| Original Article | ||
Open Vet J. 2022; 12(6): 822-829 Open Veterinary Journal, (2022), Vol. 12(6): 822–829 Original Research Clinical outcome of intravitreal gentamicin injection for the treatment of end-stage glaucoma in five rabbits (eight eyes)Gemma Turner1*, Allyson Groth1, Gerry Skinner2, Hayley Volk1, Anu O’Reilly1 and Chloe Hardman11Melbourne Eye Vet, Mulgrave, Australia 2The Rabbit Doctors, Collingwood, Australia Submitted: 19/07/2022 Accepted: 15/10/2022 Published: 11/11/2022 *Corresponding Author: Gemma Turner. Melbourne Eye Vet, Mulgrave, Australia. Email: info [at] melbourneeyevet.com.au © 2022 Open Veterinary Journal
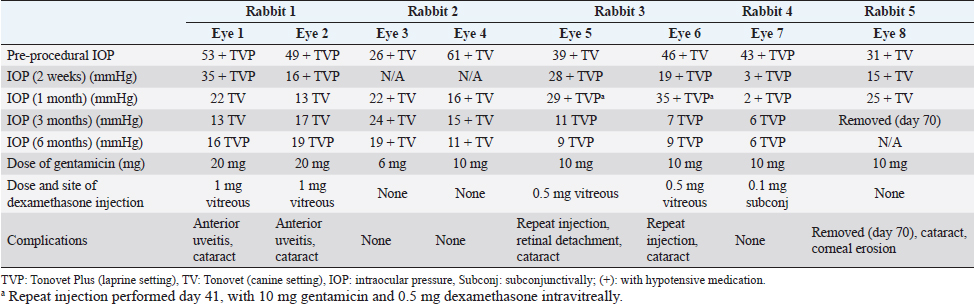
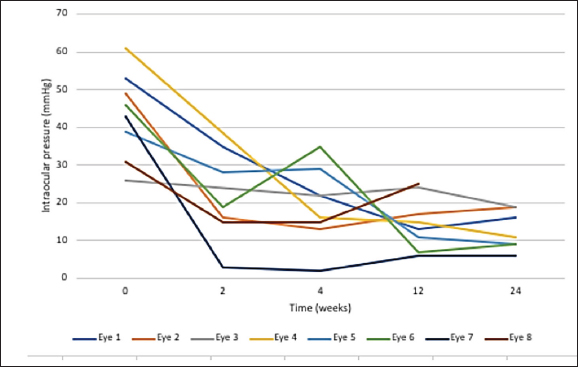
AbstractBackground: Glaucoma is a painful and blinding condition that occurs in many species, including rabbits. When medication is no longer effective in maintaining intraocular pressure (IOP), enucleation is the recognized treatment for rabbits with end-stage glaucoma. However, this procedure carries risks relating to the procedure and the anesthesia. Aim: The aim of this retrospective study was to report the efficacy of intravitreal gentamicin injection in controlling IOP in blind eyes of rabbits with end-stage glaucoma. Ocular and non-ocular complications were retrospectively assessed. Methods: Medical record review was performed to identify five client-owned rabbits (eight eyes) that were treated by intravitreal injection of 6–20 mg of gentamicin per eye (median 7.18 mg/kg) for chronic, end-stage glaucoma. Treatment was unilateral in two and bilateral in three rabbits. IOP control was assessed via rebound tonometry readings performed approximately 2 weeks, 1 month, 3 months, and 6 months after injection. Total follow-up was between 313 and 1,111 days. Ocular complications were recorded and systemic health was estimated by the owner-answered questionnaire and changes in body weight. Results: IOP was <25 mmHg in 87.5% of eyes 3 months post-injection. The most common ocular complications were cataracts (62.5%), anterior uveitis (25%), retinal detachment (12.5%), and corneal erosion (12.5%). There were no behavioral or body weight changes suggestive of systemic complications. Conclusion: 87.5% of rabbit eyes treated with intravitreal gentamicin had controlled IOP 3 months after injection. All eyes were blind at the time of injection. Ocular side effects were common. Investigation of the safety and systemic effects of intravitreal gentamicin injection is required; however, no overt complications were identified in treated rabbits in this study. Keywords: Chemical ablation, Ciliary body ablation, Gentamicin, Glaucoma, Rabbit. IntroductionGlaucoma is a painful and blinding condition in animals and people (Bouhenni et al., 2012), and affects three out of 1,000 rabbits (Innes and Williams, 2018), which is higher than their dog and cat counterparts (Innes and Williams, 2018). Despite this, there is less information available about glaucoma in rabbits. This may be because glaucoma is not as easily identifiable in rabbits as pain and distress are difficult to detect and quantify in prey species (Leach et al., 2011). Not only is glaucoma painful, but it can also cause secondary complications including buphthalmia, which can then lead to other painful and globe-threatening conditions such as non-ulcerative and/or ulcerative keratitis (Andrew, 2002). Medical treatment for glaucoma, using topical hypotensive medications, has been shown to be effective in rabbits (Yuschenoff et al., 2020). When these drugs are no longer effective, enucleation is the recognized treatment for rabbits with end-stage glaucoma (Diehl and McKinnon, 2016; Yuschenoff et al., 2020). Enucleation has its inherent risks relating to both the surgical