| Research Article | ||
Open Vet. J.. 2025; 15(2): 619-629 Open Veterinary Journal, (2025), Vol. 15(2): 619-629 Research Article Canine obesity, overweight, and adipokine serum concentration are associated with hematological, biochemical, hormonal, and cardiovascular markersAdrián Carzoli*, Ana Meikle and Paula PessinaUnidad de Imagenología, Laboratorio de Análisis Clínicos y LEMA, Facultad de Veterinaria-UdelaR, Montevideo, Uruguay *Corresponding Author: Adrián Carzoli. Unidad de Imagenología, Laboratorio de Análisis Clínicos y LEMA, Facultad de Veterinaria-UdelaR, Montevideo, Uruguay. adrian.carzoli [at] gmail.com Submitted: 26/08/2024 Accepted: 17/12/2024 Published: 28/02/2025 © 2025 Open Veterinary Journal
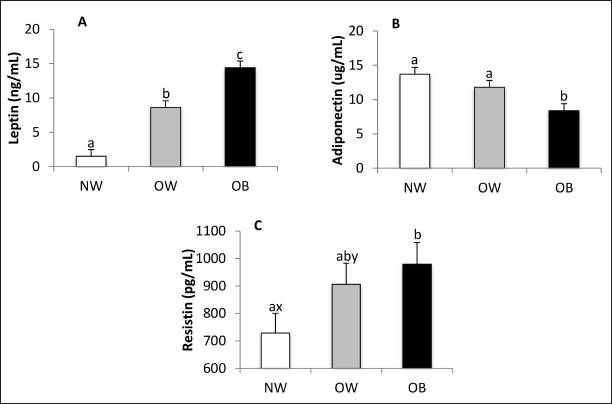
AbstractBackground: Canine obesity is an increasingly concerning issue that negatively impacts dogs’ health, quality of life, and lifespan. Aim: This study aimed to evaluate the adipokine profiles of overweight (OW) and obese (OB) dogs and their associations with various hematological, biochemical, hormonal, and cardiovascular markers. Methods: A total of 84 dogs were classified according to their body condition scores as normal weight (NW), OW, or OB, and were subsequently subjected to blood pressure measurement, blood testing, and urine sampling. Results: The findings revealed that OB dogs had higher serum concentrations of leptin and resistin while exhibiting lower levels of adiponectin when compared to NW dogs. Additionally, they showed higher systolic blood pressure. Serum creatinine levels were lower in OB dogs, and urinary specific gravity was reduced in both OW and OB dogs compared to their NW counterparts. Furthermore, total leukocyte counts and neutrophil counts were elevated in OW and OB dogs. The study also found that serum insulin levels were positively correlated with triglycerides, cholesterol, and C-reactive protein. Conclusion: Canine obesity is reflected in altered adipokine concentrations and is associated with insulin resistance, as well as changes in renal function, protein metabolism, and hematological markers. Keywords: Adipokines, Dog, Insulin resistance, Obesity, Resistin IntroductionObesity is defined as the excessive accumulation of adipose tissue, influenced by environmental, neuroendocrine, hereditary, and individual factors such as: age, sex, breed, neutering status, and comorbidities (Mao et al., 2013; Muñoz-Prieto et al., 2019). A dog is classified as obese (OB) or overweight (OW) when its body fat percentage is 40% or higher, or between 20% and 40%, respectively (Pérez-Sánchez et al., 2015). Similarly to human medicine, obesity is an increasingly common disorder in dogs, with a global prevalence estimated at between 32% and 59% (McGreevy et al., 2005; Lund et al., 2006; Courcier et al., 2010; Mao et al., 2013; Usui et al., 2016; Montoya-Alonso et al., 2017; Porsani et al., 2020). The excessive accumulation of adipose tissue is associated with various pathological conditions, including cardiovascular, respiratory, orthopedic, reproductive, and neoplastic diseases (Zoran, 2010), as well as obesity-related metabolic disorders, which encompass a group of risk factors linked to hyperinsulinemia and hypoadiponectinemia (Tvarijonaviciute et al., 2012). The relationship between obesity and obesity-related diseases, particularly metabolic disorders, appears to hinge on adipokines (Piantedosi et al., 2016). The most studied adipokine in both human and veterinary medicine is leptin, whose plasma levels rise in proportion to the mass of adipose tissue (Radin et al., 2009). In contrast, while the dynamics of certain serum adipokines, such as adiponectin and resistin, are well described in human and murine models during obesity (de Oliveira Leal and Mafra, 2013), the understanding of these dynamics in veterinary medicine remains incomplete. Some studies have indicated that serum adiponectin concentrations are lower in OB dogs compared to lean ones (Piantedosi et al., 2016; Tropf et al., 2017; Muñoz-Prieto et al., 2020), while others suggest that obesity does not significantly affect serum adiponectin levels (Verkest et al., 2011; Mori et al., 2013). Although elevated serum resistin levels in OB humans have been linked to atherosclerosis, pancreatitis, insulin resistance, and type II diabetes mellitus, there are limited reports on the relationship between serum resistin and obesity in dogs, with some studies showing no significant impact of obesity on resistin concentrations in canines (Eirmann et al., 2009; Kleine et al., 2020). This dysregulation in adipokine production associated with obesity is accompanied by insulin resistance (de Marchi et al., 2020; Ramos and Castillo, 2020) and is linked to various hematological (Safadi et al., 2021; Vieira et al., 2022), biochemical (Barić Rafaj et al., 2016; Piantedosi et al., 2016; Ramos and Castillo, 2020), hormonal (Lee et al., 2014; Cihan and Tural, 2019; Ramos and Castillo, 2020), and cardiovascular alterations (Piantedosi et al., 2016; de Marchi et al., 2020). To our knowledge, there have not been observational reports associating obesity with urinalysis in veterinary medicine concerning dogs. Furthermore, most studies do not take an integrative approach, particularly regarding urinalysis and hematology, which are essential for understanding the impact of obesity on tissues and organs. Therefore, this study aims to clarify the serum adipokine profile in OB and OW dogs, along with its associations with hematological, biochemical, hormonal, and cardiovascular parameters. Materials and MethodsInclusion and exclusion criteriaDogs aged between 1.5 and 10 years, regardless of gender or breed, were recruited for this study. Only healthy dogs (besides obesity) were included, and all dogs underwent a clinical examination, along with hematological tests, blood chemistry analysis, urinalysis, and a hormone panel assessing thyroid and adrenal function. Dogs that showed poor clinical examination results or blood values indicative of other health issues, aside from obesity, were excluded from the study. Groups conformationNinety dogs were initially recruited for the study; however, six were excluded due to comorbidities. This included five dogs that had thyroid test results indicating hypothyroidism and one dog that showed clinical signs consistent with hypercortisolism. Among the 84 dogs that remained in the study, they were classified using the nine-point body condition score (BCS) chart developed by Laflamme (1997). The classifications were as follows: normal weight (NW; BCS 4–5; n = 33), (OW; BCS 6–7; n = 28), and (OB; BCS 8–9; n = 23). The dog’s weights were measured using a Kretz Vet Scale model 150K4AOBC7RAR from Santa Fe, Argentina. Cardiovascular evaluationSystolic blood pressure (SBP), diastolic blood pressure (DBP), mean blood pressure (MBP), and cardiac frequency (CF) were assessed using an oscillometric monitor (Suntech Vet 30), following the