| Research Article | ||
Open Vet. J.. 2025; 15(2): 630-639 Open Veterinary Journal, (2025), Vol. 15(2): 630-639 Research Article Analysis of CA1, CA3, and DG areas of the hippocampus, substance-P, and brain-derived neurotrophic factors expression in the presence of Ocimum sanctum Linn on the brain of the rat model Alzheimer’s diseaseMade Bagus Auriva Mataram1,2, Ulayatul Kustiati3, Hevi Wihadmadyatami4, Dwi Aris Agung Nugrahaningsih5, Siti Isrina Oktavia Salasia6 and Dwi Liliek Kusindarta4*1Post-graduate School of Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia 2Laboratory of Clinical Pathology, Faculty of Veterinary Medicine, Brawijaya University, Malang, Indonesia 3Laboratory of Pharmacology, Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia 4Department of Anatomy, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia 5Department of Pharmacology and Therapy, Faculty of Medicine, Public Health and Nursing, Universitas Gadjah Mada, Yogyakarta, Indonesia 6Department of Clinical Pathology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia *Correspondence to: Dwi Liliek Kusindarta. Department of Anatomy, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia. indarta [at] ugm.ac.id Submitted: 19/9/2024 Accepted: 12/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
AbstractBackground: Alzheimer’s disease (AD) is a progressive neurological condition characterized by impaired cognitive dysfunction and abnormal behavior. Thirty-five million individuals worldwide suffer from dementia, making it the most frequent cause of dementia. Canine cognitive dysfunction (CCD) affects 28% of dogs aged 11–12 years. It is a prevalent disease in canines. Amyloid-β peptides accumulate neurotoxicity, resulting in problems in the central nervous system and neurotransmitters. Treatments for both AD and CCD have not yet shown acceptable outcomes. It is crucial to comprehend disease mechanisms and identify novel therapeutics using animal models. Aim: Using a rat model of AD, this study attempted to determine the effects of an ethanolic extract from Ocimum sanctum on the number and shape of neurons in the CA1, CA3, and DG sections of the hippocampus along with the expression of neurotrophic factors. Methods: The animal model will be split into five treatment groups, one of which will be a control group. The treatments will be administered for 14, 21, and 28 days, and samples will be analyzed by BDNF by ELISA, SP expression by immunohistochemical staining, and the number of neurons in CA1, CA3, and DG using cresyl violet staining. Results: Results of the study revealed increased neuronal density in the CA1, CA3, and DG regions, and these neurons were more highly expressed in the neurotrophic factor BDNF and neuropeptide SP. Conclusion: By upregulating the expression of SP and BDNF, the ethanolic extract of O. sanctum increased the neuronal counts (pyramidal and granular cells) in the hippocampal CA1, CA3, and DG regions. Keywords: AD, Hippocampus, Ocimum sanctum, Rat model IntroductionAlzheimer’s disease (AD) is a neurological condition that worsens over time and is marked by progressive loss of cognitive skills, including memory, attention, reasoning, and language. In a clinical setting, patients may exhibit disorientation, paranoia, hallucinations, and sundowning. Patients with AD frequently present with psychological symptoms such as sadness, anxiety, and psychosis, in addition to cognitive and behavioral symptoms. According to epidemiological research, AD is the most prevalent cause of dementia worldwide. Globally, there are already 35 million dementia patients, and in the next 20 years, this number is predicted to increase (Apetz et al., 2014). In contrast, canine AD or canine cognitive dysfunction (CCD), frequently referred to as canine cognitive impairment, also affects canines. Confusion, disorientation, amnesia, increased barking, mood swings, fear, loss of training, pacing, or looking into space are all signs of CCD in dogs. There are quite a few cases of pets. Specifically, CCD affects 28% of animals aged between 11 and 12 years. Neurotoxicity caused by amyloid β (Aβ) peptides, leading to changes in the morphological structure of the central nervous system and accompanied by neurotransmitter abnormalities, contributes to AD and CCD. Although the processes that lead to AD modify cognitive and behavioral functions are now well understood, few treatment methods have been effectively transferred into the clinical phase to prevent or ameliorate AD progression (Bray et al., 2023). Research in this area is essential. Blocking Aβ buildup boosts the expression of cholinergic and neurotrophic neurotransmission expression and averts oxidative stress and neuroinflammation. Recent advancements in the therapy and medication of AD have focused on various innovative approaches, particularly targeting Aβ and tau proteins, which are central to the disease’s pathology. One of the most notable developments is the use of monoclonal antibodies, such as lacenemab and gantenerumab, which have shown promise in clinical trials for slowing cognitive decline in patients with early-stage AD. Lecanemab, a humanized IgG1 monoclonal antibody, was evaluated in a phase 3 trial and demonstrated significant efficacy in reducing Aβ plaques in the brain because it correlates with cognitive improvements in patients with early AD (Dyck et al., 2023). This study showed that lacenemab targets amyloid pathology and has the potential to modify the disease course, making it a significant advancement in AD treatment options. Similarly, gantenerumab, another anti-Aβ monoclonal antibody, has been tested in two phase 3 trials, showing potential disease-modifying effects by reducing amyloid burden and improving cognitive function. These results can be observed in patients with mild cognitive impairment or mild dementia due to AD (Bateman et al., 2022; Bateman et al., 2023). However, several weaknesses and challenges have emerged regarding the efficacy and safety of these therapies. One of the primary concerns is the limited clinical efficacy observed in many trials. For instance, although antibodies such