| Review Article | ||
Open Vet. J.. 2025; 15(2): 541-555 Open Veterinary Journal, (2025), Vol. 15(2): 541-555 Review Article Lumpy skin disease: A growing threat to the global livestock industryNanik Hidayatik1*, Aswin Rafif Khairullah2, M. Gandul Atik Yuliani1, Eka Pramyrtha Hestianah3, Arindita Niatazya Novianti1, Ratna Damayanti1, Mirza Atikah Madarina Hisyam4, Ikechukwu Benjamin Moses5, Riza Zainuddin Ahmad2, Bantari Wisynu Kusuma Wardhani6, Syahputra Wibowo7 and Dea Anita Ariani Kurniasih81Division of Basic Veterinary Medicine, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 2Research Center for Veterinary Science, National Research and Innovation Agency (BRIN), Bogor, Indonesia 3Division of Veterinary Anatomy, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 4Division of Veterinary Clinic, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 5Department of Applied Microbiology, Faculty of Science, Ebonyi State University, Abakaliki, Nigeria 6Research Center for Pharmaceutical Ingredients and Traditional Medicine, National Research and Innovation Agency (BRIN), Bogor, Indonesia 7Eijkman Research Center for Molecular Biology, National Research and Innovation Agency (BRIN), Bogor, Indonesia 8Research Center for Public Health and Nutrition, National Research and Innovation Agency (BRIN), Bogor, Indonesia *Corresponding Author: Nanik Hidayatik. Division of Basic Veterinary Medicine, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: nanik-h [at] fkh.unair.ac.id Submitted: 07/11/2024 Accepted: 21/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
AbstractLumpy Skin Disease (LSD) is an infectious disease in cattle caused by the Lumpy Skin Disease Virus by the Poxviridae family. Historically, LSD was first documented in Northern Rhodesia (now Zambia) in 1929 as a symptom of bug bite poisoning or hypersensitivity, described as pseudo-urticaria. This disease, which causes widespread malaise and persistent weakness, is a serious, economically devastating, and reportable condition that lowers cattle output. The illness starts as a biphasic fever. After fever, emaciation, ocular discharge, and agalactia, the clinical signs of a minor infection develop as one or two nodular lumps appear 2–3 days later. In particular, the skin of the muzzle, back, nostrils, legs, scrotum, eyelids, lower ears, nasal, perineum, oral mucosa, and tail display painful, hyperemic, nodular lesions. A number of diagnostic methods, such as virus isolation in cell culture, transmission electron microscopy, immunohistochemistry, direct and indirect fluorescent antibody testing, agar gel immunodiffusion, enzyme-linked immunosorbent assay, western blotting, and serum neutralization test (SNT), are used to confirm the clinical diagnosis. This disease is spread by arthropods such as flies, mosquitoes, and ticks. It is most common during the rainy season, when there is a high number of biting insects, and it declines during the dry season. LSD has direct and indirect economic impacts on livestock and the industry. Since there is no effective antiviral medication to treat LSD, the only viable method of disease containment is vaccination. Keywords: Cattle, Disease, LSD, Nodules, Virus. IntroductionLumpy Skin Disease (LSD) is an infectious disease in cattle caused by the Lumpy Skin Disease Virus (LSDV) from the Poxviridae family (Ratyotha et al., 2022). There are other names for this disease, including Neethling’s virus illness, exanthema nodularis bovis, and pseudourticaria (Namazi and Tafti, 2021). The cattle industry sector is greatly impacted economically by this disease, which results in chronic weakness in affected cows, reduced milk output, miscarriage, temporary or permanent sterility, skin damage, and death (Akther et al., 2023). Nodular lesions on the skin and mucous membranes are the most typical clinical signs (Datten et al., 2023). Infected cattle frequently have skin nodular lesions on the outside of their bodies, including the head, neck, back, perineum, chest, and other parts (Liang et al., 2022). Although this disease can affect cattle of any age or breed, it primarily affects young cattle and animals that are at the height of lactation (Wilhelm and Ward, 2023). This disease is spread by arthropods such as flies, mosquitoes, and ticks. It is most common during the rainy season, when there is a high number of biting insects, and it declines during the dry season (Sprygin et al., 2019). LSDV can travel long distances, even across continents, if quarantine regulations are loosened and infected animals are transferred from one farm to another (Ratyotha et al., 2022). Notably, epidemiological evidence of zoonotic infection is lacking. Tests such as polymerase chain reaction (PCR), enzyme-linked immunosorbent assay (ELISA), serum neutralization test (SNT), and transmission electron microscopy (TEM) are used to provide conclusive diagnoses (Rouby and Aboulsoud, 2016). The incubation period for LSD is 2–5 weeks after infection with the virus, and the first lesions appear at the site of inoculation 2–3 days later. The first sign is fever, which is followed in two days by the formation of skin and mucous membrane nodules (Tuppurainen et al., 2015). LSD does not include a specific treatment. To alleviate clinical symptoms and manage any ensuing problems, infected animals should receive supportive care (Liang et al., 2022). Little is known about effective LSD prevention strategies. It is strongly recommended that diseased animals be kept in quarantine, that they be sacrificed, and that their mobility be restricted (Eom et al., 2023). The World Organization for Animal Health’s International Epizootic Office has added LSD to its list of diseases that need to be reported (Akther et al., 2023). Although it is endemic in African nations, reports of this disease have recently come from other parts of the world (Tuppurainen et al., 2021). Later, LSD made its way to Yemen, Kuwait, and Oman (Datten et al., 2023). Currently, this disease has emerged as a very major threat to large ruminant livestock in Asia, Europe, and the Middle East (Anwar et al., 2022). The disease’s recurrent outbreaks and resurgence in different regions of the world underscore the significance of reassessing the disease’s biology, virus transmission methods, and modern preventive and adaptive management strategies. The purpose of this review article is to explain the etiology, history, epidemiology, pathogenesis, immune response, clinical symptoms, diagnosis, differential diagnosis, transmission, host range, risk factors, zoonotic potential, economic impact, treatment, vaccination, and control of LSD. Planning for efficient disease management in a timely manner requires an understanding of the risks associated with LSDs in livestock husbandry. EtiologyThe virus that causes LSD illness is a member of the family Poxviridae, genus Capripoxvirus (CaPV), and subfamily Chordopoxvirinae (Akther et al., 2023). The LSD-causing virus is oval in shape, 230–260 nm in diameter, has a lipid envelope, replicates in the cytoplasm, and has a double-stranded DNA genome of approximately 151 kbp in length (Namazi and Tafti, 2021). It is believed to contain 156 genes and comprises a central coding region surrounded by identical inverted terminal repeats that are 2.4 kbp long (Rouby et al., 2024). When comparing LSD viruses to other genera of Chordopoxviruses, 146 conserved protein-coding genes were found to be involved in nucleotide metabolism, DNA replication, protein processing, virion structure creation, virulence, host range, and mRNA transcription and biogenesis (Tulman et al., 2001). Despite being phylogenetically distinct, this virus shares a high degree of genomic similarity (96%) with sheeppox and goatpox viruses (Tulman et al., 2002). The vaccine and LSD viruses share a similar morphological structure when observed under an electron microscope (Liang et al., 2022). Viruses can multiply in primary cells, including fibroblasts in chicken embryos, lung and kidney cells in sheep embryos, and kidney or testis cells in sheep and cattle (Rhazi et al., 2021). Furthermore, this virus can replicate in the kidney cells of cows and infant hamsters, although the pathological alterations are gradual. In contrast, the virus cannot reproduce in African green monkeys (Wolff et al., 2020b). LSDV is stable under alkaline conditions and can thrive at pH 6.6–8.6 (Eom et al., 2023). The virus can live for 33 days in necrotic skin nodules, 35 days in dry scabs, 6 months in infected tissue shielded from sunlight, and 18 days in dry skin at room temperature (Kumar et al., 2021). Most LSDVs may be rendered inactive at 55°C for two hours and 65°C for half an hour; however, their tolerance to high temperatures is variable (Podshibyakin et al., 2024). Additionally, this virus is susceptible to detergents containing lipid solvents and extremely basic or acidic solutions (Das et al., 2021). This virus is also vulnerable to dilution of iodine compounds, 2% phenol, 1% formalin, chloroform, 2%–3% sodium hypochlorite, ether, 0.5% quaternary ammonium, and UV light at 55°C for 1 hour (Moda et al., 2024). HistoryHistorically, LSD was first documented in Northern Rhodesia (now Zambia) in 1929 as a symptom of bug bite poisoning or hypersensitivity, described as “pseudo-urticaria” (Mazloum et al., 2023). The historical-critical events associated with LSD are mapped in Figure 1 from the very first time it was detected to date with a global timeline, regional spread, and reporting of notable outbreaks. Between 1943 and 1945, the Republic of South Africa, Botswana, and Zimbabwe (Southern Rhodesia) reported instances of contagious COVID-19 (Mafirakureva et al., 2017). Mozambique was in the east by 1946, followed by Angola and Zaire in 1950, Madagascar in 1954, and Namibia, Tanzania, and Uganda in 1956 with the disease’s spread (Elhaig et al., 2017). Numerous African countries have studied this virus during the ensuing 20 years. In 1957, LSD made its debut in Kenya, and it reemerged in 1977 (Davies, 1982). Another African nation, Sudan, reported the unexpected emergence of the LSD in 1971, after a 14-year lapse (Ali and Obeid, 1977). In 1973, an LSD inflow occurred in Chad and Niger, which contributed significantly to the 1974 pandemic in Nigeria (Wolff et al., 2021). The LSD was initially identified in Ethiopia in 1983 and progressively expanded to practically every area and agroecological zone in the nation (Gari et al., 2012). Between 1950 and 1985, numerous investigations documented a number of isolated LSD events in Tanzania, Kenya, Zimbabwe, Somalia, and Cameroon, with spontaneous reemergences in other African nations over the same time frame (Akther et al., 2023).
