| Research Article | ||
Open Vet. J.. 2025; 15(2): 746-764 Open Veterinary Journal, (2025), Vol. 15(2): 746-764 Research Article A Blend of natural phytobiotics enhances growth performance, feed efficiency, and the immuno-health status of fingerlings of Nile tilapia (Oreochromis niloticus)Shreifa G. Mabrouk1, Asmaa M. El-Nokrashy1, Nahla A. Ebied2, Bahaa H. Abdella1, Mohamed M. Zayed3,4, Sayed M. Aboleila1* and Radi A. Mohamed11Department of Aquaculture, Faculty of Aquatic and Fisheries Sciences, Kafrelsheikh University, Kafr El-Sheikh, Egypt 2Food Hygiene Unit, Animal Health Research Institute, Provisional Lab Kafr El-Sheikh, Agricultural Research Center (ARC), Kafr El-Sheikh, Egypt 3Department of Chemistry, Rabigh College of Sciences and Arts, King Abdulaziz University, Jeddah, Saudi Arabia 4Environmental and Occupational Medicine Department, National Research Centre, Cairo, Egypt *Corresponding Author: Sayed M. Aboleila. Department of Aquaculture, Faculty of Aquatic and Fisheries Sciences, Kafrelsheikh University, Kafr El-Sheikh, Egypt. Email: sayedmetwallymohamed [at] gmail.com; alsaid_metwally [at] fsh.kfs.edu.eg Submitted: 31/10/2024 Accepted: 19/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
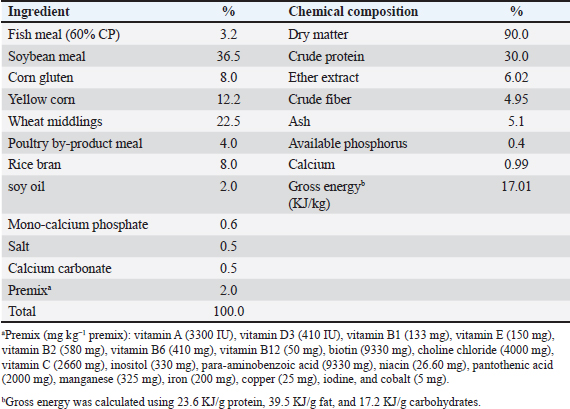
AbstractBackground: Numerous natural phytobiotic products are used as feed additives to enhance fish performance, quality, and immunity. Aim: This study evaluated the effect of a natural phytobiotics mixture [Syrena Boost (SB)] on growth performance, intestine health, immune-oxidative status, and hemato-biochemical parameters of fingerlings (Oreochromis niloticus). Methods: Fish (n=4,800, average initial weight=1.247 ± 0.047 g) were randomly distributed in 12 aquatic compartments with a total water volume of 2 m3 (2 × 1 × 1 m) for each one, representing four groups, in triplicate: control group (CG), SB1, SB2, and SB3, in which fish received a basal diet (30/6, protein/lipid ratio) containing 0, 0.1, 0.2, 0.4 g kg−1 SB, respectively, for 60 days. Results: The results showed a significant (p < 0.05) improvement in fish performance (survival rate, specific growth rate, weight gain, final weight, and feed conversion ratio) and intestine histomorphology (increased goblet cell number, intestinal villi length, but inter-villi space, decreased) in fish that received SB1, SB2, and SB3 compared with CG (p < 0.05) with the most favorable outcomes observed in treatment supplemented with SB2. Dietary inclusion of SB at different doses (SB1, SB2, and SB3) improved certain blood hematological parameters, including globulin and total protein, while decreasing aspartate aminotransferase, alanine aminotransferase, cholesterol, triglycerides, glucose, and albumin compared with CG (p < 0.05), with the most favorable outcomes observed in SB2. The fish showed improvements in digestive enzymes (lipase and amylase), antioxidant enzymes (increased catalase, superoxide dismutase), as well as a reduction in malondialdehyde, as well as showed improvement in the immunity health indicators (phagocytic index, phagocytic activity, and lysozyme activity), and gene expression (glutathione peroxidase, and catalase, tumor necrosis factor-alpha, interleukin-1β, insulin-like growth factor, interleukin-8, and growth hormone receptor) when they received SB1, SB2, and SB3 supplemented diets, with the most favorable outcomes observed in SB2 compared with CG (p < 0.05). Thus, the SB at 0.2 g kg−1 diet (SB2) can be used effectively in Tilapia diets to improve growth, intestinal health, blood health, oxidative status, and immune-related gene expression. Conclusion: Dietary supplementation with a natural phytobiotic mixture (SB®) improved fish performance, intestine health, and the immune-physiological status of Nile tilapia fingerlings. The best findings in this experiment were observed in fish that received SB2. Keywords: Immune-physiological response, Gene expression, Growth performance, Nile tilapia, Phytobiotics. IntroductionThe industrialization and sustainability of the aquaculture sector are crucial for the global population’s food security and nutrition, relying on technological advancements and effective functional feed supplementation (Khalafalla et al., 2020). Moreover, the sector is confronted with more significant encounters and challenges due to inadequate fish health management and the high cost of aquafeed (Aboleila et al., 2022). Hence, dealing with these issues through dietary supplementation of growth and health enhancers, such as phytobiotic feed additives, which function as antimicrobial, antioxidant, antiparasitic, stimulants of bile secretion, digestive enzyme activity, growth promoters, and appetite enhancers, was a highly pertinent subject in tilapia cultivation (Fawole et al., 2020). Early supplementation with microencapsulated essential oils (MEOs) improved the number, length, and width of intestinal folds and increased the number of goblet cells, positively influencing intestinal morphology and health. Therefore, growth parameters in Nile tilapia were improved after 30 days of supplementation (Roldan-Juarez et al., 2023). Including supplementation with a blend of phytobiotics for 20 days improved antioxidant protection, mitigated the effects of stressors, modulated immunity, and provided greater resistance and protection against diseases in Nile tilapia (Estaiano de Rezende et al., 2021). The use of additives, such as capsaicin, saponins, and the essential oil of star anise, in Syrena Boost (SB)® improves aquafeed efficiency, digestibility, and production costs (Abozeid et al., 2021). The saponins found in Quillaja Saponaria (QS). Additionally, Saponins exhibit a variety of effects on both animal and human biological systems and serve as active components in numerous medicinal formulations and preparations of human vaccines (Oleszek and Oleszek, 2020). Furthermore, they effectively improve nutrient intake, digestibility, and the efficiency of feed utilization in farmed fish, leading to a reduced demand for oxygen, which is vital for growth (Francis et al., 2005). Thus, it is considered one of the most important commercial sources of phenolic-binding ammonia extracts in the development aquaculture sector (Angeles Jr, et al., 2017). The primary component of hot red pepper is capsaicin, which effectively boosts the activities of intestinal and pancreatic enzymes, glucose-6-phosphate dehydrogenase, and lipoprotein lipase in adipose tissue, thereby enhancing nutrition and energy metabolism (Platel and Srinivasan, 2004). Moreover, capsaicin and cinnamaldehyde stimulate the production of amylase (Steiner and Syed, 2015). The pungent characteristics of hot red pepper (Capsicum annum L), were assessed for their antioxidant potential and effectiveness against some bacteria and fungi (Shahverdi et al., 2013; Akhtar et al., 2017). Many pharmacology studies have confirmed that star anise has active compounds responsible for antimicrobial, antioxidant, and anti-inflammatory activities (Boota et al., 2018). In addition to its aromatic, diaphoretic, anti-inflammatory, antiseptic, and stimulant properties, it can inhibit protein denaturation, prevent lipid peroxidation, and scavenge free radicals, which are related to its anti-inflammatory and antioxidant activities (Luís et al., 2019). Additionally, it promotes the digestion and immunity of fish because of its antioxidant, antiparasitic, antibacterial, and antifungal properties (Mohanasundari et al., 2022). Numerous studies have been conducted on the effects of QS, star anise, and capsaicin (hot red pepper) as a single ingredient on fish growth performance and health; however, their combined effects have not yet been examined. Consequently, this study aimed to investigate and explore the combined and synergistic effects of dietary supplementation with a natural phytobiotic mixture (SB®) as a source of QS, star anise, and capsaicin on various parameters related to Nile tilapia fingerlings, focusing on the efficiency of feed utilization, absorption efficacy, growth performance, flesh quality, intestine health, digestive enzymes, and the hemato-biochemical and immunity and health status. Materials and MethodsFish and culture systemsThe present study was conducted at a private tilapia hatchery (Tolmbat 7, Kafr Elsheikh Governorate, Egypt). Two nets measuring (6 × 3 × 1 m each) were used, with each net being divided into six equal aquatic compartments with a total water volume of 2 m3 (2 × 1 × 1 m) for each one and was filled with experimental fish and provided with a continuous supply of fresh water. Approximately 10% of the aquatic compartment water volume was replaced every day; the fish were maintained under a natural light schedule, following a 12-hour light and 12-hour dark cycle. Mono-sex fingerlings (n=4,800, average weight: 1.247 ± 0.047 g) were used in the present study. The fish were sourced from a private tilapia nursery pond in Tolmbat 7, Kafr Elsheikh, Egypt. They were transported by a special car for fish transport equipped with an oxygen supply in the morning and acclimatized for 15 days before the experiment. In addition, 30% crude protein was used as a control diet during the acclimatization phase. Thereafter, 4,800 fish were distributed randomly into four experimental treatments in triplicate (400 fish per aquatic compartment). Experiment diets and study protocolThe fish were categorized into four experimental treatments as outlined below: (1) The control group (CG) consisted of fish with a standard diet (Table 1); (2) SB1, fish was given a standard diet that included 0.1 g kg−1 of SB (Delacon Biotechnik GmbH, Austria), SB consisted of (saponin 18%, essential oils 9%, silicon dioxide 8%, and calcium carbonate 65%); (3) SB2, fish were given a standard diet containing 0.2 g kg−1 of SB; (4) SB3, fish were given a standard diet containing 0.4 g kg−1 of SB. During the feed manufacturing process, sunflower oil was used to combine the experimental diet with the tested feed additive (SB). The test diets were combined, pelleted using a 0.5-mm diameter, and dried for 24 hour at ambient temperature before being stored at 20°C until needed. The feeding protocol was conducted twice daily, administering 9% of body weight at 8:00 and 14:00 for 60 days (Huang et al., 2015; Fahrurrozi et al., 2024). A fish sample was collected from each tank every 14 days, followed by weighing, and the feed quantity was modified based on variations in body weight throughout the experimental period. The feed and total feed intakes at the end of the experiment were calculated and reported. Daily mortality was recorded and was removed from the aquatic compartments as soon as possible. Table 1. Feed formulation and proximate chemical composition of the basal diet (on a DM basis).