as aducanumab and lacenemab have demonstrated some ability to reduce amyloid plaque burden, the translation of these biological effects into meaningful clinical benefits remains contentious. Studies have shown that despite reductions in amyloid levels, improvements in cognitive function are often modest or absent (Dyck, 2018; Horosan et al., 2021). In addition, gantenerumab, despite showing some effects on cerebrospinal fluid (CSF) biomarkers, failed to demonstrate significant clinical efficacy in phase 3 trials (Tolar et al., 2020). Recent advancements in the therapy ofAD have explored various pharmacological and nonpharmacological approaches, particularly neuroprotective agents and innovative treatment modalities. This synthesis highlights several promising therapies, including natural products, neurostimulation techniques, and cognitive interventions. Natural products such as herbal medicines have emerged as a promising approach for the treatment of AD, leveraging the multi-targeting and synergistic properties of various plant-derived compounds. Several studies have highlighted the potential of specific herbal remedies to mitigate cognitive decline associated with AD. For instance, Ginkgo biloba has been extensively studied for its neuroprotective effects. It is believed to enhance cognitive function by improving blood circulation in the brain and exhibiting antioxidant properties (Singh et al., 2019). Additionally, Ginkgo biloba inhibits acetylcholinesterase, an enzyme that breaks down acetylcholine, thereby potentially improving cholinergic transmission in the brain (Perry & Howes, 2010). Another notable herbal remedy is huperzine A, derived from the Huperzia serrata. This compound has demonstrated significant effects in improving memory and cognitive function in animal models and clinical trials. Huperzine A acts as a reversible inhibitor of acetylcholinesterase, similar to donepezil, and has shown promise in enhancing memory retention and learning abilities (Ye et al., 2015). Moreover, Ocimum sanctum is one of the medicinal plants that are being used in herbal plant treatment in Southeast and South Asia, and it has become one of the most promising complementary therapy approaches (Nandini et al., 2022; Beheshti et al., 2023). However, for the prevention and therapy of AD, further research and proof of the therapeutic mechanisms are still needed. Animal models serve as a crucial connection between preclinical studies and the clinical phase to acquire a more profound understanding of the mechanisms of AD and CCD, as well as the therapeutic advantages of recently identified medications. This study employed an AD model rat approach to investigate the impact of O. sanctum ethanolic extract (OSE) on the expression of neurotransmitters (substance P-SP), and neurotrophic factors such as brain-derived neurotrophic factors (BDNF), as well as the neuronal count and neuron morphology in the cornu ammonis (CA1), cornu ammonis (CA3), and dentate gyrus (DG) regions of the hippocampus, as the centers of memory and learning. Materials and MethodsEthical learanceThe in vivo studies were conducted with the authorization of the Ethics Committee of the Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia, under reference number 062/ EC-FKH/Eks./2023. Preparation OSEO. sanctum leaves were purchased from Herbal Materia Medica Batu Laboratory, an Indonesian herbal company located in East Java. Three hundred grams of OSE were macerated in four liters of 96% ethanol (Merck, Darmstadt, Germany) to create the extract, which was then filtered. By compressing the extract with a vacuum rotary evaporator (Heidolph, Schwabach, Germany) and lowering the pressure to 60°C, the extract was concentrated. Approximately 8.82% of the weight corresponds to the measured final OSE yield. Preparation of an animal model of ADIn this research project, 60 male Sprague Dawley rats with weights ranging from 200 to 250 g were acquired. All the animals were provided by the Integrated Laboratory for Research and Testing, Universitas Gadjah Mada, Yogyakarta, Indonesia. The rats were maintained in separate cages with limitless access to water and food, a 12-hour day/night period, temperature (23°C–25°C), and relative humidity of 55%–70%. Trimethyltin (TMT, 8.0 mg/kg, Sigma, St Louis, MO, USA) was administered intraperitoneally (i.p.). The injection was diluted in physiological saline solutions. Following TMT induction, the rats exhibited specific behavioral symptoms for the AD model, including heightened assertiveness, hyperirritability, tremors, aggression, self-harm, and hyperactivity (Almasi et al., 2024). Consequently, they were kept in a single cage. Experimental designFive treatment groups were assigned to the rats: group A (n = 12) was administered saline (1 milliliter per 100 grams of body weight; group B received an induced TMT (Sigma, Steinheim, Germany); group C (n = 12) received pre-induced TMT and donepezil HCl (Novell, Jakarta, Indonesia); group D (n = 12) received pre- induced TMT and OSE; and group E (n = 12) initiated OSE at a dosage of 100 mg/kg BW orally for one week, then administered TMT and continued with OSE after induction. TMT was administered in a single dose of 8 mg/kg BW intraperitoneally (i.p.) route injection, donepezil at 0.5 mg/kg BW, and post-induced OSE at 100 mg/kg BW for 28 days peroral route. The perfusion dates were 14, 21, and 28 d. Brain tissue samples were collected, paraffin-blocked, and prepared for cresyl violet staining and substance P immunohistochemistry analysis after euthanasia. Hippocampus tissue processingEach rat received intramuscular injections of 2% and 10% ketamine hydrochloride (Ilium Ketamil Injection, New South Wales, Australia) and xylazine (Interchemie, Metaalweg, The Netherlands) before the experiment was terminated by cervical dislocation. To separate the cerebral cortex and cerebellum from the skull, the brainstem and optic chiasm were first severed. The right brain tissues were embedded in paraffin, fixed in 10% neutral-buffered formalin (Leica, Richmond, USA), and a microtome was used to slice the tissue block into 4 μm thick. The left