| Research Article | ||
Open Vet. J.. 2025; 15(2): 847-862 Open Veterinary Journal, (2025), Vol. 15(2): 847-862 Research Article The role of veterinary drug use in driving antimicrobial resistance of Staphylococcus aureus isolates in smallholder swine farms in Central VietnamNguyen Van Chao1*, Ho Thi Dung1, Vu Thi Thanh Tam2, Phan Thi Hang1 and Bui Thi Hien11Faculty of Animal Sciences and Veterinary Medicine, University of Agriculture and Forestry, Hue University, Hue city, Vietnam 2Mientrung Institute for Scientific Research, Vietnam National Museum of Nature, Vietnam Academy of Science and Technology, Hue, Vietnam *Corresponding Author: Nguyen Van Chao. Faculty of Animal Science and Veterinary Medicine, University of Agriculture and Forestry, Hue University, Hue City, Vietnam. Email: nvchao [at] hueuni.edu.vn Submitted: 05/11/2024 Accepted: 21/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
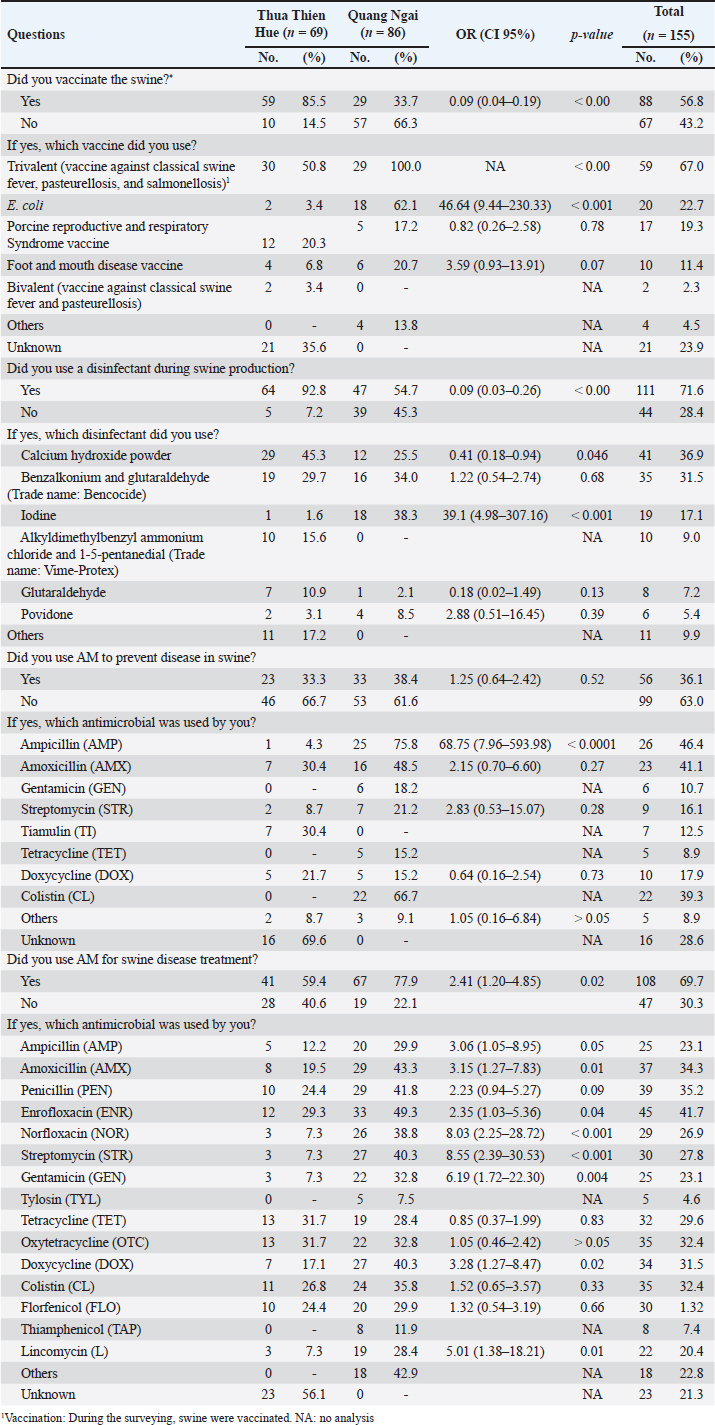
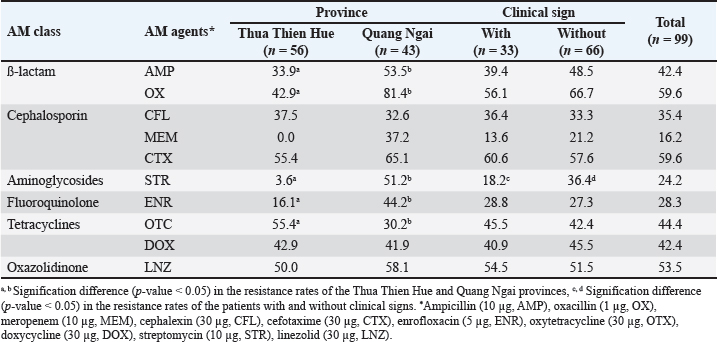
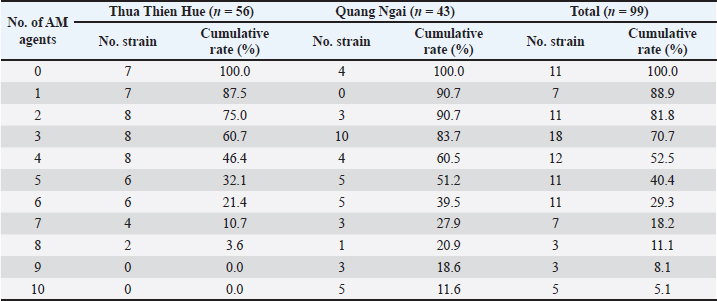
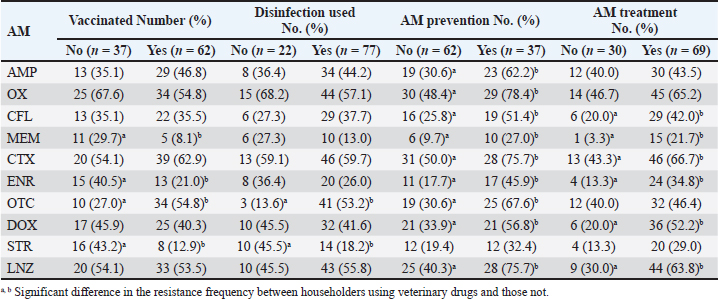
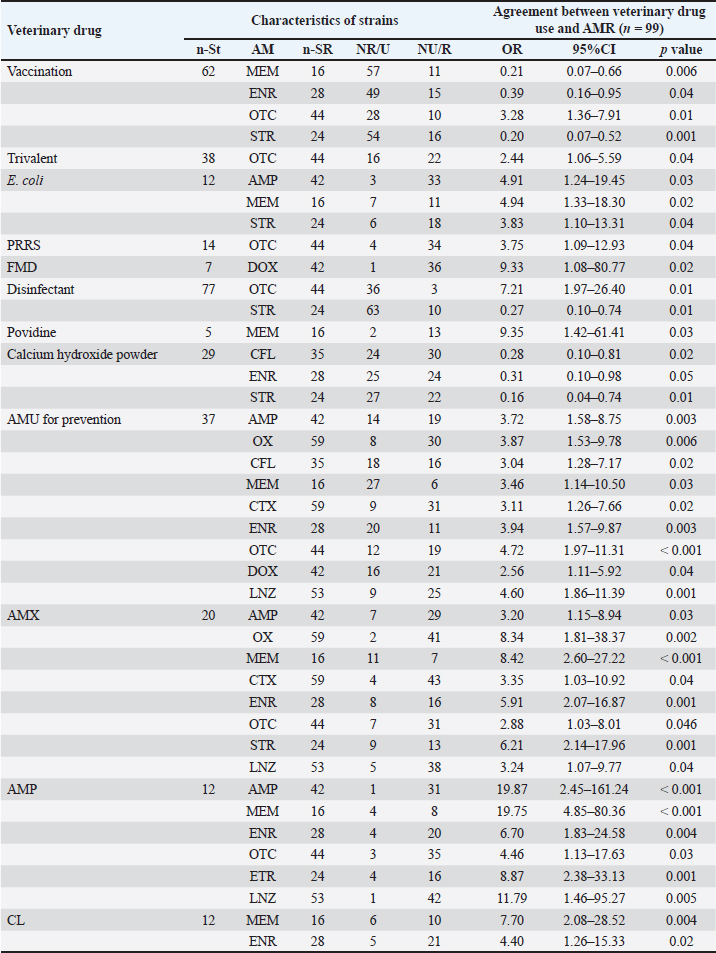
AbstractBackground: Staphylococcus aureus is a well-known opportunistic pathogen widely present in humans and food-producing animals. The emergence of antimicrobial resistance (AMR) in S. aureus represents a major challenge to animal and public health. Poor biosecurity practices and the misuse and overuse of veterinary drugs in farming settings may apply environmental pressure, which favors the selection of AMR bacteria. Aim: This study aimed to describe veterinary drug usage (VDU), prevalence of AMR phenotypes, and associations among S. aureus isolates from swine of smallholder farms in Central Vietnam. Methods: A cross-sectional survey was conducted to collect VDU data from smallholder swine farms. A total of 155 nasal swab samples were collected and used for isolating S. aureus. The AMR of S. aureus strains was tested using the disk diffusion method. Results: Approximately 56.8%, 71.6%, 36.1%, and 69.7% of farmers used vaccines, disinfectants, and antimicrobials (AMs) for prevention and treatment, respectively. Of the 155 nasal swab samples, 99 (63.9%) were positive for S. aureus. Resistance was most commonly observed against oxacillin (59.6%), cefotaxime (59.6%), and linezolid (53.5%). Positive associations were found between the use of vaccines and resistance to oxytetracycline (OR=3.28, p=0.01) and povidone usage and resistance to meropenem (OR=9.35, p=0.03). Almost all positive associations were observed between the use of AMs (for both prevention and treatment) and AMR in S. aureus. Negative associations were found between resistance to oxytetracycline and the use of gentamicin, linezolid, streptomycin, and norfloxacin. Conclusion: The present study highlights information on VDU, prevalence, AMR, and their associations with S. aureus isolated from a smallholder swine farm in Central Vietnam. These findings are expected to aid in developing countermeasures against AMR against swine production in Vietnam. Keywords: Antimicrobial, Disinfectant, Resistance, Staphylococcus aureus, Swine, Vaccine. IntroductionStaphylococcus aureus is a leading cause of infectious diseases in humans and animals. This Gram-positive, non-spore-forming, non-motile, catalase-positive bacterium is commonly found on the skin, hair, nose, and respiratory tract of humans and animals (Kluytmans et al., 1997; Graber et al., 2013). Among staphylococci, S. aureus is the most invasive species and an etiological agent of diverse human and animal maladies, including skin infections, abscesses, food poisoning, toxic shock syndrome, septicemia, and pneumonia (DeLeo and Chambers, 2009; Woodford and Livermore, 2009). Additionally, S. aureus is one of the most prominent causes of hospital-, community-, and livestock-acquired bacterial infections worldwide (Song et al., 2011; Tong et al., 2015), which underscores the importance of determining the Antimicrobial resistance (AMR) of strains from humans and animals. AMR is a serious and growing threat to global health. Evidence indicates that antimicrobial use (AMU) in livestock is a major driver of increased AMR and multidrug resistance (MDR) (Magouras et al., 2017; Collignon & Beggs, 2019). In many countries, AMs are extensively used to promote growth and prevent or treat infections in livestock (Magouras et al., 2017; Collignon and Beggs, 2019), particularly in low- and middle-income countries (Lam et al., 2019). This practice carries potential risks to human health that must be addressed (Van Boeckel et al., 2017). In the swine industry, AMU has promoted the resistance of both commensal and pathogenic bacteria (Holmer et al., 2019; Zhang et al., 2019). On farms, AMU leads to an increase in AMR bacteria, which can be transmitted directly to humans through contact or meat consumption or indirectly through environmental pathways (Silbergeld et al., 2008; Wee et al., 2020). More than four decades ago, Swann et al. (1969) discussed the association between AMU in livestock and the emergence of AMR in animals. Since then, many studies have described the association between AMU and AMR (Asai et al., 2005; Chantziaras et al., 2014; Jibril et al., 2021). In Vietnam, high levels of AMR in foodborne pathogens isolated from swine, poultry, cattle, and meat, such as Salmonella spp., Campylobacter spp., E. coli, and S. aureus, have been reported (Vo et al., 2012; Nguyen et al., 2016a; Nghiem et al., 2017; Huynh and Ly, 2018; Vu et al., 2020; Tuat et al., 2021). Farmers can buy and use AMs to prevent and treat infections that are available over the counter without a prescription. Some studies have reported the overuse of AMs in swine and