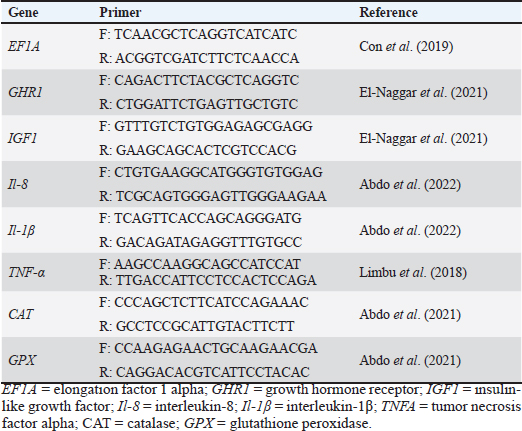
Water quality analysisThe water quality was maintained throughout the experimental period at optimal standards by daily monitoring of the water parameters. Temperature, pH, and dissolved oxygen were determined in the aquatic compartments using a Multiparameter probe meter (HI9829-03042-HANNA® insrruments). A portable photometer (Martini MI 405 MR) was also used for total ammonia measurements (Abouelenien et al., 2015). The fish growth performance, feed utilization efficiency, and various biometric indicesThe collection of fish and the measurement of their weight to determine the final weight were conducted after the experimental period, which lasted 60 days. As well as the fish’s overall length (L) was also recorded using a measuring board. In brief, the efficiency of feed utilization and fish growth performance [feed conversion ratio (FCR), condition factor (K), body weight gain (BWG), specific growth rate (SGR%)/day], viscera-somatic index (VSI), hepato-somatic index (HSI), and survival rate (SR%) were calculated using the equations and formulas described by Abozeid et al. (2021) and Elkaradawy et al. (2022). Chemical composition of the whole fish body and dietBefore the experiment began, a chemical analysis of the diet was performed. In addition, six fish were selected randomly from each aquatic compartment for chemical composition analysis after the end of the experimental period. The fish were stored at a temperature of −40°C until they were needed. The complete composition of the fish body, including carbohydrates, crude protein, lipid content, dry matter (DM), and ash, as well as the test diet’s nutritional profile, was assessed using standard methodologies (Baur and Ensminger, 1977). In addition, the standard techniques reported by Van Soest et al. (1991) were used to assess the fiber content. Intestine histomorphology and morphometric changesThe tissues of the intestinal part (posterior, middle, and anterior) were obtained by dissection of six fish/treatment and immersion in 10% neutral-buffered formalin for 3 days to achieve fixation. After fixation, the samples were dehydrated and rinsed multiple times in absolute alcohol (70%), followed by embedding in paraffin and preparation for histological investigations. On a Leica Rotary Microtome (RM 2145, Leica Microsystems, Wetzlar, Germany), serial 5 μm longitudinal sections were cut and mounted on glass slides. The slides were then routinely stained with hematoxylin and eosin (H&E) for morphometric analysis, as reported by Spencer et al. (2012). The length and width of the intestinal villi and villi surface area were assessed using the software of image analysis [National Institutes of Health (NIH), Bethesda, MD], as assessed by Suvarna and Layton (2013). As described by Al-Deriny et al. (2020), the density of goblet cells was quantified per unit surface area. Blood sampling and serum separationAccording to Jian (1986), samples of caudal vein blood (nine fish/treatment) were obtained after the experimental period and then placed in a vacuum tube containing anticoagulant (EDTA) for hematological analysis. The blood serum was collected without anticoagulants. The clotted blood was centrifuged at 3,000 rpm for 15 minutes at 4°C. After this process, the supernatant serum was carefully aspirated and stored in Eppendorf tubes at a temperature of −20°C for subsequent reliable analysis. Hematological analysisAs reported by Thrall et al. (2004), the hematological parameters [hemoglobin content, packed cell volume (PCV), red blood cells (RBCs) count, mean corpuscular hemoglobin (MCH), mean corpuscular volume (MCV), MCH concentration (MCHC), and total and differential white blood cells (WBCs)] count were measured. Serum biochemical analysisBased on the methods reported by Henry (1964), total serum protein (TP) was assessed colorimetrically at a wavelength of 546 nm, whereas albumin in the serum was assessed colorimetrically at a wavelength of 630 nm, as reported by Doumas et al. (1981). By subtracting the albumin value from the total protein value, the globulin content was mathematically estimated. The activities of AST and ALT were colorimetrically assessed using a wavelength of 540 nm according to Reitmen (1957). Total cholesterol levels and serum triglyceride levels were analyzed according to the instructions of the manufacturer’s methods of the commercial clinical kit (CHOD-PAP and GPO-PAP, Elabscience, USA), respectively (Li et al., 2018). The serum creatinine level was measured using the colorimetric method reported by (Heinegard and Tiderström, 1973). Based on the methods of Trinder (1969), glucose levels were assessed using glucose enzymatic PAP kits (Bio-Merieux, France). Antioxidant capacityELISA kits (Inova Biotechnology, China) as explained by Abdel-Tawwab et al. (2018) were used to measure the serum levels of catalase (CAT), superoxide dismutase (SOD), and malonaldehyde (MDA) using an ELISA Microplate Reader at a wavelength of 450 nm. The activities of digestive enzymesThe techniques explained by Abdel-Tawwab et al. (2018) were used to measure the levels of digestive enzymes, specifically amylase and lipase. The activities of these enzymes in fish serum (nine fish per treatment) were evaluated using diagnostic reagent kits from Cusabio Biotech, located in Wuhan, Hubei, China, following the procedures provided by the manufacturer’s guidelines. Immunity statusBased on methods explained by Demers and Bayne (1997), the activity of lysozyme in the serum was evaluated using clinical kits sourced from Sigma, USA. While the Kawahara et al. (1991) method was used to evaluate phagocytic activity (PA). The subsequent formulas were employed to quantify the number of phagocytic cells and assess the phagocytic index (PI). The PI is the ratio of the number of phagocytized cells to the number of phagocytic cells; PA is the ratio of the number of macrophages carrying yeast to the macrophages’ total number × 100. Gene expression analysisTissue samples of the fish liver (six fish/treatment) were gathered within a sterile Eppendorf tube (2 ml). The tissue samples were stored at −80°C after being shocked in liquid nitrogen until analysis. Then, 50 mg of liver tissue was used to extract the total RNA using Trizol (iNtRON Biotechnology, Inc., Korea) according to the guidelines of the manufacturer. RNA integrity was confirmed using ethidium bromide-stained 2% agarose gel electrophoresis. A Nanodrop BioDrop spectrophotometer (Biochrom Ltd., Cambridge CB23 6DW, UK) was used to determine the RNA concentration and purity using the A260/A280 nm ratio. Following the manufacturer’s instructions, 2 μg of the RNA sample were reverse-transcribed using the ABT 2X RT Mix cDNA synthesis kit (Bioline, UK). Gene expression profiling was conducted using Rotor Gene-Q (Qiagen-Germany), employing gene-specific primer sequences to amplify various genes, including growth-related genes, such as insulin-like growth factor (IGF), and growth hormone receptor (GHR), inflammatory (immunity)-related genes such as interleukin-1β (IL-1β), interleukin-8 (IL-8), and tumor necrosis factor-alpha (TNF-α), and antioxidant genes, such as CAT and glutathione peroxidase (GPX) (Table 2). The ABT 2X qPCR Mix (SYBR) kit was used for the amplification reaction. The final volume of the reaction was 20 μl, which was composed of 10 µl SYBR Green, 0.6 µl of forward and reverse specific primers, 1 µl of cDNA template, and nuclease-free water. PCR was performed at a temperature of 95°C for 15 minutes, denaturation (40 cycles) at a temperature of 95°C for 10 seconds, annealing at the primer-specific temperature for 15 seconds, and extension at a temperature of 72°C for 25 seconds. The melt curve was then analyzed to evaluate the amplification specificity at a temperature of 72°C–95°C. Each gene was examined three times. Each sample’s CT values were calculated and added to the “fold change” (2−ΔΔCT), using a calculation explained by Livak and Schmittgen (2001), as well as the expression of mRNA of each sample normalized against the elongation factor 1 alpha (EF1A) as a housekeeping gene. Statistical analysisThe GraphPad Prism software (version 8.01) was used to perform statistical analysis after the data were evaluated for homogeneity of variance and normality. One-way analysis of variance (ANOVA) and Tukey’s post-hoc multiple comparison test were used to examine the effect of SB on fish growth performance, intestine health, immune-oxidative status, and haemato-biochemical and gene expression of growth (GH and IGF-1), antioxidant (GPX, CAT), and immunity (IL-8, IL-1β, and TNF-α) for significant differences among the experimental treatments. According to the p-value below 0.05, the differences became significant and are indicated by different superscript letters. Means and standard deviations are used to express all results. Table 2. Primers used for qRT–PCR analysis.