methodology outlined by Acierno et al. (2018). Patients with a SBP greater than 160 mm Hg were classified as hypertensive. SamplingBlood samples were collected after a 12-hour fasting period, always in the morning hours. The samples were divided into three tubes: the first portion was placed in an Ethylenediaminetetraacetic acid (EDTA)-Sodium fluoride tube for glycemia analysis, the second portion was placed in a K3-EDTA tube for hematological studies, and the last portion was stored in a tube without anticoagulant for serum collection after clotting and centrifugation at 3,500 rpm for 10 minutes. The serum was then divided into two fractions; the first was used immediately for biochemical analysis, while the second fraction was frozen at –80°C for hormone processing. First morning urine samples were collected via free catch and stored in designated containers, refrigerated until processing. Adipokines and cytokinesLeptin and resistin concentrations were measured using canine-specific ELISA kits (EZCL-31K and RAB1017, respectively; Millipore, Saint Louis, USA). Adiponectin levels were assessed using a high-sensitivity human adiponectin ELISA kit (RD191023100; BioVendor R&D, Brno, Czech Republic), which has been previously validated for use in dogs (Tvarijonaviciute et al., 2010). Serum concentrations of TNF-α and IL-6 were determined using canine-specific Quantikine ELISA kits (CA6000 and CATA00, respectively; R and D Systems Inc., Minneapolis, MN). All kits were utilized according to the manufacturer’s guidelines, ensuring that the intra-assay coefficient of variation (CV) was less than 10% in all cases. HormonesInsulin concentrations were measured by radiometric assays (IRMA), using the INS-IRMA kit (DIA Source Immune Assays S.A., Belgium). Serum total T4 (TT4), free T4 (FT4), TSH, and cortisol concentrations were determined through solid-phase competitive chemiluminescent enzyme immunoassay (IMMULITE 1000), using commercial kits (Siemens, Diagnostic Product Corporation, Los Angeles, CA). The intra- assay and inter-assay CV was less than 10% for all cases. HematologyBlood counts were conducted using a Mythic 18 Vet automated system (Orphée, Geneva, Switzerland), with reagents and controls sourced from the same supplier. Subsequently, May-Grunwald Giemsa staining was performed to evaluate the blood smear, utilizing a Nikon Eclipse E100 optical microscope. Biochemistry and protein electrophoresisSerum biochemistry Serum biochemistry analysis was conducted using the CB350i automated equipment (Wiener Lab Group, Rosario, Argentina). This analysis utilized commercial reagents and controls obtained from the same supplier. The following parameters were measured: glycemia, triglycerides, alanine aminotransferase, aspartate aminotransferase, serum alkaline phosphatase, total protein, albumin, globulins, total cholesterol, High density lipoprotein (HDL) cholesterol, Low density lipoprotein (LDL) cholesterol, total bilirubin, urea, and creatinine. C-reactive protein (CRP) levels were determined using a canine-specific dry-chemistry assay (FUJI DRI CHEM SLIDE vc- CRP, Fujifilm, Japan). Protein electrophoresis was performed with an automated capillary electrophoresis instrument (Minicap, Sebia, Barcelona, Spain). For all metabolites measured, the CV for the controls used were less than 10%. UrinalysisThe chemical properties of urine were assessed using Human Combina s11 test strips, following the manufacturer’s instructions. For the evaluation of physical properties, urine specific gravity (USG) was measured using a refractometer. Additionally, turbidity and color were assessed. To obtain the urinary sediment, the sample was centrifuged at 2,000 rpm for 5 minutes, and the sediment was examined using a Nikon Eclipse optical microscope. ProxiesThe Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) and Homeostasis Model Assessment of β-cell Function (HOMA-β) indices were calculated using the HOMA calculator software version 2.2.3 (Oxford University). Statistical analysisNormality was assessed using the Kolmogorov- Smirnov test through the PROC UNIVARIATE procedure in the Statistical Analysis System (SAS). Non-normally distributed data were log-transformed for further analysis. The mixed procedure (SAS Studio, Version 3.8, Enterprise Edition) was used for data analysis, incorporating the body condition group as a fixed effect. Age category, sex, and gonadal status were examined as covariates and retained in the model if they were statistically significant. The weight of the study population was adjusted according to the distance from the ground to the scapular-humeral joint. Sex proportions were analyzed using a chi-square test. Associations between variables were evaluated using Pearson’s correlation coefficient, employing the CORR procedure (PROC CORR, SAS). A p-value of less than 0.05 was considered statistically significant, while p-values ranging from 0.05 to 0.10 were regarded as indicative of a trend. Ethical approvalThe experimental methodology was approved by the ethics committee (CHEA file number 953/19). ResultsStudy populationRegarding age, the means of the NW, OW, and OB groups were 6.3 ± 0.5, 6.9 ± 0.5, and 6.6 ± 0.6 years, respectively, with no differences between groups. The proportion of male/female in the NW group was 12/21, while that of the OW group was 10/18, and that of the OB group was 11/12, with no differences between the proportions of each group (p = 0.6). Most of the dogs (53.5%, n = 45) were mixed breed, followed by Golden Retriever (10.6%, n = 9), Labrador Retriever (9.5%, n = 8), German Shepherd and French Bull Dog (4.8%, n = 4 each), Yorkshire Terrier, Rottweiler, Poodle and Beagle (2.4%, n = 2 each), Pitbull, Pug, Border Collie, Basset Hound, Saint Bernard and Boxer (1.2%, n = 1 each). Regarding weight, which was adjusted according to the size (distance from the ground to the scapular-humeral joint), the NW group presented an average of 17.7 ± 1.6 kg, while that of the OW group was 27.6 ± 1.8 and that of the OB group was 36.9 ± 1.8 kg, all groups being different with each other (p < 0.0001). Blood pressure and CFThe OB group had higher SBP (159.9 ± 5.2 mmHg) compared to OW (144.1 ± 4.8 mmHg, p = 0.045) and NW (145.3 ± 4.8 mmHg, p = 0.030). MBP was also higher in the OB group (114.9 ± 3.5 mmHg) than OW (103.8 ± 3.3 mmHg, p = 0.03) and tended to be higher in NW (106.2 ± 3.3 mmHg, p = 0.09). DBP and CF were not affected by BCS. Adipokines, cytokines, thyroid hormones, and basal cortisolLeptin serum concentrations increased with BCS (p < 0.0001; Fig. 1A). Adiponectin was also affected by BCS (p = 0.01), being lower in the OB group compared to OW and NW (p = 0.04 and p = 0.005, respectively) (Fig. 1B). Serum resistin was higher in the OB group compared to the NW (p = 0.02). The OW group tended to have higher levels of serum resistin than the NW (p=0.08), while no differences were observed between the OW and OB groups (p = 0.60) (Fig. 1C).