hippocampus tissues were preserved in RIPA lysis buffer (Thermoscientific, Rockford, USA) and maintained at –80 °C. Cresyl violet stainThe solution containing cresyl violet (Sigma, Steinheim, Germany) was incubated at 37°C. After placing the slides within a rack, they were stained with cresyl violet for 30 minutes. The slides were dried, mounted, and coated with balsam Canada (Merck, Darmstadt, Germany) after being cleaned for a minute with distilled water. A 40× magnification light microscope (Nikon, Tokyo, Japan) was used to observe hippocampal growth. The data were analyzed using Optilab Viewer (Optilab, Yogyakarta, Indonesia). Using Optilab Image Raster software, the quantity of pyramidal and granular cells (72.351,97 μm2) inside the hippocampus region was determined. Immunohistochemical analysis of SPThe hippocampus tissue was immersed in hot salinetris–buffered saline (TBS) at pH 9 after deparaffinization with xylol and rehydration. Phosphate-buffered saline (PBS) with pH 7.4 and 0.01 M was used to wash the tissue. The tissue was then blocked with 3% H2O2 at room temperature for 20 minutes. Following a 10- minutes rinse with PBS, the samples were exposed to mouse anti-SP primary antibody (diluted at a ratio of 1:2000, Abcam, Cambridge, USA) for 1 hour at room temperature. Novolink Polymer Detection Systems (Leica, Newcastle, United Kingdom) were used for immunoreactivity analysis. For counterstaining slides, Harris hematoxylin-eosin was provided by Leica (Wetzlar, Germany. A light microscope (Olympus, Tokyo, Japan) was used to observe SP-immunoreactive cells’ development in 40× magnification. Optilab image raster software (Optilab, Yogyakarta, Indonesia) was used for comprehensive cell counting in five fields of view to count the number of SP-immunoreactive cells in the hippocampal region (72.351,97 μm2). The average cell count was calculated using SP- immunoreactive cell counts. Enzyme-linked immunosorbent assay of BDNFTo completely eliminate extra blood, the hippocampal tissues were chopped into tiny sections and washed with ice-cold PBS (0.01 M, pH=7.4). The tissue sections were weighed and then homogenized with PBS using a glass homogenizer kept on ice [tissue weight (g): PBS (ml) volume=1:9]. An ultrasonic cell disruptor is used to further elaborate on the cells. The homogenates were centrifuged for 5–10 minutes at 5,000 × g at 2°C–8°C to obtain the supernatant. Endogenous BDNF was quantified using the Sandwich enzyme- linked immunosorbent assay kit (Elbscience, Hubei, China). All materials and reagents were prepared at room temperature (18°C–25°C). Subsequently, fill the corresponding wells with 100 µl of the standard and sample. After covering the wells, incubate for 1.5 at 37°C. After removing the liquid from each well, add 100 µl of the biotinylated detection antibody to each well immediately. Shake vigorously and let the mixture stand at room temperature for an hour. Using an automaker or multichannel pipette, add 350 µl of wash buffer and discard the solution. After soaking for 1 minute, remove the solution by aspirating or decanting it from each well, and then tap dry using fresh water. The washing procedure was carried out three times. Incorporate 100 µl of HRP. A functional solution is assigned to each well. At 37°C, incubate for 30 minutes while vigorously shaking. After discarding the solution, repeat the five wash cycles. Fill each well with 90 µl of the substrate reagent. Shake well and incubate for 15 minutes at 37°C in the dark. Fill each well with 50 µl of Stop Solution. Reading at 450 nm is the last stage. Data analysisThe statistical analysis used for the obtained data was a two-way analysis of variance (ANOVA) followed by Dunnett’s post hoc test. When required, Tukey’s post hoc test can be used, followed by statistical analysis of the in vitro data using one-way ANOVA. Probability (p) values less than 0.05 were considered statistically significant. Every statistical analysis was performed using GraphPad Prism 9 (GraphPad, Inc., La Jolla, CA, USA). ResultsOSE increased neuron proliferation in the CA1, CA3, and DG areas of the hippocampus of AD model ratsCresyl violet staining was applied to brain tissue cells after tissue processing. This staining indicates that in the AD rat models with TMT induction, there is a significantly decreased number of pyramidal cells on the stratum pyramidale of the CA1 and CA3 areas. In contrast, the administration of OSE before TMT induction and after TMT induction showed a significantly increasing number of pyramidal cells on the stratum pyramidale of the CA1 and CA3 areas (Fig. 1 and 2) and granular cells on the dentate gyrus area (Fig. 3), similar to donepezil administration.
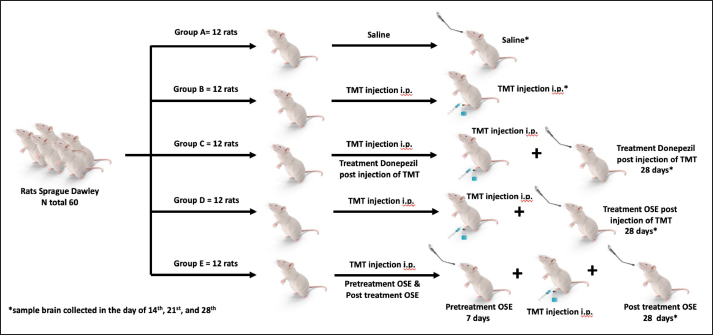
Fig. 1. Schematic diagram of the experimental design. Sixty Sprague–Dawley rats were used in the study and divided into five groups, namely group A, which was given saline solution. Group B was injected using TMT; group C was given a TMT injection (8 mg/kg BW) followed by therapy with donepezil (0.5 mg/kg BW); group D was given a TMT injection (8 mg/kg BW) followed by therapy with ethanolic extract of Ocimum sanctum Linn. (OSE) (100 mg/kg BW); and the last group, group D, was given pretreatment with OSE (100 mg/kg BW) before TMT induction (8 mg/kg BW) and continued with posttreatment with OSE (100 mg/kg BW). All treatments were provided for 28 days, and brain samples were collected on days 14, 21, and 28.
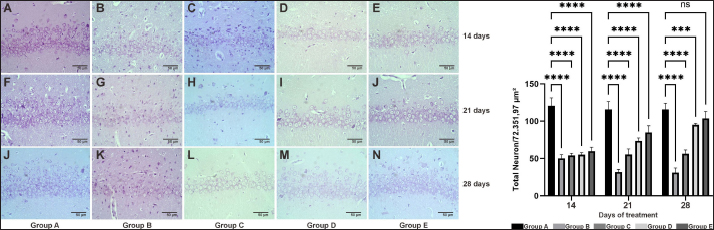
Fig. 2. The density of pyramidal cell neurons in the CA1 hippocampal of the AD rat model was measured on days 14, 21, and 28 using cresyl violet staining. Group A was observed in images A (14), F (21), and J (28); group B was observed in images B (14), G (21), and K (28); group C was observed in images C (14), H (21), and L (28); group D was observed in images D (14), I (21), and M (28); and group E was observed in images E (14), J (21), and N (28). The figure illustrates the quantification of pyramidal cells (72.351,97 μm2) in the CA1 hippocampus region. ***/****: statistically significant (p < 0.05); n.s: not statistically significant.