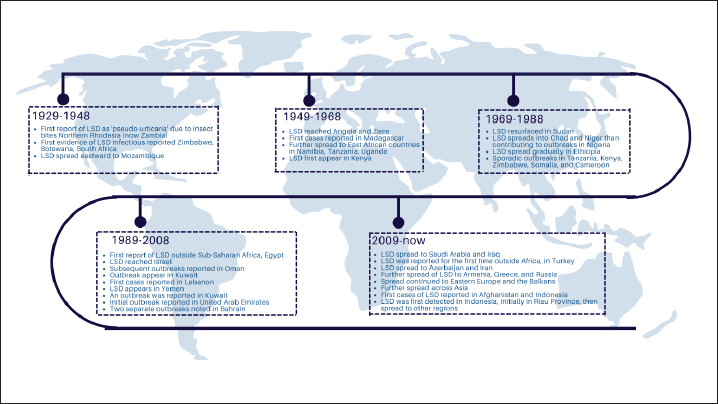
Fig. 1. Historical timeline of key events in lumpy skin disease (LSD) from 1929 to the present. The first documented LSD outbreak outside the Sub-Saharan region occurred in Egypt in 1988 (Rouby et al., 2021). LSD was first recorded in Oman in 1984, and the epizootic disease resurfaced in 2009, despite minor outbreaks with unverified cases (Tageldin et al., 2014). In Kuwait, the disease was initially discovered in 1986, and it resurfaced in 1991 (Azeem et al., 2022). Furthermore, the predominance of LSD was initially noted in 1993 in Lebanon, 1995 in Yemen, 2000 in the United Arab Emirates, 1993 and again in 2002–2003 in Bahrain, 2009 in Oman, and 2013 in Saudi Arabia and Iraq (Alkhamis and VanderWaal, 2016). The importance of exposing hosts to pathogens in an environment that supports disease spread and transmission cannot be overstated (Bianchini et al., 2023). LSD is more common in low-lying areas and near watercourses, especially during summer and autumn (Allepuz et al., 2019). This disease is considered a case of poisoning or a hypersensitivity reaction to insect bites due to the large numbers of biting insects at that time (Padha and Sofi, 2023). The illness has been reported in numerous nations worldwide. The first documented sporadic LSD occurrences occurred in 2015 in several European countries, including Greece, Albania, and the Russian Federation (Anwar et al., 2022). A significant epidemic occurred in Russia in 2017, and cases have been reported in a number of continental European nations (Byadovskaya et al., 2022). The majority of these cases have been effectively contained through widespread vaccination campaigns and appropriate management strategies. Although Georgia began vaccinating livestock in 2014 after an epidemic in neighboring Azerbaijan, an outbreak was documented in 2016 and reappeared in 2018 with six further outbreaks (Zeynalova et al., 2016). Meanwhile, outbreaks of LSD were documented in eight Balkan nations (Albania, Serbia, Kosovo, Greece, Bulgaria, and the former Yugoslav Republic of Macedonia), with a higher frequency in border regions (European Food Safety Authority, 2018). LSD has been a significant issue for Asian nations since 2019 and is one of the main risks to the continent’s food security and animal health (Li et al., 2023). The significance of LSD was highlighted by reports of a severe outbreak in Bangladesh in July 2019, and the ensuing outbreak in 2020 further demonstrated this (Uddin et al., 2024). Since then, numerous epidemics have been documented throughout Asia, including concurrent outbreaks in China and India (Lu et al., 2019; Manjunathareddy et al., 2024). LSD has spread quickly and widely throughout the nation, with outbreaks now occurring often. In 2020, outbreaks occurred in June, July, September, October, and November in Nepal, Bhutan, Sri Lanka, Vietnam, and Malaysia, respectively (Akther et al., 2023). In 2021, the World Organization for Animal Health reported one outbreak in Thailand in April, one in the Laos People’s Democratic Republic in May, and one in Pakistan in November (Saltykov et al., 2022). In March 2022, LSD was confirmed in Indonesia (Sendow et al., 2024). The emergence and reemergence of LSDs are quite concerning, particularly in light of the fact that recent outbreaks do not exhibit a distinct pattern that has been previously documented. EpidemiologyDeveloping control and prevention efforts can benefit from an understanding of the epidemiological characteristics of LSDs in infections, hosts, and the environment. LSDV does not infect nonruminant hosts and has a restricted host range. This virus can infect cattle, buffalo, and other wild ruminants (Ratyotha et al., 2022). Sendow et al. (2024) claimed that local cattle, Channel Nation cattle, and Friesland Holstein cattle could all contract LSD. In addition to the fact that buffalo always bury themselves in water or mud during the summer to lessen the likelihood of being attacked by insect vectors, cows have a greater morbidity rate than buffalo because buffalo have thick skin that makes it difficult for the LSD-spreading vector to puncture them (Di Felice et al., 2024). Furthermore, the LSD virus can infect wild animals, including giraffes, Thomson’s gazelles (Gazella thomsoni), and impala (Aepyceros melampus) (Ratyotha et al., 2022). Although there have been no reports of natural infection, LSDV has been shown to infect sheep and goats in experiments (Mazloum et al., 2023). Although LSDV can affect animals of any age, it is more likely to affect young animals. This is consistent with a study by Elhaig et al. (2017), which reported no discernible variations in the prevalence of LSD by age or sex among the 450 cows analyzed in Egypt. Moudgil et al. (2024) asserted that juvenile animals are more susceptible to disease than adult animals. Compared with adult animals, this virus can cause a significant mortality rate in young animals. The morbidity rate of this disease ranges from 5% to 45% and occasionally even 100%. Even if the death rate is less than 10%, it might reach 40% (Dubey et al., 2023). Numerous factors, including geographic location, climate, husbandry management, nutritional state, immune status, overall animal health, livestock type, virulence, population size, and insect vector distribution, influence variations in LSD mortality and morbidity rates (Akther et al., 2023). PathogenesisPoxviruses are generally epitheliotropic and can cause local or systemic disease (Modise et al., 2021). The initial viral replication occurs at the site of entry into the host. In systemic infection, further viral replication occurs in draining lymph nodes, followed by viremia and further viral replication in various organs, including the liver, spleen, and lungs (Khan et al., 2023). The latter multiplication leads to the development of secondary viremia and subsequent infection and the development of diffuse focal lesions on the skin (Liang et al., 2022). Virus replication occurs in the cytoplasm of the cell (Tulman et al., 2001). Virus particles are enveloped as mature virus particles move to the Golgi complex; however, most particles are not enveloped and are released through cell disruption (Schlosser-Perrin et al., 2023). Both enveloped and nonenveloped particles are infectious. LSD is characterized by fever and rapid spread of several small skin nodules (Adamu et al., 2024). Viral particles pass through the bloodstream and cause widespread lymphadenitis (Ren et al., 2023). Following the initial fever, viremia develops for four days. Viral replication in specific tissues, including fibroblasts, pericytes, and endothelial cells of lymphatic and blood arteries, causes subsequent skin lesions (Sanz-Bernardo et al., 2020). Acute skin injury can cause histopathological alterations, such as necrosis, edema, thrombosis, vasculitis, lymphangitis, and infarction (Gharban et al., 2019). There are nodules in the muscular fascia and subcutaneous tissue (Di Giuseppe et al., 2024). There is edema, congestion, and bleeding in the surrounding tissues of the dermis, epidermis, and core muscle, along with swollen lymph nodes (Manjunathareddy et al., 2024). Special formations known as “sit-fasts,” or necrotic cores isolated from the surrounding skin, are typically observed on a variety of body areas that are susceptible to ulceration (Datten et al., 2023). Lower levels of lymphocyte diffusion and phagocytic mobility were observed in the host’s immunological condition during the 14 days following infection (Neamat-Allah, 2015). Underweight animals, dairy cattle, and newborn calves seem to be more vulnerable to natural infection, perhaps as a result of compromised humoral immunity (Fay et al., 2022). Lifelong immunity has been demonstrated in animals that have recovered from natural viral infection. Because of maternally acquired antibodies, calves from infected dams are resistant to clinical illness for around six months (Namazi and Tafti, 2021). There is no known carrier status of LSDV, and infected animals recover from the virus. LSDV causes lymphangitis and vasculitis by infiltrating a range of cell types, including epithelial and endothelial cells, pericytes, and fibroblasts. However, the precise pathogenesis of lesions associated with cutaneous disease is not as well understood as that of the sheeppox virus (Sanz-Bernardo et al., 2020). Edema and necrosis occur during the acute phase of vasculitis and lymphangitis, along with thrombosis and infarction (Ratyotha et al., 2022). Initially, neutrophils and macrophages penetrate the lesion; fibroblasts, lymphocytes, plasma cells, and macrophages progressively replace these cells (Manjunathareddy et al., 2024). Immune responseFollowing natural or experimental infection, LSD stimulates cellular immunity and antibody production. The degree of clinical disease severity may influence the amount of antibody produced; animals with severe sickness and skin lesions typically have higher antibody levels because the high levels of viral antigen induce a B cell response (Mazloum et al., 2023). Nevertheless, after infection, some subclinically infected animals do not produce detectable antibody levels. Serum transfer tests have shown that antibodies by themselves can prevent infection with the CaPV (Tuppurainen et al., 2017). Regretfully, specific antibodies to the CaPV in cattle were not used in this investigation. An inactivated LSDV vaccination that may produce antibody responses and protect against experimental infection further illustrates the importance of antibody responses in LSDV protection (Haegeman et al., 2023). The ability of a live attenuated vaccination to protect animals against LSDV without a detectable antibody response revealed the importance of cellular immunity in defending animals against LSDV (Dhanda et al., 2024). Since research on CD4+ and CD8+ T-lymphocyte depletion in cattle has not been conducted, it is unknown how important these T cells are to the protection that results from immunization with live attenuated vaccines (Davis et al., 2024). A weak