| Review Article | ||
Open Vet. J.. 2025; 15(2): 533-540 Open Veterinary Journal, (2025), Vol. 15(2): 533-540 Review Article Global initiatives to phase-out colistin use in food-producing animalsMohamed Omar Ahmed1*, Yousef M. Abouzeed1 and Mohamed Ali Daw21Department of Microbiology and Parasitology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 2Department of Medical Microbiology and Immunology, Faculty of Medicine, University of Tripoli, Tripoli, Libya *Corresponding Author: Mohamed Omar Ahmed. Department of Microbiology and Parasitology, Faculty of Veterinary Medicine, University of Tripoli, Libya. a.mo [at] live.com; m.ahmed [at] uot.edu.ly Submitted: 04/11/2024 Accepted: 20/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
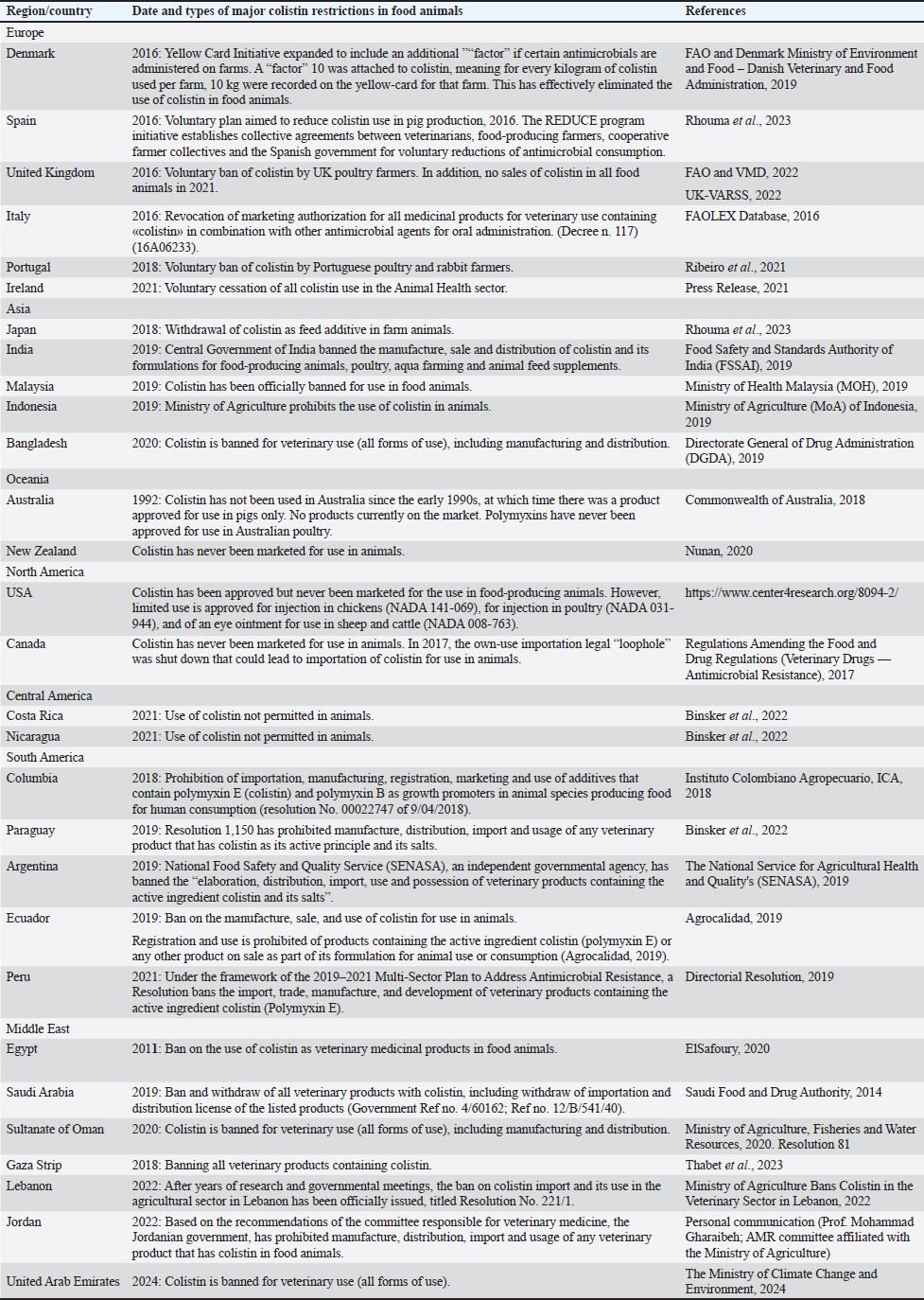
AbstractColistin (polymyxin E) is a former exclusive veterinary antimicrobial and one of the oldest antibiotics that is currently recognized as one of the highest priority and critically important antimicrobials for human medicine. For food animals, colistin is extensively used for multiple medical and nonmedical purposes, particularly for growth promotion, prophylaxis/metaphylaxis, and therapeutic purposes. As a result, colistin resistance is widespread along the food chain and is linked to multidrug-resistant bacterial infections in humans. Furthermore, human medicine is more reliant on colistin for serious infections in healthcare settings. As a result, different types of national bans/restrictions in food- producing animals have been implemented around the world. Conclusions: The testimonies of 29 countries representing global initiatives to phase-out colistin use in animal production. Keywords: Colistin (polymyxin E), Global public health, Food producing animals IntroductionOne of the oldest and previous veterinary-exclusive antibiotics, colistin (also known as Polymyxin E) is now considered one of the most significant and high- priority antibiotics for human medicine. For food animals, colistin is extensively used for several medical and nonmedical purposes, including prophylaxis/ metaphylaxis, growth promotion, and therapeutic purposes (Ortwine et al., 2015). Colistin is one of the most significant and high-priority antimicrobials, according to the World Health Organization. However, the World Organization for Animal Health (formerly known as the Office International des Epizooties (OIE) has never declared that colistin is critically important for animals. Over the past decades, the rapid global dissemination of colistin resistance has been documented in bacteria originating from animals and humans (Ali et al., 2024). Additionally, resistance to colistin was reported throughout the food chain and is linked to human clinical infections, including the environment, food- producing animals, retail meat, and asymptomatic individuals and patients whom have never been exposed to colistin, particularly young children (Peñalva et al., 2022). Following the initial reports of mobilized colistin resistance (mcr) genes conferring plasmid- mediated resistance to colistin (i.e., mcr-1 gene) in Escherichia coli in China, different variants of this gene and other numerous subvariants of mcr-2 to mcr- 10 have been reported (Shen et al., 2020; Sharma et al., 2022; Ali et al., 2024). The phylogeny and spread of mcr-1˜10 genes and variants vary greatly, suggesting different evolutionary advantages worldwide (Li et al., 2021). A major public health concern is the increased global spread of antimicrobial resistance among various bacterial species, including zoonotic pathogens, which is facilitated by the higher prevalence of mobile mcr genes among animal bacterial isolates and the significantly higher consumption of colistin in livestock compared to human medicine in several countries (Binsker et al., 2022; Tokuda et al., 2024). On the other hand, a number of independent studies have demonstrated that drastic reductions of colistin use for food-producing animals—rather than in human medicine—have resulted in significant reduction in colistin-resistant bacteria in both food-producing