| Research Article | ||
Open Vet. J.. 2025; 15(2): 804-812 Open Veterinary Journal, (2025), Vol. 15(2): 804-812 Research Article In vitro model of ischemic stroke neurons from Macaca fascicularisRatih Rinendyaputri1,a, Sela Septima Mariya1, Frans Dany1, Ariyani Noviantari1, Dwi Budiono2, Lisa Andriani Lienggonegoro1, Hasta Handayani Idrus1, Wireni Ayuningtyas3, Rachmawati Noverina3, Fathul Huda4,a and Ahmad Faried5,a*1Center for Biomedical Research, Research Organization for Health, National Research and Innovation Agency, Bogor, Indonesia 2Veterinary Paramedic Study Program, Collage of Vocational Studies, IPB University, Bogor, Indonesia 3Bio Farma Stem Cell Research and Development, Bandung, Indonesia 4Department of Neurology, Faculty of Medicine, Dr Hasan Sadikin Central General Hospital/Universitas Padjadjaran, Bandung, Indonesia 5Neurosurgery Department, Faculty of Medicine, Padjadjaran University, Bandung, Indonesia aGraduate School of Biomedical Sciences, Doctoral Program, Faculty of Medicine, Universitas Padjadjaran, Indonesia *Corresponding Author: Ahmad Faried. Neurosurgery Department, Faculty of Medicine, Padjadjaran University, Bandung, Indonesia. Email: ahmad.faried [at] unpad.ac.id Submitted: 29/10/2024 Accepted: 21/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
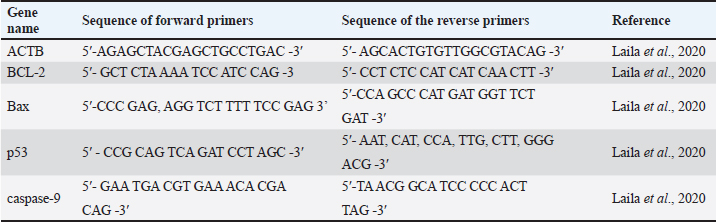
AbstractBackground: Primary cells have the same heterogeneity and differentiation capacity that has potential as an in vitro ischemic stroke models. It is hoped that primary cells from non-human primates that have genetic similarities to humans can provide molecular information and become more accurate data for use in drug screening, especially stroke therapy. B27 is a supplement commonly used in neuronal cell cultures, but there are concerns that its effects will interfere with the neuroprotective processes of the drug candidates being tested. Aim: This research will prove the demonstration of neurons as an ischemic stroke model and the effects of B27 in Macaca fascicularis (Mf) neurons as a model for ischemic stroke under oxygen glucose deprivation (OGD). Methods: Neurons were obtained from a collection of biological materials collected during previous research. Neuronal validation was performed using immunocytochemistry with the marker β-tubulin. Expression of the apoptotic response was performed by real time- polymerase chain reaction/(RT-PCR) using Bax, BCL-2, caspase-9, and p53 gene markers. Characterization of neurons in terms of positive tβubulin markers and induction of OGD in neurons can be performed for 6 h to model ischemic stroke. Results: This study showed that cultured neurons under OGD conditions can experience apoptosis, namely by increasing pro-apoptosis and decreasing anti-apoptosis. However, B27 supplementation increased the expression of anti-apoptotic Bcl-2 genes and decreased proapoptotic genes such as Bax, caspase 9, and p53. Conclusion: Neuron culture from Mf can be used as an in vitro model of ischemic stroke, and B27 supplementation in neurons exerts neuroprotective effects on the induction of OGD. Keywords: Ischemic stroke, Macaca fascicularis, Neuron, Oxygen glucose deprivation, OGD. IntroductionStroke is a degenerative disease associated with aging and a leading cause of morbidity and mortality worldwide. Ischemic ischemic stroke is caused by a blockage in the brain's arteries, resulting in a decrease in blood flow and oxygen to the affected brain tissue (Kuriakose and Xiao, 2020; Zhao et al., 2022). Brain tissue regeneration is very slow, and it is critical to use appropriate therapy to accelerate neuron cell regeneration. Researchers are still looking for the best adjuvant therapy for ischemic stroke. Preclinical research for ischemic stroke treatment has been conducted both in vivo and in vitro. Animal models ranging from rats to macaques are used to simulate ischemic stroke by performing middle cerebral occlusion artery (MCAO) and global ischemic (Bai et al., 2022; Lin et al., 2022). The oxygen-glucose deprivation (OGD) method can be used in vitro with neuron cells. Both models work by induce conditions such as ischemic stroke, by reducing the available oxygen and glucose supply to cells (Gibson and Trotman-Lucas, 2021; Babu et al., 2022). The hypoxia chamber is an incubator/chamber in which the oxygen supply is suppressed by adding nitrogen gas. Reperfusion can be simulated by reintroducing glucose and oxygen into a different chamber. The success of the in vitro brain ischemic cell model could be determined by optimizing the OGD induction time. Stroke models can be created using cell lines or primary neuronal cell cultures. Several studies have reported on the use of neural stem cells (NSCs) derived from rat fetuses, cell lines such as PC12, SH-SY5Y, BV2, HA, and HBMEC, and NSC-derived induce pluripotent stem cells, bone marrow derived mesenchymal stem cell (BMMSC), and human embryonic stem cells (Degregorio-Rocasolano et al., 2020; Goshi et al., 2020; Noviantari et al., 2020; Vagaska et al., 2020; Peng et al., 2021). The results of these studies are expected to provide an alternative for animal substitution in research involving neuronal cells or NSCs. The limitations of using cell lines are the lack of heterogeneity, do not represent the diversity of the human brain, the absence of a supporting microenvironment, and the effect on the immune system (Namekawa et al., 2019). Nevertheless, the use of neuronal cell lines, such as ReNcell, PC12, and SH-SY5Y, is widely used for research because it is cheaper and easier to genetically modify than primary cultures or animal models. (Martin et al., 2022). Several therapies were tested to determine their efficacy for neuroprotection and neurogenesis. Understanding the pathogenesis and establishing a clear pathway can facilitate the identification of therapeutic targets. In addition to the type of cells and duration of OGD induction, the composition of the induction media affects NSC in an ischemic stroke model. The use of medium and supplement compositions, such as B27, during