| Research Article | ||
Open Vet. J.. 2025; 15(2): 827-834 Open Veterinary Journal, (2025), Vol. 15(2): 827-834 Research Article Clinical outcomes and prognostic factors of repeated conventional hypofractionated radiotherapy for recurrent intranasal tumors in dogsHirona Ueno1, Yuta Nishiyama2, Takuya Maruo1*, Yohei Fukuda1, Takayuki Katayama1, Shinichiro Yoda1, Kotaro Nishi2 and Hideki Kayanuma11Laboratory of Veterinary Radiology, School of Veterinary Medicine, Azabu University, Sagamihara, Japan 2Veterinary Teaching Hospital, Azabu University, Sagamihara, Japan *Corresponding Author: Takuya Maruo. Laboratory of Veterinary Radiology, School of Veterinary Medicine, Azabu University, Sagamihara, Japan. Email: maruo [at] azabu-u.ac.jp Submitted: 30/10/2024 Accepted: 21/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
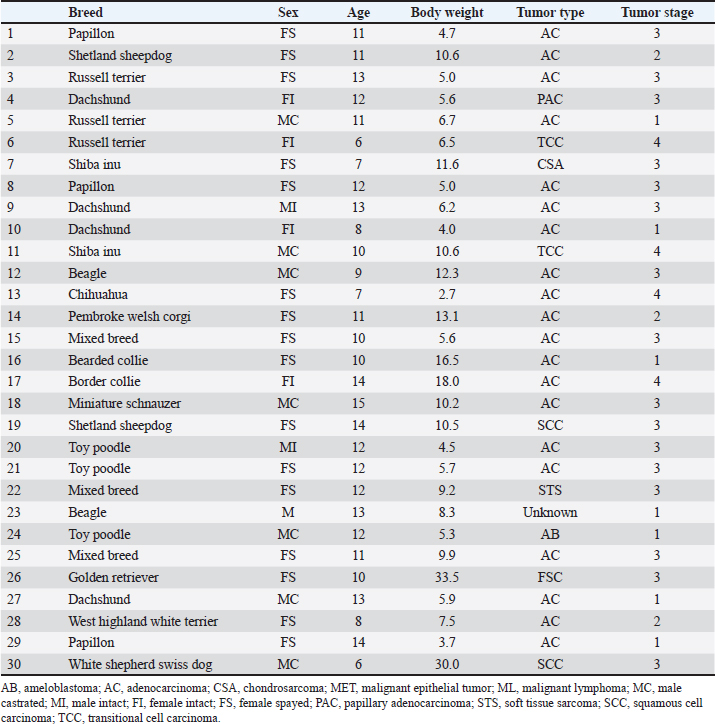
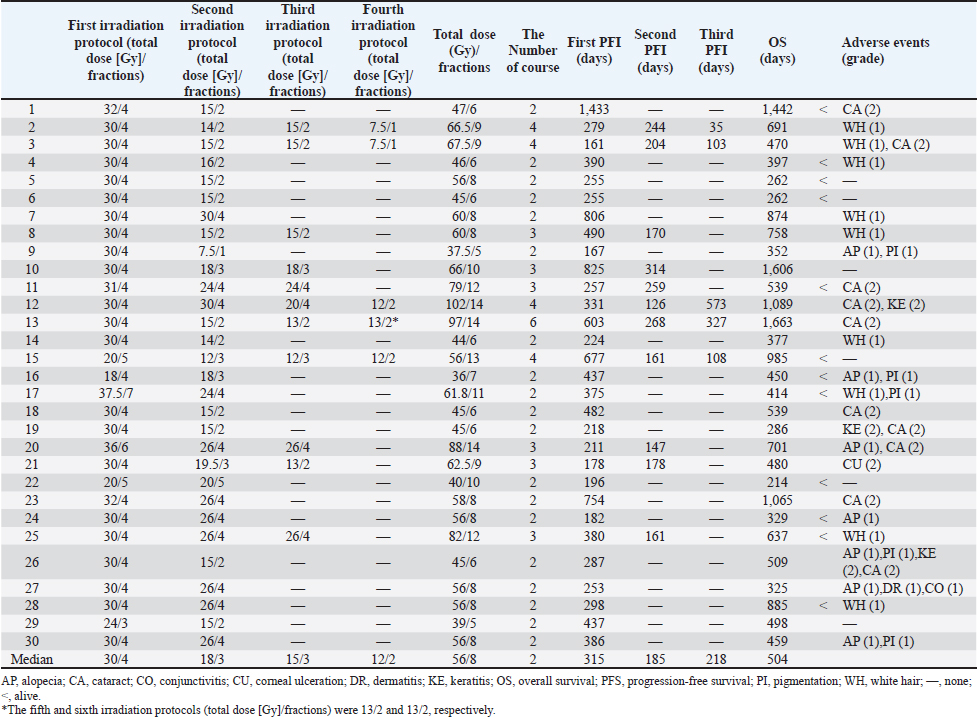
AbstractBackground: Despite an initial favorable response to hypofractionated radiotherapy (RT), intranasal neoplasia has a poor long-term prognosis. Re-irradiation (re-RT) has been shown to result in clinical remission in dogs with recurrent intranasal tumors. Hypofractionated re-RT is indicated in cases of recurrence. Aim: This study aimed to investigate the response of patients with recurrent intranasal tumors who were initially treated with repeated hypofractionated RT and subsequently underwent re-RT. Methods: Medical records of dogs with intranasal tumors that relapsed after undergoing hypofractionated RT at Azabu University Veterinary Teaching Hospital and who underwent re-RT between 2008 and 2022 were retrospectively analyzed. Results: Among the 30 dogs included, 20 had adenocarcinomas. Eleven patients underwent a second re-RT, and five patients underwent a third re-RT. The median total isocenter dose and total number of fractions were 56 Gy (range, 37.5–102) and 8 fractions (range, 5–14), respectively. The first, second, and third progression-free intervals (PFIs) were 315 (n=30), 185 (n=11), and 218 days (n=4), respectively. Overall survival (OS) was 504 days (n=30). Adverse events such as alopecia (n=7), white hair (n=8), cataracts (n=9), pigmentation (n=5), keratitis (n=3), and corneal ulceration (n=1) were limited to grades 1 and 2. The medians of the first PFI for one course and two or more courses of re-RT were 298 and 331 days, respectively, indicating no significant differences between the groups. The OS after two or more courses of re-RT (758 days) was significantly longer than after one course of re-RT alone (414 days) (p=0.048). However, the cases were not randomized, and most of the tumor types were adenocarcinomas. Conclusion: Since it is expected to prolong survival time with few life-threatening adverse events, the findings of the present study suggest that hypofractionated re-RT can be used in dogs with recurrent intranasal tumors initially treated with hypofractionated RT. Therefore, repeated hypofractionated re-RT may be a reasonable option for young dogs with a good initial response and recurrent intranasal tumors. Keywords: Dog, Hypofractionated radiotherapy, Intranasal tumor, Recurrence, Re-irradiation. IntroductionTumors of the intranasal cavity and paranasal sinuses, which account for approximately 1% of all neoplasms in dogs (Dorn et al., 