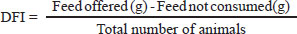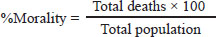| Research Article | ||
Open Vet. J.. 2026; 16(3): 1913-1924 Open Veterinary Journal, (2026), Vol. 16(3): 1913-1924 Research Article A Bacillus-based synbiotic blend improves broiler productivity and metabolic resilience under suboptimal water quality and heat stressLorena Vivas1, Marlon Brazón1, Neyo Pérez1, Moraiza Casado1, Anthony Mendoza1, David Coll2, Hennet Farias3, Danilo Zavala3 and Alberto Quintero4*1Animal Nutrition and Feeding Laboratory, Centre for Agricultural Biotechnology, Venezuelan Institute for Scientific Research, Altos de Pipe, Bolivarian Republic of Venezuela 2Computational Chemistry Laboratory, Chemistry Centre, Venezuelan Institute for Scientific Research, Altos de Pipe, Bolivarian Republic of Venezuela 3Liofilizados de Venezuela, Maracaibo, Bolivarian Republic of Venezuela 4Animal Genetics and Reproduction Laboratory, Centre for Agricultural Biotechnology, Venezuelan Institute for Scientific Research, Altos de Pipe, Bolivarian Republic of Venezuela *Corresponding Author: Alberto Quintero. Animal Genetics and Reproduction Laboratory, Centre for Agricultural Biotechnology, Venezuelan Institute for Scientific Research, Bolivarian Republic of Venezuela.Email: albertojosequinteroaraque [at] gmail.com Submitted: 18/12/2025 Revised: 11/02/2026 Accepted: 20/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
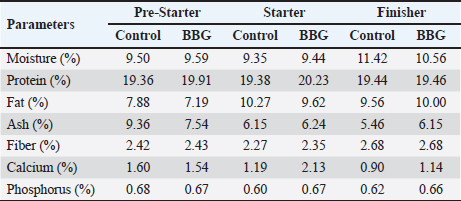
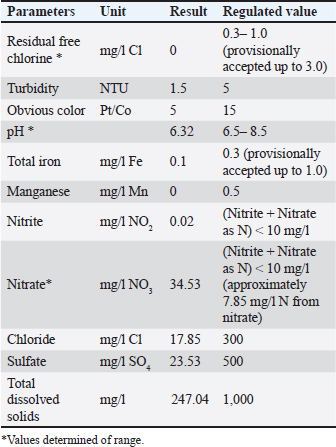
ABSTRACTBackground: Antibiotic growth promoters (AGPs) are being phased out in poultry production, leading to the search for functional non-nutritive additives. Synbiotic blends, such as the BBG complex, can enhance gut health and resilience against environmental stressors, particularly suboptimal water quality and heat stress. Aim: This study aimed to evaluate the effect of a synbiotic blend (BBG) comprising Bacillus spp., β-glucans, and β-mannans as a non-nutritive immunostimulant on the productive performance and physiological parameters of broiler chickens. Methods: A total of 264 one-day-old Ross broiler chickens were distributed into two treatments (n=132 each) with three replicates of 44 birds each, covering three phases (pre-starter, starter, and finisher). The control group received a commercial diet, whereas the BBG group received a dietary inclusion (5%) of a conglomerate comprising Bacillus subtilis (10%–14%), Bacillus toyonensis (10%–14%), Bacillus licheniformis (13%–17%), Heyndrickxia coagulans (10%–14%), Shouchella clausii (13%–17%), β-glucans (1%–5%), β-mannans (1%–5%). Data were analyzed using one-way analysis of variance. Physicochemical analyses were performed on the feed and water, while production parameters (i.e., initial and final body weight (BW), daily feed intake, BW gain, feed conversion ratio, and mortality) were evaluated. Hematological and biochemical analyses were performed weekly. Results: Physicochemical analysis confirmed that the drinking water deviated from regulatory standards, exhibiting a lack of residual chlorine, slightly acidic pH, and elevated nitrate levels. BBG administration enhanced overall productive performance, yielding higher final BW and weight gain, alongside a noteworthy reduction in mortality during the pre-starter phase. Hematological analysis showed marked leukocytosis and elevated liver enzymes (alanine aminotransferase, aspartate aminotransferase, and bilirubin) in both treatments, confirming active environmental stress. Although BBG treatment did not completely normalize these stress indicators, it improved the birds´ resilience and enhanced feed efficiency during the initial stages. Conclusion: The BBG synbiotic blend is a viable alternative to conventional AGPs, improving productivity and early viability under challenging field conditions characterized by high temperatures and suboptimal water quality. Keywords: Bacillus, Broiler chickens, Metabolic resilience, Non-nutritive additive, Productivity. IntroductionThe ban on Antibiotic growth promoters (AGPs) in several countries has driven the search for alternatives to enhance health, productivity, and intestinal integrity in broiler chickens (Udeh et al., 2020; Sanmiguel et al., 2024; Polidoro et al., 2025). Furthermore, the poultry industry faces the challenge of developing alternative feeding systems that exclude coccidiostats. The use of probiotic strains as feed additives has gained prominence among these alternatives (Fabia et al., 2021; Kalia et al., 2022; Nusairat et al., 2024; Yu et al., 2025). As poultry production capacity increases, the need for feed additives that protect bird health, improve performance, and limit foodborne pathogens is growing (El Jeni et al., 2021). Using probiotics, specifically lactic acid-producing bacteria, in poultry diets contributes to intestinal microbiota integrity and stability, thereby hampering pathogen proliferation (Mariño and Roa, 2021). Accordingly, probiotics, prebiotics, and synbiotics are used to maintain microbial balance (Szakacs et al., 2015; Markowiak and Śliżewska, 2018; Polidoro et al., 2025). These additives, often termed “natural” growth promoters (Caly et al., 2015; Sethiya, 2016), have beneficial impacts on intestinal health through the use of probiotics, prebiotics, phytobiotics, organic acids, and enzymes (Parisien et al., 2007; Roberts et al., 2015; Mehdi et al., 2018; Wickramasuriya et al., 2022; Kalia et al., 2022). Prebiotics are foods that stimulate the microbiota in the gastrointestinal tract (Muthukumaran et al., 2018). β–glucans, glucose polymers found in the cell walls of yeasts, fungi, and cereals (Muthukumaran et al., 2018), have been shown to modulate the immune response and gut health. Various biological activities have been reported for β–glucans, including antiimmunomodulatory effects. A study in piglets challenged with lipopolysaccharides found that purified β–glucans improved growth performance by enhancing intestinal barrier function and microbiota (Luo et al., 2019). Indeed, the modulation of the gut microbiota by β–glucans is a crucial factor in health regulation (Muthukumaran et al., 2018). The polymers act as a probiotic source that reduces cholesterol and triglycerides, maintains blood glucose homeostasis, and provides other health benefits (Wang et al., 2016; Muthukumaran et al., 2018; Benitez et al., 2020). In the poultry context, yeast β–glucan has been shown to be effective in mitigating the negative effects