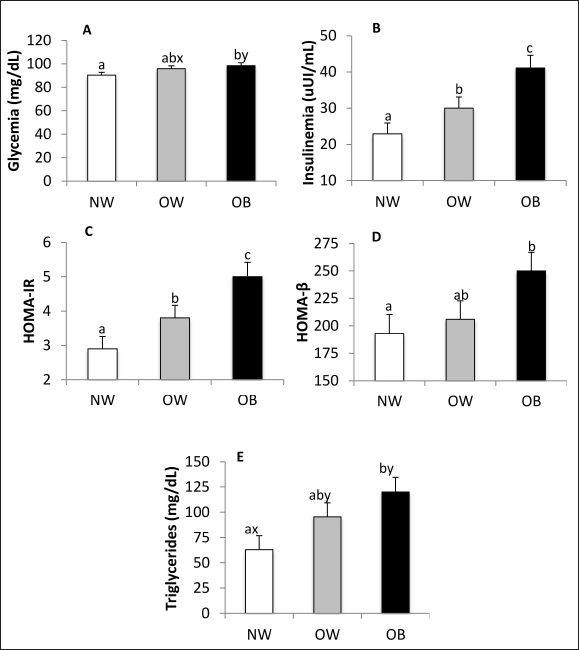
Fig. 1. Serum leptin (A), adiponectin (B), and resistin (C) concentrations in NW, BCS 4–5, OW, BCS 6–7, and OB, BCS 8–9 dogs. a versus b p < 0.05. x versus y p ≤ 0.1. The concentrations of TNF-α and IL-6 in most samples were below the sensitivity of the tests used in this study (7.8 and 31.3 pg/ml, respectively). Total T4, Free T4, TSH, and basal cortisol were not affected by BCS (data not shown). Glucose and lipid metabolismBlood glucose was higher (p = 0.02) and tended to be higher (p = 0.10) in the OB group compared to NW and OW, respectively (Fig. 2A). Insulinemia was affected by body condition (p < 0.0001), being almost two times higher in OB patients compared to NW patients (p < 0.0001, Fig. 2B). OW patients had higher insulin concentrations than NW patients (p = 0.02) and lower insulin concentrations than OB animals (p = 0.02). The HOMA-IR index increased according to BCS (p < 0.0001, Fig. 2C), being higher in OB and OW than in NW canines (p < 0.0001 and p = 0.02, respectively). OW dogs had lower HOMA-IR index values than OB dogs (p = 0.02). The HOMA-β index was higher in OB patients compared to NW patients (p = 0.03, Fig. 2D). The concentration of serum triglycerides was affected by BCS (p = 0.02), being almost two-fold greater in OB animals than in NW animals (p = 0.005, Fig. 2E). OW dogs tended to have higher concentrations than NW (p < 0.10) but did not differ from OB canines. Serum total cholesterol, HDL cholesterol, LDL cholesterol, and HDL/LDL ratio were not affected by BCS.
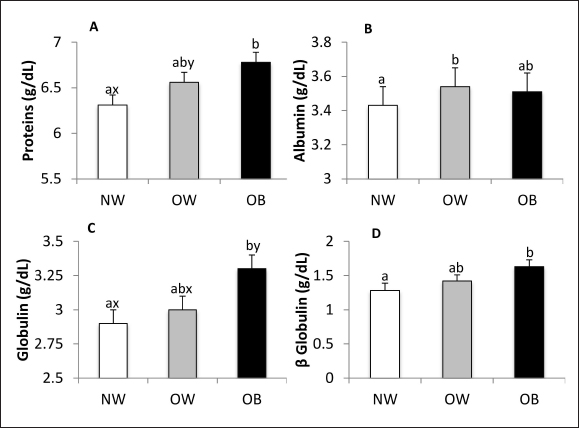
Fig. 2. Glucose (A) and insulin (B) concentrations, HOMA-IR (C) and HOMA-β (D), and triglycerides (E) concentrations in NW, BCS 4–5, OW, BCS 6–7, and OB, BCS 8–9 dogs. a versus b p < 0.05. x versus y p ≤ 0.1. Biochemistry and urinalysisSerum total protein concentration was affected by BCS (p = 0.01) (Fig. 3A), being higher in the OB group when compared to the NW group (p = 0.0027) and tending to be higher in OW canines than in NW canines (p = 0.09). Albumin tended to be affected by BCS (p = 0.07) (Fig. 3B), since its concentration was higher in OW canines in relation to the NW group (p = 0.03). Serum globulin concentration was affected by BCS (p = 0.02) (Fig. 3C), being higher in OB canines compared to the group of NW animals (p = 0.006) and tending to be higher in the group of OB canines compared to the OW group (p = 0.09). According to serum electrophoresis, beta globulins tended to be affected by BCS (p = 0.055), being higher in the OB group compared to the NW (p = 0.02) (Fig. 3D). No other protein fraction was affected (p > 0.1).
