| Research Article | ||
Open Vet. J.. 2026; 16(3): 1610-1618 Open Veterinary Journal, (2026), Vol. 16(3): 1610-1618 Research Article Evaluation of natural plant extracts as alternatives to chemical antiparasitics in Eimeria-infected chickensSomia Mustafa Elagib1,2*1Biology Department, Faculty of Science, Al-Baha University, Al Bahah, Kingdom of Saudi Arabia 2Science Department, Faculty of Teachers, Nile Valley University, Edammer, Sudan *Corresponding Author: Somia Mustafa Elagib. Biology Department, Faculty of Science, Al-Baha University, Al Bahah, Kingdom of Saudi Arabia. Email: smmhammed [at] bu.edu.sa Submitted: 12/12/2025 Revised: 15/02/2026 Accepted: 25/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractBackground: Coccidiosis caused by Eimeria spp. remains one of the most economically important diseases in poultry production worldwide. The extensive use of chemical anticoccidials has led to increasing drug resistance and concerns regarding residues, highlighting the need for effective and safe natural alternatives. Aim: This study aimed to evaluate the anticoccidial efficacy of selected natural plant extracts as alternatives to conventional chemical antiparasitic drugs in broiler chickens experimentally infected with Eimeria spp. Methods: Broiler chickens were allocated into four experimental groups: non-infected control, infected untreated control, chemically treated, and plant extract-treated groups. Birds were monitored for growth performance, fecal oocyst shedding, intestinal lesion scores, histopathological changes, hematological parameters, and immunological, biochemical, and antioxidant responses. Results: Plant extract supplementation significantly improved body weight gain and feed conversion ratio, reduced oocyst shedding, and lowered intestinal lesion scores compared with infected untreated birds (p < 0.05). Histopathological examination revealed marked preservation of intestinal architecture in extract-treated birds, comparable to the chemically treated group. Additionally, plant extracts enhanced antioxidant enzyme activities, improved immunoglobulin levels, normalized inflammatory and organ function biomarkers, and restored hematological parameters disrupted by Eimeria infection. Conclusion: The evaluated plant extracts demonstrated pronounced anticoccidial, immunomodulatory, and antioxidant effects, with efficacy comparable to that of chemical anticoccidials. These findings support the potential use of natural plant extracts as safe, effective, and sustainable alternatives for controlling coccidiosis in poultry production. Keywords: Anticoccidial activity, Broiler chickens, Coccidiosis, Drug resistance, Plant extracts. IntroductionCoccidiosis caused by protozoan parasites of the genus Eimeria remains a major global challenge in poultry production. The disease manifests as enteric lesions, impaired weight gain, poor feed conversion, and significant morbidity. It inflicts an annual economic toll of billions worldwide (Almeida et al., 2014; Qaid et al., 2021; Coroian et al., 2022; Hou et al., 2024; Sharma et al., 2024). Historically, disease control has heavily depended on chemical anticoccidials such as nicarbazin, ionophores (e.g., salinomycin, narasin), and ethopabate (Abbas et al., 2012; Drăgan et al., 2014; Barbour et al., 2015; Alvarenga et al., 2015; Muthamilselvan et al., 2016). However, the prolonged and widespread use of these drugs has accelerated the emergence of drug-resistant Eimeria strains, raising serious concerns about the environmental impact of chemical residues in poultry products. In this context, phytogenic alternatives—natural plant-based compounds including essential oils, flavonoids, tannins, and saponins—have gained attention as promising, sustainable antiparasitic agents. These botanicals act through multifaceted mechanisms: exerting direct protozoacidal effects, providing antioxidant and anti-inflammatory benefits, protecting gut barrier integrity, and modulating immune responses (Zhang et al., 2012; Surai et al., 2024). Empirical evidence supports the anticoccidial efficacy of several plant extracts. Artemisia annua, which is rich in artemisinin, has consistently demonstrated reductions in lesion scores and oocyst shedding while enhancing weight gain. Field supplementation in broilers challenged with Eimeria