animals and humans, including both asymptomatic human carriers and infected individuals (Fournier et al., 2020; Shen et al., 2020; Wang et al., 2020; Dierikx et al., 2022; Khine et al., 2022). For the majority of food-producing animal species, colistin is marketed in the European Union (EU) as a “Treatment and metaphylaxis of enteric infections caused by non-invasive E. coli susceptible to colistin sulphate.” In some non-EU regions, colistin is marketed for the treatment of Salmonella and colibacillosis in animals used for food production. Minor applications of various polymyxins for ear and eye infections in pets can be administered alone or in combination with other antibiotics. In horses/foals, off-label uses of human polymyxin B intravenous formulations are given at sub-therapeutic concentrations for endotoxemia. In human medicine, the critical need for colistin as a last-resort therapy is becoming increasingly needed. The use of colistin to treat infections caused by carbapenem-resistant Enterobacterales, multidrug- resistant (MDR)-Acinetobacter spp., MDR-Klebsiella spp., and MDR-Pseudomonas spp. is on the rise in hospitals and intensive care units (Ortwine et al., 2015). Infections by MDR Gram-negative bacteria are a growing threat to global healthcare, as reflected by greater use of colistin in hospitals/ICUs for the treatment of infections caused by carbapenem- resistant Enterobacterales, MDR-Acinetobacter spp., MDR-Klebsiella spp., and MDR-Pseudomonas spp. (Ortwine et al., 2015). Colistin is also increasingly administered via inhalation for the treatment of infections in patients with cystic fibrosis and ventilator- associated pneumonia. With an estimated 2,500 deaths (EU/EEA regions) linked to colistin-resistant Gram- negative bacteria in 2015, infections by carbapenem- resistant Gram-negative bacteria were linked to high mortalities (Cassini et al., 2019). MDR-Acinetobacter species and MDR-Klebsiella pneumonia are also reported from human clinical samples from developing countries expressing colistin resistance-carbapenemase producing phenotypes (Kieffer et al., 2018). These major concerns about agriculture colistin use have resulted in various national decisions on types of colistin bans/restrictions in food-producing animals. Several countries have taken the final decision to not allow colistin for all types of use, including therapeutic, for major food-producing animal populations. Some countries have banned colistin for use as growth promoters (feed additives), including Brazil, Chile, Uruguay, China, Japan, South Korea, Nepal, South Africa, and Vietnam. Thailand has gone a step further by further prohibiting prophylactic use of colistin sulfate for food-producing animals, from 2017 onwards (Wang et al., 2020). MethodsWeb-based methods of literature searches and internet search engines were adopted, including websites of relevant government agencies, web pages, images, information, and any other types of files. Internet-based searches of potential search engines and databases were also adopted, including Google, Google Scholar, MEDLINE, and PubMed. In addition, government agencies were contacted to confirm colistin restrictions. ResultsThe national efforts of 29 countries that have made the definitive choice to prohibit the use of colistin for any purpose, including medicinal purposes, for major food-producing animal populations have been summarized (Table 1). This has been accomplished by either totally prohibiting the use of colistin in animals, never approving its use in food-producing animals for national marketing purposes, or imposing strict regulations (voluntary or not) on large food-producing animals that have led to a significant decline in the country’s colistin sales for animals. DiscussionA growing number of countries throughout the world either forbid or severely restrict the use of colistin for major food animal populations (Table 1). Combined, these countries represent the majority of the world food-producing animal production. Since the main populations of animals used for food production stopped