| Review Article | ||
Open Vet. J.. 2026; 16(3): 1458-1467 Open Veterinary Journal, (2026), Vol. 16(3): 1458-1467 Review Article Bovine rotavirus: A narrative review of rotavirus a biology, vaccination strategies, and regional insights from MoroccoHanan El-Ouadi1,2*, Zineb Rhazzar3,4, Imane Choukri1, Nadia Touil2,4,5 and Ouafaa Fassi Fihri11Agronomic and Veterinary Institute Hassan II, Rabat, Morocco 2Mohammed VI Center for Research and Innovation (CM6RI), Rabat, Morocco 3Molecular Virology and Onco-Biology, Faculty of Medicine and Pharmacy of Rabat, Rabat, Morocco 4Cell Culture Unit, Center for Virology, Infectious and Tropical Diseases, Mohamed V Military Training Hospital, Rabat, Morocco 5Royal School of Military Health Service, Rabat BP, Morocco *Corresponding Author: Hanan El-Ouadi. Agronomic and Veterinary Institute Hassan II, Rabat, Morocco. Submitted: 25/11/2025 Revised: 11/02/2026 Accepted: 19/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
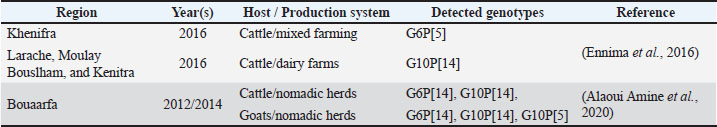
AbstractGroup A rotavirus commonly infects cattle and results in gastrointestinal disorders that are responsible for heavy losses among these animals and within the livestock industry in general. In Morocco, the G10P[14] and G6P[5] genotypes are frequently observed; however, limited data contribute to restricted vaccine choice and disease control. Preventive measures include effective colostrum management practices, hygiene, and vaccination of pregnant cows to pass on passive immunity to newborn calves. Nevertheless, the effectiveness of vaccines in Morocco has not been ascertained because there has been no proper tracking of local variants and their spread. This narrative literature review presents an updated insight into the bovine rotavirus scenario in Morocco and its impact on animal health and the cattle industry. Epidemiological data on circulating genotypes and vaccine performance remain limited and fragmented at the national level, constraining evidence-based vaccine selection and control strategies. Therefore, epidemiological investigations are recommended to identify circulating strains, along with laboratory and field studies assessing vaccine effectiveness, in addition to improved management practices and farmer education. Therefore, using these evidence-based approaches, productivity losses and the vulnerability of the Moroccan livestock industry can be minimized. This paper goes a long way in demonstrating the need for cross-disciplinary work between researchers, veterinarians, and policymakers that will help translate the findings into practical interventions in fields that not only preserve the health of animals but also the farmers’ income in Morocco’s diverse zones. Keywords: Bovine rotavirus, Neonatal calf diarrhea, G and P genotypes, Vaccine efficacy, Morocco. IntroductionRotaviruses are known to be responsible for severe diarrhea in infants and children, as well as in young mammals and avians all over the world. Bovine rotavirus (BRV) is considered to be the leading causative agent of calf diarrhea on a worldwide basis. Among all predisposing causes of diarrhea, BRV is unique in its capacity to account for 62.5% (Uddin Ahmed et al., 2022). Rotavirus diarrhea in calves is highly infectious but short-lived. Factors such as age, immunity, virus dose, other infections, and stress affect its severity. Serious complications include kidney failure and electrolyte imbalance, which may result in dehydration and death (Gichile 2022). Rotavirus is a non-enveloped virus with an Ribonucleic Acid (RNA) genome, which is segmented into 11 parts that encode both structural and non-structural proteins (Caddy et al., 2021). Among the different rotavirus groups, group A rotavirus (RVA) is the most relevant in cattle and is categorized in the genus rotavirus in the family Sedoreoviridae. They are grouped based on the antigenic and genetic differences in the outer capsid antigens, VP7 and VP4, as well as the inner capsid protein, VP6. Neutralizing antibodies target two outer capsid proteins, VP4 and VP7 (Dhama et al., 2009; Uddin Ahmed et al., 2022). The transmission of RVA between different host species, together with genetic reassortment, recombination, and point mutations, drives viral diversity and the emergence of new genotypes (Desselberger, 2014; Dóró et al., 2015). In Morocco, RVA transmission is influenced by specific livestock management practices, particularly in nomadic regions such as Bouaarfa, where close contact between humans and multiple animal species favors interspecies transmission and genetic reassortment (Alaoui Amine et al., 2020). The control of BRV relies primarily on preventive measures, including adequate colostrum intake, hygiene management, and vaccination of pregnant cows to ensure passive immunity in calves (Corl et al., 2008; Izzo et al., 2011; Gichile, 2022). However, in Morocco, challenges remain regarding vaccine strain selection and the limited availability of field-based data evaluating vaccine effectiveness under local conditions (Saif and Fernandez, 1996; Lopez and Heinrichs, 2022). This narrative literature review aims to summarize the current information regarding BRV in Morocco and its effects on health and the economy, given the limited epidemiological data available at the national level. We will also discuss the possible ways of preventing the disease with a focus on selecting the appropriate vaccine to be administered to the Moroccan cattle population. The following recommendations were made to strengthen disease control and minimize its impacts on cattle farming in Morocco. Literature search strategyThis narrative review is based on a literature search conducted between 1970 and 2025 using PubMed, Google Scholar, and Moroccan institutional databases. The keywords were related to the structure and classification of rotavirus, genetic diversification, BRV, epidemiology, genotype distribution, clinical manifestations, diagnostic techniques, maternal immunity, prophylactic and vaccination strategies, and zoonotic transmission. Eligible sources included peer-reviewed original research and relevant review articles; official regulatory and normative documents were also consulted. Studies focusing exclusively on human rotavirus without comparative relevance and those addressing aspects outside the defined thematic scope were excluded. Older studies were retained when they provided essential baseline data, particularly for Morocco, where surveillance data remain limited. More recent studies were used preferentially to discuss current classification, genotype dynamics, and vaccination strategies, while international data were included for comparative and contextual purposes. Group A rotavirusGroup A rotaviruses belong to the family Sedoreoviridae, order Reoviridae, and genus Rotavirus. Rotaviruses are icosahedral and non-enveloped viruses with 11 segments of double-stranded RNA (16–21 kbp) that are well protected by an inner and outer