| Research Article | ||
Open Vet. J.. 2025; 15(2): 1043-1049 Open Veterinary Journal, (2025), Vol. 15(2): 1043-1049 Research Article Phylogenetic analysis and genetic evaluation of the Echinococcus granulosus antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah, IraqWeam A. Hamad, Thanaa Ismael Jawad* and Wegdan H. AtiyaNursing Techniques Department, Technical Institute of Al-Dewaniyah, AL-Furat AL-Awsat Technical University, Najaf, Iraq *Corresponding Author: Thanaa Ismael Jawad. Nursing Techniques Department, Technical Institute of Al-Dewaniyah, AL-Furat AL-Awsat Technical University, Iraq. Email: than.dw [at] atu.edu.iq Submitted: 25/10/2024 Accepted: 13/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
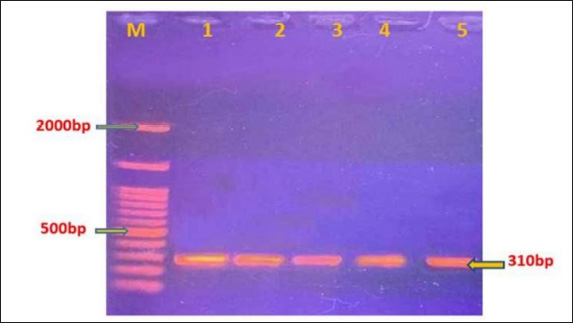
AbstractBackground: Hydatid cysts are an immature form of Echinococcus granulosus tapeworm, which causes zoonotic illness, echinococcosis (sometimes called hydatidosis). There are 10 officially recognized genotypes (G1-G10) of this parasite. Aim: The current study aimed to sequence and characterize the genotypes of E. granulosus using the antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah City, Iraq. Methods: Using specific antigen B subunit 2, 50 hydatid cysts were collected from the livers of sheep at a local slaughterhouse between March and October 2023. DNA was obtained from the hydatid fluid of sheep cysts. Protoscolices from fertile cyst fluid were also obtained. Conventional polymerase chain reaction (PCR) targeting the antigen B gene was performed on the extracted DNA of samples. PCR was performed followed by sequencing and phylogenetic analysis of PCR 7 positive isolates. Results: The predominant genotype in the sheep population was the G1 strain. Furthermore, the sequenced isolates exhibited a high degree of correlation with local and regional Iran E. granulosus sequences, demonstrating genetic diversity compared with reference strains. The current isolates were assigned accession numbers by the NCBI as follows: OR232318.1, OR232319.1, OR232320.1, OR232321.1, OR232322.1, OR232323.1, and OR232324.1. Conclusion: The findings indicate that E. granulosus has diverse genotypes and is mostly linked genetically to isolates from neighboring countries, such as Brazil and Iraq. Keywords: Echinococcus granulosus, Parasite, Sheep. IntroductionEchinococcosis, also known as hydatid disease, is a contagious disorder that results in a substantial burden of disability-adjusted life years (DALYs) globally on an annual basis (Agudelo Higuita et al., 2016). Echinococcosis is endemic to the entire Mediterranean region, including all countries in the Middle East (Andersen et al., 1997). Echinococcosis is an illness that arises from the larval form of the tapeworm E. granulosus, which is classified under the phylum Platyhelminthes. Echinococcosis can be transmitted to intermediate hosts, including domestic livestock such as sheep and goats (known as pastoral hydatidosis), as well as wild species such as moose, reindeer, and caribou (referred to as sylvan hydatidosis). Humans can contract E. granulosus infection by unintentionally consuming food or water contaminated with the eggs of the parasite. These eggs are excreted from the feces of infected carnivores. Humans are aberrant intermediate hosts of this parasite. The most common site for hydatid cysts to develop in humans is the liver, occurring in 50%–93% of cases (Ibrahim, 2010). The mature form of E. granulosus, known as the adult tapeworm stage, inhabits the canine intestines. When the eggs of this tapeworm are consumed by intermediate hosts (such as cattle, other livestock, rodents, deer, and humans), the eggs release embryos into the duodenum. Subsequently, these embryos are introduced into the portal circulation. A portion of the embryos become ensnared in the liver, while the rest