at the cranial L5 edge level. (1) Ren sinister; (2) adipose collections; (3) cecum; (4) colon ascendens; (5) jejunum; (6) pars ascendens of the duodenum; (7) colon transversum; (8) colon descendens.
Fig. 13. Pre-contrast computed tomography (CT) anatomical scan of the regio-abdominis media at the caudal L5 edge level. (1) Ren sinister; (2) adipose collections; (3) cecum; (4) colon ascendens; (5) colon transversum; (6) colon descendens. Post-contrast CT imagingCT anatomical study at the Th13 level was used to define the right kidney zones. The medulla renalis was found as a hyperattenuated soft tissue with peripheral linear and wavy margins that reached the relatively hypoattenuated renal cortex. The most peripheral structure was the capsula adiposa, characterized by hyperattenuated soft tissue and uniform relief. The hyperattenuated aorta abdominalis, v. cava caudalis, and a. renalis appeared medially to the right kidney (Fig. 14).
Fig. 14. Post-contrast computed tomography (CT) anatomical scan of regio abdominis cranialis at the Th13 level. (1) Lobus hepatis dexter; (2) ren dexter; (3) lobus caudatus (processus caudatus); (4) aorta abdominalis; (5) v. cava caudalis; (6) a. renalis; (7) ureter; (8) capsula adiposa (capsula fibrosa); (9) pars descendens of the duodenum; (10) ileum; (11) cecum; (12) colon ascendens; (13) colon transversum; (14) colon descendens; (15) gl. adrenalis dextra; (black arrowhead) XIII rib; (*) cortex renalis; (**) medulla renalis. Post-contrast CT examination at the L1 level did not clearly distinguish the cortex from the medulla of the examined organ. The kidney appeared as a uniform oval hyperattenuated soft tissue, adjacent to the hypoattenuated lobus hepatis dexter and lobus caudatus. The renal capsules—capsula fibrosa and adiposa—were not differentiated. The large blood vessels, the beginning of the ureter, and the right adrenal gland were seen medial to the renal kidney (Fig. 15).
Fig. 15. Post-contrast computed tomography (CT) anatomical scan of the regio abdominis cranialis at the L1 level. (1) Ren dexter; (2) lobus hepatis dexter; (3) lobus caudatus (proc. caudatus); (4) ureter; (5) jejunum; (6) cecum; (7) pars descendens of the duodenum; (8) ileum; (9) colon ascendens; (10) colon transversum; (11) colon descendens; (12) aorta abdominalis; (13) v. cava caudalis; (14) a. renalis; (*) gl. adrenalis dextra. At the L2 level, the renal pelvis was clearly visible as a funnel-shaped hyperattenuated soft tissue with well-demarcated hypoattenuated recesses. The transition of the renal pelvis into the relatively hypoattenuated ureter was without a distinct boundary. The post-contrast CT scan did not reveal the renal cortex and medulla margins or the presence of adipose and fibrous capsules (Fig. 16).
Fig. 16. Post-contrast computed tomography (CT) anatomical scan of the regio abdominis cranialis at the L2 level: (1) ren dexter; (2) processus caudatus; (3) pelvis renalis; (4) ureter; (5) aorta abdominalis; (6) a. renalis; (7) gl. adrenalis dextra; (8) pars descendens of the duodenum; (9) ileum; (10) cecum; (11) jejunum; (12) colon ascendens; (13) colon descendens; (14) lien; (black arrowheads) recessus renalis. The L3 scan revealed the caudal end of the kidney as a hyperattenuated oval finding. The right ureter was medially located to it. The capsula adiposa was relatively hypoattenuated and covered the fine contours of the capsula fibrosa. The beginning of the left kidney’s extremitas cranialis appeared as an oval hypoattenuated soft tissue (Fig. 17).
