| Research Article | ||
Open Vet. J.. 2025; 15(2): 901-910 Open Veterinary Journal, (2025), Vol. 15(2): 901-910 Research Article Effects of probiotic supplementation on clinicopathological alterations in Barki sheep with MangeAsmaa A. Darwish*Animal Health and Poultry Department, Animal Production and Poultry Division, Desert Research Center (DRC), Cairo, Egypt *Corresponding Author: Asmaa A. Darwish. Animal Health and Poultry Department, Animal Production and Poultry Division, Desert Research Center (DRC), Cairo, Egypt. Email: asmaa_vet25 [at] yahoo.com Submitted: 07/12/2024 Accepted: 31/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
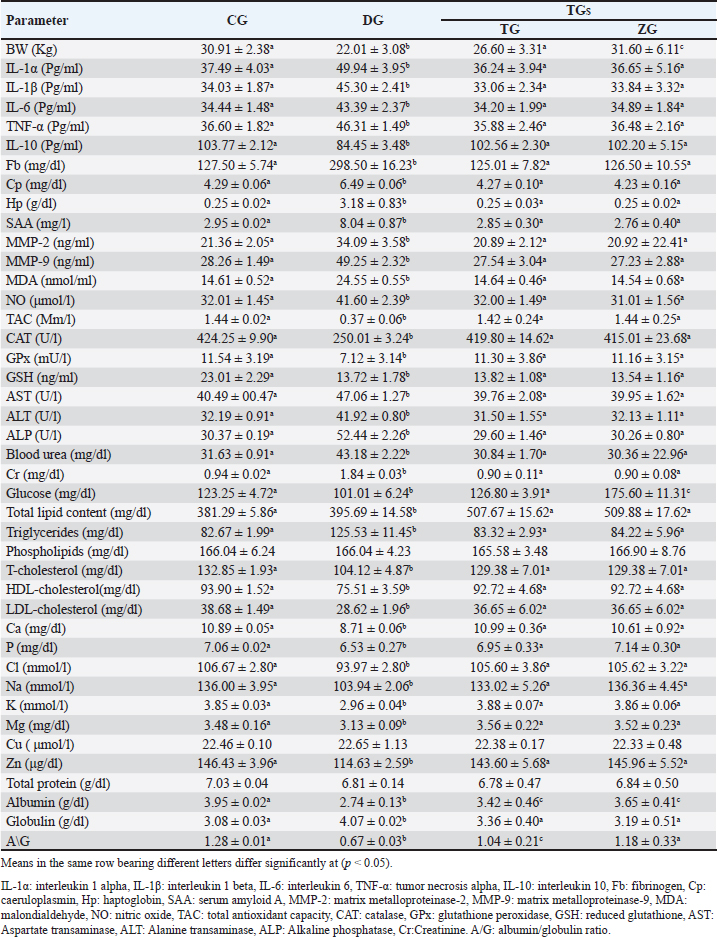
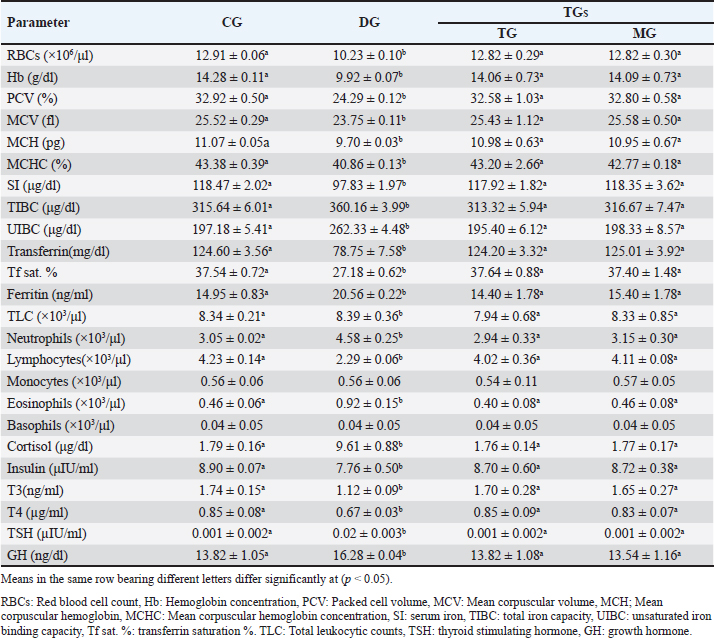
AbstractBackground: Ovine mange is a highly transmissible skin condition that typically results in significant economic losses. Aim: This research explored the clinicopathological alterations associated with mange in Barki sheep and evaluated the impact of integrating probiotics (ZAD) into the treatment protocol. Additionally, the study proposed diagnostic markers and indicators for assessing treatment prognosis. Method: Twenty healthy lambs [control group (CG)] and 20 lambs suffered from mange symptoms (confirmed by skin scrapings) in the diseased group (DG). DG was divided into two groups: the traditional group (TG) was injected once with 1 ml/50 kg ivermectin 1% (Promectine, Invesa) subcutaneously injected + topical Zinc ointment, and the ZAD Group (ZG): received the same treatment + 5 ml/ head of ZAD orally, repeated daily for 3 weeks. Skin scraping was repeated weekly until complete curing in both groups. Blood samples were collected from CG, DG (before treatment), and TG and ZG (after recovery). Result: DG (compared to CG) showed a significant (p < 0.05) innate immune response with hypercortisolemia and hypoinsulinemia, microcytic hypochromic anemia with leukocytosis and lymphocytopenia, hypoglycemia, hypoalbuminemia, hyperglobulinemia, hyperlipidemia, elevated liver and kidney function tests, decreased minerals and electrolytes, and hypozincemia. After treatment, the clinical symptoms and lesions completely recovered, and most of the estimated parameters reobtain their physiological ranges in TG and ZG on the 21st day. ZAD did not significantly impact the recovery or regression of lesions, but it markedly improved the diseased animals` body weights and serum glucose in ZG (compared to CG and TG). Total antioxidant capacity (TAC), interleukin-1β (IL-1β), and ferritin had the highest likelihood ratios among the estimated markers. Conclusion: Ovine scabies can lead to systemic and clinicopathological changes, necessitating careful monitoring and intervention. Adding ZAD to the treatment of mangy sheep might help enhance their body weights and serum glucose levels, but it does not affect the healing or reduction of lesions. TAC and IL-1β serve as dependable indicators for diagnosing ovine scabies, whereas TAC and ferritin are valuable for evaluating treatment effectiveness. Keywords: Ovine Scabies, Clinicopathological changes, Probiotics, Cytokines, Hormones, Oxidative stress, MMPs. IntroductionMange is a highly contagious skin disease noticed in all farm animals worldwide. The condition is caused by an ectoparasite called mite. Two types of mange are usually recorded in sheep: psoroptic mange (Psoroptes ovis mite infestation) and sarcoptic mange (Sarcoptes scabiei var ovis mite infestation) (Mohamed et al., 2020). Mite infestation is typically associated with scabs characterized by the rapid development of cutaneous inflammation and crusted skin lesions. The disease causes serious economic losses because of weight loss, skin and wool damage, lower milk production, worse pregnancy rate, weak lambs, increased susceptibility to other infectious diseases, additional food and acaricide costs, labor costs, and death. It is also a health hazard to breeders (Mohamed et al., 2020). In Egypt, mange infestation was noted in all farm animals in different governorates, with percentages exceeding 20% (Mohamed et al., 2020; Ahmed, et al., 2020; Dakroury and Darwish, 2021). Probiotics, which are direct-feed microbial, enhance dry matter intake and thus productivity, especially in animals under stressful conditions (Khattab et al., 2020; Abdel-Wahed et al., 2023). Since the beginning of the 21st century, numerous studies have emphasized the significant role that probiotics can play in animal husbandry. They are not just seen as simple additives anymore; instead, they are being explored as potential alternatives to conventional antibiotics. Recent research indicates that incorporating probiotics into animal feed can positively impact the gut microbiota, decrease pathogen shedding and disease symptoms, bolster gut immunity, and enhance disease resistance and overall health (Marwan and Singer, 2018, Gado, 2020). Moreover, probiotics exhibit antagonistic properties, effectively regulating gut microflora and substantially reducing the presence of foodborne pathogens such as Campylobacter, Clostridium perfringens, Escherichia coli, Listeria monocytogenes, Salmonella, and Staphylococcus aureus (Hekal, 2017). The probiotic ZAD, derived from anaerobic bacteria (Ruminococcus flavefaciens) at a concentration of 11 × 103 CFU per ml of water, is combined with specific enzymes such as cellulose (8.2 µ/g), hemicellulose (6.2 µ/mg), amylase (64.4 µ/g), and protease (12.3 µ/g) (Khattab et al., 2020). Its efficacy in improving animal productivity is notable, seen in enhanced weight gain, milk production, fertility, and bolstered immune system strength. Acting as a catalyst for existing internal bacteria within the animal’s rumen, ZAD facilitates nutrient extraction crucial for productivity, thereby enhancing feed conversion processes (Marwan and Singer, 2018; Gado, 2020). Moreover, ZAD effectively breaks down aflatoxins and elevates the immune response of animals consumed by ZAD. Furthermore, it promotes the activity of wool follicles, resulting in improved quality and quantity of wool, making it suitable for the wool industry (Hekal, 2017). This study aimed to explore the clinicopathological changes linked with mange in Barki sheep and assess the effects of incorporating ZAD into the treatment regimen for mangy ewes, focusing on the pro-inflammatory cytokines, acute phase proteins (APPs), matrix metalloproteinases (MMPs), and total antioxidant capacity (TAC) value in disease diagnosis and treatment evaluation. Materials and MethodsAfter the ethical approval of the animal and poultry health department, animal production and poultry division, Desert Research Center, Cairo, Egypt, code no. 28-10-24, 40 Barki-growing female lambs (6–8 months, 19–32 Kg B.W.) were kept in semi-closed clean well-ventilated pens on the farm of the Sustainable Development Center of Matrouh Resources. Lambs were fed mainly on concentrates (14%–16% protein), with a rate of 900–1,100 g/head/day, and hay with a rate of 350–600 g/head/day. Sufficient water was also provided. After clinical examination according to Nimmervoll et al. (2013) by inspection of each animal visually and by multiple fleece partings, followed by skin, inspection, and palpation across all parts of the animal body for the presence of gross lesions that may indicate the clinical form of mite infestation, the animals were divided into the following groups: Control group (CG): 20 apparently healthy lambs (intact skin completely covered with wool, no alopecia, physiological ranges of temp, respiratory rate, pulse, bright shiny eyes, and dry nostrils). Mangy group (DG): 20 lambs suffered from foci of dry alopecic skin in different body parts with a history of itching and pruritus, anemic mucous membrane, anorexia, and emaciation. These symptoms suggested mite infestation, and for more confirmation, skin scrapings were obtained from the periphery of the clinical lesions and placed in labeled Petri dishes. To prevent mites from escaping, vaseline was applied at the edges of the dishes. The collected samples were examined under a microscope. Skin scrapings examinationFollowing the protocol established by Kotb and Abdel-Rady (2015), Petri dishes containing skin scrapings were warmed to approximately 38°C and inspected using a stereoscopic microscope to identify various mite stages. Scrapings that tested negative for mites were transferred to test tubes containing 10 ml of 10% KOH solution and heated for 5 minutes in a water bath or beaker. Subsequently, the tubes were centrifuged for 3 minutes at 2,000 rpm, and the supernatant was discarded. 5 ml of water was added to the sediment, followed by another round of centrifugation. The supernatant was once again discarded, and a drop of the sediment was examined under a microscope to detect different mite stages. Then, the DG was divided into two treatment groups: Traditional group (TG): 10 mangy lambs were treated with a single dose of ivermectin 1% (Promectine, Invesa) and subcutaneously injected into the diseased group at a dose (1 ml/50 kg) + 10% zinc ointment was applied on the skin lesion daily until complete cure. ZAD Group (ZG): Received the same treatment + 5 ml/head of ZAD orally, repeated daily for 3 weeks (the dose and route of administration were recommended by the manufacturing company). Skin scraping was repeated on the 7th, 14th, and 21st days till the complete curing in both groups. Blood samples5-ml blood was collected from the CG, DG (before treatment), TG, and ZG (after recovery). Each blood sample was divided into three parts. The first part was collected using Na2EDTA and used for the manual estimation of hemogram and leukogram parameters according to the method described by Feldman et al. (2000). The second part was collected on citrate to prevent the coagulation process and centrifuged to obtain plasma at 3000 rpm for 20 minutes to measure Fb plasma levels using ELISA kits from IBL International Crop (Canada)®. The third part was collected in a plain test tube, allowed to coagulate, and centrifuged at 3000 rpm. for 20 minutes to obtain serum for detection of various parameters including total protein, Albumin, kidney function tests [urea, creatinine (Cr)], hepatic enzymes [alanine transaminase, aspartate transaminase, alkaline phosphatase (ALP)], minerals (Ca, P, Mg), electrolytes (Na, K, Cl), trace elements (Cu, Zn), glucose, total lipids, phospholipids, triglycerides, T/LDL/HDL-cholesterol, free radicals [nitric oxide (NO), malondialdehyde (MDA)], TAC, antioxidants [catalase (CAT), glutathione peroxidase (GPx), reduced glutathione (GSH)], iron (SI), and total iron binding capacity (TIBC) spectrophotometrically using kits from Biodiagnostic Company®, serum pro-inflammatory cytokines (IL-1α, IL-1β, IL-6, TNF-α), anti-inflammatory