procedure and the anesthetic. Rabbits have a large orbital vascular sinus in the ventronasal orbit which, if traumatized, can cause life-threatening hemorrhage (Diehl and McKinnon, 2016). Rabbits also have an increased risk of death related to anesthesia of 1.39% to 4.08% compared to 0.05% and 0.11% in dogs and cats, respectively (Brodbelt et al., 2008; Lee et al., 2018). As an alternative to enucleation for end-stage glaucoma in dogs, intravitreal gentamicin injections have been readily used with reported success rates varying from 65% to 95% (Bingaman et al., 1994; Rankin et al., 2015; Julien et al., 2020). The exact mechanism of how gentamicin affects the ciliary body epithelium is not fully understood. Intravitreal gentamicin injection procedures are often utilized in dogs due to their lower cost compared with enucleation, as well as the perceived safety advantages afforded by either requiring sedation only or requiring a shorter duration of general anesthesia as compared to enucleation (Julien et al., 2020). Systemic absorption of gentamicin, an aminoglycoside antibiotic, is of concern due to its known nephrotoxic effects via tubular necrosis (Kosek et al., 1974). Doses of 4 mg/kg subcutaneously twice daily for 4 weeks have been experimentally shown to cause renal failure and subsequent death in healthy rabbits by 20–27 days post-injection (Ginsburg et al., 1976). In dogs, a study by Rankin et al. (2015) has demonstrated that the total plasma concentration of gentamicin post intravitreal injection ranges from 0.21 to 9.71 µg/ml after 25–40 mg of gentamicin per eye with a mean gentamicin cMAX of 2.29 µg/ml at 2.54 hours post-injection. Comparatively, the systemic absorption of intravitreal gentamicin in rabbits is not known, however, it is thought to leave the eye predominantly via the aqueous humor (Kane et al., 1981). The purpose of this article was to retrospectively investigate the efficacy of intravitreal gentamicin injections in blind eyes of rabbits with end-stage glaucoma. To the authors’ knowledge, this is the first publication investigating the outcomes of intravitreal gentamicin injections for end-stage glaucoma in rabbits. Materials and MethodsCase selectionMedical records were retrospectively reviewed to identify rabbits with end-stage glaucoma that were treated with intravitreal gentamicin between 2019 and 2021 at an Australian Veterinary Ophthalmology specialist hospital. Records were included when there was documented ophthalmic examination by a board-certified Veterinary Ophthalmologist and a minimum of 6 months follow-up. End-stage glaucoma was defined as a blind eye (absent menace response, absent dazzle reflex, and absent direct and consensual pupillary light reflex) and intraocular pressure (IOP) ≥25 mmHg as estimated by rebound tonometry (TonoVet®/TonoVet Plus®) despite the use of topical glaucoma medications. Pre-procedural data gathered included signalment, body weight, eye affected, concurrent ocular disease, ocular and systemic medications, and IOP measurements. Procedural data was collected including anesthetic used, variations in treatment protocol including dosage of gentamicin, dosage, and site of administration of dexamethasone, and post-operative ocular and systemic medication. Surgical procedureWritten consent was obtained from owners prior to the procedure. Rabbits were sedated using a protocol preferred by the Veterinary Ophthalmologist performing the procedure and included ketamine (10 mg/kg), butorphanol (0.5 mg/kg), and medetomidine (0.1 mg/kg) intramuscularly for four rabbits and fentanyl (5 mcg/kg) and midazolam (0.2 mg/kg) intravenously for one rabbit. Intravitreal injection was performed as described in dogs (Rankin et al., 2015). The eye(s) were aseptically prepared with 