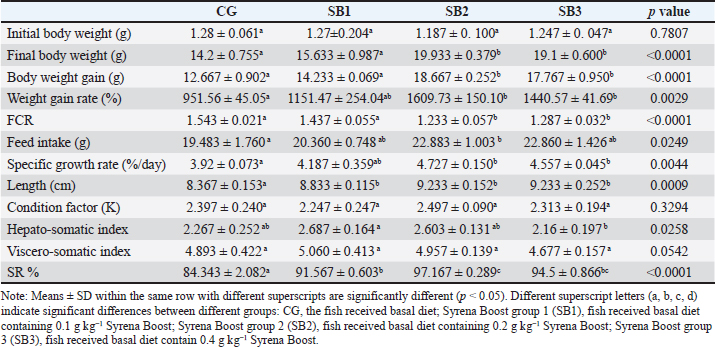
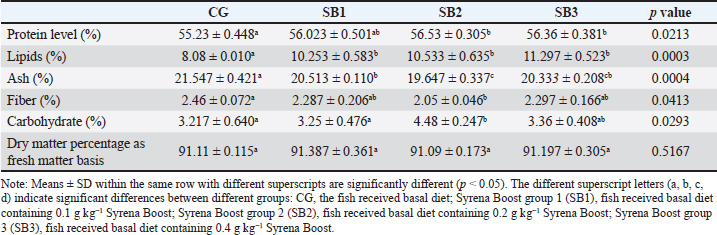
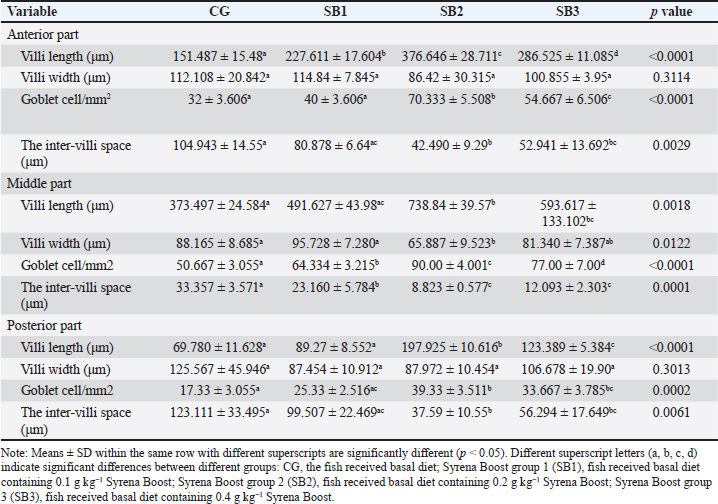
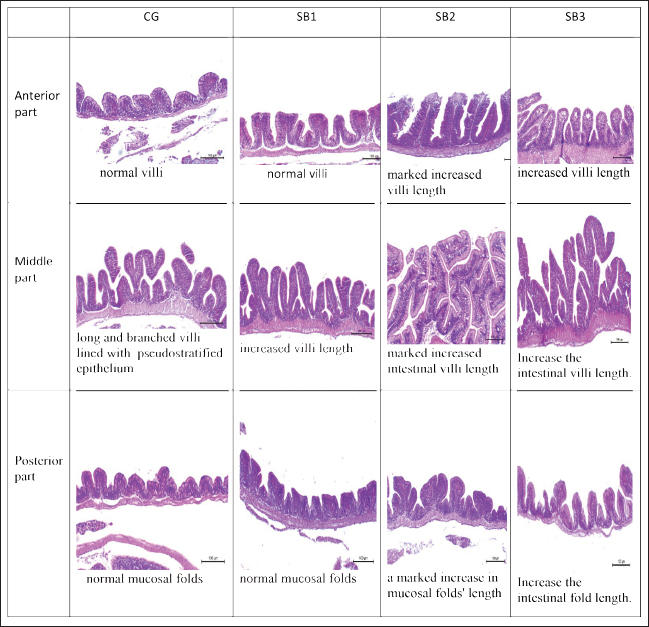
ResultsThe fish growth performance, feed utilization efficiency, and biometric indicesThe feed utilization efficiency, growth performance, and biometric indices of Nile tilapia-fed experimental diets are presented in Table 3. Treatment with SB2 or SB3 exhibited significant improvements in body weight gain and final body weight and a significant improvement in FCR compared with CG (p < 0.05). However, feed intake, weight gain rate, specific growth rate, total length, and SR significantly increased in fish fed SB1, SB2, and SB3 compared with the CG (p < 0.05), and the best findings were obtained in fish-supplemented with SB2. The HSI revealed a significant improvement in fish that acquired SB1, followed by SB2. However, VSI, initial body weight, and condition factor (K) were not significantly different between all experimental treatments (p > 0.05). Proximate body chemical compositionTable 4 lists the fish’s chemical compositions. The crude protein content and lipid level of the fish that acquired SB1, SB2, and SB3 increased significantly (p < 0.05) with respect to CG. Carbohydrate levels increased significantly (p < 0.05) in fish receiving SB2 and SB3, with respect to CG, but there were no significant differences between SB1 and CG. While fish receiving SB2 had significantly lower levels of ash and fiber, followed by SB3 and SB1, concerning CG, the DM levels of all experimental groups were not significantly different (p > 0.05). SB2 reported the best results. Histomorphology of the urinary tract and morphometric changesThe goblet cell counts and the length of the villi of different sections of the fish intestine (posterior, middle, and anterior) were significantly (p < 0.05) higher in fish that received SB2, followed by SB3, in comparison with CG (Table 5 and Fig. 1). Better outcomes were observed in fish supplemented with SB2. In contrast, the inter-villi space revealed a significant decrease in all intestinal sections (posterior, middle, and anterior) in fish receiving SB2, followed by SB3, and SB1, compared with CG. With the exception of the middle intestinal segment, where fish receiving SB2 showed a significant decrease in villi width, while the posterior and anterior intestinal segments’ villi widths were not significantly different between any of the experimental treatments(p > 0.05). Table 3. Growth performance, feed utilization, and biometric indices of fish-fed experimental diets for 60 days.
Table 4. Proximate body chemical composition (Parameter %, as DM basis) of fish-fed experimental diets for 60 days.
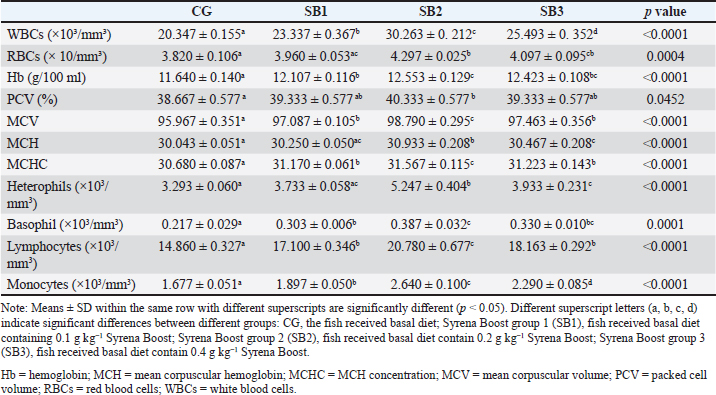
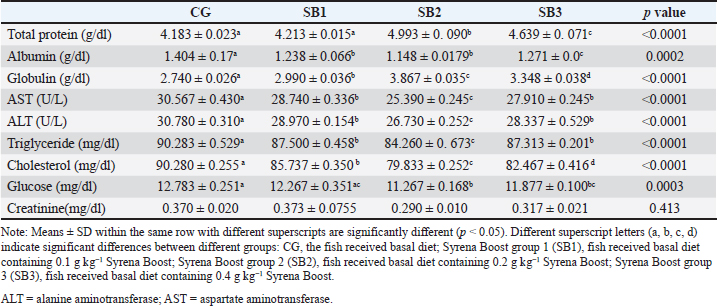
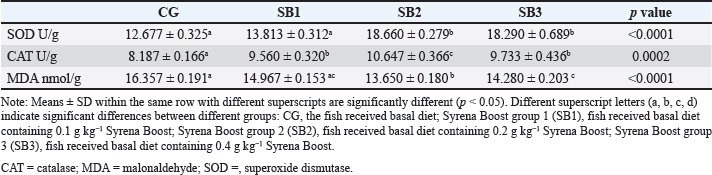
Hematological analysisTable 6 presents the blood hematological parameters of Nile tilapia-fed experimental diets. Fish treated with SB1, SB2, and SB3 had significantly higher levels of RBCs, Hb, PCV, MCV, MCH, MCHC, WBCs, heterophils, basophils, lymphocytes, and monocytes than those treated with CG (p < 0.05). The best findings were found in fish that received SB2. Serum biochemical analysisThe biochemical blood profiles of Nile tilapia diets supplemented with various SB additions are shown in Table 7. Fish treated with SB1, SB2, and SB3 had significantly lower values of AST, ALT, triglycerides, cholesterol, glucose, and albumin compared with CG (p < 0.05), in addition, the SB2 had the lowest values. Fish that received SB2 and SB3 had significantly higher total protein levels compared with CG (p < 0.05), but SB1 showed no significant differences compared with CG. Fish acquired SB1, SB2, and SB3 had significantly higher globulin levels than CG (p < 0.05), with SB2 showing the highest levels. However, creatinine levels did not differ significantly between the experimental treatments (p > 0.05). Antioxidant capacityThe antioxidant capacity profiles of Nile tilapia-fed diets treated with various inclusions of SB are presented in Table 8. The SOD values in SB2 and SB3 revealed higher differences significantly (p < 0.05) in comparison with CG, with non-significant differences between SB1 and CG. Nevertheless, the activity of CAT obtained by SB2, SB3, and SB1 exhibited more significant differences than CG (p < 0.05), as well as SB2 showed the greatest results. MDA activity was significantly decreased (p < 0.05) in fish treated with SB1, SB2, and SB3 compared to CG. The SB2 group had the highest results. Table 5. Intestinal histomorphology of fish exposed to different experimental treatments for 60 days.