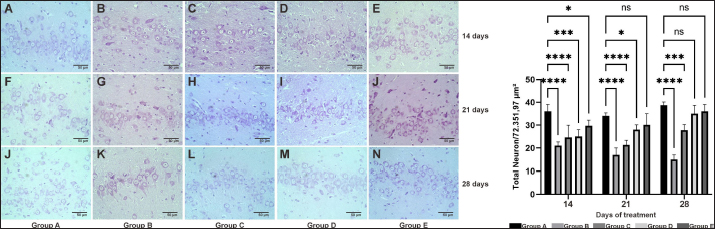
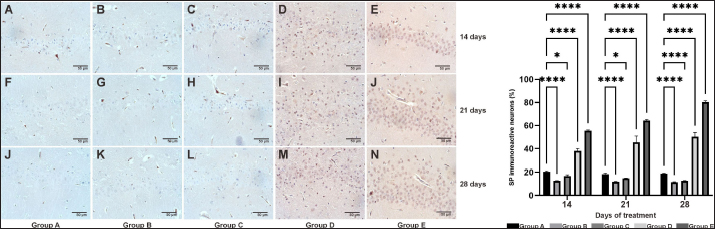
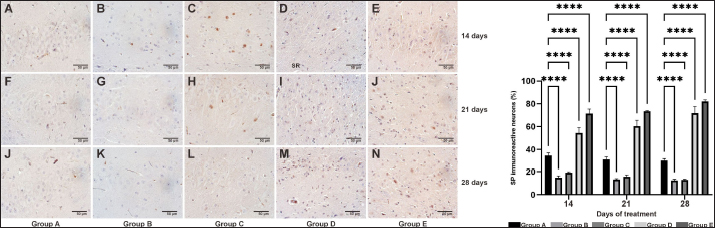
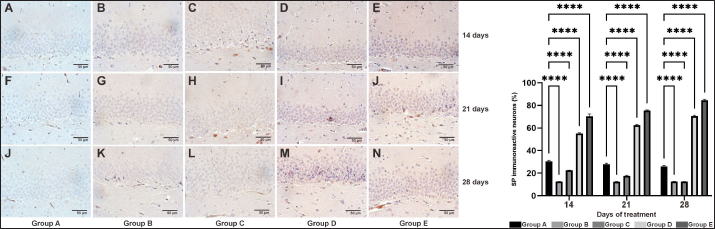
Fig. 3. The density of pyramidal cell neurons in the CA3 hippocampal of the AD rat model was measured on days 14, 21, and 28 using cresyl violet staining. Group A was observed in images A (14), F (21), and J (28); group B was observed in images B (14), G (21), and K (28); group C was observed in images C (14), H (21), and L (28); group D was observed in images D (14), I (21), and M (28); and group E was observed in images E (14), J (21), and N (28). The figure illustrates the quantification of pyramidal cells (72.351,97 μm2) in the CA3 hippocampus region. Significance level: ****/****: statistically significant (p < 0.05); n.s: not statistically significant. OSE increased substance P expression in the brains of AD model ratsThe staining results indicated that in contrast to the TMT group, there was significantly increased cell immunoreactivity against substance P on the stratum pyramidale of the CA1 and CA3 areas (Fig. 4 and 5) and granular cells on the dentate gyrus area (Fig. 6). This was the case both before and after induction of TMT as well as in the rat model of AD treated with donepezil. Immunohistochemistry was used to evaluate the concentration of SP in young and middle-aged rats following treatment.
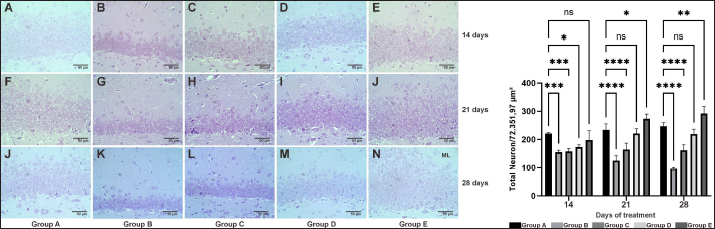
Fig. 4. The density of granular cell neurons in the DG hippocampal of the AD rat model was measured on days 14, 21, and 28 using cresyl violet staining. Group A was observed in images A (14), F (21), and J (28); group B was observed in images B (14), G (21), and K (28); group C was observed in images C (14), H (21), and L (28); group D was observed in images D (14), I (21), and M (28); and group E was observed in images E (14), J (21), and N (28). The figure illustrates the quantification of granular cells (72.351,97 μm2) in the DG hippocampus region. */**/***/****: statistically significant (p < 0.05); n.s: not statistically significant.
Fig. 5. Immunohistochemical labeling revealed the total percentage of SP-immunoreactive cells in the CA1 hippocampal region (72.351,97 μm2) of AD rat model on days 14, 21, and 28. Group A was observed in images A (14), F (21), and J (28); group B was observed in images B (14), G (21), and K (28); group C was observed in images C (14), H (21), and L (28); group D was observed in images D (14), I (21), and M (28); and group E was observed in images E (14), J (21), and N (28). The picture shows the quantification of the general percentage of SP-immunoreactive cells in the CA1 region in AD rat models, calculated by totaling all cells. */****: statistically significant (p < 0.05).
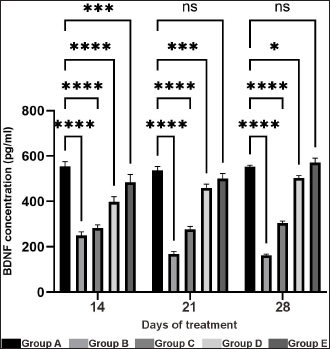
Fig. 6.Immunohistochemical labeling revealed the total percentage of SP-immunoreactive cells in the CA3 hippocampal region (72.351,97 μm2) of AD rat model on days 14, 21, and 28. Group A was observed in images A (14), F (21), and J (28); group B was observed in images B (14), G (21), and K (28); group C was observed in images C (14), H (21), and L (28); group D was observed in images D (14), I (21), and M (28); and group E was observed in images E (14), J (21), and N (28). The picture shows the quantification of the general percentage of SP-immunoreactive cells in the CA3 region in AD rat models, calculated totaling all cells. ****: statistically significant (p < 0.05). OSE increased BDNF expression in the brains of AD model ratsThe concentration of BDNF in the AD model rats in the presence of OSE was measured using ELISA. The ELISA results on day 28 showed that the rat model AD with OSE had a considerably higher concentration of BDNF (571 ± 8.04 pg/ml) before TMT induction and after treatment (503 ± 8.73 pg/ml), followed by the next lower concentration in the donepezil group levels (305 ± 6.98 pg/ml). Conversely, the AD animals treated with neither OSE nor donepezil had the lowest BDNF concentration (162 ± 4.55 pg/ml) (Fig. 7).