1% betadine (Povidone-Iodine solution 10%), followed by the application of topical proparacaine hydrochloride 0.5%. Vitreocentesis was performed using a 25-gauge, 5/8-inch hypodermic needle inserted 6–8 mm posterior to the dorsal limbus, and approximately 0.3–0.5 ml of vitreous was aspirated. Gentamicin sulfate (100 mg/ml) was then injected into the posterior segment with a dose ranging from 6 to 20 mg of gentamicin sulfate per eye (maximum of 40 mg per rabbit). Dexamethasone sodium phosphate (3 mg/ml) was injected subconjunctivally in one eye of one rabbit and intravitreally in four eyes of two rabbits. Three eyes of two rabbits were not treated with dexamethasone. Post-injection treatment varied based on IOP readings immediately after injection and included aqueocentesis and the use of topical anti-glaucoma medications. Post-operative frequency and type of hypotensive treatment varied but included latanoprost 0.005% (Xalatan; Pfizer Australia Pty Ltd), dorzolamide hydrochloride 2% (Trusopt; Mundipharma Pty Ltd), and brinzolamide 1% (Azopt; Alcon Laboratories Pty Ltd). Other topical medication included diclofenac sodium 0.1% (Voltaren; Novartis Pharmaceuticals Australia Pty Ltd), ketorolac trometamol 0.5% (Acular; Allergan Australia Pty Ltd), and tacrolimus 0.02% (Bova Australia Pty Ltd). All patients received meloxicam oral liquid 1.5 mg/ml (Metacam; Boehringer Ingelheim Pty Ltd) for pain relief. Post-procedure monitoringData from each revisit included an evaluation of systemic health, as assessed by questions to owners regarding appetite, fecal production, general well-being/activity, and any signs of ocular discomfort. Other data included the rabbit’s body weight (measured to 10 g accuracy), IOP results on the treated eye(s), and ocular examination findings, including complications, which were also recorded. The results were collated to time points at approximately 2 weeks, 1 month, 3 months, and 6 months, as well as the last recorded examination. A retrospective assessment of the rabbits’ general health was estimated by a survey answered by the owners with questions based on clinical signs pertaining to pain, as described by the Rabbit Grimace Scale (Keating et al., 2012) and general signs known to be associated with renal injury (Appendices 1 and 2). The success of the procedure was defined as lowering the IOP to <25 mmHg with or without topical hypotensive medications by 3 months post-procedure. Ethical approvalThe authors confirm that the ethical policies of the journal, as noted on the journal’s author guidelines page, have been adhered to. All animals that participated in this study were client-owned and joined the study after the owner’s written consent. ResultsClinical findingsEight eyes of five rabbits (three female, two male) were included. The median age of the five rabbits at the time of the procedure was 5.4 years (range of 1.1–7.9 years). Breeds represented included four mini lops and one Blanc de Hotot. The procedure was performed in both eyes of three rabbits, and one eye in two rabbits. Glaucoma was present for a median of 81 days (range of 3–232 days) prior to the procedure. The cause of glaucoma was not specified in all eight eyes. Procedural outcomeThe dose of gentamicin ranged from 6 to 20 mg per eye, with a median of 7.18 mg/kg of gentamicin per rabbit (ranging from 4.2–10.7 mg/kg). The dose of dexamethasone per eye ranged from 0.4 mg to 1 mg per eye, with an average of 0.53 mg/kg of dexamethasone phosphate per rabbit (ranging from 0.18–0.98 mg/kg) (Table 1). The median duration of follow-up from procedure to final re-examination was 612 days (313–1,111 days). IOP was below 25 mmHg at 2 weeks (12–17 days) in 4/6 eyes (66.7%). Results from two eyes were not available at this time. The IOP was also