using colistin, there have been no related reports from these countries indicating any issues with animal health or productivity. It is possible that other nations are considering or have already imposed comparable limitations on the use of colistin for food animals. Thus, with a wider global view, and then the combined judgement of 29 countries (Table 1) gives robust evidence that colistin is not needed for food- producing animals. In 2016, the European Commission announced the withdrawal of all marketing authorizations for colistin-containing veterinary medicinal products in combination with other antimicrobial substances to be administered orally, with no reported negative impact on European food animal production. Although sales of monotherapy colistin have decreased in Europe, 136.5 tonnes of colistin were sold in 2021, of which 99.3% were oral mass medication formulations marketed for food-producing animals administered to both healthy and diseased animals (European Medicines Agency, 2022). This corresponded to an overall aggregated EU colistin sales, in animals, of 2.2 mg/ population- corrected units (PCUs) in 2021, as compared to approximately 0.06 mg/PCU for the European human population polymyxin consumption, based on previous reports (European Centre for Disease Prevention and Control, European Food Safety Authority and European Medicines Agency, 2021). The population correction unit, referred to as PCU, is a standardized denominator for European animal sales data (PCU in 1,000 tonnes) and serves to normalize the total quantities of antibiotic- active substances sold in each country by the animal population that could be potentially treated. Collectively, these countries are shaping global One Health initiatives and efforts related to the use of antibiotics in food animals. Furthermore, no sales of colistin for animals are routinely reported by Finland, Iceland, and Norway (European Medicines Agency, 2022). Together, Denmark and Spain account for the bulk of pig production in the EU, and they achieve this without the use of colistin adopting various novel strategies that aim to eradicate colistin usage (Table 1). Table 1 Countries/regions that have e”ither banned, heavily restricted or never marketed colistin for therapeutic use in food-producing animals.
Other nations such as France, Italy, the Netherlands, and Estonia have set national targets for veterinary colistin reductions (European Medicines Agency, 2016); these targets were exceeded (European Medicines Agency, 2022). In young food-producing animals, E. coli diseases are typically manifested as gastrointestinal infections, known as colibacillosis, where only orally administered colistin achieves high gastrointestinal concentrations as it is not appreciably bioavailable to the systemic circulation. In chickens, colibacillosis differs in that the common signs are either localized (e.g., omphalitis) or systemic (e.g., colisepticemia), whereas orally administered colistin does not attain the`rapeutic concentrations at the site of infection (Eriksen et al., 2021). Colistin is effective against primary diarrheal colibacillosis, which is rare in chickens. This suggests that an alternative use of colistin is to decolonize the gastrointestinal tract of E. coli and thus reduce indoor E. coli-laden dust levels, the likely common cause of chicken colibacillosis. The most common traditional uses of colistin is for post-weaning diarrhea (PWD) in piglets, followed by colibacillosis in other food-producing animal species. PWD is a multifactorial condition occurring within the first 14 days after weaning where diarrhea (i.e., defecation with increased rate, volume, and water content) results from a combination of risk factors (e.g., breed, age/weight at weaning, colostrum intake/ quality, previous vaccination, heat/cold stress, diet characteristics, mixing unfamiliar animals) commonly enhanced by infection with specific pathogens, especially enterotoxigenic E. coli (ETEC) (Eriksen et al., 2022). However, PWD without detectable ETEC is also common, where intestinal dysbiosis from the