capsid layer. The virus has structural proteins located in the core (VP1, VP2, and VP3), inner capsid (VP6), and outer capsid layer (VP4 and VP7) (Dhama et al., 2009). Based on VP6 antigenic and genetic divergence, rotaviruses are currently classified into at least nine officially recognized species (RVA–RVI), with a tenth species (RVJ) proposed following the identification of genetically distinct rotavirus strains in bats. Among these, RVA to rotavirus C (RVC), rotavirus E (RVE), RVH, and RVI are known to infect mammals. RVA is the most widespread species in most mammalian hosts (Bányai et al., 2017) and is initially known to affect newborn calves. Several studies have reported that RVA accounts for the majority of BRV infections, often exceeding 90% in specific regions and study populations, while groups B and C are detected less frequently (Geletu et al., 2021). The nomenclature of the viral proteins identifies the structural proteins as VP and the nonstructural proteins as non-structural protein, followed by sequential numbers ranging from 1 to 6. The examination of gene coding regions reveals that there are six structural proteins (VP1–VP4, VP6, and VP7) and six nonstructural proteins (NSP1–NSP6). The viral particle is constructed from structural proteins, while NSPs play roles in the viral replication cycle or in interacting with host proteins to affect pathogenesis or the immune response (Geletu et al., 2021). In addition to VP6, two outer capsid proteins, VP4 and VP7, have historically been employed in the classification of subspecies. In order to separate group A rotaviruses into distinct serotypes, neutralizing antibodies were elicited by both VP7 and VP4, with VP7 determining the G-serotype (glycosylated protein) and VP4 the P-serotype (protease-sensitive protein) (Ericson et al., 1982; Lopez et al., 1985; Hoshino and Kapikian, 2000; Aoki et al., 2009). Genotypes classification and their diversificationRotavirus was initially classified into six serogroups: rotavirus A (RVA), rotavirus B (RVB),RVC, rotavirus D, RVE, and rotavirus F, depending on the viral capsular antigens identifiable by the serological reactions. Therefore, it is possible to refer to groups A–C for both animals and people, and groups D–F are only characteristic for animals (Damasceno et al., 2020). Currently, based on the antigenic and genetic homology of the VP6 protein, rotaviruses are classified into at least nine officially recognized species (RVA-RVI), with a tenth one (RVJ) having been proposed following the identification of genetically distinct strains (Bányai et al., 2017). The neutralization reactions of antibodies using antisera from animals that were pre-immunized with VP4 or VP7 proteins of the outer capsid of the rotavirus have also been used in the categorization of the virus into serotypes (Costa et al., 2004). G serotypes are determined by the epitopes that are located on the surface protein of VP7, while P serotypes are related to the protease-sensitive protein of VP4 (Costa et al., 2004). The current rotavirus classification system uses alphabets to define each RNA segment and protein, namely VP7-VP4-VP6-VP1-VP2-VP3 and NSP1 to NSP5/6. These are represented in the sequence Gx-P[x]-Ix-Rx-Cx-Mx-Ax-Nx-Tx-Ex-Hx, where ‟x” represents the Arabic numerals from one up to a certain (Carvalho-Costa et al., 2019). Due to the fact that point mutations occur in the genes of VP4 and VP7 during the evolution process, they have specific or atypical antigens. This enhances the genetic variation of the rotavirus strains, thereby enabling some of them to escape neutralization by the antibodies that are elicited by the vaccines (Sadiq et al., 2022). A single strain may be seen dominating a population for 2 years before being succeeded by a new emerging strain. Understanding rotavirus evolution, strain reassortment, and the corresponding evolutionary pressure in coinfection has significant implications for disease management (Sadiq et al., 2022). Inter-species transmission and genetic reassortment are the two major forces that govern the genetic variability of rotavirus and the generation of new pathogenic strains with altered pathogenicity. Furthermore, rotavirus spreads through the fecal-oral route or through direct contact (Li et al., 2016). Rotavirus A transmission between different host species contributes to the emergence of novel viral strains, increasing viral diversity in both humans and animals. However, there are other ways that lead to the development of rotavirus, such as gene reassortment, gene recombination, and the accumulation of point mutations (Desselberger, 2014). The interaction of two of these mechanisms between RVAs of different species can yield the generation and spread of new genotypes (Dóró et al., 2015). Additionally, the gene reassortment between animal and human strains adds to this genetic variability and thus calls for constant surveillance to control the viruses. However, analysis of the complete genome of animal strains (bovine S18 and caprine S19) revealed a close genetic link with the Moroccan human strain MA31. Phylogenetic analysis showed that the human strain MA31 was the result of reassortment between human and animal rotaviruses, involving interspecies transmission of RVA strains belonging to cattle, goats, and camelids. The large number of BRV serotypes makes it difficult to develop effective vaccines, and the strains used are not necessarily representative of those circulating in areas where vaccination is needed (Alaoui Amine et al., 2020). RVA transmission is influenced by specific livestock management practices, particularly in nomadic regions such as Bouaarfa, where close contact between humans and multiple animal species favors interspecies transmission and genetic reassortment (Alaoui Amine et al., 2020). Clinical symptoms in calvesRotavirus diarrhea in calves is an acute disease, and the incubation period is between 12 and 96 hours. Clinical manifestations typically progress from mild watery diarrhea and anorexia to dehydration, followed in more severe cases by electrolyte imbalance and metabolic acidosis. Although most infections are self-limiting, morbidity is generally high, and severe cases may result in death due to dehydration and cardiac failure. Variations in clinical disease in calves are attributable to several factors, such as differences in the pathogenicity of rotavirus strains, host age and immune response, amount of the inoculum used in challenge, presence of other coinfecting agents, and environmental factors, including weather, housing, overcrowding, and feeding status (Geletu et al., 2021). Laboratory diagnosisThe diagnosis of rotavirus in calves should be reliable to prevent and treat the illness properly. As clinical examination is not conclusive in establishing the causative agent, it becomes mandatory to complement it with laboratory testing. Several tests can be used for this purpose. In most cases, the identification