| Review Article | ||
Open Vet. J.. 2025; 15(2): 601-618 Open Veterinary Journal, (2025), Vol. 15(2): 601-618 Review Article Effects of stem cell therapy on preclinical strokeGita Serafika Shannon1, Ratih Rinendyaputri2, Sunarno Sunarno2 and Amarila Malik3*1Faculty of Pharmacy, Universitas Indonesia, Depok, Indonesia 2Center for Biomedical Research, National Research and Innovation Agency (BRIN), Cibinong Science Centre, Cibinong, Indonesia 3Division of Pharmaceutical Microbiology and Biotechnology, Faculty of Pharmacy, Universitas Indonesia, Depok, Indonesia *Corresponding Author: Amarila Malik. Faculty of Pharmacy, Universitas Indonesia, Depok, Indonesia. Email: amarila.malik [at] ui.ac.id Submitted: 02/12/2024 Accepted: 12/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
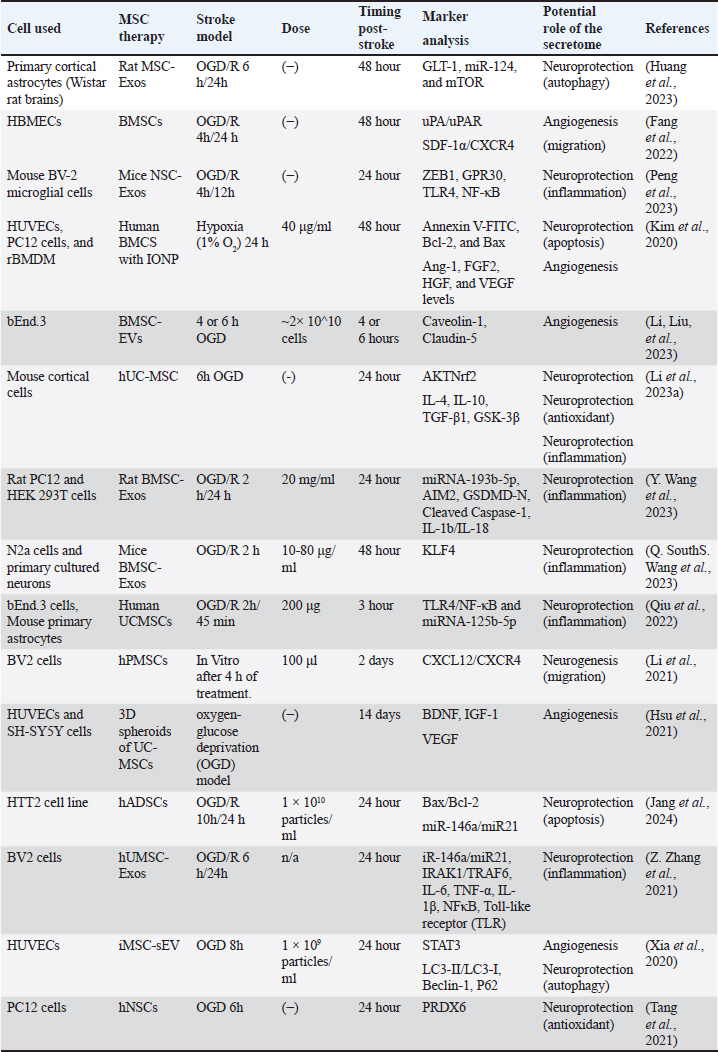
AbstractStroke, particularly ischemic stroke, is a leading cause of global mortality and disability. It is caused by blood flow obstruction and reduced oxygen delivery to brain tissue. Conventional treatments, such as tissue plasminogen activator (tPA) and mechanical thrombectomy (MT), have limited efficacy in repairing neural damage and carry risks of adverse effects. As a result, stem cell therapies, including mesenchymal stem cells (MSCs), have emerged as promising approaches for enhancing neural recovery and offering neuroprotection in ischemic stroke management. MSCs offer multifaceted benefits, such as reducing inflammation, protecting neurons, and promoting angiogenesis and neurogenesis. Recent evidence highlights the importance of MSC secretomes–extracellular vesicles (EVs) and exosomes rich in neuroprotective factors, such as microRNAs, proteins, and cytokines. These bioactive molecules demonstrated considerable efficacy in preclinical models by reducing neuroinflammation, preserving neurovascular integrity, and promoting cellular repair in ischemic environments. Preclinical in vitro and in vivo studies demonstrate the potential of the MSC secretomes to restore brain function after ischemic stroke. This is achieved by enhancing neuronal survival through mechanisms such as angiogenesis or vascular recovery, neuroprotection including modulation of immune or inflammatory responses, apoptosis, and autophagy, and promoting post-stroke neurogenesis. This review explores the translational challenges and future potential of integrating conventional ischemic stroke therapies with stem cell-based or cell-free approaches. The present study synthesizes current insights into the role of MSC-derived secretomes from both in vitro and in vivo studies. Keywords: Ischemic Stroke, Stem Cells, Preclinical, In Vitro, In Vivo. IntroductionStroke remains a leading cause of death and chronic disability worldwide, with a particularly pronounced burden in developed countries. It is characterized by abrupt cessation of cerebral blood flow caused by ischemic events (blood clots) or hemorrhagic events (ruptured blood vessels). These conditions often result in permanent brain injury and decreased motor and cognitive function (Katan and Luft, 2018). Stroke can be broadly classified into two main types: ischemic and hemorrhagic. Ischemic strokes account for 80%–87% of cases and occur due to reduced blood flow and oxygen delivery, typically due to arterial blockage. Neuronal damage in the brain caused by ischemic injury complicates treatment for patients with this condition (Chang, 2020). Hemorrhagic stroke, occurring in 10%–15% of cases, involves blood vessel rupture, causing tissue damage and cell death. The clinical manifestations of stroke vary according to location, type, and severity (An et al., 2017). The clinical manifestations of stroke depend on the brain region, in which blood circulation is disrupted. Common symptoms include body weakness, speech difficulties, vision loss, dizziness, and episodes of falling. Hemorrhagic stroke often causes more pronounced dizziness than ischemic stroke (Chugh, 2019). Diagnostic tools, such as the 6S test, sudden onset, slurred speech, side weakness, spinning (vertigo), severe headache, and second (time of onset), are used to identify strokes. Additionally, the BEFAST approach emphasizes symptoms, such as balance issues, eye (vision) problems, facial drooping, arm weakness, and speech difficulties (Chen et al., 2021). The primary objective of treatment for ischemic stroke is to rapidly reestablish cerebral blood circulation, with reperfusion being the cornerstone of management. Current interventions include intravenous tissue plasminogen activator (tPA), mechanical thrombectomy (MT), and pharmacological agents, such as aspirin, clopidogrel, and statins (Yaqubi and Karimian, 2024). Intravenous tPA is the gold standard for ischemic stroke therapy, and it is most effective when administered within the first 4.5 hours following symptom onset because its efficacy significantly declines beyond this window (Singh et al., 2020). However, tPA is associated with the risk of intracerebral hemorrhage, which may increase mortality. For cases involving large artery occlusion with substantial clot burden or when tPA is administered outside the optimal timeframe, MT is a viable alternative (Dong et al., 2020). Given the limitations of tPA and MT in promoting neural regeneration, therapies with neuroprotective neurogenic potential are needed. Cellular therapies have emerged as promising options for managing ischemic stroke, offering the potential to enhance patient recovery outcomes (Katan and Luft, 2018; Borlongan, 2019). Emerging evidence suggests that stem cell therapy can effectively improve neurological function in patients with ischemic stroke. Its potential lies in enhancing the brain’s neuroprotective mechanisms and aiding repair processes through immunomodulation and modulation of neuronal, vascular, and glial cell functions (Ejma et al., 2022). Stem cells have the potential to restore damaged brain tissue and mitigate neuroinflammation. They are distinguished by their ability for self-renewal and multipotency, which allows them to differentiate into multiple cell types (Cha et al., 2024). Several stem cell varieties—mesenchymal stem cells (MSCs), bone marrow mononuclear cells (BMMCs), neural stem cells (NSCs), and induced pluripotent stem cells (iPSCs)—have been evaluated using diverse delivery routes, such as intravenous, intra-arterial, and intracerebral methods, over durations ranging from a few hours to several months (Houkin et al., 2024). The objective of this review article was to provide a comprehensive understanding of the potential role of stem cell therapy, specifically MSCs and their derived secretomes, in enhancing post-stroke recovery, particularly in ischemic stroke. This study aimed to elucidate the underlying biological mechanisms through which MSCs exert neuroprotective effects and promote brain function recovery, focusing on the secretion of bioactive molecules, such as exosomes and extracellular vesicles (EVs) that modulate inflammation, apoptosis, and neurogenesis. By synthesizing findings from preclinical in vitro and in vivo studies, this review aims to offer insights into the translational challenges associated with integrating conventional stroke therapies with stem cell-based or cell-free approaches while also highlighting the preclinical potential of these therapeutic strategies for improving stroke recovery. The primary expectation is to provide a clear guide on the efficacy of MSC-derived secretomes in accelerating post-stroke repair, thereby paving the way for the development of more effective and safer therapeutic options in stroke management, with a focus on the