tenella yielded up to an 80% reduction in oocyst shedding and markedly improved gut health. Similarly, ethanolic blends of A. annua and Curcuma longa produced intermediate protection compared with ionophores, with observed survival benefits. Additionally, the use of Rumex nervosus leaf powder significantly reduced oocyst excretion and lesion severity in E. tenella-infected broilers. A tannin-dense herbal formula combining A. annua, Quercus infectoria, and Allium sativum decreased oocyst counts and restored intestinal morphology—presumably via direct parasite membrane disruption and immunostimulatory action. Six phytochemicals—silymarin, dihydroartemisinin, genistein, resveratrol, quercetin, and nerolidol—showed moderate efficacy against Eimeria spp. in vitro, representing diverse botanical agents for further study (Arabkhazaeli et al., 2013; Soleimani PS Delghandi; and Moallem G Karimi, 2019). The observed mechanisms of action are notable. Phenolic compounds cause direct damage to parasite membranes, while artemisinin disrupts protozoal intracellular processes via reactive oxygen species derived from peroxide bridges. Concurrently, antioxidant activity mitigates oxidative stress, supporting gut epithelial function. Immune modulation is evidenced by increased pro-inflammatory cytokines, enhanced lymphocyte proliferation, and activation of NF-κB signaling, which promote apoptosis of infected cells. Gut barrier integrity is strengthened through improved villus architecture, mucosal healing, and favorable microbiota support (Ghafouri et al., 2023; Carpio et al., 2024). Besides anticoccidial effects, phytogenic supplementation often yields enhanced growth performance and economic value. A dietary inclusion of 0.6% A. annua was found to boost growth, upregulate the expression of antioxidant genes, and improve economic returns. Similarly, Taraxacum mongolicum (dandelion) supplementation resulted in improved expression of intestinal barrier genes, elevated IL10 and occludin levels, and favorable shifts in gut microbiota. Moreover, oregano oil-based additives have been shown to improve gut histology and nutrient absorption while increasing tolerance to coccidial challenge (NRC, 1994; Dalloul and Lillehoj, 2006; Baradaran et al., 2019). However, several limitations exist. Dosage optimization remains unresolved—effective ranges vary widely, and some formulations do not match the efficacy of chemical drugs. Mechanistic understanding at the systemic and molecular levels is still incomplete. Furthermore, field validation is limited; while some promising field trials, including one in Romania evaluating A. annua, broader commercial-scale studies are scarce. Moreover, challenges in formulation standardization, stability, and delivery mechanisms remain substantial. Given the urgent need for safe, effective, and residue-free alternatives, our study focuses on three botanicals—A. annua, C. longa, and A. sativum. This study aimed to comprehensively evaluate the anticoccidial efficacy of these agents in broiler chickens under controlled experimental conditions. By measuring key performance indicators, such as oocyst excretion, lesion severity, growth parameters, immune modulation, and gut integrity, and comparing outcomes directly to standard chemical anticoccidials, this research seeks to generate robust data supporting the integration of phyto-based therapeutics into holistic coccidiosis management programs. Materials and MethodsExperimental designA total of 240 1-day-old male broiler chicks (Ross 308) were obtained from a commercial hatchery and reared under standard environmental and hygienic conditions. Upon arrival, the chicks were weighed and randomly divided into four treatment groups (n=60 per group), with each group further subdivided into 3 replicates of 20 birds each. The experimental period lasted for 42 days. The four groups were as follows:
Eimeria infection challengeAt day 14, chickens in G2–G4 were orally inoculated with a mixed suspension of Eimeria acervulina, E. maxima, and E. tenella at a total dose of 1 × 10⁵ sporulated oocysts per bird. The oocysts were previously propagated and identified through microscopy and polymerase chain reaction assays from field isolates and sporulated using 2.5% potassium dichromate solution (AOAC, 2005; Abbas et al., 2009; Wiedosari and Wardhana, 2018; Ghafouri et al., 2023). Preparation of the plant extractsDried and powdered leaves of A. annua, C. longa rhizomes, and A. sativum bulbs were obtained from certified botanical sources. Each sample was subjected to ethanol extraction (70% v/v) using a Soxhlet apparatus for 8 hours. The extracts were filtered, concentrated using a rotary evaporator at 45°C, and then spray-dried to obtain a stable powder. The prepared powders were mixed into the basal diet using a vertical mixer to ensure homogeneity (Belote et al., 2019; Ghafouri et al., 2023; Hascoët et al., 2025). Housing and feedingThe chickens were housed in floor pens with fresh wood shavings and provided ad libitum access to feed and water. The basal diet was formulated according to the recommendations of the National Research Council (1994) and was free of coccidiostats, growth promoters, or antibiotics. The temperature, humidity, and lighting were maintained according to the Ross 308 management guidelines. Data collection and evaluation criteria
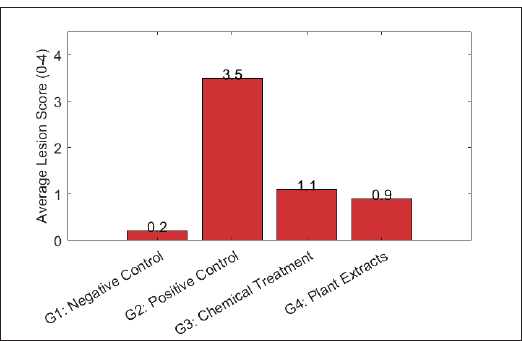
Statistical analysisAll data were analyzed using the Statistical Package for the Social Sciences v26.0 (IBM Corp.). One-way ANOVA followed by Tukey’s honestly significant difference test was used for multiple comparisons among groups. Mortality rates were analyzed using the chi-square test. A p-value <0.05 was considered statistically significant. Results are presented as mean ± standard deviation. Ethical approvalAll applicable international, national, and institutional guidelines for the care and use of animals in Al Baha University, Saudi Arabia were followed according to REF.7.1.4.2. ResultsThis study investigated the effectiveness of selected natural plant extracts (A. annua, C. longa, and A. sativum) in mitigating Eimeria spp. infections in broiler chickens compared with a standard chemical anticoccidial (salinomycin). The outcomes were evaluated based on lesion scores, oocyst shedding, BW gain, and intestinal tissue histological integrity. Lesion scoresFigure 1 shows the mean intestinal lesion scores recorded in broiler chickens from the four experimental groups following the challenge with Eimeria spp. The positive control group (G2), infected and untreated, exhibited the highest lesion score (3.5), indicating severe intestinal damage. In contrast, both the chemically treated group (G3) and the plant extract-treated group (G4) showed significantly lower lesion scores, with mean values of 1.1 and 0.9, respectively (p < 0.05) compared with G2. No significant difference was observed between the G3 and G4 groups (p > 0.05). The negative control group (G1) maintained a minimal lesion score (0.2), indicating the absence of intestinal pathology.
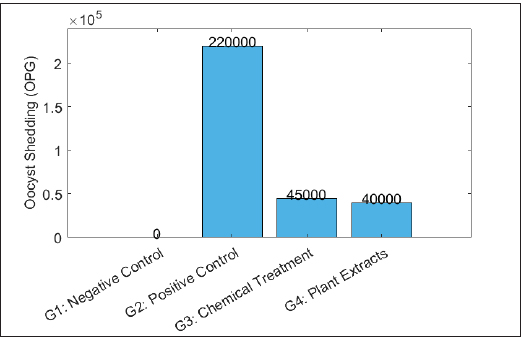
Fig. 1. Average lesion scores in broiler chickens treated with chemical and natural anticoccidials following Eimeria infection. Oocyst sheddingFigure 2 shows the mean oocyst shedding, expressed as OPG of feces, recorded for the four experimental groups. The highest oocyst output was observed in the positive control group (G2), which reached a mean value of 2.2 × 10⁵ OPG. In contrast, both treatment groups exhibited significantly lower oocyst counts (p < 0.05). The chemically treated group (G3) showed a mean oocyst shedding of 4.5 × 10⁴ OPG, whereas the plant extract-treated group (G4) recorded 4.0 × 10⁴ OPG. No significant difference was detected between G3 and G4 (p > 0.05). The negative control group (G1) exhibited no detectable oocyst shedding throughout the experimental period.
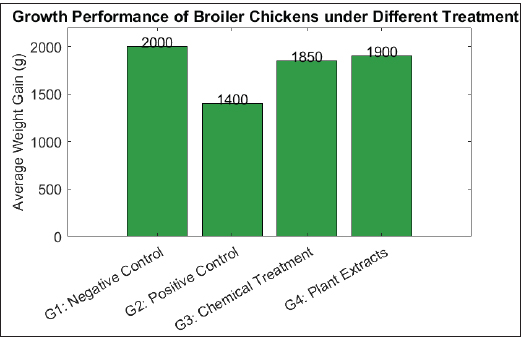
Fig. 2. Oocyst shedding in broiler chickens after Eimeria infection and treatment with chemical and natural anticoccidials. Growth performanceFigure 3 shows the average BW gain of broiler chickens across the four experimental groups. The negative control group (G1) had the highest mean weight gain (2,000 g). In contrast, the positive control group (G2) exhibited the lowest weight gain (1,400 g). Birds in the chemically treated group (G3) showed a higher mean weight gain (1,850 g) than those in the G2 group. The plant extract-treated group (G4) achieved a mean weight gain of 1,900 g, which was higher than that of G2 and G3. Statistical analysis revealed significant differences among the experimental groups (p < 0.05).