of rotavirus is performed through viral description and isolation from stool specimens. Antigen-captured enzyme-linked immunosorbent assay is a quick method of identifying pathogens in clinical specimens using antibodies that capture the antigens. Currently, molecular methods such as reverse transcription polymerase chain reaction are considered the most sensitive and widely used reference technique for rotavirus detection and genotyping, while virus isolation in cell culture is mainly reserved for research and strain characterization (Gichile, 2022). Polymerase chain reaction (PCR) is a method of identifying rotavirus by amplification of particular viral genes with specific primers, whereas double-stranded RNA can be identified by extraction, agarose gels, and silver staining. Viral strains are differentiated by examining the relative sizes of segmented RNA during electrophoresis. Although electron microscopy (EM) is a very effective technique for visualizing non-cultivable bovine enteric viruses, including bovine RVA, the technique is limited by its high cost and the need for specialized personnel. Consequently, EM is not routinely applied in most diagnostic laboratories. Currently, molecular methods such as PCR are the most widely used tools for rotavirus detection (Gichile 2022). Mechanism of calf immunityThe high risk of infection of neonates in the first months of life is one of the biggest problems in calf raising. Although neonatal calves possess a complete immune system at birth, it is functionally immature, particularly concerning adaptive immune responses. All the organs and immune cells are present in the neonates, yet, because of the immaturity of the protective mechanisms, they cannot develop an adequate immune response, and the time it takes to initiate and produce the required mechanisms for humoral and cell-mediated immunity (Cortese, 2009). In this case, the animal’s age will be the major factor that will define the effectiveness of the vaccination response. This means that the newborn calf is immunocompetent, but it may take 5–8 months for the calf’s immune system to reach maturity so that the optimal response can be elicited (Cortese, 2009). In addition, maternally derived antibodies acquired through colostrum may provide early protection against rotavirus infection while potentially interfering with the response to active vaccination in young calves. The importance of colostrum in early immune protection was further confirmed in a recent study evaluating maternal transmission of RVA and the microbial composition of colostrum in Holstein versus Hanwoo cattle (Kim et al., 2024). To have a successful colostrum management program, the producer must ensure that calves receive sufficient clean and high-quality colostrum within the first few hours of life (Godden, 2008). The quality, quantity, and speed of colostrum delivery are identified as the three major factors that define the outcomes of the passive transfer program. IgG concentration in colostrum is considered the best criterion of colostrum quality. Good practices of colostrum feeding include providing cows with colostrum that contains greater than 50 g/l of IgG to the calf within the first few hours of birth and/or within the first two hours of life and offering enough colostrum that it equals 10%–12% of the calf’s live weight (Denholm et al., 2023). This is in the range of 3–4 l, which is recommended for a Holstein calf (Godden, 2019). Additionally, colostrum should be fed to calves as soon as possible after birth to allow the calves to be able to absorb immunoglobulins. Open gut is a phenomenon that characterizes the capacity of neonatal enterocytes to non-specifically transport large molecules through pinocytosis (Broughton and Lecce, 1970). Early susceptibility to rotavirus infection and limited vaccination efficacy in newborns are partially related to the limited development of innate antiviral defenses in the neonatal intestine (López et al., 2016). Recommended prophylaxis and supportive careRotavirus infections do not have specific antiviral therapy; thus, a primary focus should be on supportive care. This attention would include the systematic substitution of fluids to alleviate the sequelae of diarrhea and vomiting, careful addressing of the acid-base and electrolyte imbalances, and rational use of adequate rehydration solutions. Fluid administration is done through esophageal catheters in neonates and intravenously in older subjects (Gichile 2022). To decrease the incidence of disease in the herd, the careful breeder must pay attention to colostrum transmission, environmental cleanliness, and the number of stress factors, such as overcrowding or feeding. Pregnant cows should be vaccinated against RVA according to manufacturer-recommended protocols, which commonly include administration at approximately 60 and 30 days before calving, although schedules may vary depending on the vaccine formulation and local practices (Izzo et al., 2011; Gichile, 2022). Bovine rotavirus can persist even in farms with good hygiene practices due to limited maternal immunity and the circulation of multiple genotypes. Continuous molecular monitoring remains essential to detect reassortant strains and to support effective vaccination strategies (Hasan et al., 2022). The recommended herd-level strategy combines proper management and vaccination of the dam a few weeks prior to calving. This approach has been successfully implemented tactically in various countries across the world (Dhama et al., 2009). The vaccinated dam should passively seal the specific antibodies within the colostrum to the young ones, affording adequate immunity just at the intestinal level (Agrawal et al., 2002; Steele et al., 2004). In a recent study, BRV was found to be bound by recombinant bovine ultra-long C-DR H3 antibodies as a match, and the evidence is in the form of Enzyme-Linked Immunosorbent Assay and neutralization tests. Under experimental conditions, these antibodies reduce diarrhea, reduce viral shedding, and increase survival rates in calves. Although these findings are promising, this approach remains experimental, and further studies are required to evaluate its safety, efficacy, and feasibility before it can be considered a choice in the prevention and treatment of BRV infections (Zhao et al., 2025). Studying the risk factors associated with BRV infection contributes to a better understanding of transmission dynamics within herds. Parameters such as herd size, colostrum management, and hygiene conditions directly influence the likelihood of infection and the severity of neonatal diarrhea (Uddin Ahmed et al., 2022). These factors condition the success of prophylactic strategies, including vaccination programs, by modulating passive immunity transfer and infection pressure at the herd level. Impact of vaccines on BRV genotypes in circulationSeveral international studies have shown that G6, G10, and G8, in