of Fusarium mycotoxins on broiler chicken performance, intestinal integrity, and liver function (Marquis et al., 2025). Mannan oligosaccharides (MOS) are prebiotics derived from the yeast cell wall of Saccharomyces cerevisiae that can be used as an alternative to AGPs (Sanmiguel et al., 2024; Polidoro et al., 2025). Additionally, β-mannanase is used in low-nutrient broiler feeds to promote better energy utilization (Ferreira et al., 2016; Sanmiguel et al., 2024). Incorporating β-mannanase can improve nutrient absorption and utilization in soybean-based diets (Yu et al., 2025). This enzyme also plays a crucial role in activating defense mechanisms and enhancing digestive enzyme activity (Ferreira et al., 2016; Benitez et al., 2020; Nusairat et al., 2024; Sanmiguel et al., 2024). Gut health and performance can be significantly modulated by the inclusion of prebiotics in the diet. According to a recent meta-analysis, MOS, which are key components of the yeast cell wall, are a viable alternative to AGPs for improving growth performance in broiler chickens (Polidoro et al., 2025). This application is not limited to broiler chickens, as the use of MOS as a substitute for antibiotics to improve production in laying hens has also been studied (Sanmiguel et al., 2024). In a broader sense, β–glucans and mannans are recognized as beneficial prebiotic additives in animals of zootechnical interest (Benitez et al., 2020). In this context, supplementation with probiotics and other biologically active components, such as postbiotics or eubiotics (immunomodulators), has emerged as a promising strategy in poultry production. Building on this, the present research evaluated the effect of a cluster of BBG as an immunostimulant on the productive performance and physiology of broiler chickens. Materials and MethodsLocation and environmental conditionsThis study was conducted in Maracay, Aragua state, Venezuela. The location had an average temperature of 30.3°C and a mean relative humidity of 57.5%. Animal and experimental design of the animalsA total of 264 one-day-old Ross broiler chickens were used. The birds were distributed into 2 treatments, each consisting of 132 birds divided into 3 replicates of 44 broilers. The study was conducted across 3 stages: pre-starter (1–10 days), starter (11–22 days), and finisher (23–42 days). Non-nutritive symbiotic additive and bacterial strainsThe experimental treatments were as follows: 1) control, receiving a commercial basal diet; and 2) BBG, receiving the commercial diet with the inclusion of 5% of a bacterial consortium consisting of B. subtilis (10%–14%), B. toyonensis (10%–14%), B. licheniformis (13%–17%), Heyndrickxia coagulans (10%–14%), S. clausii (13%–17%), β-glucans (1%–5%), and β-mannans (1%–5%). These bacterial strains were isolated experimentally from virgin soils in the Perijá region, Zulia State, Venezuela, by pre-enriching culture media with 0.5% peptone. They were then transferred to the laboratory of the Faculty of Veterinary Sciences at the University of Zulia in 2011 for molecular identification using conventional polymerase chain reaction. These strains are the property of the LIVENCA laboratory and were donated for research. Variables evaluatedThe following variables were calculated: initial body weight (IBW), final body weight (FBW), daily feed intake (DFI), body weight gain (BWG), feed conversion ratio (FCR), mortality, complete blood count, and blood chemistry. Physicochemical analysis of diet and waterDiets and raw materials were bromatologically analyzed following the Official Methods of Analysis of Association of Analytical Chemists (AOAC, 2016) (Table 1). The water quality was determined following the guidelines established by the International Organization for Standardization (ISO) 5667-3:2019 (Table 2). To characterize the environmental stress mentioned in the study, we monitored the physical and chemical parameters of the drinking water. Several indicators, including pH levels, residual chlorine, and nitrate concentrations, were found to be outside the recommended range for poultry production, establishing a challenging environmental baseline to evaluate the BBG symbiotic blend’s resilience-improving properties. Table 1. Proximate analysis of the experimental diets.
Table 2. Physical and chemical parameters of the drinking water samples.
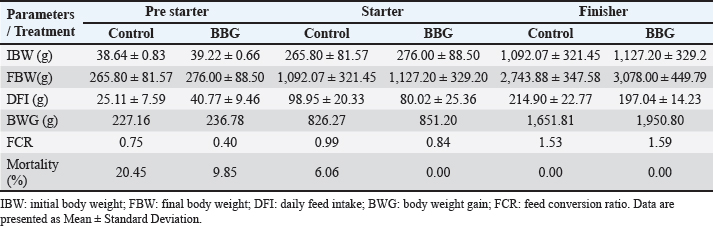
Monitoring and samplingBody weight (BW) and feed intake were measured using a CGoldenwal scale (model HZ50002B). For each treatment, 15 chickens were randomly selected and individually identified for longitudinal weighing on days 1, 4, 7, 10, 14, 21, 22, 28, 35, and 42. Body weight was recorded on days 4 and 10 to comply with the Ross management manual guidelines (Aviagen, 2021) and to monitor physiological status during weekly blood sampling. Table 3 shows the productive parameters calculated using only the weights recorded at the beginning and end of each dietary phase (days 1, 10, 22, and 42). Feed intake was measured daily. Table 3. Productive performance of chicken broilers.
For the blood profile, cardiac puncture was used for sampling. On day 1, a representative sample was taken from chickens before diet administration as a baseline value. From day 7 onwards, weekly monitoring was performed according to the quality requirements of clinical laboratories (ISO 1518). A longitudinal sampling approach was implemented for hematological and blood biochemical profiles. Because of the intensive nature of the monitoring and the pilot character of these specific physiological evaluations, samples were collected from one representative bird per treatment at each time point. Consequently, these physiological parameters are presented as a descriptive analysis aimed at identifying biological trends and temporal patterns in response to environmental stress and symbiotic intervention, rather than for comparative inferential statistics. Formulas for the production variables
DFI: daily feed intake BWG=FBW (g)-IBW(g) BWG: Body weight gain; IBW: Initial body weight; FBW: Final body weight.
FCR: Feed conversion ratio
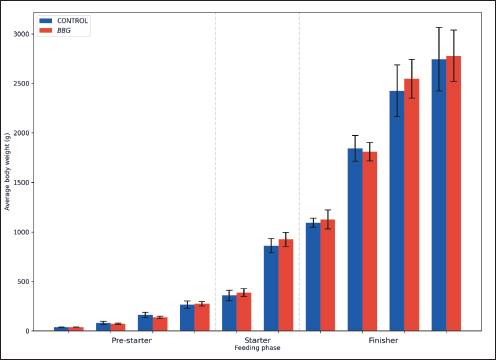
Statistical analysisThe statistical analysis was differentiated according to the nature of the variables. For productive parameters (Table 3), data were analyzed using a one-way analysis of variance using Statistical Product and Service Solutions (SPSS) via IBM SPSS (2021). BW evolution was visualized through Figure 1.