Fig. 7. Immunohistochemical labeling revealed the total percentage of SP-immunoreactive cells in the DG hippocampal region (72.351,97 μm2) of AD rat model on days 14, 21, and 28. Group A was observed in images A (14), F (21), and J (28); group B was observed in images B (14), G (21), and K (28); group C was observed in images C (14), H (21), and L (28); group D was observed in images D (14), I (21), and M (28); and group E was observed in images E (14), J (21), and N (28). The picture shows the quantification of the general percentage of SP-immunoreactive cells in the DG region in the AD rat models. ****: statistically significant (p < 0.05). DiscussionThe animal model is crucial in bridging the preclinical and clinical phases. In this study, we used male rats induction by TMT to develop an animal model of AD. The use of male rats in AD research is prevalent for several reasons, primarily related to the biological, behavioral, and experimental consistency of rats in research settings. Male rats are often chosen to minimize the variability caused by hormonal fluctuations that can occur in female rats due to their estrous cycle. Hormonal changes can significantly affect behavior, neurochemistry, and responses to treatments, potentially confounding results Almasi et al. (2024). Using male rats, we achieved more consistent and reproducible outcomes, which is crucial for understanding the pathophysiology of AD and evaluating therapeutic interventions. In addition, male rats have been effectively used to model the pathological characteristics of Aβ disease. Research indicates that Aβ-injected male rats develop senile plaques and exhibit cognitive deficits that are representative of early-onset AD (Jiang et al., 2020). These findings make them suitable for studying the mechanisms of plaque formation and the effects of potential therapeutic agents aimed at reducing Aβ levels. Furthermore, TMT is widely used to induce AD in animal models because of its specific neurotoxic effects that closely mimic the pathological features of the disease. TMT is an organotin compound that selectively induces neurodegeneration in specific brain regions, particularly the hippocampus, which is crucial for memory and learning. Studies have shown that TMT administration leads to significant neuronal cell loss and cognitive dysfunction, particularly affecting the CA1 and CA3 regions of the hippocampus (Park et al., 2019; Xi, 2024). TMT exposure results in behavioral changes that are analogous to those observed in AD patients, including impairments in spatial memory and learning abilities (Jung et al., 2013; Jeong et al., 2021). For instance, TMT-treated animals exhibit deficits in tasks such as the Morris water maze, which is commonly used to assess spatial learning and memory (Ye et al., 2020). TMT is known to induce oxidative stress and mitochondrial dysfunction, both of which are implicated in the pathogenesis of AD. The neurotoxic effects of TMT are associated with increased production of reactive oxygen species and subsequent neuronal apoptosis (Qu et al., 2011). TMT administration has been shown to lead to the accumulation of Aβ and hyperphosphorylated tau proteins, which are hallmark features of AD pathology (Jeong et al., 2021; Dragic et al., 2021). Finally, the clinical symptoms and neurobehavioral changes induced by TMT exposure in animal models closely resemble those observed in patients with AD, including memory loss, disorientation, and behavioral disturbances (Yu et al., 2016; Xi, 2024). According to our research, OSE treatment effectively acts as a neuroprotective agent in AD male rat models through the overexpression of SP and BDNF, which interact to cause neurogenesis in the hippocampus’s CA1, CA3, and DG regions. Substance P has been implicated in various cognitive processes, including learning and memory. Its role in cholinergic signaling is particularly noteworthy, as SP can modulate the activity of cholinergic neurons, which are crucial for cognitive function (Yi et al., 2020). SP has been shown to promote neuronal survival and protect against excitotoxicity, which is a significant mechanism of neuronal death in AD (Liu et al., 2022). In particular, the CA1, CA3, and DG regions of the hippocampus are crucial for memory formation and cognitive function. AD significantly decreases neurogenesis in the CA1, CA3, and DG areas, leading to cognitive decline. These hippocampal regions contribute differently to learning and memory functions, and recent research has shown that these regions are susceptible to neurodegenerative alterations linked to AD. The process that generates new neurons, or neurogenesis, is mostly observed in the DG region and is crucial for preserving the integrity of the hippocampus. Research has demonstrated that in patients with AD, neurogenesis is maintained to a modest extent in the DG, but there is a concerning decline in neurons in the CA1 and CA3 fields, which results in cognitive problems (Mu and Gage, 2011; Gomez-Nicola et al., 2014). The significance of the CA1 and CA3 regions in cognitive performance has been highlighted by the substantial correlation between neural degeneration in these regions and poor memory recall and encoding (Lazarov et al., 2010; Mu and Gage, 2011). Additionally, neurogenesis in the DG influences the functionality and connectivity of circuits in the CA1 and CA3 areas, which in turn influence the process of memory formation (Akers et al., 2014; Lazarov and Hollands, 2016). Since Aβ deposition is a pathology of AD, neuropeptides, including SP, have been shown to have neuroprotective effects in AD. These effects imply that neuropeptides protect synapses and memory, potentially preventing neurotoxicity (Johansson et al., 2015; Chen et al., 2018). The interaction between SP and other neuropeptides, such as (BDNF), is critical for the provision of neuroprotective effects (Chen et al., 2018). Prolyl oligopeptidase inhibition, for example, has been demonstrated to modify SP levels and affect cognitive performance in animal models; these findings raise the possibility of an AD treatment route (Jalkanen et al., 2007). The hippocampus’s neurogenesis and neuronal survival depend on the expression of neurotrophic factors such as BDNF. Decreased neurogenesis and cognitive function are caused by the frequently observed reduction in BDNF levels in AD (Perry et al., 2012; Lazarov and Hollands, 2016). According to Shin et al. (2019), BDNF has an impact on neurogenesis and the regulation of connections among the CA1, C3, and DG regions of the hippocampus. The involvement of BDNF in neurogenesis extends beyond the hippocampus to include the olfactory bulb area and the subventricular zone (SVZ) (Bath and Lee, 2010). The cholinergic system, particularly acetylcholine (ACh) activity, plays a role in the regulation of neurogenesis in the hippocampus. Elevated ACh levels are associated with increased pyramidal cell density in CA1 and CA3, indicating the potential neuroprotective function (Hening et al., 2018). This interaction highlights the possible therapeutic benefits of targeting cholinergic pathways to promote neurogenesis and cognitive function in AD. O. sanctum Linn, commonly known as Tulsi, has been the subject of research because of its neuroprotective properties. Research has shown that extracts from O. sanctum can stimulate the expression of choline acetyltransferase, which is essential for ACh synthesis, potentially enhancing cholinergic signaling and neurogenesis (Kusindarta et al., 2016). Additionally, O. sanctum has been found to have protective effects on neuronal cells, suggesting its potential as a therapeutic agent in reducing the neurodegenerative processes associated with AD (Kusindarta et al., 2016; Hening et al., 2018; Raditya et al., 2020; Mataram et al., 2021). The potential of natural compounds, such as O. sanctum, as neuroprotective agents highlights the importance of exploring these compounds in the context of neurodegenerative diseases, thereby inspiring further research and providing avenues for developing novel therapeutic strategies.