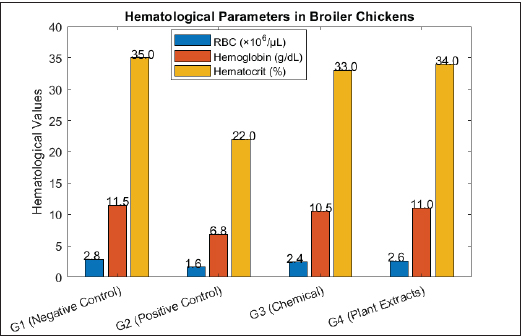
Fig. 3. Growth performance (average weight gain) of broiler chickens after infection with Eimeria and treatment with chemical and natural anticoccidials. HistopathologyIntestinal histopathological examination revealed marked differences between the experimental groups. Severe pathological alterations were observed in the positive control group (G2), including extensive epithelial necrosis, villus atrophy, ulceration, and submucosal thickening. Numerous developmental stages of Eimeria spp., including schizonts, gametocytes, and oocysts, were present within the mucosal epithelium, accompanied by dense mononuclear cell infiltration and focal hemorrhages. In the chemically treated group (G3), intestinal sections showed partial restoration of villus architecture, with reduced epithelial damage and fewer parasitic stages compared to G2. Inflammatory cell infiltration was present but less pronounced than that in the positive control group. The plant extract-treated group (G4) exhibited relatively preserved intestinal morphology. The mucosal epithelium remained largely intact, with improved villus structure, reduced inflammatory cell infiltration, and limited parasitic stages, which appeared degenerated in some sections. Lesion severity scores, quantified using established histopathological scoring criteria, were significantly lower in G4 than in G2 (p < 0.05) and were not significantly different from those observed in G3. Histopathological scoring using the Institute for Scientific Information method demonstrated consistency with conventional lesion scoring, with lower histological damage scores recorded in G3 and G4 than in G2. Immunological and biochemical parametersSignificant differences in immunological and biochemical parameters were observed between the experimental groups. Immunoglobulin levels (IgA and IgG)Serum IgA and IgG concentrations were significantly higher in the plant extract-treated group (G4) than in the positive control group (G2) (p < 0.05). The chemically treated group (G3) also showed increased immunoglobulin levels compared with G2, although the values in G4 were numerically higher. The negative control group (G1) maintained baseline Ig levels. Cytokine levelsThe positive control group (G2) exhibited significantly elevated serum levels of interleukin-1β, tumor necrosis factor-α, and IFN-γ compared to all other groups (p < 0.05). The chemically treated group (G3) and the plant extract-treated group (G4) showed significantly reduced cytokine levels relative to G2, with no significant difference between G3 and G4 (p > 0.05). Antioxidant enzyme activityThe activities of SOD, CAT, and Glutathione Peroxidase were significantly reduced in the positive control group (G2) compared to the negative control group (G1) (p < 0.05). In contrast, G3 and G4 birds exhibited significantly higher antioxidant enzyme activities than G2. Enzyme levels in G4 were comparable to those in G1. Liver and kidney function indicatorsSerum alanine aminotransferase, aspartate aminotransferase, and creatinine concentrations were significantly elevated in the positive control group (G2) relative to all other groups (p < 0.05). Both treatment groups (G3 and G4) showed significantly lower values of these biomarkers compared with G2, with no significant difference between G3 and G4. Acute phase proteinsThe highest serum levels of CRP and serum amyloid A were recorded in G2. Significant reductions in both parameters were observed in G3 and G4 compared with G2 (p < 0.05), while values in the negative control group (G1) remained within normal ranges. Hematological parametersThe hematological parameters significantly differed among the experimental groups, as shown in Figure 4. The positive control group (G2) exhibited significantly lower red blood cell count (1.6 × 10⁶/μl), hemoglobin concentration (6.8 g/dl), and hematocrit value (22%) than the other groups (p < 0.05). The negative control group (G1) recorded the highest values for red blood cell count (2.8 × 10⁶/μl), hemoglobin (11.5 g/dl), and hematocrit (35%).