biological aspects and translational hurdles in research and application. Pathophysiology of strokeIschemic stroke results from cerebral artery obstruction by a mobile embolus, which may be cardiogenic, artery-to-artery embolism, or vascular stenosis. This obstruction disrupts blood flow to the brain, leading to significant functional and neurological deficits (Shehjar et al., 2023). The resulting vascular occlusion leads to hypoxia, depriving downstream tissues of oxygen and glucose. Two distinct regions are affected: the ischemic core, where blood supply is critically insufficient for cell survival, and the ischemic penumbra, where cells remain viable for a limited time and recover if blood flow is promptly restored (Sun et al., 2024). Prolonged hypoxia and glucose deprivation deplete ATP levels, triggering membrane depolarization and excitotoxicity driven by excessive glutamate release. Overactivation of glutamate receptors causes an influx of calcium ions (Ca2+), which enhances the generation of reactive oxygen species (ROS) and reactive nitrogen species (RNS). These molecules damage lipids and DNA while releasing inflammatory mediators. Mitochondrial impairment is frequently involved in this process, leading to cytochrome C release and programmed cell death (apoptosis) (Salaudeen et al., 2024). Several pathological events can exacerbate ischemic stroke, including inflammation, energy failure, disrupted homeostasis, acidosis, increased intracellular calcium levels, excitotoxicity, and free radical toxicity. Cytokines released during the immune response contribute to cytotoxicity by activating the complement system and compromising the blood–brain barrier’s integrity. Additionally, glial cell activation, oxidative stress, and leukocyte infiltration further intensify stroke-related damage (Kuriakose and Xiao, 2020). Stroke is linked to various risk factors that can be classified as modifiable (e.g., dietary habits, preexisting comorbidities) or nonmodifiable (e.g., age, ethnicity). These factors also vary by time frame: short-term triggers (such as infections, sepsis, and psychological stress), medium-term contributors (including hypertension and hyperlipidemia), and long-term determinants (such as gender and ethnicity). It should be noted that these risk factors may differ significantly between younger and older populations (Boehme et al., 2017; George, 2020; Yoon and Bushnell, 2023). Mesenchymal stem cells (MSCs)Cell-based therapies, particularly those using stem cells, offer prospective treatments for ischemic stroke. Stem cells can regenerate damaged neurons and exert neuroprotective effects on surviving neuronal cells, thereby accelerating neurological recovery (Rahimi Darehbagh et al., 2024). These neuroprotective mechanisms include the inhibition of stroke-related pathways, such as neuroinflammation, blood–brain barrier (BBB) disruption, and apoptotic cell death, while simultaneously promoting angiogenesis and neurogenesis (Xu et al., 2017b). Several types of stem cells have been explored for stroke therapy, including MSCs, bone marrow stem cells (BMSCs), multilineage-differentiating stress-enduring (Muse) cells, and neural stem or progenitor cells. These cell types have exhibited their ability to enhance neurogenesis, promote angiogenesis, facilitate cell migration, and reduce neuroinflammation (Chrostek et al., 2019; Markowska et al., 2023). Stem cells can be sourced from four main sources: (1) embryonic tissues (Weissman, 2015); (2) fetal tissues, including the (Rosner et al., 2023) placenta (amnion and chorion), amniotic fluid, and umbilical cord (Wharton’s jelly, cord blood) (Kargozar et al., 2018; Pulido-Escribano et al., 2022; Maraldi and Russo, 2022); (3) adult tissue niches, such as adipose tissue, bone marrow, skeletal muscle, skin, and blood (Visvader and Clevers, 2016; Zou et al., 2016); and (4) iPSCs, derived from somatic cells that are genetically reprogrammed (Rony et al., 2015; Cerneckis et al., 2024). The International Society for Cellular Therapy defines MSCs based on three essential criteria: they must adhere to plastic substrates in standard culture conditions; express certain markers such as CD73, CD90, and CD105 while lacking expression of markers such as CD14, CD34, CD45, CD11b, CD79α, CD19, or HLA Class II; and demonstrate the ability to differentiate into osteoblasts, chondroblasts, and adipocytes under appropriate conditions (Ullah et al., 2015; Utama et al., 2024). Stem cells are categorized into three types: embryonic stem cells (ESCs), adult stem cells, and iPSCs (Ullah et al., 2015). While adult stem cell therapies for ischemic stroke are promising, their precise therapeutic mechanisms remain poorly understood. ESCs and iPSCs can regenerate neurons in infarcted regions, but achieving effective neuronal replacement to restore functionality remains challenging (Bang et al., 2016). Additionally, ESC and iPSC therapies carry a higher risk of tumorigenesis. Preclinical studies have shown that many transplanted cells do not survive beyond a few weeks after transplantation (Duan et al., 2021). Notably, the limited survival of transplanted cells may be an advantage of stem cell therapy because it reduces the likelihood of unregulated stem cell proliferation (Fernández-Susavila et al., 2019). Adult stem cells, particularly MSCs, are considered a more promising alternative for stroke treatment because of their ability to release bioactive compounds such as trophic factors and extracellular vesicles (EVs). EVs are tiny membrane-enclosed particles typically sized between 0.1 and 1 μm that are discharged from the cell surface to target injured brain regions. These substances can promote neurogenesis, angiogenesis, and synaptogenesis (Lai et al., 2011; Song et al., 2013). Furthermore, MSCs are believed to play several critical roles, such as reducing inflammation and mitigating scar formation, enhancing autophagy, normalizing the microenvironment and metabolic profile, and potentially restoring damaged cells across various neurological conditions (Shin et al., 2014; Yamauchi et al., 2015). Secretome mesenchymal stem cells (MSCs)MSCs secrete various substances into their culture medium, collectively known as the secretome. The secretome comprises soluble factors such as growth factors, cytokines, chemokines, and extracellular vehicles (EVs) (Ferreira et al., 2018; Foo et al., 2021). Key growth factors commonly found in stem cell secretomes include vascular endothelial growth factor (VEGF), transforming growth factor beta-1 (TGF-β1), nerve growth factor (NGF), insulin-like growth factor 1 (IGF-1), and brain-derived neurotrophic factor (BDNF) (Cunningham et al., 2018). Chemokines typically present in MSC secretomes include stromal cell-derived factor-1 (SDF-1 or C-X-C motif chemokine ligand (CXCL)12) and C-C motif chemokine ligand (CCL)2, along with cytokines such as interleukin (IL)-6, CD4+ memory T cells, and Th2 cytokines such as IL-4, IL-5, IL-6, IL-13, IL-8, and tumor necrosis factor-alpha (TNF-α)) (Han et al., 2022; Dumingan et al., 2024). Stem cell-based treatments, particularly with MSCs, have demonstrated promising results in preclinical ischemic stroke research. These treatments include improving neurological deficits, reducing infarct volume, and providing neuroprotective benefits (Zhang et al., 2021a). Numerous studies have also highlighted the therapeutic potential of MSC secretomes, focusing on their roles in immunomodulation, inflammation reduction, neuroprotection, neurotrophy, apoptosis prevention, angiogenesis regulation, and overall tissue regeneration (da Silva et al., 2023). Initially, MSCs