1968; Lana and Turek, 2020), are locally invasive and associated with a low rate of metastasis. Consequently, local treatment options, such as surgery (Adams et al., 2005; Bowles et al., 2016), photodynamic therapy (Lucroy et al., 2003; Osaki et al., 2009; Maruo et al., 2019), cryotherapy (Murphy et al., 2011), electrochemotherapy (Maglietti et al., 2017; Suzuki et al., 2017), and radiotherapy (RT) (Lana and Turek, 2020), have been recommended for the management of these tumors (Turek, 2008). RT is the first choice for the treatment of nasal tumors (Turek, 2008). RT can be divided into hypofractionated and fractionated RT based on the number of fractions, and previous studies comparing treatment efficacy according to the number of fractions have revealed that the progression-free interval (PFI) of hypofractionated RT is shorter than that of fractionated RT (Nolan and Dobson, 2018). Hypofractionated RT is often preferred for the treatment of dogs and cats because of practical considerations, such as reducing the number of anesthesia sessions required (Fujiwara et al., 2013). The resolution of clinical signs after RT has been observed in most dogs with sinonasal neoplasia; however, the long-term prognosis remains poor (Lana and Turek, 2020). Furthermore, recurrence accounts for the majority of deaths, even after undergoing RT (Adams et al., 2005, 2009; Bommarito et al., 2011; Gieger et al., 2013, 2022; Bowles et al., 2016). Re-irradiation (re-RT), fractionated re-RT (Bommarito et al., 2011), or hypofractionated re-RT (Gieger et al., 2013, 2022; Tan-Coleman et al., 2013; Rancilio et al., 2016), has been shown to result in clinical remission in dogs with recurrent intranasal tumors. However, the previously treated volume (amount of overlap with new treatment fields), dose fractionation schedule, critical tissues or organs at risk, duration since initial treatment, and feasible alternatives to re-RT must be considered before re-RT to prevent serious adverse events (Stewart, 1999). In a previous study, the PFI in dogs treated with hypofractionated RT was shorter than that of dogs treated with fractionated RT (Lana and Turek, 2020). Therefore, re-RT may be necessary after palliative hypofractionated RT. However, the prognostic factors for re-RT are not yet well defined. Furthermore, although repeated hypofractionated re-RT has been investigated in humans and is hypothesized to prolong survival, there are no corresponding reports in dogs (Notter et al., 2017). Therefore, this study aimed to clarify the response and clinical efficacy of repeated hypofractionated re-RT and to identify prognostic factors for re-RT. Materials and MethodsThe owners of the dogs were informed of the potential risk of serious radiation-related adverse effects associated with re-RT, and their consent was obtained before proceeding with the treatment. Medical records of dogs with intranasal tumors that relapsed after undergoing hypofractionated RT at Azabu University Veterinary Teaching Hospital and underwent re-RT between 2008 and 2022 were retrospectively analyzed. Data on breed, sex, age, body weight, tumor type, and tumor stage were extracted from the records. Furthermore, data on the following treatment-related factors were also collected: number of courses; initial RT protocol (total dose and number of