poultry farming, although the quantities of AMs used are unknown (Pham et al., 2013; Nguyen et al., 2016b; Nhung et al., 2016). However, these studies did not evaluate the association between AMU and AMR, and the validity of the AMU data was not confirmed by direct observations on farms. Vietnam has the fifth-highest swine production in the world. Swine production is an important economic activity in Vietnam, with most swine farmers being smallholders in rural areas. AMU in swine production is a risk factor for increased AMR. However, there is limited information on the use of AMs, vaccines, disinfectants, and AMR profiles and their association with the abundance of S. aureus. This study describes the use of AMs, vaccines, and disinfectants in smallholder swine farms in Quang Ngai and Thua Thien Hue provinces in Central Vietnam. The prevalence and AMR profiles of S. aureus strains in swine from the farms were analyzed. The associations between AMU, vaccines, disinfectants, and AMR in the isolated S. aureus strains were also investigated. These results provide a first glimpse into the use of AMs, vaccines, and disinfectants in smallholder swine farms in Central Vietnam, as well as the prevalence and epidemiology of AMR in S. aureus. This information will be useful for the clinical control of infectious diseases caused by S. aureus and for the development of policies and clinical practice guidelines to reduce AMR in swine production. Materials and MethodsExperiment and information collectionA cross-sectional survey of the use of AMs, vaccines, and disinfectants in smallholder swine farms (scale 2–20 swine) was conducted in two provinces in Central Vietnam from February 2022 to September 2023. A total of 155 smallholder swine farmers were randomly selected from Thua Thien Hue (HUE, n=69) and Quang Ngai (QN, n=86) provinces. Questionnaires were compiled after a test survey and subsequent adjustment and were used for direct interviews with farmers, animal health workers, and veterinarians. Information on veterinary drug usage, antimicrobial components, and active ingredients available on the farm was tracked and collected through labels found on remedy packs or jars left around animal housing or at a local veterinary pharmacy. To ensure the confidentiality of all remedy use information, the names and addresses of all householders were kept secure by encoding the addresses at the time of the survey. Sample collection and S. aureus identificationIn the cross-sectional survey carried out in parallel with the current study, 155 nasal swab samples were collected from swine (anorexia, fever, dermatitis, dyspnea, pneumonia, arthritis) or without clinical signs on the same swine farms (these farms were surveyed). Each sample was placed on Baird-Parker agar (Himedia Laboratories, Mumbai, India) supplemented with 5% (v/v) egg yolk tellurite emulsion and 6.5% (w/v) NaCl and incubated at 37°C for 18–24 hours. Staphylococcus aureus grows as black, shiny colonies with fine white rims surrounded by a clear zone. The colonies identified as S. aureus were confirmed by a positive coagulase result (Lin et al., 2009). In addition, each strain was confirmed to be S. aureus by polymerase chain reaction testing for the presence of the S. aureus-specific nuc gene (Brakstad et al., 1992). Antimicrobial susceptibility testingSusceptibility testing was performed on the isolated S. aureus strains (one strain per sample) using the disk diffusion method. The test was performed in accordance with the Clinical and Laboratory Standards Institute (CLSI, 2020) protocol. The common AMs used to prevent and treat infectious disease in livestock were chosen for the antimicrobial susceptibility tests, including ampicillin (10 µg, AMP), oxacillin (1 µg, OX), meropenem (10 µg, MEM), cephalexin (30 µg, CFL), cefotaxime (30 µg, CTX), enrofloxacin (5 µg, ENR), oxytetracycline (30 µg, OTX), doxycycline (30 µg, DOX), streptomycin (10 µg, STR), and linezolid (30 µg, LNZ) (Oxoid Ltd., Hampshire, UK). The inoculum was prepared from cultures in Brain Heart Infusion (BHI) broth incubated for 4–6 hours before adjusting the turbidity to 0.5 on the McFarland scale (DensiCHEKTM Plus, ALT, San Diego, USA). One hundred microliters of the bacterial suspension was spread on Mueller–Hinton agar (MHA, Merck KGaA, Darmstadt, Germany). The appropriate antimicrobial-impregnated disks were placed on the agar surface, and the plates were incubated at 37°C for 24 hours. Thereafter, the zones of inhibition were measured, and the antimicrobial susceptibility of the strains was determined using interpretative standards for Staphylococcus species (Eurl, 2018; CLSI, 2020). Strains with phenotypic resistance to two or more antimicrobial agents were defined as multiple-AMR strains. Data analysisThe data were entered into a Microsoft Excel 2016 (Microsoft Corporation, Redmond, WA, USA) spreadsheet for descriptive statistical analysis and then exported to IBM SPSS statistic 18.0 (IBM, Armonk, NY, USA) for inferential analysis. The one-way analysis of variance and Student’s unpaired t test were used to test for significance between the prevalence of S. aureus, AMR, and categorical variables (location, with or without clinical signs; use and non-use veterinary drugs). The associations between AM, vaccine, and disinfectant use and the AMR phenotype were calculated using chi-square and Fisher’s exact tests. Statistically significant associations are shown as ORs with 95% CIs. An association was considered significant when the p-value was less than 0.05. ResultsIdentification vaccination, disinfectant, and antimicrobial use on farmsThe results of veterinary drug use for swine health management are presented in Table 