| Original Article | ||
Open Vet J. 2022; 12(3): 407-413 Open Veterinary Journal, (2022), Vol. 12(3): 407–413 Original Research Adjustment of multi-leaf collimator parameters in 4-MV and 6-MV IMRT: A study of veterinary clinical casesToshie Iseri1, Yoshinori Tanabe2*, Hiro Horikirizono1, Hiroshi Sunahara1, Harumichi Itoh1, Yuki Nemoto1, Kazuhito Itamoto1, Kenji Tani1, Hidekazu Tanaka3 and Munekazu Nakaichi11Joint faculty of Veterinary Medicine, Yamaguchi University, Yamaguchi-shi, Japan 2Faculty of Medicine, Graduate School of Health Sciences, Okayama University, Okayama, Japan 3Faculty of Medicine and Health Sciences, Yamaguchi University, 1-1-1 Minami-Kogushi, Ube-shi, Japan *Corresponding Author: Yoshinori Tanabe. Faculty of Medicine, Graduate School of Health Sciences, Okayama University, 5-1 Shikata-cho, 2-chome, Kita-ku, Okayama, 700-8558, Japan. Email: tanabey [at] okayama-u.ac.jp Submitted: 20/04/2022 Accepted: 25/05/2022 Published: 21/06/2022 © 2022 Open Veterinary Journal
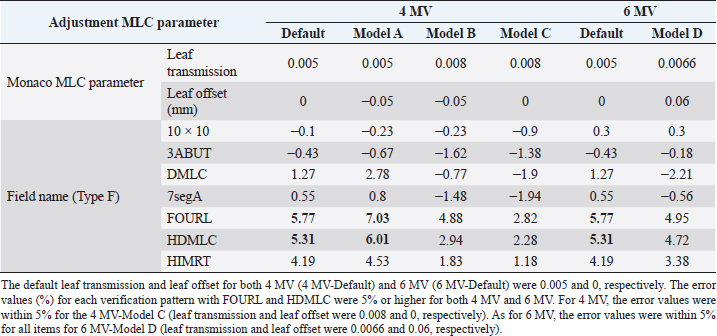
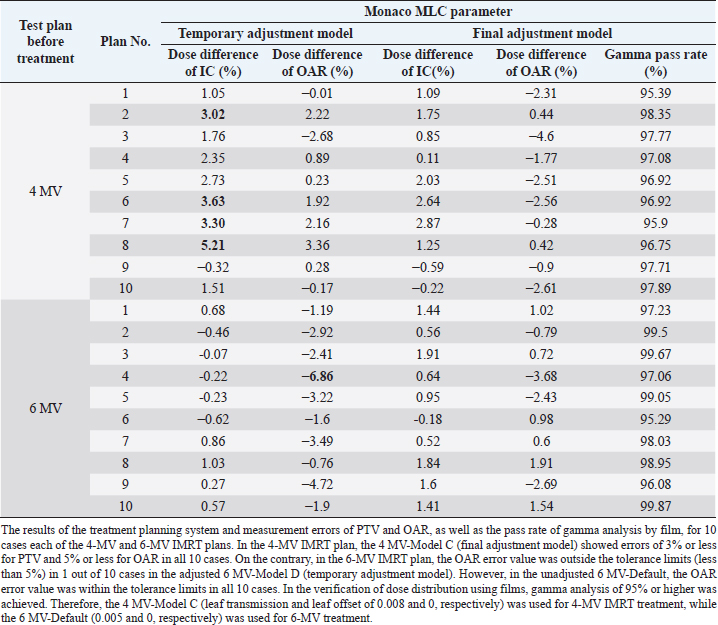
AbstractBackground: For optimal treatment, it is important to maintain optimal multi-leaf collimator (MLC) transmission in intensity-modulated radiation therapy (IMRT). However, adjustment of transmissions has not been reported in veterinary medicine. Aim: To demonstrate that appropriate MLC parameter adjustment for IMRT using 4- and 6-MV energy can reduce the need for quality assurance revalidation in real companion animal clinical cases. Methods: The MLC parameters (leaf transmission and leaf offset) of the treatment planning system were adjusted by evaluating seven plans (10 × 10 cm, 3ABUT, DMLC, 7segA, FOURL, HDMLC, and HIMRT) and 20 preclinical cases (10 cases each in 4- and 6-MV groups). Subsequently, 101 IMRT plans of 88 cases (77 dogs and 11 cats) were evaluated for absolute dose of plan target volume (PTV) and organs at risk (OAR) and were analyzed for the relative dose distribution by gamma analysis (3%/3 mm, >10%) using EBT3 film. Results: After adjustment of the MLC parameters (leaf transmission and leaf offset, 4 MV: 0.008 and 0, 6 MV: 0.005 and 0, respectively), the data from 101 plans (4 MV: 64 plans and 6 MV: 37 plans) treated with IMRT showed PTV <3%, OAR <5%, and gamma analysis pass rates ≥95% in all cases. Conclusion: Clinically meaningful dose distributions can be created even with a limited validation device if the treatment parameters are adjusted appropriately, even for tumors in canines and felines, where the irradiation field is small, the target is adjacent to the OAR, and the target is often superficial. Keywords: IMRT, Linac, MLC offset, MLC transmission, Radiation therapy. IntroductionIn veterinary medicine, as in human medicine, the number of tumor diseases is increasing due to the aging of companion animals, and the need for radiotherapy, surgery, and chemotherapy in companion animals is increasing. Radiotherapy is less invasive and causes less tissue damage than surgery and is used alone or in combination with surgery or chemotherapy. In small animal clinical practice, intensity-modulated radiation therapy (IMRT) can be used to increase therapeutic efficacy while decreasing radiation damage to normal tissues by focusing the dose on tumor tissue and decreasing the dose to critical organs (organs at risk, OAR) such as the eyes and brain. Commissioning of treatment planning systems is an important medical physics task that affects the accuracy of all plans created using the commissioned beam model (Shende and Patel, 2017). The modeling of multi-leaf collimator (MLC) transmission affects the accuracy of planning accuracy more in IMRT than in 3D conformal radiotherapy (3D CRT), thus increasing the importance of its adjustment (Kinsella et al., 2016). Patient-specific quality assurance (QA) procedures cannot