fractions); first re-RT protocol (total dose and number of fractions); second re-RT protocol (number of cases, total dose, and number of fractions); third re-RT protocol (number of cases, total dose, and number of fractions); median total tumor dose (Gy); median total dose to the ipsilateral and contralateral eyes; total isocenter dose (Gy); total number of fractions; the first, second, and third PFIs; overall survival (OS); and incidence of adverse events. The first PFI was defined as the interval between the start of the first RT course and the start of the second RT course. The second PFI was defined as the interval between the start of the second RT course (re-RT) and the commencement of the third RT course. The third PFI was defined as the interval between the initiation of the third course of RT (second re-RT) and the commencement of the fourth course. OS was defined as the duration between the start of the first course of RT and death. Patients with symptoms, such as nasal bleeding or obstruction, underwent follow-up computed tomography (CT) examination. Recurrence was determined according to the presence of symptoms and tumor growth on CT. Dogs who had undergone ≥1 re-RT and completed the first course of RT were eligible for inclusion in this study. Because the risk of complications was significantly lower in dogs when the interval between the first and second courses of RT was >5 months, the exclusion criterion was an interval of <150 days to re-RT (Turrel and Théon, 1988). The tumor stage was classified according to the modified Adams staging criteria (Adams et al., 2009). Radiation morbidity scoring criteria from the Veterinary Radiation Therapy Outcome Guidelines were used to determine toxicity (Ladue et al., 2001). Treatment began with subcutaneous administration of 0.025 mg/kg of atropine sulfate hydrate (Atropine Sulfate Injection; Mitsubishi Tanabe Pharma Co., Osaka, Japan) 15 minutes before induction of anesthesia. Intubation was performed after intravenous administration of 6–8 mg/kg propofol (Rapinovet; Takeda Schering-Plough Animal Health Corporation, Osaka, Japan) or alphaxalone (Alfaxan; dog, 3 mg/kg; Meiji Seika Pharma Co., Ltd., Tokyo, Japan). Isoflurane (Mylan Inc., Osaka, Japan) was used to induce anesthesia. Head positioning devices have also been used (Mori et al., 2009; Maruo et al., 2013; Nemoto et al., 2015). Re-RT was performed using a CT scanner (Asteon; Canon, Tokyo, Japan) with RT planning software (XiO; Elekta, Tokyo, Japan) and a 6-MV linear accelerator (Primus; Canon, Japan). A multileaf collimator with a width of 1 cm at the isocenter was used for beam shaping. The internal and set-up margins must be kept in a fixed position to facilitate the delivery of a precise dose to the entire tumor. However, because a wide irradiation margin can impair surrounding normal tissues, the irradiation margin was set at 0 mm in this study so that the planning target volume (PTV) was equal to the gross tumor volume (GTV). To correct positioning errors, CT images were acquired from the opposite side of the linear accelerator across the treatment couch prior to each treatment session. A new plan was formulated if the tumor volume showed significant changes. The clinical target volume (CTV) was defined as the GTV/mass lesion in this study, as the nasal