Fig. 17. Post-contrast computed tomography (CT) anatomical scan of the regio-abdominis cranialis at the L3 level: (1) Ren dexter; (2) ureter ; (3) aorta abdominalis; (4) V. cava caudalis; (5) adipose collections; (6) cecum; (7) colon ascendens; (8) Ren sinister; (9) colon transversum; and (10) colon descendens. The scan through L4 presented the funnel-shaped margins of the pelvis renalis as a relatively hypoattenuated soft tissue finding, whereas the kidney recesses were visualized in its central part. The crista renalis was hyperattenuated and nominated into the renal pelvis. The beginning of the ureter was medial to the left kidney (Fig. 18).
Fig. 18. Post-contrast computed tomography (CT) anatomical scan of the regio abdominis cranialis at the L4 level. (1) Ren sinister; (2) pelvis renalis; (3) ureter; (4) lien; (5) colon ascendens; (6) cecum; (7) colon transversum; (8) colon descendens; (9) mesenterium; (*) crista renalis; (black arrowhead) recessus renalis. The extremitas caudalis was clearly visible at the L5 level. The capsula adiposa was relatively hypoattenuated and covered the capsula fibrosa. Меdially, the hyperattenuated ureter was visible (Fig. 19).
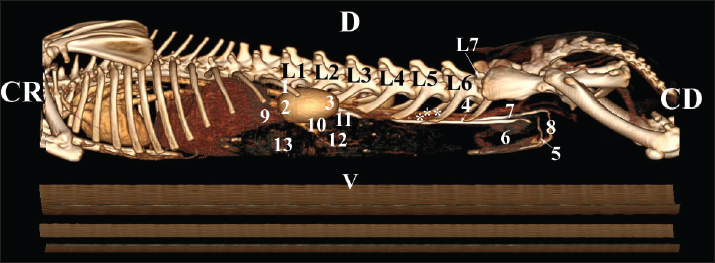
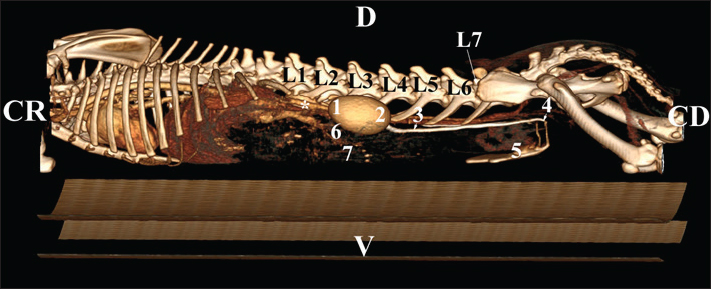
Fig. 19. Post-contrast computed tomography (CT) anatomical scan of the regio-abdominis cranialis at the L5 level: (1) Ren sinister; (2) adipose collections; (3) cecum; (4) colon ascendens; (5) colon transversum; (6) colon descendens; (7) mesenterium; (8) ureter; (9) aorta abdominalis; (10) v. cava caudalis; (*) cortex renalis; (**) capsula adiposa. Retrospective reconstructionsThe retrospective reconstruction in the Bones and Skin 1 and Bones and Skin 3 modes provided information about the anatomical features of both kidneys, including surface relief and renal pyramids. The two kidneys were multipapillar organs with 6–8 pyramids; their tips were fused into a single crista renalis, directed toward the renal pelvis. Dorsally and ventrally, the renal pelvis became wider to form recessus renalis, аnd its terminal part continued toward the ureter. The cortex renis and medulla renis were distinct soft tissue findings, and the boundary between the capsula fibrosa and capsula adiposa was clearly defined (Figs. 20 and 21).
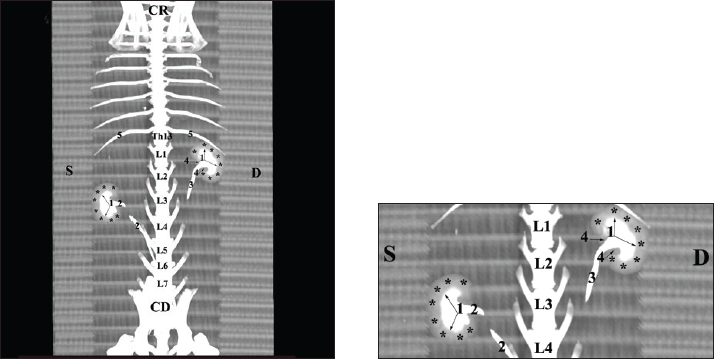
Fig. 20. Retrospective reconstruction of computed tomography (CT) data in Bones and Skin 1 mode in a rabbit: (1) Crista renalis; (2) left ureter; (3) right ureter; (4) recessus renalis; (*) papilla renalis; (5) XIII rib.