below 25 mmHg at 1 month (24–35 days) in 6/8 eyes (75%), 3 months (75–84 days) in 7/8 eyes (87.5%), and in 7/7 eyes (168–191 days) post-procedure. One eye was enucleated at 70 days post-procedure due to a persistently high IOP (30 mmHg) and an epithelial erosion and was not included in the 6-month data point. Median IOP was reduced at 2 weeks (median IOP=19 mmHg), one (median IOP=21 mmHg), three (median IOP=13 mmHg), and 6 months (median=13 mmHg) compared to the pre-operative IOP results (mean=44 mmHg) as shown in Figure 1. The number and frequency of glaucoma drops per eye per day were reduced compared to pre-procedural frequency (median of 3.8 drops) versus at 2 weeks (median 2.4 drops), 1 month (median 2 drops), 3 months (median 0.4 drops), and 6 months (median 0.4 drops). Ocular complicationsOcular complications included progression of cataracts in 5/8 eyes (62.5%), anterior uveitis in 2/8 eyes (25%), retinal detachment in 1/8 eyes (12.5%), and epithelial erosion in 1/8 eyes (12.5%). Further surgical intervention was required in 3/8 eyes (37.5%), including repeat gentamicin injection at day 41 in 2/8 eyes (25%) and enucleation at day 70 in 1/8 eyes (12.5%). Systemic complicationsBody weight was measured at each visit using the same scales. Four rabbits had a <1% weight loss and one rabbit gained 4% of its body weight from the time of the procedure to 13 days after the procedure. Table 1. Individual results for the eight eyes (five rabbits) that received intravitreal gentamicin injection for end-stage glaucoma.
Fig. 1. Intraocular pressures (mmHg) of eight rabbit eyes following intravitreal gentamicin injection. At the time of article submission, one rabbit (one eye) was euthanized 375 days after the gentamicin injection due to poor mobility secondary to hindlimb arthritis. The remaining four rabbits were alive at the time of writing the article, 313, 307, 956, and 1,111 days post-procedure. Post-procedure hematology and biochemistry were available for one of the five rabbits at 1,058 days after the procedure. The blood tests were performed for reasons unrelated to the study. No abnormalities in parameters relating to renal physiology were noted. There were no clinical records of owner-perceived discomfort or signs pertaining to an acute kidney injury based on the questions about general appetite, fecal production, demeanor, and signs of ocular discomfort (including squinting and rubbing) posed to the owners at each revisit. The retrospective questionnaire involved more in-depth questions regarding pain, formulated based on the Rabbit Grimace Scale, and in-depth questions pertaining to possible acute kidney injuries, within the immediate post-procedural period. Questions regarding changes in appetite, hiding, changes to socialization, squinting, lethargy, teeth grinding, vocalization, overt aggression, changes in fecal production, weight loss, changes in drinking, and urination habits and any overt signs of abdominal pain did not highlight any abnormalities. All five rabbit owners stated that they would recommend the injection to friends and family as a treatment option for end-stage glaucoma in rabbits. DiscussionThe success rate of intravitreal gentamicin injection for end-stage glaucoma in rabbits was 87.5% at 3 months. This was similar to the outcome as described by Rankin et al. (2015) in dogs at 1-month post-procedure, however, differed from other studies which indicated a success rate as low as 65%, in a retrospective evaluation between 1985 and 1993 (Bingaman et al., 1994), and as high as 95% at 3 months in a retrospective analysis of 108 canine eyes between 2013 and 2019 by Julien et al. (2020). The decision to describe a normal IOP as <25 mmHg was based on the literature on normal IOPs in rabbits (Williams, 