traverse the liver and disseminate to other organs, where they mature as hydatid cysts (Langer et al., 1984). In cases of cystic echinococcosis (CE), the main organs affected were the liver (in 70%–75% of cases) and the lungs (in 10%–20% of cases). However, hydatid cysts can develop in any organ of the body (Weber et al., 2023). The mature worm is of diminutive size, measuring 3–6 mm in length and 0.5 mm in breadth, and possesses 3–4 proglottids. It is evident that there are various categories of E. granulosus, with variations in the physical characteristics of mature organisms, ability to infect specific hosts, and severity of disease. Distinct strains of E. granulosus have been found to precisely indicate their inclination toward specific intermediate hosts, such as sheep and buffalo, horses, cattle, pigs, camels, and cervids (Hüttner et al., 2008). The two principal forms of echinococcosis are cystic echinococcus (CE) and alveolar echinococcus (AE). Less common variants include polycystic and unicystic echinococcosis (Thompson, 2008). CE can develop in any organ or tissue of both humans and other intermediate host species, although the majority of infections are found in the liver, followed by the lungs as the second most common site (Gottstein and Reichen, 2002). Six species of Echinococcus tapeworms were identified: E. granulosus, E. vogeli, E. oligarthrus, E. shiquicus, and E. felidis. Echinococcosis (CE) caused by E. granulosus and AE caused by E. multilocularis are two species of great medical significance. (AE) (Brunette, 2012). The diagnosis of hydatid disease typically involves serological testing. Two common tests are indirect hemagglutination (IHA) and immunoelectrophoresis. The IHA test has a sensitivity of approximately 60% for calcified or lung lesions and 88% for peritoneal or liver disease, with a specificity of 90%–95% (Maharjan et al., 2018). During infection by the larval stage of the pathogen, secretory and membrane-bound (S/M) proteins play crucial roles in avoiding the host’s immune system. An antigen is a substance that stimulates an immune response in the body (Pan et al., 2011). The native Antigen B (AgB) is a thermostable lipoprotein with a relative molecular weight (Mr) of 160 kDa. Under reducing conditions, native AgB produces three subunits when analyzed by discontinuous gel electrophoresis (Maddison et al., 1989). Antigen B subunit 2 (AgB2) is a key E. granulosus secretory antigen used in the diagnosis of hydatid disease. AgB2 belongs to the AgB lipoprotein class, is highly immunoreactive, and is a key factor in the parasite’s immune escape, making it an important diagnostic tool. Its antigenicity triggers precise antibody responses in infected hosts (measured by serological tests such as ELISAs) and is highly sensitive and specific). Molecularly, AgB2 is the gene that’s used in polymerase chain reaction (PCR)-based tests to search for E. granulosus DNA in clinical or environmental samples, and it is the one gene we need to confirm infection, especially in endemic zones. In addition to diagnosis, AgB2 immunity modulation also suggests that it could be a candidate for vaccines and therapy. These studies confirmed AgB2’s role in hydatid disease study and helped improve diagnostic and control approaches (Zhang et al, 2003; Carmena et al, 2006; Mamuti et al., 2006). Research has demonstrated that Echinococcus species exhibit variation in their genetic makeup because of differences in their geographic distribution. The various genotypes (G1-G10) of E. granulosus display unique prevalence patterns based on the geographic regions in which they are discovered (Shamsi et al., 2017). Using a variety of preventive techniques that disrupt the life cycle between definitive and intermediate hosts, echinococcosis can be effectively managed. However, the most effective