Fig. 8. The figure illustrates the BDNF concentration (pg/ ml) within the hippocampus of the rat Alzheimer’s disease model. */***/****: statistically significant (p < 0.05); n.s: not statistically significant. ConclusionIn conclusion, the ethanolic extract of O. sanctum increased the density of neurons (pyramidal and granular cells) in the CA1, CA3, and DG of the hippocampus through upregulation of SP and BDNF expression. AcknowledgmentThe authors would like to thank the Integrated Laboratory for Research and Testing, Universitas Gadjah Mada, for providing the facilities for this study. Conflict of interestThe authors declare that they have no conflicts of interest. FundingThis study was supported by grants from the Regular Fundamental Research from the Ministry of Education, Culture, Research, and Technology (grant number 2631/UN1/DITLIT/PT.01.03/2024) and the Matching Fund 2024 to Dwi Liliek Kusindarta. Author’s contributionConceptualization, H.W. and D.L.K.; Supervision, D.L.K., S.I.O.S., H.W., and D.A.A.N; Methodology, H.W. and D.L.K.; Software. M.B.A.M.; Validation, U.K. and M.B.A.M.; Formal analysis, D.L.K. and M.B.A.M.; Investigation, M.B.A.M and U.K.; Resources, D.L.K.; Data Curation, M.B.A.M.; Writing: Original Draft Preparation, Y D.L.K., S.I.O.S., H.W., D.A.A.N., and M.B.A.M; Writing: Review and Editing, H.W. and M.B.A.M; Visualization, M.B.A.M; Project Administration, U.K. and D.L.K.; and Funding Acquisition, D.L.K. The authors have reviewed and approved the published edition of this paper. ReferencesAkers, K.G., Martinez-Canabal, A., Restivo, L., Yiu, A.P., De Cristofaro, A., Hsiang, H.-L.L., Wheeler, A.L., Guskjolen, A., Niibori, Y., Shoji, H., Ohira, K., Richards, B.A., Miyakawa, T., Josselyn, S.A. and Frankland, P.W. 2014. Hippocampal neurogenesis regulates forgetting during adulthood and infancy. Science 344(6184), 598–602. Almasi, E., Heidarianpour, A. and Keshvari, M. 2024. The interactive effects of different exercises and hawthorn consumption on the pain threshold of TMT-induced Alzheimer male rats. J. Physiol. Sci. 74(36), 1–12. Apetz, N., Munch, G., Govindaraghavan, S. and Gyengesi, E. 2014. Natural compounds and plant extracts as therapeutic agents against chronic inflammation in Alzheimer’s disease--a translational perspective. CNS Neurol. Disord. Drug Targets. 13(7), 1175–1191. Bateman, R.J., Cummings, J.L., Schobel, S., Salloway, S., Vellas, B., Boada, M., Black, S.E., Blennow, K., Fountoura, P., Klein, G., Assunção, S. S., Smith, J. and Doody, R.S. 2022. Gantenerumab: an antiamyloid monoclonal antibody with potential disease-modifying effects in early Alzheimer’s disease. Alzheimer’s Res. Ther. 14(178), 1–17. Bateman, R.J., Smith, J., Donohue, M.C., Delmar, P., Abbas, R., Salloway, S., Wojtowicz, J., Blennow, K., Bittner, T., Black, S.E., Klein, G., Boada, M., Grimmer, T., Tamaoka, A., Perry, R.J., Turner, R.S., Watson, D., Woodward, M., Thanasopoulou, A., Lane, C., Baudler, M., Fox, N.C., Cummings, J.L., Fontoura, P. and Doody, R. S. 2023. Two phase 3 trials of gantenerumab in early Alzheimer’s disease. N. Engl. J. Med. 389(20), 1862–1876. Bath, K.G. and Lee, F.S., 2010. Neurotrophic factor control of adult SVZ neurogenesis. Dev. Neurobiol. 70(5), 339–349. Beheshti, F., Vakilian, A., Navari, M., Moghaddam, M., Dinpanah, H. and Ahmadi-Soleimani, S. 2023. Effects of Ocimum basilicum L. extract on hippocampal oxidative stress, inflammation, and BDNF expression in amnesic aged rats. Exp. Aging Res. 50(4), 1–16. Bray, E.E., Raichlen, D.A., Forsyth, K.K., Promislow, D.E.L., Alexander, G.E. and MacLean, E.L. 2023. Associations between physical activity and cognitive dysfunction in older companion dogs: results from the dog aging project. GeroScience 45(2), 645–661. Chen, X.-Y., Du, Y.-F. and Chen, L. 2018. Neuropeptideexert neuroprotective effects against in Alzheimer disease. Front. Mol. Neurosci. 11, 493. Dragic, M., Mitrović, N., Adžić, M., Nedeljković, N., and Grković, I., 2021. Microglial- and astrocyte-specific expression of purinergic signaling components and inflammatory mediators in the rat hippocampus during trimethyltin-induced neurodegeneration. ASN Neuro. 13, 1–18. Dyck, C.H.V. 2018. Anti-amyloid-β monoclonal antibodies for Alzheimer’s disease: pitfalls and promise. Biol. Psychiatry 83(4), 311–319. Dyck, C.H.V., Swanson, C.J., Aisen, P.S., Bateman, R.J., Chen, C., Gee, M., Kanekiyo, M., Li, D., Reyderman, L., Cohen, S., Frolich, L., Katayama, S., Sabbagh, M., Vellas, B., Watson, D., Dhadda, S., Irizarry, M., Kramer, L.D. and Iwatsubo, T. 2023. Lecanemab in early alzheimer’s disease. NEJM 388(1), 9–21. Gomez-Nicola, D., Suzzi, S., Vargas-Caballero, M., Fransen, N.L., Al-Malki, H., Cebrian-Silla, A., Garcia-Verdugo, J.M., Riecken, K., Fehse, B. and Perry, V.H. 2014. Temporal dynamics of hippocampal neurogenesis in chronic neurodegeneration. Brain 137(8), 2312–2328. Hening, P., Mataram, M.B.A., Wijayanti, N., Kusindarta, D.L. and Wihadmadyatami, H. 2018. The neuroprotective effect of Ocimum sanctum Linn. ethanolic extract on human embryonic kidney-293 cells as an in vitro model of neurodegenerative disease. Vet. World. 