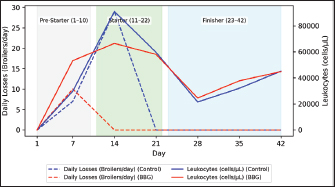
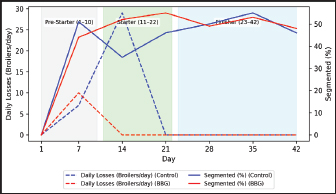
Fig. 1. Body weight (BW) performance. The model Yi=μ + Ti + εi was used, where μ=overall average, Ti=treatment effect (i=Control, BBG); and εi=experimental error. Mortality data, being binomial, were analyzed using a chi-square (χ2). All differences were considered statistically significant at p < 0.05. For biochemical data (Table 4), a descriptive longitudinal analysis was performed to visualize hematological fluctuation over time through graphical representations (Figs. 2–6).
Fig. 2. Daily mortality and total leukocyte count evolution in broilers.
Fig. 3. Daily mortality and variations in the segmented cell percentage in broilers.
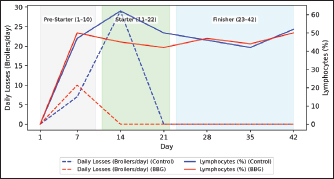
Fig. 4. Temporal dynamics of daily mortality and lymphocyte percentage.
Fig. 5. Temporal relationship between daily mortality and eosinophil count in broiler chickens.
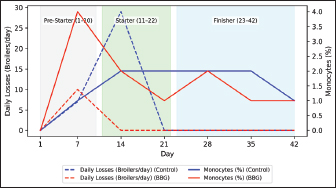
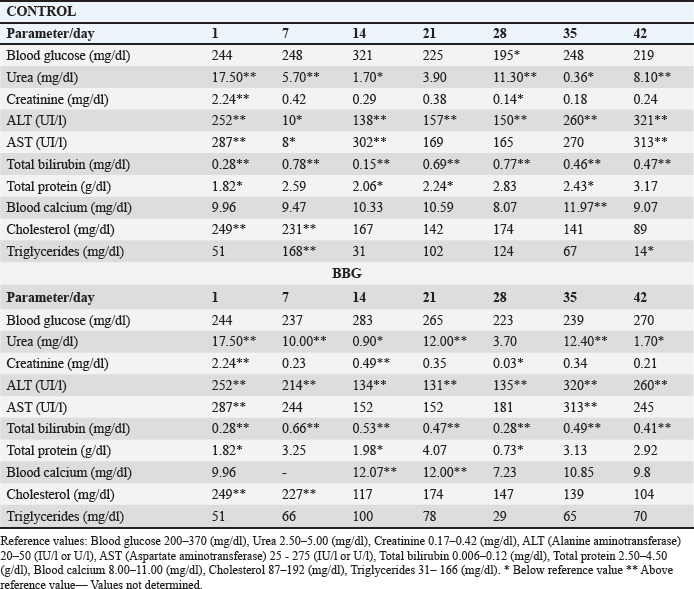
Fig. 6. Variation in the monocyte percentage and daily mortality in broilers. Table 4. Serum biochemistry parameters of broiler chickens throughout the experimental period
Ethical approvalInstitutional and national guidelines for the care and use of laboratory animals were followed in compliance with ethical standards, following the guidelines of the Bioethics Committee for Animal Research of the Venezuelan Institute for Scientific Research (COBIANIM 2025; May 13, 2025). ResultsAnalysis of diet and water qualityThe bromatological analysis of the diets developed for the three growth phases (pre-starter, starter, and finisher) (Table 1) confirmed that all nutritional levels were comparable between treatments. This consistency ensured that the diets were isoproteic and isoenergetic, meeting the specific nutritional requirements for each developmental stage. Table 2 shows the physical and chemical parameters of the water supplied to the broilers. Noncompliance with regulatory standards was identified for free residual chlorine, pH, and nitrate. Specifically, the absence of residual chlorine (0 mg/l) and the high nitrate concentration (34.53 mg/l NO3) indicate that the drinking water deviates considerably from the optimal values for poultry farming. Productive performanceTable 3 and Figure 1 summarize the production parameters evaluated throughout the three growth phases (Pre-starter, Starter, and Finisher) for both treatments (Control and BBG). In the pre-starter phase, the IBW was 38.64 ± 0.83 g for the Control and 39.22 ± 0.66 g for the BBG treatment, indicating no differences in the initial weight between the treatments. At the end of this phase, the FBW of the BBG treatment was higher (276.00 ± 88.50 g) compared to the Control (265.80 ± 81.57 g). The DFI was 25.11 ± 7.59 g for the Control and 40.77 ± 9.46 g for the BBG, while the BWG was 227.16 g and 236.78 g. The FCR was 0.75 and 0.40 for the control and BBG treatments, respectively. Mortality was notably higher in the control treatment (20.45%) than in the BBG treatment (9.85%), suggesting an increase in field challenge that affected both treatments, although the control showed a disproportionately greater impact. No significant differences were observed between treatments during this phase (p > 0.05). During the starter phase, the IBW was 265.80 ± 81.57 g for the Control and 276.00 ± 88.50 g for BBG, maintaining the trend of a higher FBW in the BBG treatment (1,127.20 ± 329.2 g) compared to the Control (1,092.07 ± 321.45 g). The DFI was 98.95 ± 20.33 g for the Control and 80.02 ± 25.36 g for the BBG, while the BWG was 826.27 g and 851.20 g. The FCR was 0.99 for the control and 0.84 for the BBG treatment, indicating better feed efficiency in the latter. Mortality in this phase was absent in the BBG treatment (0.00%) compared with 6.06% in the control group. Statistical analysis indicated no significant differences during this phase (p >0.05). In the finisher phase, the IBW was 1,092.07 ± 321.45 g for the Control and 1,127.20 ± 329.2 g for the BBG. At the end of the study (day 42), the FBW of the BBG treatment reached 3,078.00 ± 449.79 g, surpassing that of the control treatment, which recorded 2,743.88 ± 347.58 g. The DFI in this phase was 214.90 ± 22.77 g for the Control and 197.04 ± 14.23 g for the BBG, resulting in a BWG of 1,651.81 g and 1,950.80 g, respectively. The FCR was 1.53 for the control group and 1.59 for the BBG group. No mortality was observed in either treatment group during this phase. Statistical analysis showed no significant differences between treatments (p > 0.05). Overall, the treatment with BBG inclusion consistently showed higher FBW and BWG, as well as improved FCR during the pre-start (0.40 vs. 0.75) and starter phases (0.84 vs. 0.99), although the latter was slightly higher in the finisher phase (1.59 vs. 1.53). Notably, mortality was significantly lower in the early phase of BBG treatment, suggesting a protective effect against initial environmental challenges. Hematological profileIn the hematological analysis, the blood count results for broilers in both control and BBG treatments showed variations compared with the established reference values. Reference values: Leukocytes: 4,000–40,000 (cells/µl or mm³), Segmented cells: 20–50 (%), Lymphocytes: 45–75 (%), Eosinophils: 0–2 (%), Monocytes: 2–10 (%). In the Control treatment, leukocytes consistently exceeded the reference range at most sampling points, with peak values of 90,871 cells/μl (day 14), 59,583 cells/μl (day 21), and 45,117 cells/μl (day 42). This indicates a marked leukocytosis, suggesting chronic inflammation or an infectious process (Fig. 2). Segmented cells (heterophils) were above the reference range on days 7 (51%) and 35 (55%) (Fig. 3). Lymphocytes remained within the reference range, except on day 35 (42%) (Fig. 4). Eosinophils remained within the range throughout the study (Fig. 5), whereas monocytes were below the reference range on days 7 (1%) and 42 (1%) (Fig. 6). In the BBG treatment, leukocytes exceeded the reference range at all sampling points, with values ranging from 24,500 cells/μl (day 28) to 66,369 cells/μl (day 14) (Fig. 2). Segmented cells remained within the range, except on days 14 (52%), 21 (55%), and 35 (53%), when they were slightly above the upper limit (Fig. 3). Lymphocytes were within the reference range at all sampling points (Fig. 4), indicating a more stable immune