| Case Report | ||
Open Vet. J.. 2025; 15(10): 5383-5390 Open Veterinary Journal, (2025), Vol. 15(10): 5383-5390 Case Report Successful treatment and long-term follow-up of a young cat with feline infectious peritonitis and renal lymphomaIlse Beekhuis1, Goncalos Serrano2, Angel Almendros3,4 and Antonio Giuliano5*1MVetMed Anicura Haaglanden, Rijswijk, Netherlands 2Small Animal Department, Faculty of Veterinary Medicine, Ghent University, Belgium 3Department of Veterinary Clinical Sciences, Jockey Club College of Veterinary Medicine, City, China 4University of Hong Kong, Kowloon, Hong Kong 5Harvest Veterinary Oncology Center, Kwai Chung, Kowloon, Hong Kong, Hong Kong *Corresponding Author: Antonio Giuliano. Harvest Veterinary Oncology Center, Kwai Chung, Kowloon, Hong Kong, Hong Kong. Email: dr.giuliano [at] hvoc.com.hk Submitted: 22/01/2025 Revised: 27/08/2025 Accepted: 23/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
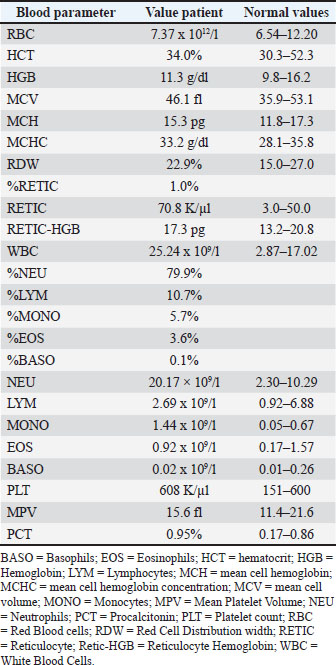
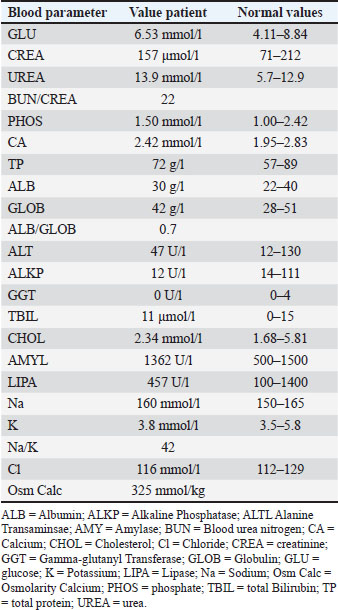
AbstractBackground: Feline lymphoma is often a fatal disease, and achieving a cure with chemotherapy is questionable. Feline infectious peritonitis (FIP) diagnosis was historically associated with a poor prognosis and high mortality, but remission is possible with the newly available treatment. Long-term follow-up data on treated patients remain limited. Case description: A 3.9-year-old neutered male cat presented with hyporexia and lethargy lasting for several days. An abdominal mass was palpated on clinical examination and confirmed to be a renal mass on a computed tomography angiography scan. Ultrasound-guided fine needle aspirates and cytological evaluation revealed a large-cell renal lymphoma. A modified Cyclophosphamide, vincristine and prednisolone protocol with vinblastine addition (Cyclophosphamide, vincristine, prednisolone and vinblastine protocol) was started, resulting in a complete response. Twenty weeks after chemotherapy initiation, the cat developed anorexia, fever, and pleural effusion. FIP was diagnosed based on a positive reverse transcription polymerase chain reaction test from the pleural effusion. Subcutaneous administration of GS441524 led to FIP resolution. Conclusion: The cat did not receive any further treatment for both FIP and lymphoma. Still alive and well, almost 5 years after the diagnosis. To the author’s knowledge, this is the first case of renal lymphoma in a cat that subsequently developed FIP. Further studies on the potential correlation between lymphoma, chemotherapy, and FIP are needed. Keywords: CHOP protocol, FIP, Remdesivir, Renal lymphoma. IntroductionThe pathogenesis of feline infectious peritonitis (FIP) is not yet entirely understood, but the virulent biotype Feline Infectious Peritonitis Virus (FIPV) is caused by a mutation in the feline enteric coronavirus (FCoV) with changes in cell tropism from enterocyte to monocyte (Müller et al., 2023; Tasker et al., 2023). Immunohistochemistry is considered the gold standard for diagnosis (Felten and Hartmann, 2019); however, a combination of clinical signs and effusion analysis, if present, including cytology, biochemistry, and FCoV RNA polymerase chain reaction testing, would strongly support the diagnosis of FIP (Thayer et al., 2022; Tasker et al., 2023). Immune-mediated vasculitis, typical of the wet form, abdominal effusion, lymphadenopathy, and subcapsular kidney abnormalities effusion have been commonly reported in infected cats (Lewis and O’Brien, 2010; Müller et al., 2023). Chest involvement with pathological lesions is less common, including pleuritis, vasculitis, perivascular parenchymal injury, and pneumonia, which can occur, leading to pleural effusion and dyspnea without the typical systemic signs of FIP (Slaviero et al., 2024). Abdominal effusions, lymphadenopathy, and pyogranulomatous abdominal lesions, the eyes, and the central nervous system are some common findings in the wet and dry (Kipar et al., 2005; Tasker, 2018, 2023). Renal abnormalities are common in younger cats, including bilateral or unilateral renomegaly and cortical hyperechogenicity, medullary rim sign, pyelectasia, and corticomedullary differentiation loss (Kipar et al., 2005; Tasker, 2018; Gülersoy et al., 2023; Müller et al., 2023). Poor prognosis and high fatality rate historically associated with FIP have improved considerably