cavity was surrounded by bone, and a 5-mm margin was secured in the cranial and caudal directions. The PTV for the first course of RT was set at an isotropic expansion of 5 mm from the CTV. The PTV for re-RT was set at a 0-mm margin. No bolus was administered during re-RT. Statistical analysisKaplan–Meier survival curves were constructed to calculate PFI and OS. Patients who survived or were lost to follow-up were also excluded. Age, body weight, tumor stage, and first PFI were compared between categories using log-rank tests to determine prognostic factors. Age, body weight, and first PFI were classified into two groups based on their respective median values. Furthermore, the study compared the duration of the first PFI and OS between the two groups, defined by the number of re-RT courses received: one group underwent a single course of re-RT, and the other underwent two or more courses of re-RT. Log-rank tests were used to assess the statistical significance of differences in the first PFI and OS between the two groups. A p-value of <0.05 was considered statistically significant. All statistical analyses were performed using the Statistical Package for the Social Sciences version 28 (IBM Corp., Armonk, NY). ResultsThirty dogs were included in this study. The signalments of the dogs included in this study are summarized in Table 1. The predominant tumor type was adenocarcinoma (n=20), and a significant proportion of cases were stage III (Table 2). The median total dose and number of administered fractions were as follows: during the initial course of RT, the protocol was 30 Gy in 4 fractions. The median total dose and number of fractions for the first, second, and third courses of re-RT were 18 Gy in 3 fractions, 15 Gy in 3 fractions, and 12 Gy in 2 fractions, respectively. The median total mean tumor dose, total dose to the affected eye, and total dose to the ipsilateral eye were 45.6 Gy (range, 32.3–70), 17 Gy (range, 0.1–40.7), and 11.8 Gy (range, 0.1–22.3), respectively. The median PFIs for the first, second, and third re-RTs were 315 (n=30), 185 (n=11), and 218 (n=4) days, respectively. The median OS was 504 days. Early adverse events of re-RT included alopecia (grade 1, n=7). Late adverse events of re-RT included skin problems, such as white hair (grade 1, n=8) and pigmentation (grade 1, n=5), and vision changes, including cataracts (grade 2, n=9), keratitis (grade 2, n=3), and corneal ulceration (grade 2, n=1). Table 1. Summary of signalments.
Table 2. Summary of treatments.
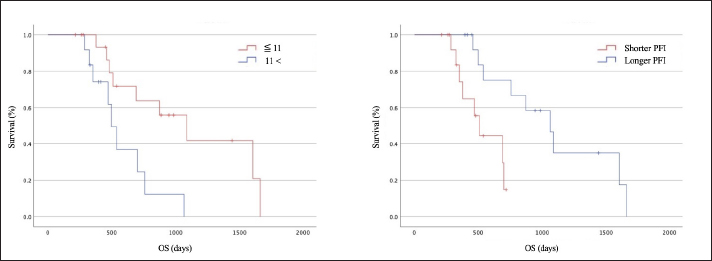
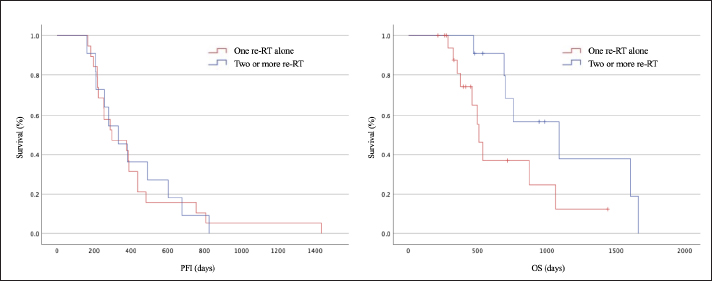
The survival periods of younger dogs (≤11 years) and those with longer PFIs were longer (p =0.009 and p=0.005, respectively; Fig. 1). Body weight, tumor stage, and re-RT fraction were not significantly associated with survival. The median PFIs after one course of re-RT alone and two or more courses of re-RT were 298 and 331 days, respectively (Fig. 2, left), indicating no significant differences between the groups. The median OS after a single course of re-RT alone and two or more courses of re-RT was 414 and 758 days, respectively (Fig. 2, right), indicating that the OS of patients who underwent two or more courses of re-RT was significantly longer than those who underwent one course of re-RT alone (p=0.048).