response compared with the control. Eosinophils were within the range throughout the study (Fig. 5), whereas monocytes were below the reference range on days 21, 35, and 42 (1%) (Fig. 6). Both treatments showed markedly elevated leukocyte levels relative to the reference values throughout the study. Although segmented cell and lymphocyte trends varied, they remained mostly within or near the established ranges for both treatments. Blood biochemistryThe results of blood biochemistry analyses for broiler chickens in the control and BBG treatments are detailed in Table 4. Various alterations were observed compared with the established reference values. In the control group, blood glucose levels remained mostly within the reference range, except on day 28 (195 mg/dl). The urea showed considerable fluctuation, exceeding the reference value on days 1, 7, 28, and 42. Creatinine was above the range on day 1 (2.24 mg/dl) and below the range on day 28 (0.14 mg/dl). The liver enzymes alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were markedly elevated. ALT was above the range at almost all sampling points, peaking at 321 IU/l (day 42). AST remained above the range on days 1, 14, and 42. The total bilirubin level remained consistently above the reference value throughout the study. Total protein level was below the range on days 1, 14, 21, and 35. Blood calcium levels remained within the range throughout the study, with a single peak of 11.97 mg/dl on day 35. Cholesterol and Triglycerides (TGs) showed transient elevation during the early stages (days 1 and 7), but subsequently stabilized within or below the range. Notably, TGs in the BBG treatment remained within the reference ranges at all sampling points. Blood glucose remained within the reference range at all sampling points in the BBG treatment, indicating superior glycemic stability compared to the Control. Urea showed variations, exceeding the range on days 1, 7, 21, and 35 (12.40 mg/dl), while falling below the range on days 14 and 42. Creatinine was above the range on days 1 (2.24 mg/dl) and 14 (0.49 mg/dl). ALT and AST enzyme levels were elevated across the board. ALT was above the range of 131 IU/l (day 21) to 320 IU/l (day 35) at all sampling points. AST levels remained above the range on days 1 (287 IU/l) and 35 (313 IU/l), while remaining within the range otherwise. The total bilirubin level was above the reference value at all sampling points. The total protein level was below the range on days 1 (1.82 g/dl), 14 (1.98 g/dl), and 28 (0.73 g/dl). Blood calcium levels were above the range on days 14 (12.07 mg/dl) and 21 (12.00 mg/dl) but dropped on day 28. Cholesterol was above the range on days 1 and 7 before stabilizing within the reference limits. Notably, triglycerides remained within the reference range at all sampling points, in contrast to the fluctuations observed in the control group. Overall, both treatments resulted in several biochemical alterations, such as constant elevation of total bilirubin, ALT, and AST enzymes. Urea and creatinine levels fluctuated in both treatments; however, the BBG treatment maintained blood glucose and triglycerides within the reference ranges more consistently than the control treatment. DiscussionNutritional formulation and compositionThe diets were developed to maintain isoproteic and isoenergetic profiles across treatments, aiming to isolate the biological effect of the BBG intervention. The nutritional parameters remained constant and aligned with the specific requirements for broiler chickens throughout various growth stages (Table 1). This nutritional standardization is consistent with reports by Mariño and Roa (2021) and Fabia et al. (2021), who emphasize that stability in energy and protein density is fundamental for validating the efficacy of eubiotic and probiotic additives (Bacillus). Furthermore, the uniformity in nutritional levels between treatments ensures that any observed productive or physiological response is a direct consequence of supplementation rather than variations in basal nutrient intake, adhering to the experimental rigor suggested by Slozhenkina et al. (2022). The impact of water qualityThe physicochemical parameters of the water (Table 2) revealed the absence of residual free chlorine. Since chlorinated drinking water has regulatory effects on the intestinal microbiota (Meng et al., 2023), its absence suggests lower oxidative stress on the bacterial strains within the BBG synbiotic additive, although it increases the risk of waterborne pathogens. A slightly acidic pH was also observed; pH is critical for maintaining intestinal homeostasis, as fluctuations can disrupt nutrient absorption and enzymatic activity (Mohamed et al., 2025). Furthermore, the nitrate level of 34.53 mg/l NO3 exceeded the recommended safety threshold, representing a potential risk factor for metabolic health and oxygen transport. Productivity and feed efficiencyThe improvement in BW and weight gain observed in the BBG treatment is a common finding in the literature investigating synbiotic additives in poultry. For example, B. licheniformis administered in drinking water was found to significantly increase BW in broilers (Liu et al., 2012). Similarly, a local probiotic was reported to have a positive effect on live weight and productivity in broilers, highlighting a 121% increase in gains (Nasimov et al., 2024). Improvements in weight gain and feed conversion have also been documented in broiler chickens that received consortia of microorganisms such as Bacillus clausii, S. cerevisiae, and Lactococcus lactis (Gutiérrez et al., 2015). These findings are particularly relevant under challenging conditions; for instance, Hashemitabar and Hosseinian (2024) highlighted the beneficial impacts of Bacillus and Lactobacillus on growth performance under heat stress, which aligns with the environmental stressors observed in this study. Furthermore, the inclusion of the symbiotic consortium improves digestion and nutrient absorption, resulting in enhanced growth, as described by Markowiak and Śliżewska (2018). Lower DFI and higher BWG in the BBG treatment are relevant from an economic perspective, as they indicate more efficient feed use. This result is consistent with studies demonstrating the ability of synbiotics to optimize intestinal health and mitigate the negative effects of poor water quality and high temperatures (Mariño and Roa, 