1. Approximately 56.8% (88/155), 71.6% (111/155), 36.1% (56/155), and 69.7% (108/155) of the farmers reported using vaccines, disinfectants, AMs for prevention and AMs for treatment, respectively. The vaccination program aimed to control six common diseases in swine, including classical swine fever, pasteurellosis, salmonellosis, E. coli, porcine reproductive and respiratory syndrome (PRRS), and FMD. The most commonly used vaccines were trivalent (67.0%), E. coli (22.7%), and PRRS (19.3%). The rates of farms in Quang Ngai (QN) using trivalent (29/29, 100%) and E. coli (18/29, 62.1%) vaccines were significantly higher than those in Thua Thien Hue (HUE) (p < 0.05). Differences in the rates of farms using disinfectants: calcium hydroxide powder, Bencocide, iodine, Povidine, glutaraldehyde, and Vime-Protex) were observed. The usage rate of calcium hydroxide powder (36.9%, 41/111) was the highest, followed by iodine (31.5%, 35/111) and Povidine (17.7%, 19/111). A significant difference (p < 0.05) in the usage rates of calcium hydroxide powder and iodine between QN and HUE was observed (Table 1). The AM use for prevention was reported in 36.1% of the farms, with the most common AMs being AMP (46.4%), followed by AMX (41.1%), CL (39.3%), and DOX (17.9%). Moreover, the AM use for treatment occurred in 69.7% (108/155) of the farms. Of the 18 AMs used across the six classes, ENR (41.7%) was the most common AM used for treatment, followed by PEN (35.2%), AMX (34.3%), OTC, and CL (32.4% each), and DOX (31.5%). Prevalence of S. aureus isolates from swineA total of 155 swine nasal swab samples were collected, of which 99 (63.9%) tested positive for Staphylococcus aureus (Fig. 1). The results revealed that the isolation rate of S. aureus varied significantly by region and presence of clinical signs (p 0.05). The prevalence of S. aureus in samples taken from HUE (56/69, 81.2%) than in QN (43/86, 50.0%) ( p < 0.0001). The prevalence of S. aureus was higher in samples with clinical signs (66/75, 88.0%) than in those without clinical signs (33/80, 41.3%) (p < 0.001). Antimicrobial resistanceOf the 99 S. aureus strains isolated from 155 samples, 56 were from HUE and 43 were from QN. Among all strains tested, resistance levels were as follows: OX and CTX (59.6% each), LNZ (53.5%), OTC (44.4%), AMP and DOX (42.4% each), CFL (35.4%), ENR (28.3%), STR (24.2%), and MEM (16.2%). The strains from QN showed a higher prevalence of resistance to OX (81.4% vs. 42.9%), AMP (53.5% vs. 33.9%), ENR (44.2% vs. 16.1%), and STR (51.2% vs. 3.6%) than the strains from HUE (p < 0.05) (Table 2). Furthermore, strains from swine without clinical signs showed significantly higher resistance to STR (36.4% vs. 18.2%) than those from swine with clinical signs. MDR was observed in 81.8% of the strains; notably, five strains from QN (two strains from swine with and three strains from swine without clinical signs) were resistant to 10 AMs (Table 3). Effects of veterinary drug use on antimicrobial resistance in S. aureus strainsThe effects of veterinary drug use on AMR in S. aureus are presented in Table 4. There was a higher prevalence of resistance to MEM (29.7% vs. 8.1%), ENR (40.5% vs. 21.0%), and STR (43.2% vs. 12.9%) in strains from farms that did not use vaccines (p < 0.05). Strains from farms that used disinfectants showed a higher prevalence of resistance to OTC (53.2% vs. 13.6%) and a lower prevalence of resistance to STR (18.2% vs. 45.5%) (p < 0.05). Furthermore, strains from farms where AMs were used preventatively showed a higher prevalence of resistance to all AMs except STR (p < 0.05) than strains from farms where AMs were not used. Staphylococcus aureus strains from farms that used AMs for treatment had a higher prevalence of resistance to CFL (42.0% vs. 20.0%), MEM (21.7% vs. 3.3%), CTX (66.7% vs. 43.3%), ENR (34.8% vs. 13.3%), DOX (52.2% vs. 20.0%), and LNZ (63.8% vs. 30.0%) than those from farms where AMs were not used (p < 0.05). The statistical analyses revealed significant associations between the use of veterinary drugs on swine farms and the prevalence of AMR in S. aureus (Table 5). A significant positive association was found between vaccination in swine farms and resistance to OTC (OR=3.28, p=0.01) in S. aureus. Specifically, the use of trivalent, E. coli, PRRS, and FMD vaccines was strongly associated with resistance to OTC (OR=2.44, p=0.04); AMP (OR=4.91, p=0.03), MEM (OR=4.94, p=0.02), and STR (OR=3.83, p=0.04); OTC (OR=3.75, p=0.04); and DOX (OR=9.33, p=0.02), respectively. For disinfectant use, Povidine had a significant positive association with MEM resistance (OR=9.35, p=0.03). In contrast, the use of calcium hydroxide powder was significantly negatively associated with resistance to CFL (OR=0.28, p=0.02), ENR (OR=0.31, p=0.05), and STR (OR=0.16, p=0.01). Regarding the use of AMs for prevention, significant positive associations were found with resistance to AMP (OR=3.72, p=0.003), OX (OR=3.87, p=0.0006), CFL (OR=3.04, p=0.02), MEM (OR=3.46, p=0.03), CTX (OR=3.11, p=0.02), ENR (OR=3.94, p=0.003), OTC (OR=4.72, p < 0.0001), DOX (OR=2.56, p=0.04), and LNZ (OR=4.60, p=0.001). Significant positive associations were also observed between the use of AMX, AMP, CL, and STR for the prevention and resistance to several AMs (e.g., AMX and AMP; AMP and AMP; CL and ENR; STR and CTX). The overall use of AMs for treatment was significantly positively associated with resistance to CFL, MEM, CTX, ENR, DOX, and LNZ in S. aureus strains. Most associations between AM use for treatment and AMR in S. aureus were positive, except for the associations between resistance to OTC and the use of GEN, LNZ, STR, and NOR, which were negative. Table 1. Use of veterinary drugs in swine farms.