detect errors in the MLC model, and the MLC must be set correctly during commissioning (Nelms et al., 2013). Therefore, MLCs need to be correctly characterized in treatment planning systems to perform IMRT accurately. IMRT requires verification to evaluate the consistency between the treatment plan results and the radiation therapy equipment before treatment, and IMRT verification includes dose evaluation and dose distribution evaluation. Optimization of MLC parameters in treatment planning systems improves the verification results and reduces the burden of re-verification. In veterinary medicine, the number of staff involved in radiotherapy is small, and efficient IMRT validation with less time burden is required. Therefore, it is important to shorten the validation time by reducing the need for revalidation through the adjustment of the MLC parameters. In veterinary medicine, the treatment field is smaller than that in humans, and distributing accurate doses is more complicated in the head and neck region because of the close proximity of important organs and tumors; it is not clear whether it is appropriate to refer to MLC parameters for human medicine. In addition, dogs and cats often use low energies, mainly 4-MV X-rays, because the irradiation field is not only small but also the target is superficial to the body surface. To the best of the authors’ knowledge, there are no reports on MLC parameter adjustment in dogs and cats, and there are no reports summarizing the clinical results of IMRT validation for 4-MV X-rays in veterinary medicine. The results of IMRT validation using 4-MV X-rays in this study indicate that IMRT can be used even at low energy, which may contribute to the application of IMRT to human cancer cases, such as pharyngeal cancer with a small irradiation field and thin body thickness, superficial mammary tumor, and pediatric cancer. The purpose of this study was to demonstrate the feasibility of IMRT at 4 MV and 6 MV in veterinary clinical patients. An additional purpose of this study was to show that appropriate MLC parameter adjustment for IMRT using 4 and 6 MV energy can reduce the need for QA revalidation in real companion animal clinical cases. Furthermore, it provides information on appropriate MLC parameters for IMRT in veterinary medicine, which have not been reported previously. Materials and MethodsSubjects and devices usedThe subjects were 88 cases (77 dogs and 11 cats weighing 8.1±6.5 kg: 1.9–34.6 [mean±SD: range]) and 101 treatment plans of oncological diseases with IMRT validation from May 2017 to December 2021. In total, 56 cases and 64 plans used 4 MV and 32 cases and 37 plans used 6 MV. MLC parameter adjustments were evaluated in 10 provisional plans of 4 MV and 6 MV prior to clinical use. We used Synergy (Elekta AB, Stockholm, Sweden) with 0.5-cm leaf Agility (Elekta AB) as the treatment device and the Monaco treatment planning system (Elekta AB, Stockholm, Sweden, ver. 5.11) as the treatment planning device. The IMRT validation equipment consisted of a 30 × 30 cm tough water phantom (Kyoto Kagaku Co., Ltd., Kyoto, Japan), a PinPoint chamber (Type 31013, PTW-Freiburg, Germany), an electrometer (RAMTEC Smart, ToyoMedic, Tokyo, Japan), an 8” × 10” Gafchromic film (GAFCHROMICTM EBT3, 04022004, Ashland Specialty Ingredients, Wilmington, DE, USA), and OmniProImRT (ver. 1.7, IBA Dosimetry, Schwarzenbruck, Germany) as the IMRT analyzer. Adjustment of MLC parametersAt the Veterinary Medical Center of Yamaguchi University, the calculated values of Monaco with the default MLC parameters were compared with the dose measurements using tuff water and PinPoint-type chambers. The MLC parameter adjustment method (Type F) published by Elekta was used to evaluate the MLC transmission and leaf end for seven different plans (10 × 10 cm, 3ABUT, DMLC, 7segA, FOURL, HDMLC, and