2021; Slozhenkina et al., 2022; Hashemitabar and Hosseinian, 2024). Several studies have shown that eubiotic additives influence accumulated intake (Timmerman et al., 2006; Gutiérrez et al., 2015; Almeida Paz et al., 2019; Fabia et al., 2021; Mariño and Roa, 2021; Kalia et al., 2022; Hashemitabar and Hosseinian, 2024). However, the reported values were higher than those obtained in this study, suggesting that the BBG intervention may promote higher feed efficiency even under suboptimal water and thermal conditions. Reduction of mortality and early survivalA notable finding was the significant reduction in mortality during the pre-starter phase in the BBG treatment group (9.85% vs. 20.45% in the control group). High mortality in the early stages is a common problem, which is often associated with factors such as transport stress, adaptation to the new environment, and susceptibility to infections. The improvement in survival, especially in this critical phase, is a strong indicator of the protective effect of the BBG synbiotic additive, which may have contributed to the better production parameters observed in the later phases. Numerous studies have found that probiotics and synbiotics maintain a low mortality rate and good animal production (Timmerman et al., 2006; Szakacs et al., 2015; Gutiérrez et al., 2015; Udeh et al., 2020; Fabia et al., 2021). The results of a recent meta-analysis indicated that dietary MOS supplementation—a component often present in synbiotic blends—was effective in increasing broiler weight gain and reducing FCR, promoting a response comparable to that of AGP-fed broilers. Although feed intake was not influenced by dietary supplementation in that study, the inclusion of MOS within a synbiotic complex, such as BBG, represents a safe and sustainable alternative for production in the absence of AGPs, enhancing early viability even under challenging environmental conditions (Polidoro et al., 2025). Physiological parameters and immune responsesRegarding physiological parameters, marked leukocytosis was the predominant finding in both treatments. These values indicate a systemic inflammatory response, a condition that can be intensified by environmental stressors such as water quality and ambient temperature (Bohatko et al., 2023). Although synbiotics are known to modulate the immune response (Markowiak and Śliżewska, 2018), the magnitude of leukocytosis suggests that the environmental challenge exceeded the BBG intervention’s capacity to completely normalize this parameter. This aligns with findings where probiotic-based additives showed variable efficacy in restoring hematological balance under severe heat stress (Zálešáková et al., 2025). The mechanism of action of the constituents within the BBG, specifically β-glucans, involves improving intestinal barrier function and modulating the gut microbiota, which contributes to inflammation control (Luo et al., 2019). Furthermore, β-glucans have been reported to possess anti-inflammatory and immunomodulatory effects (Muthukumaran et al., 2018), as well as the ability to favorably alter the gut microbiota (Wang et al., 2016). Segmented cells (key cells in the innate immune response) showed fluctuations in both treatments, occasionally exceeding the reference range; these findings differ from those obtained in various studies (Ávilez et al., 2015; Sufiriyanto et al., 2018; Kareem et al., 2024). The environment, stress levels, genetics, and nutritional sufficiency contribute to segmented cell levels (Sufiriyanto et al., 2018). Lymphocytes, on the other hand, remained mostly within the reference ranges, coinciding with various studies (Ávilez et al., 2015; Sufiriyanto et al., 2018; Kareem et al., 2024). This could indicate that the adaptive immune response was not completely compromised despite the challenges. However, the inversion of the Segmented-to-lymphocyte ratio is a commonly used indicator of stress in poultry farming (Nwaigwe et al., 2020). Elevated segmented cells and lymphocyte maintenance confirm a physiological stress response, likely linked to the thermal and water quality challenges present in the current trial. Eosinophils and leukocytes remained within or close to the reference ranges, consistent with values reported for healthy broilers (Sufiriyanto et al., 2018; Nwaigwe et al., 2020; Adetunji et al., 2022; Kareem et al., 2024). Blood glucose levels remained mostly within the reference range in both treatments, with the BBG treatment demonstrating greater stability. These values are consistent with those reported in the literature (Hagan et al., 2022; Onunkwo et al., 2022), suggesting that the birds managed to maintain glycemic homeostasis despite the environmental stress, potentially aided by the synbiotic consortium’s metabolic support. Urea and creatinine, critical indicators of renal function, showed fluctuations in both treatments, with values often exceeding the reference ranges. These results are consistent with those of other studies (Onunkwo et al., 2022; Kareem et al., 2024). The elevation of these nitrogenous metabolites could be a physiological consequence of the high nitrate levels (34.53 mg/l) detected in the drinking water, which may have placed an additional excretory burden on the kidneys, potentially leading to subclinical renal impairment regardless of the BBG intervention. Total bilirubin was also above the reference range in both treatments, a finding that differs from the results obtained on the effect of Bacillus cereus and coumarin on biochemical parameters in broiler chickens (Duskaev et al., 2020). This discrepancy suggests that the environmental conditions in the current trial exerted a stronger influence on biliary metabolism than the probiotic interventions reported in the literature. In this study, both treatments showed consistently high ALT and AST values compared with the reference ranges. This sustained elevation indicates metabolic stress or subclinical hepatic dysfunction. However, the action of the synbiotic consortium in the BBG treatment may have contributed to better overall liver function, although it did not completely normalize these enzymes. The protective role of bacterial consortia in maintaining hepatic integrity under toxicological or environmental pressure has been emphasized as a key factor in improving productive performance (Ogbuewu et al., 2022). This is supported by the observed better productive