way to prevent hydatid disease in humans and to lessen the effects of Echinococcus infections in cattle is to conduct large-scale educational campaigns directed at people who reside in areas where the disease is common (Chandrakesan and Parija, 2003). This study used the AgB subunit 2 gene from Al-Diwaniyah City, Iraq, to evaluate the genotype of the E. granulosus parasite recovered from sheep. Materials and MethodsHydatid cyst preparation and sample collectionThe current investigation entailed the acquisition of 50 specimens of hydatid cysts (cysts filled with fluid and parasite-containing structures) from the livers of sheep at abattoirs (slaughterhouses) in the Al-Diwaniya province during the period between March and October 2023. Hydatid cysts were acquired from the killed sheep and stored in containers refrigerated with ice for transportation to the Advanced Parasitology Laboratory at the College of Veterinary Medicine, University of Al-Qadisiyah. Upon arrival at the laboratory, the samples were rinsed immediately with water to eliminate any remaining soil or blood. The sterilization of each hydatid cyst surface was then performed with 70% ethanol (Smyth, 1985). Then, 10 ml medical syringes specifically designed to be free from microorganisms were used to extract the fluid from the cyst, and the protocolises were subsequently gathered (larval stage of the tapeworm) and obtained under aseptic (sterile) conditions. The cyst fluid was aspirated and collected along with the protocolises in a 250-ml beaker. The preserved samples were subsequently used for PCR and genetic sequencing to identify the prevalent strains of the parasitic tapeworm (Goksoy et al., 2008). The parasite material used for PCR was protocolises and germinal layers from hydatid cysts. Protoscolices were kept at −20°C in PBS, thoroughly washed with ethanol, and stored at −20°C in sterile microcentrifuge tubes. These storage systems preserved DNA integrity before application. DNA extractionTotal genomic DNA was extracted from either protocolises (the larval stage of a tapeworm) and germinal layers (the actively dividing cells) of sheep samples using a gSYAN DNA microkit procedure. To summarize, germinal layer tissue (20 mg) and protocolises (200 l) were homogenized and incubated at 60°C for 15 minutes with proteinase K, followed by the addition of GSB buffer and ethanol. The lysate was run through a spin column to capture DNA, washed, and eluted. The protocolises were first rinsed with 50 l of phosphate buffer saline to remove any ethanol and then centrifuged for 3 minutes to collect the sediment. Next, 20 g of the germinal layer tissue from sheep hydatid cysts and 200 l of fluid containing the protocolises were homogenized and placed into a 1.5-l microcentrifuge tube. After adding and combining 20 ml of proteinase K, the samples were incubated for 15 minutes at 60 °C. Subsequently, 200 l of GSB buffer was added, thoroughly mixed, and incubated for 5 minutes at 60°C, with the tube rotated every 3 minutes. Then, 200 l of absolute ethanol was added and mixed vigorously. Afterward, the DNA was purified by passing it through a filter column placed inside a 2-ml collection tube. To determine the concentration and purity of the extracted DNA, a Nanodrop-gel electrophoresis setting was used. Polymerase chain reactionThe extracted DNA was subjected to polymerase chain reaction using specific primers targeting the AgB gene in E. granulosus. This primer was designed using the NCBI GeneBank database and the Primer 3 plus web application based on the genomic sequence of the E. granulosus species (GenBank: AY871037.1). The base pair (500–2,000) (bp) region of the AgB gene in E. granulosus from sheep was amplified using the following primer sequences: forward primer: CGTCGTTCAAGCGTGAGTCT and reverse primer: TGTCCCGACGCATGACTTAC. All PCR