Fig. 1. The median OS of young dogs (≤11 years old) (red) and old dogs (≥12 years old) (blue) were 1,089 and 498 days, respectively (left). The OS of young dogs (red) was significantly longer than that of old dogs (blue) (p=0.009) (right). The median OS of the shorter PFI group (red) and longer PFI group (blue) were 509 and 1,065 days, respectively. The OS was significantly longer for the longer PFI group (blue) than for the shorter PFI group (red) (p=0.006). OS, overall survival; PFI, first progression-free interval.
Fig. 2. The median first PFIs of one course of re-RT alone (red) and ≥2 courses of re-RT (blue) were 298 and 331 days, respectively (left). No significant differences were observed between the two groups (right). The median OS of one course of re-RT alone (red) and ≥2 courses of re-RT (blue) were 414 and 758 days, respectively. The OS of ≥2 courses of re-RT (blue) was significantly longer than that of one course of re-RT alone (red) (p=0.048). OS, overall survival; PFI, first progression-free interval; RT, radiotherapy. DiscussionHypofractionated re-RT was performed for recurrent intranasal tumors after hypofractionated RT in dogs. In veterinary medicine, re-RT can be performed using fractionated (Turrel and Théon, 1988; Bommarito et al., 2011) or hypofractionated protocols (Gieger et al., 2013, 2022; Tan-Coleman et al., 2013; Rancilio et al., 2016;). A median total dose of 36 Gy was administered in 2-Gy fractions in a previous fractionated protocol study; the PFI was 282 days (Bommarito et al., 2011). In another study of the hypofractionated protocol, the median total dose for re-RT was 20 Gy (median dose per fraction, 8 Gy), and the PFI was 180 days (Gieger et al., 2013). The median total dose for the first re-RT was 18 Gy, with a median of 3 fractions. The dogs survived >6 months after re-RT. Therefore, re-RT with a small number of fractionations may be a reasonable option in certain situations. In previous studies, most intranasal tumors recurred after re-RT (Bommarito et al., 2011; Gieger et al., 2013), and three of 37 dogs underwent a third course of RT in one study (Gieger et al., 2013). However, these findings have not yet been validated. Eleven of the 30 dogs underwent two or more courses of re-RT in the present study. A comparison between the first PFI of dogs that received a single course of re-RT and those who received two or more courses revealed that OS was significantly longer in the group that underwent repeated re-RT (p=0.048). The findings of this study suggests that repeated hypofractionated re-RT may be a viable option, even with a small number of fractions. The CTV must be established when performing RT (ICRU Report 62, 1999; ICRU Report 50, 1993). The CTV was calculated using the GTV and the surrounding tumor invasion (margin) (ICRU Report 62, 1999; ICRU Report 50, 1993). In previous reports, the irradiation margin was often set at 1 cm, and no significant differences were observed in the treatment effect between tumors (Benson et al., 2017; Gieger et al., 2022). In this study, the CTV was equal to the GTV in all patients, and the PTV margin was set at 5 mm for the first course. Adenocarcinoma (n=20) was the most frequently observed tumor in this study; treatment was effective even 150 days after the end of the first course, with a margin of 0.5 cm. However, this margin may be insufficient for other types of tumors, such as squamous cell carcinoma, resulting in lower efficacy. Therefore, OS can be extended by increasing the margins of invasive tumors and ensuring that the prescribed dose is delivered to the CTV. Several cases of adenocarcinoma were included in the present study. Therefore, RT may be more effective in the treatment of adenocarcinomas, which are less invasive than other tumors. The adverse events associated with re-RT observed in this study were not severe. Retreatment schedules with curative