HIMRT). At 4 and 6 MV, measurements were performed using a tuff water and PinPoint chamber. Leaf transmission and offset values of the MLC parameters were adjusted to keep the error less than 5%, and the values were registered in the treatment planning system. The leaf groove width (mm) was set to 0.4; interleaf leakage was set to 3.00; leaf tip leakage was set to 1.10; and static leaf gap (mm) was set to 0.10. Preclinical evaluation of tentative plansTentative IMRT treatment plans for 20 cases (10 cases in 4 MV and 10 cases in 6 MV) were generated using the treatment planning system. The treatment planning system was compared with PinPoint chamber dosimetry at the site of planning target volume (PTV) and OAR (where the dose is approximately 1/3) to assess whether the criteria of PTV within 3% and OAR within 5% were met in all cases. Gamma path analysis using the GAFCHROMIC film was also performed to evaluate whether the criteria of gamma pass rate [dose difference (DD)/distance-to-agreement (DTA): 3%/3 mm, >10% dose threshold] of 95% or higher were met in all cases, which was defined as tolerance limits. Efficient IMRT validation in clinical plansWe measured the absolute doses using PTV and OAR chambers in IMRT of dogs and cats in 101 plans and evaluated the relative dose distribution by gamma analysis using EBT3 film. Doses were evaluated using a PinPoint-type chamber and a tough water phantom to avoid rapid changes in dose because of the large number of small-field IMRTs. The relative dosimetry for IMRT verification was performed using films, and the calculation of concentration-dose conversion curves was adopted by the segment method, in which multiple segments with different doses were formed by MLC and each film was irradiated once with a dose equivalent to the required number of measurement points (Shukla et al., 2013). The films were scanned 1 h after irradiation using an Epson Expression 10,000 XL scanner (SEIKO EPSON Co., Nagano, Japan) with 72 dpi resolution in 48-bit RGB format. The gamma-path analysis (DD/DTA: 3%/3 mm, >10% dose threshold) was evaluated by comparing the planned dose with the treatment planning system dose using OmniProImRT. The criteria of 3%/2 mm, >10%, are the most recent recommended conditions for gamma-path analysis (Miften et al., 2018). Therefore, we evaluated the impact of this change in criteria by applying this criterion of 3%/2 mm to 20 cases from September to December 2021. Statistical analysisComparison of gamma analysis pass rates between the two conditions was performed using Wilcoxon’s statistics, with a |p| < 0.05 indicating a significant difference using JMP Pro 16 statistical software (SAS, Cary, NC). Ethical approvalIn all included clinical cases, informed consent for radiotherapy was obtained from each owner. This study did not require ethical approval as the data were retrospectively analyzed. ResultsThe default leaf transmission and leaf offset for both 4 MV (4 MV-Default) and 6 MV (6 MV-Default) were 0.005 and 0, respectively (Table 1). The error (%) for each verification pattern is shown in Table 1, with FOURL and HDMLC having error values of 5% or higher for both 4 MV and 6 MV. For 4 MV, the error values were within 5% for the 4 MV-Model C (leaf transmission and leaf offset were 0.008 and 0, respectively), and for 6 MV, the error values were within 5% for all items for 6 MV-Model D (leaf transmission and leaf offset were 0.0066 and 0.06, respectively) (Table 1). Table 2 displays the results of the treatment planning system and measurement errors of PTV and OAR, as well as the pass rate of gamma analysis by film, for 10 cases each of the 4- and 6-MV IMRT plans. In the 4-MV IMRT plan, the 4 MV-Default (before adjustment) showed errors of 3% or more for PTV in 4 out of 10 cases, which was out of the tolerance limits, while the 4 MV-Model