weaning process and diet changes can lead to intestinal inflammation and diarrhea (Eriksen et al., 2022). Rotavirus and coronavirus gastrointestinal infections can be contributing pathogens. Evidence- based research studies provide several husbandry management tools to either prevent PWD or reduce the incidence/severity (Cassini et al., 2019). Furthermore, Raasch et al. (2020) assessed the effectiveness of alternative measures in pig production, such as improvement of biosecurity, vaccination, improved feeding, and health care, and found a significant reduction in colistin consumption as the result of the implementation of different measures. Widely available E. coli vaccines, for preventing colibacillosis in many food-producing animal species are marketed for third-trimester pregnant animals to boost colostral quality/immunity, or to be given to neonates prior to weaning. Live apathogenic E. coli vaccines are available as well as inactivated E. coli vaccines and vaccines against E. coli components (e.g., fimbrial adhesin antigens) that provide E. coli strains with selective advantages to attach to the intestinal wall and cause disease. Estonia is an example of a country with significant pig production that previously had low sales of doses of E. coli vaccines, but high sales of colistin (almost doubled from 2010 to 2013) (Sammul et al., 2021). A change in national policy led to a dramatic increase in sales of E. coli vaccines followed by a proportional decrease in colistin sales by 92.5% in total. It was shown on a national population scale that the consumption of colistin in pigs decreased on average by 0.23 mg/PCU (95% confidence interval: 0.06–0.39) for every 10,000 E. coli vaccine doses sold over 10 years (Sammul et al., 2021). The wealth of evidence-based husbandry management tools to either prevent colibacillosis or reduce its incidence/severity, including the use of widely available vaccines and alternative antimicrobials, demonstrates that adequate alternative medicinal products are available as well as sufficient knowledge about good husbandry practices that will reduce infection burdens on intensively driven farming. On their website news stream, the Federation of Veterinarians of Europe has taken a position that a ban on veterinary use of colistin would impact animal welfare, based on the need for treating MDR Gram-negative infections in food- producing animals. However, with the culmination of years of previous colibacillosis animal husbandry practices leading to MDR E. coli strains, then the solution is not the use of more-and-more critically important antimicrobials, but to change husbandry practices towards prevention, including vaccine use. This is especially the case with non-ETEC PWD from intestinal dysbiosis. European surveillance of veterinary antimicrobial consumption (ESVAC) (2022) reports on a European level that although veterinary colistin sales are decreased, it is not matched by increased sales of 3rd/4th generation cephalosporins and fluoroquinolones (European Medicines Agency, 2022), suggesting that treating MDR E. coli strains are a minor issue in food animals. In addition, colistin use exerts a major selection pressure on animal bacterial populations for co- selection of resistance to other antibiotic classes. Colistin resistance carried by genetic elements has been identified in a range of mobilized plasmids (IncI2, IncX4 and IncFIA type plasmids) carrying mcr genes and other MDR genes against many antibiotic classes including β-lactamases and penicillins, carbapenems, fluoroquinolones, aminoglycosides, tetracyclines, and trimethoprim sulfonamides (Mead et al., 2022). Thus, One Health perspectives of colistin use in agriculture must recognize that increases in mass medication practices correspond to increased colistin multi- resistant bacteria that negatively impact public health. The EU was the latest region to consider banning certain antimicrobials for use in animals. This was done through the establishment of a list of antimicrobials designated for human use only, specified in an EC implementing act (EU 