2021). Alternative values of normal IOP in rabbits (Pereira et al., 2011) indicate that the values obtained, following the intravitreal gentamicin injections in this study, are still outside of the normal ranges for the values set by Pereira et al. (2011). Further investigation is required to determine the true normal IOP in rabbits. The dose of gentamicin ranged from 6 to 20 mg per eye. The median dose per body weight of intravitreal gentamicin was 7.18 mg/kg and ranged from 4.2 to 10.7 mg/kg. The dose variation between rabbits was based on ophthalmologist preference. Comparatively, the study conducted in dogs by Rankin et al. (2015) had a median dose of gentamicin per body weight of 2.57 mg/kg and ranged from 0.61 to 7.50 mg/kg which correlated to a plasma concentration of 0.2–9.7 ul/ml. Comparisons between dogs and rabbits are difficult to interpret as drug metabolism and distribution differ, however, the authors believe that the total concentration of intravitreal gentamicin should be less than the known systemic dose of gentamicin for rabbits of 8 mg/kg once daily (Plumb, 2020) to reduce risk of nephrotoxicity. Caution should be exercised when considering intravitreal gentamicin in rabbits with known renal insufficiency or those that are receiving other nephrotoxic drugs. There were two eyes in one rabbit (25%) that required a second intravitreal injection, with the time from the first injection being 41 days from the first injection. In dogs, repeat injection was only required in 12 eyes (11%) of eyes with a mean time from the first injection of 158 days (Julien et al., 2020). The authors recognize that the difference in the percentage of rabbits needing repeat injections compared to dogs could be due to a number of factors including a smaller number of patients, varied gentamicin protocols, and limitations on the total dose of gentamicin per eye that could be safely administered to rabbits, due to rabbits generally being smaller in size compared to dogs. The most common ocular complication was the progression or development of cataracts in 62.5% of eyes as compared to the study in dogs where the most common ocular complication was phthisis bulbi in 59.2% of eyes (Julien et al., 2020). In this study, obvious signs of phthisis bulbi were not noted in any examination postoperatively. The authors do acknowledge, however, that any reduction in globe size could not be accurately assessed as globe diameters were not measured pre- and post-procedure. The second most common ocular complication was anterior uveitis in two out of eight eyes (25%), followed by one eye with a corneal epithelial erosion and one eye with a retinal detachment (12.5% incidence each). The authors believe that whilst not insignificant, these complications may be effectively managed through the addition of topical medication. This was shown in the two eyes with anterior uveitis which was managed with the use of topical 0.1% diclofenac twice daily and oral 1.5 mg/ml meloxicam once daily. Resolution of the uveitis was noted on days 17 and 55 post-procedure, right and left eye, respectively. Systemic complications were difficult to ascertain and were based solely on the lack of a downward trend in body weight and the lack of overt clinical signs of illness at the post-operative revisits and via a retrospective questionnaire at the time of article submission. There was a <1% reduction in body weight in any rabbit, and this was thought to be insignificant. This is a crude measure of overall health; however, it is a common clinical sign seen with many illnesses in rabbits, including kidney disease (Harcourt-Brown, 2013). Systemic complications associated with the injection were assessed via questions based on owner-perceived assessments of their rabbit. The