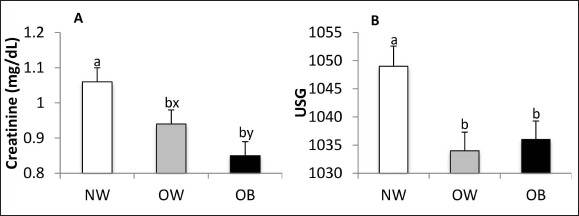
Fig. 3. Total protein (A), albumin (B), globulin (C), and beta-globulin (D) concentrations in NW, BCS 4–5, OW, BCS 6–7, and OB, BCS 8–9 dogs. a versus b p < 0.05. x versus y p ≤ 0.1. The serum concentration of hepatic enzymes, bilirubin, and CRP were not affected by BCS. While serum urea concentration was not affected by BCS, creatinine concentration was (p = 0.001). Creatinine concentrations were higher in the NW group compared to the OB (p = 0.0003) and OW groups (p=0.03) and tended to be lower in OB canines than in OW canines (p = 0.09) (Fig. 4A). USG was affected by BCS (p = 0.006), being higher in the NW group than in the OW and OB groups (p = 0.003 and p = 0.009, respectively) (Fig. 4B). Urinary pH and proteinuria were not influenced by BCS.
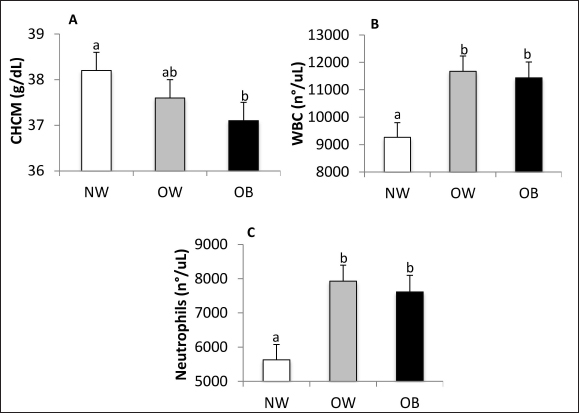
Fig. 4. Serum creatinine concentration (A) and USG (B) NW, BCS 4–5, OW, BCS 6–7, and OB, BCS 8–9 dogs. a versus b p < 0.05. x versus y p ≤ 0.1. HematologyBCS did not affect erythrocyte count, hemoglobin, hematocrit, mean corpuscular volume, or mean corpuscular hemoglobin. The mean corpuscular hemoglobin concentration (MCHC) was lower in the OB group when compared to NW (p = 0.03; Fig. 5A). Total leukocyte count was affected by BCS (p = 0.003) (Fig. 5B), being higher in OW and OB animals compared to NW (p = 0.02 and p = 0.006, respectively), with no differences between them. Neutrophils were affected by BCS (Fig. 5C), being higher in OW and OB patients when compared with NW canines (p = 0.0003 and p = 0.002, respectively). Band neutrophils, lymphocytes, monocytes, and eosinophils were not affected by BCS.
Fig. 5. MCHC (A), total leukocyte count (B), and neutrophil count (C) in NW, BCS 4–5, OW, BCS 6–7, and OB, BCS 8–9 dogs. a versus b p < 0.05. x versus y p ≤ 0.1. CHCM,MCHC. WBC, white blood cells. CorrelationsSerum insulin was positively correlated with serum triglycerides, cholesterol, and CRP (r2 = 0.73, r2 = 0.56, and r2 = 0.51, respectively, p < 0.0003 for all), and serum triglycerides were correlated with serum cholesterol (r2 = 0.81, p < 0.0001). DiscussionIn the cardiovascular evaluation, OB dogs exhibited higher SBP compared to NW and OW dogs. This finding aligns with previous studies conducted on dogs (Tebaldi et al., 2012; José Lahm et al., 2016; Piantedosi et al., 2016). However, other researchers have reported no significant difference in SBP based on BCS (Tropf et al., 2017; de Marchi et al., 2020; Piantedosi et al., 2020). In human and murine models, activation of the renin-angiotensin-aldosterone system has been observed in OB individuals, contributing to arterial hypertension (Boustany et al., 2004; Engeli et al., 2005). Additionally, leptin has been shown to stimulate the ventromedial hypothalamus in mice, which increases sympathetic activity and levels of catecholamines, potentially leading to an increase in SBP in OB animals (Satoh et al., 1999). Furthermore, insulin resistance and hyperinsulinemia associated with obesity have also been linked to elevated SBP (López et al., 2014). Leptin serum concentration increases with BCS, which aligns with several studies conducted on dogs (Jeusette et al., 2005; Ishioka et al., 2007; Lee et al., 2014; Park et al., 2014, 2015; Piantedosi et al., 2016; Palatucci et al., 2018; Piantedosi et al., 2020). In contrast, adiponectin is inversely associated with BCS, consistent with findings from several researchers (Ishioka et al., 2006; Eirmann et al., 2009; Lee et al., 2014; Park et al., 2015; Piantedosi et al., 2016; Tropf et al., 2017). Regarding resistin, there are only two published reports about this adipokine in dogs, which contradict our findings by indicating that BCS does not affect serum resistin concentration (Eirmann et al., 2009; Kleine et al., 2020). However, studies in mice and humans support our findings, showing that serum resistin levels are higher in OB individuals (Patel et al., 2003; Adeghate, 2004). Resistin influences carbohydrate metabolism and may promote diabetes by inducing insulin resistance through type 4 toll-like receptor activation (Kim et al., 2007; Tarkowski et al., 2010). In our study, the observed higher concentrations of leptin and resistin, along with lower levels of adiponectin in OB dogs, could contribute to insulin resistance. Indeed, the data on adipokines correlates with the higher serum insulin concentrations and HOMA indexes found in OB dogs, reflecting the metabolic effort to maintain glucose levels within reference ranges. Additionally, insulin resistance may explain the increased serum triglyceride concentrations observed in OB dogs, which has been previously documented by other researchers (José Lahm et al., 2016; Piantedosi et al., 2016; Tropf et al., 2017; Cihan and Tural, 2019; de Marchi et al., 2020; Safadi et al., 2021). The underlying mechanism may involve the downregulation of endothelial lipoprotein lipase, leading to decreased triglyceride clearance (Brown and Goldstein, 2008), and/or a form of selective liver insulin resistance where insulin fails to downregulate gluconeogenesis while continuing to stimulate fatty acid synthesis (Shimomura et al., 2000). A higher BCS was associated with an increased concentration of serum total proteins, primarily due to a rise in serum globulins, particularly β globulins. Previous studies have reported increases in total serum proteins and globulins in OB dogs (Piantedosi et al., 2016; Safadi et al., 2021; Vieira et al., 2022). Two research reports examined the electrophoretic protein profile of globulins in OB dogs and found that the observed differences were mainly due to the α fraction, specifically the α2 globulins (Piantedosi et al., 2016; Piantedosi et al., 2020). In contrast, studies in human medicine have noted activation of the complement system in OB individuals, which is evident from higher concentrations of C3 and C3b proteins (Oberbach et al., 2011; Al Haj Ahmad and Al-Domi, 2017; Shim et al., 2020). In dogs, these complement proteins are categorized within the β globulin fraction (Kaneko et al., 2008), which may explain the increase in this fraction observed in OB dogs in our study. Regarding renal function, serum creatinine levels decrease as BCS increases. Our findings align with those of Barić Rafaj et al. (2017), while other studies, such as those by Piantedosi et al. (2016), Bosco et al. (2018), Forster et al. (2018), Pongkan et al. (2020), Safadi et al. (2021), and Vieira et al. (2022), reported no significant effects of BCS on serum creatinine concentration. Since serum creatinine levels are associated with lean body mass, we suggest that the lower serum creatinine levels observed in the OB group may be explained by a loss of lean body mass, particularly muscle mass. In fact, muscle mass loss observed in humans with insulin resistance is attributed to ectopic fat deposits in skeletal muscle and the secretion of associated cytokines, which adversely affect muscle mass and function (Meex et al., 2019). Sarcopenic obesity in humans has been linked to insulin resistance in both men and women (Kim et al., 2013). Another possible explanation for our findings is that OB individuals tend to have higher creatinine clearance, both calculated and estimated, as reported in human studies (Tobar et al., 2013). Although still within normal ranges, lower urinary density (USG) was observed in OB and OW dogs compared to those of NW. We found no observational reports regarding the relationship between BCS and urinary density in dogs. In canines with induced obesity, an increase in glomerular filtration rate has been described (Henegar et al., 2001), which supports our findings. Overall, the data regarding USG and serum creatinine, along with serum globulin levels, suggest that even within reference values, there are functional alterations in organs and tissues due to obesity. If these alterations persist over time, they may lead to a loss of homeostasis. Research on CRP levels in canines is limited, and the findings are inconsistent. Veiga et al. (2008) reported that OB dogs have lower blood CRP levels compared to lean dogs while Barić Rafaj et al. (2017) identified significantly higher plasma CRP levels in OW and OB dogs compared to those with an ideal BCS. Besides, Tvarijonaviciute et al. (2012) found no change in CRP levels in canines following a weight loss treatment while Wakshlag et al. (2011) observed a decrease in CRP levels after a weight reduction program. In terms of hematology, MCHC was found to be lower in the OB group compared to the NW group. This observation aligns with findings in humans, where an increase in pelvic circumference was linked to a decrease in MCHC (Vuong et al., 2014). Additionally, total leukocyte counts and absolute neutrophil counts were higher in the OW and OB groups compared to the NW group. However, scientific literature on these variables presents mixed results. While several authors did not find significant differences in leukocyte counts (Barić Rafaj et al., 2017; Forster et al., 2018; Safadi et al., 2021; Vieira et al., 2022), Barbosa et al. (2019) reported a lower total leukocyte count in OB patients, with a notable decrease in lymphocytes. Furthermore, although Forster et al. (2018) and Vieira et al. (2022) concluded that there were no significant differences in total leukocyte counts, they did note a tendency for higher leukocyte counts in OB canines. Studies in humans and mice have shown that bone marrow stem cells possess leptin receptors, which directly stimulate the formation of granulocyte-monocyte colonies, the precursor cells for neutrophils (Purdy and Shatzel, 2021). Therefore, the increased leptin levels observed in OB dogs may promote granulopoiesis, resulting in elevated neutrophil and total leukocyte counts. ConclusionOB and OW canines present marked changes in carbohydrate and lipid metabolites that reflect a deterioration in insulin sensitivity as body condition increases and a compensatory hyperinsulinemia. Moreover, novel results associated with obesity are the alterations of serum resistin, the β globulin fraction, creatinine, urinary density, and neutrophils that, even within normal ranges, suggest alterations of homeostasis. AcknowledgmentsThe authors express gratitude to the “Universidad de la República” and the “Agencia Nacional de Investigación e Innovación.” Conflict of interestThe authors declare that there is no conflict of interest. FundingThis work was supported by “Agencia Nacional de Investigación e Innovación” POS_ NAC_2019_1_158146. Authors’ contributionsAdrián Carzoli: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Visualization, Writing—original draft, Ana Meikle: Conceptualization, Methodology, Validation, Writing-review and editing, Supervision, Funding acquisition. Paula Pessina: Conceptualization, Methodology, Validation, Resources, Writing-review and editing, Supervision, Project administration, Funding acquisition. Data availabilityThe data supporting this study’s findings are sensitive and not openly available. They can be obtained from the corresponding author upon reasonable request. ReferencesAcierno, M. J., Brown, S., Coleman, A. E., Jepson, R. E., Papich, M., Stepien, R. L., & Syme, H. M. 2018. ACVIM consensus statement: Guidelines for the identification, evaluation, and management of systemic hypertension in dogs and cats. Journal of Veterinary Internal Medicine, 32(6), 1803–1822. Adeghate, E. 2004. An update on the biology and physiology of resistin. Cell. Mol. Life Sci. 61, 2485–2496. Ahmad, R.M.A.H. and Al-Domi, H.A. 2017. Complement 3 serum levels as a pro-inflammatory biomarker for insulin resistance in obesity. Diabetes Metab. Syndr. 