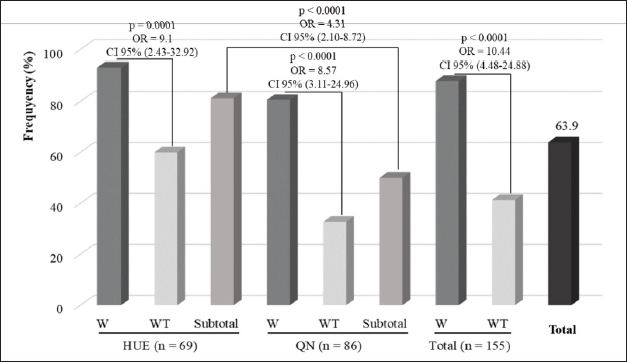
DiscussionIn the context of both reducing AMR and preventing the spread of epidemic diseases, prioritizing preventive veterinary management strategies is crucial for the sustainability of animal health (Renault et al., 2021). Previous studies indicate that improving biosecurity on livestock farms is particularly vital in combating AMR, as it can help reduce AMU and curb the dissemination and persistence of resistant microbes within farms (Davies and Wales, 2019; Kruse et al., 2020; Dhaka et al., 2023). Among the various parameters of biosecurity, the role of cleaning, disinfection, and vaccination measures has been highlighted in various studies (Martelli et al., 2017; De Lorenzi et al., 2020; Kruse et al., 2020; Dhaka et al., 2023). In Vietnam, swine farm biosecurity measures vary because of non-standardized procedures or regulations (Duong et al., 2019; Ngo et al., 2020). Moreover, data on the level of implementation of biosecurity measures on small swine farms in Vietnam are limited (Auplish et al., 2024). In the present study, the prevalence of farms using vaccines (56.8%) and disinfectants (71.6%) was higher than that of AMU for prevention (36.1%). Swine production has been facing challenges in recent years, including high animal feed prices and unpredictable epidemics, such as ASF, PRRS, and FMD, causing significant losses to small farms (Zhang & Kono, 2012; Le et al., 2019; Nguyen et al., 2021). Therefore, household livestock farms have applied various biosecurity measures to limit the impact of epidemics (Ngo et al., 2020). Moreover, some vaccines, such as those for salmonellosis, swine fever, and pasteurellosis, are included in the mandatory vaccination program according to veterinary law (National Assembly of the Socialist Republic of Vietnam, 2018). The rate of households using AMs to prevent diseases was lower than that reported in some previous studies (Pham et al., 2013; Duong and Nguyen, 2015; Nhung et al., 2016). Since 2020, the veterinary law has introduced a list of prohibited AMs added to feed for disease prevention (National Assembly of the Socialist Republic of Vietnam, 2018). Among AMUs (in both prevention and treatment), AMP, AMX, PEN, CL, GEN, and ENR were commonly used by the farmers, as reported in other studies (Pham et al., 2013; Duong and Nguyen, 2015; Nhung et al., 2016). These include AMs considered critically important to human health, such as PEN, CL, and GEN (Collignon et al., 2016). The B-lactams were the AMs of choice because they are long-acting, cheap, and have a broad spectrum (Bush and Bradford, 2016). Overreliance on these AMs can promote the development of AMR bacteria, potentially limiting their future effectiveness, especially when the drugs are underdosed in both prevention and treatment. This study observed a high prevalence of farms using CL for both prevention (39.3%) and treatment (32.4%). Previous studies have reported that CL is a popular veterinary drug used not only to treat infections but also as a growth promoter and protective agent (Duong and Nguyen, 2015; Rhouma et al., 2016; Luu et al., 2021). However, CL is considered a last-resort drug for treating drug-resistant bacterial infections (Wand et al., 2017). Fig. 1. Prevalence of S. aureus isolates from swine in Thua Thien Hue and Quang Ngai Provinces, Vietnam.
Table 2. Prevalence of antimicrobial resistance to S. aureus strains.
Table 3. Prevalence of antimicrobial resistance to S. aureus strains.
Table 4. Effects of veterinary drug usage on antimicrobial resistance in S. aureus strains (n=99).
The present study found an estimated prevalence of S. aureus of 63.9% in swine farms. Studies conducted by Linhares et al. (2015) and Sun et al. (2015) reported a higher prevalence of S. aureus isolated from swine farms (77.0% and 91.1%, respectively) than that reported in the present study. The prevalence of S. aureus strains similar to those found in this study (63.6% and 68.6%) (Nobre et al., 2021; Sineke et al., 2021) or even lower (36.2%) (Zehra et al., 2017). Staphylococcus aureus can be endemic in swine populations, and its relative prevalence varies geographically and probably temporally (Espinosa-Gongora et al., 2014). A study in Bac Ninh province, North Vietnam, found a lower prevalence of S. aureus strains in nasal swab samples of swine (13/80, 16.25%) (Vu et al., 2020) than observed in this study. In the present study, a significant difference in the prevalence of S. aureus strains was observed between QN (43/86, 50.0%) and HUE (56/69, 81.2%), with a p-value 0.05. The emergence of AMR in S. aureus represents a major challenge to animal and public health. Poor biosecurity practices and the use, misuse, and overuse of veterinary drugs in farming settings may apply environmental pressure, which favors the selection of AMR bacteria (Vestergaard et al., 2019; Urban-Chmiel et al., 2022). S. aureus is the most commonly identified AMR pathogen worldwide (Foster, 2017). It is resistant to almost all β-lactams and other major AM classes, such as fluoroquinolones (Monaco et al., 2017; Kumar et al., 2020). In this study, high resistance rates were obtained for OX, CTX (each 59.6%), and LNZ (53.5%), corroborating the findings of other studies (Nguyen et al., 2014; Guo et al., 2018; Lekagul et al., 2019; Vu et al., 2020). These AMs are frequently used in swine farming to treat diseases, and the capacity of S. aureus to acquire resistance to these drugs has already been demonstrated (Duong and Nguyen, 2015; Nhung et al., 2016; Luu et al., 2021). In addition, high frequencies of strains resistant to OTC (44.4%), AMP, DOX (each 42.4%), and CFL (35.4%) were also observed, corroborating the results of other studies (Nhung et al., 2016; Vu et al., 2020; Sineke et al., 2021). These results are worrisome because these AMs belong to classes with the highest priority among those critically important for human medicine and should be used prudently in both humans and animals (Collignon et al., 2016). MDR S. aureus strains from swine may play an important role in spreading AMR strains and ARGs among animals, humans with direct animal contact, or even pork consumers, thereby impacting the treatment of possible infections. In this study, MDR, defined as the resistance to two or more AMs, was found in 81 (81.8%) S. aureus strains. This was observed mainly on farms that used a wide variety of AMs for purposes other than treatment. Many strains showed resistance to up to 10 AMs. As in other reports, AMs are commonly used on swine farms, with usage frequency varying according to the geographic region (Foster, 2017; Abreu et al., 2019; Adesoji et al., 2019). Most of the farms where MDR S. aureus was isolated carried out disinfection of pens, used at least three AMs, and used AMs for purposes other than treatment. The broader use of AMs is expected to be associated with greater MDR colonization in swine. The use of disinfectants and downtime are biosecurity practices adopted to reduce the introduction and dissemination of infectious agents among animals. Nevertheless, disinfectant resistance may contribute to the selection of MDR bacteria, especially when the responsible genes are co-located with ARGs in mobile genetic elements (Davies & Wales, 2019; Maertens et al., 2019; Bischofberger et al., 2020). The high MDR rate highlights the need for AM stewardship to ensure prudent AMU in animal production, as it may have serious consequences for human and environmental health. Vaccination is a highly effective and valuable tool for fighting AMR. However, the present study found a significantly lower rate of S. aureus strain resistance to MEM (8.1%), ENR (21.0%), and STR (12.9%) in the vaccinated group compared with the non-vaccinated group (29.7%, 40.5%, and 43.2%, respectively). Moreover, a negative association between vaccination and AMR in S. aureus strains [resistance to MEM (OR=0.21; p=0.006), ENR (OR=0.39, p=0.04), and STR (OR=0.2, p=0.001)] was observed (Table 4). The association between vaccination and AMU/AMR has been reported (Bak and Rathkjen, 2009; Kruse et al., 2016; Temtem et al., 2016; Peiponen et al., 2018; Buchy et al., 2020; Costanzo and Roviello, 2023). Bak and Rathkjen (2009) found significantly lower AMU in the group vaccinated against Lawsonia intracellularis (LAW) than in the non-vaccinated group. However, Peiponen et al. (2018) found only a slightly, but not statistically significant, lower AMU in the group vaccinated against LAW. The associations between vaccination and AMU have also been studied at the population level, with varying results. A cross-sectional study by Temtem et al. (2016) found that herds vaccinated against Porcine Circovirus Type 2 (PCV2), Mycoplasma hyopneumoniae (MYC), and/or LAW had significantly higher AMU in weaners than herds not vaccinated against any of these infections. Similarly, Postma et al. (2016) found that the use of vaccines against higher numbers of pathogens was associated with a higher AMU in swine from birth to slaughter. Kruse et al. (2016) reported that initiating vaccination against PCV2, MYC, Actinobacillus pleuropneumoniae, PRRS, and LAW had no effect on the change in AMU in weaners and finishers. Obolski et al. (2018) found that vaccination can result in a rapid increase in the frequency of preexisting resistant variants of non-vaccine serotypes due to the removal of competition from vaccine serotypes. Similarly, the results of this study show that among the vaccines used (Table 1), none is used to prevent diseases related to S. aureus infection. Finally, vaccination is a potential weapon against AMR. However, while AMU remains at high levels, we need to understand and act to avoid increasing AMR in non-vaccine serotypes, as outlined by this study and others. Recent studies have evaluated the association between AMR and decreased susceptibility to disinfectants in bacteria isolated from livestock and the environment (Herruzo et al., 2015; Wieland et al., 2017; Basiry et al., 2022). In the present study, the positive association between disinfectant use and resistance to OTC (OR=7.21, p=0.01), as well as between the use of Povidine and resistance to MEM (OR=9.35, p=0.03), was also investigated. Previous studies have discussed cross-resistance mechanisms between AMs and disinfectants, (de Carvalho et al., 2020; Pereira et al., 2021), mostly related to general resistance responses, changes in membrane permeability (Pereira et al., 2021), efflux pumps (Nguyen et al., 2015; Blanco et al., 2016), and the structural effects of biofilms. In this regard, it is not surprising that these mechanisms cause cross-resistance because they all decrease the accessibility of disinfectants and AMs to bacterial cells. These results indicate that the cross-resistance between disinfectant resistance and AMR mechanisms is the result of general microbial adaptation to a hostile environment. Although many countries have banned the use of AMs in livestock farming (Official Journal of the European Union, 2006; FDA, 2015; The Ministry of Agriculture and Rural Development, 2020), this policy will likely increase the use of disinfectants to ensure biosecurity. However, the use of disinfectants also risks increasing resistance to disinfectants and AMR, as discussed in this study and in previous studies. Table 5. Association between vaccination, disinfectant use, and antimicrobial resistance in S. aureus.