11(9), 1237–1243. Horosan, L., Nistor, D. and Zaharia, S. 2021. Monoclonal antibodies treatment for Alzheimer’s dementia–a literature review. RJPP 23(1), 18–22. Jalkanen, A.J., Puttonen, K.A., Venäläinen, J.I., Sinervä, V., Mannila, A., Ruotsalainen, S., Jarho, E.M., Wallén, E.A.A. and Männistö, P.T. 2007. The beneficial effect of prolyl oligopeptidase inhibition on spatial memory in young but not old scopolamine-treated rats. Basic Clin. Pharmacol. Toxicol. 100(2), 132–138. Jeong, E., Bajgai, J., You, I., Rahman, M.H., Fadriquela, A., Sharma, S., Kwon, H., Lee, S., Kim, C. and Lee, K. 2021. Therapeutic effects of hydrogen gas inhalation on trimethyltin-induced neurotoxicity and cognitive impairment in the c57bl/6 mice model. Int. J. Mol. Sci. 22(24), 1–19. Jiang, W., Yao, D., Fei, X., Ai, L., Di, Y., Zhang, J., Yue, X., Zhao, S., He, R., Lyu, J. and Tong, Z. 2020. Single-point mutation from e22 to k in aβ initiates early-onset alzheimer’s disease via binding with catalase. Oxid. Med. Cell. Longev. 2020(1), 1–21. Johansson, P., Almqvist, E.G., Wallin, A., Johansson, J.-O., Andreasson, U., Blennow, K., Zetterberg, H. and Svensson, J. 2015. Cerebrospinal fluid substance P concentrations are elevated in patients with Alzheimer’s disease. Neurosci. Lett. 609, 58–62. Jung, E., Lee, M., Ahn, C.J., Cho, S., Bae, H. and Shim, I. 2013. The neuroprotective effect of gugijihwang-tang on trimethyltin-induced memory dysfunction in the rat. eCAM 2013(1), 1–6. Kusindarta, D.D.L., Wihadmadyatami, H. and Haryanto, A. 2016. Ocimum sanctum Linn. stimulate the expression of choline acetyltransferase in human cerebral microvascular endothelial cells. World 9(12), 1348–1354. Lazarov, O. and Hollands, C. 2016. Hippocampal neurogenesis: learning to remember. Prog. Neurobiol. 138–140, 1–18. Lazarov, O., Mattson, M.P., Peterson, D.A., Pimplikar, S.W. and van Praag, H. 2010. When neurogenesis encounters aging and disease. Trends Neurosci. 33(12), 569–579. Liu, X., Ganguly, P., Jin, Y., Jhatro, M.J., Shea, J., Buratto, S.K. and Bowers, M.T. 2022. Tachykinin neuropeptides and amyloid β (25–35) assembly: friend or foe. JACS 144(32), 14614–14626. Mataram, M.B.A., Hening, P., Harjanti, F.N., Karnati, S., Wasityastuti, W., Nugrahaningsih, D.A.A., Kusindarta, D.L. and Wihadmadyatami, H. 2021. The neuroprotective effect of the ethanolic extract Ocimum sanctum Linn. in the regulation of neuronal density in hippocampus areas as a central autobiography memory in the rat model of Alzheimer’s disease. J. Chem. Neuroanat. 111, 101885. Mu, Y. and Gage, F.H. 2011. Adult hippocampal neurogenesis and its role in Alzheimer disease. Mol. Neurodegener. 6(1), 85. Nandini, H.H.S., Krishna, K.L. and Apattira, C. 2022. Combination of Ocimum sanctum extract and Levetiracetam ameliorates cognitive dysfunction and hippocampal architecture in rat model of Alzheimer’s disease. J. Chem. Neuroanat. 120, 102069. Park, S.K., Kang, J.Y., Kim, J.M., Yoo, S.K., Han, H.J., Chung, D., Kim, H., Kim, G. and Heo, H.J. 2019. Fucoidan-rich substances from ecklonia cava improve trimethyltin-induced cognitive dysfunction via downregulation of amyloid β production/tau hyperphosphorylation. Mar. Drugs 17(10), 1–23. Perry, E.E.K., Johnson, M., Ekonomou, A., Perry, R.R.H., Ballard, C. and Attems, J. 2012. Neurogenic abnormalities in Alzheimer disease differ between stages and are partly related to cholinergic pathology. Neurobiol. Dis. 47(2), 155–162. Perry, E.K. and Howes, M.R. 2010. Medicinal plants and dementia therapy: herbal benefits for brain aging?. CNS Neurosci. 17(6), 683–698. Qu, M., Zhou, Z., Chen, C., Li, M., Pei, L., Chu, F., Yang, J., Wang, Y., Li, L., Liu, C., Zhang, L., Zhang, G., Yu, Z. and Deng-gao, W. 2011. Lycopene protects against trimethyltin-induced neurotoxicity in primary cultured rat hippocampal neurons by inhibiting the mitochondrial apoptotic pathway. Neurochem. Int. 59(8), 1095–1103. Raditya, M.N., Bagus, A.M.M., Kustiati, U., Wihadmadyatami, H. and Kusindarta, D.L. 2020. Expression of Serotonin in an Alzheimer’s Disease (AD) rat model under treatment of ethanolic extract Ocimum sanctum Linn. Data Br. 30, 105654. Shin, J., Kong, C., Lee, J., Choi, B.Y., Sim, J., Koh, C.S., Park, M., Na, Y.C., Suh, S.W., Chang, W.S. and Chang, J.W. 2019. Focused ultrasound-induced blood-brain barrier opening improves adult hippocampal neurogenesis and cognitive function in a cholinergic degeneration dementia rat model. Alzheimer’s. Res. Ther. 11(1), 110. Singh, S.K., Srivastav, S., Castellani, R.J., Plascencia-Villa, G. and Perry, G. 2019. Neuroprotective and antioxidant effects of ginkgo biloba extract against ad and other neurological disorders. Neurotherapeutics 16(3), 666–674. Tolar, M., Abushakra, S., Hey, J.A., Porsteinsson, A.P. and Sabbagh, M.N. 2020. Aducanumab, gantenerumab, ban2401, and alz-801—the first wave of amyloid-targeting drugs for Alzheimer’s disease with potential for near term approval. Alzheimer’s Res. Ther. 12(1), 1–10. Xi, Y., Li, West, Wang, J., Yu, M., Zeng, X., Li, H. and Li, J. 2024. Cyanidin-3- o-glucoside alleviates trimethyltinchloride–induced neurodegeneration by maintaining glutamate homeostasis through modulation of the gut microbiota. Food Sci. Hum. Wellness 13(2), 1093–1107. Ye, M., Chung, H., An, Y., Lim, S.W., Choi, W.H., Yu, A.R., Kim, J.S., Kang, M., Cho. S., Shim, I. and Bae, H. 2015. Standardized herbal formula pm012 decreases cognitive impairment and promotes neurogenesis in the 3xtg ad mouse model of Alzheimer’s disease. Mol. Neurobiol. 