performance, as a healthy liver is crucial for nutrient metabolism. The influence of synbiotic additives on liver function has been documented in the management of environmental challenges. Furthermore, the inclusion of β-glucan within the BBG likely supported the liver, as yeast-derived components have demonstrated a stabilizing effect on blood biochemical profiles in broilers subject to nutritional challenges (Marquis et al., 2025). Additionally, other authors highlight the influence of probiotics on liver function and their role in maintaining biochemical homeostasis (Duskaev et al., 2020; Udeh et al., 2020; Ogbuewu et al., 2022). The total protein in both treatments occasionally showed values below the reference range, especially in the early stages. These results are comparable with those of similar studies (Hagan et al., 2022; Onunkwo et al., 2022) and may reflect the rapid protein turnover required for growth under environmental stress (Bohatko et al., 2023). Blood calcium levels showed variations, with some peaks above the range, particularly in patients receiving BBG. Although several studies obtained values within the reference range (Yazhini et al., 2018; Hagan et al., 2022; Nwaigwe et al., 2020), the fluctuations observed here could be linked to the rapid skeletal development, metabolic demands, and homeostatic adjustments made by the birds to cope with suboptimal water quality. Finally, the levels of cholesterol and triglycerides, indicators of lipid metabolism, showed variations. Cholesterol was elevated in the early stages in both treatments, which could be related to rapid growth and lipid metabolism in young chickens, and later fell within the reference values (Yazhini et al., 2018; Hagan et al., 2022). This metabolic stability in the BBG treatment suggests that the synbiotic consortium helped regulate lipid absorption and utilization, as multi-strain probiotics can prevent excessive lipid accumulation and improve the serum profile in broilers (Zálešáková et al., 2025). Some studies have suggested that probiotics may influence lipid and cholesterol metabolism by modulating the gut microbiota, which may affect fat absorption and bile acid synthesis (Duskaev et al., 2020; Hashemitabar and Hosseinian, 2024). ConclusionBroilers receiving the dietary inclusion of the BBG treatment—a non-nutritive additive—demonstrated superior performance concerning final BW and weight gain. Although the DFI was higher in the pre-starter phase, it remained lower in the starter and finisher periods compared with the control. The use of this synbiotic blend improves the productive performance of broilers. Crucially, a noteworthy reduction in pre-starter mortality was observed in the BBG treatment compared to the control, highlighting its role in enhancing early viability. The concurrent presence of hematological abnormalities (leukocytosis) and biochemical markers (ALT and AST) confirms that environmental stressors, particularly suboptimal water quality and high temperatures, profoundly influenced the physiology of the birds. In this challenging context, BBG proved to be an effective strategy to mitigate the negative impacts of environmental stress on broiler chicken productivity and physiological stability. AcknowledgmentsC.A. Purolomo, C.A. Alimentación Balanceada Alibal, C.A. Liofilizados de Venezuela Grupo REEVEX, Instituto Nacional de Salud Agrícola Integral, Hidrológica del Centro C.A, Unidad de Producción Agrícola Nicolino Ierrobino. Conflict of interestThe authors declare that they have no conflicts of interest. FundingThis research was supported by the National Fund for Science, Technology and Innovation (Fonacit) (grant no. 2024PGP461). Authors’ contributionsLorena Vivas, Neyo Perez, Alberto Quintero, Moraiza Casado, and David Coll conceptualized and designed the study. Lorena Vivas, Anthony Mendoza, Danilo Zavala, and Alberto Quintero conducted the experimental studies (fieldwork and data acquisition experiments. Neyo Perez, Marlon Brazon, and Hennet Farias performed all statistical analyses. Lorena Vivas, Moraiza Casado, Anthony Mendoza, David Coll, Hennet Farias, and Alberto Quintero reviewed and edited the manuscript. All authors have read and approved the final version of the manuscript and agreed to be responsible for all aspects of the work. Data availabilityThe research data generated in this study are available from the authors upon reasonable request. ReferencesAdetunji, J.A., Egbere, J., Oshibanjo, D.O., Amaduruonye, W., Ediga. and B.A. 2022. Hematological profile of broiler chicken fed with lactic acid bacteria (LAB) and without LAB. Niger. J. Anim. Prod. 49, 765–767; doi:10.51791/njap.vi.4788 Almeida Paz, I.C.D.L., De Lima Almeida, I.C., De La Vega, L.T., Milbradt, E.L., Borges, M.R., Chaves, G.H.C., Dos Ouros, C.C., Lourenço Da Silva, M.I., Caldara, F.R. and Andreatti Filho, R.L. 2019. Productivity and well-being of broiler chickens supplemented with probiotic. J. Appl. Poult. Res. 28(3), 577–586; doi:10.3382/japr/pfz054 AOAC. 2016. Official Methods of Analysis of AOAC International, 20th Edition. Washington D.C.: AOAC Inc. Aviagen. 2021. Ross 308 Broiler: performance objectives. Huntsville, AL: Aviagen Group, 1–25. Ávilez, B.L., Rugeles, C.C., Jabib, L., Herrera. and Y.M. 2015. Hematological parameters in broilers raised on a closed production farm in the low tropics. Rev. Med. Vet. 29, 33–39; doi:10.19052/mv.3444 Benitez, H.R., Rondón, A.J. and Rodríguez, C.M. 2020. Effect of β-glucans and mannans in animals of zootechnical interest. Matanzas, Cuba: Universidad de Matanzas, pp: 1–12. Bohatko, A. 2023. Effect of probiotic biologics on morpho-biochemical parameters of broiler chicken blood. Ukr. J. Vet. Sci. 14(3), 9–24; doi: 10.31548/veterinary3.2023.09. Caly, D.L., D´Inca, R., Auclair, E. and Drider, D. 2015. Alternatives to antibiotics to prevent necrotic enteritis in broiler chickens: a microbiologist´s perspective. Front. Microbiol. 6, 1336; doi:10.3389/fmicb.2015.01336 Duskaev, G., Rakhmatullin, S. and Kvan, O. 2020. Effects of Bacillus cereus and coumarin on growth performance, blood biochemical parameters, and meat quality in broilers. Vet. World 13(11), 2484–2492. Fabia, K., Wolski, D., Kropisz, D., Radzki, RP., Bieńko, M., Szymańczyk, S., Kimicka, A. and Manastyrska, M. 2021. Effect of probiotic additives and Bacillus licheniformis inclusion in the diet on broiler growth. Med. Weter. 