Antimicrobial use is generally accepted as the main driver of AMR, and the relationship between AMU and AMR in animals is, therefore, a topic of considerable interest (Holmer et al., 2019; Jibril et al., 2021; Sali et al., 2021). Our investigation revealed significant associations between AMU (in both prevention and treatment) and the presence of certain AMR phenotypes. The AMR phenotype is common among E. coli indicators isolated from swine in the United States (Spronk et al., 2023) and European countries (EFSA, 2021). Similar to the findings of Kobayashi et al. (2023), the results of this study showed that AMU was associated with resistance not only to AMs belonging to the same AM class (AMX and AMP; GEN and STR) but also to AMs from other classes (AMX and ENR; FLO and DOX). Resistance genes occurring in the same mobile genetic elements and gene cassettes (Birkegård et al., 2017; Partridge et al., 2018) may explain the observed AMR phenotypes and should be addressed in more detail in future investigations. However, not all associations between AMU and AMR phenotypes in this study could be explained. For example, the use of LNZ, STR, and NOR (as treatment) was negatively associated with the presence of the OTC phenotype. In other studies, both corresponding and divergent associations between AMU and AMR, including direct and implicit resistance selection mechanisms, have been reported (Callens et al., 2015; Makita et al., 2016). Makita et al. (2016) suggested that these issues were due to natural, cross- or co-selection based on analyses of AMR in E. coli isolates from swine and AMU. ConclusionSmallholder swine farmers in the two provinces used several essential veterinary drugs (including disinfectants, vaccines, and AMs) on their farms for prevention and treatment. The S. aureus isolates from swine included those from healthy swine that threatened public and animal health. Resistance to some AMs in S. aureus isolates was associated with the use of vaccines, disinfectants, and corresponding AMs, implying that increasing the use of such AMs would increase resistance. These results should help establish countermeasures against AMR in S aureus in smallholder swine farms in Central Vietnam. The findings provide support only for analyses conducted at a single time point: on-farm veterinary drug usage is associated with on-farm AMR. Other complex multifactorial relationships may contribute to the selection of AMR bacteria, which we hope to investigate further in future studies. AcknowledgmentThe authors would like to thank Mr. Hoang Dinh Quoc, who helped us with sampling. We also thank the farmers in Quang Ngai and Hue provinces, who participated in the interviews, provided honest information, and allowed sampling from their pigs. Conflict of interestThe authors declare that they have no conflicts of interest regarding any financial, personal, or other relationships with individuals or organizations related to the material discussed in this manuscript. FundingThis work was supported by a Hue University-level research project (No. DHH2023-02-176. The authors are grateful to the president of Hue University for allowing them to conduct this study. Authors’ contributionsAll authors contributed to the study’s conception and design. Nguyen Van Chao developed the original hypotheses, designed the experiments, and collaborated in interpreting the results. Ho Thi Dung, Bui Thi Hien, and Vu Thi Thanh Tam collected the data for this study, conducted the statistical analyses, and collaborated in the interpretation of the results. Ho Thi Dung, Vu Thi Thanh Tam, Phan Thi Hang, and Bui Thi Hien collaborated in interpreting the results and finalized the manuscript. All authors have read and approved the finalized manuscript. Data availabilityData supporting the findings of this study are available from the corresponding author under the Project funding, upon reasonable request. ReferencesAbreu, R., Rodríguez-Álvarez, C., Lecuona, M., Castro, B., González, J.C., Aguirre-Jaime, A. and Arias, Á. 2019. Increased antimicrobial resistance of MRSA strains isolated from pigs in Spain between 2009 and 2018. Vet. Sci. 6(2), 38. Adesoji, A.T., Onuh, J.P., Bagu, J. and Itohan, S.A. 2019. Prevalence and antibiogram study of Staphylococcus aureus isolated from clinical and selected drinking water of Dutsin-Ma, Katsina state, Nigeria. Afr. Health Sci. 19(1), 1385–1392. Asai, T., Kojima, A., Harada, K., Ishihara, K., Takahashi, T. and Tamura, Y. 2005. Correlation between the usage volume of veterinary therapeutic antimicrobials and resistance in Escherichia coli isolated from the feces of food-producing animals in Japan. Jpn. J. Infect. Dis. 58(6), 369–372. Auplish, A., Vu, T.T.T., Pham Duc, P., Green, A., Tiwari, H., Housen, T., Stevenson, M.A. and Dhand, N. 2024. Capacity and needs assessment of veterinary services in Vietnam in biosecurity, biosafety and One Health. PLoS One 19(1), e0295898. Bak, H. and Rathkjen, P.H. 2009. Reduced use of antimicrobials after vaccination of pigs against porcine proliferative enteropathy in a Danish SPF herd. Acta Vet. Scand. 51(1), 1. Basiry, D., Entezari Heravi, N., Uluseker, C., Kaster, K.M., Kommedal, R. and Pala-Ozkok, I. 2022. The effect of disinfectants and antiseptics on co- and cross-selection of resistance to antibiotics in aquatic environments and wastewater treatment plants. Front Microbiol 13(1), 1. Birkegård, A.C., Halasa, T., Græsbøll, K., Clasen, J., Folkesson, A. and Toft, N. 2017. Association between selected antimicrobial resistance genes and antimicrobial exposure in Danish pig farms. Sci. Rep. 7(1), 9683. Bischofberger, A.M., Baumgartner, M., Pfrunder-Cardozo, K.R., Allen, R.C. and Hall, A.R. 2020. Associations between sensitivity to antibiotics, disinfectants and heavy metals in natural, clinical and laboratory isolates of Escherichia coli. Environ. Microbiol. 22(7), 2664–2679. Blanco, P., Hernando-Amado, S., Reales-Calderon, J.A., Corona, F., Lira, F., Alcalde-Rico, M., Bernardini, A., Sanchez, M.B. and Martinez, J.L. 2016. Bacterial multidrug efflux pumps: much more than antibiotic resistance determinants. Microorganisms, 4(1), 14. Brakstad, O.G., Aasbakk, K. and Maeland, J.A. 1992. Detection of Staphylococcus aureus by polymerase chain reaction amplification of the nuc gene. J. Clin. Microbiol. 30(7), 1654–1660. Buchy, P., Ascioglu, S., Buisson, Y., Datta, S., Nissen, M., Tambyah, P.A. and Vong, S. 2020. Impact of vaccines on antimicrobial resistance. Int. J. Infect. Dis. 90(1), 188–196. Bush, K. and Bradford, P.A. 2016. β-Lactams and β-Lactamase inhibitors: an overview. Cold Spring Harb. Perspect. Med. 6(8), a025247. Callens, B., Faes, C., Maes, D., Catry, B., Boyen, F., Francoys, D., de Jong, E., Haesebrouck, F. and Dewulf, J. 2015. Presence of antimicrobial resistance and antimicrobial use in sows are risk factors for antimicrobial resistance in their offspring. Microb. Drug Resist. 21(1), 50–58. Chantziaras, I., Boyen, F., Callens, B. and Dewulf, J. 2014. Correlation between veterinary antimicrobial use and antimicrobial resistance in food-producing animals: a report on seven countries. J. Antimicrob. Chemother. 