were thought to repair damaged tissues through direct cell replacement. However, it is now widely recognized that their regenerative effects are primarily mediated by paracrine mechanisms (Cunningham et al., 2018). The substances secreted by MSCs play a vital role in the recovery of injured tissues (Asgari Taei et al., 2022). Recent studies have indicated that preconditioning MSCs can alter their secretory profile, thereby enhancing the therapeutic potential of their secretome. Common in vitro preconditioning techniques include the exposure of MSCs to hypoxic environments, proinflammatory signals, and three-dimensional culture systems (Kahrizi et al., 2023). These methods modulate MSC secretions by stimulating or inhibiting the release of factors with anti-inflammatory, immunomodulatory, antitumor, and regenerative properties, depending on the stimuli. For instance: a) hypoxic conditions enhance the synthesis of growth factors and anti-inflammatory compounds; b) proinflammatory stimuli stimulate the release of immunomodulatory and anti-inflammatory molecules; and c) three-dimensional culture systems increase the yield of anti-cancer and anti-inflammatory factors compared with traditional monolayer cultures (Madrigal et al., 2014). The involvement of the MSC secretome in enhancing recovery in preclinical models of strokeIn recent decades, numerous models have been developed to address the diverse challenges of ischemic stroke, accounting for its varied etiologies and clinical presentations. These models aim to elucidate the mechanisms behind underlying ischemia and facilitate the discovery of novel therapeutic agents for ischemic stroke treatment (Fluri et al., 2015). However, despite identifying key pathophysiological processes, only a small number of experimental protective agents have been successfully applied in clinical settings (Amado et al., 2022). This gap underscores the discrepancies between the actual conditions of patients with stroke and preclinical ischemic stroke models. Therefore, it is essential to recognize the advantages and disadvantages of each stroke model to select an experimental system that aligns with specific research objectives and elements of interest (Jickling and Sharp, 2015). Preclinical in vitro test results for stroke following MSC therapyIn vitro models are commonly used in ischemic stroke research, often involving cocultures of astrocytes and endothelial cells (Wevers et al., 2021). These systems replicate key aspects of the blood–brain barrier (BBB) and provide insights into cellular interactions under ischemic conditions (Van Breedam and Ponsaerts, 2022). Some in vitro models use primary cultures obtained from animal brain tissue to study the effects of treatments on specific brain regions, such as cortical neurons. These models also facilitate investigations of cellular responses to ischemic-like conditions, including hypoxia and glucose deprivation. Furthermore, they allow researchers to explore the molecular pathways associated with cell death, such as necrosis, apoptosis, and autophagy. In vitro methods are often used alongside in vivo studies to provide deeper insights into human ischemia (Sommer, 2017; Barthels and Das, 2020). A common in vitro approach involves brain tissue slices, which are thin sections (approximately 400 micrometer) of brain tissue that facilitate detailed examination of neuronal circuits. Furthermore, organotypic culture methods that integrate brain slice cultures and primary cell cultures are used to study ex vivo brain tissue from young animals. Primary cell cultures, including brain endothelial cells (BEC) and glial cells, are also widely used in these in vitro models to investigate ischemic mechanisms (Holloway and Gavins, 2016; Singh et al., 2022). The most common in vitro method for simulating ischemic conditions, such as stroke, is oxygen–glucose deprivation (OGD). This method involves the complete removal of oxygen and glucose from the cellular environment (Trotman-Lucas and Gibson, 2021; Amado et al., 2022). Ischemic conditions can also be replicated using chemical or enzymatic agents that inhibit metabolic activity. In the OGD model, a balanced O2/CO2 medium is replaced with an N2/CO2 medium to induce hypoxia. During this process, oxygen is substituted with nitrogen, and glucose is eliminated from the medium (Babu et al., 2022). Cell cultures are used in this model to study cellular responses under ischemic conditions. Hypoxia during OGD can be induced using hypoxia chambers, anaerobic containers, incubators with hypoxic gas supplies, or anaerobic chambers equipped with palladium catalysts to remove oxygen via water formation. Among these, hypoxia chambers with gas supply are the most widely used approach for creating hypoxic environments (Sommer, 2017). The research findings related to the use of MSC therapy in in vitro models are presented in Table 1. Table 1. Overview of research examining the effectiveness of MSC therapies in preclinical in vitro models of cerebral ischemia and the roles of secretome components.
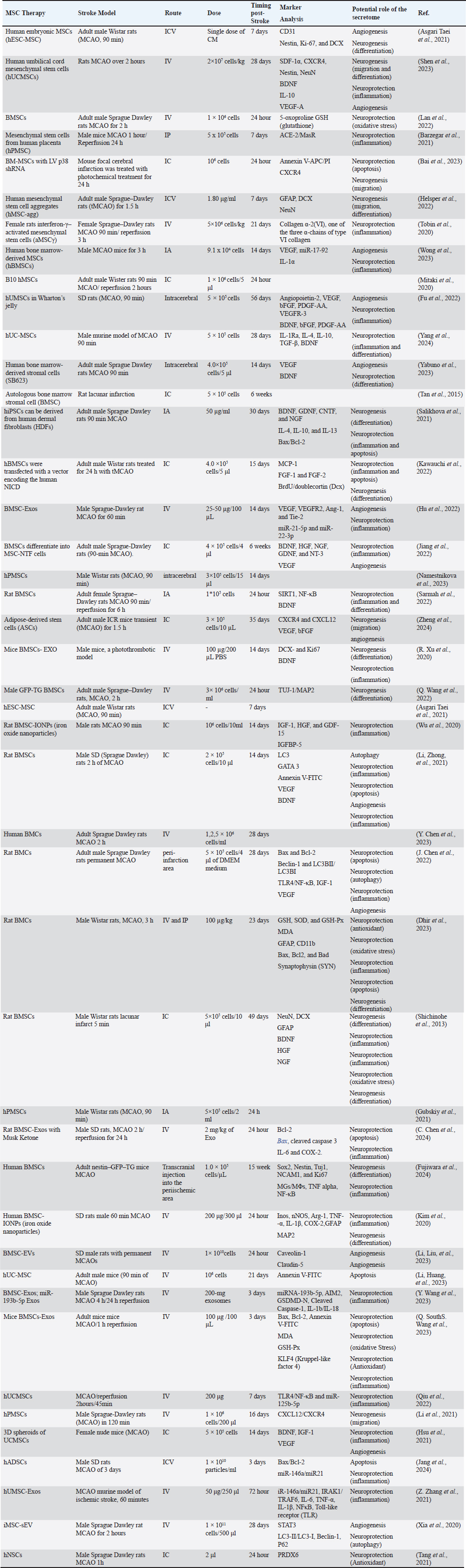
MSC therapies are advancing ischemic stroke treatment by delivering exosomes (Exos) and extracellular vesicles (EVs) that engage specific cellular pathways to confer neuroprotection, support angiogenesis, and mitigate inflammation. In astrocytes derived from Wistar rat brains, MSC-derived exosomes exert neuroprotective effects under ischemic conditions by upregulating GLT-1 via the miR-124/mTOR pathway, promoting autophagy, and reducing neuronal stress during OGD/R injury (Huang et al., 2023). Similarly, bone marrow-derived MSCs (BMSCs) modulate key pathways in human brain microvascular endothelial cells (HBMECs), specifically uPA/uPAR and SDF-1α/CXCR4, to promote angiogenesis and neurogenesis—essential for vascular repair post-stroke (Fang et al., 2022). Microglial BV-2 cells benefited from a NSC-derived exosomes, which contained ZEB1, to upregulate GPR30, suppress TLR4/NF-κB signaling, and consequently reduce inflammation (Peng et al., 2023). Further, MSC therapies enhance blood–brain barrier (BBB) integrity, as shown in bend. Three endothelial cells and primary astrocytes were treated with UCMSC-derived exosomes rich in miR-125b-5p, which blocked the TLR4/NF-κB pathway and safeguarded BBB structure and function (Qiu et al., 2022). MSC therapies also play a crucial role in apoptosis regulation and antioxidant defense. For example, under hypoxic conditions, BMSC therapies combined with iron oxide nanoparticles (IONP) in HUVECs and PC12 cells activated VEGF, Bcl-2, and TGF-β, which collectively inhibited apoptosis and reduced cellular injury (Kim et al., 2020). Moreover, adipose-derived MSCs (hADSCs) promoted HTT2 cell survival post-OGD/R by adjusting factors that promote or inhibit apoptosis Bax/Bcl-2 and miR-146a/miR-21, thus supporting neuroprotection through apoptosis inhibition (Jang et al., 2024). Notably, human NSC-derived exosomes in PC12 cells activated the antioxidant enzyme PRDX6, thereby combating oxidative stress and enhancing cell survival under ischemic conditions (Tang et al., 2021). Other models, such as PC12 and HEK293T cells, respond to rat BMSC-exosomes containing miRNA-193b-5p with reduced inflammatory responses (Wang et al., 2023b). Importantly, UC-MSC-derived three-dimensional spheroids produce paracrine factors such as BDNF, VEGF, and IGF-1 in HUVECs and SH-SY5Y cells—providing neuroprotection and support angiogenesis and anti-apoptotic mechanisms (Hsu et al., 2021). These studies, alongside others focusing on pathways such as STAT3 and miR-125b-5p, underscore the diverse molecular