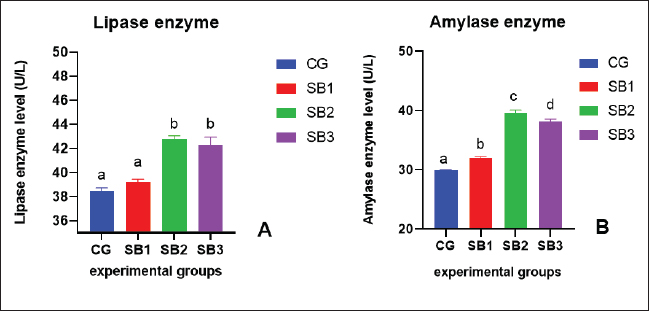
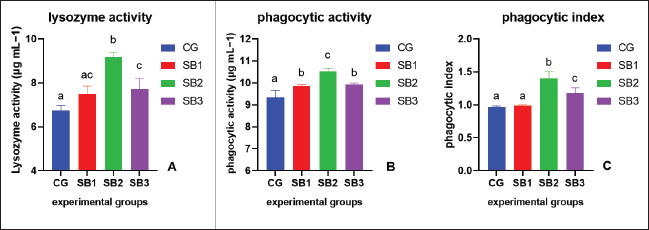
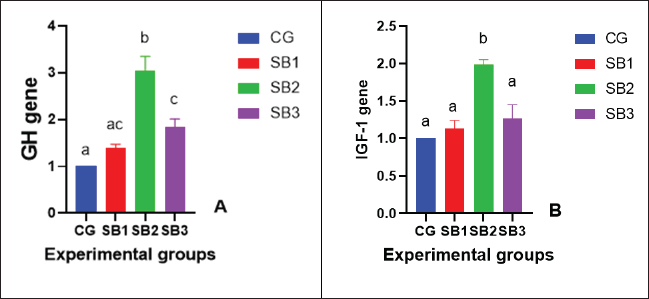
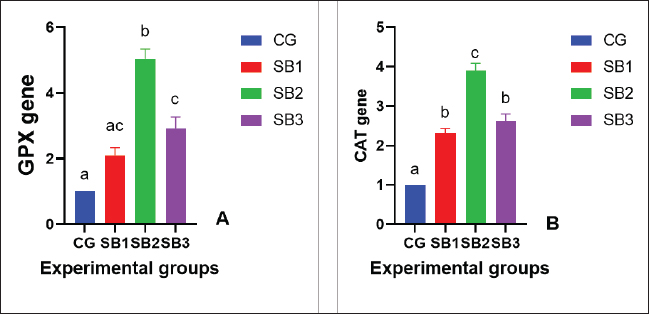
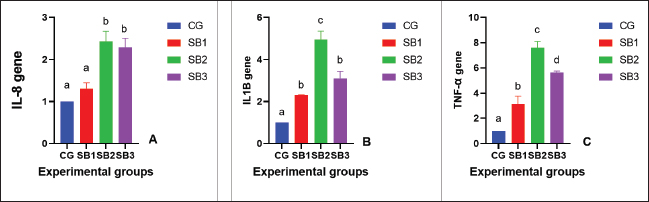
The activities of digestive enzymesSignificant variations in digestive enzyme activity (p < 0.05) were identified among Nile tilapia-fed experimental diets (Fig. 2a and b). Fish treated with SB2 and SB3 showed the most significant differences (p < 0.05) in lipase enzyme activity compared with CG, with non-significant differences between SB1 and CG. The activity of the amylase enzyme in the treatment groups (SB1, SB2, and SB3) was significantly increased (p < 0.05) compared with that of CG. Immunity statusLysozyme activity was considerably increased in fish treated with SB1, SB2, and SB3 compared to the CG (p < 0.05) (Fig. 3a). The highest levels were observed in fish supplemented with SB2. The phagocytic activities of fish-fed diets containing SB1, SB2, and SB3 differed significantly (p < 0.05) from those of CG (Fig. 3b), with SB2 exhibiting the best results. Fish treated with SB2 and SB3 had a significantly higher phagocytic index than CG (p < 0.05) (Fig. 3c), with non-significant differences between SB1 and CG. In addition, SB2 obtained the best results. Gene expression analysisExpression of the GH gene was significantly upregulated (p < 0.05) in the fish acquired SB1, SB2, and SB3 compared with CG (Fig. 4a), with the highest values observed in fish treated with SB2. Figure 4b exhibited significant elevation (p < 0.05) of the IGF-I gene in fish fed a diet supplemented with SB2 concerning CG, with non-significant differences between SB1, SB3, and CG. GPX gene expression was significantly upregulated (p < 0.05) in fish-fed SB1, SB2, and SB3 compared with CG (Fig. 5a), and CAT gene expression was significantly upregulated (p < 0.05) in fish-fed SB1, SB2, and SB3 compared with CG (Fig. 5b). The highest values were recorded in fish treated with SB2. IL-8 gene expression was significantly (p < 0.05) upregulated in fish fed SB2 and SB3 compared with CG (Fig. 6a), with non-significant differences between SB1 and CG. IL-1β and the expressions of TNF-α were up-regulated significantly (p < 0.05) in fish fed SB1, SB2, and SB3 compared with CG (Fig. 6b and c) with the highest levels observed in fish supplemented with SB2. Fig. 1. Hematoxylin-eosin-stained (H&E, bar=100 μm) photomicrographs of the anterior, middle, and posterior parts of the intestine of fish exposed to different experimental treatments for 60 days. The CG, the fish received a basal diet; Syrena Boost group 1 (SB1), fish received a basal diet containing 0.01 g kg−1 Syrena Boost; Syrena Boost group 2 (SB2), fish received a basal diet containing 0.02 g kg−1 Syrena Boost; Syrena Boost group 3 (SB3), fish received a basal diet containing 0.04 g kg−1 Syrena Boost.
DiscussionNumerous natural phytobiotic products are used as feed additives to enhance fish husbandry. Moreover, these alternative supplements have an excellent safety profile for the general health of fish and consumers (Elbialy et al., 2021; Rahman et al., 2023). The improvement in all growth performance and feed utilization parameters observed in this study reflects the power of using SB as a phytobiotic mixture in fish-supplemented feed for enhancing feed utilization indices and growth performance, and it is due to its composition of Quillaja Saponins (QSs) and capsaicin from pepper, which can improve the permeability of intestinal wall and enhance nutrient absorption (Prakash et al., 2013; Elkaradawy et al., 2022); in addition, capsaicin from pepper can enhance animal performance by stimulating digestive secretions or directly inhibiting bacteria in the gut (Citarasu, 2010). Moreover, Essential oil found in Star anise, which is rich in bioactive compounds like anethole, estragole, and anisaldehyde, has been found to enhance digestion and growth performance (Mohanasundari et al., 2022). These findings agree with those reported by Elkaradawy et al. (2022), who found an improvement in the growth performance of Nile tilapia as a response to QS supplementation (300 mg kg−1) as well as Ibrahim et al. (2024) and Yilmaz et al. (2024) recorded that growth performance also increased when using the hot red pepper “Capsicum annuum” as feed supplementation for Nile tilapia. Mohanasundari et al. (2022) also explained the role of star anise essential oil in improving and enhancing Nile tilapia fish growth. Roldan-Juarez et al. (2023) also found an improvement in the growth performance of Nile tilapia fingerlings in response to MEO supplementation. Table 6. Hematological profiles of fish-fed experimental diet for 60 days.
Table 7. Blood biochemical profiles of fish exposed to different experimental treatments for 60 days.
Supplementation with natural phytobiotics especially (SB) can affect the whole body’s proximate chemical composition of fish. Hence, the study findings revealed a general increase in protein, lipid, and carbohydrate levels and a decrease in ash and fiber levels. This notable improvement in the chemical composition of fish returned to the high activity and efficiency of digestive enzymes (lipase and amylase), enhanced growth performance, and decreased MDA production. These results are in parallel with those of Francis et al. (2002a); Abdel-Hakim et al. (2014); and Gonçalves et al., (2019). Table 8. Oxidative parameters of Nile tilapia exposed to different experimental treatments for 60 days.
Fig. 2. Digestive enzyme activity (A- lipase, B- amylase) of fish (nine fish/treatment) fed experimental diets for 60 days. The columns (mean + SD) with different letters were significantly different (p < 0.05, one-way ANOVA).
Fig. 3. Immune response (A- Lysozyme activity, B- Phagocytic activity, and C- Phagocytic index) of fish fed experimental diets for 60 days. The columns (mean + SD) with different letters were significantly different (p < 0.05, one-way ANOVA).
Fig. 4. Growth-related genes [A- growth hormone (GH) gene and B- insulin-like growth factor (IGF) gene] expression of fish exposed to different experimental treatments for 60 days. The columns (mean + SD) with different letters were significantly different (p ≤ 0.05, one-way ANOVA).
Fig. 5. Expression of antioxidant genes [A- Glutathione peroxidase (GPX) and B- and catalase (CAT) gene] expression of fish exposed to different experimental treatments for 60 days. The columns (mean + SD) with different letters were significantly different (p ≤ 0.05, one-way ANOVA).
Intestinal morphometry gives an indication of absorption potentiality, the nutrient utilization efficiency of aquatic species, and the absorption area capacity. as well as the goblet cell count and the height/width of intestinal villi serve as reliable indicators of intestinal health (Mohamed et al., 2021), as more goblet cells result in more mucins or glycoproteins being secreted, which permits intestinal microbiota modulation and prevents the growth of harmful bacteria (Abdel Rahman et al., 2019). The findings demonstrated an enhancement of the histomorphological parameters of the intestines in the experimental groups supplemented with SB. This is because dietary saponins improve the permeability of intestinal membranes to digested food ingredients (Francis et al., 2002b). Notably, improvement in intestinal morphometry was associated with the increased activity of digestive enzymes and improved growth performance. Similar to the findings of Roldan-Juarez et al. (2023), early supplementation with MEOs improved the number, length, and width of intestinal folds and increased the number of goblet cells in Nile tilapia after 30 days of supplementation. Compatible with Francis et al. (2005) and Elkaradawy et al. (2021), the QS role in enhancing intestinal health by increasing the absorption area’s capacity is also explained. These outcomes are also compatible with Kawabata et al. (2009) discussed capsaicin’s role in promoting intestinal health, as capsaicin effectively enhances the metabolism of nutrients and energy by boosting the activities of glucose-6-phosphate dehydrogenase, lipoprotein lipase in adipose tissue, and pancreatic and intestinal enzymes. Fig. 6. Expression of inflammatory (immunity)-related genes [A- Interleukin-8 (IL-8) gene, B- Interleukin-1β (IL-1β) gene, and C- tumor necrosis factor-alpha (TNF-α) gene] expression of fish exposed to different experimental treatments for 60 days. The columns (mean + SD) with different letters were significantly different (p ≤ 0.05, one-way ANOVA).