Fig. 4. Effect of treatments on hematological parameters in chickens infected with Eimeria spp. Birds in the chemically treated group (G3) showed intermediate values for all hematological parameters, which were significantly higher than those observed in G2 (p < 0.05). The red blood cell count, hemoglobin concentration, and hematocrit values of the plant extract-treated group (G4) were comparable to those of the negative control group (G1), with no significant differences between these two groups (p > 0.05). DiscussionAnticoccidial efficacy of plant extractsThis study demonstrated that the combination of A. annua, C. longa, and A. sativum exhibited strong anticoccidial activity against Eimeria spp. infection in broiler chickens. This was evidenced by significant reductions in lesion scores and oocyst shedding in the plant extract-treated group (G4), which were comparable to those observed in birds treated with the conventional anticoccidial drug salinomycin (G3). The marked decrease in oocyst output indicates effective suppression of parasite replication and transmission, which is a critical criterion for evaluating anticoccidial efficacy. These findings are consistent with those of previous reports highlighting the antiparasitic potential of phytogenic compounds. Artemisia annua contains artemisinin and related sesquiterpene lactones that interfere with the development of intracellular parasites. Curcuminoids from C. longa and organosulfur compounds from A. sativum have been shown to impair oocyst sporulation and viability. Similar reductions in oocyst shedding and lesion severity following phytogenic supplementation have been reported in broilers challenged with E. tenella and E. acervulina (Abbas et al., 2009; Ghafouri et al., 2023). Effects on intestinal integrity and immunityHistopathological findings clearly demonstrated that Eimeria infection severely compromised the intestinal architecture of untreated birds, characterized by villus atrophy, epithelial necrosis, and inflammatory infiltration. In contrast, birds treated with plant extracts showed marked preservation of intestinal morphology, including restored villus structure and reduced parasitic stages, closely resembling the chemically treated group. Enhanced immunological responses, particularly increased serum IgA and IgG levels, supported the improved intestinal integrity observed in G4. IgA plays a pivotal role in mucosal immunity by limiting parasite adhesion and invasion at the intestinal surface, whereas IgG contributes to systemic immune defense. Enhanced immunoglobulin responses following phytogenic supplementation have been previously reported and are associated with improved resistance to enteric pathogens (Lee et al., 2010; Hascoët et al., 2025). The present findings reinforce the concept that plant-derived compounds can simultaneously reduce parasitic burden and support immune competence. Antioxidant and anti-inflammatory mechanismsOxidative stress and excessive inflammation are hallmarks of coccidial infection and significantly contribute to tissue damage and performance impairment. In the current study, untreated infected birds exhibited suppressed antioxidant enzyme activity (SOD, CAT, and GPx) and elevated inflammatory markers, including proinflammatory cytokines and acute phase proteins. Plant extract treatment restored antioxidant enzyme levels and significantly reduced inflammatory mediators, indicating effective modulation of oxidative and inflammatory pathways. These effects are biologically plausible given the antioxidant and anti-inflammatory properties of the selected plants. Curcumin is a potent scavenger of reactive oxygen species and a known inhibitor of NF-κB signaling, while garlic-derived allicin and Artemisia flavonoids have demonstrated strong free radical–neutralizing capacity (Aggarwal and Harikumar, 2009; Guimarães et al., 2013). Similar improvements in antioxidant status and inflammatory markers in coccidiosis-challenged broilers have been reported in recent veterinary studies, including those indexed in OVJ and related veterinary journals (Abd-Elrahman et al., 2022). Furthermore, the normalization of liver and kidney function biomarkers in G4 indicates systemic protection beyond the intestinal tract, reinforcing the multifunctional benefits of phytogenic therapies in poultry health management. Comparison with chemical anticoccidial agentsConventional anticoccidials, such as