| Original Article | ||
Open Vet J. 2022; 12(6): 936-943 Open Veterinary Journal, (2022), Vol. 12(6): 936–943 Original Research The ability to resist Newcastle disease through inherited immunity in different strains of broilers in Nineveh governorateThamer Abdulazeez Ezzulddin, Dhyaa Mohammad Taher Jwher* and Akeel Mohammad ShareefDepartment of Veterinary Public Health, College of Veterinary Medicine, University of Mosul, Mosul, Iraq Submitted: 22/08/2022 Accepted: 06/11/2022 Published: 03/12/2022 *Corresponding Author: Dhyaa Mohammad Taher Jwher. Department of Veterinary Public Health, College of Veterinary Medicine, University of Mosul, Mosul, Iraq. Email: diaataher [at] uomosul.edu.iq © 2022 Open Veterinary Journal
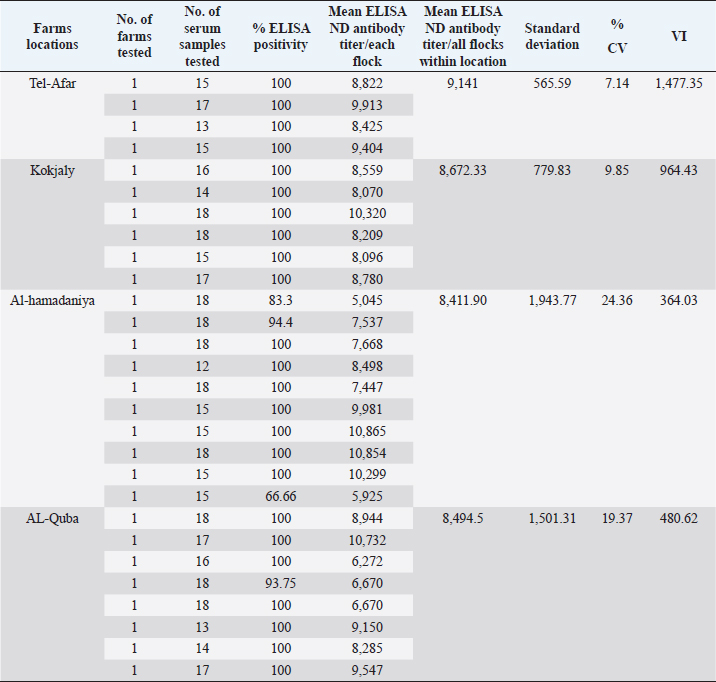
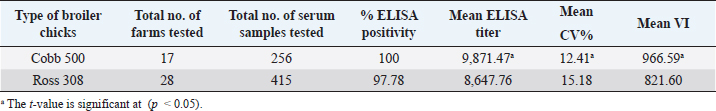
AbstractBackground: Newcastle disease (ND) is a viral disease that affects numerous bird species and is still present in many places, including Iraq. Aim: The purpose of this study was to compare maternal derived antibodies against against ND vaccination in 1-day-old imported Cobb 500 and Ross 308 chicks. Methods: From April to October 2020, four locations in Mosul governorate were chosen to compute the maternal derived antibodies from 1-day-old broiler chicks. Seven hundred and seven blood samples (451) from imported 28 Ross 308 flocks and 256 from 17 Cobb 500 flocks were obtained and tested for antibody levels using an indirect enzyme-linked immunosorbent assay assay (BioChek commercial kit, UK). Results: The Cobb 500 broiler chicks outperformed the Ross 308 broiler chicks in that all of the Cobb 500 broiler chicks had maternal antibody titers (100% positivity) versus 97.78% of the Ross 308 chicks, had a mean Maternal antibody titer of 9,871.47 versus 8,647.76, and had a mean CV percentage of 12.41% versus 15.18%, respectively. The mean vaccination index of Cobb 500 broiler chicks was 966.59 versus 821.60 for Ross 308 broiler chicks Conclusion: Although both broiler types produced chicks with high and uniform maternal antibody titers, Cobb 500 produced chicks with higher protective titers than Ross 308. Keywords: Cobb 500, Maternal antibody titers, Newcastle disease, Ross 308. IntroductionNewcastle disease (ND) is a virus in the family Paramyxoviridae and belongs to the genus Avula (Suarez et al., 2019). Even after the disease identified in 1926, it remains widespread in many nations, including Iraq (OIE, 2012). Despite the implementation of various biosecurity measures and ongoing monitoring programs, the disease—which the International Office for Animal Epidemiology lists as one of list A diseases (Alexander, 1991)—constantly poses a direct threat to the entire agricultural poultry industry and the food industry. The value of these vaccinations, which provide long-lasting immunity, has been clearly demonstrated by the use of inactivated vaccines in laying hens (Liljebjelke et al., 2008). This immunity approved on 1-day-old chicks, lowering the likelihood of ND illness infection. Enough supply of IgY antibodies is provided by the illiterately transmitted immunity through the egg yolk, allowing chicks to defend themselves until the development of their own immune systems to produce their own antibodies (Alexander, 1991; Kannaki et al., 2019). Here in Mosul Governorate, ND regarded as one of the most significant and hazardous ailments affecting broilers, laying hens, and other types of birds (Shareef and Gargees, 2008). Due to the severity of ND, the