history taken at the time of each review was less comprehensive than the retrospective questionnaire. The retrospective nature of assessing subtle behavioral and clinical signs can mean that some of these signs may have been overlooked if the owners were not actively monitoring for these specific signs before and after the procedure. Utilizing the Rabbit Grimace Scale at the time of the procedure, by presenting the image scale to the owner, maybe more useful, as owners may be more aware of the subtle signs of discomfort that may have been inadvertently overlooked. The main concern associated with the intravitreal gentamicin procedure was whether there was significant systemic absorption of gentamicin which has the potential to cause renal damage. The systemic absorption of intravitreal gentamicin is not known for rabbits with normal, healthy eyes. Furthermore, the absorption of gentamicin from an inflamed eye may be greater than that of a non-inflamed eye. Therefore, without further diagnostics, such as documenting plasma concentrations of gentamicin as performed in dogs by Rankin et al. (2015) in dogs, it is difficult to quantify and understand the potential for renal damage. A future prospective study could investigate the total plasma concentration of gentamicin after intravitreal injection in rabbits. The vitreal half-life of gentamicin in rabbits is known to be between 10 and 12 hours in normal rabbit eyes (Kane et al., 1981) and samples can be obtained around this time to determine the peak plasma concentration. Further studies could also look at the role of intravitreal dexamethasone and its systemic absorption and complications. The authors note that, although the role of intravitreal dexamethasone and its systemic absorption and complications were not investigated in this study, no outward side effects were observed. Limitations of the study were associated with its retrospective nature. These included variations in intravitreal injection protocols, sedation protocols, post-operative medication, and the timing of post-procedural examinations. Variations include, but are not limited to, variation in the volume of aspirated vitreous, dose of gentamicin, type, dose, and injection site used for concurrent glucocorticoids. Data such as histopathology on the enucleated eye in this study may have assisted in understanding the effects of gentamicin in the rabbit, including the identification of intraocular tumors, that have been documented in 39.5% and 62.5% of dogs and cats post gentamicin injection respectively (Duke et al., 2013a, 2013b). Other limitations of this study include the overall small sample size of eyes, conflicting data available on normal rabbit IOPs, and the difficulty in assessing the systemic effects of gentamicin. Overall, enucleation is the preferred treatment for a blind, glaucomatous eye refractory to medication as it negates the requirement for any post-procedural topical medication—whether that is a topical non-steroidal anti-inflammatory or topical glaucoma medication. Intravitreal gentamicin, with or without dexamethasone sodium phosphate injections, can be an appropriate alternative for rabbits as they have both a higher anesthesia risk and a higher risk of substantial blood loss during surgery. Intravitreal gentamicin may also be considered for those owners who have cost-prohibitive reasons to not consider eye removal. AcknowledgmentsNone. Conflicts of interestThe authors declare no conflicts of interest. Author contributionsGemma Turner collated data and wrote the manuscript. The procedure was performed by Chloe Hardman, Anu O’Reilly, and Hayley Volk. The manuscript was reviewed and edited by Chloe Hardman, Anu O’Reilly, Allyson Groth, Gerry Skinner, and Hayley Volk. ReferencesAndrew, S.E. 2002. Corneal diseases of rabbits. Vet. Clin. Exot. Anim. 5, 341–356. Bingaman, D.P., Lindley, M.N., Glickman, N.W., Krohne, S.G. and Bryan, G.M. 1994. Intraocular gentamicin and glaucoma: a retrospective study of 60 dog and cat eyes (1985–1993). Vet. Comp. Ophthalmol. 