pathways that MSC therapy can target to enhance post-stroke recovery and therapeutic efficacy across models (Xia et al., 2020; Qiu et al., 2022). The therapeutic potential of MSC-derived secretomes—including microRNAs and cytokines—highlights their capability to address multifaceted aspects of stroke pathology. By targeting inflammation, apoptosis, oxidative stress, and vascular repair, MSC-based therapies provide a promising platform for developing advanced ischemic stroke interventions and supporting neuronal recovery. This growing body of evidence underscores MSC therapies as a powerful approach within the field of tissue regeneration, paving the way for clinical applications in ischemic stroke recovery and beyond. Preclinical in vivo testing in stroke with MSC therapyWhile numerous animal species have been used in preclinical ischemic stroke studies, rodents are primarily favored for ischemic stroke research. This preference is due to their lower costs and the similarity of their cranial blood circulation to that of human neurovascular structures (Narayan et al., 2021). Additionally, rodents offer practical advantages in controlling ischemic models, including precise manipulation of occlusion severity, duration, and location. Other species, including rabbits, pigs, nonhuman primates, and nonprimate mammals, are also used in ischemic stroke models (Woodruff et al., 2011; Lindsey et al., 2018). These models can be broadly categorized into two types: “global” ischemia and ‟local” or “focal‟ ischemia. Global ischemia models are generated by occluding two vertebral arteries, two carotid arteries, or all four vessels simultaneously for 5–15 min. These conditions mimic scenarios such as cardiac arrest and coronary occlusion in humans (Tajiri et al., 2013). Focal ischemia models are more relevant to human ischemic events. In vivo experiments using the four-vessel occlusion (4-VO) model are commonly performed in rats, mice, dogs, and pigs. This model offers advantages such as ease of preparation, high reproducibility, and low incidence of seizures. Despite their benefits, 4-VO models have certain limitations, including the requirement for a two-step surgical procedure, permanent vertebral artery occlusion, and higher mortality rates. The two-vessel occlusion (2-VO) model is commonly performed in cats, dogs, sheep, and rabbits and involves a single surgical procedure, manageable recirculation, and lower mortality rates. However, the 2-VO model exhibits poor reproducibility and strain-dependent variability (Chung et al., 2014; Popa-Wagner et al., 2014). Middle cerebral artery occlusion (MCAO) models have been extensively developed for rodents. In aged rodents, MCAO can be induced using either permanent or transient occlusion, typically lasting 30–120 min. Ischemic stroke research uses various MCAO models, classified into seven main types: (i) non-local photothrombotic occlusion by irradiation of the right carotid artery; (ii) thermocoagulation following micro-craniotomy; (iii) intraluminal occlusion using a silicone-coated suture; (iv) mechanical occlusion with hooks attached to a micromanipulator; (v) cauterization; (vi) photothrombotic occlusion; and (vii) thrombus injection into the external carotid artery or injection of endothelin-1 (Chung et al., 2014; Popa-Wagner et al., 2014). Among these models, middle cerebral artery (MCA) occlusion is the most common, accounting for nearly 50% of ischemic stroke cases involving arterial blockages. MCA occlusion can damage the cortex and striatum, but the extent of infarction depends on several factors, including the occlusion site, duration, and collateral blood flow present in the MCA (Zeng et al., 2023). The MCAO model—particularly those using endovascular filaments—has become the preferred choice in ischemic stroke research because of its ease of use and high survival rates in experimental animals. It has been widely applied to study ischemic stroke etiopathogenesis and to develop and evaluate novel therapeutic interventions (Li and Zhang, 2021; Zeng et al., 2023). Accordingly, various mediators appear to contribute to improved functional outcomes in preclinical in vivo models of ischemic stroke. In recent preclinical in vivo ischemic stroke studies are presented in Table 2, various types of MSCs have shown notable therapeutic potential through mechanisms such as neuroprotection, angiogenesis, and antioxidative defense. For example, human embryonic MSCs (hESC-MSCs) administered intracerebroventricularly in Wistar rats MCAO showed enhanced CD31 expression, indicating increased angiogenesis (Mitaki et al., 2020). Similarly, human umbilical cord MSCs (hUCMSCs) administered intravenously in MCAO rats significantly affected neurotrophic aspects, such as BDNF, VEGF, and SDF-1, over 28 days (Yang et al., 2024). In contrast, bone marrow-derived MSCs increased glutathione synthesis, providing antioxidative effects within 24 hours (Yabuno et al., 2023). Additionally, placenta-derived MSCs demonstrated neuroprotection via the ACE-2/Ang 1-7/MasR pathway in mice (Tan et al., 2015). Furthermore, genetically modified BMSCs delivered intracardially to a focal cerebral infarction model improved CXCR4+ cell recruitment (Salikhova et al., 2021). Human MSCs in preclinical stroke models demonstrate multiple therapeutic mechanisms that contribute to recovery. MSC aggregates (hMSC-agg) reduce ischemic damage through activation of the PI3K/Akt pathway (Kawauchi et al., 2022). Interferon-γ-activated MSCs (aMSCγ) upregulate collagen α-2(VI), aiding tissue remodeling (Hu et al., 2022), whereas intra-arterial bone marrow MSCs elevate G-CSF, IL-10, and VEGF levels, supporting recovery (Hu et al., 2022). Other MSC types—such as B10 and human umbilical MSCs—improve neuroprotection and angiogenesis by increasing neurotrophic factors such as BDNF and VEGF (Namestnikova et al., 2023). Additional therapies—including modified stromal cells and exosome treatments—enhance angiogenesis, reduce apoptosis, and leverage signaling pathways for neuroprotection (Chen et al., 2023, 2024). Additionally, hBMSCs (SB623), when administered intracerebrally, elevated VEGF and BDNF levels, promoting recovery (R. Xu et al. (2020). Other approaches—such as administering autologous BMSCs into models of lacunar infarction—showed beneficial effects, particularly related to insulin-like growth factor 1 (Wang et al., 2022). Table 2. An overview of the effectiveness of MSC therapies in preclinical in vivo models of cerebral ischemia along with an emphasis on the possible roles of secretome components.