broiler industry in the Mosul governorate has suffered significant financial losses. ND avoided through immunization using vaccines. Attenuated ND vaccines and currently inactivated ND emulsion vaccines are widely used in addition to the applying biosecurity programs to control NDV (Turblin, 2009). These applications include quality check on 1-day-old chicks and the monitoring and serological assessment of antibodies in dams and chicks, which provide useful clues about the effectiveness of breeder’s vaccination and the choice of the best program for immunization against ND illness (Daniela et al., 2010). As a result, it is essential to evaluate maternal immunity in the first instance using a variety of techniques, including the agar gel precipitation test, enzyme-linked immunosorbent assay (ELISA), hemagglutination inhibition test, and virus neutralization test (Saif and Fadly, 2008). One of the diagnostic instruments widely employed in monitoring and managing antibody responses in chicken production is the enzyme immunoassay (ELISA), which is rapid, sensitive, specific, inexpensive, repeatable, and covert in its pursuit of this goal (Aldous et al., 2003). According to research, the production of antibodies against ND disease varies between broiler strains such as Ross 308 and Cobb 500 (Grindstaff et al., 2003). In recent years, the flocks of the Cobb 500 and Ross 308 are the two most popular commercial broiler strains. The former is an early-developing strain while the latter considered a late-developing strain (Danisman and Gous, 2011). As described by Yunis et al. (2000) and Cheema et al. (2003), there has been a lot of attention on growth enhancement, which is negatively associated with various elements of poultry immune performance. These and other research have shown that commercial broiler lines differ in several immune function parameters at the start. Because humoral immunity plays, a significant role in the management of infections, antibody titer used as one signal to assess the ability of immune responses (Lynne et al., 2006). By estimating the highest antibody titers transferred at 1-day-old chicks that favor prevention of ND disease and for deciding vaccination schedules in the upcoming days of chick life, we attempted to measure the level of maternal antibodies transferred from parents of these broiler strains to their progeny through yolk in order to take advantage of this ability. The research performed on Ross 308, Cobb 500 1-day-old chicks, concerned mainly the evaluation of the Newcastle maternal antibodies titers assessed by ELISA. Materials and MethodsSamplingThe study was carried out from April to October 2020 to assess the maternal derived antibody of broiler chickens. Five hundred and thirty-two thousand imported Cobb 500 (532,000) and Ross 308 (336,000) day-old broiler chicks were used for sampling. Seven hundred and seven in total, of these 451 blood samples were collected from imported 28 Ross 308 flocks (12–18 chicks/each), and 256 blood samples were collected from 17 Cobb 500 flocks (12–18 chicks/each). Jugular and brachial veins bled to collect about 1 ml of blood using a 3-ml disposable syringe, which then transferred to gel-containing tubes. For 3 hours, blood samples were allowed to coagulate in gel-containing tubes at room temperature, then centrifuged using Hettich Centrifuge (Germany)/5 minutes at 4,000 rpm for sera separation. Sera were placed into 1.5-ml tubes (Eppendorf®, Hamburg, Germany) and stored at −20°C in a freezer until processing. Indirect ELISA test (BioChek commercial kit, UK) (BioChek, 2019), used to estimate antibody levels as directed by the manufacturer. The serological analysis performed by private DNA laboratory for scientific researches/Mosul-IRAQ, using ELISA reader (biotek™ 800/TS) with a 405-nm filter, and washer (biotek™ 50/TS) (USA). Calculation of resultsFor the test result to be valid the mean negative control absorbance should read 0.349 or less and the difference between the mean negative control and the mean positive control should be greater than 0.15. Test sample values were relative to the controls. The relative amounts of antibodies in chicken samples then calculated by reference to the positive control. This relationship expressed as sample to positive ratio (S/P ratio). Interpretation of resultsSamples with an S/P of 0.35 or greater contain anti-NDV antibodies and considered positive. Calculation of S/P ratioMean of test sample − mean of negative control ---------------------------------------------------------------- Mean of positive control − mean of negative control Calculation of antibody titerThe following equation relates the S/P of a sample at a 1:500 dilutions to an end titer: Log10 titer=1.0 * Log (SP) + 3.52 Antilog=Titer S/P value Titer range Antibody status 0.349 or less 1,158 or less Negative 0.350 or greater 1,159 or greater Positive Vaccination index (VI), expresses the ratio between mean titer and CV percentage calculated to differentiate the titers estimated due to vaccination or infection. (Mean titer) 2 Mean titer VI=---------------=------------------- (St. dev. × 100) % CV Statistical analysisThe Statistical Package for the Social Sciences version 18.0 used to analyze the titers obtained by ELISA test. The significant differences in ELISA titers of chickens determined by one-way analysis of variance LSD test. Means compared at a significance level of 5%. Ethical approvalThe ethics of scientific research has been applied in dealing with animals and samples collecting. ResultsCobb 500 1-day-old broiler chicks had the highest mean ELISA Newcastle antibody titers, which were significantly (p < 0.05) higher than Ross 308 in immunological responses (Tables 1–3). Maternal antibody titers of imported Rose 308 1-day-old broiler chicks are presented in Tables 1 and 3. From tables, it is evident that there was a wide range in the maternal antibodies being from 5,045 to 10,865 among the locations studied, with a mean of 8,647.76. The uniformity of maternal antibody, expressed by CV percentage, was ranged from 7.14% to 24.36% with a mean of 15.18%. So all titers were within the excellent CV percentage limits (<40%). Out of the 451 sera sample tested for imported Ross 308 broiler chicks, 97.78% show positivity to ELISA test, and 24 flocks (85.71%) out of 28 flocks of Ross 308 tested show 100% maternal Antibodies positivity to ELISA test, while 4 flocks (14.29%) show positivity to ELISA test between 66.66% and 94.4%. The immunological response of broiler chicks to vaccination against NDV, expressed by VI ranged from 364.03 up to 1,477.35 with a mean of 821.60. Table 1. Serum samples from 1-day-old Ross 308 broilers (locations and farms), in Nineveh governorate tested and their mean ELISA ND maternal antibody titers.
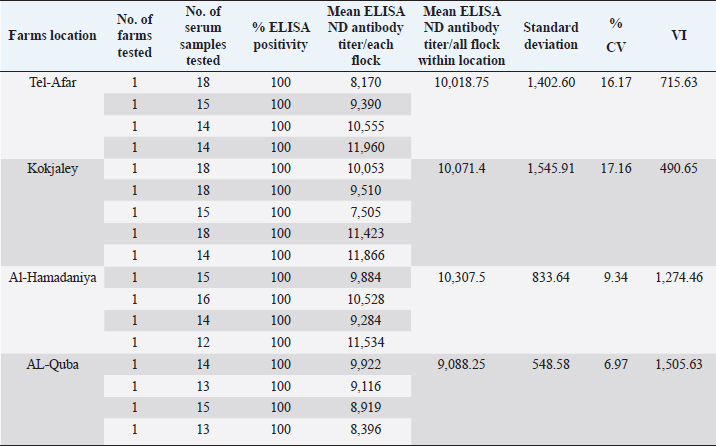
Table 2. Serum samples from 1-day-old Cobb broiler (locations and farms), in Nineveh governorate tested and their mean ELISA ND maternal antibody titer.
