| Case Report | ||
Open Vet. J.. 2024; 14(10): 2693-2699 Open Veterinary Journal, (2024), Vol. 14(10): 2678–2686 Case Report Bilateral phacoemulsification and intraocular lens implantation in a pet rabbit with cataractsFrédéric Goulle1*, Pauline Bodin2, Héloïse Cousin2, Morgane Prieto2 and Charles Cassagnes31Centre Hospitalier Vétérinaire AniCura Aquivet, Eysines, France 2Clinique Veterinaire Exotica, Le Haillan, France 3Clinique Vétérinaire Vet Riviera, Nice, France *Corresponding Author: Frédéric Goulle. Centre Hospitalier Vétérinaire AniCura Aquivet, Eysines, France. Email: frederic.goulle [at] wanadoo.fr Submitted: 24/02/2024 Accepted: 03/09/2024 Published: 31/10/2024 © 2024 Open Veterinary Journal
AbstractBackground: The occurrence of cataracts in rabbits is a common reason for consultation in veterinary ophthalmology. Case Description: A 6-year-old female Lop rabbit (Oryctolagus cuniculus) was referred with a 3-month history of bilateral cataracts. The owner noted that the rabbit was frequently bumping into obstacles. Physical examination was unremarkable. Routine hemogram and biochemistry panel were within usual values, and Encephalitozoon cuniculi were not detected by serology. A complete ophthalmological examination revealed bilateral hypermature cataracts causing total blindness. Bilateral tonometry showed normal intraocular pressures. Electroretinography (ERG) under general anesthesia revealed strong photoreceptor responses for each eye, and ocular ultrasound was normal except for bilateral hypermature cataracts. Following ERG, during the same anesthesia session, conventional phacoemulsification followed by acrylic intraocular lens (IOL; +58D) implantation was performed uneventfully in both eyes. Follow-up examinations were carried out for 1 year. Except for immediate post-operative bilateral corneal ulcers that were quickly cured with medical treatment, no other post-operative complications were observed. The 1-year follow-up examination revealed centered IOLs without posterior capsular opacity, enabling the observation of normal fundus in both eyes. The owner confirmed the rabbit had normal autonomy and moved with accuracy, as observed during follow-up visits and on videos provided by the owner. Conclusion: To the best of our knowledge, this is the first described case of successful bilateral phacoemulsification and bilateral intraocular lens implantation with prior electroretinographic evaluation of the retinal function in a pet rabbit with cataracts. Keywords: Cataract, Electroretinography, Intraocular lens, Phacoemulsification, Rabbit. IntroductionCataract, defined as an opacity of the lens or lens capsule is one of the most common causes of visual impairment and blindness in humans and animals (Glover and Constantinescu, 1997). Phacoemulsification cataract extraction with intraocular lens implantation is the standard surgical procedure in veterinary and human medicine. There is little information about the occurrence of spontaneous cataracts in rabbits (Williams, 2012; Gomes et al., 2018). The occurrence of cataracts in rabbits is low, a study of 1000 rabbits showed a low proportion of animals with cataracts with only 45 animals from that 1,000 having lens changes (Williams, 2021). The clinical case presented here describes the successful surgical management of bilateral cataracts in a healthy pet rabbit (Oryctolagus cuniculus) by bilateral phacoemulsification and bilateral intraocular lens implantation with prior electroretinographic evaluation of the retinal function. Case DetailsHistoryA 6-year-old female lop rabbit was referred to our hospital with a 3-month history of bilateral cataracts. The owner reported that the rabbit had frequently bumped into obstacles for the past 2 weeks, but no discomfort had been detected. The rabbit lived indoors with exits in the garden, the owners did not report any traumatic incident. The referring veterinarian had diagnosed bilateral mature cataracts and performed routine bloodwork and serum submission for indirect immunofluorescence for Encephalitozoon cuniculi. Results of routine hemogram and biochemistry were unremarkable and E. cuniculi was not detected by serology. Ophthalmic examinationA thorough ophthalmologic examination was performed. Palpebral, dazzle, direct, and indirect pupillary light reflexes were present in both eyes (oculus uterque OU). There was no pupil size asymmetry. The Schirmer tear test (Schirmer Tear Test, MSD Animal Health, Merck &Co., Inc., Rahway, NJ, USA) values were 13 mm/minute for the right eye (oculus dexter OD) and 11 mm/minute for the left eye (oculus sinister OG). Fluorescein stain (Fluoresceine 0.5% TVM, Dômes Pharma, Lempdes, France) was negative OU. Tonometry (Tonovet, Icare, Vantaa, Finland) revealed intraocular pressure (IOP) of 12 mm Hg OD and 11 mm Hg OS. Slit-lamp biomicroscopy examination (Hawk-eye, Dioptrix, Toulouse, France) revealed bilateral hypermature cataracts (Fig. 1), that prevented examination of the fundus OU by indirect ophthalmoscopy. No aqueous flare was apparent OU. On physical examination, the rabbit was in excellent overall condition. Electroretinography (ERG)Electroretinography under general anesthesia was scheduled to assess retinal function. Mydriasis was obtained with tropicamide eye drops (Mydriaticum 2 mg/0.4 ml, Thea Pharma, Clermont-Ferrand, France) (1 drop every 5 minutes for 20 minutes). The rabbit was premedicated with medetomidine (Dormilan, Axience, Barcelona, Spain) intramuscularly (0.07 mg/kg) and morphine hydrochloride (Morphine, CDM Lavoisier, Paris, France) subcutaneously (0.1 mg/kg). Afterward, general anesthesia was induced with ketamine (Anesketin, Dechra, AE Bladel, The Netherlands) intramuscularly (5 mg/kg) and intubation with a 2.0 endotracheal tube was performed. Anesthesia was maintained with 1.5% isoflurane (IsoFlo, Zoetis, Malakoff, France) delivered in oxygen. A 24-gauge catheter was set in the caudal auricular vein and sterile saline solution was delivered throughout the procedure. Electrodes were placed classically on each superior eyelid conjunctiva and subcutaneously at the base of each ear (Fig. 2). A reference electrode was placed subcutaneously on the dorsal neck. ERG (Visiosystem, Siem Bio-Medicale, Nîmes, France) was then performed: first an achromatic stimulation (1 Hz) in photopic conditions, followed by a blue stimulation (0.1 Hz) in scotopic conditions, another blue stimulation (0.1 Hz) after 8 minutes of dark adaptation, and then an achromatic flicker stimulation (30 Hz) in the photopic atmosphere. Bilateral recordings revealed strong photoreceptor responses for each eye (Fig. 3). Ocular ultrasonographyVitreoretinal ultrasonography (Vu Max HD, Sonomed Escalon, Lake Success, NY, USA) was performed OU by use of a 20-MHz ultrasound probe revealing bilateral hypermature cataracts, without retinal detachment neither vitreous anomaly nor lens displacement (Fig. 4). Surgical procedureFollowing ERG and ultrasonography, during the same anesthesia session, bilateral conventional phacoemulsification was performed. The anesthetized rabbit was placed in lateral recumbency; the right eye was prepared aseptically with diluted 5% povidone-iodine solution and draped routinely. After a 3.2 mm clear corneal incision using an angled microsurgical knife (Unique Technologies, Inc., Mohnton, USA) and inflation of the anterior chamber with 1.6% sodium hyaluronate viscoelastic substance (Ophteis Bio 1.6, Rayner, Worthing, UK), anterior capsulorhexis was performed. Then phacoemulsification (Infiniti Vision System, Alcon laboratories, Inc. Fort Worth, USA) using a one-handed technique was completed, followed by meticulous irrigation-aspiration of the residual cortical material and polishing of the posterior capsule (Fig. 5). Implantation inside the capsular bag of an acrylic intraocular lens (IOLs) with a power of 58 diopters (An-lens MR1-13, An-Vision GmbH, Hennigsdorf, Germany) was then performed in a conventional manner using an injection cartridge through the limbal keratotomy. After centering IOL in the capsular bag and meticulously removing the viscoelastic gel with the irrigation/aspiration handpiece (Fig. 5), the corneal incision was closed with simple interrupted absorbable sutures of 9-0 polyglactin 910 (Vicryl 9-0, Ethicon, Puerto Rico, USA). An intracameral injection of tissue plasminogen activator (tPA, 25 μg) (Actilyse, Boehringer Ingelheim, Paris, France) was then completed with a 30-gauge needle next to the incision to avoid fibrin formation. The animal was repositioned and the same procedure was repeated for the left eye. An Elizabethan collar was placed at the end of the surgery and the animal recovered uneventfully.