2022/1255) (eur-lex.europa.eu/ legal-content/EN/TXT/?uri=CELEX:32022R1255). The criteria for establishing the list was based on a European Parliament (EP) Delegated Act (EU 2021/1760) (eur-lex.europa.eu/legal-content/EN/ TXT/?uri=CELEX%3A32021R1760), whereby conditions were applied to assess if an antimicrobial/ antimicrobial class fulfills all three established criteria (A-High importance for human health, B-Risk of transmission of resistance from animals to humans, C-Non-essential need for animal health) to be designated for human-use only. Although this EU approach led to the conclusion that colistin did not qualify for the human-reserved list, based on not fulfilling Criterion C, it is still worthwhile to re- examine colistin from a global One Health perspective, since the Criteria are published public information and represent the latest high-level government scheme to assess if antimicrobials are essential for animal health. In conclusion, colistin is marketed for the treatment and metaphylaxis in food-producing animals. However, alternative and preventative strategies, including vaccines, can be used for the important food- producing animals to limit the morbidity and mortality from clinical infection and drug-resistant microbes. Testimonies of 29 countries, representing the bulk of global food-producing animal production, provide robust evidence that colistin is not essential for its marketed use, clearly demonstrating that even intensive animal production can be maintained without the use of this antibiotic. Their combined national responses are in line with the clinical significance of colistin for human medicine and the broad spread of antimicrobial resistance in food animals. AcknowledgmentsNot applicable. Conflict of interestsThe authors declare that they have no competing interests. FundingThe authors received no financial support for the research, authorship, and/or publication of this article. Author’s contributionsMOA designed the plan of the manuscript and prepared the manuscript. MAD and YMA validated the study and revised the manuscript. All authors read and approved the final manuscript. ReferencesAgrocalidad. 2019. The executive director of the phyto and zoosanitary regulation and control agency – resolución 0003. Available via https://www.agrocalidad.gob.ec/wp-content/uploads/2020/05/dol6.pdf Ali, R.I., El-Abdelaziz, S.A., Kamel, M.A., Murad, S.K., Abdallah, H.M. and Salem, G.A. 2024. Phenotypic and genotypic characterization of extended spectrum beta-lactamase producing E. coli harboring carbapenem and colistin-resistant genes from poultry farms in Egypt. Open Vet. J. 14(1), 459–469. Argentina Ministry of Justice and Human Rights. 2019. RESOL-2019-22-APN-PRES # SENASA - Veterinary products: prohibition of processing, distribution, import, use and possession. Binsker, U., Käsbohrer, A. and Hammerl, J.A. 2022. Global colistin use: a review of the emergence of resistant Enterobacterales and the impact on their genetic basis. FEMS Microbiol. Rev. 46, fuab049. Cassini, A., Högberg, L.D., Plachouras, D., Quattrocchi, A., Hoxha, A., Simonsen, G.S., Colomb-Cotinat, M., Kretzschmar, M.E., Devleesschauwer, B., Cecchini, M. and Ouakrim, D.A. 2019. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: a population-level modelling analysis. Lancet Infect. Dis. 19(1), 56–66. Commonwealth of Australia. 2018. Importance ratings and summary of antibacterial uses in human and animal health in Australia. Available via amr.gov.au/about-amr/amr-australia/amr-and-animal-health-australia Directorate General of Drug Administration (DGDA). 2019. Default suspended/cancel products. Available via http://dgdagov.info/index.php/registered-products/suspend-cancel-products/1532-notifucation-07-05-2019-2/file Dierikx, C.M., Meijs, A.P., Hengeveld, P.D., van der Klis, F.R., van Vliet, J., Gijsbers, E.F., Rozwandowicz, M., van Hoek, A.H., Hendrickx, A.P., Hordijk, J. and Van Duijkeren, E. 2022. Colistin-resistant Enterobacterales among veterinary healthcare workers and in the Dutch population. JAC Antimicrob. Resist. 4(2), dlac041. Directorial Resolution. 