| Case Report | ||
Open Vet J. 2022; 12(5): 622-627 Open Veterinary Journal, (2022), Vol. 12(5): 622–627 Case Report Thymic squamous cell carcinoma in a catRichard Chung Yi Looi1*, Johanna Todd2, Veronika Langova2, David Taylor3 and Amanda Miller11Surgery Department, Small Animal Specialist Hospital, North Ryde, Australia 2Medical Oncology Department, Small Animal Specialist Hospital, North Ryde, Australia 3Vetnostics, Laverty Pathology, Macquarie Park, Australia *Corresponding Author: Richard Chung Yi Looi. Surgery Department, Small Animal Specialist Hospital, North Ryde, Australia. Email: rlooi [at] sashvets.com Submitted: 10/05/2022 Accepted: 10/08/2022 Published: 07/09/2022 © 2022 Open Veterinary Journal
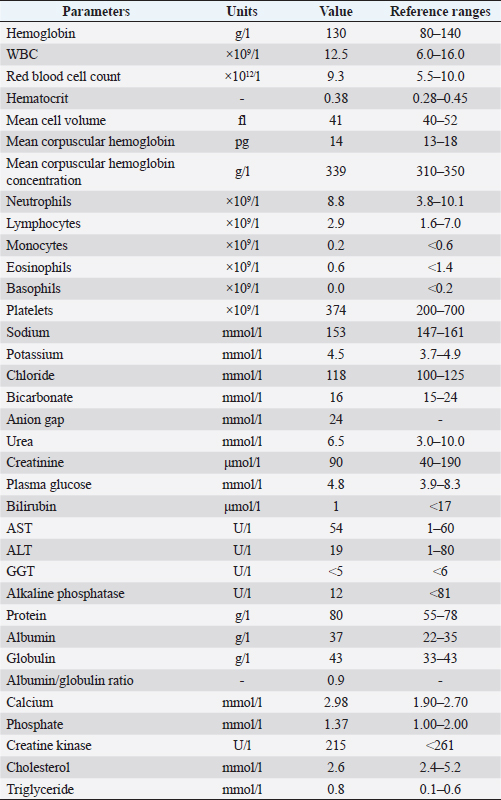
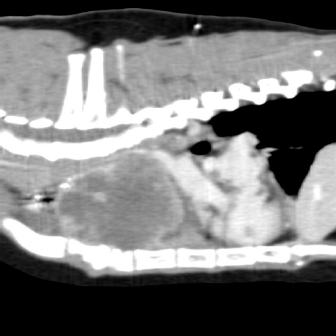
AbstractBackground: The most common cranial mediastinal masses affecting dogs and cats include lymphoma and thymic epithelial tumors. In this report, a cat with a cranial mediastinal mass was diagnosed with a thymic carcinoma subtype squamous cell carcinoma, which has rarely been reported in cats. Management of this subtype with a combination of surgery and chemotherapy has so far not been reported. This case report describes the treatment with surgical management followed by intracavitary carboplatin. Case Description: A 12-year-old male neutered domestic short hair cat was referred for lethargy, hyporexia, and weight loss, and was diagnosed with a cranial mediastinal mass radiographically. Initial cytology through fine needle aspirates was suggestive of carcinoma. Computed tomography was performed which did not show evidence of other primary tumor sources or metastases, and hence surgical resection was recommended. Intraoperative findings revealed local invasion of the surrounding tissues, including major vasculature and nerves, although histopathological assessment showed no local lymph node involvement. Intracavitary carboplatin chemotherapy was administered 2 weeks postsurgery. The patient was humanely euthanized 4 weeks postsurgery due to evidence of local recurrence causing significant respiratory compromise. Conclusion: A combination of surgical excision as well as intracavitary carboplatin does not seem to be effective for the treatment of this thymic carcinoma subtype, with evidence of early recurrence and return of clinical signs. Keywords: Thymic carcinoma, Squamous cell carcinoma, Mediastinal mass, Carboplatin. IntroductionThe most common cranial mediastinal masses affecting both dogs and cats include lymphoma and thymic epithelial tumors (TETs), with other differentials being relatively rare (Withrow, 2013). The term TETs has been used to outline a number of histologic subtypes of thymoma in humans, and has been recently been described in dogs (Burgess et al., 2016; Oura et al., 2019). TETs are currently classified according to the World Health Organization’s histopathological classification which differentiates thymomas from thymic carcinomas, with thymic carcinomas differentiated into subtypes (Marx et al., 2022). In the present case, the cat had been diagnosed with thymic carcinoma with a subtype of squamous cell carcinoma which has rarely been reported in cats (Carpenter and Valentine, 1992; Anilkumar et al., 1994). Of the only