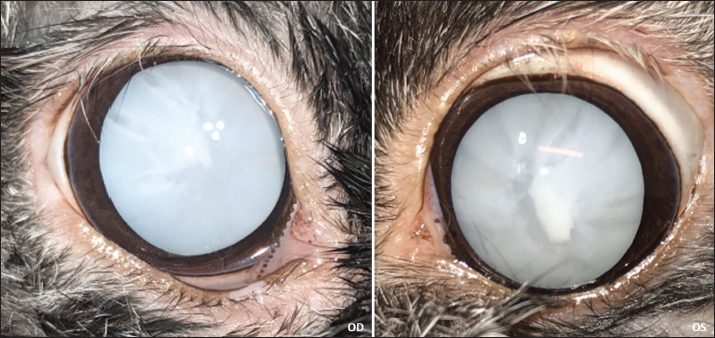
Fig. 1. Appearance of both eyes after chemical dilation. Note the bilateral total hypermature cataracts.
Fig. 2. Position of anesthetized rabbit and electrodes for electroretinographic examination. The electrodes were placed on each upper eyelid conjunctiva and subcutaneously, at the base of each ear and on the dorsal neck for the reference electrode. Postoperative managementImmediately after surgery, buprenorphine (Bupaq, VetViva Richter GmbH, Wels, Austria) 0.025 mg/kg, q6h, and meloxicam (Metacam, Boehringer Ingelheim Vetmedica GmbH, Ingelheim/Rhein, Germany) 1 mg/kg, q12 hours, were injected subcutaneously for 24 hours. Immediate postoperative topical medication was dexamethasone-neomycin sulfate-polymixin B sulfate eye drops (Maxidrol, Novartis Pharma S.A.S., Rueil-Malmaison, France), q8h. Feeding was resumed rapidly during the first 24 hours by force-feeding according to spontaneous food intake. During the first 24 hours, no post-operative ocular hypertension was noted OU, a very slight 1+ flare was observed OU with no particular discomfort and no corneal ulcers were present OU. The rabbit was discharged the day after the surgery with an Elizabethan collar. The oral postoperative treatment included meloxicam oral solution (Metacam, Boehringer Ingelheim Vetmedica GmbH, Ingelheim/Rhein, Germany) 0.5 mg/kg, q12h, for 5 days, tramadol hydrochlorhyde oral solution (Topalgic 100 mg/ml, Sanofi, Paris, France) 10 mg/kg, q12h, for 2 days and oral doxycycline hyclate (Doxyval 20, Ceva Santé Animale, Libourne, France) 10 mg/kg, q24h, for 7 days. The topical postoperative treatment included dexamethasone-neomycin sulfate-polymixin B sulfate eye drops (Maxidrol, Novartis Pharma S.A.S., Rueil-Malmaison, France), q8h, for 1 week then q12h for 3 weeks and hyaluronic acid-vitamin B12-trophic factors eye drops (Twelve TVM, Dômes Pharma, Lempdes, France) q8h, for 4 weeks. After 48 hours, the