77(6), 304–308; doi: 10.21521/mw.6534 Ferreira, H.C., Hannas, M.I., Albino, L.F.T., Rostagno, H.S., Neme, R., Faria, B.D., Xavier, M.L. and Rennó, L.N. 2016. Effect of the addition of β-mannanase on the performance, metabolizable energy, amino acid digestibility coefficients, and immune functions of broilers fed different nutritional levels. Poult. Sci. 95, 1848–1857. Gutiérrez, L., Bedoya, O. and Arenas, J. 2015. Evaluation of production parameters in broiler chickens supplemented with probiotic microorganisms. Temas. Agrarios. 20(2), 81–85. Hagan, JK., Hagan, BA., Ofori, SA. and Etim, N.N. 2022. Haematological and serum biochemical profiles of two broiler strains fed rations with varying levels of palm kernel oil residue. Ghana J. Anim. Sci. 13(2), 30–41. Available via https://www.scirp.org/reference/referencespapers?referenceid=4110542 Hashemitabar, S.H. and Hosseinian, S.A. 2024. The comparative effects of probiotics on growth, antioxidant indices and intestinal histomorphology of broilers under heat stress condition. Sci. Rep. 14(1), 1–13; doi:10.1038/s41598-024-66301-9 IBM Corp. 2021. IBM SPSS Statistics for Windows, Version 28.0. Armonk, NY: IBM Corp. International Organization for Standardization. 2019. ISO 5667-3:2019. Water quality—Sampling—Part 3: Preservation and handling of water samples. Geneva, Switzerland: ISO. International Organization for Standardization. 2022. ISO 15189:2022. Medical laboratories—Requirements for quality and competence. Geneva, Switzerland: ISO. Jeni, R.E., Dittoe, D.K., Olson, E.G., Lourenco, J., Corcionivoschi, N., Ricke, S.C. and Callaway, T.R. 2021. Probiotics and potential applications for alternative poultry production systems. Poult. Sci. 103, 101156; doi:10.1016/j.psj.2021.101156 Kalia, V.C., Shim, W.Y., Patel, S.K.S., Gong, C. and Lee, J.K. 2022. Recent developments in antimicrobial growth promoters in chicken health: opportunities and challenges. Sci. Total Environ. 834, 155300; doi:10.1016/j.scitotenv.2022.155300 Kareem, D.U., Amos, A.T., Idowu, O.P.A., Bonagurio, L.P. and Idowu, O.M.O. 2024. Blood profile as a health indicator in broiler chickens fed diets of different particle sizes supplemented with multienzyme. Agric. Trop. Subtrop. 57, 45–59; doi:10.2478/ats-2024-0005 Liu, X., Yan, H., Lv, L., Xu, Q., Yin, C., Zhang, K., Wang, P. and Hu, J. 2012. Growth performance and meat quality of broiler chickens supplemented with Bacillus licheniformis in drinking water. Asian-Australas. J. Anim. Sci. 25(5), 682–689; doi:10.5713/ajas.2011.11334 Luo, J., Chen, D., Mao, X., He, J., Yu, B., Cheng, L. and Zeng, D. 2019. Purified β-glucans of different molecular weights enhance growth performance of LPS-challenged piglets via improved gut barrier function and microbiota. Animals 9(9), 602; doi:10.3390/ani9090602 Mariño, IE. and Roa, ML. 2021. Production parameters and digestibility of chickens using cayenne pepper (Hibiscus rosa-sinensis) and a probiotic (Lactobacillus acidophilus) plus pectin. Orinoquia 25(1), 35–46; doi:10.22579/20112629.654 Markowiak, P. and Śliżewska, K. 2018. The role of probiotics, prebiotics and synbiotics in animal nutrition. Gut Pathog. 10, 21; doi:10.1186/s13099-018-0250-0 Marquis., V., Schulthess., J., Molst., F., Santos. and RR. 2025. Effect of yeast β-glucan on the performance, intestinal integrity, and liver function of broiler chickens fed a diet naturally contaminated with Fusarium mycotoxins. Toxins 17(2), 51; doi:10.3390/toxins17020051 Mehdi, Y., Létourneau-Montminy, M.P., Gaucher, M.L., Chorfi, Y., Suresh, G., Rouissi, T., Brar, S.K., Côté, C., Ramirez, A.A. and Godbout, S. 2018. Use of antibiotics in broiler production: global impacts and alternatives. Anim. Nutr. 4(2), 170–178; doi:10.1016/j.aninu.2018.03.002 Meng, W.S., Zou, Q., Xiao, Y., Ma, W., Zhang, J., Wang, T. and Li, D. 2023. Growth performance and cecal microbiota of broiler chicks as affected by drinking water disinfection and/or herbal extract blend supplementation. Poult. Sci. 102(6), 102707; doi:10.1016/j.psj.2023.102707 Mohamed, A., Khalil, M., Soliman, F. and El-Sabrout, K. 2025. The effect of drinking ionized water on the productive performance, physiological status, and carcass characteristics of broiler chicks. Animals 15(2), 229; doi:10.3390/ani15020229 Muthukumaran, J., Jialic, C., Stephen, S. and Baojun, X. 2018. A critical review on the impacts of β-glucans on gut microbiota and human health. J. Nutr. Biochem. 61, 101–110; doi:10.1016/j.jnutbio.2018.06.010 Nasimov, S., Mamatova, Z., Sattorov, J., Safarov, X. and Azimova, D. 2024. Effect of the use of a probiotic Innoprovet on the growth parameters of broiler chickens (Uzbekistan). In: BIO Web of Conferences, Les Ulis, France: EDP Sciences, Vol. 95, p. 01035; doi: 10.1051/bioconf/20249501035 Nusairat, B., Odetallah, N., Tsai, C.Y. and Wang, J.J. 2024. Effect of dietary β-mannanase supplementation on broiler performance. Poult. Sci. 103(4), 103452; doi:10.1016/j.psj.2024.103452 Nwaigwe, C.U., Ihedioha, J.I., Shoyinka, S.V. and Nwaigwe, C.O. 2020. Evaluation of the hematological and clinical biochemical markers of stress in broiler chickens. Vet. World 13(10), 2294–2300; doi:10.14202/vetworld.2020.2294-2300 Ogbuewu, I.P., Mabelebele, M., Sebola, N.A., Mbajiorgu, C. 2022. Bacillus probiotics as alternatives to in-feed antibiotics and their influence on growth, serum chemistry, antioxidant status, intestinal histomorphology and lesion scores in disease-challenged broiler chickens. Front. Vet. Sci. 9, 876725; doi:10.3389/fvets.2022.876725 Onunkwo, D., Ufot, U., Ezenyilimba, B., Omumuabuike, J., Ezeoke, F. 2022. Blood profile of starter broiler chickens fed a diet containing leaf meal composite as an alternative to commercial broiler premix. Niger. J. Anim. Sci. 24(2), 103–110. Parisien, A., Allain, B., Zhang, J., Mandeville, R. and Lan, C.Q. 2007. Novel alternatives to antibiotics: bacteriophages, bacterial cell wall hydrolases, and antimicrobial peptides. J. Appl. Microbiol. 