C (after adjustment) showed errors of 3% or less for PTV and 5% or less for OAR in all 10 cases. On the contrary, in the 6-MV IMRT plan, the OAR error value was outside the tolerance limits (less than 5%) in 1 out of 10 cases in the adjusted 6 MV-Model D, whereas in the unadjusted 6 MV-Default, the OAR error value was within the tolerance limits in all 10 cases. In the verification of dose distribution using film, gamma analysis of 95% or higher was achieved. Therefore, the 4 MV-Model C (leaf transmission and leaf offset of 0.008 and 0, respectively) for 4 MV and 6 MV-Default (0.005 and 0, respectively) for 6 MV will be used in future studies. After adjustment of the MLC parameters, we examined the data from 88 cases and 101 plans (76 dogs and 12 cats) treated with IMRT from May 2017 to December 2021. Overall, 56 cases and 64 plans were treated with 4 MV and 32 cases and 37 plans were treated with 6 MV. In the irradiation area, the median length (head–tail direction) was 10.5 cm (width=2.4–26.1) and the median width (left–right direction) was 7.0 cm (width=1.7–15.0). The error values were within 3% for all PTVs [4 MV: median=0.45%; width=−2.96 to 2.88%; Fig. 1(a); and 6 MV: median=0.15%, width=−2.96 to 2.83%; Fig. 1(d)] and within 5% for all OARs [4 MV: median=−1.22%, width=−4.75 to 4.63%; Fig. 1(b); and 6 MV: median=−0.64%, width=−4.18 to 3.14%; Fig. 1(e)], and all film-based gamma analysis pass rates were above 95% [4 MV: median=99.00%, width=95.90–99.98%; Fig. 1(c); and 6 MV: median=99.07%, width=95.29–99.99%; Fig. 1(f); 3 mm, 3%, threshold 10%], which allowed us to create a plan within the tolerance limits and implement treatment without the need for revalidation. In the study of changing the criteria of gamma-path analysis, using 20 animals from August to December 2021, the median value of gamma-path analysis was 99.01% (range=96.56–99.94%) under the conditions of gamma 3%/3 mm, >10%, and the median value of gamma-path analysis was 98.02% (range=95.01–99.87%) under the conditions of gamma 3%/2 mm, >10%, which showed no significant differences (p=0.08; Fig. 2). DiscussionIMRT is widely used in veterinary radiotherapy (Kitagawa et al., 2020), and it is important to maintain optimal MLC transmission in IMRT to provide the best treatment. Various methods for adjusting the transmission required for IMRT have been validated in human medicine, but none has been reported in veterinary medicine. This is the first study to report the adjustment of MLC parameters in veterinary medicine. In addition, although veterinary medicine has limited validation equipment compared to human medicine, we were able to adjust the MLC parameters using basic detectors and films at our facility. Therefore, no cases required revalidation of the IMRT plan due to prior adjustment of the MLC parameters, and it was thought that QA was possible with an emphasis on time efficiency. A previous human study reported the results of the MLC parameters using Elekta Synergy and Agility as follows: leaf transmission=0.0032, leaf offset=−0.05, leaf groove width=0.4, interleaf leakage=7.0, leaf tip leakage=1.18, and MLC corner leakage=0 (Roche et al., 2018). Naturally, it is necessary to make adjustments for each device; however, the conditions required for clinical cases in veterinary medicine are thought to be different, and the data of our study provide useful information for examining MLC parameters used in veterinary medicine. In addition, although there are few reports on QA in veterinary medicine, our report is expected to contribute to information on MLC parameter values in veterinary medicine, as well as data on QA reports in other institutions. Table 1. Data of the default transmission, leaf transmission after adjustment, and leaf offset for both 4-MV and 6-MV IMRT and the results error (%) for each verification pattern.