11, 229–232. Barbosa, A.D.A., Martins, N.F., Rosário, S.A., da Silva Nunes, P.C., Passarelli, D. and Leite-Dellova, D.C.A. 2019. Evaluation of coagulation parameters in dogs with overweight or obesity. Acta Sci. Vet. 47, 1–8. Barić Rafaj, R., Kuleš, J., Marinculić, A., Tvarijonaviciute, A., Ceron, J., Mihaljević, Ž., Tumpa, A. and Mrljak, V. 2017. Plasma markers of inflammation and hemostatic and endothelial activity in naturally overweight and obese dogs. BMC Vet. Res. 13, 1–7. Barić Rafaj, R., Kuleš, J., Turković, V., Rebselj, B., Mrljak, V. and Kučer, N. 2016. Prospective hematologic and biochemical evaluation of spontaneously overweight and obese dogs. Vet. Arh. 86, 383–394. Bosco, A.M., Almeida, B.F.M., Valadares, T.C., Baptistiolli, L., Hoffmann, D.J., Pereira, A.A.F., Lima, V.M.F. and Ciarlini, P.C. 2018. Preactivation of neutrophils and systemic oxidative stress in dogs with hyperleptinemia. Vet. Immunol. Immunopathol. 202, 18–24. Boustany, C.M., Bharadwaj, K., Daugherty, A., Brown, D.R., Randall, D.C. and Cassis, L.A. 2004. Activation of the systemic and adipose renin-angiotensin system in rats with diet-induced obesity and hypertension. Am. J. Physiol. Regul. Integr. Comp. Physiol. 287, 943–949. Brown, M.S. and Goldstein, J.L. 2008. Selective versus total insulin resistance: a pathogenic paradox. Cell Metab. 7(2), 95–96. Cihan, H. and Tural, M. 2019. Assessment of asymmetric dimethyl arginine, cardiac troponin I, thyroxine, cholesterol, and triglyceride levels in obese dogs and dogs with normal body condition. Turk. J. Vet. Anim. Sci. 43(2), 271–275. Courcier, E.A., Thomson, R.M., Mellor, D.J. and Yam, P.S. 2010. An epidemiological study of environmental factors associated with canine obesity. J. Small Anim. Pract. 51, 362–367. De Marchi, P.N., de Araújo Machado, L.H., Holsback, L., Ragazzi, J.C., Fagnani, R., Zacarias Junior, A., Zahn, F.S. and Lahm Cardoso, M.J. 2020. Metabolic profile and adipokine levels in overweight and obese dogs. Turk. J. Vet. Anim. Sci. 44, 1093–1099. de Oliveira Leal, V. and Mafra, D. 2013. Adipokines in obesity. Clin. Chim. Acta 419, 87–94. Eirmann, L.A., Freeman, L.M., Laflamme, D.P., Michel, K.E. and Satyaraj, E. 2009. Comparison of adipokine concentrations and markers of inflammation in obese versus lean dogs. Intern. J. Appl. Res. Vet. Med. 7(4), 196–205. Engeli, S., Böhnke, J., Gorzelniak, K., Janke, J., Schling, P., Bader, M., Luft, F.C. and Sharma, A.M. 2005. Weight loss and the renin-angiotensin-aldosterone system. Hypertension 45, 356–362. Forster, G.M., Stockman, J., Noyes, N., Heuberger, A.L., Broeckling, C.D., Bantle, C.M. and Ryan, E.P. 2018. A comparative study of serum biochemistry, metabolome and microbiome parameters of clinically healthy, normal weight, overweight, and obese companion dogs. Top. Companion Anim. Med. 33(4), 126–135. Henegar, J.R., Bigler, S.A., Henegar, L.K., Tyagi, S.C. and Hall, J.E. 2001. Functional and structural changes in the kidney in the early stages of obesity. J. Am. Soc. Nephrol. 12, 1211–1217. Ishioka, K., Hosoya, K., Kitagawa, H., Shibata, H., Honjoh, T., Kimura, K. and Saito, M. 2007. Plasma leptin concentration in dogs: effects of body condition score, age, gender and breeds. Res. Vet. Sci. 82, 11–15. Ishioka, K., Omachi, A., Sagawa, M., Shibata, H., Honjoh, T., Kimura, K. and Saito, M. 2006. Canine adiponectin: cDNA structure, mRNA expression in adipose tissues and reduced plasma levels in obesity. Res. Vet. Sci. 80, 127–132. Jeusette, I.C., Lhoest, E.T., Istasse, L.P. and Diez, M.O. 2005. Influence of obesity on plasma lipid and lipoprotein concentrations in dogs. Am. J. Vet. Res. 66, 81–86. José Lahm Cardoso, M., Fagnani, R., Zaghi Cavalcante, C., de Souza Zanutto, M., Júnior, A.Z., Holsback da Silveira Fertonani, L., Calesso, J.R., Melussi, M., Pinheiro Costa, H. and Yudi Hashizume, E. 2016. Blood pressure, serum glucose, cholesterol, and triglycerides in dogs with different body scores. Vet. Med. Int. 2016(1), 8675283. Kaneko, J.J., Harvey, J.J. and Bruss, M.L. 2008. Clinical biochemistry of domestic animals, San Diego, California. Academic Press, p: 273. Kim, F., Pham, M., Luttrell, I., Bannerman, D.D., Tupper, J., Thaler, J., Hawn, T.R., Raines, E.W. and Schwartz, M.W. 2007. Toll-like receptor-4 mediates vascular inflammation and insulin resistance in diet-induced obesity. Circ. Res. 100, 1589–1596. Kim, T.N., Park, M.S., Lim, K.I., Choi, H.Y., Yang, S.J., Yoo, H.J., Kang, H.J., Song, W., Choi, H., Baik, S.H. and Choi, D.S. 2013. Relationships between sarcopenic obesity and insulin resistance, inflammation, and vitamin D status: the Korean sarcopenic obesity study. Clin. Endocrinol. 78, 525–532. Kleine, S.A., Gogal, R.M., Krunkosky, T.M., Sanderson, S.L., George, C., Norton, M. and Budsberg, S.C. 2020. Resistin concentrations in serum and stifle synovial fluid from normal and cruciate deficient dogs with osteoarthritis. Vet. Surg. 49, 1164–1173. Laflamme, D. P. 1997. Development and Validation of a Body Condition Score System for Dogs Canine Practice, 22(1), 10–15. Lee, S.H., Lim, S.J., Park, H.J. and Song, K.H. 2014. Leptin, adiponectin levels, and thyroid hormones in normal and obese dogs. Korean J. Vet. Res. 54, 165–169. López, M.S., Maskin de Jensen, A.N., Mir, C.N., Ibañez de Pianesi, M.E. and Manulak, M.A. 2014. Adiposidad abdominal, insulino-resistencia, hipertensión y sodio urinarioen adolescentes obesos. Acta Bioquím. Clín. Latinoam. 