combination with P[1], P[5], P[11], P[15], and P[21], are the most epidemiologically relevant RVA genotypes in cattle worldwide (Martella et al., 2010). This provides key information for the development of appropriate bovine vaccines. The most effective rotavirus vaccination strategy in cattle involves promoting higher and prolonged levels of protective anti-rotavirus antibodies in the mammary secretions of vaccinated pregnant cows, thereby conferring passive protection to newborn calves (Crouch et al., 2001). Indeed, it has been demonstrated that when calves are fed colostrum from vaccinated mothers during the first hours of life, until they develop their own immunity, the specific anti-rotavirus antibodies present in the colostrum reduce the incidence of rotavirus-induced diarrhea and viral shedding in infected calves (Kaplon et al., 2013). Bovine rotavirus vaccine does not contribute to the development of new rotavirus strains with genotypes or variants that are not included in the vaccine (Kaplon et al., 2013). Furthermore, it does not put selective pressure that leads to the development of other pathogens, such as caliciviruses implicated in diarrhea in calves. This narrative underscores the effectiveness of the vaccine in controlling diarrheal infections without passing on other disease-associated pathogens (Kaplon et al., 2013). However, the detection of new rotavirus genotypes not included in current vaccines reflects an antigenic difference between vaccine strains and circulating field strains rather than a vaccine-induced viral mutation. Some recent studies have shown that other new genotypes of rotavirus that are not included in the vaccine have appeared, and therefore, vaccines need to be modified to cover the expanded range of strains and genotypes (Papp et al., 2013; Desselberger, 2017; Amin et al., 2024; Jampanil et al., 2025). Two BRV genotypes, G6P[11] and G10P[11], were detected in six vaccinated dairy cattle herds that were vaccinated with G6P[5] (Fritzen et al., 2019). Although this result exhibits a high level of passive immunity against the homologous BRV strain of the vaccine, it also identifies heterologous infections. However, in other papers, we found that the overall frequency and severity of neonatal diarrhea were significantly decreased in all herds (Fritzen et al., 2019). The current study highlights the relevance of systematic sampling and genomic sequencing of BRV strains in neonatal diarrhea outbreaks in dairy herds to not only estimate the current control program and the cost-effectiveness of BRV vaccination approaches but also identify possible pathogenic new strains with cross-species infection potential (Fritzen et al., 2019). Choice of the vaccine strainThe choice of the vaccine strain is normally determined by the strains that have been identified in the field, with the aim of achieving the best vaccine results. However, such genetic variation remains an issue that global health officials and scientists have yet to overcome when developing vaccines with broad and consistent cross-protection against multiple circulating strains (Golaviya et al., 2024). The detection of new G/P genotypes in cattle due to genetic mutation or genetic shift suggests that humans and other animals may act as reservoirs of BRV evolution. Hence, the identification of the genotype(s) of the BRV strain(s) associated with the infections is useful in the development of vaccines and in reducing the incidence of severe dehydrating diarrhea in young calves (Golaviya et al., 2024). Fluctuations in the prevalence of different viral strains have been reported. However, a dominant genotype (G6P[5]) has been frequently identified as one of the major circulating genotypes in certain regions and production systems, although its prevalence varies geographically and temporally (Papp et al., 2013; Shin et al., 2023). In the Moroccan context, where genotype data remain limited to a small number of regional studies (Ennima et al., 2016; Alaoui Amine et al., 2020), this epidemiological pattern conceals a crucial vaccine challenge: The mismatch between the genotypes present in the commercially administered vaccine and those of the rotavirus strains currently circulating in both vaccinated and unvaccinated herds serves to underscore the vital necessity for unceasing monitoring to forestall potential causes of vaccine failure (Rocha et al., 2017). The genetic diversity of BRV varies across production systems and regions, reflecting distinct evolutionary lineages and historical viral exchanges. This highlights the need for continuous molecular surveillance to update vaccines according to locally circulating strains (Louge Uriarte et al., 2023). Evolution of BRV vaccination strategiesThe BRV vaccine aims to induce mucosal immunity in the small intestine of calves. In some studies, an oral, live, attenuated formulation was used on newborn animals and gave promising results. However, its efficiency was significantly reduced in the case of a double-blind trial, primarily due to the availability of maternal antibodies and logistical challenges linked to large herds. The risk is that the calves will contract virulent viral strains before developing a sufficient immune response, thereby undermining the overall effectiveness of the vaccinations (Saif and Fernandez, 1996). Due to the failure of the oral rotavirus vaccine, the decision was taken to pursue a passive immunization strategy. This involves the administration of an antigen to pregnant cows to increase the levels of antibodies against rotavirus in colostrum and milk (Saif and Fernandez, 1996). Antibodies are transferred to calves during lactation, providing temporary protection. However, antibody levels rapidly decline in milk, leaving young calves vulnerable to BRV-induced diarrhea. Given that serum-derived Immunoglobulin G subclass 1 is the predominant antibody to BRV in colostrum and milk, parenteral vaccination of cows is a logical approach to increase these antibodies in mammary secretions and prolong their presence in milk, thus offering better protection to calves (Saif and Fernandez, 1996; Pinheiro et al., 2022). More recent field-based studies, mainly evaluating inactivated multivalent vaccines used for maternal immunization, have consistently demonstrated a significant reduction in the severity of neonatal diarrhea, morbidity, and viral shedding in vaccinated herds. Nevertheless, these studies also showed that vaccination does not completely prevent BRV infection or viral circulation, particularly when circulating field genotypes differ from vaccine strains (Kaplon et al., 2013; Fritzen et al., 2019). Molecular analyses further highlighted the persistence and emergence of heterologous BRV genotypes in vaccinated herds, emphasizing the influence of local epidemiological context and viral genetic diversity on vaccine performance (Fritzen et al., 2019; Louge Uriarte et al., 2023). Optimized inactivated multivalent vaccines