OGD expansion and treatment is clearly different to obtain optimal results (Babu et al., 2022; Martin et al., 2022). Neural cell culture techniques emphasize the need to carefully control the metabolic environment to mimic brain function and the cellular and molecular pathophysiology of brain disorders in culture. Cell viability is decreased by up to 50% in glucose deprivation, even in normoxic conditions, but B27 supplementation can affect neuronal metabolism, preserve neurons in hypoxic conditions, and inhibit glycolysis (Wohnsland et al., 2010; Sünwoldt et al., 2017). The effects of B27, neurotrofic growth factor (NGF), and basic firoblast growth factor (b-FGF) supplementation in serum-free media increased the efficiency of human cord mesenchymal stem cells from developing into neuron-like cells. Nestin, neuron-specific enolase, neurofilament heavy polypeptide, and glial fibrillary acidic protein expression show these findings (Li et al., 2020). The induction of the differentiation of SH-SY5Y cells into glutaminergic neuron cells with retinoic acid (RA) and BDNF was extremely successful. The ability of RA and BDNF to promote neurite elongation was not further enhanced by the addition of B27. However, B27 promoted the generation and survival of many differentiated glutamatergic SH-SY5Y cells. By maintaining no apoptotic process and increased cell death in the treatment without fetal bovine serum (FBS)/serum deprivation in serum-free media, B27’s safety was shown (Martin et al., 2022). Drugs or adjuvant for ischemic stroke therapy are still being explored. Several studies have reported success in stroke therapy using rats as animal models (Bai et al., 2022). The success of adjuvant stroke therapy using mouse models lacks genetic differences, and metabolism is a consideration for translational research. In addition, the number of animal models must consider animal ethics and welfare concerns. The macaque as a non-human primate (NHP) animal model is required in drug discovery research because of its phylogenetic similarity to humans (Mariya et al., 2019). NHP cells can be used to reduce the use of macaques as an animal model. For neuroscience research, neurons isolated from macaque brain tissue can be used. We were able to culture astrocytes and neuron cells from macaque brain tissue. The brains of the NHP animal model are structurally and functionally similar (Lin et al., 2022). The use of primary culture neurons derived from the NHP presents a challenge for neuroscience research. The most difficult aspect of utilizing using as an in vitro model for ischemic stroke is to determine the best time and culture conditions. Neuron viability is maintained by the application of nutrients and culture media such as B27. This supplement protects neurons and cells from damage. To reduce bias in component assays, the culture medium must be optimized during OGD induction. The successful use of neurons from NHP obtained from the NHP contributed to the development of ischemic stroke therapy. The goal of this study was to obtain a neuronal model of Macaca fascicularis (Mf) as an in vitro model of ischemic stroke, as well as to investigate the influence of B27 on the neuronal culture medium under OGD conditions. Materials and MethodsNeuron cultureThis research uses neurons that have been stored as biological material from Biofarama, which received ethics from the Institutional Animal Care and Use Committee (IACUC) of PT Biofarma (01/IACUC_BF/X/22). The culture media used were neurobasal media (Gibco), 1% B27 (Gibco), 1% gentamicin (GibcoTM), 10% FBS(GibcoTM), 1% Non-essential amino acids, and N2. (18) NSCs were separated into groups with and without B27 in the culture medium for the hypoxia treatment group. Neuron induction with Oxygen and/or Glucose Deprivation (OGD)Hypoxic conditions for NSC culture can be used as a brain ischemic cell model. Macaque NSCs were cultured in phosphate-buffered saline (PBS) medium (no glucose, GIBCO) at 5% O2 (95% N2 and 5% CO2). After the third passage on day 4, the culture medium was replaced with a treatment medium under nomoxic conditions (21% O2 and 5% CO2) for 6 hours (Zhang et al., 2022). Neuron Surface Marker AnalysisImmunocytochemistry (ICC)NSCs were characterized by using the β-tubulin marker in passages two or three. On the third day of culture, the NSCs were fixed with 4% w/v paraformaldehyde (PFA), washed with PBS according to the kit, and added β-III tubulin antibody (sc-5274) was added for 12 h at 4oC. Rinsing with PBS and the solution was performed according to kit instructions. The antibodies were diluted to 1:50. Staining with hematoxylin for 20 s was followed by rinsing with PBS and observation under a microscope (Budiariati et al., 2019). Characterization of post-induction neurons by quantitative real time-polimerase chain reaction (RT-qPCR)The gene expressions of apoptosis biomarkers (Bax, BCL-2, caspase 9, and p53) were evaluated between the hypoxia-treated group and the non-hypoxia-treated, B27 supplemented, and non-supplemented groups. The total mRNA was extracted from the NSC samples according to the kit procedure (RNA easy Mini Kit, Qiagen, Germany). The mRNA concentration was measured using Nanodrops. A 10-ng/L concentration of mRNA was reversed to cDNA using a cDNA synthesis kit (Sensifast Bioline, Meridian) according to the manufacturer’s protocol. cDNA was used as a template for RT-qPCR to evaluate gene expression levels of apoptosis biomarkers. The apoptosis biomarker primers used in this study were shown in Table 1. RT-qPCR was performed using Sensifast Bioline SYBR Green Mix (Meridian, USA) in Quantistudio 5 (Applied Biosystem 7500) thermal cycler. The RT-qPCR program was designed for pre-denaturation at 95oC for 2 min, denaturation at 95oC for 30 s, and annealing at 95oC for 30 s. The denaturation and annealing steps were repeated for 40 cycles. We analyzed the cycle threshold to measure the relative quantitative (RQ) using the ∆CT (Livak) method (Darusman et al., 2022). Table 1. The primer sequences used in this study.