Furthermore, MSCs derived from human adipose tissue, administered intracranially, increased miR-146a and miR-21 expression, contributing to neuroprotection (Hsu et al., 2021). Studies utilizing three-dimensional spheroids of MSCs in MCAO models revealed the capacity to deliver paracrine factors, such as BDNF and IGF-1, further supporting recovery mechanisms (Li et al., 2021). In summary, these results emphasize the varied therapeutic potential of MSCs and their derivatives in promoting stroke recovery while underscoring the need to improve delivery techniques and gain a deeper understanding of the underlying mechanisms for future clinical use. ConclusionThe use of MSCs and their secretomes represents a groundbreaking approach to managing ischemic stroke, effectively addressing the limitations of traditional therapies. Preclinical research has demonstrated that MSCs and their secretomes offer substantial neuroprotective, anti-inflammatory, and angiogenic benefits, significantly supporting functional recovery in ischemic stroke models. MSC-secretome-based therapies, particularly those involving extracellular vesicles and exosomes, have yielded promising results in enhancing neuronal survival and facilitating vascular repair. These effects are primarily mediated by the regulation of inflammation and stimulation of neurogenesis pathways. In vitro and in vivo models provide essential insights into the interaction of secretomes with diverse cell types implicated in ischemic stroke pathology. Despite these promising findings, the translation of MSC therapies into clinical practice requires further refinement, particularly in terms of delivery routes, dosing protocols, and preconditioning strategies. Addressing these translational challenges could facilitate the integration of MSC-based therapies into standard clinical practice for stroke, providing a safer and more versatile treatment option to enhance recovery and improve patient outcomes. AcknowledgmentThe authors would like to express their gratitude to the National Research and Innovation Agency of Indonesia (BRIN) for their invaluable support throughout the preparation of this review article. Competing interestThe authors declare no competing interests related to the research, authorship, or publication of this article. FundingNo funding Author’s contributionsGita Serafika Shannon drafted the manuscript. Ratih Rinendyaputri and Amarila Malik revised and edited the manuscript. Ratih Rinendyaputri, Amarila Malik, and Sunarno critically checked the manuscript. Gita Serafika Shannon, Ratih Rinendyaputri, and Amarila Malik edited the manuscript. All authors have read and approved the final version of the manuscript. Data availabilityAll references are open-access, so data can be obtained from the online literature. ReferencesAmado, B., Melo, L., Pinto, R., Lobo, A., Barros, P. and Gomes, J.R. 2022. Ischemic stroke, lessons from the past towards effective preclinical models. Biomedicines 10, 2561. An, S.J., Kim, T.J. and Yoon, B. W. 2017. Epidemiology, risk factors, and clinical features of intracerebral hemorrhage: an update. J. Stroke. 19, 3–10. Asgari Taei, A., Dargahi, L., Khodabakhsh, P., Kadivar, M. and Farahmandfar, M. 2022. Hippocampal neuroprotection mediated by secretome of human mesenchymal stem cells against experimental stroke. CNS Neurosci. Ther. 28, 1425–1438. Asgari Taei, A., Nasoohi, S., Hassanzadeh, G., Kadivar, M., Dargahi, L. and Farahmandfar, M. 2021. Enhancement of angiogenesis and neurogenesis by intracerebroventricular injection of secretome from human embryonic stem cell-derived mesenchymal stem cells in ischemic stroke model. Biomed. Pharmacother. 140, 111709. Babu, M., Singh, N. and Datta, A. 2022. In vitro oxygen glucose deprivation model of ischemic stroke: a proteomics-driven systems biological perspective. Mol. Neurobiol. 59, 2363–2377. Bai, Y., Wang, L., Xu, R. and Cui, Y. 2023. Mesenchymal stem cells with p38 mitogen-activated protein kinase interference ameliorate mouse ischemic stroke. Exp. Biol. Med. 248, 2481–2491. Bang, O. Y., Kim, E. H., Cha, J. M. and Moon, G. J. 2016. Adult stem cell therapy for stroke: challenges and progress. J. Stroke. 18, 256. Barthels, D. and Das, H. 2020. Current advances in ischemic stroke research and therapies. Biochim. Biophys. Acta Mol. Basis Dis. 1866, 165260. Barzegar, M., Vital, S., Stokes, K.Y., Wang, Y., Yun, J.W., White, L.A., Chernyshev, O., Kelley, R.E. and Alexander, J.S. 2021. Human placenta mesenchymal stem cell protection in ischemic stroke is angiotensin converting enzyme-2 and masR receptor-dependent. Stem Cells. 39, 1335–1348. Boehme, A.K., Esenwa, C. and Elkind, M.S.V. 2017. Stroke risk factors, genetics, and prevention. Circ. Res. 120, 472. Borlongan, C.V. 2019. Concise review: stem cell therapy for stroke patients: are we there yet? Stem Cells Transl. Med. 8, 983–988. Cerneckis, J., Cai, H. and Shi, Y. 2024. Induced pluripotent stem cells (iPSCs): molecular mechanisms of induction and applications. Signal Transduct. Target Ther. 9, 1–26. Cha, Z., Qiao, Y., Lu, Q., Wang, Q., Lu, X., Zhou, H. and Li, T. 2024. Research progress and challenges of stem cell therapy for ischemic stroke. Front. Cell Dev. Biol. 12, 1410732. Chang, J.C. 2020. Stroke classification: critical role of unusually large von Willebrand factor multimers and tissue factor on clinical phenotypes based on novel “two-path unifying theory” of hemostasis. Clin. Appl. Thromb. Hemost. 26, 1076029620913634. Chen, C., Feng, D., Lu, F., Qin, J., Dun, L., Liao, Z., Tao, J. and Zhou, Z. 2024. Neuroprotective effects of exosomes derived from bone marrow mesenchymal stem cells treated by Musk Ketone on ischemic stroke. J. Stroke Cerebrovasc. Dis. 33, 107628. Chen, J., Lin, X., Yao, C., Bingwa, L.A., Wang, H., Lin, Z., Jin, K., Zhuge, Q. and Yang, S. 2022. Transplantation of Roxadustat-preconditioned bone marrow stromal cells improves neurological function recovery through enhancing grafted cell survival in ischemic stroke rats. CNS Neurosci. Ther. 28, 1519–1531. Chen, X., Zhao, X., Xu, F., Guo, M., Yang, Y., Zhong, L., Weng, X. and Liu, X. 2021. A systematic review and meta-analysis comparing FAST and BEFAST in acute stroke patients. Front. Neurol. 12, 765069. Chen, Y., Peng, D., Li, J., Zhang, L., Chen, J., Wang, L. and Gao, Y. 2023. A comparative study of different doses of bone marrow-derived mesenchymal stem cells improve post-stroke neurological outcomes via intravenous transplantation. Brain Res. 1798, 148161. Chrostek, M.R., Fellows, E.G., Crane, A.T., Grande, A.W. and Low, W.C. 2019. Efficacy of stem cell-based therapies for stroke. Brain Res. 1722, 146362. Chugh, C. 2019. Acute ischemic stroke: management approach. Indian J. Crit. Care Med. 23, S140. Chung, J.W., Park, .H., Kim, N., Kim, W.J., Park, J.H., Ko, Y., Yang, M.H., Jang, M.S., Han, M.K., Jung, C., Kim, J.H., Oh, C.W. and Bae, H.J. 2014. Trial of ORG 10172 in Acute Stroke Treatment (TOAST) classification and vascular territory of ischemic stroke lesions diagnosed by diffusion-weighted imaging. J. Am. Heart Assoc. 3, e001119. Cunningham, C.J., Redondo-Castro, E. and Allan, S.M. 2018. The therapeutic potential of the mesenchymal stem cell secretome in ischaemic stroke. J. Cereb. Blood Flow Metab. 38, 1276–1292. da Silva, A.V., Serrenho, I., Araújo, B., Carvalho, A.M. and Baltazar, G. 2023. Secretome as a tool to treat neurological conditions: are we ready? Int. J. Mol. Sci. 24, 16544. Dhir, N., Jain, A., Sharma, A.R., Sharma, S., Mahendru, D., Patial, A., Malik, D., Prakash, A., Attri, S.V., Bhattacharyya, S., Das Radotra, B. and Medhi, B. 2023. Rat BM-MSCs secretome alone and in combination with stiripentol and ISRIB, ameliorated microglial activation and apoptosis in experimental stroke. Behav. Brain Res. 449, 114471. Dong, X., Gao, J., Su, Y. and Wang, Z. 2020. Nanomedicine for ischemic stroke. Int. J. Mol. Sci. 21, 7600. Duan, R., Gao, Y., He, R., Jing, L., Li, Y., Gong, Z., Yao, Y., Luan, T., Zhang, C., Li, L. and Jia, Y. 2021. Induced pluripotent stem cells for ischemic stroke treatment. Front. Neurosci. 15, 628663. Dumingan, A., A. Malik, R. Rinendyaputri, H. A. N. Utama, S. Sunarno, Y. R. Purwaningtyas, H. H. Idrus, R. Noverina, F. Huda, and A. Faried. 2024. The Characteristics of Umbilical Cord Derived Mesenchymal Stem Cell/UCMSC from Macaca fascicularis and Its Secretome Under Hypoxic Conditions. 17, 1–13. Ejma, M., Madetko, N., Brzecka, A., Alster, P., Budrewicz, S., Koszewicz, M., Misiuk-Hojło, M., Tomilova, I.K., Somasundaram, S.G. and Kirkland, C.E. 2022. The role of stem cells in the therapy of stroke. Curr. Neuropharmacol. 