cytokine (IL-10), and hormones (Cortisol, growth hormone (GH), thyroid stimulating hormone (TSH), T3, T4, Insulin) levels were measured in serum by ELISA kits of MyBioSource company®, serum MMPs concentrations by using Cloud-Clone Corp company® ELISA kits, serum amyloid A (SAA), and serum haptoglobin (Hp) using ELISA kits from IBL International Crop (Canada)®, Serum caeruloplasmin (Cp), and serum transferrin (Tf) by a turbidimetric method using Elabscience USA® kits, and serum ferritin by CLIA method using Abnova® (Taipei) kits. All manual instructions were carefully followed. - Transferrin saturation percent (TF sat. %)=SI/TIBC*100. - Unsaturated iron binding capacity (UIBC)=TIBC-SI. Statistical analysisMeans of different statistical parameters between the studied groups were compared by one-way ANOVA test, and post hoc differences between means were determined by a multiple comparison Tukey`s HSD test using SPSS version 24 at a 0.05 level of probability. GraphPad Prism version 8 software was used to evaluate the area under the curve (AUC), cut-off points, sensitivity, specificity, and likelihood ratio (LR) for the measured proinflammatory cytokines, APPs, MMPs, and TAC between DG and CG as well as between TGs and DG. The positive predictive value (PPV), negative predictive value (NPV), and accuracy rate were calculated according to the next equations: PPV=True positive ÷ Total positive × 100. NPV=True negative ÷ Total negative × 100. Accuracy rate=(True positive + True negative) ÷ Total population × 100. ResultsThe microscopical examination of the skin scraps confirmed the presence of mite infestation in DG. DG (compared to CG) displayed a significant (p < 0.05) decrease in body weights as well as a prominent innate immune response represented by the significant (p < 0.05) elevation of the serum pro-inflammatory cytokines (IL-1α, IL-1β, IL-6, TNF-α), APPs (Fb, Cp, SAA, Hp), free radicals (NO, MDA), MMPs (MMP-2, MMP-9) concentrations, and the significant (p < 0.05) decline of the IL-10, TAC and antioxidant enzymes (GSH, GPx, CAT) concentrations (Table 1). The serum levels of glucose, Alb, A/G, T/HDL/LDL-cholesterol, minerals (Ca, P, Mg), electrolytes (Na, K, Cl), and trace elements (Zn) were significantly (p < 0.05) downregulated in DG compared with CG, whereas the serum levels of Glob, Urea, Cr, liver enzymes, total lipids, and triglycerides significantly (p < 0.05) upregulated in DG compared with CG. TP, Cu, and phospholipids were non-significantly (p ≥ 0.05) changed in DG compared to CG (Table 1). The hemogram of DG (compared to CG) revealed a significant (p < 0.05) diminishing in (RBCs, Hb, PCV, MCV, MCH, MCHC) and lymphocyte counts, with a significant (p < 0.05) increment in neutrophil and eosinophil counts. TLC, monocytes, and basophils non-significantly (p ≥ 0.05) changed between DG and CG. In addition, DG (compared to CG) exhibited a significant (p < 0.05) decrease in SI, transferrin, Tf. Sat. %, insulin, T3, and T4, as well as a significant (p < 0.05) increase in serum levels of ferritin, TIBC, UIBC, cortisol, GH, and TSH (Table 2). Post-treatmentA marked gradual improvement in scabies clinical signs and a prominent regression of intense lesions were noticed in both treated groups (TG and ZG) from the seventh day post-treatment until the end of the experiment. Microscopically, a gradual decline in the living mite count was observed in TG and ZG compared with the zeroth day. By the 21st day, all treated lambs recovered completely, all clinical signs disappeared, skin scrapes were clean (no living mites), and wool follicles reappeared. There were no differences in the recovery rate or lesion regression between the two groups. In turn, most of the prior clinicopathological parameter levels significantly (p < 0.05) increased or decreased in TG and ZG until approaching the CG levels [non-significantly (p ≥ 0.05) changed with CG]. Interestingly, ZG showed a significant increase in body weight and serum glucose compared with CG and TG, and both groups exhibited a significant decrease in Alb compared with CG, while TG demonstrated a significant decrease in A/G compared with CG and ZG (Tables 1 and 2). Regarding the value of the estimated pro-inflammatory cytokines, APPs, TAC, and MMPs as indicators for ovine scabies and its treatment follow-up, all of them yielded AUC=1, sensitivity, and NPV as 100%, and high specificity, PPV, and AR ≥70% (except Hp in the treatment monitoring its specificity=65%), but their LRs differed, TAC had a high LR as 20 in both the disease diagnosis and its treatment monitoring, IL-1β had LR as 20 in the disease indication only while ferritin had LR as 20 in the disease treatment evaluation, the rest markers yielded low to moderate LRs either in the disease diagnosis or its treatment evaluation (Table 3). DiscussionMange is a distressing condition characterized by a severe hypersensitive response of sheep to the fecal material of parasitic mites. In this study, as well as in previous studies, mangy lambs suffered from dermatitis, which is marked by intense itching and self-inflicted injuries due to scratching. Anorexia, emaciation, and pale mucous membranes were also noted in mangy animals (Mohamed et al., 2020; Ahmed, et al., 2020; Dakroury and Darwish, 2021). Table 1. Body weight, immunological, and biochemical parameters of the studied groups. Value=mean ± SD.