reported case where surgical intervention was pursued, persistent pleural effusion after surgical resection was a feature, which eventually led to humane euthanasia of the patient (Carpenter and Valentine, 1992). Chemotherapy has not been reported as a management for this subtype of thymic carcinoma. To our knowledge, this is the first case describing surgical management with postoperative usage of intracavitary carboplatin. Case DetailsA 12-year-old male neutered domestic short hair cat weighing 5.4 kg presented to the Small Animal Specialist Hospital (SASH, North Ryde) oncology department for further workup of a cranial mediastinal mass diagnosed radiographically by the referring veterinarian. Initial presenting complaints were recent hyporexia, lethargy, and weight loss. Complete blood count, serum biochemistry, and urinalysis were performed. Hyperproteinemia 80 g/l [reference interval (RI) 55–78 g/l], hyperalbuminemia 37 g/l (RI 22–35 g/l), hypertriglyceridemia 0.8 mmol/l (RI 0.1–0.6 mmol/l), and hypercalcemia 1.60 mmol/l (RI 1.10–1.40, ionized calcium) were noted on the blood work (Table 1). Urinalysis showed mild proteinuria and hematuria, which was considered to be clinically insignificant on a cystocentesis sample. The mass was diagnosed via three-view thoracic radiographs and sampled via fine need aspiration guided by ultrasound. Cytological findings were suggestive of carcinoma. On physical examination, the cat had an unkept coat, reduced respiratory sounds cranially, and persistent stridor, but was otherwise bright and ambulatory. Computed tomography (CT) was performed using a Phillips Brilliance six slice CT. Multiphase contrast-enhanced CT images of the head, thorax, and abdomen were acquired with the cat in sternal recumbency (Figs. 1 and 2). A cranial mediastinal mass measuring 60 × 40 mm causing dorsal tracheal displacement toward the right was identified. With contrast administration, there was moderate enhancement of the periphery (+60 Hu) and poor central enhancement (+10 Hu). There was moderate to marked compression of the cranial vena cava, with a focal intraluminal contrast-filling defect at the cranial aspect of the vena cava just caudal to the mass. Here, the path of the cranial vena cava could not be completely traced, suggesting almost complete compression or invasion. There was no evidence of subcutaneous edema of the head or cervical region. Moderate volume of pleural fluid was bilaterally present. Multifocal, slightly branching soft tissue conglomerates largely following the bronchi of the caudal lobes toward the apex was noted, most likely to be bronchial disease with bronchiolar consolidation. Pulmonary metastatic neoplasia was considered to be unlikely. Surgical resection in the form of debulking was recommended on the basis that the large cranial mediastinal mass was most likely the cause of the cat’s clinical signs, and that reduction of gross disease should improve survival time as well as being more responsive to adjunctive treatment with chemotherapy. The cat was then started on mirtazapine (1.875 mg orally every 48 hours) and prednisolone (5 mg orally every 24 hours). Table 1. Complete blood count and biochemistry.
Fig. 1. Computerized tomography of the cranial mediastinal mass (sagittal plane).