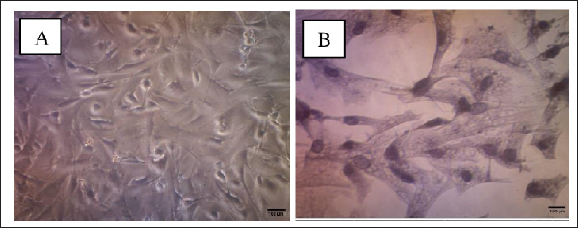
Data AnalysisQuantitative relative (RQ) data of apoptotic biomarker genes (Bax, BCL-2, caspase 9, and p53) under hypoxic and normoxic conditions, with or without B27 supplementation under NSC hypoxia conditions were calculated using the Livak formula. Ethical approvalNot needed for this study. ResultsCulture and characterization of neuronsThe population of neurons cultured with the appropriate medium, namely basal-medium neurons, morphologically has cell bodies and neurites that have different branches and lengths. After confluence and the number of cells increases for further treatment, each cell population trajectory exhibits unipolar, bipolar, and multipolar forms until the third passage. ICC with the β-III tubulin marker was used to determine neuronal populations in cultures of Mf fetal brain hippocampal tissue (Fig. 1).
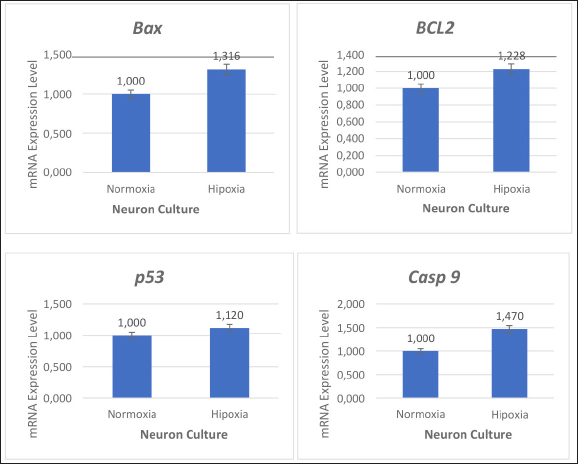
Figure 1. Characterization of Macaca fascicularis brain neuron cultures. Neuron culture on day 6 after the first culture (A), after fixation and administration of β-tubulin antibodies and hematoxylin (B) (using 100x and 400x magnification). Induction of Oxygen and Glucose Deprivation (OGD) on NeuronOnce the cells reached 70–80% confluency, the neuron culture medium was rinsed with PBS and replaced with the glucose-free medium before being transported to an anaerobic chamber containing 95% N2 and 5% CO2 for 6 h at 37° C. After OGD induction in the third passage of the neuron population, cell morphology remained unchanged (Fig. 1). The primer sequences used in this study are listed below. These primers were utilized to determine the apoptotic process in the NSC population before and after OGD induction. ACTB was used as a housekeeping gene control, and anti-apoptotic primers, such as BCL-2 and pro-apoptosis, using p53, Bax, and caspase-9 (Table 1). The effects of oxygen and glucose deprivation (OGD) and re-exposure to normal culture conditions are expected to mimic hypoxic ischemia and reperfusion of NSC cultures of Mf. To determine the effect of apoptosis on NSCs, gene expression was detected and evaluated for various gene markers for the apoptotic process. This study showed that hypoxic conditions resulted in increased expression levels of Bax, BCL-2, caspase 9, and p53 which were almost 2-fold higher than those detected in nonhypoxic conditions (Fig. 2).
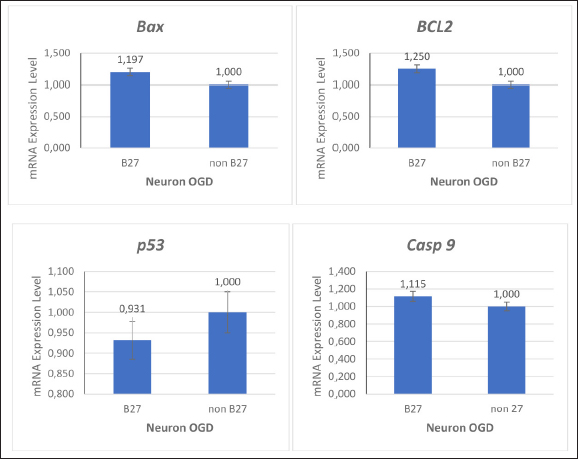
Figure 2. Effects of oxygen and glucose deprivation (OGD) on neuronal populations undergoing apoptosis. In hypoxic conditions, neuronal cells exhibit increased Bax, p53, and caspase-9 expression and decreased BCL-2 expression. To determine the effect of B27 supplementation in NSC cultures treated with OGD, markers for the apoptosis process were examined using the RT-qPCR. The expression of the BCL-2 detection gene increased and the expression of the Bax, p53, and caspase-9 genes decreased with B27 supplementation (Fig. 3).