Table 2. Erthyrogram, iron profile parameters, leukogram, and serum hormone levels of the studied groups. Value=mean ± SD.
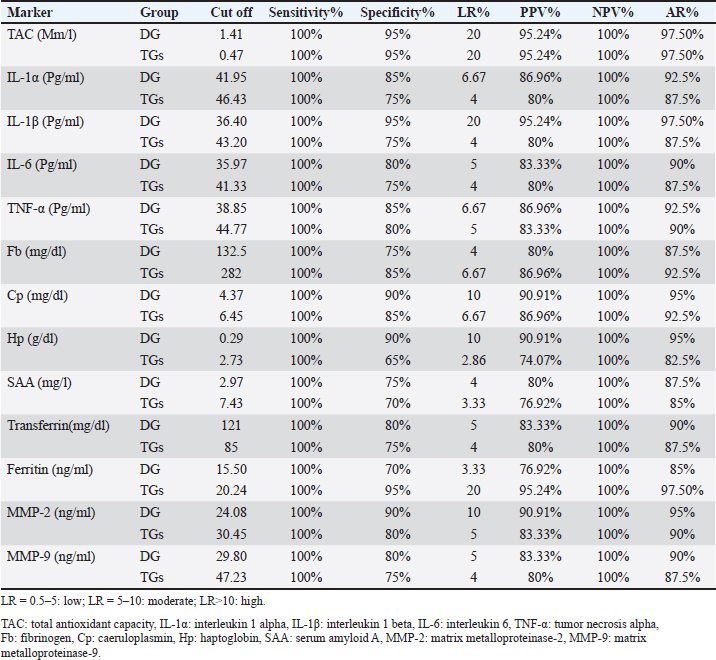
Consistent with these symptoms, DG displayed a noticeable innate immune response, which started with an elevation of the serum pro-inflammatory cytokines (IL-1α, IL-1β, IL-6, TNF-α) levels in DG. Whereas the living mite usually deposits its allergic excretory/secretory products onto the host`s skin’s outer layer. These products disrupt skin barrier function through their proteolytic activity. They cleave intercellular tight junctions, thus breaching the epidermis and increasing epithelial permeability. This digestion activity may enable mite antigens to enter the subepidermal layer and interact with antigen-presenting cells, leading to further allergic sensitization and inflammatory immune response induction (Bhat et al., 2017; Chen, 2021; Chen et al., 2021; Dinçer et al., 2022; Sharaf, 2024). The inflammatory immune response to mite infestation is performed, maintained, and reinforced through the upregulation of pro-inflammatory cytokines. These pro-inflammatory cytokines promote keratinocyte proliferation, resulting in hyperkeratotic scabs, and increase the expression of adhesion molecules, such as selecting, integrins, and intercellular adhesion molecules, on endothelial cells, potentially leading to immune cell extravasation of neutrophils, eosinophils, and basophils to the site of skin damage. In this manner, these pro-inflammatory cytokines provide for innate defense and influence the type of adaptive response that follows. On the contrary, IL-10 production is reduced in DG, leading to the expansion of pro-inflammatory cytokine action and intensifying the situation (Bhat et al., 2017; Abdallah et al., 2023; Hassan et al., 2020; Chen, 2021; Chen et al., 2021; Dinçer et al., 2022; Sharaf, 2024). Table 3. Cut off points, sensitivity, specificity, likelihood ratio (LR), positive predictive value (PPV%), negative predictive value (NPV%), and accuracy rate% (AR%)of the suggested markers in DG (compared to CG) and in TGs (compared to DG).