69(3), 827–834. CLSI, 2020. Performance standards for antimicrobial susceptibility testing. 30th ed. CLSI supplement M100S. Wayne, PA: CLSI. Collignon, P. and Beggs, J.J. 2019. Socioeconomic enablers for contagion: factors impelling the antimicrobial resistance epidemic. Antibiotics 8(3), 86. Collignon, P.C., Conly, J.M., Andremont, A., McEwen, S.A., Aidara-Kane, A., Agerso, Y., Andremont, A., Collignon, P., Conly, J., Dang Ninh, T., Donado-Godoy, P., Fedorka-Cray, P., Fernandez, H., Galas, M., Irwin, R., Karp, B., Matar, G., McDermott, P., McEwen, S., Mitema, E., Reid-Smith, R., Scott, H.M., Singh, R., DeWaal, C.S., Stelling, J., Toleman, M., Watanabe, H. and Woo, G.J. 2016. World Health Organization ranking of antimicrobials according to their importance in human medicine: a critical step for developing risk management strategies to control antimicrobial resistance from food animal production. Clin. Infect. Dis. 63(8), 1087–1093. Costanzo, V. and Roviello, G.N. 2023. The potential role of vaccines in preventing antimicrobial resistance (AMR): an update and future perspectives. Vaccines, 11(2), 333. Davies, R. and Wales, A. 2019. Antimicrobial resistance on farms: a review including biosecurity and the potential role of disinfectants in resistance selection. Compr. Rev. Food Sci. Food Saf. 18(3), 753–774. de Carvalho, C.C., Teixeira, R. and Fernandes, P. 2020. Mycobacterium vaccae adaptation to disinfectants and hand sanitisers, and evaluation of cross-tolerance with antimicrobials. Antibiotics, 9(9), 544. De Lorenzi, G., Borella, L., Alborali, G., Prodanov-Radulović, J., Štukelj, M. and Bellini, S. 2020. African swine fever: a review of cleaning and disinfection procedures in commercial pig holdings. Res. Vet. Sci. 132(1), 262–267. DeLeo, F.R. and Chambers, H.F. 2009. Reemergence of antibiotic-resistant Staphylococcus aureus in the genomics era. J. Clin. Investig. 119(9), 2464–2474. Dhaka, P., Chantziaras, I., Vijay, D., Bedi, J.S., Makovska, I., Biebaut, E. and Dewulf, J. 2023. Can improved farm biosecurity reduce the need for antimicrobials in food animals? a scoping review. Antibiotics, 12(5), 893. Duong, T.T., Brewer, T.D., Luck, J. and Zander, K.K. 2019. Understanding biosecurity threat perceptions across Vietnamese smallholder farmers in Australia. Crop Protect. 117, 147–155. Duong, T.T. and Nguyen, V.L. 2015. Survey on antibiotics for porker and broiler chicken in commercial farm in Bac Giang Province. J. Sci. Dev. 13(5), 717–722. EFSA. 2021. European Food Safety Authority. The European Union Summary Report on Antimicrobial Resistance in zoonotic and indicator bacteria from humans, animals and food in 2018/2019. EFSA J. 19(4), e8583. Espinosa-Gongora, C., Moodley, A., Lipinska, U., Broens, E.M., Hermans, K., Butaye, P., Devriese, L.A., Haesebrouck, F. and Guardabassi, L. 2014. Phenotypes and genotypes of old and contemporary porcine strains indicate a temporal change in the S. aureus population structure in pigs. PLoS One 9(7), e101988. Eurl, A. 2018. Isolation of methicillin-resistant Staphylococcus aureus (MRSA) from food producing animals and farm environment. DTU Natl. Food Inst. 1(1), 1–8. FDA. 2015. Veterinary feed directive, Fed Regist. 80, 31708–31735. Foster, T.J. 2017. Antibiotic resistance in Staphylococcus aureus. Current status and future prospects. FEMS Microbiol. Rev. 41(3), 430–449. Graber, H.U., Pfister, S., Burgener, P., Boss, R., Meylan, M. and Hummerjohann, J. 2013. Bovine Staphylococcus aureus: diagnostic properties of specific media. Res. Vet. Sci. 95(1), 38–44. Guo, D., Liu, Y., Han, C., Chen, Z. and Ye, X. 2018. Phenotypic and molecular characteristics of methicillin-resistant and methicillin-susceptible Staphylococcus aureus isolated from pigs: implication for livestock-association markers and vaccine strategies. Infect. Drug Resist. 11(1), 1299–1307. Herruzo, I., Herruzo, R. and Vizcaino, M. 2015. Is there a correlation between antibiotic resistance and decreased susceptibility to biocides in different genus of bacterial genera. J. Antibiotics Resist. 1(1), 1–7. Holmer, I., Salomonsen, C.M., Jorsal, S.E., Astrup, L.B., Jensen, V.F., Høg, B.B. and Pedersen, K. 2019. Antibiotic resistance in porcine pathogenic bacteria and relation to antibiotic usage. BMC Vet. Res. 15(1), 449. Huynh, T.T.A. and Ly, T.L.K. 2018. The prevalence and antibiotic resistance of Salmonella spp. isolated from pigs and farm environments in vinh long province. Can Tho Univ. J. Sci. 54, 26–33. Jibril, A.H., Okeke, I.N., Dalsgaard, A. and Olsen, J.E. 2021. Association between antimicrobial usage and resistance in Salmonella from poultry farms in Nigeria. BMC Vet. Res. 17(1), 234. Kluytmans, J., van Belkum, A. and Verbrugh, H. 1997. Nasal carriage of Staphylococcus aureus: epidemiology, underlying mechanisms, and associated risks. Clin. Microbiol. Rev. 10(3), 505–520. Kobayashi, S., Tamamura-Andoh, Y., Yamane, I., Kusumoto, M. and Katsuda, K. 2023. The association between farm-level antimicrobial usage and resistance of Staphylococcus spp., as the major genus isolated from aerosol samples, in Japanese piggeries. Front. Vet. Sci. 10(1), 1127819. Kruse, A.B., de Knegt, L.V., Nielsen, L.R. and Alban, L. 2016. No clear effect of initiating vaccination against common endemic infections on the amounts of prescribed antimicrobials for danish weaner and finishing pigs during 2007-2013. Front. Vet. Sci. 3(1), 120. Kruse, A.B., Nielsen, L.R. and Alban, L. 2020. Herd typologies based on multivariate analysis of biosecurity, productivity, antimicrobial and vaccine use data from Danish sow herds. Prev. Vet. Med. 181, 104487. Kumar, S., Singh, S., Kumar, V., Datta, S., Dhanjal, D.S., Sharma, P. and Singh, J. 2020. Pathogenesis and antibiotic resistance of Staphylococcus aureus. In Model Organisms for Microbial Pathogenesis, Biofilm Formation and Antimicrobial Drug Discovery. Eds., Siddhardha, B., Dyavaiah, M. and Syed, A. Singapore, Springer Singapore, pp: 99–115. Lam, Y., Fry, J.P. and Nachman, K.E. 2019. Applying an environmental public health lens to the industrialization of food animal production in ten low-and middle-income countries. Global. Health 15(1), 1–20. Le, V.P., Jeong, D.G., Yoon, S.W., Kwon, H.M., Trinh, T.B.N., Nguyen, T.L., Bui, T.T.N., Oh, J., Kim, J.B., Cheong, K.M., Van Tuyen, N., Bae, E., Vu, T.T.H., Yeom, M., Na, W. and Song, D. 2019. Outbreak of African Swine Fever, Vietnam, 2019. Emerg. Infect. Dis. 25(7), 1433–1435. Lekagul, A., Tangcharoensathien, V. and Yeung, S. 2019. Patterns of antibiotic use in global pig production: a systematic review. Vet. Anim. Sci 7(1), 100058. Lin, J., Yeh, K.S., Liu, H.T. and Lin, J.H. 2009. Staphylococcus aureus Isolated from Pork and chicken Carcasses in Taiwan: prevalence and antimicrobial susceptibility. J. Food Prot. 72(3), 608–611. Linhares, L.L., Yang, M., Sreevatsan, S., Munoz-Zanzi, C.A., Torremorell, M. and Davies, P.R. 2015. The effect of anatomic site and age on detection of Staphylococcus aureus in pigs. J. Vet. Diagn. Invest. 