response in either immunity can be compensated for, according to current experimental research, which indicates that immunity to LSDV is a combination of cellular and antibody immunological responses (Milovanović et al., 2019). Clinical symptomsThis disease takes 2–5 weeks to incubate in natural settings, whereas it takes 7–14 days in experimental settings (Wolff et al., 2020a). There are three types of LSD: chronic, subacute, and acute. The illness starts as biphasic fever (Di Felice et al., 2024). After fever, emaciation, ocular discharge, and agalactia, the clinical signs of a minor infection develop as one or two nodular lumps appear 2–3 days later (Datten et al., 2023). In particular, the skin of the muzzle, back, nostrils, legs, scrotum, eyelids, lower ears, nasal, perineum, and oral mucosa, and tail display painful, hyperemic, nodular lesions (Pandey et al., 2022). In severe cases, the stage lasts for 7–12 days and involves the development of over 100 nodules on the skin over the body (Kumar et al., 2021). A narrow hemorrhagic ring separates the firm and slightly elevated nodules from the surrounding skin. The nodules involve the dermis, epidermis, adjacent subcutis, and muscle (Amin et al., 2021). Subsequently, the lesions transform into exuding papules, vesicles, and pustules before gradually crusting over (Ratyotha et al., 2022). Lesions heal quite slowly. Lesions gradually form on the mucous membranes of the mouth, vulva, respiratory tract, and nose. After 2–3 weeks, the skin lesions become harder and necrotic, which causes discomfort to the animals, and they become reluctant to move (Gupta et al., 2020). Septicemia may result from bacterial and screwworm fly invasion caused by the lesion’s characteristic “sitfast”-shaped hole, which is created as the peels of the lesion (Al-Salihi, 2014). Animals with this infection also exhibit widespread lymph node enlargement. Lesions of skin disease exhibit bulging epithelial cell degeneration and eosinophilic intracytoplasmic inclusion bodies on histopathology (Parvin et al., 2022). Pneumonia is a side effect of LSDs that occurs when animals inhale necrotic material (Shumilova et al., 2023). The acute stage of infection is when abortion occurs. Another effect of this illness in male and female animals is infertility (Hasib et al., 2021). Female animals spend a lot of time in anestrus. Male-infected animals with genital sores also stay sterile for months (Bianchini et al., 2023). Recovery is very slow due to secondary bacterial infections, pneumonia, mastitis, and fly attacks in necrotic lesions, which leave deep holes in the body (Murti et al., 2024). DiagnosisTypical clinical symptoms are typically used to make a preliminary diagnosis of LSD. A number of diagnostic methods, such as virus isolation in cell culture, TEM, immunohistochemistry, direct and indirect fluorescent antibody testing, agar gel immunodiffusion, ELISA, western blotting, and SNT, are used to confirm the clinical diagnosis (Amin et al., 2021). Among the molecular diagnostic techniques employed are dot blot hybridization, real-time PCR, and conventional PCR (Elhaig et al., 2017). LSDV can be cultured in a variety of tissue cultures: sheep and calf kidney cells, sheep kidney cells, sheep and calf adrenal, and thyroid cultures, sheep and calf fetal muscle cells, rabbit fetal kidney and skin cells, sheep and calf testis cells, sheep embryonic kidney and lung cells, chicken embryo fibroblasts, on the chorioallantoic membrane of embryonated chicken eggs, African green monkey (Vero) kidney cells, baby hamster kidney cells, primary cell cultures of the bovine dermis, and horse lung cells (Alemu, 2024). The formation of the cytopathic effect, which is characterized by rounding, shrinking, and shedding of cells to give the monolayer a moth-eaten appearance, can take up to 14 days after primary isolation (Kumar et al., 2021). The gold standard and most specific serological test for identifying LSDV antibodies is SNT, although it takes a long time to do (Krešić et al., 2020). When interpreting results, it is always important to consider the sensitivity of SNT in the presence of low amounts of neutralizing antibodies in the tested serum, as documented previously (Ntombela et al., 2023). As a result, a negative test result does not necessarily indicate that the animal has not encountered the virus. SNT’s specificity and sensitivity were 97% and 78%, respectively (Senthilkumar et al., 2024). This is because a cell-mediated immune response is primarily triggered by LSDV infection. LSDV can be detected using fluorescence antibody methods. However, there is a possibility that this method will react with other Parapoxviruses (Zeedan et al., 2019). SNT has not been shown to exhibit such cross-reactions. In addition, this method is not as specific as SNT. Western blotting is another method for reliably detecting LSDV with sensitivity and specificity, but it is costly and necessitates specialized equipment and expertise to conduct (Chapman et al., 2021). Differential diagnosisNumerous illnesses have symptoms that resemble those of LSD. Veterinarians must make an honest diagnosis to guarantee basic preventive measures for sensitive livestock. Many illnesses, including pseudo-scabiei, bovine viral diarrhea/mucosal disease, demodicosis (Demodex), photosensitization, malignant catarrhal fever of the bovine, besnoitiosis, insect bite allergy, pseudo-cowpox, oncocercariasis, urticaria, infectious tuberculosis, Rinderpest, and vaccinia virus/cowpox, can be mistaken for LSD (Sukoco et al., 2023). TransmissionLSDs can harm wild ruminants, cattle, and buffalo (Akther et al., 2023). Goats and sheep do not appear to be affected by this virus. Indirect transmission is less common in LSD, and it occurs only in recombinant LSDV strains. In dry scabs, LSDV can survive for extended periods at room temperature (Liang et al., 2022). According to reports, the virus can live for at least 18 days in air-dried skin, up to 33 days or longer in necrotic skin nodules, and up to 35 days in dry crusts (Guyassa, 2022). This virus can be rendered inactive by heating it to 55°C for two hours and then to 65°C for half an hour. Because the virus remains in lesions or scabs for extended periods, skin lesions are believed to be the primary source of infection (Kumar et al., 2021). Additionally, the virus is expelled from milk (which can infect nursing calves), saliva, semen, nasal secretions, tears, and blood (Ratyotha et al., 2022). In addition to contaminated food and water, LSDV can spread directly in the later stages of the disease through saliva, nasal secretions, and semen, as well as by arthropods, particularly blood-sucking insects (Annandale et al., 2014). Numerous studies have found no positive relationship between infection rates and cattle density, suggesting that indirect transmission of the virus is more significant than direct transmission, at least in the early stages of the disease (Ochwo et al., 2019; Bianchini et al., 2023). This suggests that several vector species, especially blood-sucking insects, are involved in the virus’s dissemination, as the majority of LSD outbreaks take place during the summer when arthropod activity is at its highest. The transmission routes of LSD among cattle are depicted in Figure 2. This explains the disease’s repercussions for populations that are not sick, as well as the vector and nonvector channels that aid in its transmission. Direct contact through body fluids such as the mouth, nose, eyes, milk, semen, and intrauterine fluids is considered nonvector transmission. Indirect transmission occurs through contaminated objects and medical equipment, which can serve as fomites for the virus (Aleksandr et al., 2020). Although sharing feeding and watering facilities can be dangerous, newborn calves are particularly susceptible to infection by coming into contact with their mothers’ contaminated bodily fluids (Eom et al., 2023). LSDV spreads mostly through vector transmission, which is made possible by blood-feeding insects that bite people to spread the virus from infected animals (Sprygin et al., 2019). While the movement of sick livestock can generate outbreaks in new places, such transmission can occur at a limited, localized level within herds or over large territories (Mat et al., 2021). Therefore, these transmission methods increase the amount of LSD that uninfected cow herds are exposed to, creating ongoing problems for disease containment and control. Hard ticks may play a role in the spread of viruses, as reported by several studies (Lubinga et al., 2015; El-Ansary et al., 2022; Zeedan et al., 2024). Hemocytes, salivary glands, and the midgut are among the tick organs that contain LSDV and viral antigens (Lubinga et al., 2014). Additionally, molecular evidence indicates that ticks transmit the virus mechanically and transstadially (El-Ansary et al., 2022). However, the rapid emergence of broad epidemics cannot be explained by their extended attachment to the host. As a result, ticks appear to serve as virus reservoirs (Tuppurainen et al., 2013). The only dipteran that can completely spread the virus to vulnerable animals is Aedes aegypti (Riana et al., 2024). The virus cannot be spread by mosquitoes such as Anopheles stephensi Liston, Culex quinquefasciatus Say, and Culicoides nubeculosus (Chihota et al., 2003). The transmission of LSDV to susceptible animals has not been successful despite the fact that Stomoxys calcitrans has been observed in LSD outbreaks and has infected sheep and goats with the CaPV (Mazloum et al., 2023). Given that LSDV has been found in Culicoides punctatus, it could contribute to the spread of viruses (Paslaru et al., 2022). Additionally, the probability of transmission was reported to be strongly connected with the ratio of biting insects to the host population (Owada et al., 2024). PCR and virus isolation, experimental investigations showed that LSDV was persistent in bovine semen (Irons et al., 2005). Furthermore, the virus is transferred to inseminated heifers through semen (Annandale et al., 2014).