Table 3. Summary of the total number of imported 1-day-old Ross 308 and local Cobb 500 broiler chicks, serum samples, mean ELISA titers, mean CV percentage (CV%), and mean VI.
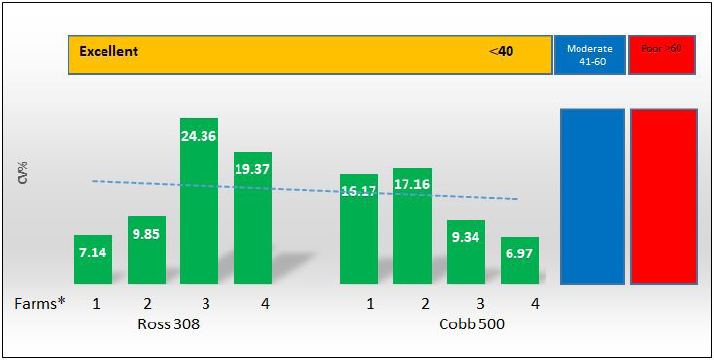
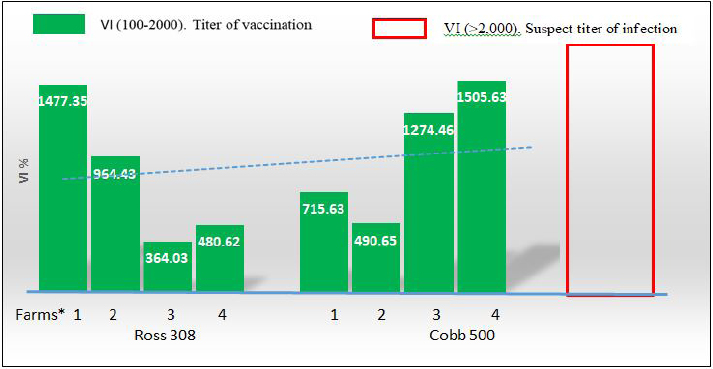
On the other hand, the results obtained from testing sera belonging to imported Cobb 500 broiler chicks were better than imported Ross 308 broiler which are shown in Tables 2 and 3. First of all the mean maternal antibodies ELISA titers, was ranged from 7,505 to 11,960, with a significant (p < 0.05) higher mean ELISA value up to 9,871.47. These relatively closest titers to each other in Cobb chicks were in the same time reflected by the better CV percentage in Cobb 500 chicks, being 12.41% versus that of 15.18% in Ross 308 chicks. Although both occurred within the excellent CV percentage (i.e., less than 40%), but at the same time were significantly differ (p < 0.05) from each other. Seropositivity of Cobb sera tested revealed that all of the chicks had maternal antibody titers (100% positivity). The VIs obtained in Cobb broiler chick flocks were significantly (p < 0.05) higher than those seen with Ross 308 chicks, and ranged from 490.65 to 1,505.63, with a mean of 966.59. A diagrammatic representation of the CV percentages and VI index as scores of vaccination for both strains, Ross 308 and Cobb 500, is presented in Figures 1 and 2. Figure 1 concerning CV percentage shows that Cobb 500 strain had better score of vaccination through their lower CV percentage compared with Ross 308 strain. Although Cobb 500 strain, had higher VI index when compared with Ross 308 strain, but both strains were within the vaccination titers’ limit (<2,000) (Fig. 2). DiscussionHens vaccinated by attenuated ND vaccine at early and late breeding stages resulted in activation of the mucosal immunity in the reproductive tract causing direct secretion of NDV antibodies into the eggs. Transfer of maternal antibody of NDV and other diseases is important for early chicken survival. A killed-type vaccine to NDV which is usually used in broiler breeders vaccination programs in both broiler types was successful in enhancing the immune response of high and uniform maternal antibody in one-day-old chicks. Genotypic structure of modern fast-growing chickens has been changed, particularly after decades of breeding for higher production (Emam et al., 2014), and antibody values could be affected by intrinsic and extrinsic factors related to the birds and their management in addition to the vaccination programs, since genetics significantly influences the immune system, so immunocompetence of livestock plays an important role in farm profitability because it directly affects health maintenance. In this trail, it is evident that there was a significant difference in the mean maternal antibody titers with both broiler types of imported Cobb 500 and Ross 308 but the diversity in these titers was less widely in Cobb 500 chicks in comparison to those of Ross 308 1-day-old chicks. This situation was expressed by a significant (p < 0.05) lower CV percentage in titers of Cobb chicks compared to percent in Ross 308. Moreover, all the tested Cobb 500 1-day-old chicks had maternal antibody titers, a situation not recorded with all the tested sera of Ross chicks. The results logically being moved to the favor of Cobb 500 chicks with a good vaccination response Tables 1–3. Mansour et al. (2016) supported this finding about the effect of genetic strains (Ross 308 and Cobb 500) on immune response against ND vaccine in broiler chickens.