Figure 3. Effects of B27 supplementation on neurons following oxygen and glucose deprivation (OGD). These data showed that B27 supplements give effect in BCL-2 gene expression as antiapoptotic and Bax, p53, and caspase-9 as proapoptotic genes. DiscussionOur study successfully demonstrated the successful use of neurons Mf neurons vitro ischemic stroke model and the neuroprotective effect of B27 supplementation in culture media in response to OGD. Under OGD conditions, there is an increase in apoptosis factors such as Bax, p53, and caspase9 and low expression of BCL-2 as an antiapoptotic marker. However, apoptosis is characterized by reduced expression of pro-apoptotic genes such as p53 and caspase 9, as well as an increase in the BCL-2/Bax ratio when B27 is used. B27 has been used to successfully induce the differentiation of MSCs and SH-SY5Y cells into glutaminergic neurons (Li et al., 2020; Martin et al., 2022). Control of neuron metabolism under in vitro culture conditions can be achieved by supplementing culture media. Different supplements will respond differently to glycolysis, which is something to consider when using them as a cell model. Different responses due to changes in the microenvironment or treatment results are important points (Sünwoldt et al., 2017). Similarly, the presence or absence of glucose in culture media has a significant impact on neuronal survival (Wohnsland et al., 2010). This study successfully isolated and characterized neurons from the hippocampal region of the Mf fetus for an ischemic stroke model using the OGD method. OGD induction in neurons can be performed for 6 h to create an ischemic stroke model. Previous research has reported the isolation of neurons from the cortex of the rat fetus using the OGD model (Liu et al., 2014). This study used βtubulin as the neuron marker to characterize the heterogeneity of cell populations isolated from the hippocampus. The culture in this study used a neurobasal medium was supplemented with 10% FBS, N2, and B27, which were used for dominant neuronal cell culture. To obtain the neron population, we added 20 µg/mL b-FGF, 20 µg/mL EGF, 10 µg/mL insulin-transferrin-sodium selenite supplement, and 10 µg/mL heparin (Liu et al., 2014; Ye et al., 2022). In the inflammatory model, agriculture consisting of neurons, astrocytes, and microglia isolated from neonatal rats faithfully mimics the neuroinflammatory response rather than mono and standard co-cultures (Chiang et al., 2022). Astrocytes culture from adult macaque brain organs have been successfully isolated and characterized to determine changes in DNA damage repair patterns due to ionization radiation at gene locus-specific and genome-wide levels (Yong et al., 2014). Macaque brain cells from fetuses aged 80 days gestation were successfully cultured by adding 10 ng/ml BDNF to the culture medium, thereby increasing the survival effect (Negishi et al., 2003). This study isolated NSCs from the hippocampus of a fetus aged 125 days of digestion, which is expected to be a source of neuron progenitor populations to become a stroke model. Animal models of ischemic stroke have also been used in small animals, such as rats and mice, as well as in non-human primates (Tuo et al., 2017; Lin et al., 2022). The issue of animal welfare is a serious concern. Animal models are sacrificed, and ligation is performed on the arteries that supply blood to the brain using the transient middle cerebral artery occlusion (MCAO) method. The use of cell lines and primary cells is projected potential research productions for neurodegenerative therapies, particularly ischemic stroke (Bai et al., 2022). As an in vitro model of stroke, OGD simulates neuronal cell damage. The time it takes for OGD to become an in vitro stroke model varies depending on the cell type; in primary culture, it takes just 2, 4, or 6 hours, whereas cell lines like SH-SY5Y require 24 hours (Blinova et al., 2022; Chiang et al., 2022; Mariana et al., 2022). Neurons isolated from the rat fetal cortex experienced significant death after 1 hour of OGD (zero glucose and 2% oxygen concentration) treatment by showing an increase in death-associated protein kinase 1 (Degregorio-Rocasolano et al., 2020). One of the method’s flaws was the inability to maintain intracellular oxygen levels even when the environment was controlled to 2–3% oxygen (Babu et al., 2022). There was an increase in pro-apoptosis gene expression after OGD/hypoxia compared with normal circumstances in the study that succeeded in creating a stroke model utilizing NSCs with the OGD approach (5% oxygen for 6 hours). Apoptosis may be responsible for most of the cell death after acute brain ischemia. Acute brain ischemia is characterized by critical chemical processes that cause apoptosis in many cells, such as free radical overproduction, Ca2+ overload, and excitotoxicity. These alterations in cellular homeostasis may result in necrosis or apoptosis, depending on cell type, age, and brain location (Broughton et al., 2009). In this study under hypoxic conditions, Bax, BCL-2, caspase-9, and p53 gene expression were increased (Fig. 3). The onset of ischemic stroke reduces substrate delivery, particularly oxygen and glucose, and compromises the energetics required to maintain