Fig. 2. Computerized tomography of the cranial mediastinal mass (coronal plane). Prior to surgery, the cat was premedicated with 1.5 mg methadone, 30 mg ketamine, and 1.5 mg midazolam intramuscularly. Anesthesia was induced with alfaxalone intravenously to effect and the cat intubated with a 4-mm cuffed endotracheal tube. Perioperative antibiotics in the form of 150 mg cephazolin were administered intravenously. Analgesia was provided by continuous rate infusion of fentanyl (5 ug/kg/hour) and ketamine (0.6 mg/kg/hour) and also received three 2 ug/kg fentanyl boluses. Anesthetic complications encountered included mild hypotension, hypothermia, and tachycardia. Intraoperatively the cat was mechanically ventilated with intermittent positive pressure ventilation. The cranial mediastinal mass was approached through a cranial median sternotomy extending caudally from the manubrium. The xiphoid was left intact. Upon entry, there was a moderate amount of serosanguinous fluid. The mass was positioned immediately cranial to the heart, incorporating the cranial vena cava, brachiocephalic trunk, and branches, including the common carotid arteries, as well as both phrenic nerves. These structures were preserved during debulking of the mass via blunt and sharp dissections, leaving approximately 10% of the tumor volume in situ around the cranial vena cava at the level of the brachiocephalic branches due to the significant risk of fatal hemorrhage with further dissection. A representative mediastinal lymph node was extirpated and was submitted for histopathological analysis alongside the cranial mediastinal mass. Hemostasis was achieved through a combination of a bipolar vessel sealing device (Caiman, Aesculap), gelatin hemostatic agent (Gelita, B. Braun), and bipolar electrocautery (Bovie, Symmetry Surgical). Prior to closure, a 10 Fr thoracic drain (Argyle, Cardinal Health) was placed through the left thoracic wall at the seventh intercostal space and secured to the skin with a Chinese finger trap suture pattern of 2–0 polypropylene. The cat recovered uneventfully from anesthesia and postoperatively it received a constant rate infusion of fentanyl (2–5 ug/kg/hour) and ketamine (0.2–0.5 mg/kg/hour), and fluid therapy in the form of Hartmann’s intravenously. The cat was initially supported in an oxygen chamber with an oxygen level starting at 60%, which was gradually reduced and successfully discontinued 14 hours later. The fentanyl and ketamine were also weaned the next day and the cat was started on buprenorphine (0.1 mg intravenously every 8 hours) for the initial 24 hours, followed by transmucosal administration for 4 days. Meloxicam (0.25 mg orally every 24 hours) for 7 days was commenced once the cat was eating well. The thoracic drain was nonproductive within 12 hours of surgery and was removed without complications. Three days postoperatively, the cat was discharged for home care.
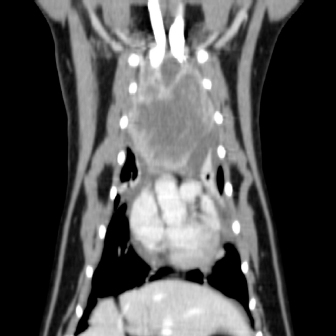
Fig. 3. Histopathological image of the biopsy from the cranial mediastinal mass showing neoplasia replacing normal thymic architecture. Necrotic areas not seen in this image. Histopathological report on the cranial mediastinal mass established the diagnosis of squamous cell carcinoma. The neoplasm extensively replaced normal architecture (Fig. 3) and was associated with broad zones of necrosis. The squamous cells had pleomorphic features, including variation in cell and nuclear size and shape, as well as elevated mitotic activity. The mediastinal lymph node had reactive lymphoid elements without evidence of metastatic neoplasia. Two days after discharge, the cat re-presented to the emergency department due to disinterest in food and dyspnea. Point of care ultrasound (POCUS) revealed a large volume of pleural effusion bilaterally, as well as scant pericardial effusion. Thoracocentesis of the left hemithorax yielded 250 ml of milky effusion. The effusate was assumed to be chylous in nature due to the gross appearance, additional testing of the fluid was declined due to financial constraints. After thoracocentesis, the cat’s respiratory rate and respiratory effort improved dramatically and no oxygen supplementation was required. The cat began eating voluntarily with a good appetite. Repeat POCUS was performed the next