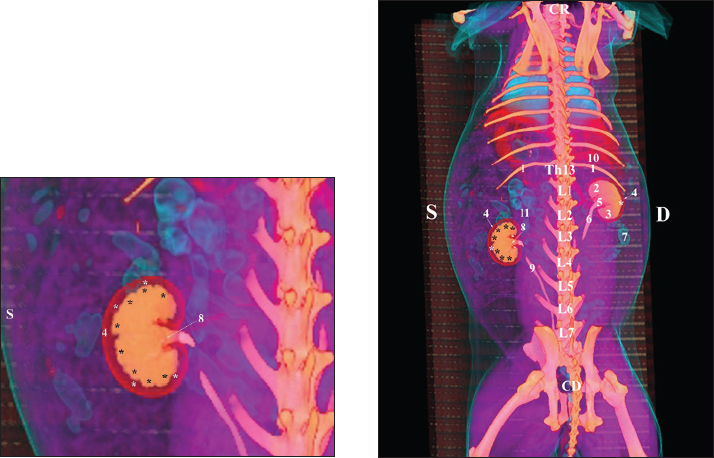
Fig. 21. Retrospective reconstruction of computed tomography data in Bones and Skin 3 mode in a rabbit. (1) XIII h rib; (2) extremitas cranialis; (3) extremitas caudalis; (4) capsula adiposa; (5) pelvis renalis (right kidney); (6) right ureter (pars abdominalis); (7) pars descendens of the duodenum; (8) pelvis renalis (left kidney); (9) left ureter (pars abdominalis); (10) lobus caudatus (proc. caudatus); (11) pars ascendens of the duodenum. Retrospective reconstruction in the Angio–Right and Left mode (right and left sides) yielded information about the localization of both kidneys in the RA. The right kidney was located in the region abdominis cranialis and region abdominis media, between Th13 (L1) and L2–L3, whereas the left kidney was located between L2 (L3) and L4 (L5) (Figs. 22 and 23).
Fig. 22. Retrospective reconstruction of computed tomography (CT) data in Angio–Right and Left 1 mode in a rabbit (right side). (1) 13th rib; (2) extremitas cranialis; (3) extremitas caudalis; (4) right ureter (pars abdominalis); (5) right ureter (pars pelvina); (6) colon; (7) left ureter (pars abdominalis); (8) left ureter (pars pelvina); (9) lobus caudatus (proc. caudatus); (10) pars descendens of the duodenum; (11) ileum; (12) colon ascendens; (13) cecum; (*) v. cava caudalis; (**) aorta abdominalis.
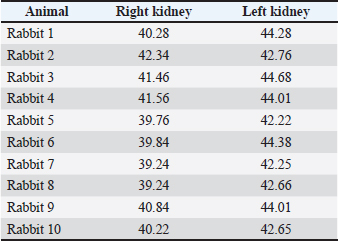
Fig. 23. Retrospective reconstruction of computed tomography data in Angio–Right and Left 1 mode in a rabbit (left side). (1) Extremitas cranialis; (2) Extremitas caudalis; (3) Left ureter (pars abdominalis); (4) Left ureter (pars pelvina); (5) Vesica urinaria; (6) Cecum; (7) Colon ascendens; (*) Aorta abdominalis. Statistical and metric analysesSoft tissue density was measured on pre-contrast CT scans to obtain objective data. The density of the right kidney was 40.28 ± 6.6 HU, and that of the left kidney was 43.36 ± 5.2 HU (Table 1). Table 1. CT values of rabbit kidneys in the HU.