Table 2. Results of the treatment planning system and measurement errors of PTV and OAR, as well as the pass rate of gamma analysis by film for 10 cases each of the 4-MV and 6-MV IMRT plans.
In human medicine, 6 and 10 MV are often used for IMRT clinically (Kleiner and Podgorsak, 2016; Mazonakis et al., 2021), and many reports on the adjustment of MLC parameters are also related to 6 and 10 MV (Shende and Patel, 2017). However, in tumors close to the body surface, such as pharyngeal cancer, mammary glands, and pediatric cancer, IMRT with low-energy X-rays is a possible treatment option when OAR is adjacent to the target. In this study, we performed IMRT for the treatment of various tumors in various locations in animals with spontaneous tumors. In IMRT using 6 MV, which is frequently used in humans, QA was within the tolerance limits in all cases in this study, and it was thought that the MLC parameters could be adjusted without the need for re-examination. In addition, 4-MV IMRT, which is rarely used in human medicine, could be performed at the same level without the need for QA review in all cases. Although the MLC parameters were adjusted for cats and dogs in this study, it was thought that the values of these parameters would be useful for IMRT with small irradiation fields or superficial targets in human medicine.
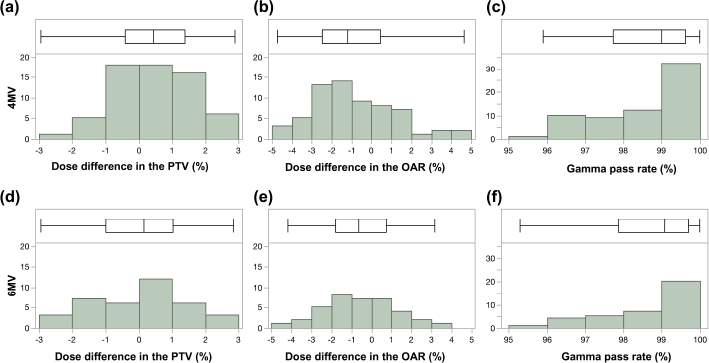
Fig. 1. Box-and-whisker diagram and histogram of error values in PTV, OAR, and gamma pass rate in clinical cases receiving 4-MV and 6-MV IMRT. The results of error values in PTV (a; 4 MV, d; 6 MV), OAR (b; 4 MV, e; 6 MV), and gamma pass rate (c; 4 MV, f; 6 MV). The error values were within 3% for all PTVs (4 MV: median=0.45%; width=−2.96% to 2.88%; 6 MV: median=0.15%, width=−2.96% to 2.83%) and within 5% for all OARs (4 MV: median=−1.22%, width=−4.75 to 4.63%; 6 MV: median=−0.64%, width=−4.18 to 3.14%), and all film-based gamma analysis pass rates were above 95% (4 MV: median=99.00%, width=95.90%–99.98%; 6 MV: median=99.07%, width=95.29%–99.99%; 3 mm/3%, threshold 10%), which allowed us to create a plan within the acceptable range and implement treatment without the need for revalidation. The top of each graph shows a box-and-whisker diagram with the middle line of the box indicating the median value; the left end of the box indicating the first quartile; the right end of the box indicating the third quartile, the left end of the whisker indicating the minimum value; and the right end of the whisker indicating the maximum value. Below each graph is a histogram showing the dose difference (%) or gamma pass rate (%) on the X-axis and the cumulative number of plans on the Y-axis To adjust the MLC parameters in the human medicine, diodes, such as MapCHECK 2, Gafchromic EBT3 film, and ionization chambers were used in the study (Grofsmid et al., 2010; Kinsella et al., 2016; Roche et al., 2018). Dosimetric validation of a commercial AAPM TG-106 proposes the use of films, portal images, or diodes as measurement devices to determine MLC parameters (Das et al., 2008). In veterinary medicine, compared to human medicine, it is economically more difficult to purchase expensive diathermy instruments, and there are associated limitations such as fewer radiation therapists and less manpower. Therefore, it is desirable to verify the results using basic dosimeters and films. In the present study, verification was performed using films and an ionization chamber, and it was thought that verification was possible without using relatively expensive measurement equipment. However, verification using film is difficult because