The blood hematological parameters of Nile tilapia are important indicators of fish health. The improvement in all hematological parameters in fish treated with SB observed in this study indicates the ability of the phytobiotic mixture to improve the general health and immunity of fish. This outcome could be explained by QS’s capacity to modify immunity by boosting antigen absorption, inducing the generation of cytotoxic T lymphocytes and cytokines in response to various antigens, and acting as an immunostimulant (de Groot and Müller-Goymann, 2016; Fleck et al., 2019), as well as the anti-inflammatory and immunomodulatory properties of capsaicin and the essential oil of star anise (Luís et al., 2019; Valdivieso-Ugarte et al., 2019). The current findings align with those of Güroy et al. (2016), who reported that adding QS to the diet of young striped catfish led to an improvement in PCV. Additionally, Amiri and Bahrekazemi (2017) noted an increase in the percentage of WBCs, heterophils, basophils, monocytes, and lymphocytes in fish fed a diet containing 0.5% QS. The increase in lymphocytes coincided with the findings of Elkaradawy et al. (2022), who observed a higher lymphocyte percentage in Nile tilapia fingerlings that were given diet-acquired QS (300 mg kg−1) beyond those given the control diet. Therefore, SB may enhance the hematological profile of Nile tilapia, as well as bolster their overall health and immune response. Serum proteins are crucial components, and their concentration serves as a fundamental indicator of fish health status (Alexander et al., 2011). Our research revealed that dietary supplementation with SB increased TP and globulin levels although the albumin value decreased. Notably, the most favorable outcomes were observed in fish that received SB2. These results may be ascribed to the ability of QS saponin to augment the secretion of trypsin, which leads to the rise of TP by improving protein absorption, as well as its role in enhancing protein metabolism (Wang et al., 2020) and to the ability of the essential oil star anise to reduce protein denaturation (Luís et al., 2019). These findings are consistent with those reported by Elkaradawy et al. (2022), who reported an increase in TP and globulin levels via QS supplementation (300 mg kg−1) into fish feed. The transfer of amino group functions of alpha-amino acids to alpha-keto acids is controlled by ALT and AST, which are essential liver enzymes for monitoring liver health and function (Abdo, 2020). Fish that received SB2 showed lower serum levels of ALT and AST compared with CG, suggesting that the use of natural phytobiotics enhances liver health. In addition, this improvement is likely due to the antioxidant effects of capsaicin, star anise-essential oil, and QS-containing flavonoids, which prevent cell membrane lipid peroxidation and stop the release of liver damage enzymes (ALT and AST) into the plasma (Shahverdi et al., 2013; Luís et al., 2019). The protective effect of saponin on the liver is attributed to its ability to mitigate oxidative stress and maintain hepatocyte integrity and function (Fleck et al., 2019). These findings are consistent with Fleck et al. (2019) and Elkaradawy et al. (2022), who confirmed a reduction in AST and ALT levels in response to dietary QS supplementation. The concentrations of glucose and triglycerides in fish can indicate their stress response and are important for maintaining body balance (Mommsen et al., 1999). Our research showed that fish receiving SB2 had significantly reduced levels of glucose, triglycerides, and cholesterol compared to CG, which is attributed to reduced cholesterol absorption in the intestine and increased activity of β-hydroxy β-methylglutaryl-CoA reductase and low-density lipoprotein receptors in the liver, as explained by Rao and Gurfinkel (2000). These findings align with those of Angeles Jr et al. (2017), who found decreased glucose and triglyceride concentrations in Nile tilapia blood when fed with QS additives (150 mg kg−1). Additionally, McCain (2013) observed that capsaicin can lower serum glucose and raise serum insulin levels by enhancing pancreatic function and insulin secretion. The measurement of creatinine levels in the blood reflects the kidney’s ability to regulate creatinine levels in fish muscles (Campbell, 2004). Our results indicate that there was no significant variation in creatinine levels across all experimental treatments, suggesting that SB does not adversely affect renal function or fish health, consistent with the results of Angeles Jr et al. (2017) and Elkaradawy et al. (2022). The oxidative status and cellular immunity of fish are assessed using SOD, CAT, and MDA concentrations to determine the damage caused by free radicals in different fish tissues and organs (Mohamed et al., 2021). SOD and CAT play crucial roles in neutralizing free radicals and preventing lipid peroxidation, whereas MDA release indicates the extent of cell destruction and lipid peroxidation in tissues (Salama et al., 2019). In this study, Nile tilapia fingerlings showed the highest CAT and SOD activity when supplemented with SB, with the most favorable results observed in SB2. Moreover, the addition of SB to Nile Tilapia significantly decreased MDA activity, demonstrating the antioxidant properties of phytobiotics. In parallel with Elkaradawy et al. (2022), QS supplementation increased CAT and SOD activity and decreased MDA activity. Additionally, Fleck et al. (2019) found that QS-derived saponin notably reduced the production of MDA in the liver, suggesting that the antioxidant properties of QS saponin can mitigate oxidative stress by scavenging excessive radicals, as explained by Yu et al. (2014). In addition, Estaiano de Rezende et al. (2021) reported that supplementation with a blend of phytobiotics for 20 days provided greater antioxidant protection in Nile tilapia. As well as Luís et al. (2019) also stated that star anise essential oils can prevent both lipid peroxidation and protein denaturation and can scavenge free radicals due to bioactive components such as estragole, anisaldehyde, and anthole (Newberne et al., 1999). Capsaicin has also been observed to effectively prevent lipid peroxidation and protein oxidation induced by gamma (γ) radiation, highlighting its role against oxidation and radiation (Gangabhagirathi and Joshi, 2015). The activity of digestive enzymes is directly correlated with the digestion abilities of aquatic animals. This study demonstrated a notable elevation in the values of lipase and amylase activity in Nile tilapia fingerlings that received SB2 compared with the CG. This can be ascribed to the ability of QS, capsaicin, and star anise essential oils to improve the functions of pancreatic and intestinal enzymes, thereby enhancing the digestion process (Citarasu, 2010; Serrano Jr, 2013). In line with our study, Elkaradawy et al. (2022) confirmed that QS significantly improved lipase and amylase activities in fish. The lysis of pathogenic bacteria and the induction of specific and nonspecific immune responses are caused by lysozyme activity, which serves as an important natural defense mechanism (Chen et al., 2014). As reported in this study, fish treated with SB exhibited the highest lysozyme activity compared with the control. The increase in lysozyme levels may be due to enhanced lymphocyte production, which in turn modulates the immune status of fish (Ruchin, 2020). This increase could be attributed to the antibacterial activity of QS and its ability to enhance antibody formation or modify immunity to favor a more cell-mediated response, as well as the roles of aglycone amphiphilic structures and branched sugar radicals in boosting the immune system (Oda et al., 2000). This effect is also attributed to the antimicrobial activity of capsaicin (Ibrahim et al., 2024) and star anise essential oils (Tewksbury et al., 2008; Boota et al., 2018; Oleszek & Oleszek, 2020). This result is also consistent with the findings of Abozeid et al. (2021) and Mohanasundari et al. (2022), who confirmed that natural plant supplementation enhances lysozyme activity and improves the general immunity of fish. Phagocytosis has a fundamental function in the cellular immune system of fish, helping them to more effectively defend against pathogen attacks by identifying and containing existing pathogens (Harikrishnan et al., 2011). Our results revealed that the highest significant phagocytic index/activity was reported in fish receiving SB2, which is related to the ability of QS to control diseases (Ng’ambi et al., 2016). It may also be attributed to the higher production of RBCs, white blood cells, and lymphocytes, which modulate the fish’s immune status (Amiri and Bahrekazemi, 2017; Ruchin, 2020). These results correspond with those of McCain (2013); Zhang et al. (2020); and Mohanasundari et al. (2022), who recommended the use of natural plant supplementation as an immune stimulant for fish. The findings from the gene expression analysis conducted in this research indicated an increase in the expression of genes related to fish growth (GH and IGF-1), immune-related cytokines (IL-8, IL-1β, and TNF-α), and antioxidant enzymes (GPX and CAT) in fish that acquired an SB-enriched diet compared with fish that acquired a control diet, with the most significant results observed in the group receiving SB2. The expression of GH and IGF-1 serve as key indicators of enhanced growth performance in Nile tilapia, as noted by Hassaan et al. (2019) and Mohamed et al. (2021). This can be viewed as an additional rationale for the improved growth rates observed in fish-fed SB. These findings are similar to those of Elkaradawy et al. (2022) and Khieokhajonkhet et al. (2023). The expression of GPX and CAT are essential markers for assessing antioxidant activity in animals, as these genes help neutralize free radicals and decrease lipid peroxidation (Kandeil et al., 2020). SB supplementation in feed led to a boost in the expression of GPX and CAT, likely due to enhanced oxidative status in the fish (Yu et al., 2014; Luís et al., 2019). This observation aligns with the findings of Elkaradawy et al. (2022) and Deng et al. (2023). Pro-inflammatory cytokines secreted by immune cells, particularly IL-8, TNF-α, and IL-1β, have an essential function in modulating the innate immune response; therefore, the expression patterns