day, revealing a minimal volume of pleural effusion and unchanged volume of the pericardial effusion. Echocardiographic examination was indicative of restrictive filling in the diastolic phase, suggestive of pericarditis. The cat was discharged later that day. Two weeks postoperatively, the cat presented to the oncology department for commencement of chemotherapy. The client reported normal energy levels, consistently good appetite, and no evidence of respiratory compromise. Clinically relevant findings on complete blood count included an anemia without reticulocytosis of PCV 24% (RI 26%–46%) and reticulocyte count 4.8 K/ul (RI 2.0–50.0 K/ul), with normal white blood cell count (WBC) 4.81 × 109/l (RI 2.87–17.02 × 109/l), and normal neutrophil count 3.36 × 109/l (RI 2.30–10.29 × 109/l). Partial serum biochemistry abnormalities included decreased creatinine 39 umol/l (RI 71–212 umol/l), decreased blood urea nitrogen 5.4 mmol/l (RI 5.7–12.9 mmol/l), and decreased alanine aminotransferase <10 U/l (RI 12–130 U/l). Under sedation, POCUS revealed mild pleural effusion bilaterally and subsequent thoracocentesis yielded 60 ml of serous effusion. Intracavitary chemotherapy was administered with carboplatin 62 mg. Approximately 2 weeks after initiation of chemotherapy, the cat represented to the SASH emergency department for dyspnea. POCUS revealed a moderate volume of pleural fluid in both hemithoraxes, no pericardial effusion, and mass-like lesions in the cranial and caudal portion of the thorax. This was suspected to be pleural effusion due to carcinomatosis. Due to the cat’s clinical deterioration and unknown future prognosis, the owners elected for humane euthanasia. Full necropsy and further sampling and testing of the new lesions in the chest were not performed. DiscussionThe incidence of TETs in humans is rare with a reported rate of 2.2/1,000,000 for thymomas, and only 0.3/1,000,000 for thymic carcinomas, both associated with paraneoplastic syndromes including myasthenia gravis and other immune-mediated syndromes in up to 45% of cases (de Jong et al., 2008; Venuta et al., 2010). Myasthenia gravis and megaesophagus have been reported in up to 40% of dogs with thymoma as well as in cats, but was not seen in this case (Zitz et al., 2008; Robat et al., 2013). TETs are staged according to the Masaoka–Koga staging system, where local invasiveness is taken into consideration and the invasiveness correlates well with overall survival (Detterbeck et al., 2012; Girard et al., 2015). Recurrence rates are high in humans, ranging from 25% to 76% depending on the stage (Girard et al., 2015). In dogs, recurrence rates have been reported to be around 25%–40% (Martano et al., 2021; Yale et al., 2022). Due to the presence of macroscopic invasion into cranial vena cava on CT imaging and evidence of tumor infiltration into vascular connective tissue on histopathology, this cat would be stage III based on the Masaoka–Koga staging system (Detterbeck et al., 2012). A multi-institutional study showed that the median overall survival for human patients with thymic carcinoma was 6.6 years, with a recurrence rate of 35% at 5 years (Ahmad et al., 2015). In dogs with thymic carcinoma, the median survival time was 69 days (Burgess et al., 2015). In a reported case of a dog with metastatic thymic carcinoma, the dog was started on toceranib phosphate at initial diagnosis, with stable disease at restaging 6 months later; however, it died at 12 months postdiagnosis and treatment where local disease progression was noted (O’Connell et al., 2018). Postoperative chemotherapy as well as radiotherapy is recommended in cases of incompletely resected thymic carcinomas in humans (Girard et al., 2015). The use of tyrosine kinase inhibitors has been described in the human literature (Pagano et al., 2014; Remon et al., 2016), and in a single canine case (O’Connell et al., 2018). The first-line treatment for humans for nonresectable or recurrent disease is multiagent combinations containing platinum-based therapies such as cisplatin, doxorubicin and cyclophosphamide, or carboplatin and paclitaxel (Girard et al., 2015; Marx et al., 2015). Response rates to chemotherapeutic regimens including platinum derivatives were noted to be above 50% in the human literature (Berghmans