CT anatomical dimensionsThe mean right kidney length was 3.45 ± 0.09 cm compared to 3.50 ± 0.07 cm for the left kidney. An excellent positive correlation was found between both kidney length (Y variable) and L2 length (X variable). The Pearson correlation coefficient was 0.957 for the right kidney and 0.961 for the left kidney at 95% CI. The length of both kidneys was strongly positively correlated with L2 length. The right and left kidney width was 2.36 ± 0.06 cm and 2.18 ± 0.07 cm, respectively, at p < 0.001. In all of the studied animals, the aforementioned specifics of the rabbit kidneys were found. The investigated organs had common features, including the cortex renis, medulla renis, eight papillae renales, well-developed pelvis renalis, macrodimensions, and soft tissue CT values. DiscussionThe presented information about the bilateral localization of both kidneys and the cranial position of the right kidney in relation to the left one confirmed Bensley’s (1948) thesis on the topography of these organs in rabbits. Furthermore, the retrospective reconstruction of CT data in Bones and Skin 1 and Bones and Skin 3 modes demonstrated the presence of well-formed renal papillae, which disagrees with the existing theories about the unipyramidal structure of the rabbit kidney (Bensley, 1948; Brewer, 2006; Dyce et al., 2010). The presence of well-developed capsula adiposa, capsula fibrosa, and distinctive cortex renis, medulla renis, and pelvis renalis in the Bones and Skin 3 reconstruction mode was added to the anatomical structure of rabbit kidneys (Bensley, 1948; Dyce et al., 2010). The presented data on the right kidney’s localization in the regio abdominis cranialis and that of the left kidney in the regio abdominis cranialis and regio abdominis media supplemented the information on the mutual placement of kidneys in the abdominal cavity. The observed localization of the right kidney in relation to the thoracic wall after Angio–Right and Left mode reconstruction agrees with the hypothesis on the intrathoracic localization of this organ in the rabbit (Harcourt-Brown, 2002). The presented evidence on the multipapillary structure of the two rabbit kidneys differs from the theory about the primitive structure of these organs (Dimitrov and Chaprazov, 2012a,b). On the other hand, the established bean-like shape and the sharply demarcated boundary between the cortex and medulla in the Bones and Skin 3 reconstruction correspond to the existing data on the structure of these organs (Dimitrov and Chaprazov, 2012a,b). We affirm that the medulla renis is built of renal papillae, which does not confirm the unipyramidal structure of the kidney, similar to that in rabbits and rodents (Vella and Donnelly, 2012). The eight papillae, the presence of a distinct crista renalis, the topographic location of the right kidney between Th13 (L1) and L2–L3, and the left kidney between L2 (L3) and L4 (L5) expand the theory (Barone, 2020) about the pyramidal structure of these organs, as well as their topographic location in the abdominal cavity. The obtained data are intravital and largely influenced by the inspiration and expiration phases. The eight papillae and smooth surfaces of both kidneys are consistent with published data describing the smooth and multipyramidal nature of the rabbit kidney (Stamatova-Yovcheva et al., 2022). Our results, showing the anatomical contact between the right kidney and the liver, the proximity of the right kidney with the duodenal pars descendens, the cecum and the colon ascendens, and finally the close proximity of the left kidney to the jejunum and colon descendens, support previously reported information for the kidneys in the rabbit (Barone, 2001; Hristov et al., 2006). We affirm that the used bone landmarks, e.g., Th13 (L1) and L2(L3) for the topographic position of the right