of these cytokine genes can indicate changes in immune responses (Van der Meide and Schellekens, 1996; Furman and Davis, 2015). Results from this study demonstrated upregulated expression of immune-related genes (TNF-α, IL-1β, and IL-8) in Nile tilapia; TNF-α, primarily released by activated macrophages, coordinates defense mechanisms against pathogen invasion and promotes neutrophil-based immunity (Bilen et al., 2019; Mohamed et al., 2021), while IL-1β is considered a pro-inflammatory cytokine that activates macrophages and lymphocytes to combat pathogens (Low et al., 2003). This outcome may be returned to the ability of QS saponins to stimulate the formation of inflammasome, which subsequently leads to the release of IL-1β and IL-6 from antigen-presenting cells (Marty-Roix et al., 2016). The inflammasome consists of various sensors and receptors that initiate inflammation by activating caspase-1. Additionally, saponins can enhance the expression of TLR interferon-α, resulting in the generation of active forms of IL-18 and IL-1β (Guo et al., 2015). Furthermore, the immunomodulatory and anti-inflammatory effects of capsaicin (C. annuum) increased the gastrointestinal mucosal barrier permeability in fish and finfish, which enhanced the intake of immunomodulatory substances, such as vaccine particles (Gaafar et al., 2018), and the essential oil from star anise may also contribute to these results (Luís et al., 2019; Valdivieso-Ugarte et al., 2019). Our findings are consistent with those of Attia et al. (2022) and Ibrahim et al. (2024). Thus, the present results indicate that dietary SB has upregulated expression of genes correlated with fish growth (GH and IGF-1), genes associated with antioxidant enzyme activity (GPX and CAT), and proinflammatory cytokine genes correlated with immune response (IL-1β, IL-8, and TNF-α), thereby improving growth performance, health status, and immune response in Nile tilapia. Further research investigating the specific gene expression profiles of fish that consume SB is required to validate this hypothesis. ConclusionFish performance, meat quality, intestinal health, digestive enzymes, hemato-biochemical profile, and immune health of fingerling Nile tilapia (Oreochromis niloticus) were improved by dietary supplementation with SB®. Furthermore, the most favorable outcomes in this research were observed with the natural phytobiotic blend “SB®” at a concentration of 0.2 g kg−1 (SB2) for improving fish performance, intestine health, and the immune-physiological status of Nile tilapia fingerlings. Therefore, incorporating a natural phytobiotic blend into feed could improve fish health and performance. Further research is required to explore the effects of SB® during the growth and reproduction phases of Nile tilapia. Conflict of interestThe authors declare that they have no conflicts of interest. FundingNo funding organization provided funding for this study. Authors’ contributionsSayed M. Aboleila: planned the experiments and wrote the original draft. Bahaa H. Abdella: designed and organized the experiments. Shreifa G. Mabrouk conducted the experiments and procedures. Asmaa M. El-Nokrashy: contributed to sample preparation. Nahla A. Ebied: Investigation and Formal analysis. Mohamed M Zayed: contributed to data curation and Investigation. Radi A. Mohamed: Conceptualization, supervision, writing, review, and editing. Each author also contributed to the interpretation of the findings, offered valuable critiques, and contributed to the development of the research, analysis, and manuscript. Data availabilityData can be provided upon request. ReferencesAbdel-Hakim, N., Lashin, M., Al-Azab, A.-D., Goda, A. and Nazmi, H. 2014. The effect of using some natural growth promoters on growth performance and feed utilization of monosex Nile tilapia (Oreochromis niloticus), fingerlings. Egypt J. Aquat. Biol. Fish. 18(3), 67–78; doi:10.21608/EJABF.2014.2219. Abdel Rahman, A., Hassanin, M. and ElHady, M. 2019. Growth performance, haematology and intestinal histo-morphology of Nile tilapia fed on Indian Lotus (Nelumbo nucifera Gaertn.) leaf powder at different concentrations. Aquac. Res. 50(11), 3211–3222; doi:10.1111/are.14276. Abdel-Tawwab, M., Samir, F., Abd El-Naby, A.S. and Monier, M.N. 2018. Antioxidative and immunostimulatory effect of dietary cinnamon nanoparticles on the performance of Nile tilapia, Oreochromis niloticus (L.) and its susceptibility to hypoxia stress and Aeromonas hydrophila infection. Fish Shellfish Immunol. 74, 19–25; doi:10.1016/j.fsi.2017.12.033. Abdo, H.S. 2020. Effect of dietary phytobiotics supplementation on growth, serum biochemical and chemical composition of Nile Tilapia (Oreochromis Niloticus). Ann. Agric. Sci. Moshtohor. 58(2), 273–282; doi:10.21608/assjm.2020.114834. Abdo, S.E., El-Nahas, A.F., Abdelmenam, S., Elmadawy, M.A., Mohamed, R., Helal, M.A. and El-Kassas, S. 2022. The synergetic effect of Bacillus species and Yucca shidigera extract on water quality, histopathology, antioxidant, and innate immunity in response to acute ammonia exposure in Nile tilapia. Fish Shellfish Immunol. 128, 123–135. https://doi.org/10.1016/j.fsi.2022.07.058. Abdo, S.E., Gewaily, M.S., Abo-Al-Ela, H.G., Almeer, R., Soliman, A.A., Elkomy, A.H. and Dawood, M.A.O. 2021. Vitamin C rescues inflammation, immunosuppression, and histopathological alterations induced by chlorpyrifos in Nile tilapia. Environ. Sci. Pollut. Res. Int. 28(22), 28750–28763; doi:10.1007/s11356-021-12711-5. Aboleila, S.M., Salah, A., Mohamed, R.A. and Diab, A.M. 2022. Combined effect of a mixture of Bacillus species and vaccination on haemato-biochemical parameters, immune response and transcriptomic changes of Nile tilapia challenged with Streptococcus iniae. Aquac. Res. 53(14), 5029–5044; doi:10.1111/are.15989. Abouelenien, F., Elsaidy, N., Kirrella, G.A. and Mohamed, R.A. 2015. Hygienic effect of supplementing Orechromis Niloticus farm with fresh or fermented chicken manure on: water, fish quality and performance. Alex. J. Vet. Sci. 45, 79–90; doi:10.5455/ajvs.181937. Abozeid, A.M., Abdel-Rahim, M.M., Abouelenien, F., Elkaradawy, A. and Mohamed, R.A. 2021. Quillaja saponaria and/or Yucca schidigera ameliorate water quality, growth performance, blood health, intestine and gills histomorphology of Nile tilapia, Oreochromis niloticus. Aquac. Res. 52(12), 6117–6613; doi:10.1111/are.15474. Akhtar, F., Sharif, H.M., Mallick, M.A., Zahoor, F., Abdulmalik, A., Baig, W., Shujaat, N., Gul, S., Bibi, G. and Ramzan, R. 2017. Capsaicin: its biological activities and in silico target fishing. Acta. Pol. Pharm. Res. 74(2), 321–329. Al-Deriny, S.H., Dawood, M.A., Abou Zaid, A.A., Wael, F., Paray, B.A., Van Doan, H. and Mohamed, R.A. 2020. The synergistic effects of Spirulina platensis and Bacillus amyloliquefaciens on the growth performance, intestinal histomorphology, and immune response of Nile tilapia (Oreochromis niloticus). Aquac. Rep. 17, 100390; doi:10.1016/j.aqrep.2020.100390. Alexander, C., Sahu, N., Pal, A. and Akhtar, M. 2011. Haemato-immunological and stress responses of Labeo rohita (Hamilton) fingerlings: effect of rearing temperature and dietary gelatinized carbohydrate. J. Anim. Physiol. Anim. Nutr. 95(5), 653–663; doi:10.1111/j.1439-0396.2010.01096.x. Amiri, Z. and Bahrekazemi, M. 2017. Effect of oral administration of Levamisole, Quil-A and Cinnamon in growth amount, hematological and immune parameters of Marmalade cichlid, Labeotrophus fuelleborni (Ahl, 1926). Iran J. Aquat. Anim. Health. 3(2), 86–97; doi:10.29252/ijaah.3.2.86. Angeles Jr, I.P., Gallego, L.M., Navarro, M.A.M. and Chien, Y.-H. 2017. Dietary effects of Quillaja saponaria and Yucca schidigera extract on rearing performance of Nile tilapia Oreochromis niloticus l. and its antioxidant capacity and metabolic response following hypoxic stress. Int. J. Agric. Technol. 13, 2249–2266; doi:10.5555/ ijat.20183304212. Attia, M.M., Alzahrani, A.M., Hanna, M.I., Salem, H.M., Abourehab, M.A., El-Saadony, M.T. and Thabit, H. 2022. The biological activity of Illicium verum (Star Anise) on Lernaea cyprinacea-infested Carassius auratus (Goldfish): in vivo study. Life, 12(12), 2054; doi:10.3390/life12122054. Baur, F.J. and Ensminger, L.G. 1977. The Association of Official Analytical Chemists (AOAC). J. Am. Oil. Chem. Soc. 54(4), 171–172. https://doi.org/10.1007/BF02670789. Bilen, S., Sirtiyah, A.M.A. and Terzi, E. 2019. Therapeutic effects of beard lichen, Usnea barbata extract against Lactococcus garvieae infection in rainbow trout (Oncorhynchus mykiss). Fish Shellfish Immunol. 87, 401–409; doi:10.1016/j.fsi.2019.01.046. Boota, T., Rehman, R., Mushtaq, A. and Kazerooni, E. 2018. Star anise: a review on benefits, biological activities and potential uses. Int. J. Chem. Biochem. Sci. 14, 110–114. Campbell, T. 2004. Clinical chemistry of fish and amphibians. In: Veterinary Hematology and Clinical Chemistry. Eds., Thrall, M.A., Baker, D.C., Campbell, T.W., DeNicola, D., Fettman, M.J., Lassen, E.D., Rebar, A. and Weiser, G. Pennsylvania, Lippincott Williams & Wilkins, pp 499–517. Available via https://www.cabidigitallibrary.org/doi/full/10.5555/20133232959 Chen, Y., Zhu, X., Yang, Y., Han, D., Jin, J. and Xie, S. 2014. Effect of dietary lysozyme on growth, immune response, intestine microbiota, intestine morphology and resistance to Aeromonas hydrophilia in gibel carp (C arassius auratus gibelio). Aquac. Nutr. 20(3), 229–241; doi:10.1111/anu.12069. Citarasu, T. 2010. Herbal biomedicines: a new opportunity for aquaculture industry. Aquac. Int. 18(3), 403–414; doi:10.1007/s10499-009-9253-7. Con, P., Nitzan, T., Slosman, T., Harpaz, S. and Cnaani, A. 2019. Peptide transporters in the primary gastrointestinal tract of pre-feeding Mozambique tilapia larva. Front. Physiol. 10, 808; doi:10.3389/fphys.2019.0080. de Groot, C. and Müller-Goymann, C.C. 2016. Saponin interactions with model membrane systems–Langmuir monolayer studies, hemolysis and formation of ISCOMs. Planta. Med. 82(18), 1496–1512; doi:10.1055/s-0042-118387. Demers, N.E. and Bayne, C.J. 1997. The immediate effects of stress on hormones and plasma lysozyme in rainbow trout. Dev. Comp. Immunol. 