intent require high-dose RT. However, this is associated with an increased risk of late response damage to normal tissues (Stewart, 1999). The risk of complications was significantly lower in cats and dogs when the interval between the first and second courses of RT was > 5 months (Turrel and Théon, 1988). Incidences of keratoconjunctivitis sicca, corneal ulceration, and cataracts have been reported after re-RT for intranasal tumors (Gieger et al., 2013). Most of the adverse events associated with re-RT observed in this study were grades 1 or 2, which can be attributed to the re-RT margin set at 0 mm and the interval until re-RT of ≥150 days. This study has several limitations. First, the sample size is small. Second, the re-RT protocol was not uniform, and prognostic factors could not be determined because the grouping criteria for one course of re-RT alone and two or more courses of re-RT were unclear. Third, the response to treatment was not classified in this study. Because the re-RT margin was 0 mm, the treatment effect and adverse events were likely to be less than expected with a wider margin. Fourth, the incidence of minor adverse events may not have been recorded due to the retrospective nature of this study. Finally, as most of the tumors included in the study were adenocarcinomas, an unintended case-selection bias may have been introduced. The results of this study suggest that hypofractionated re-RT may help prolong OS, particularly in younger patients (<11 years) and those who show a favorable response to initial treatment. Furthermore, this study indicates that adenocarcinomas may have a lower CTV than other tumors and that repeated hypofractionated re-RT may be a reasonable option in dogs with recurrent intranasal tumors. Conflicts of interestThe authors declare no conflict of interest. FundingThis research received no specific grant. Authors’ contributionsH.U., Y.N., and T.M. designed the study; H.U., Y.N., T.M., Y.F., T.K., S.Y., and K.N. collected the data; H.U., Y.F., and T.K. analyzed and interpreted the data; Y.N. and T.M. drafted the manuscript; T.M. and H.K. supervised and directed the study; and H.U., Y.N., T.M., Y.F., T.K., S.Y., K.N., and H.K. approved the final version for publication. Data availabilityAll data supporting the findings of this study are available within the manuscript. ReferencesAdams, W.M., Bjorling, D.E., McAnulty, J.E., Green, E.M., Forrest, L.J. and Vail, D.M. 2005. Outcome of accelerated radiotherapy alone or accelerated radiotherapy followed by exenteration of the nasal cavity in dogs with intranasal neoplasia: 53 cases (1990-2002). J. Am. Vet. Med. Assoc. 227(6), 936–941. Adams, W.M., Kleiter, M.M., Thrall, D.E., Klauer, J.M., Forrest, L.J., La Due, T.A. and Havighurst, T.C. 2009. Prognostic significance of tumor histology and computed tomographic staging for radiation treatment response of canine nasal tumors. Vet. Radiol. Ultrasound 50(3), 330–335. Benson, R., Giridhar, P., Venkatesulu, B.P., Mallick, S., Raza, M.W. and Rath, G.K. 2017. Re-irradiation for head and neck squamous cell carcinoma. J. Egypt. Natl. Cancer Inst. 29(1), 1–9. Bommarito, D.A., Kent, M.S., Selting, K.A., Henry, C.J. and Lattimer, J.C. 2011. Reirradiation of recurrent canine nasal tumors. Vet. Radiol. Ultrasound 52(2), 207–212. Bowles, K., DeSandre-Robinson, D., Kubicek, L., Lurie, D., Milner, R. and Boston, S.E. 2016. Outcome of definitive fractionated radiation followed by exenteration of the nasal cavity in dogs with sinonasal neoplasia: 16 cases. Vet. Comp. Oncol. 14(4), 350–360. Dorn, C.R., Taylor, D.O., Frye, F.L. and Hibbard, H.H. 1968. Survey of animal neoplasms in Alameda and Contra Costa Counties, California. I. Methodology and description of cases. J. Natl. Cancer Inst. 40(2), 295–305. Fujiwara, A., Kobayashi, T., Kazato, Y., Yayoshi, N. and Fujita, M. 2013. Efficacy of hypofractionated radiotherapy for nasal tumours in 38 dogs (2005–2008). J. Small Anim. Pract. 