of the storage and handling conditions of the film, the performance of the scanning equipment, and the reproducibility of the analysis. It also has the disadvantages of low initial costs and high running costs. Therefore, it is necessary to consider the balance between these two factors and to find a method that is easy to use. In the present study, it was considered that the gamma analysis path rate worsened, especially in the low-dose range in complicated plans such as those with two or more PTVs, and the absolute dosimetry with PTVs and OARs was within the tolerance limits in all cases. This may be because of the handling of the films. The fact that the verification was performed using films was considered a limitation of this study. Another limitation of this study was that we could not compare the validation results of diode arrays and films. Some veterinary hospitals use a diode array for QA of the plan (Kitagawa et al, 2020); however, it is expensive, and our hospital did not have one. Since it may be clinically easier to perform QA with diode arrays than with film and chamber measurements, information on differences in the QA results of film and diode arrays even in dogs and cats would be useful. A comparison between diodes and films has been reported (Kinsella et al., 2016), and further study is necessary. However, since the purpose of this study was to adjust MLC parameters for veterinary patients, a verification method with films is also recommended, and the fact that diode arrays could not be used was not considered to have a significant impact on this study. The third limitation is that the absolute values of the MLC parameters in this study are considered to be different for each machine, so the MLC parameter needs to be adjusted for each machine. However, the values of the MLC parameters of 4 MV and 6 MV for veterinary clinical patients in this study were considered important information at the beginning of the adjustment process.
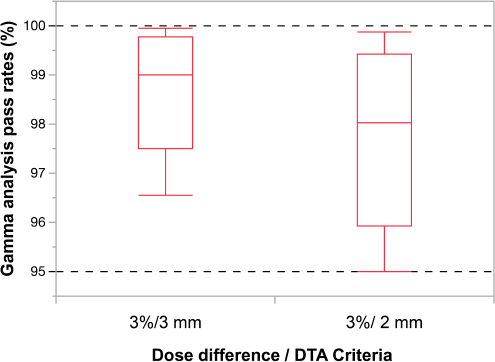
Fig. 2. Box-and-whisker diagram of the differences in gamma-path analysis for two criteria. In the study of changing the criteria of gamma-path analysis, using 20 animals from August to December 2021, the median value of gamma-path analysis was 99.01% (range=96.56%–99.94%) under the conditions of gamma 3%/3 mm, >10%, and the median value of gamma-path analysis was 98.02% (range=95.01%–99.87%) under the conditions of Gamma 3%/2 mm, >10%. There were no statistically significant differences (p=0.08). In recent years, it has been recommended that the gamma analysis pass rate conditions for relative dose distribution verification be 2 mm/3%, and 10% threshold, instead of 3 mm/3%, and 10% threshold (Miften et al., 2018). In this study, we examined the respective gamma analysis pass rates in the last 20 cases and found that the median gamma analysis pass rate was 99.01% (range=96.56–99.94%) for the 3 mm/3%, and threshold 10% conditions, and 98.02% (range=95.01–99.87%) for the 2 mm/3%, and threshold 10% conditions. Although the results of the two conditions were not significantly different and the recommended gamma analysis pass rate of 95% was exceeded in all cases, more stringent conditions will eventually be required as with human medicine. In the future, further study on the conditions of the gamma analysis pass rate is needed in the veterinary field. In this study of IMRT in dogs and cats, we were able to show that the validation results were convincing by adjusting the MLC parameters in all cases of 4 MV and 6 MV. Therefore, in the future, for cases with changes in tumor size