owner reported by telephone that the animal was eating well spontaneously, urinating and defecating normally. Outcome and follow-upFollow-up visits were carried out at 1, 3, and 12 weeks, then at 4, 8, and 12 months post-operatively. Between visits, the owner provided regular updates. A complete ophthalmologic examination was performed at every visit, including neuro-ophthalmologic evaluation, slit-lamp examination, rebound-tonometry, and fluorescein dye test. At the 1-week postoperative follow-up, tonometry was in normal values OU, no aqueous flare was noted and IOL was well-centered OU. A paracentral upper epithelial corneal ulcer was present in both eyes, a few millimeters below the keratotomy site. Dexamethasone-neomycin sulfate-polymixin B sulfate eye drops were therefore discontinued, replaced by tobramycin eye drops (Tobrex 0.3%, Novartis Pharma S.A.S., Rueil-Malmaison, France) q8h, for 2 weeks and hyaluronic acid-vitamin B12-trophic factors eye drops (Twelve TVM, Dômes Pharma, Lempdes, France) q8h, were continued for 4 weeks. At the 3-week follow-up visit, the epithelial ulcers were healed. Except for these bilateral corneal ulcers quickly healed, no other post-operative complications were observed. Follow-up examinations up to 1 year post-operatively revealed no inflammation, IOP in normal values OU, well-centered IOLs (Fig. 6), and normal fundus OU. Slight linear and vertical posterior capsular folds were observed OU at the 3-month visit, but no posterior capsular opacification (PCO) was present OU. At 8 and 12-month visits, these capsular folds were stable and no PCO was still observed (Fig. 6). The owner confirmed that the rabbit had normal autonomy and moved with accuracy, as observed during follow-up visits and on videos provided by the owner.
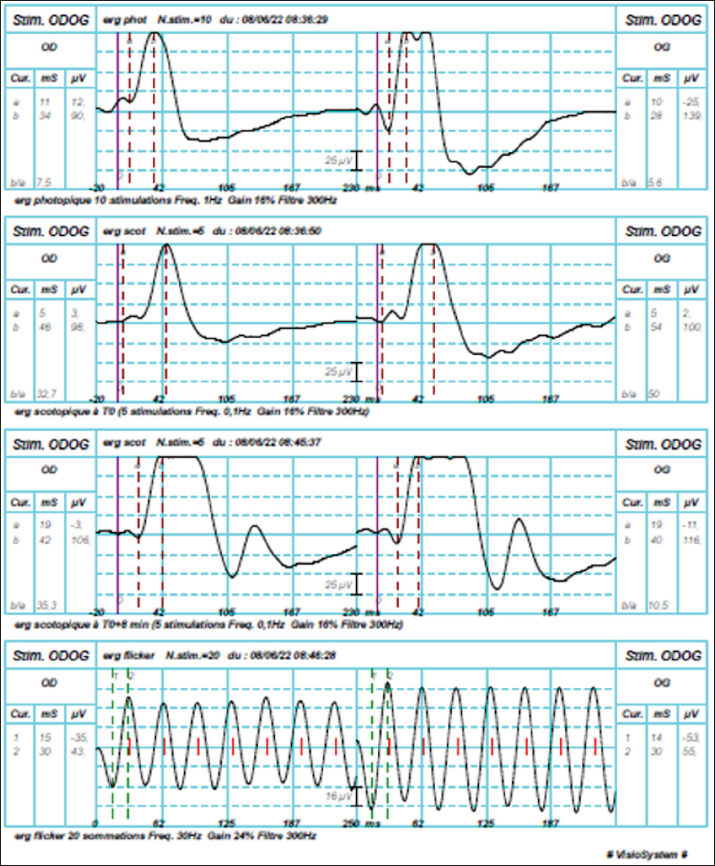
Fig. 3. Electroretinograms of the rabbit in different atmospheres, showing strong photoreceptor responses for each eye.