Another component of the inflammatory immune response is acute phase proteins. APPs are hepatic proteins that circulate at low levels in the serum of healthy individuals. Their titer increases rapidly following the onset of the disease and persists in the blood for a long time. They return to preinfestation levels soon after successful treatment or disease resolution. These properties suggest that APPs are excellent biomarkers for various animal and human diseases. In the current data, APPs production markedly enhanced in DG. Similar observations were recorded in camels and sheep with scabies by Hassan et al. (2020) and Wells et al. (2013), respectively. They emphasized APPs (especially SAA) values as indicators of both infestation and successful treatment. MMPs are zinc-containing endopeptidases responsible for extracellular matrix breakdown and cleavage. Among them, MMP-2 and MMP-9 are known as gelatinases, which specialize in degrading gelatin but can also target other proteins such as collagens and elastin (Darwish, 2020). They play crucial roles in various physiological processes, including angiogenesis, endothelial cell apoptosis, vascular basement membrane degradation, cellular infiltration facilitation, and pro-inflammatory mediator modulation. The activities of MMPs are tightly regulated by tissue inhibitors of metalloproteinases (TIMPs) (Darwish, 2020). Any imbalance between MMPs and TIMPs is closely associated with the pathogenesis of numerous diseases, notably malignancies and inflammatory conditions. MMP-2 and MMP-9 displayed a marked increase in DG in the present data, which mimicked previous opinions that nearly all parasite infections use MMP-2 and MMP-9 to remodel tissue, which often slows down the production of ECM molecules, including collagen II and aggrecan (Darwish and Eldakroury, 2020; Elmaidomy et al., 2023). In our study, mangy lambs exhibited elevated MDA, and NO levels, accompanied by reduced antioxidant and TAC. This result implies potent oxidative stress. Inflammation associated with ovine mange enhances the proliferation of inflammatory cells. These cells, especially neutrophils and macrophages, are recruited during inflammation and release free radicals that exert potent cytotoxic effects against parasites and pathogens. The presence of oxidative stress is further evidenced by lipid peroxidation, which is a well-established mechanism of cellular damage. MDA, a major by-product of lipid peroxidation, is elevated in various diseases, including parasitic infestations. Increased lipid peroxidation can compromise skin integrity by altering its structure and permeability. Oxidative stress is a common finding in scabies and other ectoparasite infestations in different animals (Aktas et al., 2017; Mohamed et al., 2017; Al-Hosary et al., 2018; Ural et al., 2018; Atteya et al., 2019; Darwish and Eldakroury, 2020; Hassan et al., 2020; Abdallah et al., 2023). The reduced levels of antioxidants undoubtedly worsened the disease, intensifying the disease’s severity. The buildup of free radicals due to the lack of antioxidant control targets various body cells, including red blood cells, hepatocytes, and kidney cells. This resulted in the depicted anemia and elevated liver and kidney function tests in DG in the current study (Mohamed et al., 2017; Darwish and Eldakroury, 2020). Rationally, liver injury in DG was associated with the recorded hypoglycemia, hypoalbuminemia, and T/HDL/LDL-hypocholesterolemia in DG. The liver plays a crucial role in blood glucose regulation, albumin synthesis, and cholesterol formation (Darwish and Eldakroury, 2020). Similarly, renal insufficiency leads to a disturbance in mineral and electrolyte levels in DG blood because the kidneys are unable to effectively retain minerals and electrolytes from urine. Anorexia and subsequent malnutrition are also possible causes of noticed anemia, hypoglycemia, hypoalbuminemia, T/HDL/LDL-hypocholesterolemia, decreased minerals and electrolytes, and hypozincemia in DG (Vishe et al., 2012; Abdalrahman and Mustafa, 2018; Darwish and Eldakroury, 2020; Hassan et al., 2020). On the other hand, hypertriglyceridemia and subsequent total hyperlipidemia in DG are attributed to intense fat body lipolysis to save energy and maintain the body’s vital processes (Darwish and Eldakroury, 2020). Hypoglycemia, stress related to mite infestation, and activated pro-inflammatory cytokines induce multiple endocrine changes, mainly hypercortisolemia. Hypercortisolemia was reported before in noticed before in mange infestation in rabbits by Dakroury and Darwish (2021). Pro-inflammatory cytokines directly affect the HPA axis (hypothalamus-pituitary-adrenal axis) and increase pituitary adrenocorticotropic hormone secretion. This, in turn, increases cortisol production from the adrenal cortex, resulting in significant hypercortisolemia in DG. Cortisol has two primary functions in this context. First, it stimulates hepatic gluconeogenesis, enhances the transfer of fatty acids from adipose tissue to the liver, and suppresses thyroid and pancreatic β cell activity, which leads to hypoinsulinemia and decreased T3 and T4 levels in DG (Francisco et al., 2018; Dakroury and Darwish, 2021). Consequently, cellular glucose uptake decreases, hypoglycemia related to the disease is corrected, and the energy necessary for immune response and host survival is provided. Second, cortisol inhibits the activity of pro-inflammatory cytokines, thereby protecting the body from their adverse effects. The activation of the hypothalamus-pituitary unit also triggers the release of GH, which improves the immune response and counteracts the immunosuppressive effects of cortisol (Francisco et al., 2018; Dakroury and Darwish, 2021). The observed