et al., 2018); however, these regimens in addition to surgical excision as a treatment option for thymic carcinomas have not been evaluated in the veterinary literature. To our knowledge, this is the first reported case of a feline thymic carcinoma subtype squamous cell carcinoma treated initially with surgical management followed by adjuvant intracavitary carboplatin. Intracavitary carboplatin has been described in the veterinary literature for the treatment of both pleural and peritoneal carcinomatosis, sarcomatosis, or mesothelioma (Spugnini et al., 2008; Floch et al., 2020). Two out of three dogs with carcinomatosis of unknown origin showed complete resolution of effusion and no measurable tumor for at least 255 days (Moore et al., 1991). In the current case, intracavitary carboplatin did not seem effective in managing the neoplastic pleural effusion as clinical signs recurred in the cat 17 days postadministration. This echoes the findings of Floch et al. (2020) where 87.5% of cats with pleural carcinomatosis had recurrence of effusion within 2 weeks, and all cats died or were euthanized within 16 days. ConclusionsFeline thymic carcinomas are rare and there is limited literature describing their behavior, clinical course, and histopathologic characteristics. Diagnosis of thymic carcinoma relays a poor prognosis in dogs, and likely can be extrapolated similarly in cats. Intracavitary carboplatin seemed ineffective at treating pleural effusion caused by neoplastic causes in this case. Further cases would be needed to more accurately characterize outcomes as well as the usefulness of adjuvant carboplatin or other platinum-based chemotherapy in this disease. AcknowledgmentsThe authors thank Dr Richard Lam from the SASH Radiology Department for aiding with the diagnostics for this case and providing the necessary images for the publication. Conflict of interestThe authors declared no potential conflicts of interests with respect to the research, authorship, and/or publication of this article. Authors’ contributionsAll authors conceived and carried out the procedures; drafted and revised the manuscript; and approved the final version of the manuscript. ReferencesAhmad, U., Yao, X., Detterbeck, F., Huang, J., Antonicelli, A., Filosso, P.L., Ruffini, E., Travis, W., Jones, D.R., Zhan, Y., Lucchi, M. and Rimner, A. 2015. Thymic carcinoma outcomes and prognosis: results of an international analysis. J. Thorac. Cardiovasc. Surg. 149(1), 95–100. Anilkumar, T.V., Voigt, R.P., Quigley, P.J., Krausz, T., Sarraf, C.E. and Alison, M.R. 1994. Squamous cell carcinoma of the feline thymus with widespread apoptosis. Res. Vet. Sci. 56(2), 208–215. Berghmans, T., Durieux, V., Holbrechts, S., Jungels, C., Lafitte, J.J., Meert, A.P., Moretti, L., Ocak, S., Roelandts, M. and Girard, N. 2018. Systemic treatments for thymoma and thymic carcinoma: a systematic review. Lung. Cancer. 126, 25–31. Burgess, K.E., DeRegis, C.J., Brown, F.S. and Keating, J.H. 2016. Histologic and immunohistochemical characterization of thymic epithelial tumours in the dog. Vet. Comp. Oncol. 14(2), 113–121. Carpenter, J.L. and Valentine, B.A. 1992. Squamous cell carcinoma arising in two feline thymomas. Vet. Pathol. 29(6), 541–543. de Jong, W.K., Blaauwgeers, J.L.G., Schaapveld, M., Timens, W., Klinkenberg, T.J. and Groen, H.J. 2008. Thymic epithelial tumours: a population-based study of the incidence, diagnostic procedures and therapy. Eur. J. Cancer. 44(1), 123–130. Detterbeck, F.C., Nicholson, A.G., Kondo, K., Van Schil, P. and Moran, C. 2011. The Masaoka-Koga stage classification for thymic malignancies: clarification and definition of terms. J. Thorac. Oncol. 6(7 Suppl. 3), S1710–1716. Floch, F., Boissy, L., Lanore, D., Sayag, D. and Serres, F. 2020. Evaluation of intracavitary carboplatin chemotherapy for treatment of pleural carcinomatosis in cats: a retrospective study of eight cases. J. Feline. Med. Surg. 22(2), 84–90. Girard, N., Ruffini, E., Marx, A., Faivre-Finn, C., Peters, S. and Committee, E.G. 2015. Thymic epithelial tumours: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 26(Suppl. 