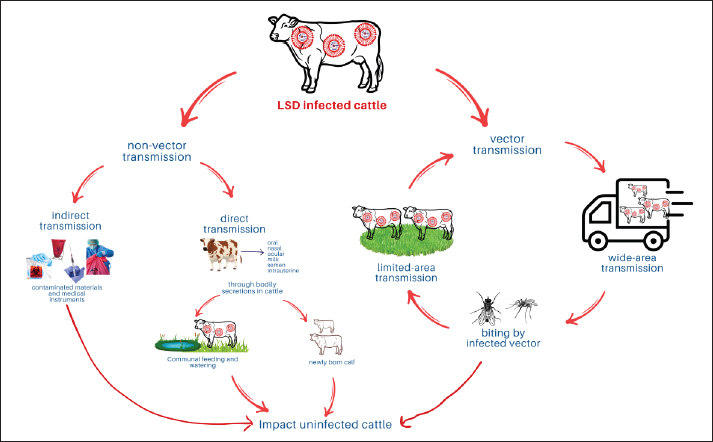
Fig. 2. Transmission pathways of lumpy skin disease virus. Host rangeSusceptible hosts include buffalo (Bubalus bubalis) and cattle (Bos taurus and Bos indicus) (Eom et al., 2023). Native cattle breeds are less vulnerable than B. taurus. All ages of animals are vulnerable, but calves are particularly vulnerable and develop lesions in as little as 24 to 48 hours (Fay et al., 2022). Natural wild animals are immune to infection, but experimental infection causes clinical lesions in Thomson’s gazelle, Arabian oryx (Oryx leucoryx), springbok (Antidorcas marsupialis), giraffe (Giraffe camelopardalis), and impala (A. melampus) (Bianchini et al., 2023). The contribution of wildlife to the spread and upkeep of LSDV is typically small. Additionally, humans are immune to the virus. Risk factorsWarm and humid weather, environments that sustain large populations of vectors, like those following periodic rains, and the addition of additional animals to the herd are risk factors linked to the development of LSD (Issimov et al., 2022). Other risk variables that have been identified as potentially increasing the occurrence of the disease include herd size, vector population, distance to lakes, herd migration, transportation of diseased animals to disease-free areas, shared pastures, and water supplies (Namazi and Tafti, 2021). Furthermore, wind strength and direction probably play a role in the virus’s dissemination (Sprygin et al., 2019). This disease can affect cattle of all sexes, of any age, and of any variety. Furthermore, age, gender, management type, average annual rainfall, and common water supply are risk factors linked to LSDV seropositivity (Hailu et al., 2014). Zoonotic potentialThere is currently no scientific evidence that a skin condition characterized by bumps is a zoonotic illness that can infect humans or animals. LSDV can spread to nursing calves during the infectious phase by contaminated milk or skin sores on the teats (Rittipornlertrak et al., 2024). Nevertheless, there is no evidence that milk can infect people with LSDV. However, to prevent many other foodborne diseases, it is advised to adhere to fundamental food hygiene rules, such as properly boiling milk (Gallo et al., 2020). Economic impactNumerous significant sectors and businesses have documented the direct or indirect economic impact of LSD. In South Asia, home to 21% of the world’s dairy herd, the most obvious and significant effect directly associated with LSD is a severe drop in milk production (Vinitchaikul et al., 2023). A study conducted in Turkey found that the average milk output of impacted cows decreased by 159 liters per lactation (Tuppurainen and Oura, 2012). Although there is a chance that meat will contract secondary bacterial infections, meat from cattle infected with LSDV is not forbidden from entering the food chain. In Ethiopia, LSDV infection has been linked to estimated yearly decreases in beef production of 1.2% and 6.2% among native breeds and Friesian cattle, respectively (Molla et al., 2017). Furthermore, a raw cowhide’s value may be lowered by any cracks, lesions, or scars, as is the situation with hides from animals exposed to high levels of LSD (Khattab et al., 2022). The high quality of Bangladeshi cowhide is well known, and the country exports 3.5% of its leather annually, with 56% of the leather coming from cows (Hasib et al., 2021). The same is true for India, which ranks ninth globally in exports and generates $8,500 million annually from the sale of leather and cowhide goods (Kumar and Tripathi, 2022). Lameness and fever made it impossible to use animals for forced labor. Reduced conception rates can arise from artificial insemination of breeding cattle with diseased bull semen, which can spread LSD (Givens, 2018). Additionally, a number of health issues that affect bulls, such as mastitis, orchitis, abortion, and infertility, result in significant financial losses for ranch owners. LSD’s indirect economic effects of LSDs are estimated for culling, trade restrictions, vaccination, quarantine, and treatment expenses, feed and labor costs, and agricultural biosecurity (Molla et al., 2017). In addition to the lengthy fattening period, farm owners must incur additional expenses for feed supplements for ailing animals during the recuperation phase (Das et al., 2021). The projected cost of treating afflicted livestock with broad-spectrum antibiotics and anti-inflammatory medications for LSD in Jordan is $35.04 (Abutarbush et al., 2015). Large numbers of afflicted animals may occasionally need to be put down, as was the case in Greece and Bulgaria, where Bulgaria experienced the worst economic catastrophe, with each herd costing approximately US$8,000 (Agianniotaki et al., 2017; Casal et al., 2018). Because LSD is a transboundary infectious disease, there is a good chance that it will spread quickly through the channels of production and commercialization (Eom et al., 2023). Taking into account the degree of culling and the quantity of bulls at risk, a risk assessment study for LSD carried out at the Ethiopian bull market calculated the financial loss to be US$667,785.6 (Alemayehu et al., 2013). Cost-effective quarantine is not always the best option for MLPs. Manual work, animal nutrition, diagnostic testing, disposing of positive test findings, and other expenses are all included in the estimated $145,000 cost of quarantine in the United States (Eom et al., 2023). TreatmentSince there is no effective antiviral medication to treat LSD, the only viable method of disease containment is vaccination. In epidemic settings, LSD prophylaxis is rarely tried outside of symptomatic and supportive treatments like antibiotics to prevent secondary bacterial infections in abraded skin and wound healing sprays (Datten et al., 2023). Although little feedback exists, intravenous hydration treatment and anti-inflammatory medications may be administered to boost hunger (Liang et al., 2022). VaccinationVaccination is a successful and efficient way to manage LSD. According to Tuppurainen et al. (2020), vaccination is the only method that can stop the spread of the LSD virus in both endemic and newly infected areas. The majority of LSD vaccines currently on the market are live attenuated vaccines based on strains of the LSD virus, goatpox virus (GTPV), or sheeppox virus (SPPV) (Vandenbussche et al., 2022). On the other hand, the first inactivated vaccination was just released. Compared with live attenuated LSD virus vaccines, GTPV/SPPV-based vaccines are less effective; nevertheless, they do not result in fever or the development of clinical symptoms following vaccination (Sprygin et al., 2020). Although live vaccinations help prevent disease and can generate a robust and sustained immune response, they can also result in modest symptoms, including skin lesions and local inflammation (Datten et al., 2023). Using live attenuated vaccines that are either homologous (the Neethling strain of LSDV) or heterologous (sheeppox virus or goatpox virus), animals can be protected against LSDV infection due to the genus CaPV’s known ability to provide cross-protection (Morgenstern and Klement, 2020). The following vaccine strains are commercially available: goat pox vaccine virus (KSGP) O-240 and O-180, Yugoslavian sheep pox (SPP) virus RM65, CaPV, which include the Romanian SPP virus, Kenyan goat and sheep pox viruses, and Neethling virus LSD strain (Tuppurainen et al., 2014). According to Tekilegiorgis and Tamir (2019), the SPPV vaccine in the Middle East and Horn of Africa had unfavorable effects on cattle following vaccination and only partially protected them. In contrast, Gari et al. (2015) showed that neither the Kenyan goat and sheep pox vaccination (strain vaccine (KSGP) O-180) nor the Ethiopian Neethling virus vaccine could protect cattle against LSD. Meanwhile, cows can be protected from LSD by goat pox. These findings, however, are in contrast to those of a study by Hakobyan et al. (2023), which found that the heterologous vaccine (sheep poxvirus) used in Armenia protects 86.09% of the cow population and has no negative effects on the cattle. Vaccine failure has been attributed to a number of factors, including low vaccine titers, vaccination of animals that are experiencing a temporary incubation period for the disease, handling, and storage errors, and differences between the vaccine strain and the virus strain that infects animals in the field (Bamouh et al., 2021). ControlLSDs can be prevented from spreading by implementing stringent quarantine regulations, vector management strategies, and restrictions on cattle movement (Akther et al., 2023). Vectors can contribute to the mechanical spread of illness; in