increase in TSH levels may be attributed to the pituitary response to hypothyroidism. Regarding the protein profile of DG, it illustrated a noticeable hyperglobulinemia, which indicates the body’s response to the antigenic stimuli of the mites and its metabolic products and toxins by different immune protein production (cytokines, APPs, MMPs, Igs) (Vishe et al., 2012; Darwish and Eldakroury, 2020). In contrast, during infection, the body ignores albumin synthesis, concentrating on other globulin formations (cytokines, APPs, MMPs, Igs) (Abdalrahman and Mustafa, 2018, Dakroury and Darwish, 2021). This explained the hypoalbuminemia in DG in the current data and the subsequent decrease in the A/G ratio. It is worth mentioning that the insignificant alterations in TP in DG here mean that albumin and globulin experienced similar percentage shifts. In agreement with previous studies, the hemogram of DG in our work exhibited microcytic hypochromic anemia (Vishe et al., 2012; Onoja and Shoyinka, 2016; Darwish and Eldakroury, 2020; Salvi et al., 2022). In addition to prior oxidative stress and anorexia, this result was assigned to pro-inflammatory cytokines activity, which inhibits the erythropoiesis process through several mechanisms. They interfere with iron gut absorption, prevent iron binding with transferrin by increasing hepcidin secretion, enhance ferritin formation, and decrease transferrin production. This is a protective mechanism aimed at lowering iron bioavailability in infested mites. Thus, it is prevented from reproducing and threatening its survival. In chronic, prolonged infestation, this action unfortunately causes outstanding anemia (Vishe et al., 2012; Onoja and Shoyinka, 2016; Darwish and Eldakroury, 2020; Salvi et al., 2022). The iron profile of DG confirmed this theory, as it displayed pronounced hypoferemia, hypotransferrinemia, and hyperferritinemia. The above-described APR may also be involved in iron profile changes, as transferrin is a negative APP (decreased during infection) and ferritin is a positive APP (increased during infection). Logically, increased TIBC and UIBC and decreased Tf sat. % in DG in our results are consequences of hypoferemia. Similar findings were noticed in camels infested with scabies by Darwish and Eldakroury (2020). The leukogram of DG in our data revealed leukocytosis due to neutrophilia and eosinophilia. Pro-inflammatory cytokines likely drive this response by promoting the production of leukocytes in bone marrow, their release into circulation, and their migration to skin damage sites, particularly neutrophils and eosinophils, to combat and eliminate mite infestation. Eosinophilia is a common response to parasitic infestation, whereas neutrophilia may indicate a secondary bacterial infection (Vishe et al., 2012; Onoja and Shoyinka, 2016; Salvi et al., 2022). In addition, the recorded hypercortisolemia in DG in this study may play a role in the observed alterations in the DG leukogram results. Hypercortisolemia is known to redistribute circulatory leukocytes, leading to a shift of neutrophils from the marginating pool to the circulating pool at the expense of lymphocytes. The lymphopenia noted in DG supported this hypothesis Darwish and Eldakroury (2020). Ivermectin effectively controlled the mange infestation, leading to complete recovery of skin lesions in both treated groups by the 21st day, with no living mites detected in skin scrapings, and restoring the normal ranges of most of the above-described immunological and clinicopathological parameters. This outcome aligns with previous findings highlighting ivermectin’s efficacy against various mite infestations (Darwish and Eldakroury, 2020; Mohamed et al., 2020; Dakroury and Darwish, 2021). The mechanism involves the induction of parasite death and paralysis by enhancing membrane permeability to chloride ions, thereby disrupting nerve cell activity in nematodes and muscle cells in arthropods (Darwish and Eldakroury, 2020). In addition, ivermectin promotes the production of the inhibitory neurotransmitter GABA in presynaptic neurons. Its broad tissue distribution, particularly in adipose tissue and skin, ensures prolonged action against diverse ectoparasites across various developmental stages (Dakroury and Darwish, 2021). Notably, zinc ointment at 10% concentration speeds up the reappearance of wool follicles, besides its soothing anti-inflammatory effect on the skin in both groups (Gupta et al., 2014; Onoja et al., 2021). Although the addition of ZAD to the treatment protocol for mangy sheep did not significantly impact the recovery or regression of lesions, it markedly improved the diseased animals` body weights and serum glucose (compared to CG and TG). This is because of its high content of digestive enzymes such as cellulose, hemicellulose, amylase, and protease (Khattab et al., 2020). Thus, it facilitates nutrient extraction and enhances feed conversion processes and body weight (Marwan and Singer, 2018; Gado, 2020). The slight hypoalbuminemia detected in TG and ZG (compared to CG) after treatment may be assigned to oxidative stress connected with ivermectin administration and related liver injury. Similarly, Darwish and Eldakroury (2020) reported hypoalbuminemia in mangy she-camels until the 28th day after ivermectin injection. Interestingly, the degree of this hypoalbuminemia was greater in TG than in ZG, so A/G decreased in TG only. The present study found that pro-inflammatory cytokines, APPs, MMPs, and TAC serve as valuable biomarkers for diagnosing ovine scabies and assessing treatment effectiveness. These markers demonstrated high AUCs, sensitivities, and NPVs and acceptable specificities, PPVs, and ARs, with TAC emerging as the most reliable indicator