48, 301–310. Lund, E.M., Armstrong, P.J., Kirk, C.A. and Klausner, J.S. 2006. Prevalence and risk factors for obesity in adult dogs from private US veterinary practices. Intern. J. Appl. Res. Vet. Med. 4(2), 177–186. Mao, J., Xia, Z., Chen, J. and Yu, J. 2013. Prevalence and risk factors for canine obesity surveyed in veterinary practices in Beijing, China. Prev. Vet. Med. 112(3–4), 438–442. McGreevy, P.D., Thomson, P.C., Pride, C., Fawcett, A., Grassi, T. and Jones, B. 2005. Prevalence of obesity in dogs examined by Australian veterinary practices and the risk factors involved. Vet. Rec. 156, 695–702. Meex, R.C., Blaak, E.E. and van Loon, L.J. 2019. Lipotoxicity plays a key role in the development of both insulin resistance and muscle atrophy in patients with type 2 diabetes. Obes. Rev. 20, 1205–1217. Montoya-Alonso, J.A., Bautista-Castaño, I., Peña, C., Suárez, L., Juste, M.C. and Tvarijonaviciute, A. 2017. Prevalence of canine obesity, obesity-related metabolic dysfunction, and relationship with owner obesity in an obesogenic region of Spain. Front. Vet. Sci. 4, 2–5. Mori, N., Takemitsu, H., Okada, Y., Yamamoto, I. and Arai, T. 2013. A comparison of metabolic parameters between obese and non-obese healthy domestic dogs in Japan. Asian J. Anim. Vet. Adv. 8, 863–873. Muñoz-Prieto, A., Martínez-Subiela, S., Caldin, M., Cerón, J.J. and Tvarijonaviciute, A. 2020. Use of proteases for the evaluation of the different adiponectin isoforms in the dog. Domest. Anim. Endocrinol. 70, 106380. Muñoz-Prieto, A., Martínez-Subiela, S., Cerón, J.J. and Tvarijonaviciute, A. 2019. A new highly sensitive immunoassay for the detection of adiponectin in serum and saliva of dogs and its application in obesity and canine leishmaniosis. Res. Vet. Sci. 125, 374–381. Oberbach, A., Blüher, M., Wirth, H., Till, H., Kovacs, P., Kullnick, Y., Schlichting, N., Tomm, J.M., Rolle-Kampczyk, U., Murugaiyan, J. and Binder, H. 2011. Combined proteomic and metabolomic profiling of serum reveals association of the complement system with obesity and identifies novel markers of body fat mass changes. J. Proteome Res. 10, 4769–4788. Palatucci, A.T., Piantedosi, D., Rubino, V., Giovazzino, A., Guccione, J., Pernice, V., Ruggiero, G., Cortese, L. and Terrazzano, G. 2018. Circulating regulatory T cells (Treg), leptin and induction of proinflammatory activity in obese Labrador Retriever dogs. Vet. Immunol. Immunopathol. 202, 122–129. Park, H.J., Lee, S.E., Kim, H.B., Isaacson, R.E., Seo, K.W. and Song, K.H. 2015. Association of obesity with serum leptin, adiponectin, and serotonin and gut microflora in beagle dogs. J. Vet. Intern. Med. 29, 43–50. Park, H.J., Lee, S.E., Oh, J.H., Seo, K.W. and Song, K.H. 2014. Leptin, adiponectin and serotonin levels in lean and obese dogs. BMC Vet. Res. 10, 1–8. Patel, L., Buckels, A.C., Kinghorn, I.J., Murdock, P.R., Holbrook, J.D., Plumpton, C., Macphee, C.H. and Smith, S.A. 2003. Resistin is expressed in human macrophages and directly regulated by PPAR γ activators. Biochem. Biophys. Res. Commun. 300(2), 472–476. Pérez-Sánchez, A.P., Del-Angel-Caraza, J., Quijano-Hernández, I.A. and Barbosa-Mireles, M.A. 2015. Obesity-hypertension and its relation to other diseases in dogs. Vet. Res. Commun. 39, 45–51. Piantedosi, D., Di Loria, A., Guccione, J., De Rosa, A., Fabbri, S., Cortese, L., Carta, S. and Ciaramella, P. 2016. Serum biochemistry profile, inflammatory cytokines, adipokines and cardiovascular findings in obese dogs. Vet. J. 216, 72–78. Piantedosi, D., Palatucci, A.T., Giovazzino, A., Ruggiero, G., Rubino, V., Musco, N., Carriero, F., Farina, F., Attia, Y.A.E.W., Terrazzano, G. and Lombardi, P. 2020. Effect of a weight loss program on biochemical and immunological profile, serum leptin levels and cardiovascular parameters in obese dogs. Front. Vet. Sci. 7, 398. Pongkan, W., Jitnapakarn, W., Phetnoi, W., Punyapornwithaya, V. and Boonyapakorn, C. 2020. Obesity-Induced heart rate variability impairment and decreased systolic function in obese male dogs. Animals 10, 1383. Porsani, M.Y.H., Teixeira, F.A., Oliveira, V.V., Pedrinelli, V., Dias, R.A., German, A.J. and Brunetto, M.A. 2020. Prevalence of canine obesity in the city of São Paulo, Brazil. Sci. Rep. 10(1), 14082. Purdy, J.C. and Shatzel, J.J. 2021. The hematologic consequences of obesity. Eur. J. Haematol. 106(3), 306–319. Radin, M.J., Sharkey, L.C. and Holycross, B.J. 2009. Adipokines: a review of biological and analytical principles and an update in dogs, cats, and horses. Vet. Clin. Pathol. 38, 136–156. Ramos, J.R. and Castillo, V. 2020. Evaluation of insulin resistance in overweight and obese dogs. Int. J. Vet. Sci. Res. 6(1), 058–063. Safadi, D.M., Falbo, M.K., Sandini, I.E. and Stremel, H.F. 2021. Preventive blood evaluation in obese dogs (Canis lupus familiares). Acta Vet. Bras. 15, 41–45. Satoh, N., Ogawa, Y., Katsuura, G., Numata, Y., Tsuji, T., Hayase, M., Ebihara, K., Masuzaki, H., Hosoda, K., Yoshimasa, Y. and Nakao, K. 1999. Sympathetic activation of leptin via the ventromedial hypothalamus leptin-induced increase in catecholamine secretion. Diabetes 48, 1787–1793. Shim, K., Begum, R., Yang, C. and Wang, H., 2020. Complement activation in obesity, insulin resistance, and type 2 diabetes mellitus. World J. Diabetes 11, 1–12. Shimomura, I., Matsuda, M., Hammer, R.E., Bashmakov, Y., Brown, M.S. and Goldstein, J.L. 2000. Decreased IRS-2 and increased SREBP-1c lead to mixed insulin resistance and sensitivity in livers of lipodystrophic and ob/ob mice. Mol. Cell. 6(1), 77–86. Steppan, C.M., Bailey, S.T., Bhat, S., Brown, E.J., Banerjee, R.R., Wright, C.M., Patel, H.R., Ahima, R.S. and Lazar, M.A. 2001. The hormone resistin links obesity to diabetes. Nature 409, 307–312. Tarkowski, A., Bjersing, J., Shestakov, A. and Bokarewa, M.I. 2010. Introduction resistin competes with lipopolysaccharide for binding to toll-like receptor 4. J. Cell. Mol. Med. 14, 1419–1431. Tebaldi, M., Lourenço, M.L.G., Machado, L.H.A., Sudano, M.J. and Carvalho, L.R. 2012. Estudo da pressão arterial pelo método indireto oscilométrico (petmap) em cães domésticos não anestesiados. Arq. Bras. Med. Vet. Zootec. 