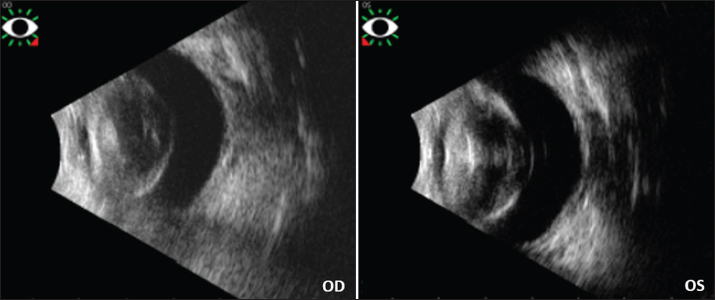
Fig. 4. Ultrasound images of the rabbit’s both eyes. Note for each eye the thickened and opaque lens, without lens displacement nor retinal detachment nor vitreous anomaly.
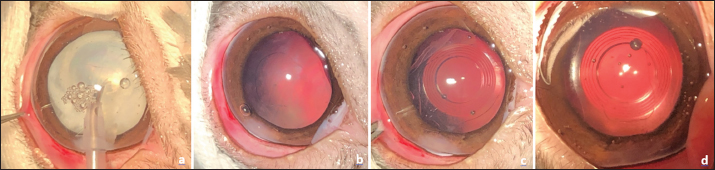
Fig. 5. Surgical steps pictures. One-handed phacoemulsification for right eye (a), aspect after irrigation-aspiration of the cortical material and polishing of the posterior capsule for the same eye (b). Aspect after implantation of intraocular lens in the capsular bag, before corneal incision suture, for the right eye (c), for the left eye (d). Ethical approvalNo ethical approval was needed for this article. DiscussionThere are different causes of cataracts in rabbits: congenital, juvenile, senile, Encephalitozoon cuniculi infection, and genetic (Geeraets et al., 1965; Gelatt, 1975; Ashton et al., 1976; Felchle and Sigler, 2002; Giordano et al., 2005; Künzel et al., 2008; Williams, 2013; Peng et al., 2015). Spontaneous cataracts have been described in rabbits, especially in young animals and are consistent with an inherited origin (Munger et al., 2002). In our case described here, serologic tests were negative for Encephalitozoon cuniculi and there were no neurological abnormalities or phacoclastic uveitis. The cause for cataracts was not determined in our case, but was supposed inherited. Retinal diseases in rabbits are poorly described; one case of spontaneous retinal degeneration in a laboratory animal (Reichenbach and Baar, 1985) and an isolated case of retinal toxicity following oral administration of ofloxacin (Rampal et al., 2008) have been reported. Although retinopathies are therefore rare in rabbits, we carried out an electroretinographic examination, as we routinely do for domestic carnivores prior to any cataract surgery. Moreover, cataract surgery is not as routinely performed in rabbits as in dogs. As little is known about spontaneous retinal diseases in rabbits, and as the bilateral cataract procedure is quite expensive and risky (considering the anesthetic risk), we think that retinal function evaluation by ERG is relevant. Furthermore, the retinal function evaluation by ERG is very common in laboratory animal medicine such as rabbits to assess the retinal toxicity of xenobiotics used in ophthalmology. In rabbits, the photoreceptor population is essentially dominated by rods (95%) (Famiglietti and Sharpe, 1995). The ERG performed in our case showed a strong photoreceptor response in both photopic and scotopic atmospheres. During phacoemulsification in rabbits, cases of iris prolapse into the keratotomy wound have been described due to the convexity of the iris in this species, this complication being favored by suboptimal mydriasis (Gomes et al., 2018), and by a very shallow anterior chamber in rabbits, worsened by the mature cataract and the higher lens volume. We did not encounter any intraoperative difficulties of this type in our case, the mydriasis having been total and stable during the surgery.