overall (for the disease diagnosis and treatment evaluation). IL-1β proved effective for diagnosing the disease, while ferritin was effective in predicting the treatment outcomes. Although these findings align with prior research emphasizing the significance of these markers in various animal and human diseases, they differ in their ranking (Wells et al., 2013; Darwish and Eldakroury, 2020; Hassan et al., 2020; Abdallah et al., 2023; Sharaf, 2024). This variance may stem from differences in animal species, disease progression, and geographical factors. ConclusionInfestation of sheep with mites not only harms the skin but also induces systemic alterations that can profoundly affect the animals’ physiology and health. For full recovery, monitoring all systemic changes and intervening as needed are essential, such as addressing anemia, oxidative stress, and decreased minerals and electrolytes. ZAD addition to the treatment of mangy sheep may be useful in improving the diseased animals` body weights and serum glucose but it does not impact the recovery or regression of lesions. TAC is a reliable indicator for the diagnosis of ovine scabies and its treatment evaluation, IL-1β is an effective tool for diagnosing the disease only, and ferritin is good for the treatment evaluation only. AcknowledgmentThe author acknowledges all members of the Animal Health and Poultry Department (DRC) for their sincere guidance and cooperation. Conflict of interestThe authors declare no conflict of interest. Authors` contributionAD designed the research, collected the blood samples, performed the clinicopathological, and statistical analyses of the data, and wrote the paper. ReferencesAbdallah, M.S., Sallam, N.H., Abdelnaeim, N.S., Mandour, M.A., Saad Eldin, W.F. and Abouelhassan, E.M. 2023. Therapeutic management, clinicopathological, molecular and cost studies on Sarcoptes scabiei infestation in rabbit. J. Adv. Vet. Res. 13(3), 333–338. Available via https://www.advetresearch.com/index.php/AVR/article/view/1223 Abdel-Wahed, A.M., Khattab, I.M., Zaher, M., Khattab, A.S., El-Damrawy, S.Z., El-Keredy, A. and Soltan, Y.A. 2023. Growth performance, nutrient utilization, rumen fermentation, blood biochemistry, and carcass traits of lambs fed Atriplex nummularia L. hay-based diet supplemented with yeast or bacterial direct- fed microbial. Anim. Biotechnol. 34(7), 2132–2140; doi:10.1080/10495398.2022.2077739. Abdalrahman, B.M. and Mustafa B.H.S. 2018. Effect of tick and lice on some blood constitute and trace elements of local black goat in Sulaimani, Kurdistan Region of Iraq. KJAR. 3(2), 42–46. Ahmed, M.A., Elmahallawy, E.K., Gareh, A., Abdelbaset, A.E., El-Gohary, F.A., Elhawary, N.M., Dyab, A.K., Elbaz, E. and Abushahba, M.F.N. 2020. Epidemiological and histopathological investigation of Sarcoptic Mange in Camels in Egypt. Animals 10, 1485. doi:10.3390/ani10091485. Aktas, M.S., Kandemir, F.M. Kirbas, A. Hanedan, B. and Aydin, M.A. 2017. Evaluation of oxidative stress in sheep infected with Psoroptes ovis using total antioxidant capacity, total oxidant status, and malondialdehyde level. J. Vet. Res. 61, 197–201; doi:10.1515/jvetres-2017-0025. Atteya, M.M., Ghobashy, M.A., Wahba, A.A. and Abouelhassan, E.M. 2019. Evaluation of the prevalence and oxidative status in Sheep infected with Sarcoptic Scabiei in Ismailia Governorate, Egypt. EVMSPJ. 15,114–129. Al-Hosary, A.A., Abd Ellah, M.R. and Ahmed, L.S. 2018. Evaluation of oxidative stress in sheep infested with ticks and concurrent diagnosis of Theileriosis. Asian J. Anim. Vet. Adv. 13, 263–268; doi:10.3923/ajava.2018.263.268. Bhat, S.A., Mounsey, K.E., Liu, X. and Walton, S.F. 2017. Host immune responses to the itch mite, Sarcoptes scabiei, in humans. Parasite Vectors. 10(1), 385; doi:10.1186/s13071-017-2320-4. Chen, Z. 2021. Investigations into potential alternatives for sustainable control of psoroptic mange in Belgian Blue cattle. PhD dissertation, Ghent University. Faculty of Veterinary Medicine, Merelbeke, Belgium. Chen, Z., Claerebout, E., Chiers, K., Pas, M., Pardon, B., van Mol, W., Casaert, S., De Wilde, N., Duchateau, L. and Geldhof, P. 2021. Dermal immune responses against Psoroptes ovis in two cattle breeds and effects of anti-inflammatory dexamethasone treatment on the development of psoroptic mange. Vet. Res. 52, 1; 10.1186/s13567-020-00874-x. Dakroury, M.F. and Darwish, A.A. 2021. A comparative pharmacological study on moxidectin and propolis ointment in rabbits naturally infested with Psoroptes cuniculi. Iraqi J. Vet. Sci. 35(4), 725–731. Darwish, A.A. 2020. Investigation of matrix metalloproteinase-2 and matrix metalloproteinase-9 activity in some inflammatory conditions in Barki sheep. Vet. Sci. Res. Rev. 6(1), 25–32; doi:10.17582/journal.vsrr/2020/6.1.25.32. Darwish, A.A. and Eldakroury, M.F. 2020. The effect of ivermectin injection on some clinicopathological parameters in camels naturally infested with scabies. Adv. Anim. Vet. Sci. 8(s2), 34–40; doi:10.17582/journal.aavs/2020/8.s2.34.40. Dinçer, P.F., Gökce- cik, Ö.F. and Başa, A. 2022. Evaluation of interleukin and vitamin levels in sheep infested with sarcoptes scabiei. Vet. Sci. Pract. 17(1), 11–15. Elmaidomy, A.H., Abdel-Maqsoud, N.M.R., Tammam, O.Y., Abdel-Rahman, I.M., Elrehany, M.A., Bakhsh, H.T., Altemani, F.H., Algehainy, N.A., Alzubaidi, M.A., Alsenani, F., Sayed, A.M., Abdelmohsen, U.R. and Zahran, E.M. 2023. Egyptian mandarin peel oil’s anti-scabies potential via downregulation-of-inflammatory/immune-cross-talk: GC-MS and PPI network studies. Sci. Rep. 13(1), 14192; 10.1038/s41598-023-38390-5. Feldman, B.F., Zink, J.C. and Jain, N.C. 2000. “Schalm’s Veterinary Hematology”, 5th ed. Philadelphia, PA: Lippincott Williams & Wilkins. Francisco, J.R., Naïma, M. and Martins J.O. 2018. Interplay between Hormones, the Immune System, and Metabolic Disorders. Mediators Inflamm. 