and structures during the treatment period, it is recommended that online adaptive radiotherapy is used (Glide-Hurst et al., 2021). We adjusted the MLC parameters necessary for 4-MV and 6-MV IMRT. Our study showed that 4-MV IMRT could be used clinically without complications in dogs and cats. Furthermore, the need for revalidation was reduced when all the dose distributions were within tolerance limits, and IMRT was considered to be possible within appropriate tolerances. AcknowledgmentsNone. FundingThis publication was prepared without any external source of funding. Conflict of interestThe authors declare that there is no conflict of interest. Authors’ contributionsAdjusting the MLC parameters was performed by Toshie Iseri and Yoshinori Tanabe. Radiation therapy was performed by Toshie Iseri, Hiro Horikirizono, and Munekazu Nakaichi. Data organization and analysis were performed by Harumichi Itoh, Hiroshi Sunahara, Yuki Nemoto, Kazuhito Itamoto, and Kenji Tani. Toshie Iseri, Yoshinori Tanabe, and Hidekazu Tanaka were involved in the writing of the first draft, and all authors read and approved the final draft. ReferencesDas, I.J., Cheng, C.W., Watts, R.J., Ahnesjö, A., Gibbons, J., Li, X.A., Lowenstein, J., Mitra, R.K., Simon, W.E. and Zhu, T.C. 2008. TG-106 of the Therapy Physics Committee of the AAPM. Accelerator beam data commissioning equipment and procedures: report of the TG-106 of the Therapy Physics Committee of the AAPM. Med. Phys. 35, 4186–4215. Glide-Hurst, C.K., Lee, P., Yock, A.D., Olsen, J.R., Cao, M., Siddiqui, F., Parker, W., Doemer, A., Rong, Y., Kishan, A.U., Benedict, S.H., Li, X.A., Erickson, B.A., Sohn, J.W., Xiao, Y. and Wuthrick, E. 2021. Adaptive radiation therapy (ART) strategies and technical considerations: a state of the ART review from NRG Oncology. Int. J. Radiat. Oncol. Biol. Phys. 109, 1054–1075. Grofsmid, D., Dirkx, M., Marijnissen, H., Woudstra, E. and Heijmen, B. 2010. Dosimetric validation of a commercial Monte Carlo based IMRT planning system. Med. Phys. 37, 540–549. Kinsella, P., Shields, L., McCavana, P., McClean, B. and Langan, B. 2016. Determination of MLC model parameters for Monaco using commercial diode arrays. J. Appl. Clin. Med. Phys. 17, 37–47. Kitagawa, K., Reich, S.N., Desai, N., Policelli, R., Zimmerman, A., Sledge, D. and Vilar Saavedra, P. 2020. Outcome of a dog undergoing definitive-intent intensity-modulated radiation therapy for an intranasal ganglioneuroma. Vet. Radiol. Ultrasound. 61, E50–E54. Kleiner, H. and Podgorsak, M.B. 2016. The dosimetric significance of using 10 MV photons for volumetric modulated arc therapy for post-prostatectomy irradiation of the prostate bed. Radiol. Oncol. 50, 232–237. Mazonakis, M., Kachris, S. and Damilakis, J. 2021. VMAT for prostate cancer with 6-MV and 10-MV photons: Impact of beam energy on treatment plan quality and model-based secondary cancer risk estimates. Mol. Clin. Oncol. 14, 89. Miften, M., Olch, A., Mihailidis, D., Moran, J., Pawlicki, T., Molineu, A., Li, H., Wijesooriya, K., Shi, J., Xia, P., Papanikolaou, N. and Low, D.A. 2018. Tolerance limits and methodologies for IMRT measurement-based verification QA: Recommendations of AAPM Task Group No. 218. Med. Phys. 45, e53–e83. Nelms, B.E., Chan, M.F., Jarry, G., Lemire, M., Lowden, J., Hampton, C. and Feygelman, V. 2013. Evaluating IMRT and VMAT dose accuracy: practical examples of failure to detect systematic errors when applying a commonly used metric and action levels. Med. Phys. 40, 111722. Roche, M., Crane, R., Powers, M. and Crabtree, T. 2018. Agility MLC transmission optimization in the Monaco treatment planning system. J. Appl. Clin. Med. Phys. 19, 473–482. Shende, R. and Patel, G. 2017. Validation of Dosimetric Leaf Gap (DLG) prior to its implementation in Treatment Planning System (TPS): TrueBeam™ millennium 120 leaf MLC. Rep. Pract. Oncol. Radiother. 22, 485–494. Shukla, A.K., Oinam, A.S., Kumar, S., Sandhu, I.S. and Sharma, S.C. 2013. A calibration method for patient specific IMRT QA using a single therapy verification film. Rep. Pract. Oncol. Radiother. 18, 235–240. | ||