53(8), 5401–5412. Ye, M., Han, B.H., Kim, J.S., Kim, K. and Shim, I. 2020. Neuroprotective effect of bean phosphatidylserine on tmt-induced memory deficits in a rat model. Int. J. Mol. Sci. 21(14), 1–13. Yi, J.H., Whitcomb, D.J., Park, S.J., Martinez-Perez, C., Barbati, S.A., Mitchell, S.J. and Cho, K. 2020. M1 muscarinic acetylcholine receptor dysfunction in moderate Alzheimer’s disease pathology. Brain Commun. 2(2), 1–13 Yu, J., Ding, D., Sun, H., Salvi, R. and Roth, J.A. 2016 Trimethyltin-induced cochlear degeneration in rat. J. Otol. 11(3), 118–126. | ||
| How to Cite this Article |
| Pubmed Style Mataram MBA, Kustiati U, Wihadmadyatami H, Nugrahaningsih DAA, Salasia SIO, Kusindarta DL. Analysis of CA1, CA3, and DG areas of the hippocampus, substance-P, and brain-derived neurotrophic factors expression in the presence of Ocimum sanctum Linn on the brain of the rat model Alzheimer’s disease. Open Vet. J.. 2025; 15(2): 630-639. doi:10.5455/OVJ.2025.v15.i2.11 Web Style Mataram MBA, Kustiati U, Wihadmadyatami H, Nugrahaningsih DAA, Salasia SIO, Kusindarta DL. Analysis of CA1, CA3, and DG areas of the hippocampus, substance-P, and brain-derived neurotrophic factors expression in the presence of Ocimum sanctum Linn on the brain of the rat model Alzheimer’s disease. https://www.openveterinaryjournal.com/?mno=221227 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.11 AMA (American Medical Association) Style Mataram MBA, Kustiati U, Wihadmadyatami H, Nugrahaningsih DAA, Salasia SIO, Kusindarta DL. Analysis of CA1, CA3, and DG areas of the hippocampus, substance-P, and brain-derived neurotrophic factors expression in the presence of Ocimum sanctum Linn on the brain of the rat model Alzheimer’s disease. Open Vet. J.. 2025; 15(2): 630-639. doi:10.5455/OVJ.2025.v15.i2.11 Vancouver/ICMJE Style Mataram MBA, Kustiati U, Wihadmadyatami H, Nugrahaningsih DAA, Salasia SIO, Kusindarta DL. Analysis of CA1, CA3, and DG areas of the hippocampus, substance-P, and brain-derived neurotrophic factors expression in the presence of Ocimum sanctum Linn on the brain of the rat model Alzheimer’s disease. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 630-639. doi:10.5455/OVJ.2025.v15.i2.11 Harvard Style Mataram, M. B. A., Kustiati, . U., Wihadmadyatami, . H., Nugrahaningsih, . D. A. A., Salasia, . S. I. O. & Kusindarta, . D. L. (2025) Analysis of CA1, CA3, and DG areas of the hippocampus, substance-P, and brain-derived neurotrophic factors expression in the presence of Ocimum sanctum Linn on the brain of the rat model Alzheimer’s disease. Open Vet. J., 15 (2), 630-639. doi:10.5455/OVJ.2025.v15.i2.11 Turabian Style Mataram, Made Bagus Auriva, Ulayatul Kustiati, Hevi Wihadmadyatami, Dwi Aris Agung Nugrahaningsih, Siti Isrina Oktavia Salasia, and Dwi Liliek Kusindarta. 2025. Analysis of CA1, CA3, and DG areas of the hippocampus, substance-P, and brain-derived neurotrophic factors expression in the presence of Ocimum sanctum Linn on the brain of the rat model Alzheimer’s disease. Open Veterinary Journal, 15 (2), 630-639. doi:10.5455/OVJ.2025.v15.i2.11 Chicago Style Mataram, Made Bagus Auriva, Ulayatul Kustiati, Hevi Wihadmadyatami, Dwi Aris Agung Nugrahaningsih, Siti Isrina Oktavia Salasia, and Dwi Liliek Kusindarta. "Analysis of CA1, CA3, and DG areas of the hippocampus, substance-P, and brain-derived neurotrophic factors expression in the presence of Ocimum sanctum Linn on the brain of the rat model Alzheimer’s disease." Open Veterinary Journal 15 (2025), 630-639. doi:10.5455/OVJ.2025.v15.i2.11 MLA (The Modern Language Association) Style Mataram, Made Bagus Auriva, Ulayatul Kustiati, Hevi Wihadmadyatami, Dwi Aris Agung Nugrahaningsih, Siti Isrina Oktavia Salasia, and Dwi Liliek Kusindarta. "Analysis of CA1, CA3, and DG areas of the hippocampus, substance-P, and brain-derived neurotrophic factors expression in the presence of Ocimum sanctum Linn on the brain of the rat model Alzheimer’s disease." Open Veterinary Journal 15.2 (2025), 630-639. Print. doi:10.5455/OVJ.2025.v15.i2.11 APA (American Psychological Association) Style Mataram, M. B. A., Kustiati, . U., Wihadmadyatami, . H., Nugrahaningsih, . D. A. A., Salasia, . S. I. O. & Kusindarta, . D. L. (2025) Analysis of CA1, CA3, and DG areas of the hippocampus, substance-P, and brain-derived neurotrophic factors expression in the presence of Ocimum sanctum Linn on the brain of the rat model Alzheimer’s disease. Open Veterinary Journal, 15 (2), 630-639. doi:10.5455/OVJ.2025.v15.i2.11 |