used for maternal immunization represent the most advanced and widely applied for BRV strategy control under field conditions, while improvements mainly focus on vaccination schedules, strain selection, colostrum management, and molecular surveillance (Kaplon et al., 2013; Fritzen et al., 2019; Pinheiro et al., 2022; Louge Uriarte et al., 2023). In parallel, alternative and next-generation approaches, including DNA vaccines, virus-like particles, subunit vaccines, and recombinant bovine antibodies such as ultra-long Complementarity-Determining Region H3 antibodies, have been explored. Although these strategies have shown promising results in the experimental setting, including reduced viral shedding and disease severity, they remain experimental or adjunct approaches and have not yet been implemented as routine vaccines in cattle (Dhama et al., 2009; Zhao et al., 2025). One health approach to zoonosisNew genotypes (G6P[14] and G10P[15]) and a known genotype (G8P[14, 12]) were revealed in free-ranging ruminants in Slovenia between 2017 and 2021. G6P[14] was identified in red deer and roe deer, G8P[14] in chamois, and G10P[15] in red deer and roe deer. Genotypes G6P[14] and G8P[14] have zoonotic features and are closely related to human RVA strains. The source of RVA remains a mystery (Šenica et al., 2024). Zoonotic transmission and gene segment reassortment between human and animal rotavirus strains increase the diversity of human rotavirus strains (Luchs et al., 2012). The literature on zoonotic rotavirus transmission in animals is limited, mainly because of the lack of genomic sequencing information. Enhanced monitoring of rotavirus in domestic animal populations is essential to improve understanding of its epidemiology. Future studies should focus on the prevalence of rotavirus infection and circulating BRV genotype distribution among nomadic livestock in Morocco. It is recommended to implement maternal rotavirus vaccination programs against nomadic calves and goats, as well as create a strong surveillance system to allow rotavirus detection and genotyping (Alaoui Amine et al., 2020). Zoonotic transmission events, combined with environmental dissemination through contaminated water, food, and surfaces, create favorable conditions for viral persistence, interspecies transmission, and genomic reassortment. This interconnected transmission network complicates the identification of the primary source of infection and contributes to the continuous diversification of human rotavirus strains, particularly in regions where close human–animal contact and inadequate sanitation coexist (Sadiq and Khan, 2023). Neonatal diarrhea in calves in MoroccoLivestock production in Morocco is a productive sector of undeniable socio-economic interest, accounting for approximately 34% of agricultural GDP, 4% of total GDP, contributing 30% of agricultural added value, and providing income for 2/3 of the active rural population (Onicl, 2023). However, this livestock sector is constantly threatened by infectious diseases, which are a major obstacle to maintaining and improving the productivity of livestock farms. Animal diseases are a real brake on the development of livestock farming and therefore on its contribution to the economy (Dhama et al., 2009). The RVAs, alone or in combination, are the major causes of viral-induced neonatal diarrhea and are a significant cause of mortality and morbidity in newborn calves (Berber et al., 2021; Ghonaim et al., 2025). Rotavirus is reported as the most common cause of diarrhea in calves less than 1 month old (Reynolds et al. 1986; Snodgrass et al. 1986; Radostits et al., 2007). Neonatal diarrhea is the leading cause of morbidity and mortality in newborn calves in most countries in the world (Radostits et al., 2001). It is a worldwide problem with considerable economic impact. In Morocco, neonatal diarrhea is responsible for 10%–14% of preweaning calf mortality and 20% of lamb and kid mortality (Zouagui et al., 2017). In an earlier study, neonatal diarrhea in calves and lambs was predominantly caused by rotavirus (29.7% and 30% prevalence, respectively) and Escherichia coli K99 (26.9% and 10%, respectively), with cases declining markedly after 20 days in calves and 11 days in lambs, although 34.8% and 55% of cases remain etiologically unexplained (Fassi Fihri et al., 1988). More recent studies have provided laboratory-based confirmation of RVA circulation in Moroccan cattle, as group A rotavirus was successfully isolated and identified from neonatal diarrheic calves, reinforcing the role of RVA as an active etiological agent in current field conditions (Ennima et al., 2016). Moroccan nomadic livestock systems represent an understudied reservoir of rotavirus due to herd mobility and interspecies contact, promoting the emergence of potentially zoonotic strains (Alaoui Amine et al., 2020). Historical serological surveys have also suggested that RVA infections persist under two different transmission paths. The introduction occurs through imported cattle and by local breed circulation, which can be verified by serological detection in specific herds. Seropositive samples predominate at 65% in intensive animal farming, 40% in small-scale farms, and 79% in imported Friesian calves (Schwers et al., 1984). Taken together, these findings indicate that rotavirus-associated neonatal diarrhea remains a persistent and economically significant problem in Morocco. The limited availability of recent national data underscores the need for integrated clinical, virological, and molecular surveillance to better characterize current BRV dynamics and guide control strategies. Bovine rotavirus serotypes in MoroccoAvailable data on BRV genotypes in Morocco remain limited and are largely based on a small number of regional studies conducted over the past decade, highlighting the need for updated national surveillance epidemiology (Table 1). Table 1. Rotavirus A genotypes in cattle and small ruminants in Morocco.