2019. Nº 0091-2019-MINAGRI-SENASA-DIAIA, Ministry of Agriculture and Irrigation (Minagri). ElSafoury, H. 2020. Legislative status of veterinary medicinal products in Egypt. Technical report under action A2 of the Egyptian Vulture New LIFE project. Egypt: Nature Conservation Egypt, 55 p. Available via https://www.lifeneophron.eu/back2/public/files/documents/elsafoury-et-al-2020-5e5cf4605ddd7.pdf Eriksen, E.Ø., Kudirkiene, E., Christensen, A.E., Agerlin, M.V., Weber, N.R., Nødtvedt, A., Nielsen, J.P., Hartmann, K.T., Skade, L., Larsen, L.E., Pankoke, K., Olsen, J.E., Jensen, H.E. and Pedersen, K.S. 2021. Post-weaning diarrhea in pigs weaned without medicinal zinc: risk factors, pathogen dynamics, and association to growth rate. Porcine Health Manag. 7(1), 54. Eriksen, E.Ø., Pedersen, K.S., Larsen, I. and Nielsen, J.P. 2022. Evidence-based recommendations for herd health management of porcine post-weaning diarrhea. Animals 12(14), 1737. European Centre for Disease Prevention and Control, European Food Safety Authority and European Medicines Agency. 2021. Third joint inter-agency report on integrated analysis of consumption of antimicrobial agents and occurrence of antimicrobial resistance in bacteria from humans and food-producing animals in the EU/EEA, JIACRA III. 2016–2018. Stockholm, Parma, Amsterdam: ECDC, EFSA, EMA. European Medicines Agency (EMA). 2022. European surveillance of veterinary antimicrobial consumption (ESVAC). ‘Sales of veterinary antimicrobial agents in 31 European countries in 2021’ (EMA/795956/2022). European Medicines Agency (EMA). 2016. Updated advice on the use of colistin products in animals within the European Union: development of resistance and possible impact on human and animal health EMA/231573. FAO and Denmark Ministry of Environment and Food – Danish Veterinary and Food Administration. 2019. Tackling antimicrobial use and resistance in pig production: lessons learned from Denmark. Rome, 52 pp. Licence: CC BY-NC-SA 3.0 IGO. FAO and VMD. 2022. Tackling antimicrobial use and resistance in food-producing animals – Lessons learned in the United Kingdom. Rome; doi:https://doi.org/10.4060/cc0927en FAOLEX Database. 2016. Decree 25 July 2016-Revocation of the marketing authorizations of all veterinary medicinal products containing “colistin” in combination with other antimicrobial agents for oral administration. Official Gazette of the Italian Republic 197/2016. Available via https://www.fao.org/faolex/results/details/en/c/LEX-FAOC176432/ Food Safety and Standards Authority of India (FSSAI). 2019. Direction under section 16(5)read with 18(2)(d)of Food Safety and Standard Act, 2006 regarding operationalisation of draft food safety and standard (contaminants, toxins and residues) amendment regulations. Available via https://www.fssai.gov.in/upload/advisories/2019/08/5d4c042779d77Direction_Colistin_Ban_FSSAI_08_08_2019.pdf (Accessed 8 September 2019). Fournier, C., Aires-de-Sousa, M., Nordmann, P. and Poirel, L. 2020. Occurrence of CTX-M-15- and MCR-1-producing Enterobacterales in pigs in Portugal: evidence of direct links with antibiotic selective pressure. Int. J. Antimicrob. Agents 55, 105802. Instituto Colombiano Agropecuario, ICA. 2018. Resolución No. 00022747 (09/04/2018). Por medio de la cual se prohíbe la importación, fabricación, registro, comercialización y uso de aditivos que contengan polimixina E (colistina) y polimixina B como promotores de crecimiento en especies animales productoras de alimentos para el consumo humano. Available via https://www.ica.gov.co/getattachment/4972ba67-e1ba-4b2a-89ed-09f54f5c62b4/2018R22747.aspx (Accessed 17 August 2021). Khine, N.O., Lugsomya, K., Niyomtham, W., Pongpan, T., Hampson, D.J. and Prapasarakul, N. 2022. Longitudinal monitoring reveals persistence of colistin-resistant Escherichia coli on a pig farm following cessation of colistin use. Front. Vet. Sci. 9, 845746. Kieffer, N., Ahmed, M.O., Elramalli, A.K., Daw, M.A., Poirel, L., Álvarez, R. and Nordmann, P. 2018. Colistin-resistant carbapenemase-producing isolates among Klebsiella spp. and Acinetobacter baumannii in Tripoli, Libya. J. Glob. Antimicrob. Resist. 