salinomycin, remain effective but are increasingly associated with drug resistance, residue accumulation, and regulatory restrictions. In the present study, the plant extract combination achieved anticoccidial efficacy comparable to that of salinomycin across multiple parameters, including lesion scores, oocyst shedding, growth performance, histopathology, immune response, and hematological indices (Eldeeb et al., 2025 and Salah et al., 1995). Notably, birds treated with plant extracts exhibited growth performance and hematological values approaching those of uninfected controls, highlighting the additional health-promoting effects of chemical anticoccidials. This aligns with emerging evidence that phytogenic additives can provide broader physiological benefits, including improved gut health and systemic resilience, rather than acting solely as antiparasitic agents. The present findings support the integration of phytogenic strategies as effective, safe, and environmentally friendly alternatives to synthetic anticoccidials into modern poultry production systems. ConclusionThe present study provides robust evidence that selected natural plant extracts, particularly A. sativum and A. annua, exert significant protective effects against Eimeria spp. infection in broiler chickens. Compared with untreated infected birds, phytogenic supplementation resulted in marked reductions in intestinal lesion scores and oocyst shedding, along with significant improvements in growth performance and hematological parameters. Histopathological evaluation further confirmed intestinal architecture preservation and reduced tissue damage in birds receiving plant-based treatment. The anticoccidial efficacy of the plant extract combination was comparable to that of the conventional chemical anticoccidial salinomycin across multiple parameters. In addition to controlling parasitic infection, phytogenic treatment was associated with improved immune status, enhanced antioxidant enzyme activity, and normalization of liver and kidney function-related biochemical markers, indicating broad systemic benefits during coccidial challenge. These findings support the use of natural plant extracts as effective and sustainable alternatives to synthetic anticoccidials, particularly in the context of increasing concerns regarding drug resistance, chemical residues, and consumer demand for residue-free poultry products. Although the present results are promising, further investigations are recommended to elucidate underlying molecular mechanisms, optimize dosing strategies, and validate efficacy under large-scale commercial production systems. Overall, the integration of phytogenic compounds into poultry health management programs may represent a viable approach for improving disease control, animal welfare, and production efficiency in a sustainable manner. AcknowledgmentsNo specific acknowledgment is required. Conflict of interestThe author declares that there are no conflicts of interest. FundingThis research received no specific grant from any funding agency, commercial, or not-for-profit sector. Authors' contributionsSomia Mustafa: Conceptualization, Methodology, Investigation, Data curation, Formal analysis, Original draft writing, Review and editing, Visualization, Supervision. Data availabilityThe data supporting the findings of this study are available from the corresponding author upon reasonable request. All data were generated and analyzed during the current study. ReferencesAbbas, R.Z., Colwell, D.D. and Gilleard, J. 2012. Botanicals: an alternative approach for the control of avian coccidiosis. World’s Poult. Sci. J. 68, 203–215. Abbas, R.Z., Iqbal, Z., Khan, M.N., Zafar, M.A. and Zia, M.A. 2009. Anticoccidial activity of Curcuma longa L. in broilers. Braz. Arch. Biol. Technol. 52(4), 963–967. Abd-Elrahman, S.M., Mohamed, S.A.A., Mohamed, S.E., El-Khadragy, M.F., Dyab, A.K., Hamad, N., Safwat, M.M., Nasr, A.A.E., Alkhaldi, A.A.M., Gareh, A. and et al. 2022. Comparative effect of allicin and alcoholic garlic extract on the morphology and infectivity of chicken Eimeria tenella oocysts. Animals 12, 3185; doi: 10.3390/ani12223185 Aggarwal, B.B. and Harikumar, K.B. 2009. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune, and neoplastic diseases. Cell Biol. 41(1), 40–59; doi:10.1016/j.biocel.2008.06.010 Almeida, G.F.D., Thamsborg, S.M., Madeira, A.M.B.N. and et al. 2014. Effects of combining ethanolic extracts of Artemisia annua and Curcuma longa in broilers challenged with Eimeria acervulina and Eimeria maxima infective oocysts. Parasitology 141(3), 347–355; doi:10.1017/S0031182013001443 Almeida, F.N., Htoo, J.K., Thomson, J. and Stein, H.H. 2013. Amino acid digestibility in camelina products fed to growing pigs. Can. J. Anim. Sci. 93 (3), 335–343. Alvarenga, R.R., Rodrigues, P.B., Zangeronimo, M.G., Oliveira, E.C., Mariano, F.C.M.Q., Lima, E.M.C., Garcia Jr, A.A.P., Naves, L.P. and Nardelli, N.B.S. 2015. Validation of prediction equations of energy values of a single ingredient or their combinations in male broilers. Asian-Aust. J. Anim. Sci. 28, 1335–1344. AOAC. 2005. Official Methods of Analysis of AOAC International, 18th ed. Gaithersburg, MD: AOAC International. Arabkhazaeli, F., Modrisanei, M., Nabian, S., Mansoori, B. and Madani, A. 2013. Evaluating the resistance of field isolates of Eimeria spp to anticoccidial drugs using three different indices. Iran. J. Parasitol. 8(2), 234–241. Baradaran, A., Samadi, F., Ramezanpour, S.S. and Yousefdoust, S. 2019. Hepatoprotective effects of silymarin on CCl4-induced hepatic damage in broiler chickens model. Toxicol. Rep. 6, 788–794. Barbour, E.K., Ayyash, D.B., Iyer, A. and Harakeh, S.T.K.A. 2015. Review of approaches targeting the replacement of coccidiostat application in poultry production. Poultry Sci. 17(4), 405–418. Belote, B.L., Soares, I., Tujimoto-Silva, A., Sanches, A.W.D., Kraieski, A.L. and Santin, E. 2019. Applying “I see inside” histological methodology to evaluate gut health in broilers challenged with Eimeria. Parasitol 276S, 100004; doi:10.1016/j.vpoa.2019.100004 Carpio, M.B., Valdes-Pena, M.A., Molina, D.A., Cabello, S.E.E., Guerrero, C.A.S., Cribillero, G., Coca, K.F.V. and Icochea, E. 2024. Evaluation of commercial feed additive and silymarin doses on broiler performance with and without CCl4-induced liver damage. Poult. Sci. 103, 103567. Coroian, M., Pop, L.M., Popa, V., Friss, Z., Oprea, O., Kalmár, Z., Pintea, A., Borșan, S.D., Mircean, V., Lobonțiu, I., Militaru, D., Vârban, R. and Györke, A. 2022. Efficacy of Artemisia annua against coccidiosis in broiler chickens: a field trial. Microorganisms 10, 2277; doi:10.3390/microorganisms10112277 Dalloul, R.A. and Lillehoj, H.S. 2006. Poultry coccidiosis: recent advancements in control measures and vaccine development. Expert Rev. Vaccines 5(1), 143–163; doi:10.1586/14760584.5.1.143 Drăgan, L., Györke, A., Ferreira, J.F., Pop, I.A., Dunca, I., Drăgan, M., Mircean, V., Dan, I. and Cozma, V. 2014. Effects of Artemisia annua and Foeniculum vulgare on Eimeria tenella (phylum Apicomplexa)-infected chickens. Acta. Vet. Scand. 56, 22; doi:10.1186/1751-0147-56-22 Eldeeb, F.A., Noseer, E.A., Abdelazeem, S., Ali, E., Basher, A.W., Abdalla, M.A.A. and Ibrahim, H.H. 2025. Effect of dietary supplementation of Lawsonia inermis and Acacia nilotica extract on growth performance, intestinal histopathology, and antioxidant status of broiler chickens challenged with coccidiosis. BMC Vet. Res. 21(2), doi: 10.1186/s12917-024-04409-w Ghafouri, S.A., Ghaniei, A., Sadr, S., Amiri, A.A., Tavanaee Tamannaei, A.E., Charbgoo, A., Ghiassi, S. and Dianat, B. 2023. Anticoccidial effects of tannin-based herbal formulation (Artemisia annua, Quercus infectoria, and Allium sativum) against coccidiosis in broilers. Dis 47(4), 820–828; doi:10.1007/s12639-023-01627-1 Guimarães, M.R., Leite, F.R., Spolidorio, L.C., Kirkwood, K.L. and Rossa C Jr. 2013. Curcumin abrogates LPS-induced pro-inflammatory cytokines in RAW 264.7 macrophages. Oral Biol. 58(10), 1309–1317; doi:10.1016/j.archoralbio.2013.07.005 Hascoët, A.S., Torres-Celpa, P., Riquelme-Neira, R. and Hidalgo-Olate, H. 2025. Evaluation of the efficacy of a phytogenic supplement (alkaloids and flavonoids) in the control of Eimeria spp. in experimentally challenged broiler chickens. Animals (Basel) 15(6), 847; doi:10.3390/ani15060847 Hassan Abd-Elrahman, S., Abd-Elhamid Hashem, F. and Awny Farag, A. 2022. Mitigation of heat stress effects on snake cucumber plants by silicon additions and rice straw soil cover. Commun. Soil Sci. Plant Anal. 53(6), 759–772. Hou, Y., Han, B., Lin, Z., Liu, Q., Liu, Z., Si, H. and Hu, D. 2024. Effects of six natural compounds and their derivatives on coccidiosis control in chickens. Microorganisms 12, 601; doi:10.3390/microorganisms12030601 Johnson, J. and Reid, W.M. 1970. Anticoccidial drugs: lesion scoring techniques in battery and floor-pen experiments with chickens. Exp. Parasitol. 