64, 1456–1464. Tobar, A., Ori, Y., Benchetrit, S., Milo, G., Herman-Edelstein, M., Zingerman, B., Lev, N., Gafter, U. and Chagnac, A. 2013. Proximal tubular hypertrophy and enlarged glomerular and proximal tubular urinary space in obese subjects with proteinuria. PLOS one 8(9), e75547. Tropf, M., Nelson, O.L., Lee, P.M. and Weng, H.Y. 2017. Cardiac and metabolic variables in obese dogs. J. Vet. Intern. Med. 31, 1000–1007. Tvarijonaviciute, A., Ceron, J.J., Holden, S.L., Cuthbertson, D.J., Biourge, V., Morris, P.J. and German, A.J. 2012. Obesity-related metabolic dysfunction in dogs: a comparison with human metabolic syndrome. BMC Vet. Res. 8, 1–8. Tvarijonaviciute, A., Martínez-Subiela, S., & Cerón, J. J. 2010. Validation of 2 commercially available enzyme-linked immunosorbent assays for adiponectin determination in canine serum samples. The Canadian Journal of Veterinary Research, 74, 279–285. Usui, S., Yasuda, H. and Koketsu, Y. 2016. Characteristics of obese or overweight dogs visiting private Japanese veterinary clinics. Asian Pac. J. Trop. Biomed. 6(4), 338–343. Veiga, A.P., Price, C.A., de Oliveira, S.T., Dos Santos, A.P., Campos, R., Barbosa, P.R. and Gonzalez, F.H. 2008. Association of canine obesity with reduced serum levels of C-reactive protein. J. Vet. Diagn. Invest. 20, 224–228. Verkest, K.R., Rand, J.S., Fleeman, L.M., Morton, J.M., Richards, A.A., Rose, F.J. and Whitehead, J.P. 2011. Distinct adiponectin profiles might contribute to differences in susceptibility to type 2 diabetes in dogs and humans. Domest. Anim. Endocrinol. 41, 67–73. Vieira, A.B., Restrepo, M.A., Auzenne, D., Molina, K., O’Sullivan, M., Machado, M.V. and Cavanaugh, S.M. 2022. Mild to moderate overweight in dogs: is there an impact on routine hematological and biochemical profiles, echocardiographic parameters and cardiac autonomic modulation? Vet. Res. Commun. 46, 527–535. Vuong, J., Qiu, Y., La, M., Clarke, G., Swinkels, D.W. and Cembrowski, G. 2014. Reference intervals of complete blood count constituents are highly correlated to waist circumference: Should obese patients have their own “normal values?” Am. J. Hematol. 89, 671–677. Wakshlag, J.J., Struble, A.M., Levine, C.B., Bushey, J.J., Laflamme, D.P. and Long, G.M. 2011. The effect of weight loss on adipokines and markers of inflammation in dogs. Br. J. Nutr. 106, S11–S14. Zoran, D.L. 2010. Obesity in dogs and cats: a metabolic and endocrine disorder. Vet. Clin. North Am. Small Anim. Pract. 40(2), 221–239. | ||
| How to Cite this Article |
| Pubmed Style Carzoli A, Meikle A, Pessina P. Canine obesity, overweight, and adipokine serum concentration are associated with hematological, biochemical, hormonal, and cardiovascular markers. Open Vet. J.. 2025; 15(2): 619-629. doi:10.5455/OVJ.2025.v15.i2.10 Web Style Carzoli A, Meikle A, Pessina P. Canine obesity, overweight, and adipokine serum concentration are associated with hematological, biochemical, hormonal, and cardiovascular markers. https://www.openveterinaryjournal.com/?mno=217363 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.10 AMA (American Medical Association) Style Carzoli A, Meikle A, Pessina P. Canine obesity, overweight, and adipokine serum concentration are associated with hematological, biochemical, hormonal, and cardiovascular markers. Open Vet. J.. 2025; 15(2): 619-629. doi:10.5455/OVJ.2025.v15.i2.10 Vancouver/ICMJE Style Carzoli A, Meikle A, Pessina P. Canine obesity, overweight, and adipokine serum concentration are associated with hematological, biochemical, hormonal, and cardiovascular markers. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 619-629. doi:10.5455/OVJ.2025.v15.i2.10 Harvard Style Carzoli, A., Meikle, . A. & Pessina, . P. (2025) Canine obesity, overweight, and adipokine serum concentration are associated with hematological, biochemical, hormonal, and cardiovascular markers. Open Vet. J., 15 (2), 619-629. doi:10.5455/OVJ.2025.v15.i2.10 Turabian Style Carzoli, Adrián, Ana Meikle, and Paula Pessina. 2025. Canine obesity, overweight, and adipokine serum concentration are associated with hematological, biochemical, hormonal, and cardiovascular markers. Open Veterinary Journal, 15 (2), 619-629. doi:10.5455/OVJ.2025.v15.i2.10 Chicago Style Carzoli, Adrián, Ana Meikle, and Paula Pessina. "Canine obesity, overweight, and adipokine serum concentration are associated with hematological, biochemical, hormonal, and cardiovascular markers." Open Veterinary Journal 15 (2025), 619-629. doi:10.5455/OVJ.2025.v15.i2.10 MLA (The Modern Language Association) Style Carzoli, Adrián, Ana Meikle, and Paula Pessina. "Canine obesity, overweight, and adipokine serum concentration are associated with hematological, biochemical, hormonal, and cardiovascular markers." Open Veterinary Journal 15.2 (2025), 619-629. Print. doi:10.5455/OVJ.2025.v15.i2.10 APA (American Psychological Association) Style Carzoli, A., Meikle, . A. & Pessina, . P. (2025) Canine obesity, overweight, and adipokine serum concentration are associated with hematological, biochemical, hormonal, and cardiovascular markers. Open Veterinary Journal, 15 (2), 619-629. doi:10.5455/OVJ.2025.v15.i2.10 |