neuronal ionic gradients (Broughton et al., 2009). Accelerated energy depletion during brain ischemia causes membrane potential loss and neuronal depolarization. Voltage-dependent Ca2+ channels are then triggered, resulting in the release of excitatory amino acids into the extracellular area. Following this, cytotoxic Ca2+ buildup develops, which is expected to launch a sequence of cytoplasmic and nuclear processes, including the activation of the intrinsic apoptotic pathway (Uzdensky, 2019). In this study, neurons of Mf Indonesia origin under hypoxic conditions triggered apoptosis pathways compared with neurons in normal conditions. Our study evaluated gene expressions. The use of neurons from the cortex area is predicted to be imitated in vivo settings, in which the neuron area survives OGD conditions and regenerates the infarcted area of Bax, BCL-2, caspase-9, and p53, which encode proteins involved in the intrinsic pathway of apoptosis. Based on our molecular study, we suggest that neurons should be treated with the OGD approach (5% oxygen for 6 hours) in NSCs (Wang et al., 2018). Successful use of SH-SY5Y and PC12 in an in vitro stroke model has been reported. Neural apoptosis in strokes is triggered by direct miR-186-5p suppression of insulin-like growth factor 1 (IGF-1) expression; this experiment was performed with SH-SY5Y after 10 hours of OGD. PC12 is also a model of ischemic stroke after inducing neuronal differentiation using NGF with OGD for 3 hours (Chua and Lim, 2021). The use of NSC and cell line as a stroke model has been reported in the prevention of neuronal death through inflammatory processes and the ability of NSCs to differentiate into neurons (Liu et al., 2014; Budiariati et al., 2019). The neuroprotective impact of resveratrol (48 hours) was determined by using SH-SY5Y with 1% oxygen for 24 hours. Neuronal damage is caused by elevated reactive oxygen species and inflammation after an ischemic stroke. The AMP-activated Protein Kinase (AMPK) and nuclear factor erythroid 2-related factor (Nrf2) pathways are activated, which modifies the action of antioxidants like superoxide dismutase (SOD) on ischemic stroke. By boosting Nrf2 expression, resveratrol protects against ischemia damage. This study found that resveratrol has a neuroprotective impact in the OGD model by acting as an antioxidant, lowering ROS and activating Nrf2, as well as suppressing the activation of nuclear factor-kappa B (NF-B) and NOD-, LRR-, and pyrin domain-containing protein 3 (NLRP3) inflammasome and reducing the generation of inflammatory cytokines (Chiang et al., 2022). The SH-SY5Y cell line was also used to prove the efficacy of monomethyl lithospermate (MOL) for neuroprotection and infarction reduction. MOL plays a role in increasing SH-SY5Y cell viability by inhibiting mitochondrial membrane potential, alleviating oxidative stress, and activating the fosfatidilinositol 3-kinase (PI3K)/ protein kinase B (AKT) pathway through the promotion of PI3K and AKT phosphorylation (Yang et al., 2022). These results indicate that neurons from the Macaca brain can be used for in vitro drug testing or stem cell applications in ischemic stroke through the apoptosis pathway. In this study, B27 was used to provide neuroprotection under OGD conditions ((Figure 3). The results of this study indicate that the expression of Bax, BCL-2, p53, and caspase-9 in clarified Mf NSC is correlated with B27 supplementation. B27 neuroprotection is indicated by an increase in the expression of the BCL-2 ratio. Interestingly, the mRNA expression level of p53 decreased (Figure 3). Emerging evidence has shown that p53 inhibits neuronal differentiation and reduces apoptosis (Xiong et al., 2020; Darusman et al., 2022). Oxidative cellular status, increased PI3K/Akt signaling, elevated ID1, and metabolism are implicated in p53-loss-induced neurogenesis. However, p53 has also been shown to promote neuronal differentiation. In this study, we demonstrated that a decrease in p53 gene expression in NSC cynomolgus monkeys may occur because B27 was induced (Liu et al., 2013). B27 supports nerve cell metabolic diseases in the primary culture of neurons and cell lines as an in vitro model. The effects of culture microenvironment on neuronal function are important for supporting survival and metabolic activity, especially in deficient neuronal metabolism models using OGD. Another study found that using 2% B27 and 5% FBS can create a microenvironment that preserves the neuron-like characteristics of dental pulp stem cells, as evidenced by the high levels of Nestin and MAP-2 expression (Luo et al., 2020). Under serum-free culture conditions, B27 can be used to stimulate the population of glutamatergic neurons in SH-SY5Y (Martin et al., 2022). B27 is a cofactor of ketoacid dehydrogenases, which plays a crucial role in mitochondrial energy metabolism and can inhibit glycolysis in vitro via Akt/JNK signaling. B27 is a serum-free nerve cell supplement containing a combination of vitamins, proteins, fatty acids, and antioxidants. Initially, enzymes were created to improve primary neuronal survival, as well as preserve NSC development and differentiation (Sünwoldt et al., 2017; Martin et al., 2022). Cell lines with immortal and homogenous features, such as