27(1), 55–60. Luu, Q.H., Nguyen, T.B.T., Ta, H.L., Erickson, V.I. and Padungtod, P. 2021. Quality testing of veterinary antimicrobial products used for livestock in Vietnam, 2018–2019. PLoS One 16(3), e0247337. Maertens, H., De Reu, K., Meyer, E., Van Coillie, E. and Dewulf, J. 2019. Limited association between disinfectant use and either antibiotic or disinfectant susceptibility of Escherichia coli in both poultry and pig husbandry. BMC Vet. Res. 15(1), 310. Magouras, I., Carmo, L.P., Stärk, K.D. and Schüpbach-Regula, G. 2017. Antimicrobial usage and-resistance in livestock: where should we focus? Front. Vet. Sci. 4(1), 148. Makita, K., Goto, M., Ozawa, M., Kawanishi, M., Koike, R., Asai, T. and Tamura, Y. 2016. Multivariable analysis of the association between antimicrobial use and antimicrobial resistance in Escherichia coli isolated from apparently healthy pigs in Japan. Microb. Drug Resist. 22(1), 28–39. Martelli, F., Lambert, M., Butt, P., Cheney, T., Tatone, F.A., Callaby, R., Rabie, A., Gosling, R.J., Fordon, S. and Crocker, G. 2017. Evaluation of an enhanced cleaning and disinfection protocol in Salmonella contaminated pig holdings in the United Kingdom. PLoS One 12(6), e0178897. Monaco, M., Pimentel de Araujo, F., Cruciani, M., Coccia, E.M. and Pantosti, A. 2017. Worldwide epidemiology and antibiotic resistance of Staphylococcus aureus. In Staphylococcus aureus: microbiology, pathology, immunology, therapy and prophylaxis. Eds., Bagnoli, F., Rappuoli, R. and Grandi, G. Cham, Switzerland: Springer International Publishing, pp: 21–56. National Assembly of the Socialist Republic of Vietnam, 2018. Law on Animal Husbandry, 32/2018/QH14. Available via https://www.economica.vn/Content/files/LAW%20%26%20REG/Law%20on%20Animal%20Husbandry%202018.pdf, Aoa, (Accessed 12 January 2021). Nghiem, M.N., Nguyen, V.T., Nguyen, T.T.H., Nguyen, T.D. and Vo, T.T.B. 2017. Antimicrobial resistance gene expression associated with multidrug resistant Salmonella spp. isolated from retail meat in Hanoi, Vietnam. Int. Microbiol. 20(2), 85–93. Ngo, T.K.C., Nguyen, C.D., Ngo, T.L.Q. and Ha, M.T. 2020. Biosecurity level practices in pig and poultry production in Vietnam. Adv. Anim. Vet. Sci. 8(10), 1068–1074. Nguyen, T.N., Cao, T.T., Nguyen, T.V., Campbell, J., Baker, S., Thwaites, G., Ngo, H.T. and Carrique-Mas, J. 2015. Induction of antimicrobial resistance in Escherichia coli and non-typhoidal Salmonella strains after adaptation to disinfectant commonly used on farms in Vietnam. Antibiotics 4(4), 480–494. Nguyen, T.N., Hotzel, H., El-Adawy, H., Tran, H.T., Le, M.T., Tomaso, H., Neubauer, H. and Hafez, H.M. 2016a. Genotyping and antibiotic resistance of thermophilic Campylobacter isolated from chicken and pig meat in Vietnam. Gut Pathog. 8(1), 19. Nguyen, T.T., L., P.T.N., Nguyen, N.Q., S., D.X., Lee, H.S., Nguyen, V.H., Padungtod, P., Nguyen, T.T., Nguyen, T.T., Tran, C.T. and Rich, K.M. 2021. An assessment of the economic impacts of the 2019 African swine fever outbreaks in Vietnam. Front. Vet. Sci. 8(1), 686038. Nguyen, V.C., Nguyen, T.N., Nguyen, H.N., Nguyen, T.M.H., Nguyen, V.T., Thwaites, G. and Carrique-Mas, J. 2016b. Antimicrobial consumption in medicated feeds in Vietnamese pig and poultry production. EcoHealth 13(3), 490–498. Nguyen, V.K., Zhang, T., Vu, T.B.N., Dao, T.T., Tran, K.T., Nguyen, T.D.N., Tran, T.H.K., Nguyen, T.C.K., Fox, A. and Horby, P. 2014. Staphylococcus aureus nasopharyngeal carriage in rural and urban northern Vietnam. Trans. R. Soc. Trop. Med. Hyg. 108(12), 783–790. Nhung, T.N., Nguyen, H.M., Nguyen, C.V., Nguyen, T.V., Nguyen, M.T., Thai, H.Q., Ho, M.H., Thwaites, G., Ngo, H.T., Baker, S., Carrique-Mas, J. and Elkins, C.A. 2016. Use of colistin and other critical antimicrobials on pig and chicken farms in Southern Vietnam and its association with resistance in commensal Escherichia coli bacteria. Appl. Environ. Microbiol. 82(13), 3727–3735. Nobre, M., Santos, L., Silva, D., Oliveira, F., Araújo, A., Campos, M., Sousa, B., Figueirêdo, A., Muratori, M. and Soares, M. 2021. Multiresistance and virulence factors of Staphylococcus aureus isolated from pigs. Arq. Bras. Med. Vet. Zootec. 73(1), 343–351. Obolski, U., Lourenço, J., Thompson, C., Thompson, R., Gori, A. and Gupta, S. 2018. Vaccination can drive an increase in frequencies of antibiotic resistance among nonvaccine serotypes of of Streptococcus pneumoniae. Proc. Natl. Acad. Sci. 115(12), 3102–3107. Official Journal of the European Union. 2006. Regulation (EC) No 1831/2003 of the European Parliament and of the Council of 22 September 2003 on Additives for Use in Animal Nutrition. Off J Eur Union. Oct 18; L 268: p. 29. lastly amended by Regulation (EC) No. 767/2009. Partridge, S.R., Kwong, S.M., Firth, N. and Jensen, S.O. 2018. Mobile genetic elements associated with antimicrobial resistance. Clin. Microbiol. Rev. 31(4), e00088–e00017. doi:10.1128/cmr. 00088-00017. Peiponen, K.S., Tirkkonen, B.T., Junnila, J.J.T. and Heinonen, M.L. 2018. Effect of a live attenuated vaccine against Lawsonia intracellularis in weaned and finishing pig settings in Finland. Acta Vet. Scand. 60(1), 18. Pereira, B.M.P., Wang, X. and Tagkopoulos, I. 2021. Biocide-induced emergence of antibiotic resistance in Escherichia coli. Front. Microbiol. 12(1), 640923. Pham, K.D., Saegerman, C., Douny, C., Vu, D.T., Ha, V.B. and Dang, V.B. 2013. First survey on the use of antibiotics in pig and poultry production in the Red River Delta region of Vietnam. Food Public Health 3(1), 247–256. Postma, M., Backhans, A., Collineau, L., Loesken, S., Sjölund, M., Belloc, C., Emanuelson, U., grosse Beilage, E., Nielsen, E.O. and Stärk, K.D. 2016. Evaluation of the relationship between the biosecurity status, production parameters, herd characteristics and antimicrobial usage in farrow-to-finish pig production in four EU countries. Porcine Health Manag. 2(9), 1–11. Renault, V., Humblet, M.F. and Saegerman, C. 2021. Biosecurity concept: origins, evolution and perspectives. Animals, 12(1), 63. Rhouma, M., Beaudry, F. and Letellier, A. 2016. Resistance to colistin: what is the fate for this antibiotic in pig production? Int. J. Antimicrob. Agents 48(2), 119–126. Sali, V., Nykäsenoja, S., Heikinheimo, A., Hälli, O., Tirkkonen, T. and Heinonen, M. 2021. Antimicrobial use and susceptibility of indicator Escherichia coli in finnish integrated pork production. Front. Microbiol. 12(1), 754894. Silbergeld, E.K., Graham, J. and Price, L.B. 2008. Industrial food animal production, antimicrobial resistance, and human health. Annu. Rev. Public Health 29(1), 151–169. Sineke, N., Asante, J., Amoako, D.G., Abia, A.L.K., Perrett, K., Bester, L.A. and Essack, S.Y. 2021. Staphylococcus aureus in intensive pig production in South Africa: antibiotic resistance, virulence determinants, and clonality. Pathogens (Basel, Switzerland), 10(3), 317. Song, J.H., Hsueh, P.R., Chung, D.R., Ko, K.S., Kang, C.I., Peck, K.R., Yeom, J.S., Kim, S.W., Chang, H.H., Kim, Y.S., Jung, S.I., Son, J.S., So, T.M., Lalitha, M.K., Yang, Y., Huang, S.G., Wang, H., Lu, Q., Carlos, C.C., Perera, J.A., Chiu, C.H., Liu, J.W., Chongthaleong, A., Thamlikitkul, V. and Van, P.H. 2011. Spread of methicillin-resistant Staphylococcus aureus between the community and the hospitals in Asian countries: an ANSORP study. J. Antimicrob. Chemother. 