104(1), 1–13; doi:10.1111/j.1365-2672.2007.03498.x Polidoro, B.R., De Oliveira, M.J.K., Braga, F.D.S.C. and Polycarpo, G.D.V. 2025. Mannan oligosaccharide as an alternative to infeed antibiotics to improve growth performance of broilers: a meta-analysis. Br. Poult. Sci. 66(1), 98–106; doi:10.1080/00071668.2024.2391764 Roberts, T., Wilson, J., Guthrie, A., Cookson, K., Vancraeynest, D., Schaeffer, J., Moody, R. and Clark, S. 2015. New issues and science in broiler chicken intestinal health: emerging technology and alternative intervention. J. Appl. Poult. Res. 24(2), 257–266; doi:10.3382/japr/pfv023 Sanmiguel, R.A., Barreto, O.T., Javierre, J.A., Mogollon, D.C., León, M.L., Sosa, M.A. and Carvajal,E.L. 2024. Utilization of mannan oligosaccharides as antibiotic substitutes in laying hens. Vet. Res. Forum 15(12), 645–650; doi:10.30466/vrf.2024.2024105.4229 Sethiya, N.K. 2016. Review on natural growth promoters available for improving gut health of poultry: an alternative to antibiotic growth promoters. Asian. J. Poult. Sci. 10(1), 1–29; doi:10.3923/ajpsaj.2016 Slozhenkina, M.I., Frolova, M.V., Gorlov, I.F., Nikolaev, D.V., Mosolova, N.I. and Myasnikova, O.V. 2022. Improving the quality of broiler chicken meat without the use of antibiotics. IOP Conference Series: Earth and Environmental Science, Bristol, United Kingdom: IOP Publishing; Vol. 965(1), 012033; doi: 10.1088/1755-1315/965/1/012033 Sufiriyanto, S., Iriyanti, N. and Susanti, E. 2018. Haematology profiles and performance of broiler chickens fed on commercial feed. Anim. Prod. 20(3), 183–190; doi:10.20884/1.jap.2018.20.3.732 Szakacs, A.R., Matei, S., Ştefănuţ, L., Simcsak, R. and Macri, A. 2015. The impact of pre and probiotic on growing performance and haematological parameters in Ross 708 broiler chickens. Bull. UASVM. Vet. Med. 72(2), 335; doi:10.15835/buasvmcn-vm:11448 Timmerman, H.M., Veldman, A., Van Den Elsen, E., Rombouts, F.M. and Beynen, A.C. 2006. Mortality and growth performance of broilers given drinking water supplemented with chicken-specific probiotics. Poult. Sci. 85(8), 1383–1388; doi:10.1093/ps/85.8.1383 Udet, F.U., Udeh, V.C. and Chukwudi, P. 2020. Serum biochemistry profile and liver function indices of broiler chickens served with dietary inclusion of probiotics (Saccharomyces cerevisiae) and enzymes. Niger. J. Anim. Prod. 47(6), 120–128. Wang, Y., Ames, N.O., Tun, H.M., Tosh, S.M., Jones, P.J. and Khafipour, E. 2016. High molecular weight barley β-Glucan alters gut microbiota toward reduced cardiovascular disease risk. Front. Microbiol. 7, 129; doi:10.3389/fmicb.2016.00129 Wickramasuriya, S.S., Park, I., Lee, K., Lee, Y., Kim, W.H., Nam, H. and Lillehoj, H.S. 2022. Role of physiology, immunity, microbiota, and infectious diseases in the gut health of poultry. Vaccine 10(2), 172; doi:10.3390/vaccines10020172 Yazhini, P., Visha, P., Selvaraj, P., Vasanthakumar, P. and Chandran, V. 2018. Dietary encapsulated probiotic effect on broiler serum biochemical parameters. Vet. World 11(9), 1344–1348; doi:10.14202/vetworld.2018.1344-1348 Yu, M., Oketch, E.O., Nawarathne, S.R., Chathuranga, N.C., Maniraguha, V., Sta. Cruz, B.G., Seo, E., Lee, J., Park, H., Choi, H., Yi, Y.J., Chae, K.S., Yu, J. and Heo, J.M. 2025. Metabolizable energy and amino acid-deficient diets supplemented with β-mannanase in response to growth performance, intestinal health, and immune response in broilers. Poult. Sci. 104(1), 105222; doi:10.1016/j.psj.2025.105222 Zálešáková, D., Novotný, J., Řiháček, M., Horáková, L., Mrkvicová, E., Šťastník, O. and Pavlata, L. 2025. The blood biochemical parameters intervals and dynamics in modern broiler chickens. Vet. Anim. Sci. 29, 100465; doi:10.1016/j.vas.2025.100465 | ||
| How to Cite this Article |
| Pubmed Style Vivas L, Brazón M, Pérez N, Casado M, Mendoza A, Coll D, Farias H, Zavala D, Quintero A. A Bacillus-based synbiotic blend improves broiler productivity and metabolic resilience under suboptimal water quality and heat stress. Open Vet. J.. 2026; 16(3): 1913-1924. doi:10.5455/OVJ.2026.v16.i3.46 Web Style Vivas L, Brazón M, Pérez N, Casado M, Mendoza A, Coll D, Farias H, Zavala D, Quintero A. A Bacillus-based synbiotic blend improves broiler productivity and metabolic resilience under suboptimal water quality and heat stress. https://www.openveterinaryjournal.com/?mno=303701 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.46 AMA (American Medical Association) Style Vivas L, Brazón M, Pérez N, Casado M, Mendoza A, Coll D, Farias H, Zavala D, Quintero A. A Bacillus-based synbiotic blend improves broiler productivity and metabolic resilience under suboptimal water quality and heat stress. Open Vet. J.. 2026; 16(3): 1913-1924. doi:10.5455/OVJ.2026.v16.i3.46 Vancouver/ICMJE Style Vivas L, Brazón M, Pérez N, Casado M, Mendoza A, Coll D, Farias H, Zavala D, Quintero A. A Bacillus-based synbiotic blend improves broiler productivity and metabolic resilience under suboptimal water quality and heat stress. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1913-1924. doi:10.5455/OVJ.2026.v16.i3.46 Harvard Style Vivas, L., Brazón, . M., Pérez, . N., Casado, . M., Mendoza, . A., Coll, . D., Farias, . H., Zavala, . D. & Quintero, . A. (2026) A Bacillus-based synbiotic blend improves broiler productivity and metabolic resilience under suboptimal water quality and heat stress. Open Vet. J., 16 (3), 1913-1924. doi:10.5455/OVJ.2026.v16.i3.46 Turabian Style Vivas, Lorena, Marlon Brazón, Neyo Pérez, Moraiza Casado, Anthony Mendoza, David Coll, Hennet Farias, Danilo Zavala, and Alberto Quintero. 2026. A Bacillus-based synbiotic blend improves broiler productivity and metabolic resilience under suboptimal water quality and heat stress. Open Veterinary Journal, 16 (3), 1913-1924. doi:10.5455/OVJ.2026.v16.i3.46 Chicago Style Vivas, Lorena, Marlon Brazón, Neyo Pérez, Moraiza Casado, Anthony Mendoza, David Coll, Hennet Farias, Danilo Zavala, and Alberto Quintero. "A Bacillus-based synbiotic blend improves broiler productivity and metabolic resilience under suboptimal water quality and heat stress." Open Veterinary Journal 16 (2026), 1913-1924. doi:10.5455/OVJ.2026.v16.i3.46 MLA (The Modern Language Association) Style Vivas, Lorena, Marlon Brazón, Neyo Pérez, Moraiza Casado, Anthony Mendoza, David Coll, Hennet Farias, Danilo Zavala, and Alberto Quintero. "A Bacillus-based synbiotic blend improves broiler productivity and metabolic resilience under suboptimal water quality and heat stress." Open Veterinary Journal 16.3 (2026), 1913-1924. Print. doi:10.5455/OVJ.2026.v16.i3.46 APA (American Psychological Association) Style Vivas, L., Brazón, . M., Pérez, . N., Casado, . M., Mendoza, . A., Coll, . D., Farias, . H., Zavala, . D. & Quintero, . A. (2026) A Bacillus-based synbiotic blend improves broiler productivity and metabolic resilience under suboptimal water quality and heat stress. Open Veterinary Journal, 16 (3), 1913-1924. doi:10.5455/OVJ.2026.v16.i3.46 |