human glioblastoma and rat pheochromocytoma, have distinct qualities from primary cells. The ability of glioblastoma (SH-SY5Y) to differentiate into neuronal cells and to become an ischemic stroke model is highly variable (Liu et al., 2018). In vivo, the condition of ischemic stroke in the brain organ is related to communication with other cells and the surrounding microenvironment, and not just neuron cells (Pei et al., 2019; Chen et al., 2020; Jiao et al., 2023). Although NSCs predominate in the isolated hippocampus, other cell types such as astrocytes, microglia, and oligodendrocytes can communicate with each other to migrate to the infarcted area (Wu et al., 2012; Budiariati et al., 2019). The ischemic stroke model of neuron Mf was expected to be a better vitro model with genetic affinity to humans. This will provide better data if neuron from Mf is used as an allogenic model of therapy compared with stroke models from rats, zebrafish, or cell lines with different genetics (Kuang et al., 2020; Rinendyaputri et al., 2023). Neurons may be developed into a cell line and made into a stock cell line, and further research is needed to prove this, but from this research, neurons from Mf can be used as an ischemic stroke model. ConclusionNeurons were effectively extracted and employed as an in vitro ischemic stroke model. The use of B27 supplementation in stroke models with OGD induction in NSCs results in an increase in neuroprotective activity; therefore, it should be evaluated when used for in vitro studies in both drug screening and neurodegenerative physiopathology, particularly in stroke. AcknowledgementsWe thank Prof. Dr. NLP Indi Darmayati, M.Biomed, and Prof. Sunarno who supported this research, team who helped obtain data and technical support for this research, and Department of Medicine Science, Padjadjaran University. Conflict of interestsThe authors declare that there is no conflict of interest. FundingThis research was funded by the Rumah Program Research Organization for Health, National Research and Innovation Agency (BRIN). Authors’ contributionsWireni Ayuningtyas was corresponding for the treatment and obtaining the NSC samples. Ratih Rinendyaputri was corresponding for PCR and Ariyani Noviantari for staining of NSC. Article writing was done equally by the authors. Data availabilityAll data are provided in the manuscript. ReferencesBabu, M., Singh, N. and Datta, A. 2022. In vitro oxygen-glucose deprivation model of ischemic stroke: a proteomics-driven systems biological perspective. Mol. Neurobiol. 59(4), 2363–2377. Bai, S., Lu, X., Pan, Q., Wang, B., Pong U,K., Yang, Y., Wang, H., Lin, S., Feng, L., Wang, Y., Li, Y., Lin, W., Wang, Y., Zhang, X., Li, Y., Li, L., Yang, Z., Wang, M., Lee, W.Y.W., Jiang, X. and Li, G. 2022. Cranial bone transport promotes angiogenesis, neurogenesis, and modulates meningeal lymphatic function in middle cerebral artery occlusion rats. Stroke 53(4), 1373–1385. Blinova, E., Turovsky, E., Eliseikina, E., Igrunkova, A., Semeleva, E., Golodnev, G., Termulaeva, R., Vasilkina, O., Skachilova, S., Mazov, Y., Zhandarov, K., Simakina, E., Belanov, K., Zalogin, S. and Blinov, D. 2022. A novel hydroxypyridine compound protects brain cells against ischemic damage in vitro and in vivo. Int. J. Mol. Sci. 23(21), 1–19. Broughton, B.R.S., Reutens, D.C. and Sobey, C.G. 2009. Apoptotic mechanisms after cerebral ischemia. Stroke 40(5), 1–9. Budiariati, V., Rinendyaputri, R., Noviantari, A., Budiono, D., Fahrudin, M., Juliandi, B. and Boediono, A. 2019. Heterogeneity of cell populations and secretome profiles of differentiated cells from E17 rat neural progenitor cells. J.Stem Cells Regen. Med. 15(2), 35–45. Chen, W., Zheng, P., Hong, T., Wang, Y., Liu, N., He, B., Zou, S., Ren, D., Duan, J., Zhao, L. and Feng, J. 2020. Astrocyte-derived exosomes induce neuronal recovery after TBI by delivering gap junction alpha 1-20 k. J. Tissue Eng. Regen. Med. 14, 412–423. Chiang, M.C., Nicol, C.J.B., Lo, S.S., Hung, S.W., Wang, C.J. and Lin, C.H. 2022. Resveratrol mitigates oxygen- and glucose deprivation-induced inflammation, NLRP3 inflammasome, and oxidative stress in 3D neuronal culture. Int. J. Mol. Sci. 23(19), 1–20. Chua, P.F. and Lim, W.K. 2021. Optimisation cell-based in vitro ischemic stroke model for screening neuroprotective agents. Sci. Rep. 11(1), 1–15. Darusman, H.S., Mariya, S.S., Sari, I.K., Nisa, M.A., Sari, K., Mariya, S., Mustopa, A.Z. and Saepuloh, U. 2022. Spontaneous expression of KI67 and P53 genes in cynomolgus monkeys infected with papillomavirus. Vet. World 15(4), 962–967. Degregorio-Rocasolano, N., Guirao, V., Ponce, J., Melià-Sorolla, M., Aliena-Valero, A., García-Serran, A., Salom, J.B., Dávalos, A., Martí-Sistac, O. and Gasull, T. 2020. Comparative proteomics unveils lrrfip1 as a new player in the dapk1 interactome of neurons exposed to oxygen and glucose deprivation. Antioxidants 9(12), 1–21. Gibson, C.L. and Trotman-Lucas, M. 2021. A review of experimental models of focal cerebral ischemia, focusing on the middle cerebral artery occlusion model. F1000Res. 10, 1-17. Goshi, N., Morgan, R.K., Lein, P.J. and Seker, E. 2020. A primary neural cell culture model to study neuron, astrocyte, and microglial interactions in neuroinflammation. J. Neuroinflammation. 