reactions were performed using Taq DNA Polymerase (Thermo Fisher Scientific, USA), reaction buffer (1X), and dNTP solution (200 M each), both provided by the same manufacturer. The primers were created by Bioneer Company (South Korea), and the PCR solution was free of nucleases. These parameters make the amplification process reproducible and accurate. The PCR reaction was performed in a total volume of 20 l. This included 5 l of purified genomic DNA, 1 l (10 picomoles) of each forward and reverse primer, and 10 l of deionized PCR water. This reaction mixture was briefly mixed using an ExiSpin vortex A thermocycler (Mygene Bioneer, Korea) was used to perform PCR with the following cycling conditions: initial denaturation at 94°C for 5 minutes, denaturation at 94°C for 30 seconds, annealing at 54°C for 30 seconds, extension at 72°C for 30 seconds, and a final extension at 72°C for 5 minutes. DNA sequencing and phylogenetic tree analysisSeven purified positive PCR products of the AgB subunit 2 gene were sequenced. AgB2 forward primers were used for DNA sequencing, which was conducted using the AB DNA sequencing system provided by the Bioneer Company in Korea. The DNA sequencing data were then used for phylogenetic analysis using the MEGA –X program. In summary, a 310 (bp) PCR product of the AgB2 gene was sequenced by the Bioneer Company in South Korea, and the DNA sequence data were subsequently analyzed using bioinformatics tools to construct a phylogenetic tree. The phylogenetic tree using the maximum likelihood method was analyzed by bioinformatics using MEGA-X (version 10.2.6): Multiple sequence alignment was performed, and the alignments were precise with reference sequences downloaded from GenBank. The sequences were compared with local and global isolates, and parameters were established for model selection and bootstrapping. ResultsFrom a total of 50 samples of hydatid cysts, 22 positive samples were positive by specific primer with 44%, and seven samples out of 22 positive samples were sent for sequencing. Amplification of antigen B subunit 2 by PCR and sequencingPCR amplification of hydatid cyst isolates was effective. After the electrophoresis, the PCR result revealed distinct 310 bp bands in all samples, as clearly demonstrated in Figure 1 under UV light. The G1 genotypes were identified in the isolates selected.
Fig. 1. PCR analysis of antigen B subunit 2 in Echinococcus granulosus hydatid cysts obtained from sheep using agarose gel electrophoresis. Lane M: Ladder, Lanes 1–5: Positive samples. Sequencing and phylogenetic tree construction of antigen B subunit 2 geneSheep phylogenetic analysis of seven amplicons of sheep samples revealed that G1 is the genotype that infects sheep in Iraq (multiple sequence). The tree was built using the sequences collected here and reference sequences from GenBank, including those from global isolates (Brazil bovine isolates: AY496952.1 and AY314985.1 and Argentina human isolates: AY569358.1 and AY569366.1). Local sequences were aligned to these references with BioEdit, and all seven isolates were of the G1 genotype (the sheep strain of choice). The alignment was performed using Bioedit, a DNA analysis application, and the results were compared with previously published data. Gene references. The findings indicated that 99.69% and 100.00% of the alignment results were exactly the same as the typical sheep strain G1 genotype. The AgB subunit 2 gene of local isolates E. granulosus was aligned with global reference strains for E. granulosus recorded in GenBank. The detected isolates of E. granulosus were deposited in GeneBank using the following accession numbers: OR232318.1, OR232319.1, OR232320.1, OR232321.1, OR232322.1, OR232323.1, and OR232324.1 and submitted to GenBank. The phylogenetic tree based on sequence analysis is shown in Figure 2.