kidney and L2 (L3) and L4 (L5) for the topography of the left kidney, are of higher informative value than the others (Barone, 2001; Hristov et al., 2006), according to which the ribs and intercostal spaces are reference anatomical landmarks. The proposed algorithm established the bilateral placement of both kidneys relative to the median plane and the cranial localization of the right kidney relative to the left one. Therefore, our study protocol differed significantly from those of previous studies (Tortora and Derrickson, 2012) that aimed to describe the mutual position of these organs in a dorsoventral aspect. The intravital pre-contrast and post-contrast CT scans offer wider ranges for the topography of the rabbit kidney—from Th13(L1) to L2(L3) for the right one and from L2(L3) to L4(L5) for the left one, compared to Th13-L2 that was previously described (Dimitrov and Chaprazov, 2012a,b). Prospective and retrospective reconstructions established the anatomical contact between the right kidney and the caudate liver lobe, which adds to the research algorithm applied in rabbits (Dimitrov and Chaprazov, 2012a,b). Given the appropriateness of rabbit kidneys in testing and monitoring various drugs, our hypothesis enables us to propose our results as an anatomical imaging model for studying important human diseases (Wu et al., 2003; Saeed et al., 2020). The intravital imaging results obtained are highly informative and can be used successfully in the interpretation and diagnosis of kidney diseases specific to this species (Percy and Barthold, 2007; Harcourt-Brown, 2013). The results accurately reflect the in vivo anatomical visualization of the rabbit kidney. They clearly show the structure of the renal parenchyma and the proximity of the organs to adjacent soft tissue structures. The topographic boundaries of the kidneys are presented in relation to the corresponding anatomical bone markers. Therefore, we propose CT as a modern, intravital, fast, and accurate method for imaging the kidneys, in support of its application in mammals (Yoon et al., 2010). The obtained pre-contrast and post-contrast anatomical images, based on the visualization of the capsula adiposa and capsula fibrosa, the clear demonstration of the renal pelvis, recesses, and crista renalis, the presence of distinct renal papillae, and finally the sharp definition between the kidney’s cortex and medulla were detailed and with high resolution. The presented facts support the hypothesis that CT is an appropriate and highly valuable method for visualizing parenchymal organs (Kleiter et al., 1999; Gielen et al., 2003; Yewon et al., 2023). In our opinion, the application of CT in the study of localization of the rabbit kidneys, their anatomical boundaries, adjacencies, including the contact between the right kidney and the caudate lobe, the intermediate attenuation degree and the lack of difference in the density between the cortex renis and the medulla renis adds up to the statements on the application of this technique in the rabbit (El-Ghazali et al., 2021). The soft tissue normoattenuated homogeneous images of both kidneys correspond to earlier reports on the kidneys in dogs (Serra-Gomez de la Serna et al., 2024). The crista renalis was hypoattenuated relative to the medulla renis in the precontrast study and hyperattenuated in the postcontrast study. This specific feature highlights the advantages of the algorithm used in this study, as well as the comprehensiveness of the results, which contrasts with the description of the crista renalis in dogs (Serra-Gomez de la Serna et al., 2024). The equal density of the cortex renis and medulla renis, as well as the absence of a distinct border between these two structures in prospective reconstructions and the presence of a border in retrospective reconstructions, is different