A study conducted in Morocco in 2016 described the distribution of G6P [5] in the field in the Khenifra region and G10P[14] on dairy farms in Laaraych, Moulay Bouslham, and Kenitra (Ennima et al., 2016). In 2012 and 2014, caprine and BRVs isolated from nomadic livestock in the Bouaarfa region revealed the existence of three circulating genotypes G6P[14], G10P[14], and G10P[5] in ruminants, with a predominance of G6P[14] (Alaoui Amine et al., 2020). BRV is not listed among the diseases in Chapter 1.3 of the World Organization for Animal Health’s terrestrial animal health code, which defines internationally notifiable terrestrial animal diseases (World Organization for Animal Health, 2023). In Morocco, BRV is not included among the contagious diseases subject to mandatory reporting under Dahir No. 1-75-292 issued by the National Office for Food Safety (Royaume du Maroc, 1977). Consequently, no specific mandatory control measures are applied. BRV control is indirectly insured through general animal health and biosecurity regulations, while veterinary rotavirus vaccines are authorized and listed under the supervision of the ONSSA. Although this regulatory situation is consistent with global practice, the lack of structured national surveillance and genotype-based monitoring limits the assessment of vaccine strain matching and field efficacy under Moroccan conditions. Vaccines used in MoroccoVeterinary vaccines against rotavirus in cattle on the Moroccan market are inactivated mixed vaccines against neonatal diarrhea containing other agents, especially coronavirus and Escherichia coli available vaccines do not all contain identical rotavirus strains or genotypes. Authorized formulations include vaccines based on G6 and/or G in general, the rotavirus strain used genotypes, G6P[5] strains, as well as products formulated with reference BRV strains such as Lincoln (G6[1]) or Rol strain (G6 lineage), depending on the manufacturer according to the positive list of veterinary drugs issued by the “Office National de Sécurité Sanitaire des Produits Alimentaires” (ONSSA, 2025). Studies on the epidemiology of rotavirus in Morocco are still scarce; therefore, estimating the incidence of rotavirus infection and its effect on animal health is difficult. The available epidemiological data are limited to a few regional studies, which are detailed in Section “Bovine rotavirus serotypes in Morocco,” these studies were conducted in specific geographic areas and time periods and do not provide nationwide prevalence, genotype distribution, or temporal trend data. These data are not consolidated and may not encompass all parts of the country. Besides, the rotavirus vaccine, while available and in use, has not been established as effective in particular states within the regions. Lack of information regarding the national levels of vaccination and the real influence of the vaccination campaign on the reduction of the frequency of infections makes it possible to remain rather skeptical about the role of vaccination in controlling rotavirus in animals in Morocco. ConclusionBRV remains an important contributor to neonatal calf diarrhea in Morocco, with measurable implications for animal health and livestock productivity. Although genotypes such as G6P[5], G10P[14], and G6P[14] have been documented, current epidemiological data are geographically limited and do not yet allow a comprehensive assessment of national genotype dynamics. Considering the segmented genome of rotavirus and its capacity for genetic reassortment and interspecies transmission, particularly within nomadic and mixed production systems, continuous molecular monitoring is essential to better characterize circulating strains and anticipate evolutionary shifts. Maternal vaccination combined with optimized colostrum management constitutes the primary preventive strategy currently applied. However, the effectiveness of structured postmarketing evaluation of vaccines and systematic assessment of vaccine strain matching under Moroccan field conditions remains limited. Furthermore, while the national and international regulatory framework does not classify BRV as a notifiable disease, the absence of coordinated national surveillance constrains evidence-based vaccine selection and long-term control planning. Therefore, strengthening BRV management requires the integration of systematic molecular surveillance, harmonized diagnostic capacity, structured epidemiological reporting, and periodic reassessment of vaccine composition in relation to circulating strains. Enhanced collaboration among academic institutions, veterinary services, and regulatory authorities would support data-driven decision-making and improve herd-level disease control. Within a One Health perspective, the documented interspecies transmission potential of rotaviruses further supports the implementation of longitudinal surveillance programs linking livestock, wildlife, and environmental interfaces. Establishing a coordinated national BRV monitoring framework would strengthen preventive strategies and contribute to Morocco’s livestock sector’s resilience and sustainability in the context of evolving viral diversity. AcknowledgmentThe authors would like to acknowledge the Agronomic and Veterinary Institute Hassan II (IAV Hassan II) and the Mohammed VI Center for Research and Innovation (CM6RI) for providing a supportive academic and research environment that facilitated the preparation of this review. Conflict of interestThe authors declare no conflict of interest. FundingThis study received no specific grant. Authors' contributionHEO made the study concept, performed the literature search, and prepared the original manuscript. It was interpreted with the help of ZR and IC and critically revised by NT and OFF. Data availabilityAll data supporting this study’s findings are available within the manuscript. ReferencesAgrawal, D.K., Singh, N.P. and Chauhan, R.S. 2002. Colostral antibodies against rotavirus infection in neonatal calves. J. Immunol. Immunopathology. 4(1and2), 107–109. Alaoui Amine, S., Melloul, M., El Alaoui, M.A., Boulahyaoui, H., Loutfi, C., Touil, N. and El Fahime, E. 2020. Evidence for zoonotic transmission of species A rotavirus from goat and cattle in nomadic herds in Morocco, 2012–2014. Virus Genes 56(5), 582–593. Amin, A.B., Cates, J.E., Liu, Z., Wu, J., Ali, I., Rodriguez, A., Panjwani, J., Tate, J.E., Lopman, B.A. and Parashar, U.D. 2024. Rotavirus genotypes in the postvaccine era: a systematic review and meta-analysis of global, regional, and temporal trends by rotavirus vaccine introduction. J. Infect. Dis. 229(5), 1460–1469. Aoki, S.T., Settembre, E.C., Trask, S.D., Greenberg, H.B., Harrison, S.C. and Dormitzer, P.R. 2009. Structure of rotavirus outer-layer protein VP7 bound with a neutralizing Fab. Science 324(5933), 1444–1447. Bányai, K., Kemenesi, G., Budinski, I., Földes, F., Zana, B., Marton, S., Varga-Kugler, R., Oldal, M., Kurucz, K. and Jakab, F. 2017. Candidate new rotavirus species in Schreiber's bats, Serbia. Infect. Genet. Evol. 48, 19–26. Berber, E., Çanakoğlu, N., Sözdutmaz, I., Simsek, E., Sursal, N., Ekinci, G., Kökkaya, S., Arıkan, E., Ambarcıoğlu, P., Göksu, A. and Keleş, I. 2021. Seasonal and age-associated pathogen distribution in newborn calves with diarrhea admitted to ICU. Vet. Sci. 8(7), 128. Broughton, C.W. and Lecce, J.G. 1970. Electron-microscopic studies of the jejunal epithelium from neonatal pigs fed different diets. J. Nutr. 100(4), 445–449. Caddy, S., Papa, G., Borodavka, A. and Desselberger, U. 2021. Rotavirus research: 2014–2020. Virus Res. 304, 198499. Carvalho-Costa, F.A., De Assis, R.M.S., Fialho, A.M., Araújo, I.T., Silva, M.F., Gómez, M.M., Andrade, J.S., Rose, T.L., Fumian, T.M., Volotão, E.M., Miagostovich, M.P. and Leite, J.P.G. 2019. The evolving epidemiology of rotavirus A infection in Brazil a decade after the introduction of universal vaccination with Rotarix®. BMC. Pediatr. 