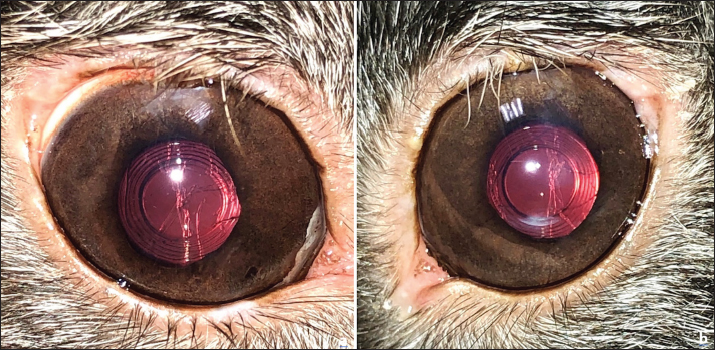
Fig. 6. Appearance of both eyes at 8 months post-operatively. Note well-centered intraocular lenses and presence of slight posterior capsular folds, in the right eye (a), in the left (b). Post-operative complications described in rabbits include post-operative ocular hypertension, post-operative inflammation with fibrinous reaction, particularly in young rabbits, and non-infected superficial corneal ulcers (Gomes et al., 2018). In our case, an ulcer of this type developed in both corneas one week after surgery. Conventional topical treatment allowed healing in each eye. The rabbit lens has a high potential for lens fiber re-growth and rabbits are known to produce posterior capsular opacities (PCO), more intensively in young animals (Wallentin et al., 2002; Gwon, 2008; Sandmeyer et al., 2011). In rabbits, PCO could be reduced by IOL implantation (Hazra et al., 2012; Ollerton et al., 2012; Sanchez et al., 2017). A prototype IOL with a size of 13 mm and a refractive power of +58 diopters has been shown to be of suitable size in rabbits and to achieve emmetropia in this species (Sanchez et al., 2017). Thanks to this latter study, an IOL designed for rabbits is now commercially available and was used for each eye in our case. Although it was formerly thought that a blind rabbit could apparently lead a normal life, it is now clear that for rabbits as for other animals, cataract surgery can greatly improve the rabbit's quality of life (Sanchez et al., 2018). It has been shown that rabbits can see both in front and behind them (Williams, 2013); therefore, a cataract, especially bilateral, greatly handicaps the animal in its movements. In our case, the owner confirmed that the rabbit had now regained autonomy and normal activity, and was moving with precision, with runs in the garden, as observed during follow-up visits and on videos provided by the owner. ConclusionTo the best of our knowledge, this is the first described case of successful bilateral phacoemulsification and bilateral intraocular lens implantation with prior electroretinographic evaluation of the retinal function in a pet rabbit with cataracts. AcknowledgmentNone. Conflict of interestThe authors declare no conflict of interest. FundingThis work received no specific grant or funding. Authors’ contributionsFG contributed the conception and design of the study. FG performed the examinations, surgery, and follow-up. MP, PB, and HC performed the anesthesia and its monitoring, and managed the anesthetic recovery and the return to feeding. FG wrote the manuscript. CC participated in the literature search and contributed to writing part of the discussion. All the authors approved the final draft of the manuscript. Data availabilityThe data supporting the findings of this study are available within the manuscript. Any other data are available from the corresponding author upon reasonable request. ReferencesAshton, N., Cook, C. and Clegg, F. 1976. Encephalitozoonosis (nosematosis) causing bilateral cataract in a rabbit. Br. J. Ophthalmol. 