from what was reported for the kidneys in dogs and cats (Oliveira et al., 2013). The CT imaging of the anatomical contact between the right kidney and the proc. caudatus on transverse sections was in wider limits (from Th13 to L3) compared to those described in previous studies on rabbits and chinchillas (Stamatova-Yovcheva et al., 2012; Stamatova-Yovcheva et al., 2013; Dilek et al., 2024), where the topographic relationship between the two organs was reported at the Th13 level. The existing correlation between the length of both rabbit kidneys and the length of the second lumbar vertebra, regardless of the biological subject’s body weight, agrees with a study conducted in dogs (Hoey et al., 2016). Our results, which describe a smaller length of the right kidney compared to the left one and a statistically significantly greater width of the right kidney relative to the left one, contradict the published data in dogs, which report higher values of these macrometric parameters for the right kidney compared to the left one (Atalar et al., 2017). Our data on the successful application of 3D reconstruction in various modes for anatomical imaging of the kidneys in rabbits contribute to the existing information for dogs (Atalar et al., 2017). CT is a definitive method for obtaining real and in vivo information about the macro-dimensions of rabbit kidneys. The obtained results are precise and accurate because the anatomic data are obtained using CT slices and reconstructions. Thus, CT is more accurate than conventional radiology when studying rabbit kidneys (Dorotea et al., 2016). The pre-contrast and post-contrast CT prospective reconstruction as well as 3D retrospective reconstruction allow the acquisition of highly informative anatomical imaging data. ConclusionThe applied CT algorithms demonstrated the topography of the studied organs in relation to the other abdominal organs and vessels and provided data about the renal cortex and medulla. Both kidneys of all examined rabbits were smooth and multi-papillary, with eight papillae. The obtained CT anatomical results can be used as appropriate models for investigation in veterinary and human medicine because the method is rapid and the ionization is minimized. All safety principles are considered. AcknowledgmentsWe would like to express our gratitude to Mrs. Daniela Ivanova, Department of Veterinary Surgery, Faculty of Veterinary Medicine, Trakia University, Stara Zagora, Bulgaria. Conflict of interestThe Authors declare that there is no conflict of interest. FundingThis research was funded by the Bulgarian Ministry of Education and Science in the framework of the Bulgarian National Recovery and Resilience Plan, Component “Innovative Bulgaria,” Project No. BG-RRP-2.004-0006-C02 “Development of research and innovation at Trakia University in service of health and sustainable well-being.” Authors' contributionsConceptualization: Avche Dineva, Kamelia Stamatova-Yovcheva, and Rosen Dimitrov. Validation: Diyana Vladova, David Yovchev, Ömer Gurkan Dilek. Methodology: Hristo Hristov, Vladi Nedev, Nikolay Goranov, and Tihomir Dinev. Data availabilityHowever, data are available from the authors upon reasonable request and with permission from the corresponding author, Kamelia Stamatova-Yovcheva. ReferencesAtalar., Koç, M., Alklay, A. and Hüseyin, H. 2017. Three-Dimensional Analysis with Computed Tomography of Kidneys in the Kangal Dogs. Dicle Üniv. Vet. Fak. Derg. 10(1), 24–29. Balikci, S., Banzato, T., Bellini, L., Contiero, B. and Zotti, A. 2016. Radiographic anatomy of dwarf rabbit abdomen with normal measurements. B. J. V. M. 19(2), 96–107. Barone, R. 2001. Chapitre V. In Anatomie comparée des mammifères domestiques. Barone Paris: Vigot, pp: 843–59. Barone, R. 2020. Chapitre, I.In Anatomie comparée des mammifères domestiques. Splanchnologie II. Barone. and R Paris: Vigot, pp: 3–80. Bensley, B. 1948. Rabbit Dissection. The Urogenital system.In Practical Anatomy of the Rabbit: an Elementary Laboratory Textbook in Mammalian Anatomy. Bensley Toronto: The University Press, pp: 127–33. Brewer. 