fact, blood-sucking insect vectors are mostly responsible for the dissemination of LSDV; hence, it is important to regulate their reproduction and spread (Sprygin et al., 2019). Vector control techniques include the use of ectoparasites, pesticides, and vector traps (Socha et al., 2022). According to Punyapornwithaya et al. (2023), effective vector management can mechanically reduce disease transmission rates; however, this is not the case for large-scale livestock management. Additionally, widespread pesticide use may upset the natural equilibrium and harm beneficial insects like honey bees (Tudi et al., 2021). As a result, environmentally friendly vector control measures must be taken, such as reducing vector reproduction by cleaning up excrement and preventing water collection in the cage and its surroundings and members (Wilson et al., 2020). In addition to vectors, contaminated equipment and animals with disease can spread disease (Akther et al., 2023). It is possible to confine the animal population and sterilize the cages and equipment more thoroughly before an outbreak occurs. The spread of LSDs can be stopped by limiting animal movements or requiring animals to be quarantined before being imported for a minimum of 3–4 weeks (European Food Safety Authority et al., 2018). ConclusionLSD poses a significant economic threat to the cattle industry because of its impact on animal health and productivity, necessitating urgent attention to its management and prevention strategies. Understanding the disease’s transmission and clinical manifestations and effective control measures are crucial for safeguarding livestock and mitigating its widespread effects. Future worksThe scientific community has taken notice of recurrent LSD breakouts in susceptible areas. Therefore, the widespread implementation of emergency preparedness is necessary to stop the spread of this cross-border disease. To prevent the onset and spread of infectious diseases, attention should be paid to vector control, movement limitations, stringent quarantine, improved vaccination programs, appropriate animal care, and general farm cleanliness management. Therefore, more research is required to determine the origin of the infection, identify the causative agent through genetic detection and characterization, and then examine the ecology and epidemiology of LSDV. AcknowledgmentsThe authors would like to thank the Lembaga Penelitian dan Pengabdian Masyarakat (LPPM) Universitas Airlangga and all the parties who have helped in the preparation of this article. Conflict of interestThe authors declare no conflict of interest. FundingThe authors thank Universitas Airlangga for funding support (Grant number 397/UN3.14/PT/2020). Author’s contributionsNH, ARK, and RD drafted the manuscript. IBM, DAAK, and MGAY revised and edited the manuscript. EPH, BWKW, and SW participated in preparing and critical checking of the manuscript. ANN, RZA, and MAMH edited the manuscript. All authors have read and approved the final manuscript. Data availabilityAll references are open-access, so data can be obtained from the online web. ReferencesAbutarbush, S.M., Ababneh, M.M., Al Zoubi, I.G., Al Sheyab, O.M., Al Zoubi, M.G., Alekish, M.O. and Al Gharabat, R.J. 2015. Lumpy skin disease in Jordan: disease emergence, clinical signs, complications and preliminary-associated economic losses. Transbound. Emerg. Dis. 62(5), 549–554. Adamu, K., Abayneh, T., Getachew, B., Mohammed, H., Deresse, G., Zekarias, M., Chala, W. and Gelaye, E. 2024. Lumpy skin disease virus isolation, experimental infection, and evaluation of disease development in calf. Sci. Rep. 14(1), 20460. Agianniotaki, E.I., Tasioudi, K.E., Chaintoutis, S.C., Iliadou, P., Mangana-Vougiouka, O., Kirtzalidou, A., Alexandropoulos, T., Sachpatzidis, A., Plevraki, E., Dovas, C.I. and Chondrokouki, E. 2017. Lumpy skin disease outbreaks in Greece during 2015-16, implementation of emergency immunization and genetic differentiation between field isolates and vaccine virus strains. Vet. Microbiol. 201(1), 78–84. Akther, M., Akter, S.H., Sarker, S., Aleri, J.W., Annandale, H., Abraham, S. and Uddin, J.M. 2023. Global burden of lumpy skin disease, outbreaks, and future challenges. Viruses 15(9), 1861. Al-Salihi, K.A. 2014. Lumpy skin disease: review of literature. Mirror Res. Vet. Sci. Anim. 3(3), 6–23. Aleksandr, K., Olga, B., David, W.B., Pavel, P., Yana, P., Svetlana, K., Alexander, N., Vladimir, R., Dmitriy, L. and Alexander, S. 2020. Nonvector-borne transmission of lumpy skin disease virus. Sci. Rep. 10(1), 7436. Alemayehu, G., Zewde, G. and Admassu, B. 2013. Risk assessments of lumpy skin diseases in the Borena bull market chain and their implications for livelihoods and international trade. Trop. Anim. Health Prod. 45(5), 1153–1159. Alemu, T.Z. 2024. Review on epidemiology and diagnosis of lumpy skin disease. J. Vet. Med. Anim. Sci. 7(1), 1138. Ali, B.H. and Obeid, H.M. 1977. Investigation of the first outbreaks of lumpy skin disease inSudann. Br Vet. J. 133(2), 184–189. Alkhamis, M.A. and VanderWaal, K. 2016. Spatial and temporal epidemiology of lumpy skin disease in the Middle East, 2012-2015. Front. Vet. Sci. 3(1), 19. Allepuz, A., Casal, J. and Beltrán-Alcrudo, D. 2019. Spatial analysis of lumpy skin disease in Eurasia-predicting areas at risk of further spread within the region. Transbound. Emerg. Dis. 66(2), 813–822. Amin, D.M., Shehab, G., Emran, R., Hassanien, R.T., Alagmy, G.N., Hagag, N.M., Abd-El-Moniem, M.I.I., Habashi, A.R., Ibraheem, E.M. and Shahein, M.A. 2021. Diagnosis of naturally occurring lumpy skin disease virus infection in cattle using virological, molecular, and immunohistopathological assays. Vet. World 14(8), 2230–2237. Annandale, C.H., Holm, D.E., Ebersohn, K. and Venter, E.H. 2014. Seminal transmission of lumpy skin disease virus in heifers. Transbound. Emerg. Dis. 61(5), 443–448. Anwar, A., Na-Lampang, K., Preyavichyapugdee, N. and Punyapornwithaya, V. 2022. Lumpy skin disease outbreaks in Africa, Europe, and Asia (2005-2022): multiple change point analysis and time series forecast. Viruses 14(10), 2203. Azeem, S., Sharma, B., Shabir, S., Akbar, H. and Venter, E. 2022. Lumpy skin disease is expanding its geographic range: a challenge for Asian livestock management and food security. Vet. J. 279(1), 105785. Bamouh, Z., Hamdi, J., Fellahi, S., Khayi, S., Jazouli, M., Tadlaoui, K.O., Fihri, O.F., Tuppurainen, E. and Elharrak, M. 2021. Post-vaccination reactions of two live attenuated vaccines against lumpy skin disease of cattle. Vaccines 9(6), 621. Bianchini, J., Simons, X., Humblet, M.F. and Saegerman, C. 2023. Lumpy skin disease: a systematic review of mode of transmission, risk of emergence and risk entry pathway. Viruses 15(8), 1622. Byadovskaya, O., Prutnikov, P., Shalina, K., Babiuk, S., Perevozchikova, N., Korennoy, F., Chvala, I., Kononov, A. and Sprygin, A. 2022. Changes in the epidemiology of lumpy skin disease in Russia since its first introduction from 2015 to 2020. Transbound. Emerg. Dis. 69(5), e2551–e2562. Casal, J., Allepuz, A., Miteva, A., Pite, L., Tabakovsky, B., Terzievski, D., Alexandrov, T. and Beltrán-Alcrudo, D. 2018. Economic cost of lumpy skin disease outbreaks in three Balkan countries: Albania, Bulgaria and the Former Yugoslav Republic of Macedonia (2016-2017). Transbound. Emerg. Dis. 65(6), 1680–1688. Chapman, R., van Diepen, M., Douglass, N., Galant, S., Jaffer, M., Margolin, E., Ximba, P., Hermanus, T., Moore, P.L. and Williamson, A.L. 2021. Assessment of an LSDV-vectored vaccine for heterologous prime-boost immunization against HIV. Vaccines (Basel) 9(11), 1281. Chihota, C.M. Rennie, L.F. Kitching, R.P. and Mellor, P.S. 2003. Attempted mechanical transmission of lumpy skin disease virus by biting insects. Med. Vet. Entomol. 17(3), 294–300. Das, M., Chowdhury, S.R., Akter, S., Mondal, A.K., Uddin, J., Rahman, M. and Rahman, M. 2021. An updated review of lumpy skin disease: perspective of Southeast Asian countries. J. Adv. Biotechnol. Exp. Ther. 4(3), 322–333. Datten, B., Chaudhary, A.A., Sharma, S., Singh, L., Rawat, K.D., Ashraf, M.S., Alneghery, L.M., Aladwani, M.O., Rudayni, H.A., Dayal, D., Kumar, S. and Chaubey, K.K. 2023. Extensive examination of the significance, symptoms, diagnosis, available therapies, and prognosis of lumpy skin disease. Viruses 15(3), 604. Davies, F.G. 1982. Observations on the epidemiology of lumpy skin disease in Kenya. J. Hyg. (Lond) 88(1), 95–102. Davis, S.K., Jia, F., Wright, Q.G., Islam, M.T., Bean, A., Layton, D., Williams, D.T. and Lynch, S.E. 2024. Correlations of protection for mammalian livestock vaccines against high-priority viral diseases. Front. Immunol. 