54(2), 80–86. Gieger, T., Siegel, S., Rosen, K., Jackson, D., Ware, K., Kiselow, M. and Shiomitsu, K. 2013. Reirradiation of canine nasal carcinomas treated with coarsely fractionated radiation protocols: 37 cases. J. Am. Anim. Hosp. Assoc. 49(5), 318–324. Gieger, T.L., Haney, S.M. and Nolan, M.W. 2022. Re-irradiation of canine non-lymphomatous nasal tumours using stereotactic radiation therapy (10 Gy x 3) for both courses: assessment of outcome and toxicity in 11 dogs. Vet. Comp. Oncol. 20(2), 502–508. International Commission on Radiation Units and Measurements (ICRU) Report 62. 1999. Prescribing, recording and reporting photon beam therapy (supplement to ICRU Report 50). Bethesda, MD: ICRU Publications. International Commission on Radiation Units and Measurements (ICRU) Report 50. 1993. Prescribing, recording and reporting photon beam therapy. Bethesda, MD: ICRU Publications. Ladue, T., Klein, M.K. and Veterinary Radiation Therapy Oncology Group. 2001. Toxicity criteria of the veterinary Radiation Therapy Oncology Group. Vet. Radiol. Ultrasound 42(5), 475–476. Lana, S.E. and Turek, M.M. 2020. Nasal cavity and sinus tumors. In Small animal clinical oncology, 6th ed. Eds., Vail, D.M., Thamm, D.H. and Liptak, J.M. St. Louis, MO: Saunders Elsevier, pp: 494–523. Lucroy, M.D., Long, K.R., Blaik, M.A., Higbee, R.G. and Ridgway, T.D. 2003. Photodynamic therapy for the treatment of intranasal tumors in 3 dogs and 1 cat. J. Vet. Intern. Med. 17(5), 727–729. Maglietti, F., Tellado, M., Olaiz, N., Michinski, S. and Marshall, G. 2017. Minimally invasive electrochemotherapy procedure for treating nasal duct tumors in dogs using a single needle electrode. Radiol. Oncol. 51(4), 422–430. Maruo, T., Fukuyama, Y., Nagata, K., Yoshioka, C., Nishiyama, Y., Kawarai, S., Kayanuma, H., Orito, K. and Nakayama, T. 2019. Intraoperative acridine orange photodynamic therapy and cribriform electron-beam irradiation for canine intranasal carcinomas: 14 cases. Can. Vet. J. 60(5), 509–513. Maruo, T., Nakamura, S., Fukuyama, Y. and Kawarai, S. 2013. Validation of new bite block-type head-immobilization devices for radiotherapy in dogs. Vet. Radiol. Ultrasound 54(6), 674–679. Mori, A., Shida, T., Maruo, T., Fukuyama, Y., Imai, R., Ito, T., Kayanuma, H. and Suganuma, T. 2009. Examination of the utility of a bite block-type head immobilization device in dogs and cats. J. Vet. Med. Sci. 71(4), 453–456. Murphy, S.M., Lawrence, J.A., Schmiedt, C.W., Davis, K.W., Lee, F.T. Jr, Forrest, L.J. and Bjorling, D.E. 2011. Image-guided transnasal cryoablation of a recurrent nasal adenocarcinoma in a dog. J. Small Anim. Pract. 52(6), 329–333. Nemoto, Y., Maruo, T., Fukuyama, Y., Kawarai, S., Shida, T. and Nakayama, T. 2015. A novel support device for head immobilization during radiation therapy that is applicable to both cats and dogs. Vet. Radiol. Ultrasound 56(6), 680–686. Nolan, M.W. and Dobson, J.M. 2018. The future of radiotherapy in small animals - should the fractions be coarse or fine? J. Small Anim. Pract. 59(9), 521–530. Notter, M., Piazena, H. and Vaupel, P. 2017. Hypofractionated re-irradiation of large-sized recurrent breast cancer with thermography-controlled, contact-free water-filtered infra-red-A hyperthermia: a retrospective study of 73 patients. Int. J. Hyperthermia. 33(2), 227–236. Osaki, T., Takagi, S., Hoshino, Y., Okumura, M., Kadosawa, T. and Fujinaga, T. 2009. Efficacy of antivascular photodynamic therapy using benzoporphyrin derivative monoacid ring A (BPD-MA) in 14 dogs with oral and nasal tumors. J. Vet. Med. Sci. 71(2), 125–132. Rancilio, N.J., Custead, M.R. and Poulson, J.M. 2016. Radiation therapy communication-reirradiation of a nasal tumor in a brachycephalic dog using intensity modulated radiation therapy. Vet. Radiol. Ultrasound 57(5), E46–E50. Stewart, F.A. 1999. Re-treatment after full-course radiotherapy: is it a viable option? Acta Oncol. 