4, 113–119. Bouhenni, R.A., Dunmire, J., Sewell, A. and Edward, D.P. 2012. Review article: animal models of glaucoma. J. Biomed. Biotechnol. 2012, 692609. Brodbelt, D.C., Blissitt, K.J., Hammond, R.A., Neath, P.J., Young, L.E., Pfeiffer, D.U. and Wood, J.L. 2008. The risk of death: the confidential enquiry into perioperative small animal fatalities. Vet. Anaesth. Anal. 35, 365–373. Diehl, K.A. and McKinnon, J-A. 2016. Eye removal surgeries in exotic pets. Vet. Clin. Exot. Anim. 19, 245–267. Duke, F.D., Strong, T.D., Bentley, E. and Dubielzig, R.R. 2013a. Canine ocular tumors following ciliary body ablation with intravitreal gentamicin. Vet. Ophthalmol. 16(2), 159–162. Duke, F.D., Strong, T.D., Bentley, E. and Dubielzig, R.R. 2013b. Feline ocular tumors following ciliary body ablation with intravitreal gentamicin. Vet. Ophthalmol. 16(1), 188–190. Ginsburg, D.S., Quintanilla, A.P. and Levin, M. 1976. Renal glycosuria due to gentamicin in rabbits. J. Infect. Dis. 134, 119–132. Harcourt-Brown, F.M. 2013. Diagnosis of renal disease in rabbits. Vet. Clin. North Am. Exot. Anim. Pract. 16, 145–174. Innes, S. and Williams, D.L. Prevalence of ocular disease in 1000 normal rabbits: preliminary findings. In British Small Animal Veterinary Conference, Birmingham, UK, 2018. Julien, M.E., Schechtmann, S.A., Michau, T.M., Welihozkiy, A., Baldwin, T.L. and Stine, JM. 2020. Pharmacologic ciliary body ablation for chronic glaucoma in dogs: a retrospective review of 108 eyes from 2013 to 2018. Vet. Ophthalmol. 24, 125–130. Kane, A., Barza, M. and Baum, J. 1981. Intravitreal injection of gentamicin in rabbits. Effect of inflammation and pigmentation on half-life and ocular distribution. Invest. Ophthalmol. Vis. Sci. 20, 593–597. Keating, S.C., Thomas, A.A., Flecknell, P.A. and Leach, M.C. 2012. Evaluation of EMLA cream for preventing pain during tattooing of rabbits: changes in physiological, behavioural and facial expression responses. PLoS One 7(9), e44437. Kosek, J.C., Mazze, R.I. and Cousins, M.J. 1974. Nephrotoxicity of gentamicin. Lab. Invest. 30(1), 48–57. Leach, M.C., Coulter, C.A., Richardson, C.A. and Flecknell, P.A. 2011. Are we looking in the wrong place? Implications for behavioural-based pain assessment in rabbits (Oryctolagus cuniculi) and beyond? PLoS One 6(3), e13347. Lee, H.W., Machin, H. and Adami, C. 2018. Peri-anaesthetic mortality and nonfatal gastrointestinal complications in pet rabbits a retrospective study on 210 cases. Vet. Anaesth. Analg. 45, 520–528. Pereira, F.Q., Bercht, B.S., Soares, M.G., Bittencourt da Mota, M.G. and Pigatto, J.A.T. 2011. Comparison of a rebound and an applanation tonometer for measuring intraocular pressure in normal rabbits. Vet. Ophthalmol. 14(5), 321–326. Plumb, D.C. 2020. Gentamicin. Plumb’s veterinary drugs. Available via https://app.plumbs.com/drug-monograph/vTu5mODx1yPROD (Accessed 19 April 2022). Rankin, A.J., Lanuza, R., KuKanich, B., Crumley, W.C., Pucket, J.D., Allbaugh, R.A. and Meekins, J.M. 2015. Measurement of plasma gentamicin concentrations post chemical ciliary body ablation in dogs with chronic glaucoma. Vet. Ophthalmol. 19(1), 57–62. Williams, D.L. 2021. The rabbit. In Veterinary ophthalmology, 6th ed. Ed., Gelatt, K. Hoboken, NJ: Wiley-Blackwell, pp: 2179–2190. Yuschenoff, D., Graham, J. and Pumphrey, S.A. 2020. Diagnosis and treatment of glaucoma in client-owned rabbits (Oryctolagus cuniculus): 16 eyes from 11 rabbits (2008–2019). J. Exot. Pet. Med. 34, 67–71. Appendix 1.Questionnaire for rabbit owners.