15(1), 1397780. Dhanda, S., Sharma, D.K., Kamboj, H., Kumar, G., Mittal, P., Kumar, R., Verma, A., Rathore, K., Gaur, M., Barua, S., Tripathi, B.N., Sharma, S. and Kumar, N. 2024. Evaluation of the immune responses in buffaloes vaccinated with a live-attenuated lumpy skin disease vaccine (Lumpi-ProVacInd). Trop. Anim. Health Prod. 56(7), 226. Di Felice, E., Pinoni, C., Rossi, E., Amatori, G., Mancuso, E., Iapaolo, F., Taraschi, A., Di Teodoro, G., Di Donato, G., Ronchi, G.F., Mercante, M.T., Di Ventura, M., Morelli, D. and Monaco, F. 2024. Susceptibility of the mediterranean buffalo (Bubalus bubalis) following experimental infection with lumpy skin disease virus. Viruses 16(3), 466. Di Giuseppe, A., Zenobio, V., Dall’Acqua, F., Di Sabatino, D. and Calistri, P. 2024. Lumpy Skin Disease. Vet. Clin. North Am. Food Anim. Pract. 40(2), 261–276. Dubey, A., Ghosh, N.S., Gupta, A. and Singh, S. 2023. A review of current epidemiology and molecular studies of lumpy skin disease virus—an emerging worldwide threat to domestic animals. J. Med. Pharm. Allied Sci. 12(1), 5635–5643. El-Ansary, R.E., El-Dabae, W.H., Bream, A.S. and El Wakil, A. 2022. Isolation and molecular characterization of lumpy skin disease virus from hard ticks, Rhipicephalus (Boophilus) annulatus in Egypt. BMC Vet. Res. 18(1), 302. Elhaig, M.M., Selim, A. and Mahmoud, M. 2017. Lumpy skin disease in cattle: frequency of occurrence in a dairy farm and a preliminary assessment of its possible impact on Egyptian buffaloes. Onderstepoort. J. Vet. Res. 84(1), e1–e6. Eom, H.J., Lee, E.S. and Yoo, H.S. 2023. Lumpy skin disease as an emerging infectious disease. J. Vet. Sci. 24(3), e42. European Food Safety Authority (EFSA). 2018. Lumpy skin disease II. Data collection and analysis. EFSA J. 16(2), e05176. European Food Safety Authority (EFSA), Calistri, P., DeClercq, K., De Vleeschauwer, A., Gubbins, S., Klement, E., Stegeman, A., Abrahantes, J.C., Antoniou, S.E., Broglia, A. and Gogin, A. 2018. Lumpy skin disease: scientific and technical assistance in the control and surveillance activities. EFSA J. 16(10), e05452. Fay, P.C., Wijesiriwardana, N., Munyanduki, H., Sanz-Bernardo, B., Lewis, I., Haga, I.R., Moffat, K., van Vliet, A.H.M., Hope, J., Graham, S.P. and Beard, P.M. 2022. The immune response to lumpy skin disease virus in cattle is influenced by the inoculation route. Front. Immunol. 13(1), 1051008. Gallo, M., Ferrara, L., Calogero, A., Montesano, D. and Naviglio, D. 2020. Relationships between food and diseases: what to know to ensure food safety. Food Res. Int. 137(1), 109414. Gari, G., Abie, G., Gizaw, D., Wubete, A., Kidane, M., Asgedom, H., Bayissa, B., Ayelet, G., Oura, C.A., Roger, F. and Tuppurainen, E.S. 2015. Evaluation of the safety, immunogenicity, and efficacy of three capripoxvirus vaccine strains against lumpy skin disease virus. Vaccine 33(28), 3256–3261. Gari, G., Grosbois, V., Waret-Szkuta, A., Babiuk, S., Jacquiet, P. and Roger, F. 2012. Lumpy skin disease in Ethiopia: a seroprevalence study across different agro-climate zones. Acta Trop. 123(2), 101–106. Gharban, H.A.J., Al-Shaeli, S.J.J., Al-Fattli, H.H.H. and Altaee, M.N.K. 2019. Molecular and histopathological confirmation of clinically diagnosed lumpy skin disease in cattle, Baghdad Province, Iraq. World 12(11), 1826–1832. Givens, M.D. 2018. Review: risks of disease transmission through semen in cattle. Animal 12(s1): s165–s171. Gupta, T., Patial, V., Bali, D., Angaria, S., Sharma, M. and Chahota, R. 2020. A review: Lumpy skin disease and its emergence in India. Vet. Res. Commun. 44(1), 111–118. Guyassa, C. 2022. Epidemiology and diagnostic methods of lumpy skin disease: a short review. Int. J. Vet. Sci. Res. 8(2), 64–70. Haegeman, A., De Leeuw, I., Mostin, L., Van Campe, W., Philips, W., Elharrak, M., De Regge, N. and De Clercq, K. 2023. Duration of immunity after vaccination of cattle with a live attenuated or inactivated lumpy skin disease virus vaccine. Microorganisms 11(1), 210. Hailu, B., Tolosa, T., Gari, G., Teklue, T. and Beyene, B. 2014. Estimated prevalence and risk factors associated with clinical Lumpy skin disease in north-eastern Ethiopia. Prev. Vet. Med. 115(1–2), 64–68. Hakobyan, V., Sargsyan, K., Kharatyan, S., Elbakyan, H., Sargsyan, V., Markosyan, T., Vardanyan, T., Badalyan, M. and Achenbach, J.E. 2023. The serological response in cattle following administration of a heterologous sheep pox virus strain vaccine for protection from lumpy skin disease; current situation in Armenia. Vet. Sci. 10(2), 102. Hasib, F.M.Y., Islam, M.S., Das, T., Rana, E.A., Uddin, M.H., Bayzid, M., Nath, C., Hossain, M.A., Masuduzzaman, M., Das, S. and Alim, M.A. 2021. Lumpy skin disease outbreak in cattle population of Chattogram, Bangladesh. Vet. Med. Sci. 7(5), 1616–1624. Irons, P.C., Tuppurainen, E.S. and Venter, E.H. 2005. Excretion of lumpy skin disease virus in bull semen. Theriogenology 63(5), 1290–1297. Issimov, A., Kushaliyev, K., Abekeshev, N., Molla, W., Rametov, N., Bayantassova, S., Zhanabayev, A., Paritova, A., Shalmenov, M., Ussenbayev, A., Kemeshov, Z., Baikadamova, G. and White, P. 2022. Risk factors associated with lumpy skin disease in cattle in West Kazakhstan. Prev. Vet. Med. 207(1), 105660. Khan, H.M., Sajid, H.B., Manan, A., Awan, M.H., Hussain, I. and Raheem, M.A. 2023. A systematic review of lumpy skin disease virus (LSDV) and its emergence in Pakistan. Middle East J. Appl. Sci. Technol. 6(3), 19–30. Khattab, M.S., Osman, A.H., AbuBakr, H.O., Azouz, R.A., Salem, N.Y. and Farag, H.S. 2022. Economic effects of lumpy skin lesions on cowhide industry in Egypt. Int. J. Vet. Sci. Med. Diagn. 3(2), 123. Krešić, N., Šimić, I., Bedeković, T., Acinger-Rogić, Ž. and Lojkić, I. 2020. Evaluation of serological tests for detection of antibodies against lumpy skin disease virus. J. Clin. Microbiol. 58(9), e00348-20. Kumar, N., Chander, Y., Kumar, R., Khandelwal, N., Riyesh, T., Chaudhary, K., Shanmugasundaram, K., Kumar, S., Kumar, A., Gupta, M.K., Pal, Y., Barua, S. and Tripathi, B.N. 2021. Isolation and characterization of lumpy skin disease virus from cattle in India. PLoS One 16(1), e0241022. Kumar, N. and Tripathi, B.N. 2022. A severe skin virus epidemic sweeping through the Indian subcontinent is a threat to the livelihood of farmers. Virulence 13(1), 1943–1944. Li, Y., An, Q., Sun, Z., Gao, X. and Wang, H. 2023. Risk factors and spatiotemporal distribution of lumpy skin disease occurrence in the asian continent during 2012–2022: an ecological niche model. Transbound. Emerg. Dis. 2023(1), 6207149. Liang, Z., Yao, K., Wang, S., Yin, J., Ma, X., Yin, X., Wang, X. and Sun, Y. 2022. Understanding research advances in lumpy skin disease: a comprehensive literature review of experimental evidence. Front. Microbiol. 13(1), 1065894. Lu, G., Xie, J., Luo, J., Shao, R., Jia, K. and Li, S. 2021. Lumpy skin disease outbreaks in China since 3 August 2019. Transbound. Emerg. Dis. 68(2), 216–219. Lubinga, J.C., Clift, S.J., Tuppurainen, E.S., Stoltsz, W.H., Babiuk, S., Coetzer, J.A. and Venter, E.H. 2014. Immunohistochemical demonstration of lumpy skin disease virus infection in Amblyomma hebraeum and Rhipicephalus appendiculatus ticks using immunohistochemistry. Ticks Tick Borne Dis. 5(2), 113–120. Lubinga, J.C., Tuppurainen, E.S., Mahlare, R., Coetzer, J.A., Stoltsz, W.H. and Venter, E.H. 2015. Evidence of transstadial and mechanical transmission of lumpy skin disease virus by Amblyomma hebraeum ticks. Transbound. Emerg. Dis. 62(2), 174–182. Mafirakureva, P., Saidi, B. and Mbanga, J. 2017. Incidence and molecular characterization of lumpy skin disease virus in Zimbabwe using the P32 gene. Trop Anim Health Prod. 49(1), 47–54. Manjunathareddy, G.B., Saminathan, M., Sanjeevakumar, L., Rao, S., Dinesh, M., Dhama, K., Singh, K.P. and Tripathi, B.N. 2024. Pathological, immunological, and molecular epidemiological analysis of lumpy skin disease virus in Indian cattle during a high-mortality epidemic. Vet. Q. 44(1), 1–22. Mat, B., Arikan, M.S., Akin, A.C., Çevrimli, M.B., Yonar, H. and Tekindal, M.A. 2021. Determination of production losses related to lumpy skin disease among cattle in Turkey and analysis using the SEIR epidemic model. BMC Vet. Res. 17(1), 300. Mazloum, A., Van Schalkwyk, A., Babiuk, S., Venter, E., Wallace, D.B. and Sprygin, A. 2023. Lumpy skin disease: history, current understanding, and research gaps in the context of recent geographic expansion. Front. Microbiol. 