2006. Biology of rabbit. J. Am. Assoc. Lab. Anim. Sci. 45(1), 8–24. Dilek, O.G., Dimitrov, R., Stamatova‐Yovcheva, K., Ersen, M., Yovchev, D., Sabancı, S.S. and Karakurum, E. 2024. Computed tomography and three-dimensional anatomical study of the liver in the chinchilla (Chinchilla lanigera). Anat. Histol. Embryol. 53(2), e13025; doi: 10.1111/ahe.13025 Dimitrov, R. and Chaprazov, T. 2012. An anatomic and contrast enhanced radiographic investigation of the rabbit kidneys, ureters and urinary bladder. Rev. Med. Vet. 163(10), 469–474. Dimitrov, R., Kostov, D., Stamatova, K. and Yordanova, V. 2012. Anatomotopographical and morphological analysis of normal kidneys of rabbit (Oryctolagus cuniculus). Trakia J. Sci. 10(2), 79–84. Dyce, K., Sack, W. and Wensing, C. 2010. The urogenital apparatus.In Textbook of Veterinary Anatomy. Singh, B.W Philadelphia: W. B. Saunders Company, pp: 167–215. El-Ghazali, H.M., Ammar, S.M.S., Mohamed, S.K.A., Gomaa, M. and Ibrahim, S.M.E. 2021. Topographic and morphometric study on the kidneys of Balady rabbit enhanced by ultrasonographic, radiographic and computed tomography Scan. Int. J. Vet. Sci. Res. 11(1), 28–36. Farhana, S. and Wibowo, F.A. 2019. Anatomical and histological study of male uropoetic organs of common rabbit (Oryctolagus cuniculus). In Proc. Internat. Conf. Sci. Engin. 2, 149–152. Gielen, I., Caelenberg, A. and Van Bree, H. 2003. Computed Tomography (CT) in small animals. Part 1. Technical Aspects. Vlaams Diergeneeskd. Tijdschr. 72(3), 158–167. Harcourt-Brown, F.M. 2013. Diagnosis of renal disease in rabbits. Vet. Clin. Exot. Anim. 16(1), 145–174. Hoey, S.E., Heder, B.L., Hetzel, S.J. and Waller, K.R. 2016. Use of computed tomography for measurement of kidneys in dogs without renal disease. Small. Animals. &. Exotics. 248(3), 282–287. Hristov, H., Kostov, D. and Vladova, D. 2006. Topographical anatomy of some abdominal organs in rabbits. Trakia. J. Sci. 4(3), 7–10. Ji, Y., Hwang, G., Lee, S.J., Lee, K. and Yoon, H. 2023. A deep learning model for automated kidney calculi detection on non-contrast computed tomography scans in dogs. Front. Vet. Sci. 10, 1236579; doi:10.3389/fvets.2023.1236579 Kleiter, M., Henninger, W., Hirt, R. and Lorinson, D. 1999. Portosystemic shunt in a dog - computed tomography as a successful imaging method. Wien. Tierarztl. Monat. 86, 64–70. Oliveira, D.C., Costa, L.A.V.S., Silva, I.C.C., Teixeira, M.W. and Costa, F.S. 2013. Quantitative computed tomography of the renal cortex in healthy dogs and cats. Arq. Bras. Med. Vet. Zootec. 65(4), 1253–1256. Percy, D. and Barthold, S. 2007. Rabbit.In Pathology of laboratory rodents and rabbits. Percy, D. and Barthold, S Oxford: Blackwell Publishing, pp: 253–308. Saeed, M.I., Nicklas, R.D., Kumar, V., Kapoor, R. and Gani, I.Y. 2020. Severe intraoperative anaphylaxis related to thymoglobulin during living donor kidney transplantation. Antibodies 9(3), 43. Serra‐gomez De La Serna, B., Schiborra, F. and Marwood, R. 2024. Prevalence of canine renal crest hyperattenuation in precontrast computed tomography. Vet. Radiol. Ultrasound. 