Appendix 2. Rabbit Grimace Scale (15).
| ||
| How to Cite this Article |
| Pubmed Style Turner G, Groth A, Oreilly A, Volk H, Skinner G, Hardman C. Clinical outcome of intravitreal gentamicin injection for the treatment of end-stage glaucoma in five rabbits (eight eyes). Open Vet J. 2022; 12(6): 822-829. doi:10.5455/OVJ.2022.v12.i6.6 Web Style Turner G, Groth A, Oreilly A, Volk H, Skinner G, Hardman C. Clinical outcome of intravitreal gentamicin injection for the treatment of end-stage glaucoma in five rabbits (eight eyes). https://www.openveterinaryjournal.com/?mno=81848 [Access: April 03, 2025]. doi:10.5455/OVJ.2022.v12.i6.6 AMA (American Medical Association) Style Turner G, Groth A, Oreilly A, Volk H, Skinner G, Hardman C. Clinical outcome of intravitreal gentamicin injection for the treatment of end-stage glaucoma in five rabbits (eight eyes). Open Vet J. 2022; 12(6): 822-829. doi:10.5455/OVJ.2022.v12.i6.6 Vancouver/ICMJE Style Turner G, Groth A, Oreilly A, Volk H, Skinner G, Hardman C. Clinical outcome of intravitreal gentamicin injection for the treatment of end-stage glaucoma in five rabbits (eight eyes). Open Vet J. (2022), [cited April 03, 2025]; 12(6): 822-829. doi:10.5455/OVJ.2022.v12.i6.6 Harvard Style Turner, G., Groth, . A., Oreilly, . A., Volk, . H., Skinner, . G. & Hardman, . C. (2022) Clinical outcome of intravitreal gentamicin injection for the treatment of end-stage glaucoma in five rabbits (eight eyes). Open Vet J, 12 (6), 822-829. doi:10.5455/OVJ.2022.v12.i6.6 Turabian Style Turner, Gemma, Allyson Groth, Anu Oreilly, Hayley Volk, Geraldine Skinner, and Chloe Hardman. 2022. Clinical outcome of intravitreal gentamicin injection for the treatment of end-stage glaucoma in five rabbits (eight eyes). Open Veterinary Journal, 12 (6), 822-829. doi:10.5455/OVJ.2022.v12.i6.6 Chicago Style Turner, Gemma, Allyson Groth, Anu Oreilly, Hayley Volk, Geraldine Skinner, and Chloe Hardman. "Clinical outcome of intravitreal gentamicin injection for the treatment of end-stage glaucoma in five rabbits (eight eyes)." Open Veterinary Journal 12 (2022), 822-829. doi:10.5455/OVJ.2022.v12.i6.6 MLA (The Modern Language Association) Style Turner, Gemma, Allyson Groth, Anu Oreilly, Hayley Volk, Geraldine Skinner, and Chloe Hardman. "Clinical outcome of intravitreal gentamicin injection for the treatment of end-stage glaucoma in five rabbits (eight eyes)." Open Veterinary Journal 12.6 (2022), 822-829. Print. doi:10.5455/OVJ.2022.v12.i6.6 APA (American Psychological Association) Style Turner, G., Groth, . A., Oreilly, . A., Volk, . H., Skinner, . G. & Hardman, . C. (2022) Clinical outcome of intravitreal gentamicin injection for the treatment of end-stage glaucoma in five rabbits (eight eyes). Open Veterinary Journal, 12 (6), 822-829. doi:10.5455/OVJ.2022.v12.i6.6 |