38(7), 855–862. Suzuki, D.O.H., Berkenbrock, J.A., de Oliveira, K.D., Freytag, J.O. and Rangel, M.M.M. 2017. Novel application for electrochemotherapy: immersion of nasal cavity in dog. Artif. Organs 41(8), 767–773. Tan-Coleman, B., Lyons, J., Lewis, C., Rosenberg, M. and Ruiz, A. 2013. Prospective evaluation of a 5 × 4 Gy prescription for palliation of canine nasal tumors. Vet. Radiol. Ultrasound 54(1), 89–92. Turek, M. M. 2008. Tumors of the respiratory system. In Decision making in small animal oncology. Eds., Argyle, D.J., Brearley, M.J. and Turek, M.M. Ames, IA: Wiley-Blackwell, pp: 239–282. Turrel, J.M. and Théon, A.P. 1988. Reirradiation of tumors in cats and dogs. J. Am. Vet. Med. Assoc. 193(4), 465–469. | ||
| How to Cite this Article |
| Pubmed Style Ueno H, Nishiyama Y, Maruo T, Fukuda Y, Katayama T, Yoda S, Nishi K, Kayanuma H. Clinical outcomes and prognostic factors of repeated conventional hypofractionated radiotherapy for recurrent intranasal tumors in dogs. Open Vet. J.. 2025; 15(2): 827-834. doi:10.5455/OVJ.2025.v15.i2.32 Web Style Ueno H, Nishiyama Y, Maruo T, Fukuda Y, Katayama T, Yoda S, Nishi K, Kayanuma H. Clinical outcomes and prognostic factors of repeated conventional hypofractionated radiotherapy for recurrent intranasal tumors in dogs. https://www.openveterinaryjournal.com/?mno=226692 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.32 AMA (American Medical Association) Style Ueno H, Nishiyama Y, Maruo T, Fukuda Y, Katayama T, Yoda S, Nishi K, Kayanuma H. Clinical outcomes and prognostic factors of repeated conventional hypofractionated radiotherapy for recurrent intranasal tumors in dogs. Open Vet. J.. 2025; 15(2): 827-834. doi:10.5455/OVJ.2025.v15.i2.32 Vancouver/ICMJE Style Ueno H, Nishiyama Y, Maruo T, Fukuda Y, Katayama T, Yoda S, Nishi K, Kayanuma H. Clinical outcomes and prognostic factors of repeated conventional hypofractionated radiotherapy for recurrent intranasal tumors in dogs. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 827-834. doi:10.5455/OVJ.2025.v15.i2.32 Harvard Style Ueno, H., Nishiyama, . Y., Maruo, . T., Fukuda, . Y., Katayama, . T., Yoda, . S., Nishi, . K. & Kayanuma, . H. (2025) Clinical outcomes and prognostic factors of repeated conventional hypofractionated radiotherapy for recurrent intranasal tumors in dogs. Open Vet. J., 15 (2), 827-834. doi:10.5455/OVJ.2025.v15.i2.32 Turabian Style Ueno, Hirona, Yuta Nishiyama, Takuya Maruo, Yohei Fukuda, Takayuki Katayama, Shinichiro Yoda, Kotaro Nishi, and Hideki Kayanuma. 2025. Clinical outcomes and prognostic factors of repeated conventional hypofractionated radiotherapy for recurrent intranasal tumors in dogs. Open Veterinary Journal, 15 (2), 827-834. doi:10.5455/OVJ.2025.v15.i2.32 Chicago Style Ueno, Hirona, Yuta Nishiyama, Takuya Maruo, Yohei Fukuda, Takayuki Katayama, Shinichiro Yoda, Kotaro Nishi, and Hideki Kayanuma. "Clinical outcomes and prognostic factors of repeated conventional hypofractionated radiotherapy for recurrent intranasal tumors in dogs." Open Veterinary Journal 15 (2025), 827-834. doi:10.5455/OVJ.2025.v15.i2.32 MLA (The Modern Language Association) Style Ueno, Hirona, Yuta Nishiyama, Takuya Maruo, Yohei Fukuda, Takayuki Katayama, Shinichiro Yoda, Kotaro Nishi, and Hideki Kayanuma. "Clinical outcomes and prognostic factors of repeated conventional hypofractionated radiotherapy for recurrent intranasal tumors in dogs." Open Veterinary Journal 15.2 (2025), 827-834. Print. doi:10.5455/OVJ.2025.v15.i2.32 APA (American Psychological Association) Style Ueno, H., Nishiyama, . Y., Maruo, . T., Fukuda, . Y., Katayama, . T., Yoda, . S., Nishi, . K. & Kayanuma, . H. (2025) Clinical outcomes and prognostic factors of repeated conventional hypofractionated radiotherapy for recurrent intranasal tumors in dogs. Open Veterinary Journal, 15 (2), 827-834. doi:10.5455/OVJ.2025.v15.i2.32 |