Fig. 2. Phylogenetic tree analysis based on antigen B subunit 2 using the likelihood process (MEGA X version) for Echinococcus granulosus typing detection. Colored dots: Current study sequences. The genetic sequence similarity between the local isolates with accession numbers (OR232323.1, OR232324.1) obtained from sheep liver samples was 100% compared with the Brazilian isolates obtained from cattle (AY496952.1). This high sequence identity suggests a close genetic relationship between local and Brazilian isolates. In contrast, the sequence similarity between the local and Argentine isolates obtained from human and animal samples was 90.46%. Similarly, the sequence similarity among the Argentine isolates obtained from human and animal samples was 89.85%. The phylogenetic network analysis revealed clear genetic distinctions between the various genotypes examined in this study. The 310-bp AgB subunit 2 gene sequence was aligned to global reference sequences in GenBank. The area was identified as a valid genetic marker for strain differentiation in E. granulosus. All seven sequenced amplicons of the hydatid cysts were mapped to international sequences, and the isolates were all G1 genotypes, 100% identical to Brazilian strains (AY496952.1), and references worldwide. DiscussionThe study conclusions showed that among the sheep samples collected, the G1 genotype of E. granulosus, i.e., the strain prevalent in sheep, was the most often detected genotype. Our analysis showed that the E. granulosus strain that was most frequently found in Al-Diwaniyah corresponded with the results reported in a previous study (Nejad et al., 2011). However, it does conflict with the data reported in reference (Fadhil and Agha, 2011), which indicates the presence of two different strains identified from sheep samples: strain (G1) and buffalo strain (G3) depending on cox1 and nad1. The reason for this difference may be due to the difference in the target, as AgB subunit 2 was used in the current study. The methodologies used for the molecular identification and genetic categorization of E. granulosus can influence the outcomes of analyzing and distinguishing the strains of this parasite. Variations in the procedures employed for isolating and cultivating the parasite, as well as the genetic sequencing techniques used, may result in differences in the identification of the specific strains. The results mismatched prior research conducted in Iraq, specifically by Hama et al. (2013), which identified the presence of G1 in both humans and animals. The results indicated a complete match with the sheep strain with 100% similarity (G1). Al-Diwaniyah city sheep as hosts are also pertinent for studying the E. granulosus AgB2 gene because the area is very rich in hydatid disease (probably due to environmental and animal farming characteristics). The urban farming and pastoral landscapes, where sheep are a major animal, make it easy for final hosts (dogs) and intermediaries (sheep) to close up their interactions and thus the parasite’s life cycle. Additionally, conventional animal production methods such as home slaughtering and improper sewage disposal of infected organs aid in the spread of the disease. These findings and the fact that the environment is optimal for parasite growth make Al-Diwaniyah an ideal location to study the genetic and epidemiological biology of E. granulosus and to establish specific diagnostic and control measures. The local isolates from sheep liver (with accession numbers OR232323.1 and OR232324.1) have a 100% genetic sequence match with the Brazilian isolates from cattle (AY496952.1). This high degree of sequence identity suggests that the local isolates are genetically similar to international breeds found in Brazilian cattle, indicating potential shared origins or close evolutionary relationships. This is because Iraq imports live animals from Brazil for rearing here for a period of time before they are sent to slaughterhouses. This gives a good amount of time for sharing different microorganisms, including the current