20, 630. Fang, C.N., Song, T., Tao, X., Liu, S.F. and Feng, Z. 2022. Bone marrow stromal cells attenuate oxygen and glucose deprivation followed by re-oxygenation-induced brain microvascular endothelial cell injury. Acta Neurobiol. Exp. 82, 398–407. Fernández-Susavila, H., Bugallo-Casal, A., Castillo, J. and Campos, F. 2019. Adult stem cells and induced pluripotent stem cells for stroke treatment. Front. Neurol. 10, 908. Ferreira, J.R., Teixeira, G.Q., Santos, S.G., Barbosa, M.A., Almeida-Porada, G. and Gonçalves, R.M. 2018. Mesenchymal stromal cell secretome: influencing therapeutic potential by cellular pre-conditioning. Front. Immunol. 9, 2837. Fluri, F., Schuhmann, M.K. and Kleinschnitz, C. 2015. Animal models of ischemic stroke and their application in clinical research. Drug Des. Devel. Ther. 9, 3445–3454. Foo, J.B., Looi, Q.H., Chong, P.P., Hassan, N.H., Yeo, G.E.C., Ng, C.Y., Koh, B., How, C.W., Lee, S.H. and Law, J.X. 2021. Comparing the therapeutic potential of stem cells and their secretory products in regenerative medicine. Stem Cells Int. 2021, 2616807. Fu, Y.S., Yeh, C.C., Chu, P.M., Chang, W.H., Lin, M.Y.A. and Lin, Y.Y. 2022. Xenograft of human umbilical mesenchymal stem cells promotes recovery from chronic ischemic stroke in rats. Int. J. Mol. Sci. 23, 3149. Fujiwara, S., Nakano-Doi, A., Sawano, T., Kubo, S., Doe, N. and Nakagomi, T. 2024. Administration of human-derived mesenchymal stem cells activates locally stimulated endogenous neural progenitors and reduces neurological dysfunction in mice after ischemic stroke. Cells 13, 939. George, M.G. 2020. Risk factors for ischemic stroke in younger adults. Stroke 51, 729–735. Gubskiy, I.L., Namestnikova, D.D., Sukhinich, K.K., Revkova, V.A., Melnikov, P.A., Gubsky, L.V., Chekhonin, V.P. and Yarygin, K.N. 2021. MRI-based and histologically verified 3D modeling of spatial distribution of intra-arterially transplanted cells in rat brain. Bull. Exp. Biol. Med. 171, 517–522. Han, Y., Yang, J., Fang, J., Zhou, Y., Candi, E., Wang, J., Hua, D., Shao, C. and Shi, Y. 2022. The secretion profile of mesenchymal stem cells and potential applications in treating human diseases. Signal Transduct. Target Ther. 7, 1–19. Helsper, S., Bagdasarian, F.A., Yuan, X., Xu, K., Lee, J.Y., Rosenberg, J.T., Borlongan, C.V., Ma, T. and Grant, S.C. 2022. Extended ischemic recovery after implantation of human mesenchymal stem cell aggregates indicated by sodium MRI at 21.1 T. Transl. Stroke Res. 13, 543–555. Holloway, P.M. and Gavins, F.N.E. 2016. Modeling ischemic stroke in vitro: status quo and future perspectives. Stroke 47, 561–569. Houkin, K., Osanai, T., Uchiyama, S., Minematsu, K., Taguchi, A., Maruichi, K., Niiya, Y., Asaoka, K., Kuga, Y., Takizawa, K., Haraguchi, K., Yoshimura, S., Kimura, K., Tokunaga, K., Aoyama, A., Ikawa, F., Inenaga, C., Abe, T., Tominaga, A., Takahashi, S., Kudo, K., Fujimura, M., Sugiyama, T., Ito, M., Kawabori, M., Hess, D.C., Savitz, S.I., Hirano, T., Investigators. 2024. Allogeneic stem cell therapy for acute ischemic stroke: the phase 2/3 TREASURE randomized clinical trial. JAMA Neurol. 81, 154–162. Hsu, T.W., Lu, Y.J., Lin, Y.J., Huang, Y.T., Hsieh, L.H., Wu, B.H., Lin, Y.C., Chen, L.C., Wang, H.W., Chuang, J.C., Fang, Y.Q. and Huang, C.C. 2021. Transplantation of 3D MSC/HUVEC spheroids with neuroprotective and proangiogenic potentials ameliorates ischemic stroke brain injury. Biomaterials 272, 120765. Hu, H., Hu, X., Li, L., Fang, Y., Yang, Y., Gu, J., Xu, J. and Chu, L. 2022. Exosomes derived from bone marrow mesenchymal stem cells promote angiogenesis in ischemic stroke mice via upregulation of MiR-21-5p. Biomolecules 12, 883. Huang, W., Fan, Y., Jiang, C., Jiao, J., Ji, W., Huang, H. and Shao, J. 2023. Marrow mesenchymal stem cell-derived exosomes upregulate astrocytic glutamate transporter-1 expression via miR-124/mTOR pathway against oxygen-glucose deprivation/reperfusion injury. J. Integr. Neurosci. 22, 144. Jang, E., Yu, H., Kim, E., Hwang, J., Yoo, J., Choi, J., Jeong, H.S. and Jang, S. 2024. The therapeutic effects of blueberry-treated stem cell-derived extracellular vesicles in ischemic stroke. Int. J. Mol. Sci. 25, 6362. Jiang, F., Zhou, H., Cheng, Y., He, Z., Meng, P., Sun, K., Wang, P., Han, X., Wang, L., Yang, M., Jiang, N., Liu, Y., Yuan, C., Yang, Q. and An, Y. 2022. Various detailed characteristics of a new enhanced neurotrophic factor secreting rat derived bone marrow mesenchymal stem cells and its preliminary application in rat models of ischemic stroke. Exp. Cell Res. 416, 113140. Jickling, G.C. and Sharp, F.R. 2015. Improving the translation of animal ischemic stroke studies to humans. Metab. Brain Dis. 30, 461–467. Kahrizi, M.S., Mousavi, E., Khosravi, A., Rahnama, S., Salehi, A., Nasrabadi, N., Ebrahimzadeh, F. and Jamali, S. 2023. Recent advances in pre-conditioned mesenchymal stem/stromal cell (MSCs) therapy in organ failure; a comprehensive review of preclinical studies. Stem Cell Res. Ther. 14, 155. Kargozar, S., Mozafari, M., Hashemian, S.J., Brouki Milan, P., Hamzehlou, S., Soleimani, M., Joghataei, M.T., Gholipourmalekabadi, M., Korourian, A., Mousavizadeh, K. and Seifalian, A.M. 2018. Osteogenic potential of stem cells-seeded bioactive nanocomposite scaffolds: A comparative study between human mesenchymal stem cells derived from bone, umbilical cord Wharton’s jelly, and adipose tissue. J. Biomed. Mater. Res. B Appl. Biomater. 106, 61–72. Katan, M. and Luft, A. 2018. Global burden of stroke. Semin. Neurol. 38, 208–211. Kawauchi, S., Yasuhara, T., Kin, K., Yabuno, S., Sugahara, C., Nagase, T., Hosomoto, K., Okazaki, Y., Tomita, Y., Umakoshi, M., Sasaki, T., Kameda, M., Borlongan, C.V. and Date, I. 2022. Transplantation of modified human bone marrow-derived stromal cells affords therapeutic effects on cerebral ischemia in rats. CNS Neurosci. Ther. 28, 1974–1985. Kim, H.Y., Kim, T.J., Kang, L., Kim, Y.J., Kang, M.K., Kim, J., Ryu, J.H., Hyeon, T., Yoon, B.W., Ko, S.B. and Kim, B.S. 2020. Mesenchymal stem cell-derived magnetic extracellular nanovesicles for targeting and treatment of ischemic stroke. Biomaterials 243, 119942. Kuriakose, D. and Xiao, Z. 2020. Pathophysiology and treatment of stroke: present status and future perspectives. Int. J. Mol. Sci. 21, 1–24. Lai, R.C., Chen, T.S. and Lim, S.K. 2011. Mesenchymal stem cell exosome: a novel stem cell-based therapy for cardiovascular disease. Regener. Med. 6, 481–492. Lan, X.Y., Sun, Z.W., Xu, G.L., Chu, C.Y., Qin, H.M., Li, S., Geng, X., Gao, P., Boltze, J. and Li, S. 2022. Bone marrow mesenchymal stem cells exert protective effects after ischemic stroke through upregulation of glutathione. Stem Cell Rev. Rep. 18, 585–594. Li, Y. and Zhang, J. 2021. Animal models of stroke. Animal Model Exp. Med. 4, 204. Li, Y., Dong, Y., Ran, Y., Zhang, Y., Wu, B., Xie, J., Cao, Y., Mo, M., Li, S., Deng, H., Hao, W., Yu, S. and Wu, Y. 2021a. Three-dimensional cultured mesenchymal stem cells enhance repair of ischemic stroke through inhibition of microglia. Stem Cell Res. Ther. 12, 358. Li, Y., Huang, J., Wang, J., Xia, S. Ran, H., Gao, L., Feng, C., Gui, L., Zhou, Z. and Yuan, J. 2023a. Human umbilical cord-derived mesenchymal stem cell transplantation supplemented with curcumin improves the outcomes of ischemic stroke via AKT/GSK-3β/β-TrCP/Nrf2 axis. J. Neuroinflammation. 20, 49. Li, Y., Liu, B., Zhao, T., Quan, X., Han, Y., Cheng, Y., Chen, Y., Shen, X., Zheng, Y. and Zhao, Y. 2023b. Comparative study of extracellular vesicles derived from mesenchymal stem cells and brain endothelial cells attenuating blood-brain barrier permeability via regulating Caveolin-1-dependent ZO-1 and Claudin-5 endocytosis in acute ischemic stroke. J. Nanobiotechnology. 