17(155), 1–16. Jiao, Q., Wang, L., Zhang, Z., Chen, X., Lu, H. and Liu, Y. 2023. The biological behaviors of neural stem cells affected by the microenvironment of host organotypic brain slices under different conditions. Int. J. Mol. Sci. 24(4), 1–13. Kuang, Y., Zheng, X., Zhang, L., Ai, X., Venkataramani, V., Kilic, E., Hermann, D.M., Majid, A., Bähr, M. and Doeppner, T.R. 2020. Adipose-derived mesenchymal stem cells reduce autophagy in ischemic stroke mice by extracellular vesicle transfer of miR-25. J. Extracell. Vesicles. 10(1), 1–20. Kuriakose, D. and Xiao, Z. 2020. Pathophysiology and treatment of stroke: present status and future perspectives. Int. J. Mol. Sci. 21(20), 1–24. Laila, F., Fardiaz, D., Yuliana, N.D., Damanik, M.R.M., and Nur Annisa Dewi, F. 2020. Methanol Extract of Coleus amboinicus (Lour) Exhibited Antiproliferative Activity and Induced Programmed Cell Death in Colon Cancer Cell WiDr. Int J Food Sci. 1–12 Li, Y., Yang, J., Fu, G., Zhou, P., Liu, Y., Li, Z. and Jiao, G. 2020. Human umbilical cord mesenchymal stem cells differentiate into neuron-like cells after induction in B27-supplemented serum-free medium. Nan Fang Yi Ke Da Xue Xue Bao 40(9), 1340–1345. Lin, X., Wang, H., Chen, J., Zhao, P., Wen, M., Bingwa, L.A., Jin, K., Zhuge, Q. and Yang, S. 2022. Nonhuman primate models of ischemic stroke and neurological evaluation after ischemic stroke. J. Neuroscience Methods. 376 (2022), 1–12. Liu, H., Jia, D., Li, A., Chau, J., He, D., Ruan, X., Liu, F., Li, J., He, L. and Li, B. 2013. P53 regulates neural stem cell proliferation and differentiation via bmp-smad1 signaling and ID1. Stem Cells Dev. 22(6), 913–927. Liu, Q., Fan, X., Zhu, J., Xu, G., Li, Y. and Liu, X. 2014. Coculturing improves OGD-injured neuronal repair and NSC differentiation via Notch pathway activation. Neurosci. Lett. 559, 1–6. Liu, Y., Eaton, E.D., Wills, T.E., McCann, S.K., Antonic, A. and Howells, D.W. 2018. Human ischemic cascade studies using SH-SY5Y cells: a systematic review and meta-analysis. Transl. Stroke Res. 9(6), 564–574. Luo, L., Wang, X., Zhang, Y., Wu, Y., Hu, F., Xing, Z., Wang, L., Xiao, J., Guastaldi, F., He, Y. and Ye, Q. 2020. Biological behavioral alterations of postneural differentiated dental pulp stem cells through an in situ microenvironment. Front. Cell Dev. Biol. 8, 1–13. Mariana, M., Roque, C., Baltazar, G. and Cairrao, E. 2022. In vitro model of ischemic stroke: functional analysis of vascular smooth muscle cells. Cell Mol. Neurobiol. 42(7), 2289–2304. Mariya, S.S., Dewi, F.N., Villiandra, V., Paramastri, Y.A., Iskandriati, D., Saepuloh, U., Hayes, E., Pamungkas, J. and Sajuthi, D. 2019. Isolation and characterization of c-c chemokine ligand 7 (Ccl7) in cynomolgus macaques. Hayati 26(3), 129–132. Martin, E.R., Gandawijaya, J. and Oguro-Ando, A. 2022. A novel method for generating glutamatergic SH-SY5Y neuron-like cells utilizing B-27 supplement. Front. Pharmacol. 13, 1–20. Namekawa, T., Ikeda, K., Horie-Inoue, K. and Inoue, S. 2019. Application of prostate cancer models in preclinical studies: advantages and limitations of cell lines, patient-derived xenografts, and three-dimensional culture of patient-derived cells. Cells 8(74), 1–24. Negishi, T., Ishii, Y., Kyuwa, S., Kuroda Y. and Yoshikawa Y 2003. Primary culture of cortical neurons, type 1 astrocytes, and microglial cells from cynomolgus monkey (Macaca fascicularis) fetuses. J. Neurosci. Methods 131(1–2): 133–140. Noviantari, A., Antarianto, R.D., Rif’ati, L., Rinendyaputri, R., Zainuri, M. and Dany, F. 2020. Expression of nestin during differentiation into neurons of rat bone marrow mesenchymal stem cells by neurotrophin-3 (NT-3). Int. J. Appl. Pharm. 12(3), 44–49. Pei, X., Li, Y., Zhu, L. and Zhou, Z. 2019. Astrocyte-derived exosomes suppress autophagy and ameliorate neuronal damage in experimental ischemic stroke. Exp. Cell. Res. 382(2), 111–174. Peng, Y., Chu, S., Yang, Y., Zhang, Z., Pang, Z. and Chen, N. 2021. Neuroinflammatory in vitro cell culture models and their potential applications for neurological disorders. Front. Pharmacol. 12, 1–13. Rinendyaputri, R., Lienggonegoro, L.A., Idrus, H.H., Noverina, R. and Faried, A. 2023. Role of neuroprotection in zebrafish (Danio rerio) animal model. AIP Conf. Proc. 2956(1), 1–8. Sünwoldt, J., Bosche, B., Meisel A. and Mergenthaler, P 2017. Neuronal culture microenvironments determine preferences in bioenergetic pathway use. Front. Mol. Neurosci. 10, 1–11. Tuo, Q.Z., Lei, P., Jackman, K.A., Li, X.L., Xiong, H., Li, X.L., Liuyang, Z.Y., Roisman, L., Zhang, S.T., Ayton, S., Wang, Q., Crouch, P.J., Ganio, K., Wang, X.C., Pei, L., Adlard, P.A., Lu, Y.M., Cappai, R., Wang, J.Z., Liu, R. and Bush, A.I. 2017. Tau-mediated iron export prevents ferroptotic damage after ischemic stroke. Mol. Psychiatry. 