13, 37–39. Li, W., Liu, Z., Yin, W., Yang, L., Qiao, L., Song, S., Ling, Z., Zheng, R., Wu, C., Wang, Y. and Shen, J. 2021. MCR expression conferring varied fitness costs on host bacteria and affecting bacteria virulence. Antibiotics 10(7), 872. Mead, A., Billon-Lotz, C., Olsen, R., Swift, B., Richez, P., Stabler, R. and Pelligand, L. 2022. Epidemiological prevalence of phenotypical resistances and mobilised colistin resistance in avian commensal and pathogenic E. coli from Denmark, France, The Netherlands, and the UK. Antibiotics 11(5), 631. Ministry of Agriculture (MoA) of Indonesia. 2019. Through the directorate general of livestock and animal health services (DGLAHS) issuing regulation No 09160/PK.350/F/12/. Ministry of Agriculture Bans Colistin in the Veterinary Sector in Lebanon. 2022. Resolution No. 221/1 of 2022 banning the Import, Circulation, and Use of Colistin in the Veterinary Sector in Lebanon. Available via leb210250E.pdf and https://faolex.fao.org/docs/pdf/leb210250.pdf Ministry of Agriculture, Fisheries and Water Resources. 2020. Prohibiting and restricting the import, circulation and use of certain veterinary preparations. Resolution 81/2020. Available via https://www.mafwr.gov.om:4442/Uploads/Documents/637266032993614990.pdf Ministry of Health Malaysia (MOH). 2019. Joint press statement Ministry of Health Malaysia and Ministry of Agriculture and Agro-based Industry: national antibiotic awareness week campaign. Available via www.moh.gov.my/index.php/database_stores/attach_download/657/1259 Nunan, C. 2020. Farm antibiotics and trade deals—Could UK standards be undermined? Alliance to save our antibiotics. Available via https://www.saveourantibiotics.org/media/1864/farm-antibiotics-and-trade-could-uk-standards-be-undermined-asoa-nov-2020.pdf Ortwine, J.K., Kaye, K.S., Li, J. and Pogue, J.M. 2015. Colistin: understanding and applying recent pharmacokinetic advances. Pharmacotherapy 35(1), 11–16. Peñalva, G., Crespo-Robledo, P., Molvik, M., López-Navas, A., Kacelnik, O., Cisneros, J.M. and EU-JAMRAI WP7.4.1 Group. 2022. A step forward in antibiotic use and resistance monitoring: a quarterly surveillance system pilot in 11 European Union/European Economic Area countries, September 2017 to May 2020. Euro. Surveill. 27(46), 2200082. Press Release. 2021. Minister for Agriculture in the Department of Agriculture, Food and the Marine. Government of Ireland. Available via gov.ie/en/press-release/1480e-minister-mcconalogue-welcomes-the-announcement-of-a-statement-of-intent-around-the-voluntary-cessation-of-use-of-colistin-in-the-animal-health-sector-in-ireland/ Raasch, S., Collineau, L., Postma, M., Backhans, A., Sjölund, M., Belloc, C., Emanuelson, U., Beilage, E.G., Stärk, K., Dewulf, J. and on the behalf of the MINAPIG Consortium. 2020. Effectiveness of alternative measures to reduce antimicrobial usage in pig production in four European countries. Porcine Health Manag. 6, 6. Regulations Amending the Food and Drug Regulations (Veterinary Drugs — Antimicrobial Resistance). 2017. Food and Drugs Act. Vol. 151, No. 10: SOR/2017-76 (Canada gazette – Regulations amending the food and drug regulations (Veterinary Drugs — Antimicrobial Resistance). Rhouma, M., Madec, J.Y. and Laxminarayan, R. 2023. Colistin: from the shadows to a One Health approach for addressing antimicrobial resistance. Int. J. Antimicrob. Agents 61(2), 106713. Ribeiro, S., Mourão, J., Novais, Â., Campos, J., Peixe, L. and Antunes, P. 2021. From farm to fork: colistin voluntary withdrawal in Portuguese farms reflected in decreasing occurrence of mcr-1-carrying Enterobacteriaceae from chicken meat. Environ. Microbiol. 23, 7563–7577. Sammul, M., Mõtus, K. and Kalmus, P. 2021. The use of colistin in food-producing animals in Estonia-vaccination as an effective alternative to consumption of critically important antimicrobials in pigs. Antibiotics 10(5), 499. Saudi Food and Drug Authority. 