28(1), 30–36. Lee, S.H., Lillehoj, H.S., Hong, Y.H., Jang, S.I., Lillehoj, E.P., Ionescu, C., Mazuranok, L. and Bravo, D. 2010. In vitro effects of plant and mushroom extracts on immunological function of chicken lymphocytes and macrophages. Br. Poult. Sci. 51(2), 213–221; doi:10.1080/00071661003745844 Muthamilselvan, T., Kuo, T.F., Wu, Y.C. and Yang, W.C. 2016. Herbal remedies for coccidiosis control: a review of plants, compounds, and anticoccidial actions. Evid-Based. Complement. Altern. Med. 2016(1), 657981; doi: 10.1155/2016/657981 National Research Council. 1994. Nutrient requirements of poultry, 9th revised ed. Washington, DC: National Academy Press. Qaid, M.M., Al-Mufarrej, S.I., Azzam, M.M., Al-Garadi, M.A., Albaadani, H.H., Alhidary, I.A. and Aljumaah, R.S. 2021. Anti-coccidial effect of Rumex nervosus leaf powder on broiler chickens infected with Eimeria tenella oocyst. J. Vaccines 11(Animals), 167; doi:10.3390/ani11010167 Salah, N., Miller, N.J., Paganga, G., Tijburg, L., Bolwell, G.P. and Rice-Evans, C. 1995. Polyphenolic flavanols as scavengers of aqueous phase radicals and as chain-breaking antioxidants. Acta 1222, 103–108. Sharma, M.K., Liu, G., Choppa, V.S.R., Rafieian-Naeini, H.R., Mahdavi, F.S., Marshall, B., Gogal RM Jr. and Kim, W.K. 2024. Effects of supplementation with Artemisia annua on the performance and gut health of laying hens challenged with mixed Eimeria species. Front. Physiol. 15, 1381548; doi:10.3389/fphys.2024.1381548 Soleimani, V., Delghandi, P.S., Moallem, S.A. and Karim,i G. 2019. Safety and toxicity of silymarin, the major constituent of milk thistle extract: an updated review. Phytother. Res. 33, 1627–1638; doi:10.1016/j.phyt.2019.01.003 Surai, P.F., Surai, A. and Earle-Payne, K. 2024. Silymarin and inflammation: food for thought. Antioxidants 13, 98. Wiedosari, E. and Wardhana, A.H. 2018. Anticoccidial activity of artemisinin and extract of Artemisia annua leaves in chicken infected by Eimeria tenella. Ilmu Ternak Dan Veter. 22, 196–204. Zhang, D., Sun, B., Yue, Y., Zhou, Q. and Du, A. 2012. Anticoccidial activity of Dichroa febrifuga Lour. extract against Eimeria tenella infection in chickens. Parasitol. Res. 111, 2229–2233. | ||
| How to Cite this Article |
| Pubmed Style Somia Mustafa Elagib. Evaluation of natural plant extracts as alternatives to chemical antiparasitics in Eimeria-infected chickens. Open Vet. J.. 2026; 16(3): 1610-1618. doi:10.5455/OVJ.2026.v16.i3.18 Web Style Somia Mustafa Elagib. Evaluation of natural plant extracts as alternatives to chemical antiparasitics in Eimeria-infected chickens. https://www.openveterinaryjournal.com/?mno=303978 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.18 AMA (American Medical Association) Style Somia Mustafa Elagib. Evaluation of natural plant extracts as alternatives to chemical antiparasitics in Eimeria-infected chickens. Open Vet. J.. 2026; 16(3): 1610-1618. doi:10.5455/OVJ.2026.v16.i3.18 Vancouver/ICMJE Style Somia Mustafa Elagib. Evaluation of natural plant extracts as alternatives to chemical antiparasitics in Eimeria-infected chickens. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1610-1618. doi:10.5455/OVJ.2026.v16.i3.18 Harvard Style Somia Mustafa Elagib (2026) Evaluation of natural plant extracts as alternatives to chemical antiparasitics in Eimeria-infected chickens. Open Vet. J., 16 (3), 1610-1618. doi:10.5455/OVJ.2026.v16.i3.18 Turabian Style Somia Mustafa Elagib. 2026. Evaluation of natural plant extracts as alternatives to chemical antiparasitics in Eimeria-infected chickens. Open Veterinary Journal, 16 (3), 1610-1618. doi:10.5455/OVJ.2026.v16.i3.18 Chicago Style Somia Mustafa Elagib. "Evaluation of natural plant extracts as alternatives to chemical antiparasitics in Eimeria-infected chickens." Open Veterinary Journal 16 (2026), 1610-1618. doi:10.5455/OVJ.2026.v16.i3.18 MLA (The Modern Language Association) Style Somia Mustafa Elagib. "Evaluation of natural plant extracts as alternatives to chemical antiparasitics in Eimeria-infected chickens." Open Veterinary Journal 16.3 (2026), 1610-1618. Print. doi:10.5455/OVJ.2026.v16.i3.18 APA (American Psychological Association) Style Somia Mustafa Elagib (2026) Evaluation of natural plant extracts as alternatives to chemical antiparasitics in Eimeria-infected chickens. Open Veterinary Journal, 16 (3), 1610-1618. doi:10.5455/OVJ.2026.v16.i3.18 |