21, 70. Li, Y., Zhong, W., Huang, Q., Lang, B. and Tang, X. 2021b. GATA3 improves the protective effects of bone marrow-derived mesenchymal stem cells against ischemic stroke induced injury by regulating autophagy through CREG. Brain Res. Bull. 176, 151–160. Lindsey, M.L., Bolli, R., Canty, J.M., Du, X.J., Frangogiannis, N.G., Frantz, S., Gourdie, R.G., Holmes, J.W., Jones, S.P., Kloner, R.A., Lefer, D.J., Liao, R., Murphy, E., Ping, P., Przyklenk, K., Recchia, F.A., Longacre, L.S., Ripplinger, C.M., Van Eyk, J.E. and Heusch, G. 2018. Guidelines for experimental models of myocardial ischemia and infarction. Am. J. Physiol. Heart Circ. Physiol. 314, H812–H838. Madrigal, M., Rao, K.S. and Riordan, N.H. 2014. A review of therapeutic effects of mesenchymal stem cell secretions and induction of secretory modification by different culture methods. Transl. J. Med. 12, 260. Maraldi, T. and Russo, V. 2022. Amniotic fluid and placental membranes as sources of stem cells: progress and challenges. Int. J. Mol. Sci. 23, 5362. Markowska, A., Koziorowski, D. and Szlufik, S. 2023. Microglia and stem cells for ischemic stroke treatment-mechanisms, current status, and therapeutic challenges. Front. Biosci. 28, 269. Mitaki, S., Nagai, A., Wada, Y., Onoda, K., Md. Sheikh, A., Adachi, E., Ichi Matsumoto, K. and Yamaguchi, S. 2020. iTRAQ-based proteomic analysis after mesenchymal stem cell line transplantation for ischemic stroke. Brain Res. 1742, 146900. Namestnikova, D.D., Gubskiy, I.L., Cherkashova, E.A., Sukhinich, K.K., Melnikov, P.A., Gabashvili, A.N., Kurilo, V.V., Chekhonin, V.P., Gubsky, L.V. and Yarygin, K.N. 2023. Therapeutic efficacy and migration of mesenchymal stem cells after intracerebral transplantation in rats with experimental ischemic stroke. Bull. Exp. Biol. Med. 175, 116–125. Narayan, S.K., Grace Cherian, S., Babu Phaniti, P., Babu Chidambaram, S., Rachel Vasanthi, A.H. and Arumugam, M. 2021. Preclinical animal studies in ischemic stroke: Challenges and some solutions. Animal Model Exp. Med. 4, 104. Peng, J., Yu, Z., Xiao, R., Hu, X. and Xia, Y. 2023. Exosomal ZEB1 derived from neural stem cells reduces inflammation injury in OGD/R-treated microglia via the GPR30-TLR4-NF-κB axis. Neurochem. Res. 48, 1811–1821. Popa-Wagner, A., Buga, A.M., Doeppner, T.R. and Hermann, D.M.. 2014. Stem cell therapies in preclinical models of stroke associated with aging. Front. Cell Neurosci. 8, 1–10. Pulido-Escribano, V., Torrecillas-Baena, B., Camacho-Cardenosa, M., Dorado, G., Gálvez-Moreno, M.Á. and Casado-Díaz, A. 2022. Role of hypoxia preconditioning in therapeutic potential of mesenchymal stem-cell-derived extracellular vesicles. World J. Stem Cells 14, 453. Qiu, L., Cai, Y., Geng, Y., Yao, X., Wang, L., Cao, H., Zhang, X., Wu, Q., Kong, D., Ding, D., Shi, Y., Wang, Y. and Wu, J. 2022. Mesenchymal stem cell-derived extracellular vesicles attenuate tPA-induced blood–brain barrier disruption in murine ischemic stroke models. Acta Biomater. 154, 424–442. Rahimi Darehbagh, R., Seyedoshohadaei, S.A., Ramezani, R. and Rezaei, N. 2024. Stem cell therapies for neurological disorders: current progress, challenges, and future perspectives. Eur. J. Med. Res. 29, 1–20. Rony, I.K., Baten, A., Bloomfield, J.A., Islam, M.E., Billah, M.M. and Islam, K.D. 2015. Inducing pluripotency in vitro: Recent advances and highlights in induced pluripotent stem cells generation and pluripotency reprogramming. Cell Prolif. 48, 140–156. Rosner, M., Horer, S., Feichtinger, M. and Hengstschläger, M. 2023. Multipotent fetal stem cells in reproductive biology research. Stem Cell Res. Ther. 14, 1–26. Salaudeen, M.A., Bello, N., Danraka, R.N. and Ammani, M.L. 2024. Understanding the pathophysiology of ischemic stroke: the basis of current therapies and opportunity for new ones. Biomolecules 14, 305. Salikhova, D., Bukharova, T., Cherkashova, E., Namestnikova, D., Leonov, G., Nikitina, M., Gubskiy, I., Akopyan, G., Elchaninov, A., Midiber, K., Bulatenco, N., Mokrousova, V., Makarov, A., Yarygin, K., Chekhonin, V., Mikhaleva, L., Fatkhudinov, T. and Goldshtein, D. 2021. Therapeutic effects of hiPSC-derived glial and neuronal progenitor cells-conditioned medium in experimental ischemic stroke in rats. Int. J. Mol. Sci. 22, 4694. Sarmah, D., Datta, A., Kaur, H., Kalia, K., Borah, A., Rodriguez, A.M., Yavagal, D.R. and Bhattacharya, P. 2022. Sirtuin-1 - Mediated NF-κB pathway modulation to mitigate inflammasome signaling and cellular apoptosis is one of the neuroprotective effects of intra-arterial mesenchymal stem cell therapy following ischemic stroke. Stem Cell Rev. Rep. 18, 821–838. Shehjar, F., Maktabi, B., Rahman, Z.A., Bahader, G.A., James, A.W., Naqvi, A., Mahajan, R. and Shah, Z.A. 2023. Stroke: molecular mechanisms and therapies: update on recent developments. Neurochem. Int. 162, 105458. Shen, D., Wang, H., Zhu, H., Jiang, C., Xie, F., Zhang, H., Lv, Q., Liu, Q., Wang, Z., Qi, N. and Wang, H. 2023. Pre-clinical efficacy evaluation of human umbilical cord mesenchymal stem cells for ischemic stroke. Front. Immunol. 13, 1095469. Shichinohe, H., Yamauchi, T., Saito, H., Houkin, K. and Kuroda, S. 2013. Bone marrow stromal cell transplantation enhances recovery of motor function after lacunar stroke in rats. Acta Neurobiol. Exp. 73, 354–363. Shin, J.Y., Park, H.J., Kim, H.N., Oh, S.H., Bae, J.S., Ha, H.J. and Lee, P.H. 2014. Mesenchymal stem cells enhance autophagy and increase β-amyloid clearance in Alzheimer disease models. Autophagy 10, 32–44. Singh, A.A., Kharwar, A. and Dandekar, M.P.. 2022. A review on preclinical models of ischemic stroke: insights into the pathomechanisms and new treatment strategies. Curr. Neuropharmacol. 20, 1667. Singh, M., Pandey, P.K., Bhasin, A., Padma, M.V. and Mohanty, S. 2020. Application of stem cells in stroke: a multifactorial approach. Front. Neurosci. 14, 532599. Sommer, C.J. 2017. Ischemic stroke: experimental models and reality. Acta Neuropathol. 133, 245–261. Song, M., Mohamad, O., Gu, X., Wei, L. and Yu, S.P. 2013. Restoration of intracortical and thalamocortical circuits after transplantation of bone marrow mesenchymal stem cells into the ischemic brain of mice. Cell Transplant. 22, 2001–2015. Sun, Y., Jiang, X. and Gao, J. 2024. Stem cell-based ischemic stroke therapy: novel modifications and clinical challenges. Asian J. Pharm Sci. 19, 100867. Tajiri, N., Dailey, T., Metcalf, C., Mosley, Y.I., Lau, T., Staples, M., van Loveren, H., Kim, S.U., Yamashima, T., Yasuhara, T., Date, I., Kaneko, Y. and Borlongan, C.V. 2013. In vivo animal stroke models: a rationale for rodent and non-human primate models. Transl Stroke Res. 4, 308. Tan, C., Shichinohe, H., Abumiya, T., Nakayama, N., Kazumata, K., Hokari, M., Hamauchi, S. and Houkin, K. 2015. Short-, middle- and long-term safety of superparamagnetic iron oxide-labeled allogeneic bone marrow stromal cell transplantation in rat model of lacunar infarction. Neuropathology 35, 197–208. Tang, B., Ni, W., Zhou, J., Ling, Y., Niu, D., Lu, X., Chen, T., Ramalingam, M. and Hu, J. 2021. Peroxiredoxin 6 secreted by Schwann-like cells protects neuron against ischemic stroke in rats via PTEN/PI3K/AKT pathway. Tissue Cell. 73, 101635. Tobin, M.K., Stephen, T.K.L., Lopez, K.L., Pergande, M.R., Bartholomew, A.M., Cologna, S.M. and Lazarov, O. 2020. Activated mesenchymal stem cells induce recovery following stroke via regulation of inflammation and oligodendrogenesis. J. Am. Heart Assoc. 9, e013583. Trotman-Lucas, M. and Gibson, C.L. 2021. A review of experimental models of focal cerebral ischemia focusing on the middle cerebral artery occlusion model. F1000Res. 10, 242. Ullah, I., Subbarao, R.B. and Rho, G.J. 2015. Human mesenchymal stem cells - current trends and future prospective. Biosci. Rep. 35, e00191. Utama, H. A. N., S. S. Mariya, A. Dumingan, R. Rinendyaputri, Sunarno, and A. Malik. 