21(4), 363–373; doi:10.1016/s0145-305x(97)00009-8. Deng, H., Zhang, J., Yang, Q., Dong, X., Zhang, S., Liang, W., Tan, B. and Chi, S. 2023. Effects of dietary steroid saponins on growth performance, serum and liver glucose, lipid metabolism and immune molecules of hybrid groupers (♀ Epinephelus fuscoguttatus×♂ Epinephelus lanceolatu) fed high-lipid diets. Metabolites 13(2), 305; doi:10.3390/metabo13020305. Doumas, B.T., Bayse, D.D., Carter, R.J., Peters Jr, T. and Schaffer, R. 1981. A candidate reference method for determination of total protein in serum. I. Development and validation. Clin. Chem. 27(10), 1642–1650; doi:10.1093/clinchem/27.10.1651. Elbialy, Z.I., Rizk, M., Al-Hawary, I.I., Salah, A.S., Mohammed, R.A., Assar, D.H., Almeer, R. and Dawood, M.A. 2021. Yucca schidigera extract mediated the growth performance, hepato-renal function, antioxidative status and histopathological alterations in Nile tilapia (Oreochromis niloticus) exposed to hypoxia stress. Aquac. Res. 52(5), 1965–1976; doi:10.1111/are.15045. Elkaradawy, A., Abdel-Rahim, M.M., Albalawi, A.E., Althobaiti, N.A., Abozeid, A.M. and Mohamed, R.A. 2021. Synergistic effects of the soapbark tree, Quillaja saponaria and Vitamin E on water quality, growth performance, blood health, gills and intestine histomorphology of Nile tilapia, Oreochromis niloticus fingerlings. Aquac. Rep. 20, 100733; doi:10.1016/j.aqrep.2021.100733. Elkaradawy, A., Abdel-Rahim, M.M. and Mohamed, R.A. 2022. Quillaja saponaria and/or linseed oil improved growth performance, water quality, welfare profile and immune-oxidative status of Nile tilapia, Oreochromis niloticus fingerlings. Aquac. Res. 53(2), 576–589; doi:10.1111/are.15602. El-Naggar, K., Mohamed, R., El-katcha, M.I., Abdo, S.E. and Soltan, M.A. 2021. Plant ingredient diet supplemented with lecithin as fish meal and fish oil alternative affects growth performance, serum biochemical, lipid metabolism and growth-related gene expression in Nile tilapia. Aquac. Res. 52(12), 6308–632; doi:10.1111/are.15494. Estaiano de Rezende, R.A., Soares, M.P., Sampaio, F.G., Cardoso, I.L., Ishikawa, M.M., Lima Dallago, B.S., Rantin, F.T. and Teixeira Duarte, M.C. 2021. Phytobiotics blend as a dietary supplement for Nile tilapia health improvement. Fish Shellfish Immunol. 114, 293–300; doi:10.1016/j.fsi.2021.05.010. Fahrurrozi, A., Madusari, B.D., Syakirin, M.B., Mardiana, T.Y. and Ariadi, H. 2024. Optimization feeding rate towards saline tilapia production in stagnant waters, Pekalongan City. J. Aquac. Fish. Health. 13(1), 1–12; doi:10.20473/jafh.v13i1.49072. Fawole, F.J., Adeoye, A.A., Tiamiyu, L.O., Samuel, F.C., Omosuyi, O.M. and Amusa, M.T. 2020. Combinación dietética de semillas de papaya y polvo de cáscara de cebolla: impacto en el crecimiento, la hematología, los parámetros bioquímicos y el estado antioxidante de Clarias gariepinus. Aquac. Res. 51(7), 2903–2912; doi:10.1111/are.14629. Fleck, J.D., Betti, A.H., Da Silva, F.P., Troian, E.A., Olivaro, C., Ferreira, F. and Verza, S.G. 2019. Saponins from Quillaja saponaria and Quillaja brasiliensis: particular chemical characteristics and biological activities. Molecules 24(1), 171; doi:10.3390/molecules24010171. Francis, G., Levavi-Sivan, B., Avitan, A. and Becker, K. 2002a. Effects of long term feeding of Quillaja saponins on sex ratio, muscle and serum cholesterol and LH levels in Nile tilapia (Oreochromis niloticus (L.)). Comp. Biochem. Physiol. C. Toxicol. Pharmacol. 133(4), 593–603; doi:10.1016/s1532-0456(02)00167-9. Francis, G., Makkar, H.P. and Becker, K. 2002b. Dietary supplementation with a Quillaja saponin mixture improves growth performance and metabolic efficiency in common carp (Cyprinus carpio L.). Aquaculture 203(3–4), 311–320; doi:10.1016/S0044-8486(01)00628-7. Francis, G., Makkar, H.P. and Becker, K. 2005. Quillaja saponins—a natural growth promoter for fish. Anim. Feed. Sci. Technol. 121(1–2), 147–157; doi:10.1016/j.anifeedsci.2005.02.015. Furman, D. and Davis, M.M. 2015. New approaches to understanding the immune response to vaccination and infection. Vaccine 33(40), 5271–5281; doi:10.1016/j.vaccine.2015.06.117. Gaafar, A.Y., Yamashita, H., Istiqomah, I., Kawato, Y., Ninomiya, K., Abd, E.Y. and Nakai, T. 2018. An oral vaccination method with the aid of capsaicin against viral nervous necrosis (VNN). Fish. Pathol. 53(3), 110–113; doi:10.3147/jsfp.53.110. Gangabhagirathi, R. and Joshi, R. 2015. Antioxidant activity of capsaicin on radiationinduced oxidation of murine hepatic mitochondrial membrane preparation. Res. Rep. Biochem. 5, 163–171; doi:10.2147/RRBC.S84270. Gonçalves, R.A., Serradeiro, R., Machado, M., Costas, B., Hunger, C. and Dias, J. 2019. Interactive effects of dietary fishmeal level and plant essential oils supplementation on European sea bass, Dicentrarchus labrax: growth performance, nutrient utilization, and immunological response. J. World. Aquac. Soc. 50(6), 1078–1092; doi:10.1111/jwas.12616. Guo, H., Callaway, J.B. and Ting, J.P. 2015. Inflammasomes: mechanism of action, role in disease, and therapeutics. Nat. Med. 21(7), 677–687; doi:10.1038/nm.3893. Güroy, B., Mantoğlu, S., Merrifield, D.L. and Guroy, D. 2016. Effects of dietary Nutrafito Plus on growth, haemotological parameters and total ammonia-nitrogen excretion of juvenile striped catfish Pangasianodon hypophthalmus. Aquac. Res. 47(6), 1770–1777; doi:10.1111/are.12634. Harikrishnan, R., Kim, J.-S., Kim, M.-C., Balasundaram, C. and Heo, M.-S. 2011. Prunella vulgaris enhances the non-specific immune response and disease resistance of Paralichthys olivaceus against Uronema marinum. Aquaculture 318(1–2), 61–66; doi:10.1016/j.aquaculture.2011.05.020. Hassaan, M.S., Mohammady, E.Y., Soaudy, M.R., El-Garhy, H.A., Moustafa, M.M., Mohamed, S.A. and El-Haroun, E.R. 2019. Effect of Silybum marianum seeds as a feed additive on growth performance, serum biochemical indices, antioxidant status, and gene expression of Nile tilapia, Oreochromis niloticus (L.) fingerlings. Aquaculture 509, 178–187; doi:10.1016/j.aquaculture.2019.05.006. Heinegård, D. and Tiderström, G. 1973. Determination of serum creatinine by a direct colorimetric method. Clin. Chim. Acta. 43(3), 305–310; doi:10.1016/0009-8981(73)90466-X. Henry, R. 1964. Clinical chemistry, principles and techniques. New York: Harber and Row Publishers, pp: 181. Huang, Q., Huang, K., Ma, Y., Qin, X., Wen, Y., Sun, L. and Tang, L. 2015. Feeding frequency and rate effects on growth and physiology of juvenile genetically improved farmed Nile Tilapia. N. Am. J. Aquac, 77(4), 503-512. https://doi.org/10.1080/15222055.2015.1066472. Ibrahim, R.E., Rhouma, N.R., Elbealy, M.A., Abdelwarith, A.A., Younis, E.M., Khalil, S.S. and Rahman, A.N.A. 2024. Effect of dietary intervention with Capsicum annuum extract on growth performance, physiological status, innate immune response, and related gene expression in Nile tilapia. Comp. Biochem. Physiol. B. Biochem. Mol. Biol. 270, 110914; doi:10.1016/j.cbpb.2023.110914. Jain, N.C. 1986. Schalm’s veterinary hematology. Philadelphia, PA: Lea and Febiger, Available via https://www.cabidigitallibrary.org/doi/full/10.5555/19872289576. Kandeil, M.A., Mohammed, E.T., Hashem, K.S., Aleya, L. and Abdel-Daim, M.M. 2020. Moringa seed extract alleviates titanium oxide nanoparticles (TiO 2-NPs)-induced cerebral oxidative damage, and increases cerebral mitochondrial viability. Environ. Sci. Pollut. Res. Int. 27, 19169–19184; doi:10.1007/s11356-019-05514-2. Kawabata, F., Inoue, N., Masamoto, Y., Matsumura, S., Kimura, W., Kadowaki, M., Higashi, T., Tominaga, M., Inoue, K. and Fushiki, T. 2009. Non-pungent capsaicin analogs (capsinoids) increase metabolic rate and enhance thermogenesis via gastrointestinal TRPV1 in mice. Biosci. Biotechnol. Biochem. 73(12), 2690–2697; doi:10.1271/bbb.90555. Kawahara, E., Ueda, T. and Nomura, S. 1991. In vitro phagocytic activity of white-spotted char blood cells after injection with Aeromonas salmonicida extracellular products. Fish Pathol, 26(4), 213-214; doi:10.3147/jsfp.26.213. Khalafalla, M.M., Ibrahim, S.A., Zayed, M.M., Awad, M.N. and Mohamed, R.A. 2020. Effect of a dietary mixture of beneficial bacteria on growth performance, health condition, chemical composition, and water quality of Nile Tilapia, Oreochromis niloticus fingerlings. J. Aquat. Food. Prod. Technol. 29(8), 823–835; doi:10.1080/10498850.2020.1764685. Khieokhajonkhet, A., Suwannalers, P., Aeksiri, N., Ratanasut, K., Chitmanat, C., Inyawilert, W., Phromkunthong, W. and Kaneko, G. 2023. Effects of dietary red pepper extracts on growth, hematology, pigmentation, disease resistance, and growth-and immune-related gene expressions of goldfish (Carassius auratus). Anim. Feed. Sci. Technol. 301, 115658; doi:10.1016/j.anifeedsci.2023.115658. Limbu, S.M., Zhou, L., Sun, S.X., Zhang, M.L. and Du, Z.Y. 2018. Chronic exposure to low environmental concentrations and legal aquaculture doses of antibiotics cause systemic adverse effects in Nile tilapia and provoke differential human health risk. Environ. Int. 115, 205–219; doi:10.1016/j.envint.2018.03.034. Li, S., Sang, C., Zhang, J., Li, Z. and Chen, N. 2018. Molecular cloning, expression profiling of adipose triglyceride lipase (ATGL) and forkhead box O1 (FoxO1), and effects of dietary carbohydrate level on their expression in hybrid grouper (Epinephelus fuscoguttatus♀× E. lanceolatus♂). Aquaculture 492, 103–112; doi:10.1016/j.aquaculture.2018.03.062. Livak, K.J. and Schmittgen, T.D. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25(4), 402–408; doi:10.1006/meth.2001.1262. Low, C., Wadsworth, S., Burrells, C. and Secombes, C. 2003. Expression of immune genes in turbot (Scophthalmus maximus) fed a nucleotide-supplemented diet. Aquaculture 221(1–4), 23–40; doi:10.1016/S0044-8486(03)00022-X. Luís, Â., Sousa, S., Wackerlig, J., Dobusch, D., Duarte, A.P., Pereira, L. and Domingues, F. 2019. Star anise (Illicium verum Hook. f.) essential oil: antioxidant properties and antibacterial activity against Acinetobacter baumannii. Flavour. Fragr. J. 34(4), 260–270; doi:10.1002/ffj.3498. Marty-Roix, R., Vladimer, G.I., Pouliot, K., Weng, D., Buglione-Corbett, R., West, K., MacMicking, J.D., Chee, J.D., Wang, S. and Lu, S. 2016. Identification of QS-21 as an inflammasome-activating molecular component of saponin adjuvants. J. Biol. Chem. 291(3), 1123–1136; doi:10.1074/jbc.M115.683011. McCain, P.D. 2013. Capsaicin supplemented feed and its effects on stress modulation in Nile tilapia and Coho salmon. West Lafayette, IN: Purdue University. Mohamed, R.A., Yousef, Y.M., El-Tras, W.F. and Khalafallaa, M.M. 2021. Dietary essential oil extract from sweet orange (Citrus sinensis) and bitter lemon (Citrus limon) peels improved Nile tilapia performance and health status. Aquac. Res. 52(4), 1463–1479; doi:10.1111/are.15000. Mohanasundari, L., Devi, G.B., Musthafa, M.S. and Madhavi, M. 2022. Effects of Illicium verum Hook. f.