19, 42. Corl, B.A., Oliver, S.A.M., Lin, X., Oliver, W.T., Ma, Y., Harrell, R.J. and Odle, J. 2008. Conjugated linoleic acid reduces body fat accretion and lipogenic gene expression in neonatal pigs fed low-or high-fat formulas. J. Nutr. 138, 449–454. Cortese, V.S. 2009. Neonatal immunology. Vet. Clinics North Amer. Food Anim. Pract. 25(1), 221–227. Costa, P.S.S., Cardoso, D.D.P., Grisi, S.J.F.E., Silva, P.A., Fiaccadori, F., Souza, M.B.L.D. and Santos, R.A.T. 2004. Rotavirus A infections and reinfections: genotyping and vaccine implications. J. J. Pediatr. 80, 119–122. Crouch, C.F., Oliver, S. and Francis, M.J. 2001. Serological, colostral and milk responses of cows vaccinated with a single dose of a combined vaccine against rotavirus, coronavirus and Escherichia coli F5 (K99). Vet. Rec. 149(4), 105–108; doi:10.1016/j.vr.2001.01.009 Damasceno, J.L., Andrade, G., Santiago, M.B., Martins, C.H.G. and Pires, R.H. 2020. Rotavirus: a review and emergence of new genotypes. Revista Brasileira Multidisciplinar 23, 173–189. Denholm, K., Baxter-Smith, K., Simpson, R. and Williams, P. 2023. A survey of colostrum management practices on UK dairy farms. Livestock 28(4), 150–159. Desselberger, U. 2014. ‘Rotaviruses’. Virus Res. 190, 75–96. Desselberger, U. 2017. Differences in rotavirus vaccine effectiveness by country: likely causes and contributing factors. Pathogens 6(4), 65; doi:10.1016/j.pathogens.2016.09.010 Dhama, K., Chauhan, R.S., Mahendran, M. and Malik, S.V.S. 2009. Rotavirus diarrhea in bovines and other domestic animals. Vet. Res. Commun. 33(1), 1–23. Dóró, R., Farkas, S.L., Martella, V. and Bányai, K. 2015. Zoonotic transmission of rotavirus: surveillance and control. Expert. Rev. Anti-Infect. Ther. 13, 1337–1350. Ennima, I., Sebbar, G., Harif, B., Amzazi, S., Loutfi, C. and Touil, N. 2016. Isolation and identification of group A rotaviruses among neonatal diarrheic calves, Morocco. BMC. Res. Notes. 9(1), 1–6; doi: 10.1016/j.bmcrn.2016.01.003 Ericson, B.L., Graham, D.Y., Mason, B.B. and Estes, M.K. 1982. Identification, synthesis, and modifications of simian rotavirus SA11 polypeptides in infected cells. J. Virol. 42(3), 825–839. Fassi Fihri, M., Johnson, D. and Taoudi, A. 1988. Epidemiologie des diarrhees a Escherichia coli et a rotavirus chez le veau et l’agneau au Maroc. Annales. De. Recherches. Vétérinaires. 19, 59–64; doi:10.1016/j.anvrv.1988.1.59 Fritzen, J.T.T., Lorenzetti, E., Oliveira, M.V., Bon, V.R., Ayres, H., Alfieri, A.F. and Alfieri, A.A. 2019. Cross-sectional study of the G and P genotypes of rotavirus A field strains circulating in regularly vaccinated dairy cattle herds. Trop. Anim. Health. Prod. 51(4), 887–892. Geletu, U.S., Usmael, M.A. and Bari, F.D. 2021. Rotavirus in calves and its zoonotic importance. Vet. Med. Intl. 2021, 6639701. Ghonaim, A.H., Lei, M., Zeng, Y., Xu, Q., Hong, B., Li, D., Yang, Z., Zhou, J., Liu, C., He, Q., Zhang, Y. and Li, W. 2025. Molecular surveillance and whole genomic characterization of bovine rotavirus A G6P 1 reveals interspecies reassortment with human and feline strains in China. Vet. Sci. 12(8), 742. Gichile, A.G. 2022. Review on the epidemiology of bovine rotavirus and its public health significance. Int. J. Vet. Sci. Res. 8(1), 5–10. Godden, S. 2008. Colostrum management for dairy calves. Vet. Clinics North Amer. Food Anim. Pract. 24(1), 19–39. Godden, S. 2019. Colostrum management for dairy calves. Vet. Clinics. North. Amer. Food. Anim. Pract. 35(3), 535. Golaviya, A., Mathakiya, R., Jakhesara, S. and Koringa, P. 2024. Determining genetic diversity of prevalent G and P genotype of bovine rotavirus A from neonatal calves of Gujarat, India. J. Vet. Sci. 25(4), 55; doi:10.1016/j.jvs.2024.04.055 Hasan, M.A., Kabir, M.H., Miyaoka, Y., Yamaguchi, M. and Takehara, K. 2022. G and P genotype profiles of rotavirus a field strains circulating in a vaccinated bovine farm as parameters for assessing biosecurity level. J. Vet. Med. Sci. 84(7), 929–937; doi:10.1016/j.jvms.2022 Hoshino, Y. and Kapikian, A.Z. 2000. Rotavirus serotypes: classification and importance in epidemiology, immunity, and vaccine development. J. Health. Popul. Nutr. 59, 514–516; doi:10.1016/j.jhpn.2004.04.014 Izzo, M.M., Kirkland, P.D., Mohler, V.L., Perkins, N.R., Gunn, A.A. and House, J.K. 2011. Prevalence of major enteric pathogens in Australian dairy calves with diarrhea. Austral. Vet. J. 89(5), 167–173. Jampanil, N., Kumthip, K., Longum, T., Xie, Z., Yodmeeklin, A., Sirilert, S., Ukarapol, N., Sansaard, N., Promping, C., Okitsu, S., Kobayashi, T., Ushijima, H., Maneekarn, N. and Khamrin, P. 2025. Rotavirus genotype dynamics and the emergence of G3P 8 in Thailand following nationwide vaccine implementation. Int. J. Mol. Sci. 26(18), 9249. Kaplon, J., Fremy, C., Bernard, S., Rehby, L., Aho, S., Pothier, P. and Ambert-Balay, K. 2013. Impact of rotavirus vaccine on rotavirus genotypes and caliciviruses circulating in French cattle. Vaccine 31(20), 2433–2440; doi:10.1016/j.vaccine.2013.09.007 Kim, S.H., Miguel, M., Naing, Y.P., Cho, Y.I. and Lee, S.S. 2024. Maternal transmission of rotavirus to calves and comparison of colostrum and fecal microbiota in Holstein and Hanwoo Cattle. Vet. Sci. 11(12), 606. Li, K., Lin, X.D., Huang, K.Y., Zhang, B., Shi, M., Guo, W.P., Wang, M.R., Wang, W., Xing, J.G., Li, M.H., Hong, W.S., Holmes, E.C. and Zhang, Y.Z. 2016. Identification of novel and diverse rotaviruses in rodents and insectivores, and evidence of cross-species transmission into humans. Virology 494, 168–177; doi:10.1016/j.virology.2016.01.015 Lopez, A.J. and Heinrichs, A.J. 2022. Invited review: the importance of colostrum in the newborn dairy calf. J. Dairy Sci. 105(4), 2733–2749. Lopez, S., Arias, C.F., Bell, J.R., Strauss, J.H. and Espejo, R.T. 1985. Primary structure of the cleavage site associated with trypsin enhancement of rotavirus SA11 infectivity. Virology 144(1), A11–A19; doi:10.1016/j.virology.1985.11.011 López, S., Sánchez-Tacuba, L., Moreno, J. and Arias, C.F. 2016. Rotavirus strategies against the innate antiviral system. Annu. Rev. Virol. 3, 591–609. Louge Uriarte, E.L., Badaracco, A., Spetter, M.J., Miño, S., Armendano, J.I., Zeller, M., Heylen, E., Späth, E., Leunda, M.R., Moreira, A.R., Matthijnssens, J., Parreño, V. and Odeón, A.C. 2023. Molecular epidemiology of rotavirus A in calves: evolutionary analysis of a bovine G8P 11 strain and spatio-temporal dynamics of G6 lineages in the Americas. Viruses 15(10), 2115. Luchs, A., Cilli, A., Morillo, S.G., Carmona, R.D.C.C. and Timenetsky, M.D.C.S.T. 2012. Rare G3P 3 rotavirus strain detected in Brazil: possible human–canine interspecies transmission. J. Clin. Virol. 54(1), 89–92. Martella, V., Bányai, K., Matthijnssens, J., Buonavoglia, C. and Ciarlet, M. 2010. Zoonotic aspects of rotaviruses. Vet. Microbiol. 