60, 618–631. Famiglietti, E.V. and Sharpe, S.J. 1995. Regional topography of rod and immunocytochemically characterized "blue" and "green" cone photoreceptors in rabbit retina. Vis. Neurosci. 12, 1151–1175. Felchle, L.M. and Sigler, R.L. 2002. Phacoemulsification for the management of Encephalitozoon cuniculi-induced phacoclastic uveitis in a rabbit. Vet. Ophthalmol. 5, 211–215. Geeraets, W.J., Harrel, W., Guerry, D., Ham, W.T. and Mueller, H.A. 1965. Aging anomalies and radiation effect of rabbit lens. Acta Ophthalmol. 43, 3–21. Gelatt, K.N. 1975. Congenital cataracts in a litter of rabbits. J. Am. Vet. Med. Assoc. 167, 598–599. Giordano, C., Weigt, A., Vercelli, A., Rondena, M., Grilli, G. and Giudice, C. 2005. Immunohistochemical identification of Encephalitozoon cuniculi in phacoclastic uveitis in four rabbits. Vet. Ophthalmol. 8, 271–275. Glover, T.D. and Constantinescu, G.M. 1997. Surgery for cataracts. Vet. Clin. North. Am. Small. Anim. Pract. 27, 1143–1173. Gomes, F.E., de Matos, R. and Ledbetter, E. 2018. Phacoemulsification of bilateral cataracts in two pet rabbits. Open. Vet. J. 8(2), 125–130. Gwon, A. 2008. The rabbit in cataract/IOL surgery. In: Animal Models in Eye Research 1st Edn. (Ed Tsonis, P.A.). Elsevier, London, UK, pp: 184–204. Hazra, S., Palui, H. and Vemuganti, G.K. 2012. Comparison of design of intraocular lens versus the material for PCO prevention. Int. J. Ophthalmol. 5, 59–63. Künzel, F., Gruber, A., Tichy, A., Edelhofer, R., Nell, B., Hassan, J., Leschnik, M., Thalhammer, J.G. and Joachim, A. 2008. Clinical symptoms and diagnosis of encephalitozoonosis in pet rabbits. Vet. Parasitol. 151, 115–124. Munger, R.J., Langevin, N. and Podval, J. 2002. Spontaneous cataracts in laboratory rabbits. Vet. Ophthalmol. 5, 177–181. Ollerton, A., Werner, L., Fuller, S.R., Kavoussi, S.C., McIntyre, J.S. and Mamalis, N. 2012. Evaluation of a new single-piece 4% water content hydrophobic acrylic intraocular lens in the rabbit model. J. Cataract Refract. Surg. 38, 1827–1832. Peng, X.W., Roshwalb, S., Cooper, T.K., Zimmerman, H. and Christensen, N.D. 2015. High incidence of spontaneous cataracts in aging laboratory rabbits of an inbred strain. Vet. Ophthalmol. 18, 186–190. Rampal, S., Kaur, R., Sethi, R., Singh, O. and Sood, N. 2008. Ofloxacin-associated retinopathy in rabbits: role of oxidative stress. Hum. Exp. Toxicol. 27, 409–415. Reichenbach, A. and Baar, U. 1985. Retinitis-pigmentosa-like tapetoretinal degeneration in a rabbit breed. Doc. Ophthalmol. 60, 71–78. Sanchez, R.F., Becker, R., Dawson, C., Escanilla, N. and Lam, R. 2017. Calculation of posterior chamber intraocular lens (IOL) size and dioptric power for use in pet rabbits undergoing phacoemulsification. Vet. Ophthalmol. 20, 242–249. Sanchez, R.F., Everson, R., Hedley, J., Dawson, C., Lam, R., Priestnall, S.L., Garcia de Carellan, A., de Miguel, C. and Seymour, C. 2018. Rabbits with naturally occurring cataracts referred for phacoemulsification and intraocular lens implantation: a preliminary study of 12 cases. Vet. Ophthalmol. 