5), 40–55. Martano, M., Buracco, P. and Morello, E.M. 2021. Canine epithelial thymic tumors: outcome in 28 dogs treated by surgery. Animals 11(12), 3444. Marx, A., Chan, J.K.C., Chalabreysse, L., Dacic, S., Detterbeck, F., French, C.A., Hornick, J.L., Inagaki, H., Jain, D., Lazar, A.J., Marino, M., Marom, E.M., Moreira, A.L., Nicholson, A.G., Noguchi, M., Nonaka, D., Papotti, M.G., Porubsky, S., Sholl, L.M., Tateyama, H., Thomas de Montpreville, V., Travis, W.D., Rajan, A., Roden, A.C. and Strobel, P. 2022. The 2021 WHO classification of tumors of the thymus and mediastinum: what is new in thymic epithelial, germ cell, and mesenchymal tumors? J. Thorac. Oncol. 17(2), 200–213. Moore, A.S., Kirk, C. and Cardona, A. 1991. Intracavitary cisplatin chemotherapy experience with six dogs. J. Vet. Intern. Med. 5(4), 227–231. O'Connell, E., Harper, A., Blundell, R. and Batchelor, D. 2018. Paraneoplastic immune mediated neutropenia in a dog following thymoma excision with later development of metastatic thymic carcinoma treated with toceranib phosphate. Vet. Rec. 6(3), e000548. Oura, T.J., Hamel, P.E., Jennings, S.H., Bain, P.J., Jennings, D.E. and Berg, J. 2019. Radiographic differentiation of cranial mediastinal lymphomas from thymic epithelial tumors in dogs and cats. J. Am. Anim. Hosp. Assoc. 55(4), 187–193. Pagano, M., Sierra, N.M.A., Panebianco, M., Rossi, G., Gnoni, R., Bisagni, G. and Boni, C. 2014. Sorafenib efficacy in thymic carcinomas seems not to require c-KIT or PDGFR-alpha mutations. Anticancer. Res. 34(9), 5105–5110. Remon, J., Girard, N., Mazieres, J., Dansin, E., Pichon, E., Greillier, L., Dubos, C., Lindsay, C.R. and Besse, B. 2016. Sunitinib in patients with advanced thymic malignancies: cohort from the French RYTHMIC network. Lung. Cancer. 97, 99–104. Robat, C.S., Cesario, L., Gaeta, R., Miller, M., Schrempp, D. and Chun, R. 2013. Clinical features, treatment options, and outcome in dogs with thymoma: 116 cases (1999–2010). J. Am. Vet. Med. Assoc. 243(10), 1448–1454. Spugnini, E.P., Crispi, S., Scarabello, A., Caruso, G., Citro, G. and Baldi, A. 2008. Piroxicam and intracavitary platinum-based chemotherapy for the treatment of advanced mesothelioma in pets: preliminary observations. J. Exp. Clin. Cancer. Res. 27, 6. Venuta, F., Anile, M., Diso, D., Vitolo, D., Rendina, E.A., De Giacomo, T., Francioni, F. and Coloni, G.F. 2010. Thymoma and thymic carcinoma. Eur. J. Cardiothorac. Surg. 37(1), 13–25. Withrow, S.J. 2013. Withrow and MacEwen's small animal clinical oncology. Saunders. Yale, A.D., Priestnall, S.L., Pittaway, R. and Taylor, A.J. 2022. Thymic epithelial tumours in 51 dogs: histopathologic and clinicopathologic findings. Vet. Comp. Oncol. 20(1), 50–58. Zitz, J.C., Birchard, S.J., Couto, G.C., Samii, V.F., Weisbrode, S.E. and Young, G.S. 2008. Results of excision of thymoma in cats and dogs: 20 cases (1984–2005). J. Am. Vet. Med. Assoc. 232(8), 1186–1192. | ||
| How to Cite this Article |
| Pubmed Style Richard Chung Yi Looi|. Thymic squamous cell carcinoma in a cat.. Open Vet J. 2022; 12(5): 622-627. doi:10.5455/OVJ.2022.v12.i5.5 Web Style Richard Chung Yi Looi|. Thymic squamous cell carcinoma in a cat.. https://www.openveterinaryjournal.com/?mno=30591 [Access: July 03, 2025]. doi:10.5455/OVJ.2022.v12.i5.5 AMA (American Medical Association) Style Richard Chung Yi Looi|. Thymic squamous cell carcinoma in a cat.. Open Vet J. 2022; 12(5): 622-627. doi:10.5455/OVJ.2022.v12.i5.5 Vancouver/ICMJE Style Richard Chung Yi Looi|. Thymic squamous cell carcinoma in a cat.. Open Vet J. (2022), [cited July 03, 2025]; 12(5): 622-627. doi:10.5455/OVJ.2022.v12.i5.5 Harvard Style Richard Chung Yi Looi| (2022) Thymic squamous cell carcinoma in a cat.. Open Vet J, 12 (5), 622-627. doi:10.5455/OVJ.2022.v12.i5.5 Turabian Style Richard Chung Yi Looi|. 2022. Thymic squamous cell carcinoma in a cat.. Open Veterinary Journal, 12 (5), 622-627. doi:10.5455/OVJ.2022.v12.i5.5 Chicago Style Richard Chung Yi Looi|. "Thymic squamous cell carcinoma in a cat.." Open Veterinary Journal 12 (2022), 622-627. doi:10.5455/OVJ.2022.v12.i5.5 MLA (The Modern Language Association) Style Richard Chung Yi Looi|. "Thymic squamous cell carcinoma in a cat.." Open Veterinary Journal 12.5 (2022), 622-627. Print. doi:10.5455/OVJ.2022.v12.i5.5 APA (American Psychological Association) Style Richard Chung Yi Looi| (2022) Thymic squamous cell carcinoma in a cat.. Open Veterinary Journal, 12 (5), 622-627. doi:10.5455/OVJ.2022.v12.i5.5 |