| How to Cite this Article |
| Pubmed Style Iseri T, Tanabe Y, Horikirizono H, Sunahara H, Itoh H, Nemoto Y, Itamoto K, Tani K, Tanaka H, Nakaichi M, . Adjustment of multi-leaf collimator parameters in 4-MV and 6-MV IMRT: A study of veterinary clinical cases. Open Vet J. 2022; 12(3): 407-413. doi:10.5455/OVJ.2022.v12.i3.15 Web Style Iseri T, Tanabe Y, Horikirizono H, Sunahara H, Itoh H, Nemoto Y, Itamoto K, Tani K, Tanaka H, Nakaichi M, . Adjustment of multi-leaf collimator parameters in 4-MV and 6-MV IMRT: A study of veterinary clinical cases. https://www.openveterinaryjournal.com/?mno=5527 [Access: July 26, 2024]. doi:10.5455/OVJ.2022.v12.i3.15 AMA (American Medical Association) Style Iseri T, Tanabe Y, Horikirizono H, Sunahara H, Itoh H, Nemoto Y, Itamoto K, Tani K, Tanaka H, Nakaichi M, . Adjustment of multi-leaf collimator parameters in 4-MV and 6-MV IMRT: A study of veterinary clinical cases. Open Vet J. 2022; 12(3): 407-413. doi:10.5455/OVJ.2022.v12.i3.15 Vancouver/ICMJE Style Iseri T, Tanabe Y, Horikirizono H, Sunahara H, Itoh H, Nemoto Y, Itamoto K, Tani K, Tanaka H, Nakaichi M, . Adjustment of multi-leaf collimator parameters in 4-MV and 6-MV IMRT: A study of veterinary clinical cases. Open Vet J. (2022), [cited July 26, 2024]; 12(3): 407-413. doi:10.5455/OVJ.2022.v12.i3.15 Harvard Style Iseri, T., Tanabe, Y., Horikirizono, H., Sunahara, H., Itoh, H., Nemoto, Y., Itamoto, K., Tani, K., Tanaka, H., Nakaichi, M. & (2022) Adjustment of multi-leaf collimator parameters in 4-MV and 6-MV IMRT: A study of veterinary clinical cases. Open Vet J, 12 (3), 407-413. doi:10.5455/OVJ.2022.v12.i3.15 Turabian Style Iseri, Toshie, Yoshinori Tanabe, Hiro Horikirizono, Hiroshi Sunahara, Harumichi Itoh, Yuki Nemoto, Kazuhito Itamoto, Kenji Tani, Hidekazu Tanaka, Munekazu Nakaichi, and . 2022. Adjustment of multi-leaf collimator parameters in 4-MV and 6-MV IMRT: A study of veterinary clinical cases. Open Veterinary Journal, 12 (3), 407-413. doi:10.5455/OVJ.2022.v12.i3.15 Chicago Style Iseri, Toshie, Yoshinori Tanabe, Hiro Horikirizono, Hiroshi Sunahara, Harumichi Itoh, Yuki Nemoto, Kazuhito Itamoto, Kenji Tani, Hidekazu Tanaka, Munekazu Nakaichi, and . "Adjustment of multi-leaf collimator parameters in 4-MV and 6-MV IMRT: A study of veterinary clinical cases." Open Veterinary Journal 12 (2022), 407-413. doi:10.5455/OVJ.2022.v12.i3.15 MLA (The Modern Language Association) Style Iseri, Toshie, Yoshinori Tanabe, Hiro Horikirizono, Hiroshi Sunahara, Harumichi Itoh, Yuki Nemoto, Kazuhito Itamoto, Kenji Tani, Hidekazu Tanaka, Munekazu Nakaichi, and . "Adjustment of multi-leaf collimator parameters in 4-MV and 6-MV IMRT: A study of veterinary clinical cases." Open Veterinary Journal 12.3 (2022), 407-413. Print. doi:10.5455/OVJ.2022.v12.i3.15 APA (American Psychological Association) Style Iseri, T., Tanabe, Y., Horikirizono, H., Sunahara, H., Itoh, H., Nemoto, Y., Itamoto, K., Tani, K., Tanaka, H., Nakaichi, M. & (2022) Adjustment of multi-leaf collimator parameters in 4-MV and 6-MV IMRT: A study of veterinary clinical cases. Open Veterinary Journal, 12 (3), 407-413. doi:10.5455/OVJ.2022.v12.i3.15 |