65(4), 352–358. Stamatova Yovcheva, K., Dimitrov, R., Toneva, Y., Yonkova, P., Kostov, D., Rusenov, A., Uzunova, K. and Yordanova, V. 2013. Helical computed tomography application in rabbit liver anatomy: comparison with frozen cross-sectional cuts. Turkish. J. Vet. Anim. Sci. 37(5), 553–558. Stamatova-Yovcheva, K., Dimitrov, R., Dilek, Ö. G., Tsandev, N., Kostadinov, G., Russenov, A. and Hristov, Ts. 2022. A case of multipyramidal kidneys with smooth surface in a New Zealand white rabbit. MAE Vet. Fak. Derg. 7(1), 58-61. Stamatova-Yovcheva, K., Dimitrov, R., Yonkova, P., Russenov, A., Yovchev, D. and Kostov, D. 2012. Comparative imaging anatomic study of domestic rabbit liver (Oryctolagus cuniculus). Trakia J. Sci. 10(1), 57–63. Vella, D. and Donnelly, T.M. 2004. Basic Anatomy, Physiology, and Husbandry.In Ferrets, Rabbits, and Rodents. Clinical Medicine and Surgery. Quesenberry, K. and Carpenter St. Louis: Elsevier Saunders, pp: 157–73. Wu, J., Ge, X. and Fahy, G.M. 2003. Ultrarapid nonsuture mated cuff technique for renal transplantation in rabbits. Microsurgery 23(4), 369–373. Yoon, W., Lee, M.Y., Ryu, J.M., Moon, Y.J., Lee, S.H., Park, J.H., Yun, S.P., Jang, M.W., Park, S.S. and Han, H.J. 2010. Multidetector row computed tomography evaluation of the micropig kidney as a potential renal donor. J. Vet. Sci. 11(1), 9–13. | ||
| How to Cite this Article |
| Pubmed Style Dineva A, Stamatova-yovcheva K, Dimitrov R, Vladova D, Yovchev D, Dilek OG, Hristov H, Nedev V, Goranov N, Dinev T. Computed tomography in the vivo anatomy of rabbit kidneys (Oryctolagus cuniculus). Open Vet. J.. 2026; 16(2): 1297-1314. doi:10.5455/OVJ.2026.v16.i2.47 Web Style Dineva A, Stamatova-yovcheva K, Dimitrov R, Vladova D, Yovchev D, Dilek OG, Hristov H, Nedev V, Goranov N, Dinev T. Computed tomography in the vivo anatomy of rabbit kidneys (Oryctolagus cuniculus). https://www.openveterinaryjournal.com/?mno=297189 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.47 AMA (American Medical Association) Style Dineva A, Stamatova-yovcheva K, Dimitrov R, Vladova D, Yovchev D, Dilek OG, Hristov H, Nedev V, Goranov N, Dinev T. Computed tomography in the vivo anatomy of rabbit kidneys (Oryctolagus cuniculus). Open Vet. J.. 2026; 16(2): 1297-1314. doi:10.5455/OVJ.2026.v16.i2.47 Vancouver/ICMJE Style Dineva A, Stamatova-yovcheva K, Dimitrov R, Vladova D, Yovchev D, Dilek OG, Hristov H, Nedev V, Goranov N, Dinev T. Computed tomography in the vivo anatomy of rabbit kidneys (Oryctolagus cuniculus). Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1297-1314. doi:10.5455/OVJ.2026.v16.i2.47 Harvard Style Dineva, A., Stamatova-yovcheva, . K., Dimitrov, . R., Vladova, . D., Yovchev, . D., Dilek, . O. G., Hristov, . H., Nedev, . V., Goranov, . N. & Dinev, . T. (2026) Computed tomography in the vivo anatomy of rabbit kidneys (Oryctolagus cuniculus). Open Vet. J., 16 (2), 1297-1314. doi:10.5455/OVJ.2026.v16.i2.47 Turabian Style Dineva, Avche, Kamelia Stamatova-yovcheva, Rosen Dimitrov, Diyana Vladova, David Yovchev, Omer Gurkan Dilek, Hristo Hristov, Vladi Nedev, Nikolay Goranov, and Tihomir Dinev. 2026. Computed tomography in the vivo anatomy of rabbit kidneys (Oryctolagus cuniculus). Open Veterinary Journal, 16 (2), 1297-1314. doi:10.5455/OVJ.2026.v16.i2.47 Chicago Style Dineva, Avche, Kamelia Stamatova-yovcheva, Rosen Dimitrov, Diyana Vladova, David Yovchev, Omer Gurkan Dilek, Hristo Hristov, Vladi Nedev, Nikolay Goranov, and Tihomir Dinev. "Computed tomography in the vivo anatomy of rabbit kidneys (Oryctolagus cuniculus)." Open Veterinary Journal 16 (2026), 1297-1314. doi:10.5455/OVJ.2026.v16.i2.47 MLA (The Modern Language Association) Style Dineva, Avche, Kamelia Stamatova-yovcheva, Rosen Dimitrov, Diyana Vladova, David Yovchev, Omer Gurkan Dilek, Hristo Hristov, Vladi Nedev, Nikolay Goranov, and Tihomir Dinev. "Computed tomography in the vivo anatomy of rabbit kidneys (Oryctolagus cuniculus)." Open Veterinary Journal 16.2 (2026), 1297-1314. Print. doi:10.5455/OVJ.2026.v16.i2.47 APA (American Psychological Association) Style Dineva, A., Stamatova-yovcheva, . K., Dimitrov, . R., Vladova, . D., Yovchev, . D., Dilek, . O. G., Hristov, . H., Nedev, . V., Goranov, . N. & Dinev, . T. (2026) Computed tomography in the vivo anatomy of rabbit kidneys (Oryctolagus cuniculus). Open Veterinary Journal, 16 (2), 1297-1314. doi:10.5455/OVJ.2026.v16.i2.47 |