140(3–4), 246–255. ONICL , 2023. Situation de l’agriculture marocaine N°15 (2019–2020). Morocco: Office National Interprofessionnel des Céréales et des Légumineuses (ONICL). Available via https://www.onicl.org.ma/portail/wp-content/uploads/2023/02/rapport-situation-agriculture-marocaine-n%C2%B015-2019-2020.pdf ONSSA, 2025. Liste positive des médicaments vétérinaires (version 06 mai 2025). Rabat, Morocco: Office National de Sécurité Sanitaire des Produits Alimentaires (ONSSA). Available via (https://www.onssa.gov.ma/wp-content/uploads/2025/05/LISTE-POSITIVE-DES-MEDICAMENTS-VETERINAIRES-06-MAI-2025.pdf) Papp, H., László, B., Jakab, F., Ganesh, B., De Grazia, S., Matthijnssens, J., Ciarlet, M., Martella, V. and Bányai, K. 2013. Review of group A rotavirus strains reported in swine and cattle. Vet. Microbiol. 165(3–4), 190–199. Pinheiro, F.A., Decaris, N., Parreño, V., Brandão, P.E., Ayres, H. and Gomes, V. 2022. Efficacy of prepartum vaccination against neonatal calf diarrhea in Nelore dams as a prevention measure. BMC. Vet. Res. 18, 323. Radostits, O.M., Gay, C.C., Blood, D.C. and Hinchcliff, K.W. 2001. Veterinary Medicine, A Textbook of the Diseases of Cattle, Sheep, Pigs, Goats and Horses,. Radostits, O.M., Gay, C.C., Hinchcliff, K.W. and Constable, P.D. 2007. Veterinary Medicine: a textbook of the diseases of cattle, horses, sheep, pigs, and goats, Reynolds, D.J., Morgan, J.H., Chanter, N., Jones, P.W., Bridger, J.C., Debney, T.G. and Bunch, K.J. 1986. Microbiology of calf diarrhea in southern Britain. Vet. Rec. 119(2), 34–39. Rocha, T.G., Silva, F.D.F., Gregori, F., Alfieri, A.A., Buzinaro, M.D.G. and Fagliari, J.J. 2017. Longitudinal study of bovine rotavirus group A in newborn calves from vaccinated and unvaccinated dairy herds. Trop. Anim. Health. Prod. 49, 783–790. Royaume du Maroc. 1977. Dahir portant loi n° 1-75-292 du 5 Chaoual 1397 (19 septembre 1977) édictant des mesures propres à garantir les animaux domestiques contre les maladies contagieuses. Bull. Officiel n 3388, 1079. Sadiq, A. and Khan, J. 2023. Rotavirus in developing countries: molecular diversity, epidemiological insights, and strategies for effective vaccination. Front. Microbiol. 14, 1297269. Sadiq, A., Bostan, N., Jadoon Khan. and Aziz, A. 2022. Effect of rotavirus genetic diversity on vaccine impact. Rev. Med. Virol. 32(1), 2259; e2259 Saif, L.J. and Fernandez, F.M. 1996. Group A rotavirus veterinary vaccines. J. Infect. Dis. 174(Supplement 1), S98–S106. Schwers, A., Mahin, L., Pastoret, P., Dewulf, M., Mazouz, A., Maenhoudt, M. and Michaux, C. 1984. Prevalence of antibody to rotavirus in Moroccan cattle. Comparative. Immunol. Microbiol. Infect. Dis. 7(1), 69–73. Šenica, P., Žele Vengušt, D., Vengušt, G. and Kuhar, U. 2024. Genomic revelations: investigating rotavirus A presence in wild ruminants and its zoonotic potential. Front. Vet. Sci. 11, 1429654. Shin, J., Park, G.N., Choe, S., Cha, R.M., Kim, K.S., An, B.H., Kim, S.Y., Moon, S.H., Hyun, B.H. and An, D.J. 2023. Phylogenetic analysis of G and P genotypes of bovine group A rotavirus strains isolated from diarrheic Vietnam cows in 2017 and 2018. Animals 13(14), 2314. Snodgrass, D.R., Terzolo, H.R., Sherwood, D., Campbell, I., Menzies, J.D. and Synge, B.A. 1986. Etiology of diarrhea in young calves. Vet. Rec. 119(2), 31–34. Steele, A.D., Geyer, A. and Gerdes, G.H. 2004. Rotavirus infections//Coetzer JAW, Tustin RC, editors. Infectious diseases of livestock, Cape Town, South Africa: Oxford University Press. Uddin Ahmed, N., Khair, A., Hassan, J., Khan, M.A.H.N.A., Rahman, A.K.M.A., Hoque, W., Rahman, M., Kobayashi, N., Ward, M.P. and Alam, M.M. 2022. Risk factors for bovine rotavirus infection and genotyping of bovine rotavirus in diarrheic calves in Bangladesh. PLos One 17(2), e0264577. World Organization for Animal Health. 2023. The Terrestrial Animal Health Code. Chapter 1.3: Diseases, infections, and infestations listed by the WOAH. Paris, France: WOAH. Zhao, Q., Li, P., Wang, B., Li, B., Gao, M., Ren, G., Rile, G., Rila, S., Ma, K. and Bao, F. 2025. Bovine Ultra-Long CDR H3 Specific for Bovine Rotavirus Displays Potent Virus Neutralization and Therapeutic Effects in Infected Calves. Biomolecules 15(5), 15; doi:10.1016/j.biomol.2020 Zouagui, Z., Elbay, S., Alali, S., and Lbacha, H.A. 2017. ‘Diarrhées néonatales chez le veau au Maroc: prévalence des causes infectieuses majeures (Escherichia coli F5, Coronavirus, Rotavirus et Cryptosporidium parvum). | ||
| How to Cite this Article |
| Pubmed Style El-ouadi H, Rhazzar Z, Choukri I, Touil N, Fihri OF. Bovine rotavirus: A narrative review of rotavirus a biology, vaccination strategies, and regional insights from Morocco. Open Vet. J.. 2026; 16(3): 1458-1467. doi:10.5455/OVJ.2026.v16.i3.5 Web Style El-ouadi H, Rhazzar Z, Choukri I, Touil N, Fihri OF. Bovine rotavirus: A narrative review of rotavirus a biology, vaccination strategies, and regional insights from Morocco. https://www.openveterinaryjournal.com/?mno=299353 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.5 AMA (American Medical Association) Style El-ouadi H, Rhazzar Z, Choukri I, Touil N, Fihri OF. Bovine rotavirus: A narrative review of rotavirus a biology, vaccination strategies, and regional insights from Morocco. Open Vet. J.. 2026; 16(3): 1458-1467. doi:10.5455/OVJ.2026.v16.i3.5 Vancouver/ICMJE Style El-ouadi H, Rhazzar Z, Choukri I, Touil N, Fihri OF. Bovine rotavirus: A narrative review of rotavirus a biology, vaccination strategies, and regional insights from Morocco. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1458-1467. doi:10.5455/OVJ.2026.v16.i3.5 Harvard Style El-ouadi, H., Rhazzar, . Z., Choukri, . I., Touil, . N. & Fihri, . O. F. (2026) Bovine rotavirus: A narrative review of rotavirus a biology, vaccination strategies, and regional insights from Morocco. Open Vet. J., 16 (3), 1458-1467. doi:10.5455/OVJ.2026.v16.i3.5 Turabian Style El-ouadi, Hanan, Zineb Rhazzar, Imane Choukri, Nadia Touil, and Ouafaa Fassi Fihri. 2026. Bovine rotavirus: A narrative review of rotavirus a biology, vaccination strategies, and regional insights from Morocco. Open Veterinary Journal, 16 (3), 1458-1467. doi:10.5455/OVJ.2026.v16.i3.5 Chicago Style El-ouadi, Hanan, Zineb Rhazzar, Imane Choukri, Nadia Touil, and Ouafaa Fassi Fihri. "Bovine rotavirus: A narrative review of rotavirus a biology, vaccination strategies, and regional insights from Morocco." Open Veterinary Journal 16 (2026), 1458-1467. doi:10.5455/OVJ.2026.v16.i3.5 MLA (The Modern Language Association) Style El-ouadi, Hanan, Zineb Rhazzar, Imane Choukri, Nadia Touil, and Ouafaa Fassi Fihri. "Bovine rotavirus: A narrative review of rotavirus a biology, vaccination strategies, and regional insights from Morocco." Open Veterinary Journal 16.3 (2026), 1458-1467. Print. doi:10.5455/OVJ.2026.v16.i3.5 APA (American Psychological Association) Style El-ouadi, H., Rhazzar, . Z., Choukri, . I., Touil, . N. & Fihri, . O. F. (2026) Bovine rotavirus: A narrative review of rotavirus a biology, vaccination strategies, and regional insights from Morocco. Open Veterinary Journal, 16 (3), 1458-1467. doi:10.5455/OVJ.2026.v16.i3.5 |