Fig. 1. Coefficient correlation (CV %) of 1-day-old Ross 308 and Cobb 500 broiler chicks. (*) Farms: (1): Tel-Afar; (2): Kokjaly; (3): Al-Hamadaniya; (4): Al-Quba.
Fig. 2. VI of 1-day-old Ross 308 AND Cobb 500 broiler chicks. (*) Farms: (1): Tal-Afar; (2): Kokgaley; (3): Al-hamadania; (4): Al-quba. This is may be due to the difference in the strain genotypes of Ross breed in comparison with Cobb 500 breeds or due to massive selection to disease resistance breeding by the primary breeders, these genes are carried on major histocompatibility complex genes class IV region responsible for the difference in immune response between chicken breeds. This agrees with Lwelamira et al. (2009) who explain that the production of antibodies in the humoral immune response is controlled by many genes responsible for genetic resistance. Both hybrids Cobb 500 and Ross 308 are excellent to keep by following the housing and feeding requirements (Strakova et al., 2015). They achieve weights of 2 kg and above in 42 days, but the superior genetics of Cobb 500 give an extra edge to this hybrid breed, since, Cobb 500 has better average daily weight gains and growth performance (El-Tahawy et al., 2017; Pascalau et al., 2017) and better immune defense system to counter diseases than Ross 308 (Mayahi et al., 2016; Gholami et al., 2020). Immune responses are crucial for optimal health and affected by host genetics. Some genes in broiler chickens regulate disease resistance (Mansour et al., 2016). Monitoring of vaccination responses in poultry is the mean corner stone for the successful vaccination programs by estimating the mean titer of antibodies in a given flock and their measurement of uniformity using for example ELISA technology (Hamal et al., 2006; BioChek, 2012). Our current results further supported by the study of Cheema et al. (2003) who reported that Ross 308 was associated with lower serum IgA levels than those of Cobb 500. Although serum sampling from 1-day-old chicks applied here in both broiler types, was at the minimum rate of 12–18 chicks per flock (the optimal sample size is 23 samples/flock) (BioChek, 2012), but ELISA mean titers obtained here occurred within the reproducible and predictable ELISA titers of 1-day-old chicks (5,000–10,000), CV under 40% and VI less than 2,000 (BioChek, 2012; Cobb-Vantres, 2013). However, ELISA titer for NDs and other respiratory viral agents can be differentiated as a vaccination response or probable infection response by measuring the relationship between mean titer and its uniformity expressed by VI (Cardoso et al., 2006). The VI seems to be a logical parameter to compare the serological response between Cobb 500 and Ross 308 strains in their maternal antibody titer transmitted to their off-springs, as VI gives a high score with a good vaccination (high mean titer, low percentage CV) in Cobb 500 strain, and a lower score with a less titer after vaccination (low mean titer, high percentage CV) in Ross 308 strain. The effect of broiler strain on the immune response was also reported by Mayahi et al. (2016), who showed that Cobb 500 had the highest immune responses against ND vaccine than Ross 308, which was in the second order. In the diagrammatic