(Chinese herb) enriched diet on growth performance, immune response and disease resistance in Catla catla [Hamilton] fingerlings against Aeromonas hydrophila. Fish Shellfish Immunol. 127, 455–462; doi:10.1016/j.fsi.2022.06.050. Mommsen, T.P., Vijayan, M.M. and Moon, T.W. 1999. Cortisol in teleosts: dynamics, mechanisms of action, and metabolic regulation. Rev. Fish. Biol. Fish. 9(3), 211–268; doi:10.1023/A:1008924418720. Newberne, P., Smith, R., Doull, J., Goodman, J., Munro, I., Portoghese, P., Wagner, B., Weil, C., Woods, L. and Adams, T. 1999. The FEMA GRAS assessment of trans-anethole used as a flavouring substance. Food Chem. Toxicol. 37(7), 789–811; doi:10.1016/S0278-6915(99)00037-X. Ng’ambi, J.W., Li, R., Mu, C., Song, W., Liu, L. and Wang, C. 2016. Dietary administration of saponin stimulates growth of the swimming crab Portunus trituberculatus and enhances its resistance against Vibrio alginolyticus infection. Fish Shellfish Immunol. 59, 305–311; doi:10.1016/j.fsi.2016.10.041. Oda, K., Matsuda, H., Murakami, T., Katayama, S., Ohgitani, T. and Yoshikawa, M. 2000. Adjuvant and haemolytic activities of 47 saponins derived from medicinal and food plants. Biol. Chem. 381(1), 67–74; doi:10.1515/BC.2000.009. Oleszek, M. and Oleszek, W. 2020. Saponins in food. Singapore: Springer. Prakash, U.N. and Srinivasan, K. 2013. Enhanced intestinal uptake of iron, zinc and calcium in rats fed pungent spice principles–piperine, capsaicin and ginger (Zingiber officinale). J. Trace. Elem. Med. Biol. 27(3), 184–190; doi:10.1016/j.jtemb.2012.11.003. Platel, K. and Srinivasan, K. 2004. Digestive stimulant action of spices: a myth or reality? Indian J. Med. Res. 119(5), 167. Rahman, A.N.A., Amer, S.A., Masoud, S.R., El-Saber, M.M., Osman, A., Younis, E.M., Abdelwarith, A.A., Davies, S.J., Khamis, T. and Ibrahim, R.E. 2023. Neem seed protein hydrolysate as a fishmeal substitute in Nile tilapia: effects on antioxidant/immune pathway, growth, amino acid transporters-related gene expression, and Aeromonas veronii resistance. Aquaculture 573, 739593; doi:10.1016/j.aquaculture.2023.739593. Rao, Α. and Gurfinkel, D. 2000. The bioactivity of saponins: triterpenoid and steroidal glycosides. Drug Metab. Dispos. 17(1–4), 211–236; doi:10.1515/dmdi.2000.17.1-4.211. Reitman, S. 1957. Un método colorimétrico para la determinación de la glutamato oxaloacetato sérico y la glutamato piruvato transaminasa sérica. Am. J. Clin. Pathol. 28, 56; doi:10.1093/ajcp/28.1.56. Roldan-Juarez, J., Pinares, R., Smith, C.E., Llerena, C.A., Machaca, V. and Pizarro, D.M. 2023. Microencapsulated essential oils influence the growth and foregut histomorphometry of Nile tilapia (Oreochromis niloticus) fingerlings. Vet. Anim. Sci, 22, 100316. https://doi.org/10.1016/j.vas.2023.100316. Ruchin, A.B. 2020. Environmental colour impact on the life of lower aquatic vertebrates: development, growth, physiological and biochemical processes. Rev. Aquac. 12(1), 310–327; doi:10.1111/raq.12319. Salama, M.S., Morsy, W.A., Mohamed, R.A. and El-Midany, S.A. 2019. Effect of some feed-additives on the growth performance, physiological response and histopathological changes of rabbits subjected to ochratoxin-A feed contamination. Sloven. Vet. Res. 56, 499–508; doi:10.26873/SVR-787-2019. Serrano Jr, A. 2013. Effects of Quillaja saponins on growth, feed efficiency, digestive enzyme activities and metabolism of common carp (Cyprinus carpio L). Aquac. Nutr. 19(4), 468–474; doi:10.1111/j.1365-2095.2012.00980.x. Shahverdi, A., Kheiri, F., Faghani, M., Rahimian, Y. and Rafiee, A. 2013. The effect of use red pepper (Capsicum annum L.) and black pepper (Piper nigrum L.) on performance and hematological parameters of broiler chicks. Eur. J. Zool Res, 2(6), 44–48. Spencer, L., Bancroft, J., Bancroft, J. and Gamble, M. 2012. Tissue processing. Bancroft’s theory and practice of histological techniques. 7th ed. Netherlands, Amsterdam: Elsevier Health Sciences, pp: 105–123. Steiner, T. and Syed, B. 2015. Phytogenic feed additives in animal nutrition. In Medicinal and aromatic plants of the world. Eds. Máthé, Á. Dordrecht, Springer, pp: 403–423; doi:10.1007/978-94-017-9810-5_20. Suvarna, S., Layton, C. and Bancroft, J. 2013. The hematoxylins and eosin. Bancroft’s Theory and Practice of Histological Techniques, 7th ed. London, UK: Churchill Livingstone, pp: 172–186; doi:10.1016/B978-0-7020-4226-3.00010-X. Tewksbury, J.J., Reagan, K.M., Machnicki, N.J., Carlo, T.A., Haak, D.C., Peñaloza, A.L.C. and Levey, D.J. 2008. Evolutionary ecology of pungency in wild chilies. PNAS 105(33), 11808–11811; doi:10.1073/pnas.0802691105. Thrall, M., Baker, D. and Lassen, E. 2004. Veterinary haematology and clinical chemistry. Philadelphia, USA: Lippincott Williams and Wilkins. Trinder, P. 1969. Determination of glucose in blood using glucose oxidase with an alternative oxygen acceptor. Ann. Clin. Biochem. 6(1), 24–27; doi:10.1177/000456326900600108. Valdivieso-Ugarte, M., Gomez-Llorente, C., Plaza-Díaz, J. and Gil, Á. 2019. Antimicrobial, antioxidant, and immunomodulatory properties of essential oils: a systematic review. Nutrients 11(11), 2786; doi:10.3390/nu11112786. Van der Meide, P. and Schellekens, H. 1996. Cytokines and the immune response. Biotherapy 8(3-4), 243–249; doi:10.1007/BF01877210. Van Soest, P.J., Robertson, J.B. and Lewis, B.A. 1991. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy. Sci. 74(10), 3583–3597; doi:10.3168/jds.S0022-0302(91)78551-2. Wang, L., Wu, D., Fan, Z., Li, H., Li, J., Zhang, Y., Xu, Q., Wang, G. and Zhu, Z. 2020. Effect of Yucca schidigera extract on the growth performance, intestinal antioxidant status, immune response, and tight junctions of mirror carp (Cyprinus carpio). Fish Shellfish Immunol. 103, 211–219; doi:10.1016/j.fsi.2020.05.039. Yilmaz, S., Çelik, E.Ş., Ergün, S., Gürkan, M., Kesbic, F.I. and Abdel-Latif, H.M. 2024. The effects of Capsicum annuum oleoresin, as a dietary carotenoid, on growth, gut microbiome, intestinal histomorphometry, and sensory characteristics of Oncorhynchus mykiss. J. World Aquac. Soc. 55(1), 149–168; doi:10.1111/jwas.13039. Yu, H., Zheng, L., Yin, L., Xu, L., Qi, Y., Han, X., Xu, Y., Liu, K. and Peng, J. 2014. Protective effects of the total saponins from Dioscorea nipponica Makino against carbon tetrachloride-induced liver injury in mice through suppression of apoptosis and inflammation. Int. Immunopharmacol. 19(2), 233–244; doi:10.1016/j.intimp.2014.01.019. Zhang, X., Huang, K., Zhong, H., Ma, Y., Guo, Z., Tang, Z., Liang, J., Luo, Y., Su, Z. and Wang, L. 2020. Effects of Lycium barbarum polysaccharides on immunological parameters, apoptosis, and growth performance of Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 97, 509–514; doi:10.1016/j.fsi.2019.12.068. | ||
| How to Cite this Article |
| Pubmed Style Mabrouk SG, El-nokrashy AM, Ebied NA, Abdella BH, Zayed MM, Aboleila SM, Mohamed RA. A Blend of natural phytobiotics enhances growth performance, feed efficiency, and the immuno-health status of fingerlings of Nile tilapia (Oreochromis niloticus). Open Vet. J.. 2025; 15(2): 746-764. doi:10.5455/OVJ.2025.v15.i2.24 Web Style Mabrouk SG, El-nokrashy AM, Ebied NA, Abdella BH, Zayed MM, Aboleila SM, Mohamed RA. A Blend of natural phytobiotics enhances growth performance, feed efficiency, and the immuno-health status of fingerlings of Nile tilapia (Oreochromis niloticus). https://www.openveterinaryjournal.com/?mno=226841 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.24 AMA (American Medical Association) Style Mabrouk SG, El-nokrashy AM, Ebied NA, Abdella BH, Zayed MM, Aboleila SM, Mohamed RA. A Blend of natural phytobiotics enhances growth performance, feed efficiency, and the immuno-health status of fingerlings of Nile tilapia (Oreochromis niloticus). Open Vet. J.. 2025; 15(2): 746-764. doi:10.5455/OVJ.2025.v15.i2.24 Vancouver/ICMJE Style Mabrouk SG, El-nokrashy AM, Ebied NA, Abdella BH, Zayed MM, Aboleila SM, Mohamed RA. A Blend of natural phytobiotics enhances growth performance, feed efficiency, and the immuno-health status of fingerlings of Nile tilapia (Oreochromis niloticus). Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 746-764. doi:10.5455/OVJ.2025.v15.i2.24 Harvard Style Mabrouk, S. G., El-nokrashy, . A. M., Ebied, . N. A., Abdella, . B. H., Zayed, . M. M., Aboleila, . S. M. & Mohamed, . R. A. (2025) A Blend of natural phytobiotics enhances growth performance, feed efficiency, and the immuno-health status of fingerlings of Nile tilapia (Oreochromis niloticus). Open Vet. J., 15 (2), 746-764. doi:10.5455/OVJ.2025.v15.i2.24 Turabian Style Mabrouk, Shreifa G., Asmaa M. El-nokrashy, Nahla A. Ebied, Bahaa H. Abdella, Mohamed M. Zayed, Sayed M. Aboleila, and Radi A. Mohamed. 2025. A Blend of natural phytobiotics enhances growth performance, feed efficiency, and the immuno-health status of fingerlings of Nile tilapia (Oreochromis niloticus). Open Veterinary Journal, 15 (2), 746-764. doi:10.5455/OVJ.2025.v15.i2.24 Chicago Style Mabrouk, Shreifa G., Asmaa M. El-nokrashy, Nahla A. Ebied, Bahaa H. Abdella, Mohamed M. Zayed, Sayed M. Aboleila, and Radi A. Mohamed. "A Blend of natural phytobiotics enhances growth performance, feed efficiency, and the immuno-health status of fingerlings of Nile tilapia (Oreochromis niloticus)." Open Veterinary Journal 15 (2025), 746-764. doi:10.5455/OVJ.2025.v15.i2.24 MLA (The Modern Language Association) Style Mabrouk, Shreifa G., Asmaa M. El-nokrashy, Nahla A. Ebied, Bahaa H. Abdella, Mohamed M. Zayed, Sayed M. Aboleila, and Radi A. Mohamed. "A Blend of natural phytobiotics enhances growth performance, feed efficiency, and the immuno-health status of fingerlings of Nile tilapia (Oreochromis niloticus)." Open Veterinary Journal 15.2 (2025), 746-764. Print. doi:10.5455/OVJ.2025.v15.i2.24 APA (American Psychological Association) Style Mabrouk, S. G., El-nokrashy, . A. M., Ebied, . N. A., Abdella, . B. H., Zayed, . M. M., Aboleila, . S. M. & Mohamed, . R. A. (2025) A Blend of natural phytobiotics enhances growth performance, feed efficiency, and the immuno-health status of fingerlings of Nile tilapia (Oreochromis niloticus). Open Veterinary Journal, 15 (2), 746-764. doi:10.5455/OVJ.2025.v15.i2.24 |