66(5), 1061–1069. Spronk, T., Green, A.L., Vuolo, M., Ruesch, L., Edler, R., Haley, C., Scaria, J., Hennings, J., Dee, S. and Shivley, C.B. 2023. Antimicrobial use and antimicrobial resistance monitoring in pig production in the United States of America. Revue Sci. et Tech. (International Office of Epizootics), 42(1), 52–64. Sun, J., Yang, M., Sreevatsan, S. and Davies, P.R. 2015. Prevalence and characterization of Staphylococcus aureus in growing pigs in the USA. PLoS One 10(11), e0143670. Swann, M., Baxter, K. and Field, H. 1969. Report of the Joint Committee on the use of Antibiotics in Animal Husbandry and Veterinary medicine. London, UK. London Her Majesty’s Stationery Office. Temtem, C., Kruse, A.B., Nielsen, L.R., Pedersen, K.S. and Alban, L. 2016. Comparison of the antimicrobial consumption in weaning pigs in Danish sow herds with different vaccine purchase patterns during 2013. Porcine Health Manag. 2(1), 23. The Ministry of Agriculture and Rural Development, 2020. Circular No. 12/2020/TT-BNNPTNT of the Minister of Agriculture and Rural Development Providing for Management of Veterinary Drugs Containing Narcotic Substances and Precursors; Veterinary Prescribing; Amendments to Circular No. 18/2018/TT-BNNPTNT. Development, TMoAaR, Tong, S.Y., Davis, J.S., Eichenberger, E., Holland, T.L. and Fowler Jr, V.G. 2015. Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 28(3), 603–661. Tuat, C.V., Hue, P.T., Loan, N.T.P., Thuy, N.T., Hue, L.T., Giang, V.N., Erickson, V.I. and Padungtod, P. 2021. Antimicrobial resistance pilot surveillance of pigs and chickens in Vietnam, 2017-2019. Front. Vet. Sci. 8(1), 618497. Urban-Chmiel, R., Marek, A., Stępień-Pyśniak, D., Wieczorek, K., Dec, M., Nowaczek, A. and Osek, J. 2022. Antibiotic resistance in bacteria—a review. Antibiotics, 11(8), 1079. Van Boeckel, T.P., Glennon, E.E., Chen, D., Gilbert, M., Robinson, T.P., Grenfell, B.T., Levin, S.A., Bonhoeffer, S. and Laxminarayan, R. 2017. Reducing antimicrobial use in food animals. Science 357(6358), 1350–1352. Vestergaard, M., Frees, D. and Ingmer, H. 2019. Antibiotic resistance and the MRSA problem. Microbiol. Spectr. 7(2), 57. Vo, T.D.T., Huynh, A.D.M., Ngo, T.H., Nguyen, D.T. and Tran, T.M.T. 2012. An investigation of the carriage rate of methicillin-resistant Staphylococcus aureus in pigs in the Western Province of Vietnam. J. Biomimet. Biomat. Tissue Eng. 12(1), 91–98. Vu, T.K.H., Truong, T.H.G., Tran, T.N., Huynh, T.M.L. and Dang, T.T.S. 2020. Prevalence and antibiotic resistance of Staphylococcus aureus and Methicillin resistant S. aureus isolated from pigs. J. Vet. Sci. Technol. XXVII(2), 45–53. Wand, M.E., Bock, L.J., Bonney, L.C. and Sutton, J.M. 2017. Mechanisms of increased resistance to chlorhexidine and cross-resistance to colistin following exposure of Klebsiella pneumoniae clinical isolates to chlorhexidine. Antimicrob Agents Chemother. 61(1). Wee, B.A., Muloi, D.M. and van Bunnik, B.A.D. 2020. Quantifying the transmission of antimicrobial resistance at the human and livestock interface with genomics. Clin. Microbiol. Infect. 26(12), 1612–1616. Wieland, N., Boss, J., Lettmann, S., Fritz, B., Schwaiger, K., Bauer, J. and Hölzel, C.S. 2017. Susceptibility to disinfectants in antimicrobial-resistant and -susceptible isolates of Escherichia coli, Enterococcus faecalis and Enterococcus faecium from poultry-ESBL/AmpC-phenotype of E. coli is not associated with resistance to a quaternary ammonium compound, DDAC. J. Appl. Microbiol. 122(6), 1508–1517. Woodford, N. and Livermore, D.M. 2009. Infections caused by Gram-positive bacteria: a review of the global challenge. J. Infect. 59 Suppl 1(S4-16), 19766888. Zehra, A., Singh, R., Kaur, S. and Gill, J.P.S. 2017. Molecular characterization of antibiotic-resistant Staphylococcus aureus from livestock (bovine and swine). Vet. World 10(6), 598–604. Zhang, B., Ku, X., Yu, X., Sun, Q., Wu, H., Chen, F., Zhang, X., Guo, L., Tang, X. and He, Q. 2019. Prevalence and antimicrobial susceptibilities of bacterial pathogens in Chinese pig farms from 2013 to 2017. Sci. Rep. 9(1), 1–11. Zhang, H. and Kono, H. 2012. Economic impacts of porcine reproductive and respiratory syndrome (PRRS) outbreak in Vietnam pig production. Trop. Agri. Res. 23(2), 152–159. | ||
| How to Cite this Article |
| Pubmed Style Chao NV, Dung HT, Tam VTT, Hang PT, Hien BT. The role of veterinary drug use in driving antimicrobial resistance of Staphylococcus aureus isolates in smallholder swine farms in Central Vietnam. Open Vet. J.. 2025; 15(2): 847-862. doi:10.5455/OVJ.2025.v15.i2.34 Web Style Chao NV, Dung HT, Tam VTT, Hang PT, Hien BT. The role of veterinary drug use in driving antimicrobial resistance of Staphylococcus aureus isolates in smallholder swine farms in Central Vietnam. https://www.openveterinaryjournal.com/?mno=227411 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.34 AMA (American Medical Association) Style Chao NV, Dung HT, Tam VTT, Hang PT, Hien BT. The role of veterinary drug use in driving antimicrobial resistance of Staphylococcus aureus isolates in smallholder swine farms in Central Vietnam. Open Vet. J.. 2025; 15(2): 847-862. doi:10.5455/OVJ.2025.v15.i2.34 Vancouver/ICMJE Style Chao NV, Dung HT, Tam VTT, Hang PT, Hien BT. The role of veterinary drug use in driving antimicrobial resistance of Staphylococcus aureus isolates in smallholder swine farms in Central Vietnam. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 847-862. doi:10.5455/OVJ.2025.v15.i2.34 Harvard Style Chao, N. V., Dung, . H. T., Tam, . V. T. T., Hang, . P. T. & Hien, . B. T. (2025) The role of veterinary drug use in driving antimicrobial resistance of Staphylococcus aureus isolates in smallholder swine farms in Central Vietnam. Open Vet. J., 15 (2), 847-862. doi:10.5455/OVJ.2025.v15.i2.34 Turabian Style Chao, Nguyen Van, Ho Thi Dung, Vu Thi Thanh Tam, Phan Thi Hang, and Bui Thi Hien. 2025. The role of veterinary drug use in driving antimicrobial resistance of Staphylococcus aureus isolates in smallholder swine farms in Central Vietnam. Open Veterinary Journal, 15 (2), 847-862. doi:10.5455/OVJ.2025.v15.i2.34 Chicago Style Chao, Nguyen Van, Ho Thi Dung, Vu Thi Thanh Tam, Phan Thi Hang, and Bui Thi Hien. "The role of veterinary drug use in driving antimicrobial resistance of Staphylococcus aureus isolates in smallholder swine farms in Central Vietnam." Open Veterinary Journal 15 (2025), 847-862. doi:10.5455/OVJ.2025.v15.i2.34 MLA (The Modern Language Association) Style Chao, Nguyen Van, Ho Thi Dung, Vu Thi Thanh Tam, Phan Thi Hang, and Bui Thi Hien. "The role of veterinary drug use in driving antimicrobial resistance of Staphylococcus aureus isolates in smallholder swine farms in Central Vietnam." Open Veterinary Journal 15.2 (2025), 847-862. Print. doi:10.5455/OVJ.2025.v15.i2.34 APA (American Psychological Association) Style Chao, N. V., Dung, . H. T., Tam, . V. T. T., Hang, . P. T. & Hien, . B. T. (2025) The role of veterinary drug use in driving antimicrobial resistance of Staphylococcus aureus isolates in smallholder swine farms in Central Vietnam. Open Veterinary Journal, 15 (2), 847-862. doi:10.5455/OVJ.2025.v15.i2.34 |