representation (Fig. 1), there are three categories, excellent, medium, and poor CV percentages. Although all ELISA maternal antibody titers for both strains of broiler chicks occur within the excellent level (<40%), but there was lesser CV percentage and higher ELISA maternal antibody titers recorded to the favor Cobb 500 1-day-old chicks in comparison to Ross 308 chicks. In the same direction, VI index was within the suspected vaccination titer for both strains of broiler chicks (VI suspect of infection >2,000). The results of this study suggest that the variation observed in immune responses between these strains of chickens is most likely due to differences in the genetic background between each strain of chicken. In conclusion, it could be said that both broiler types had high and uniform maternal antibody titers. Although in general, more protective titer was found with Cobb 500 than those in Ross 308. However, on the basis of our results veterinarians may expect less sever field cases of ND (morbidity and mortality) in Cobb 500 broilers than Ross 308 chicks. This may have attributed to the difference in the strain genotypes of Cobb breed in comparison with Ross under the recurrent exposure to the local widely spread endemic Newcastle virus, in addition to the vaccine strains used in breeders’ vaccination and the schedule and the vaccination application method used. In the same direction, VI index was within the suspected vaccination titer for both strains of broiler chicks (VI suspect of infection >2,000) (BioChek), but VI in Cobb 500 1-day-old chicks give a high score with higher ELISA mean titer and low percentage CV in comparison to Ross 308 strain with lower score through their lower ELISA mean titer and higher percentage of CV (Fig. 2). AcknowledgmentsNone. Conflict of interestThe authors declare that they have no conflict of interest. ReferencesAldous, E.W., Mynn, J.K., Banks, J. and Alexander, D.J. 2003. A molecular epidemiological study of avian paramyxovirus type 1 (Newcastle disease virus) isolates by phylogenetic analysis of a partial nucleotide sequence of the fusion protein gene. Avian Pathol. 32(3), 239–257. Alexander, D.J. 1991. Newcastle disease and other paramyxovirus infection. In Disease of poultry, 9th edition. Eds., Calnek, B.W., Dames H.J., Beared C.W., Reid W.N. and Jorder Jr, H.W. Ames, IO: Iowa state University Press, pp: 496–519. BioChek. 2019. Smart veterinary diagnostic. NDV Antibody detection assay. Available via www.biochek.com/ringtrail. BioChek. 2012. Smart veterinary diagnostic. Interpretation and application of results manual. Immunology 1979 36 427 genetic analysis of antibody responsiveness to sheep erythrocytes in crosses between lines of mice selected for high or low antibody synthesis. Reeuwijk, Netherlands: BioChek. Cardoso, A., Albuquerque, R. and Tessari, E. 2006. Humoral immunological response in broilers vaccinated against Newcastle disease and supplemented with dietary zinc and vitamin E. Rev. Brasil. Cienc. Avic. 8(2), 99–103. Cheema, M.A., Qureshi, M.A. and Havenstein, G.B. 2003. A comparison of the immune response of a 2001 commercial broiler with a 1957 randombred broiler strain when fed representative 1957 and 2001 broiler diets. Poult. Sci. 82(10), 1519–1529. Cobb: vaccination procedure guide. Vaccination. Available via Cobb-Vantres.Com. L-1070-03 Daniela, B., Virgilia, P., Radulescu1, O. and Jenica, B. 