2024. Veterinary antimicrobial products classifications. Version 0.1. Available via https://www.sfda.gov.sa/sites/default/files/2024-11/VeterinaryAntimicrobialProductsClassifications.pdf Sharma, J., Sharma, D., Singh, A. and Sunita, K. 2022. Colistin resistance and management of drug resistant infections. Can. J. Infect. Dis. Med. Microbiol. 2022, 4315030. Shen, C., Zhong, L.L., Yang, Y., Doi, Y., Paterson, D.L., Stoesser, N., Ma, F., El-Sayed, M.A., Feng, S., Huang, S. and Li, H.Y. 2020. Dynamics of mcr-1 prevalence and mcr-1-positive Escherichia coli after the cessation of colistin use as a feed additive for animals in China: a prospective cross-sectional and whole genome sequencing-based molecular epidemiological study. Lancet Microbe. 1(1), e34–e43. Thabet, A.M., Alzuheir, I.M., Laham, N.A.A., Helal, B.Y.A., Fayyad, A.F., Jalboush, N.H. and Gharaibeh, M.H. 2023. First report of mobile colistin resistance gene mcr-1 in avian pathogenic Escherichia coli isolated from turkeys in the Gaza Strip, Palestine. Vet. World 16(6), 1260–1265. The Ministry of Climate Change and Environment. 2024. United Arab Emirates. Administrative Decision No. 6 of 2024 regarding the prohibition of importing, trading and registering the active ingredient colistin in the veterinary sector. Available via https://www.moccae.gov.ae/ar/legislations.aspx#page=1 The National Service for Agricultural Health and Quality’s (SENASA). 2019. Resolution 22/2019. Available via https://www.boletinoficial.gob.ar/detalleAviso/primera/200151/20190115 (Accessed 24 February 2021). Tokuda, M. and Shintani, M. 2024. Microbial evolution through horizontal gene transfer by mobile genetic elements. Microb. Biotechnol. 17(1), e14408. UK-VARSS. 2022. Veterinary antibiotic resistance and sales surveillance report (UK-VARSS 2021). Addlestone, UK: Veterinary Medicines Directorate. Wang, Y., Xu, C., Zhang, R., Chen, Y., Shen, Y., Hu, F., Liu, D., Lu, J., Guo, Y., Xia, X. and Jiang. 2020. Changes in colistin resistance and mcr-1 abundance in Escherichia coli of animal and human origins following the ban of colistin-positive additives in China: an epidemiological comparative study. Lancet Infect. Dis. 20(10), 1161–1171. | ||
| How to Cite this Article |
| Pubmed Style Ahmed MO, Abouzeed YM, Daw MA. Global initiatives to phase-out colistin use in food-producing animals. Open Vet. J.. 2025; 15(2): 533-540. doi:10.5455/OVJ.2025.v15.i2.4 Web Style Ahmed MO, Abouzeed YM, Daw MA. Global initiatives to phase-out colistin use in food-producing animals. https://www.openveterinaryjournal.com/?mno=227387 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.4 AMA (American Medical Association) Style Ahmed MO, Abouzeed YM, Daw MA. Global initiatives to phase-out colistin use in food-producing animals. Open Vet. J.. 2025; 15(2): 533-540. doi:10.5455/OVJ.2025.v15.i2.4 Vancouver/ICMJE Style Ahmed MO, Abouzeed YM, Daw MA. Global initiatives to phase-out colistin use in food-producing animals. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 533-540. doi:10.5455/OVJ.2025.v15.i2.4 Harvard Style Ahmed, M. O., Abouzeed, . Y. M. & Daw, . M. A. (2025) Global initiatives to phase-out colistin use in food-producing animals. Open Vet. J., 15 (2), 533-540. doi:10.5455/OVJ.2025.v15.i2.4 Turabian Style Ahmed, Mohamed Omar, Yousef Mohamed Abouzeed, and Mohamed Ali Daw. 2025. Global initiatives to phase-out colistin use in food-producing animals. Open Veterinary Journal, 15 (2), 533-540. doi:10.5455/OVJ.2025.v15.i2.4 Chicago Style Ahmed, Mohamed Omar, Yousef Mohamed Abouzeed, and Mohamed Ali Daw. "Global initiatives to phase-out colistin use in food-producing animals." Open Veterinary Journal 15 (2025), 533-540. doi:10.5455/OVJ.2025.v15.i2.4 MLA (The Modern Language Association) Style Ahmed, Mohamed Omar, Yousef Mohamed Abouzeed, and Mohamed Ali Daw. "Global initiatives to phase-out colistin use in food-producing animals." Open Veterinary Journal 15.2 (2025), 533-540. Print. doi:10.5455/OVJ.2025.v15.i2.4 APA (American Psychological Association) Style Ahmed, M. O., Abouzeed, . Y. M. & Daw, . M. A. (2025) Global initiatives to phase-out colistin use in food-producing animals. Open Veterinary Journal, 15 (2), 533-540. doi:10.5455/OVJ.2025.v15.i2.4 |