parasite. Molecular and immunological studies have shown that the parasitic tapeworm E. granulosus expresses the (AgB) protein. This is significant because AgB is a key antigenic target in the host’s immune response to cystic echinococcosis (CE) caused by E. granulosus. AgB subunit 2 aggregation in cyst fluid. Due to the nature of CE cystic lesions, AgB subunit 2 may accumulate in cyst fluid after being secreted by the parasite. Using antigen B subunit 2 (AgB2) in this study could identify genotypic differences from those detected by mitochondrial genes, such as cox1 and nad1, because of differences in genetic and functional properties. AgB2, which is encoded by nuclear DNA, participates directly in immune modulation and is subject to selection pressures from host immune responses, which may lead to greater genetic variation and adaptation to specific hosts (Zhang et al., 2003; Carmena et al., 2006). This is why AgB2 can be useful for spotting very subtle genotypic differences in antigenic or host–parasite interactions, such as what is happening in regions such as Al-Diwaniyah where there are different livestock and environmental factors. Instead, the genes cox1 and nad1 for mitochondria are very conserved and are used primarily for large-scale phylogenetic classes of Echinococcus granulosus genotypes (e.g., G1-G10) (Hüttner et al., 2008). These markers can be useful for international strain identification, but they might miss host-specific adaptations or functional differences that could be used to interpret local epidemiology and immune behavior. The focus on AgB2 aims not only to characterize E. granulosus genetic diversity locally but also to improve diagnostic and control methods by understanding immune and pathogenic dynamics (Mamuti et al., 2006). This accumulation allows AgB subunit 2 to aggregate into a conformation that is more immunogenic (able to provoke a stronger immune response) than the individual monomer form. Hejazi et al. (2024) and Mohaghegh et al. (2018), both of whom, found Echinococcus granulosus with the G1 genotype to be predominant in northeastern Iran. Hejazi et al. focused on definitive host (dogs) isolates and sheep and dogs during the parasite lifecycle. Mohaghegh et al. used cox1-based qPCR-HRM for fast, precise sheep genotyping but focused on mitochondrial genes, which are conserved and can be phylogenetically classified. The aggregated form of AgB subunit 2 may be a more potent immunogenic target than the monomer form, potentially influencing the host’s immune response to the parasite. The diagnostic potential of recombinant AgB has been demonstrated. These findings suggest that recombinant AgB should be recommended for the serological diagnosis of cystic echinococcosis. In summary, the key points highlight the genetic similarities between local and international parasite isolates, the expression and potential immunogenic properties of the AgB antigen, and the diagnostic utility of recombinant AgB, all of which are important considerations in understanding and managing cystic echinococcosis (Dintzis et al., 1976). The similarity between the E. granulosus strains under study and global strains may be attributed to the fact that the aforementioned gene does not show any difference in its nucleotide sequence (Moro and Schantz, 2009). ConclusionOur research revealed that Echinococcus granulosus has many genotypes, with the predominant genotype being G1 in sheep from Al-Diwaniyah City, Iraq. The 310-bp AgB2 gene sequences were highly similar in genetic content to isolates from Brazilian strains. These findings suggest that E. granulosus strains in these areas are genetically linked and might share common evolutionary roots because of trade and migration. These results support the use of antigen B subunit 2 as a molecular marker in genomic and epidemiological studies. These data allow us to make more sense of the parasite’s distribution and, therefore, can be used to target control and diagnostic interventions in endemic areas. Further studies with more data and geographic comparisons are