2010. Serological data of avian infectious diseases. Lucrari Stiinlifice Med. Vet. 333(1), 187–196. Danisman, R. and Gous, R.M. 2011. Effect of dietary protein on the allometric relationships between some carcassportions and body protein in three broiler strains. S. Afr. J. Anim. Sci. 41(3), 195–208. El-Tahawy, A.S., Taha, A.E. and Adel, S.A. 2017. Effect of flock size on the productive and economic efficiency of Ross 308 and Cobb 500 broilers. Eur. Poult. Sci. 81, 1–10. Emam, M., Mehrabani-Yeganeh, H., Barjesteh, N., Nikbakht, G., Thompson-Crispi, K. and Charkhkar, S. 2014. The influence of genetic background versus commercial breeding programs on chicken immunocompetence. Poult Sci. 93(1), 77–84. Gholami, M., Chamani, M., Alireza Seidavi, A., Ali Asghar Sadeghi, A.A. and Mehdi Aminafschar, A. 2020. Effects of stocking density and environmental conditions on performance, immunity, carcase characteristics, blood constitutes, and economical parameters of Cobb 500 strain broiler chickens. Ital. J. Anim. Sci. 19(1), 524–535. Grindstaff, J.L., Brodie, E.D. and Ketterson, E.D. 2003. Immune function across generations: integrating mechanism and evolutionary process in maternal antibody transmission. Proc. Biol. Sci. 270(1531), 2309–2319. Hamal, K.R., Burgess, S.C., Pevzner, I.Y. and Erf, G.F. 2006. Maternal antibody transfer from dams to their egg yolks, egg whites, and chicks in meat lines of chickens. Poult. Sci. 85(8), 1364–1372. Kannaki, T.R., Priyanka, E. and Haunshi, S. 2019. Concanavalin A- sandwich-ELISA for the detection of antibodies against Newcastle disease virus (NDV) in chicken. Indian J. Anim. Res. 53(12), 1656–1660. Liljebjelke, K.A., King, D.J. and Kapczynski, D.R. 2008. Determination of minimum hemagglutinin units in an inactivated Newcastle disease virus vaccine for clinical protection of chickens from exotic Newcastle disease virus challenge. Avian Dis. 52(2), 260–268. Lwelamira, J., Kifaro, G.C. and Gwakisa, P.S. 2009. Genetic parameters for body weights, egg traits and antibody response against Newcastle disease virus (NDV) vaccine among two Tanzania chicken ecotypes. Trop. Anim. Health Prod. 41, 51–59. Lynne, A.M., Foley, S.L., Nolan, L.K., Foley, A.S.L. and Nolanac, L.K. 2006. Immune response to recombinant Escherichia coli Iss protein in poultry. Avian Dis. 50(2), 273–276. Mansour, M., Forough, T. and Mahya, A. 2016. Effect of genetic strains (Ross 308, Cobb 500 and Hubbard F15) on immune response against Newcastle disease vaccine in broiler chickens. Int. J. Enteric. Pathog. 4(4), 1–3. Mayahi, M., Talazadeh, F. and Abdolshah, M. 2016. Effect of genetic strains (Ross 308, Cobb 500 and Hubbard F15) on immune response against Newcastle disease vaccine in broiler chickens. Int. J. Enteric. Pathog. 4(4), 37–39. OIE. 2012. Terrestrial manual. Newcastle disease chapter 2.3.14.2012. Paris, France: OIE. Pascalau, S., Cadar, M., Răducu, C. and Marchis, Z. 2017. Evaluation of productive performances in Ross 308 and Cobb 500 hybrids. ABAH Bioflux, 9(1), 22–27. Saif, Y.M. and Fadly, A.M. 2008. Diseases of poultry. Ames, IO: Iowa State University Press, Blackwell Publishing. Available via https://www.academia.edu/38368719/Diseases_of_Poultry_Y_M_saif_pdf Shareef, A.M. and Gargees, M.T. 2008. Mycofix ameliorative effect on Newcastle disease antibody production in broiler chickens during aflatoxicosis. Iraqi J. Vet. Sci. 22(1), 29–34. Strakova, E., Suchy, E., Navratil, P., Karel, T. and Herzig, I. 2015. Comparison of the content of crude protein and amino acids in the whole bodies of cocks and hens of Ross 308 and Cobb 500 hybrids at the end of fattening. Czech J. Anim. Sci. 60(2), 67–74. Suarez, D.L., Miller, P.J., Koch, G., Mundt, E. and Rautenschlein, S. 2019. Newcastle disease, other avian paramyxoviruses, and avian metapneumovirus infections. Dis. Poult. 