14(1), 1266759. Milovanović, M., Dietze, K., Milićević, V., Radojičić, S., Valčić, M., Moritz, T. and Hoffmann, B. 2019. Humoral immune response to repeated lumpy skin disease virus vaccination and serological test performance. BMC Vet. Res. 15(1), 80. Moda, M., Sharma, N., Thakur, M., Sharma, I. and Sharma, K. 2024. A Overview on Lumpy Skin Disease: A Review. Asian J. Res. Pharm. Sci. 14(1), 43–50. Modise, B.M., Settypalli, T.B.K., Kgotlele, T., Xue, D., Ntesang, K., Kumile, K., Naletoski, I., Nyange, J.F., Thanda, C., Macheng, K.N., Marobela-Raborokgwe, C., Viljoen, G.J., Cattoli, G. and Lamien, C.E. 2021. First molecular characterization of poxviruses in cattle, sheep, and goats in Botswana. Virol. J. 18(1), 167. Molla, W., de Jong, M.C.M., Gari, G. and Frankena, K., 2017. Economic impact of lumpy skin disease and cost effectiveness of vaccination for the control of outbreaks in Ethiopia. Prev. Vet. Med. 147(1), 100–107. Morgenstern, M. and Klement, E. 2020. The effect of vaccination with a live-attenuated Neethling lumpy skin disease vaccine on milk production and mortality-an analysis of 77 dairy farms in Israel. Vaccines (Basel) 8(2), 324. Moudgil, G., Chadha, J., Khullar, L., Chhibber, S. and Harjai, K. 2024. Lumpy skin disease: insights into the current status and geographical expansion of a transboundary viral disease. Microb. Pathog. 186(1), 106485. Murti, N.T., Safitri, E., Srianto, P., Madyawati, S.P. and Rofikoh, A.R. 2024. Prevalence and progression of lumpy skin disease in cattle over six months leading up to Eid al-Adha in 2023 in the Cirebon District of West Java Province, Indonesia. IOP Conf. Ser. Earth Environ. Sci. 1292(1), 012037. Namazi, F. and Tafti, A.K. 2021. Lumpy skin disease, an emerging transboundary viral disease: a review. Vet. Med. Sci. 7(3), 888–896. Neamat-Allah, A.N. 2015. Immunological, hematological, biochemical, and histopathological studies on cows naturally infected with lumpy skin disease. Vet. World 8(9), 1131–1136. Ntombela, N., Matsiela, M., Zuma, S., Hiralal, S., Naicker, L., Mokoena, N. and Khoza, T. 2023. Production of recombinant lumpy skin disease virus A27L and L1R proteins for diagnostics and vaccine development. Vaccine X 15(1), 100384. Ochwo, S., VanderWaal, K., Munsey, A., Nkamwesiga, J., Ndekezi, C., Auma, E. and Mwiine, F.N. 2019. Seroprevalence and risk factors for lumpy skin disease virus seropositivity in cattle in Uganda. BMC Vet. Res. 15(1), 236. Owada, K., Mahony, T.J., Ambrose, R.K., Hayes, B.J. and Magalhães, R.J.S. 2024. Epidemiological risk factors and modeling approaches for risk assessment of lumpy skin disease virus introduction and spread: methodological review and implications for risk-based surveillance in Australia. Transbound. Emerg. Dis. 2024(1), 3090226. Padha, S. and Sofi, O.M. 2023. Lumpy skin disease as an alarming transboundary disease: implications and consequences. Indian J. Anim. Health 62(2), 257–261. Pandey, N., Hopker, A., Prajapati, G., Rahangdale, N., Gore, K. and Sargison, N. 2022. Observations of presumptive lumpy skin disease in native cattle and Asian water buffaloes around the tiger reserves of the central Indian highlands. N. Z. Vet. J. 70(2), 101–108. Parvin, R., Chowdhury, E.H., Islam, M.T., Begum, J.A., Nooruzzaman, M., Globig, A., Dietze, K., Hoffmann, B. and Tuppurainen, E. 2022. Clinical epidemiology, pathology, and molecular investigation of lumpy skin disease outbreaks in Bangladesh during 2020-2021 indicate the re-emergence of an old African strain. Viruses 14(11), 2529. Paslaru, A.I., Maurer, L.M., Vögtlin, A., Hoffmann, B., Torgerson, P.R., Mathis, A. and Veronesi, E. 2022. Putative roles of mosquitoes (Culicidae) and biting midges (Culicoides spp.) as mechanical or biological vectors of lumpy skin disease virus. Med. Vet. Entomol. 36(3), 381–389. Podshibyakin, D., Padilo, L., Agoltsov, V., Chernykh, O., Popova, O., Mutalif, K. and Solotova, N. 2024. Analysis of environmental factors influencing the seasonality of lumpy skin disease outbreaks and assessment of the risk of spread in the Saratovskaya oblast of Russia. Vet. World 17(3), 630–644. Punyapornwithaya, V., Salvador, R., Modethed, W., Arjkumpa, O., Jarassaeng, C., Limon, G. and Gubbins, S. 2023. Estimating transmission kernel for lumpy skin disease virus from outbreak data in Thailand in 2021. Viruses 15(11), 2196. Ratyotha, K., Prakobwong, S. and Piratae, S. 2022. Lumpy skin disease: a newly emerging disease in Southeast Asia. Vet. World 15(12), 2764–2771. Ren, S., Chen, H., Yuan, L., Yang, X., Afera, T.B., Rehman, Z.U., Wang, H., Wang, X., Ma, C., Lin, Y., Qiu, X., Yin, X. and Sun, Y. 2023. Phylogenetic and pathogenic characterization of lumpy skin disease virus circulating in China. Virology 585(1), 127–138. Rhazi, H., Safini, N., Mikou, K., Alhyane, M., Lenk, M., Tadlaoui, K.O. and Elharrak M. 2021. Comparative sensitivity study of primary cells, vero, OA3.Ts, and ESH-L cell lines to lumpy skin disease, sheeppox, and goatpox virus detection and growth. J. Virol. Methods 293(1), 114164. Riana, E., Sri-In, C., Songkasupa, T., Bartholomay, L.C., Thontiravong, A. and Tiawsirisup, S. 2024. Infection, dissemination, and transmission of lumpy skin disease virus in Aedes aegypti (Linnaeus), Culex tritaeniorhynchus (Giles), and Culex quinquefasciatus (Say) mosquitoes. Acta Trop. 254(1), 107205. Rittipornlertrak, A., Modethed, W., Sangkakam, K., Muenthaisong, A., Vinitchaikul, P., Boonsri, K., Pringproa, K., Punyapornwithaya, V., Kreausukon, K., Sthitmatee, N. and Singhla, T. 2024. Persistence of passive immunity in calves receiving colostrum from cows vaccinated with a live attenuated lumpy skin disease vaccine and the performance of serological tests. Front. Vet. Sci. 11(1), 1303424. Rouby, S. and Aboulsoud, E. 2016. Evidence of intrauterine transmission of lumpy skin disease virus. Vet. J. 209(1), 193–195. Rouby, S.R. Ghonaim, A.H. Chen, X. and Li, W. 2024. The current epizootiological situation of three major viral infections affecting cattle in Egypt. Viruses 16(10), 1536. Rouby, S.R., Safwat, N.M., Hussein, K.H., Abdel-Ra’ouf, A.M., Madkour, B.S., Abdel-Moneim, A.S. and Hosein, H.I. 2021. Lumpy skin disease outbreaks in Egypt during 2017-2018 among sheeppox vaccinated cattle: epidemiological, pathological, and molecular findings. PLoS One 16(10), e0258755. Saltykov, Y.V. Kolosova, A.A. and Feodorova, V.A. 2022. Update on lumpy skin disease: emergence in Asian part of Eurasia. Acta Vet. 72(3), 287–299. Sanz-Bernardo, B., Haga, I.R., Wijesiriwardana, N., Hawes, P.C., Simpson, J., Morrison, L.R., MacIntyre, N., Brocchi, E., Atkinson, J., Haegeman, A., De Clercq, K., Darpel, K.E. and Beard, P.M. 2020. Lumpy skin disease is characterized by severe multifocal dermatitis with necrotizing fibrinoid vasculitis following experimental infection. Vet. Pathol. 57(3), 388–396. Schlosser-Perrin, L., Holzmuller, P., Fernandez, B., Miotello, G., Dahmani, N., Neyret, A., Bertagnoli, S., Armengaud, J. and Caufour, P. 2023. Constitutive proteins of lumpy skin disease virion assessed by next-generation proteomics. J. Virol. 97(10), e0072323. Sendow, I., Meki, I.K., Dharmayanti, N.L.P.I., Hoerudin, H., Ratnawati, A., Settypalli, T.B.K., Ahmed, H.O., Nuradji, H., Saepulloh, M., Adji, R.S., Fairusya, N., Sari, F., Anindita, K., Cattoli, G. and Lamien, C.E. 2024. Molecular characterization of recombinant LSDV isolates from the 2022 outbreak in Indonesia through phylogenetic networks and whole-genome SNP-based analysis. BMC Genomics 25(1), 240. Senthilkumar, C., Sindhu, C., Vadivu, G. and Neethirajan, S. 2024. Early detection of lumpy skin disease in cattle using deep learning—a comparative analysis of pretrained models. Vet. Sci. 11(10), 510. Shumilova, I., Sprygin, A., Mazloum, A., Pronin, V., Byadovskaya, O., Babiuk, S., Donnik, I. and Chvala, I. 2023. Comparison of gross pathology between classical and recombinant lumpy skin disease viruses. Viruses 15(9), 1883. Socha, W., Kwasnik, M., Larska, M., Rola, J. and Rozek, W. 2022. Vector-Borne viral diseases as a current threat for human and animal health-one health perspective. J. Clin. Med. 11(11), 3026. Sprygin, A., Pestova, Y., Bjadovskaya, O., Prutnikov, P., Zinyakov, N., Kononova, S., Ruchnova, O., Lozovoy, D., Chvala, I. and Kononov, A. 2020. Evidence of the recombination of vaccine strains of lumpy skin disease virus with field strains, causing disease. PLoS One 15(5), e0232584. Sprygin, A., Pestova, Y., Wallace, D.B., Tuppurainen, E. and Kononov, A.V. 2019. Transmission of lumpy skin disease virus: a short review. Virus Res. 269(1), 197637. Sukoco, H., Fahrodi, D.U., Said, N.S., Marsudi, Irfan, M., Salmin, Wahyuni, S. and Hardyanti, K. 2023. Lumpy skin disease (LSD): etiology, pathogenesis, prevention and control. J. Educ. Technol. Inf. Soc. Sci. Health 2(1), 549–560. Tageldin, M.H., Wallace, D.B., Gerdes, G.H., Putterill, J.F., Greyling, R.R., Phosiwa, M.N., Al Busaidy, R.M. and Al Ismaaily, S.I. 2014. Lumpy skin disease of cattle: an emerging problem in the Sultanate of Oman. Trop. Anim. Health Prod. 46(1), 241–246. Tekilegiorgis, T. and Tamir, D. 2019. Review of alternative LSD vaccines. Apprch. Poult. Dairy Vet. Sci. 7(2), 618–619. Tudi, M., Ruan, H.D., Wang, L., Lyu, J., Sadler, R., Connell, D., Chu, C. and Phung, D.T. 2021. Agriculture development, pesticide application, and its impact on the environment. Int. J. Environ. Res. Public Health 18(3), 1112. Tulman, E.R., Afonso, C.L., Lu, Z., Zsak, L., Kutish, G.F. and Rock, D.L. 2001. Genome of lumpy skin disease virus. J. Virol. 