needed to confirm these results and investigate the dynamics of E. granulosus transmission. AcknowledgmentsThe authors would like to thank the Department of Nursing techniques, Technical Institute of Al-Diwaniyah, AL-Furat AL-Awsat Technical University, Iraq. Conflict of interestThere are no conflicts of interest to declare. FundingThe study did not receive any financial support, but the authors’ personal assets funded the study. Authors’ contributionsAll authors participated in all parts of the study. Data availabilityThe data presented in this study are available upon request from the corresponding author. ReferencesAgudelo Higuita, N.I., Brunetti, E. and McCloskey, C. 2016. Cystic echinococcosis. J. Clin. Microbiol. 54, 518–523. Andersen, F.L., Ouhelli, H. and Kashani, M. 1997. Compendium on cystic echinococcosis. Provo: Brigham Young University. Brunette, G.W. 2012. The yellow book: CDC and health information for international travel. Oxford: Oxford University. pp: 640. Carmena, D., Benito, A. and Eraso, E. 2006. Antigen B from Echinococcus granulosus: an overview of its immune escape mechanisms and role as a diagnostic marker. Parasitol. Res. 98(5), 357–364. Chandrakesan, S.D. and Parija, S.C. 2003. Latex agglutination test (LAT) for antigen detection in the cystic fluid for the diagnosis of cystic echinococcosis. Diagn. Microbiol. Infect. Dis. 45(2), 123–126. Dintzis, H.M., Dintzis, R.Z. and Vogelstein, B. 1976. Molecular determinants of immunogenicity: an immunon model of immune response. Proc. Natl Acad. Sci. 73, 371. Fadhil, S.A. and Agha, A.M. 2011. Genotyping of cystic echinococcosis isolates from human and animals clinical samples. A Thesis Submitted to the Council of Veterinary Medicine College. Al Khobar, Saudi Arabia: Al-Qadisiyah University. Goksoy, E., Saklak, M., Saribeyoglu, K. and Schumpelick, V. 2008. Surgery for Echinococcus cysts in the liver. Der Chirurg. 79(8), 729–737. Gottstein, B. and Reichen, J. 2002. Hydatid lung disease (echinococcosis/hydatidosis). Clin. Chest Med. 23, 397–408. Hama, A.A., Mero, W.M. and Jubrael, J.M., 2013. Molecular identification of Echinococcus granulosus (G1) strain in human and animals. J. Univ. Zakho. 1(1), 1–6. Hejazi, S.H., Mirbadie, S.R., Jafari, R., Rezaiemanesh, M.R., Azizi, O., Badmasti, F. and others. 2024. Echinococcus granulosus sheep strain (G1) as the predominant genotype in definitive host (dogs) isolates in northeastern Iran. Vet. Parasitol. Reg. Stud. Rep. 48, 100975. Hüttner, M., Nakao, M., Wassermann, T., Siefert, L., Boomker, J.D., Dinkel, A., Sako, Y., Mackenstedt, U., Romig, T. and Ito, A. 2008. Genetic characterization and phylogenetic position of Echinococcus felidis (Cestoda: Taeniidae) from the African lion. Int. J. Parasitol. 38, 861–868. Hüttner, M., Siefert, L., Mackenstedt, U. and Romig, T. 2008. A survey of Echinococcus species in wild carnivores and livestock in East Africa. Int. J. Parasitol. 39(11), 1269–1276. Ibrahim, Z.A. 2010. Attenuation of human hydatid cyst protoscolices viability by 1Hydroxyphenazin (1-HP) pigment: in vitro and in vivo study. J. Fac. Med. Baghdad. 52(3), 338–341. Langer, J.C., Rose, D.B. and Kestone, J.S. 1984. Diagnosis and management of hydatid disease of the liver. Ann. Surg. 199, 412. Maddison, S.E., Slemenda, S.B., Schantz, P.M., Fried, J.A., Wilson, M. and Tsang, V.C. 1989. A specific diagnostic antigen of Echinococcus granulosus with an apparent molecular weight of 8 kDA. Am. J. Trop. Med. Hyg. 40(4), 377–383. Maharjan, S.B., Paudyal, S., Shah, S., Dahal, R. and Shah, J.N. 2018. Clinical profile and surgical outcome of abdominal hydatid cyst at a university hospital in Nepal. J. Patan Acad. Health Sci. 5(2), 52–57. Mamuti, W., Sako, Y., Nakao, M., Xiao, N., Nakaya, K., Ishikawa, Y. and Ito, A. 2006. Recent advances in characterization of Echinococcus antigen B. Parasitol. Int. 55(S), S57–S62. Mohaghegh, M.A., Yousefi-Darani, H., Azami, M., Ghomashlooyan, M., Hashemi, N., Jabalameli, Z. and others. 2018. Analysis of the cox1 gene in Echinococcus granulosus from sheep in northeast Iran using PCR high-resolution melting (qPCR-HRM) curve analysis. Trop. Biomed. 