22(1), 66–109. Turblin, V. 2009. The benefits of Newcastle killed vaccines in broilers. Petaling Jaya, Malaysia: CEVA Animal Health Asia Pacific-Selangor, no. 24. Yunis, R., Ben-David, A., Heller, E.D. and Cahaner, A. 2000. Immunocompetence and viability under commercial conditions of broiler group differing in growth rate and in antibody response to Escherichia coli vaccine. Poult. Sci. 79, 810–816. | ||
| How to Cite this Article |
| Pubmed Style Ezzulddin TA, Jwher DMT, Shareef AM. The ability to resist Newcastle disease through inherited immunity in different strains of broilers in Nineveh governorate. Open Vet J. 2022; 12(6): 936-943. doi:10.5455/OVJ.2022.v12.i6.20 Web Style Ezzulddin TA, Jwher DMT, Shareef AM. The ability to resist Newcastle disease through inherited immunity in different strains of broilers in Nineveh governorate. https://www.openveterinaryjournal.com/?mno=108005 [Access: July 05, 2025]. doi:10.5455/OVJ.2022.v12.i6.20 AMA (American Medical Association) Style Ezzulddin TA, Jwher DMT, Shareef AM. The ability to resist Newcastle disease through inherited immunity in different strains of broilers in Nineveh governorate. Open Vet J. 2022; 12(6): 936-943. doi:10.5455/OVJ.2022.v12.i6.20 Vancouver/ICMJE Style Ezzulddin TA, Jwher DMT, Shareef AM. The ability to resist Newcastle disease through inherited immunity in different strains of broilers in Nineveh governorate. Open Vet J. (2022), [cited July 05, 2025]; 12(6): 936-943. doi:10.5455/OVJ.2022.v12.i6.20 Harvard Style Ezzulddin, T. A., Jwher, . D. M. T. & Shareef, . A. M. (2022) The ability to resist Newcastle disease through inherited immunity in different strains of broilers in Nineveh governorate. Open Vet J, 12 (6), 936-943. doi:10.5455/OVJ.2022.v12.i6.20 Turabian Style Ezzulddin, Thamer Abdulazeez, Dhyaa Mohammad Taher Jwher, and Akeel Mohammad Shareef. 2022. The ability to resist Newcastle disease through inherited immunity in different strains of broilers in Nineveh governorate. Open Veterinary Journal, 12 (6), 936-943. doi:10.5455/OVJ.2022.v12.i6.20 Chicago Style Ezzulddin, Thamer Abdulazeez, Dhyaa Mohammad Taher Jwher, and Akeel Mohammad Shareef. "The ability to resist Newcastle disease through inherited immunity in different strains of broilers in Nineveh governorate." Open Veterinary Journal 12 (2022), 936-943. doi:10.5455/OVJ.2022.v12.i6.20 MLA (The Modern Language Association) Style Ezzulddin, Thamer Abdulazeez, Dhyaa Mohammad Taher Jwher, and Akeel Mohammad Shareef. "The ability to resist Newcastle disease through inherited immunity in different strains of broilers in Nineveh governorate." Open Veterinary Journal 12.6 (2022), 936-943. Print. doi:10.5455/OVJ.2022.v12.i6.20 APA (American Psychological Association) Style Ezzulddin, T. A., Jwher, . D. M. T. & Shareef, . A. M. (2022) The ability to resist Newcastle disease through inherited immunity in different strains of broilers in Nineveh governorate. Open Veterinary Journal, 12 (6), 936-943. doi:10.5455/OVJ.2022.v12.i6.20 |