2024. Biotechnology-based therapy for stroke treatment: review. 22, 248–260. Van Breedam, E. and Ponsaerts, P. 2022. Promising strategies for the development of advanced in vitro models with high predictive power in ischaemic stroke research. Int. J. Mol. Sci. 23, 7140. Visvader, J.E. and Clevers, H. 2016. Tissue-specific designs of stem cell hierarchies. Nat. Cell Biol. 18, 349–355. Wang, Q., Zhao, S.F., Xiao, X., Liu, Y.N., Wang, X.L. and Du, Y.X. 2022. Combined treatment of bone marrow mesenchymal stem cells and fasudil promotes neurovascular remodeling and neurological function recovery in ischemic stroke. Appl. Biochem. Biotechnol. 194, 801–812. Wang, Q.S., Xiao, R.J., Peng, J., Yu, Z.T., Fu, J.Q. and Xia, Y. 2023a. Bone marrow mesenchymal stem cell-derived exosomal KLF4 alleviated ischemic stroke through inhibiting N6-methyladenosine modification level of Drp1 by targeting lncRNA-ZFAS1. Mol. Neurobiol. 60, 3945–3962. Wang, Y., Chen, H., Fan, X., Xu, C., Li, M., Sun, H., Song, J., Jia, F., Wei, W., Jiang, F., Li, G. and Zhong, D. 2023b. Bone marrow mesenchymal stem cell-derived exosomal miR-193b-5p reduces pyroptosis after ischemic stroke by targeting AIM2. J. Stroke Cerebrovasc. Dis. 32, 107235. Weissman, I.L. 2015. Stem cells are units of natural selection for tissue formation, for germline development, and in cancer development. Proc. Natl. Acad. Sci. U S A. 112, 8922–8928. Wevers, N.R., Nair, A.L., Fowke, T.M., Pontier, M., Kasi, D.G., Spijkers, X.M., Hallard, C., Rabussier, G., van Vught, R., Vulto, P., de Vries, H.E. and Lanz, H.L. 2021. Modeling ischemic stroke in a triculture neurovascular unit on-a-chip. Fluids Barriers CNS 18, 1–18. Wong, R., Smith, C.J., Allan, S.M. and Pinteaux, E. 2023. Preconditioning with interleukin-1 alpha is required for the neuroprotective properties of mesenchymal stem cells after ischemic stroke in mice. J. Cereb. Blood Flow Metab. 43, 2040–2048. Woodruff, T.M., Thundyil, J., Tang, S.C., Sobey, C.G., Taylor, S.M. and Arumugam, T. V. 2011. Pathophysiology, treatment, and animal and cellular models of human ischemic stroke. Mol. Neurodegener. 6, 1–19. Wu, M.R., Lee, C.H. and Hsiao, J.K. 2020. Bidirectional enhancement of cell proliferation between iron oxide nanoparticle-labeled mesenchymal stem cells and choroid plexus in a cell-based therapy model of ischemic stroke. Int. J. Nanomedicine 15, 9181–9195. Xia, Y., Ling, X., Hu, G., Zhu, Q., Zhang, J., Li, Q., Zhao, B., Wang, Y. and Deng, Z. 2020. Small extracellular vesicles secreted by human iPSC-derived MSC enhance angiogenesis through inhibiting STAT3-dependent autophagy in ischemic stroke. Stem Cell Res. Ther. 11, 313. Xu, R., Bai, Y., Min, S., Xu, X., Tang, T. and Ju, S. 2020. In vivo monitoring and assessment of exogenous mesenchymal stem cell-derived exosomes in mice with ischemic stroke by molecular imaging. Int. J. Nanomedicine 15, 9011–9023. Xu, W., Zheng, J., Gao, L., Li, T., Zhang, J. and Shao, A. 2017. Neuroprotective effects of stem cells in ischemic stroke. Stem Cells Int. 2017, 4653936. Yabuno, S., Yasuhara, T., Nagase, T., Kawauchi, S., Sugahara, C., Okazaki, Y., Hosomoto, K., Sasada, S., Sasaki, T., Tajiri, N., Borlongan, C.V. and Date, I. 2023. Synergistic therapeutic effects of intracerebral transplantation of human modified bone marrow-derived stromal cells (SB623) and voluntary exercise with running wheel in a rat model of ischemic stroke. Stem Cell Res. Ther. 14, 10. Yamauchi, T., Kuroda, Y., Morita, T., Shichinohe, H., Houkin, K., Dezawa, M. and Kuroda, S. 2015. Therapeutic effects of human multilineage-differentiating stress enduring (MUSE) cell transplantation into infarct brain of mice. PLoS One 10, e0116009. Yang, Y., Liu, Q., Deng, S., Shao, Q., Peng, L., Ling, Y., Huang, Y., Zheng, S., Jiang, Q., Nie, D. and Chen, J. 2024. Human umbilical cord derived mesenchymal stem cells overexpressing HO-1 attenuate neural injury and enhance functional recovery by inhibiting inflammation in stroke mice. CNS Neurosci Ther. 30, e14412. Yaqubi, S. and Karimian, M. 2024. Stem cell therapy as a promising approach for ischemic stroke treatment. Curr. Res. Pharmacol. Drug Discov. 6, 100183. Yoon, C.W. and Bushnell, C.D. 2023. Stroke in women: a review focused on epidemiology, risk factors, and outcomes. J Stroke. 25, 2–15. Zeng, L., Hu, S., Zeng, L., Chen, R., Li, H., Yu, J. and Yang, H. 2023. Animal models of ischemic stroke with different forms of middle cerebral artery occlusion. Brain Sci. 13, 1007. Zhang, Le, X., Zhang, X.G., Huang, Y.R., Zheng, Y.Y., Ying, P.J., Zhang, X.J., Lu, X., Wang, Y.J. and Zheng, G.Q. 2021a. Stem cell-based therapy for experimental ischemic stroke: a preclinical systematic review. Front. Cell Neurosci. 15, 628908. Zhang, Z., Zou, X., Zhang, R., Xie, Y., Feng, Z., Li, F., Han, J., Sun, H., Ouyang, Q., Hua, S., Lv, B., Hua, T., Liu, Z., Cai, Y., Zou, Y., Tang, Y. and Jiang, X. 2021b. Human umbilical cord mesenchymal stem cell-derived exosomal miR-146a-5p reduces microglial-mediated neuroinflammation via suppression of the IRAK1/TRAF6 signaling pathway after ischemic stroke. Aging 13, 3060–3079. Zheng, H., Haroon, K., Liu, M., Hu, X., Xu, Q., Tang, Y., Wang, Y., Yang, G.Y. and Zhang, Z. 2024. Monomeric CXCL12-engineered adipose-derived stem cells transplantation for the treatment of ischemic stroke. Int. J. Mol. Sci. 25, 792. Zou, T., Fan, J., Fartash, A., Liu, H. and Fan, Y. 2016. Cell-based strategies for vascular regeneration. J. Biomed. Mater. Res. A. 104, 1297–1314. | ||
| How to Cite this Article |
| Pubmed Style Shannon GS, Rinendyaputri R, Sunarno S, Malik A. Effects of stem cell therapy on preclinical stroke. Open Vet. J.. 2025; 15(2): 601-618. doi:10.5455/OVJ.2025.v15.i2.9 Web Style Shannon GS, Rinendyaputri R, Sunarno S, Malik A. Effects of stem cell therapy on preclinical stroke. https://www.openveterinaryjournal.com/?mno=231121 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.9 AMA (American Medical Association) Style Shannon GS, Rinendyaputri R, Sunarno S, Malik A. Effects of stem cell therapy on preclinical stroke. Open Vet. J.. 2025; 15(2): 601-618. doi:10.5455/OVJ.2025.v15.i2.9 Vancouver/ICMJE Style Shannon GS, Rinendyaputri R, Sunarno S, Malik A. Effects of stem cell therapy on preclinical stroke. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 601-618. doi:10.5455/OVJ.2025.v15.i2.9 Harvard Style Shannon, G. S., Rinendyaputri, . R., Sunarno, . S. & Malik, . A. (2025) Effects of stem cell therapy on preclinical stroke. Open Vet. J., 15 (2), 601-618. doi:10.5455/OVJ.2025.v15.i2.9 Turabian Style Shannon, Gita Serafika, Ratih Rinendyaputri, Sunarno Sunarno, and Amarila Malik. 2025. Effects of stem cell therapy on preclinical stroke. Open Veterinary Journal, 15 (2), 601-618. doi:10.5455/OVJ.2025.v15.i2.9 Chicago Style Shannon, Gita Serafika, Ratih Rinendyaputri, Sunarno Sunarno, and Amarila Malik. "Effects of stem cell therapy on preclinical stroke." Open Veterinary Journal 15 (2025), 601-618. doi:10.5455/OVJ.2025.v15.i2.9 MLA (The Modern Language Association) Style Shannon, Gita Serafika, Ratih Rinendyaputri, Sunarno Sunarno, and Amarila Malik. "Effects of stem cell therapy on preclinical stroke." Open Veterinary Journal 15.2 (2025), 601-618. Print. doi:10.5455/OVJ.2025.v15.i2.9 APA (American Psychological Association) Style Shannon, G. S., Rinendyaputri, . R., Sunarno, . S. & Malik, . A. (2025) Effects of stem cell therapy on preclinical stroke. Open Veterinary Journal, 15 (2), 601-618. doi:10.5455/OVJ.2025.v15.i2.9 |