35(1), 91–99. Moro, P.L. and Schantz, P.M. 2009. Echinococcosis: a review. Int. J. Infect. Dis. 13, 125–133. Nejad, M.R., Roshani, M., Lahmi. and Mojarad, F.E. 2011. Evaluation of four DNA extraction methods for the detection of Echinococcus granulosus genotype. J. Gastrol. Hepatol. FBB. 4(2), 91–94. Pan, D., Bera, A.K., Bandyopadhyay, S., Das, S., Rana, T., Das, S.K., Bandyopadhyay, S., Manna, B. and Bhattacharya, D. 2011. Molecular characterization of antigen B2 subunit in two genotypes of Echinococcus granulosus from Indian bubaline isolates, its stage specific expression and serological evaluation. Mol. Biol. Rep. 38, 2067–2073. Shamsi, M., Dalimi, A., Khosravi, A. and Pourahmad, F. 2017. Determination and sequencing of genotypes of Echinococcus granulosus from stray dogs isolates with cox1 gene in Ilam, west of Iran. J. Ilam Univ. Med. Sci. 24, 97–106. Smyth, J. 1985. In vitro culture of Echinococcus spp. In Proceedings of the 13th Int. Congr. Hydatidology. Madrid. pp: 84–89. Thompson, R.C. 2008. The taxonomy, phylogeny and transmission of Echinococcus. Exp. Parasitol. 119(4), 439–446. Weber, T.F., Junghanss, T. and Stojković, M. 2023. Pulmonary cystic echinococcosis. Curr. Opin. Infect. Dis. 36(5), 318–325. Zhang, W., Li, J. and McManus, D.P. 2003. Concepts in immunology and diagnosis of hydatid disease. Clin. Microbiol. Rev. 16(1), 18–36. | ||
| How to Cite this Article |
| Pubmed Style Hamad WA, Jawad TI, Atiya WH. Phylogenetic analysis and genetic evaluation of the Echinococcus granulosus antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah, Iraq. Open Vet. J.. 2025; 15(2): 1043-1049. doi:10.5455/OVJ.2025.v15.i2.52 Web Style Hamad WA, Jawad TI, Atiya WH. Phylogenetic analysis and genetic evaluation of the Echinococcus granulosus antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah, Iraq. https://www.openveterinaryjournal.com/?mno=226126 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.52 AMA (American Medical Association) Style Hamad WA, Jawad TI, Atiya WH. Phylogenetic analysis and genetic evaluation of the Echinococcus granulosus antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah, Iraq. Open Vet. J.. 2025; 15(2): 1043-1049. doi:10.5455/OVJ.2025.v15.i2.52 Vancouver/ICMJE Style Hamad WA, Jawad TI, Atiya WH. Phylogenetic analysis and genetic evaluation of the Echinococcus granulosus antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah, Iraq. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 1043-1049. doi:10.5455/OVJ.2025.v15.i2.52 Harvard Style Hamad, W. A., Jawad, . T. I. & Atiya, . W. H. (2025) Phylogenetic analysis and genetic evaluation of the Echinococcus granulosus antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah, Iraq. Open Vet. J., 15 (2), 1043-1049. doi:10.5455/OVJ.2025.v15.i2.52 Turabian Style Hamad, Weam A., Thanaa Ismael Jawad, and Wegdan H. Atiya. 2025. Phylogenetic analysis and genetic evaluation of the Echinococcus granulosus antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah, Iraq. Open Veterinary Journal, 15 (2), 1043-1049. doi:10.5455/OVJ.2025.v15.i2.52 Chicago Style Hamad, Weam A., Thanaa Ismael Jawad, and Wegdan H. Atiya. "Phylogenetic analysis and genetic evaluation of the Echinococcus granulosus antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah, Iraq." Open Veterinary Journal 15 (2025), 1043-1049. doi:10.5455/OVJ.2025.v15.i2.52 MLA (The Modern Language Association) Style Hamad, Weam A., Thanaa Ismael Jawad, and Wegdan H. Atiya. "Phylogenetic analysis and genetic evaluation of the Echinococcus granulosus antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah, Iraq." Open Veterinary Journal 15.2 (2025), 1043-1049. Print. doi:10.5455/OVJ.2025.v15.i2.52 APA (American Psychological Association) Style Hamad, W. A., Jawad, . T. I. & Atiya, . W. H. (2025) Phylogenetic analysis and genetic evaluation of the Echinococcus granulosus antigen B subunit 2 gene isolated from sheep in Al-Diwaniyah, Iraq. Open Veterinary Journal, 15 (2), 1043-1049. doi:10.5455/OVJ.2025.v15.i2.52 |