21, 399–412. Sandmeyer, L.S., Bauer, B.S. and Grahn B.H. 2011. Diagnostic ophthalmology. Can. Vet. J. 52(3), 327–328. Wallentin, N., Lundgren, B., Holmén, J.B. and Lundberg, C. 2002. Development of posterior capsule opacification in the rabbit. Ophthalmic Res. 34, 14–22. Williams, D.L. 2012. Ophthalmology of exotic pets. Chichester, West Sussex, UK: Wiley-Blackwell, pp: 44–45. Williams, D.L. 2013. The rabbit. In: Veterinary ophthalmology. 5th ed. Eds., Gelatt, K.N., Gilger, B.C. and Kern, T.J. Hoboken, NJ: John Wiley & Sons, Inc., pp: 1725–1749. Williams, D.L. 2021. Small mammal ophthalmology. In: Veterinary ophthalmology. 6th ed. Eds., Gelatt, K.N., Ben-Shlomo, G., Gilger, B.C., Hendrix, D.V.H., Kern, T.J. and Plummer, C.E. Hoboken, NJ: Wiley-Blackwell, pp: 2179–2199. | ||
| How to Cite this Article |
| Pubmed Style Goulle F, Bodin P, Cousin H, Prieto M, Cassagnes C. Bilateral phacoemulsification and intraocular lens implantation in a pet rabbit with cataracts. Open Vet. J.. 2024; 14(10): 2693-2699. doi:10.5455/OVJ.2024.v14.i10.19 Web Style Goulle F, Bodin P, Cousin H, Prieto M, Cassagnes C. Bilateral phacoemulsification and intraocular lens implantation in a pet rabbit with cataracts. https://www.openveterinaryjournal.com/?mno=190367 [Access: January 25, 2026]. doi:10.5455/OVJ.2024.v14.i10.19 AMA (American Medical Association) Style Goulle F, Bodin P, Cousin H, Prieto M, Cassagnes C. Bilateral phacoemulsification and intraocular lens implantation in a pet rabbit with cataracts. Open Vet. J.. 2024; 14(10): 2693-2699. doi:10.5455/OVJ.2024.v14.i10.19 Vancouver/ICMJE Style Goulle F, Bodin P, Cousin H, Prieto M, Cassagnes C. Bilateral phacoemulsification and intraocular lens implantation in a pet rabbit with cataracts. Open Vet. J.. (2024), [cited January 25, 2026]; 14(10): 2693-2699. doi:10.5455/OVJ.2024.v14.i10.19 Harvard Style Goulle, F., Bodin, . P., Cousin, . H., Prieto, . M. & Cassagnes, . C. (2024) Bilateral phacoemulsification and intraocular lens implantation in a pet rabbit with cataracts. Open Vet. J., 14 (10), 2693-2699. doi:10.5455/OVJ.2024.v14.i10.19 Turabian Style Goulle, Frédéric, Pauline Bodin, Héloïse Cousin, Morgane Prieto, and Charles Cassagnes. 2024. Bilateral phacoemulsification and intraocular lens implantation in a pet rabbit with cataracts. Open Veterinary Journal, 14 (10), 2693-2699. doi:10.5455/OVJ.2024.v14.i10.19 Chicago Style Goulle, Frédéric, Pauline Bodin, Héloïse Cousin, Morgane Prieto, and Charles Cassagnes. "Bilateral phacoemulsification and intraocular lens implantation in a pet rabbit with cataracts." Open Veterinary Journal 14 (2024), 2693-2699. doi:10.5455/OVJ.2024.v14.i10.19 MLA (The Modern Language Association) Style Goulle, Frédéric, Pauline Bodin, Héloïse Cousin, Morgane Prieto, and Charles Cassagnes. "Bilateral phacoemulsification and intraocular lens implantation in a pet rabbit with cataracts." Open Veterinary Journal 14.10 (2024), 2693-2699. Print. doi:10.5455/OVJ.2024.v14.i10.19 APA (American Psychological Association) Style Goulle, F., Bodin, . P., Cousin, . H., Prieto, . M. & Cassagnes, . C. (2024) Bilateral phacoemulsification and intraocular lens implantation in a pet rabbit with cataracts. Open Veterinary Journal, 14 (10), 2693-2699. doi:10.5455/OVJ.2024.v14.i10.19 |