2018, 8654212; 10.1155/2018/8654212 Gado, H.M. 2020. Utilization of anaerobic microbiology to improve animal production. Egyptian J. Anim. Prod. 57, 81–86. Gupta, M., Mahajan, V.K., Mehta, K.S. and Chauhan, P.S. 2014. Zinc therapy in dermatology: a review. Dermatol Res Pract. 2014, 709152; doi:10.1155/2014/709152. Hassan, H., Gadallah, S., Kamr, A. and Arbaga, A. 2020. Acute phase proteins, trace elements and cytokines expression as a diagnostic and prognostic biomarker in diseased camel. J. Curr. Vet. Res. 2(1), 93–100; doi:10.21608/jcvr.2020.90229. Hekal S.A. 2017. Effect of biological additives supplementation on skin and coat characteristics of Barki sheep. Int. J. Adv. Res. 5, 951–962. Khattab, I.M., Abdel-Wahed, A.M., Khattab, A.S., Anele, U.Y., El-Keredy, A. and Zaher, M. 2020. Effect of dietary probiotics supplementation on intake and production performance of ewes fed Atriplex hay-based diet. Livestock Sci. 237, 104065; doi:10.1016/j.livsci.2020.104065. Kotb, S. and Abdel-Rady, A. 2015. Sarcoptic mange of camel in upper Egypt: prevalence, risk assessment, and control measures. J. Adv. Vet. Anim. Res. 2(4), 410–417. Marwan, A.A. and Singer, A.M. 2018. Influence of exogenous enzymes supplementation on in vitro ruminal fermentation and reproductive performance of sheep. Egyptian J. Nutr. Feeds 21(3), 645–656. Mohamed, R.I., Mohamed, D.I., Khalil, M., ELmasry, N.M. and Rasheed, N. 2017. Histopathological, cinico-biochemical and therapeutic studies on different types of mange in domestic rabbits. Assiut Vet. Med. J. 63, 90–101. Mohamed, E., Khalafallah, S. and Metawea, Y. 2020. Epidemiology and control of mange in sheep and goat in menoufia governorate. Benha Vet. Med. J. 39(2), 111–116; doi:10.21608/bvmj.2020.53293.1313. Nimmervoll, H., Hoby, S., Robert, N., Lommano, E., Welle, M. and Ryser-Degiorgis, M. 2013. Pathology of Sarcoptic mange in red foxes (Vulpes Vulpes): Macroscopic and histologic characterization of three disease stages. J. Wildlife Dis. 49, 91–102. Onoja, R.I. and Shoyinka, S.V.O. 2016. Effects of lesion distribution on the hematology and some biochemical indices of Sarcoptes scabiei infested West African Dwarf goat. J. Adv. Parasitol. 3(3), 66–74; doi:10.14737/journal.jap/2016/3.3.66.74. Onoja, R.I., Anyogu, D.C. and Shoyinka, S.V. 2021. The relationship between Serum Zinc, Copper and vitamin A concentration and the severity of lesions in Sarcoptic Mange of goat. AJVS. 69(2), 126–132; doi:10.5455/ajvs.45643. Salvi, R.V., Devi, S., Prajapati, A.S., Parmar, M.P., Bharai, M.V., Parikh R. and Patel. J.R. 2022. Clinico-hemato-biochemical findings and therapeutic management of MEHSANA goats infested with scabies. Pharm. Inno. J. 11(11), 2018–2020. Sharaf M.S. 2024. Scabies: immunopathogenesis and pathological changes. Parasitol. Res. 123(3), 149; doi:10.1007/s00436-024-08173-6. Ural, K., Ural, T.R., Çamkerten, I., Erdoğan, H., Ayan, A., Gültekin, M., Haydardedeoğlu, A.E., Aysul, N. and Erdoğan S. 2018. Interpretation of Serum 25-Hydroxy vitamin D3 concentrations in sheep with naturally occurring sarcoptic mange. JAVST 3(1), 35–40. Vishe, H.P., Pawar, K., Gupta, H.K. and Rao, G.S. 2012. Prevalence and hemato-biochemical studies in parasitic and non parasitic dermatological disorders in Surti buffalo and buffalo calves. Vet. World. 5(4), 230–235; doi:10.5455/vetworld.2012.230-235. Wells, B., Innocent, G.T., Eckersall, P.D., McCulloch, E., Nisbet, A.J. and Burgess, S.T. 2013. Two major ruminant acute phase proteins, haptoglobin and serum amyloid A, as serum biomarkers during active sheep scab infestation. Vet. Res. 44, 103; doi:10.1186/1297-9716-44-103. | ||
| How to Cite this Article |
| Pubmed Style Asmaa A. Darwish. Effects of probiotic supplementation on clinicopathological alterations in Barki sheep with mange. Open Vet. J.. 2025; 15(2): 901-910. doi:10.5455/OVJ.2025.v15.i2.39 Web Style Asmaa A. Darwish. Effects of probiotic supplementation on clinicopathological alterations in Barki sheep with mange. https://www.openveterinaryjournal.com/?mno=232017 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.39 AMA (American Medical Association) Style Asmaa A. Darwish. Effects of probiotic supplementation on clinicopathological alterations in Barki sheep with mange. Open Vet. J.. 2025; 15(2): 901-910. doi:10.5455/OVJ.2025.v15.i2.39 Vancouver/ICMJE Style Asmaa A. Darwish. Effects of probiotic supplementation on clinicopathological alterations in Barki sheep with mange. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 901-910. doi:10.5455/OVJ.2025.v15.i2.39 Harvard Style Asmaa A. Darwish (2025) Effects of probiotic supplementation on clinicopathological alterations in Barki sheep with mange. Open Vet. J., 15 (2), 901-910. doi:10.5455/OVJ.2025.v15.i2.39 Turabian Style Asmaa A. Darwish. 2025. Effects of probiotic supplementation on clinicopathological alterations in Barki sheep with mange. Open Veterinary Journal, 15 (2), 901-910. doi:10.5455/OVJ.2025.v15.i2.39 Chicago Style Asmaa A. Darwish. "Effects of probiotic supplementation on clinicopathological alterations in Barki sheep with mange." Open Veterinary Journal 15 (2025), 901-910. doi:10.5455/OVJ.2025.v15.i2.39 MLA (The Modern Language Association) Style Asmaa A. Darwish. "Effects of probiotic supplementation on clinicopathological alterations in Barki sheep with mange." Open Veterinary Journal 15.2 (2025), 901-910. Print. doi:10.5455/OVJ.2025.v15.i2.39 APA (American Psychological Association) Style Asmaa A. Darwish (2025) Effects of probiotic supplementation on clinicopathological alterations in Barki sheep with mange. Open Veterinary Journal, 15 (2), 901-910. doi:10.5455/OVJ.2025.v15.i2.39 |