22(11), 1520–1530. Uzdensky, A.B. 2019. Apoptosis regulation in the penumbra after ischemic stroke: expression of pro- and antiapoptotic proteins. Apoptosis 24, 687–702. Vagaska, B., Gillham, O. and Ferretti, P. 2020. Modeling human CNS injury using human neural stem cells in 2- and 3-Dimensional cultures. Sci. Rep. 10(1), 1–14. Wang, R., Bao, H., Zhang, S., Li, R., Chen, L. and Zhu, Y. 2018. Mir-186-5p promotes apoptosis by targeting IGF-1 in the SH-SY5Y OGD/R model. Int. J. Biol. Sci. 14(13), 1791–1799. Wohnsland, S., Bürgers, H.F., Kuschinsky, W. and Maurer, M.H. 2010. Neurons and neuronal stem cells survive in glucose-free lactate and high-glucose cell culture medium during normoxia and anoxia. Neurochem. Res. 35(10), 1635–1642. Wu, Y., Chen, Q., Peng, H., Dou, H., Zhou, Y., Huang, Y. and Zheng, J.C. 2012. The direct migration of human neural progenitor cells to interleukin-1β is promoted by chemokines stromal cell-derived factor-1 and monocyte chemotactic factor-1 in mouse brains. Transl. Neurodegener. 1(15), 1–111. Xiong, Y., Zhang, Y., Xiong, S. and Williams-Villalobo, A.E. 2020. Figure 1. Overview of p53 functions in brain development, neural stem cells, and brain cancer. Biology (Basel) 1–13. Yang, F., Chen, Z.R., Yang, X.H., Xu, Y., Ran, N.J., Liu, M.J., Jin, S.G., Jia, H.N. and Zhang, Y. 2022. Monomethyl lithospermate alleviates ischemic stroke injury in middle cerebral artery occlusion mice in vivo and protects oxygen glucose-deprivation/reoxygenation-induced SHSY-5Y cells in vitro via activation of PI3K/Akt signaling. Front. Pharmacol. 13(13), 1–14. Ye, Y., Feng, Z., Tian, S., Yang, Y., Jia, Y., Wang, G., Wang, J., Bai, W., Li, J. and He, X. 2022. HBO alleviates neural stem cell pyroptosis via the H19/miR-423-5p/NLRP3 axis and improves neurogenesis after oxygen glucose deprivation. Oxid. Med. Cell Longev. 7, 1–15. Yong, R.L., Yang, C., Lu, J., Wang, H., Schlaff, C.D., Tandle, A., Graves, C.A., Elkahloun, A.G., Chen, X., Zhuang, Z. and Lonser, R.R. 2014. Cell transcriptional state alters the genomic patterns of DNA double-strand break repair in human astrocytes. Nat. Commun. 17(5), 1–12. Zhang, T., Liu, W., Yang, J., Xu, H., Cao, Y., Guo, L., Sun, J., Liang, B., Du, X., Chai, L., Yuan, Q. and Hu, L. 2022. Components of Salvia miltiorrhiza and Panax notoginseng protect pericytes against OGD/R-induced injury by regulating the PI3K/AKT/mTOR and JNK/ERK/P38 signaling pathways. J. Mol. Neurosci. 72(12), 2377–2388. Zhao, Y., Zhang, X., Chen, X. and Wei, Y.U.N. 2022. Neuronal injuries in cerebral infarction and ischemic stroke: mechanisms to treatment (Review). Int. J. Mol. Med. 49(15), 1–9. | ||
| How to Cite this Article |
| Pubmed Style Rinendyaputri R, Mariya SS, Dany F, Noviantari A, Budiono D, Lienggonegoro LA, Idrus HH, Ayuningtyas W, Noverina R, Huda F, Faried A. In vitro model of ischemic stroke neurons from Macaca fascicularis. Open Vet. J.. 2025; 15(2): 804-812. doi:10.5455/OVJ.2025.v15.i2.29 Web Style Rinendyaputri R, Mariya SS, Dany F, Noviantari A, Budiono D, Lienggonegoro LA, Idrus HH, Ayuningtyas W, Noverina R, Huda F, Faried A. In vitro model of ischemic stroke neurons from Macaca fascicularis. https://www.openveterinaryjournal.com/?mno=226601 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.29 AMA (American Medical Association) Style Rinendyaputri R, Mariya SS, Dany F, Noviantari A, Budiono D, Lienggonegoro LA, Idrus HH, Ayuningtyas W, Noverina R, Huda F, Faried A. In vitro model of ischemic stroke neurons from Macaca fascicularis. Open Vet. J.. 2025; 15(2): 804-812. doi:10.5455/OVJ.2025.v15.i2.29 Vancouver/ICMJE Style Rinendyaputri R, Mariya SS, Dany F, Noviantari A, Budiono D, Lienggonegoro LA, Idrus HH, Ayuningtyas W, Noverina R, Huda F, Faried A. In vitro model of ischemic stroke neurons from Macaca fascicularis. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 804-812. doi:10.5455/OVJ.2025.v15.i2.29 Harvard Style Rinendyaputri, R., Mariya, . S. S., Dany, . F., Noviantari, . A., Budiono, . D., Lienggonegoro, . L. A., Idrus, . H. H., Ayuningtyas, . W., Noverina, . R., Huda, . F. & Faried, . A. (2025) In vitro model of ischemic stroke neurons from Macaca fascicularis. Open Vet. J., 15 (2), 804-812. doi:10.5455/OVJ.2025.v15.i2.29 Turabian Style Rinendyaputri, Ratih, Sela Septima Mariya, Frans Dany, Ariyani Noviantari, Dwi Budiono, Lisa Andriani Lienggonegoro, Hasta Handayani Idrus, Wireni Ayuningtyas, Rachmawati Noverina, Fathul Huda, and Ahmad Faried. 2025. In vitro model of ischemic stroke neurons from Macaca fascicularis. Open Veterinary Journal, 15 (2), 804-812. doi:10.5455/OVJ.2025.v15.i2.29 Chicago Style Rinendyaputri, Ratih, Sela Septima Mariya, Frans Dany, Ariyani Noviantari, Dwi Budiono, Lisa Andriani Lienggonegoro, Hasta Handayani Idrus, Wireni Ayuningtyas, Rachmawati Noverina, Fathul Huda, and Ahmad Faried. "In vitro model of ischemic stroke neurons from Macaca fascicularis." Open Veterinary Journal 15 (2025), 804-812. doi:10.5455/OVJ.2025.v15.i2.29 MLA (The Modern Language Association) Style Rinendyaputri, Ratih, Sela Septima Mariya, Frans Dany, Ariyani Noviantari, Dwi Budiono, Lisa Andriani Lienggonegoro, Hasta Handayani Idrus, Wireni Ayuningtyas, Rachmawati Noverina, Fathul Huda, and Ahmad Faried. "In vitro model of ischemic stroke neurons from Macaca fascicularis." Open Veterinary Journal 15.2 (2025), 804-812. Print. doi:10.5455/OVJ.2025.v15.i2.29 APA (American Psychological Association) Style Rinendyaputri, R., Mariya, . S. S., Dany, . F., Noviantari, . A., Budiono, . D., Lienggonegoro, . L. A., Idrus, . H. H., Ayuningtyas, . W., Noverina, . R., Huda, . F. & Faried, . A. (2025) In vitro model of ischemic stroke neurons from Macaca fascicularis. Open Veterinary Journal, 15 (2), 804-812. doi:10.5455/OVJ.2025.v15.i2.29 |