75(15), 7122–7130. Tulman, E.R., Afonso, C.L., Lu, Z., Zsak, L., Sur, J.H., Sandybaev, N.T., Kerembekova, U.Z., Zaitsev, V.L., Kutish, G.F. and Rock, D.L. 2002. The genomes of sheeppox and goatpox viruses. J. Virol. 76(12), 6054–6061. Tuppurainen, E., Dietze, K., Wolff, J., Bergmann, H., Beltran-Alcrudo, D., Fahrion, A., Lamien, C.E., Busch, F., Sauter-Louis, C., Conraths, F.J., De Clercq, K., Hoffmann, B. and Knauf, S. 2021. Review: vaccines and treatments for lumpy skin disease. Vaccines (Basel) 9(10), 1136. Tuppurainen, E.S., Lubinga, J.C., Stoltsz, W.H., Troskie, M., Carpenter, S.T., Coetzer, J.A., Venter, E.H. and Oura, C.A. 2013. Mechanical transmission of lumpy skin disease virus by Rhipicephalus appendiculatus male ticks. Epidemiol. Infect. 141(2), 425–430. Tuppurainen, E.S. and Oura, C.A. 2012. Review: lumpy skin disease: an emerging threat to Europe, the Middle East and Asia. Transbound. Emerg. Dis. 59(1), 40–48. Tuppurainen, E.S., Pearson, C.R., Bachanek-Bankowska, K., Knowles, N.J., Amareen, S., Frost, L., Henstock, M.R., Lamien, C.E., Diallo, A. and Mertens, P.P. 2014. Characterization of sheep pox virus vaccines for fighting lumpy skin disease virus. Antiviral Res. 109(1), 1–6. Tuppurainen, E.S., Venter, E.H., Coetzer, J.A. and Bell-Sakyi, L. 2015. Lumpy skin disease: attempts at propagation in tick cell lines and the presence of viral DNA in field ticks collected from naturally-infected cattle. Ticks Tick Borne Dis. 6(2), 134–140. Tuppurainen, E.S.M., Antoniou, S.E., Tsiamadis, E., Topkaridou, M., Labus, T., Debeljak, Z., Plavšić, B., Miteva, A., Alexandrov, T., Pite, L., Boci, J., Marojevic, D., Kondratenko, V., Atanasov, Z., Murati, B., Acinger-Rogic, Z., Kohnle, L., Calistri, P. and Broglia, A. 2020. Field observations and experiences gained from the implementation of control measures against lumpy skin disease in Southeast Europe between 2015 and 2017. Prev. Vet. Med. 181(1), 104600. Tuppurainen, E.S.M., Venter, E.H., Shisler, J.L., Gari, G., Mekonnen, G.A., Juleff, N., Lyons, N.A., De Clercq, K., Upton, C., Bowden, T.R., Babiuk, S. and Babiuk, L.A. 2017. Review: capripoxvirus diseases: current status and opportunities for control. Transbound. Emerg. Dis. 64(3), 729–745. Uddin, M.A., Hossain, M.T., Rahman, A.K.M.A., Siddique, M.P., Kafi, M.A., Hossain, M.G., Chakraborty, S., Rahman, M.M., Khasruzzaman, A.K.M., Ward, M.P. and Islam, M.A. 2024. Characterization, histopathology, a, andmmunogenicity of thlumpykin disease virus isolated during 2019-2020 in Bangladesh. Front. Microbiol. 15(1), 1324243. Vandenbussche, F., Mathijs, E., Philips, W., Saduakassova, M., De Leeuw, I., Sultanov, A., Haegeman, A. and De Clercq, K. 2022. Recombinant LSDV Strains in Asia: vaccine spillover or natural emergence? Viruses 14(7), 1429. Vinitchaikul, P., Punyapornwithaya, V., Seesupa, S., Phuykhamsingha, S., Arjkumpa, O., Sansamur, C. and Jarassaeng, C. 2023. The first study to investigate the impact of lumpy skin disease outbreaks on monthly milk production on dairy farms in Khon Kaen, Thailand. Vet. World 16(4), 687–692. Wilhelm, L. and Ward, M.P. 2023. The spread of lumpy skin disease virus across Southeast Asia: insights from surveillance. Transbound. Emerg. Dis. 2023(1), 3972359. Wilson, A.L., Courtenay, O., Kelly-Hope, L.A., Scott, T.W., Takken, W., Torr, S.J. and Lindsay, S.W. 2020. The importance of vector control for the control and elimination of vector-borne diseases. PLoS Negl. Trop. Dis. 14(1), e0007831. Wolff, J., Krstevski, K., Beer, M. and Hoffmann, B. 2020a. Minimum infective dose of a lumpy skin disease virus field strain from North Macedonia. Viruses 12(7), 768. Wolff, J., Moritz, T., Schlottau, K., Hoffmann, D., Beer, M. and Hoffmann, B. 2020b. Development of a safe and highly efficient inactivated vaccine candidate against lumpy skin disease virus. Vaccines (Basel) 9(1), 4. Wolff, J., Tuppurainen, E., Adedeji, A., Meseko, C., Asala, O., Adole, J., Atai, R., Dogonyaro, B., Globig, A., Hoffmann, D., Beer, M. and Hoffmann, B. 2021. Characterization of a Nigerian lumpy skin disease virus isolate after experimental infection of cattle. Pathogens 11(1), 16. Zeedan, G.S.G., Abdalhamed, A.M., Allam, A.M. and Abdel-Shafy, S. 2024. Molecular detection of lumpy skin disease virus in naturally infected cattle and buffaloes: unveiling the role of tick vectors in disease spread. Vet. Res. Commun. 48(6), 3921–3939. Zeedan, G.S.G., Mahmoud, A.H., Abdalhamed, A.M., El-Razik, K.A.E.A., Khafagi, M.H. and Zeina, H.A.A.A. 2019. Detection of lumpy skin disease virus in cattle using real-time polymerase chain reaction and serological diagnostic assays in different governorates of Egypt in 2017. Vet. World 12(7), 1093–1100. Zeynalova, S., Asadov, K., Guliyev, F., Vatani, M. and Aliyev, V. 2016. Epizootology and molecular diagnosis of lumpy skin disease in livestock in Azerbaijan. Front. Microbiol. 7(1), 1022. | ||
| How to Cite this Article |
| Pubmed Style Hidayatik N, Khairullah AR, Yuliani MGA, Hestianah EP, Novianti AN, Damayanti R, Hisyam MAM, Moses IB, Ahmad RZ, Wardhani BWK, Wibowo S, Kurniasih DAA. Lumpy skin disease: A growing threat to the global livestock industry. Open Vet. J.. 2025; 15(2): 541-555. doi:10.5455/OVJ.2025.v15.i2.5 Web Style Hidayatik N, Khairullah AR, Yuliani MGA, Hestianah EP, Novianti AN, Damayanti R, Hisyam MAM, Moses IB, Ahmad RZ, Wardhani BWK, Wibowo S, Kurniasih DAA. Lumpy skin disease: A growing threat to the global livestock industry. https://www.openveterinaryjournal.com/?mno=227712 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.5 AMA (American Medical Association) Style Hidayatik N, Khairullah AR, Yuliani MGA, Hestianah EP, Novianti AN, Damayanti R, Hisyam MAM, Moses IB, Ahmad RZ, Wardhani BWK, Wibowo S, Kurniasih DAA. Lumpy skin disease: A growing threat to the global livestock industry. Open Vet. J.. 2025; 15(2): 541-555. doi:10.5455/OVJ.2025.v15.i2.5 Vancouver/ICMJE Style Hidayatik N, Khairullah AR, Yuliani MGA, Hestianah EP, Novianti AN, Damayanti R, Hisyam MAM, Moses IB, Ahmad RZ, Wardhani BWK, Wibowo S, Kurniasih DAA. Lumpy skin disease: A growing threat to the global livestock industry. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 541-555. doi:10.5455/OVJ.2025.v15.i2.5 Harvard Style Hidayatik, N., Khairullah, . A. R., Yuliani, . M. G. A., Hestianah, . E. P., Novianti, . A. N., Damayanti, . R., Hisyam, . M. A. M., Moses, . I. B., Ahmad, . R. Z., Wardhani, . B. W. K., Wibowo, . S. & Kurniasih, . D. A. A. (2025) Lumpy skin disease: A growing threat to the global livestock industry. Open Vet. J., 15 (2), 541-555. doi:10.5455/OVJ.2025.v15.i2.5 Turabian Style Hidayatik, Nanik, Aswin Rafif Khairullah, M. Gandul Atik Yuliani, Eka Pramyrtha Hestianah, Arindita Niatazya Novianti, Ratna Damayanti, Mirza Atikah Madarina Hisyam, Ikechukwu Benjamin Moses, Riza Zainuddin Ahmad, Bantari Wisynu Kusuma Wardhani, Syahputra Wibowo, and Dea Anita Ariani Kurniasih. 2025. Lumpy skin disease: A growing threat to the global livestock industry. Open Veterinary Journal, 15 (2), 541-555. doi:10.5455/OVJ.2025.v15.i2.5 Chicago Style Hidayatik, Nanik, Aswin Rafif Khairullah, M. Gandul Atik Yuliani, Eka Pramyrtha Hestianah, Arindita Niatazya Novianti, Ratna Damayanti, Mirza Atikah Madarina Hisyam, Ikechukwu Benjamin Moses, Riza Zainuddin Ahmad, Bantari Wisynu Kusuma Wardhani, Syahputra Wibowo, and Dea Anita Ariani Kurniasih. "Lumpy skin disease: A growing threat to the global livestock industry." Open Veterinary Journal 15 (2025), 541-555. doi:10.5455/OVJ.2025.v15.i2.5 MLA (The Modern Language Association) Style Hidayatik, Nanik, Aswin Rafif Khairullah, M. Gandul Atik Yuliani, Eka Pramyrtha Hestianah, Arindita Niatazya Novianti, Ratna Damayanti, Mirza Atikah Madarina Hisyam, Ikechukwu Benjamin Moses, Riza Zainuddin Ahmad, Bantari Wisynu Kusuma Wardhani, Syahputra Wibowo, and Dea Anita Ariani Kurniasih. "Lumpy skin disease: A growing threat to the global livestock industry." Open Veterinary Journal 15.2 (2025), 541-555. Print. doi:10.5455/OVJ.2025.v15.i2.5 APA (American Psychological Association) Style Hidayatik, N., Khairullah, . A. R., Yuliani, . M. G. A., Hestianah, . E. P., Novianti, . A. N., Damayanti, . R., Hisyam, . M. A. M., Moses, . I. B., Ahmad, . R. Z., Wardhani, . B. W. K., Wibowo, . S. & Kurniasih, . D. A. A. (2025) Lumpy skin disease: A growing threat to the global livestock industry. Open Veterinary Journal, 15 (2), 541-555. doi:10.5455/OVJ.2025.v15.i2.5 |