in recent years due to the novel use of nucleoside analogs, which have been successfully cleared and completed long-term resolution of clinical signs (Dickinson et al., 2020; Coggins et al., 2023; Green et al., 2023). However, the long-term follow-up of these patients with this new drug remains limited. Lymphoma is a hematopoietic neoplasia that occurs due to malignant cell proliferation of lymphocytes (Ambrosini et al., 2021). It originates from DNA mutations during primary differentiation of secondary lymphoid tissues, with potential viral and environmental factors and possible genetic predisposition (Argyle and Pecceu, 2016). Lymphomas are mainly categorized according to their topography (Sato Y Fujino and Chino, 2014). There are alimentary, mediastinal, multicentric, and extranodal lymphomas (Sato Y Fujino and Chino, 2014), where extranodal lymphoma refers to a disease confined to locations other than the alimentary, mediastinal, or multicentric sites, including the nasal, central nervous system, cutaneous, Ocular, laryngeal, and renal lymphoma (Argule and Pecceu, 2016).Renal lymphoma can be primary or associated with gastrointestinal or multicentric involvement (Ambrosini et al., 2021). Clinical signs are widely variable and nonspecific because they are associated with the affected organ (Ambrosini et al., 2021). Cats diagnosed with renal lymphoma often exhibit signs of renal disease, such as polyuria, polydipsia, azotemia, and nonregenerative anemia (Moore, 2013 ). Ultrasound can be used for the diagnosis of renal lymphoma and is also used to guide fine needle aspiration. Cytology is highly sensitive and specific for diagnosis (Moore et al., 2013). Neoplastic lymphocytes are large with relatively abundant cytoplasm and large nuclei that contain pale chromatin and one or more prominent nucleoli (Twomey and Alleman, 2005). Chemotherapy is the treatment of choice for large, high-grade lymphoma (Moore, 2013). The prognosis for large-cell/high-grade lymphoma mainly depends on the response to chemotherapy treatment and anatomical form. Nasal and mediastinal lymphoma carry the best prognosis, most likely due to the high response rate (Pui Yung Anna et al., 2025). Long-term follow-up of more than 2–3 years for patients treated for renal lymphoma is only rarely reported (Williams et al., 2021). In this report, we present a case of FIP infection following chemotherapy in a young cat treated for renal lymphoma. The cat achieved a complete response to both FIP treatment and lymphoma, and he is currently free of both diseases 5 years after diagnosis. Case detailsA neutered 3.9-year-old domestic short-haired male cat was referred for further investigation of a large mass in the cranial abdomen. At presentation, a 2-week history of hyporexia and lethargy was reported. On physical examination, besides a huge palpable mass in the mid-cranial abdomen, occupying two-thirds of the abdomen, no other abnormalities were observed. Hematology (Table 1), full biochemistry profile (Table 2), and urine protein to creatinine ratio (UPC) (Table 3) showed no significant abnormalities. Immunodeficiency virus and feline leukemia virus were both negative on the SNAP test (4Dx Plus; IDEXX Laboratories, Westbrook, ME). CT angiography of the thorax and abdomen revealed left renomegaly measuring approximately 80 mm in width and 65 mm in height and 90 mm in length with a markedly deformed structure, no corticomedullary definition, and a large lobulated expansile appearance (Fig. 1). The mass effect led to severe lateral intestine displacement into the right abdomen and craniolateral displacement of the spleen, which curled in the left cranial abdomen. The right kidney was mildly enlarged, approximately 50 mm in length, with several expansile, rounded lesions within the cortex and medulla. The rest of the CT examination was unremarkable. Ultrasound-guided fine needle angiography aspirates of the mass arising from the left kidney revealed a cell population of large-cell lymphoma (Fig. 2). A Cyclophosphamide, vincristine, prednisolone and vinblastine (COVP) protocol was initiated. This comprises a modified COP protocol with vinblastine addition (Pui Yung Anna., 2025). Complete remission was achieved after the second week of the COVP protocol. No significant AEs attributed to chemotherapy were reported during the chemotherapy course.
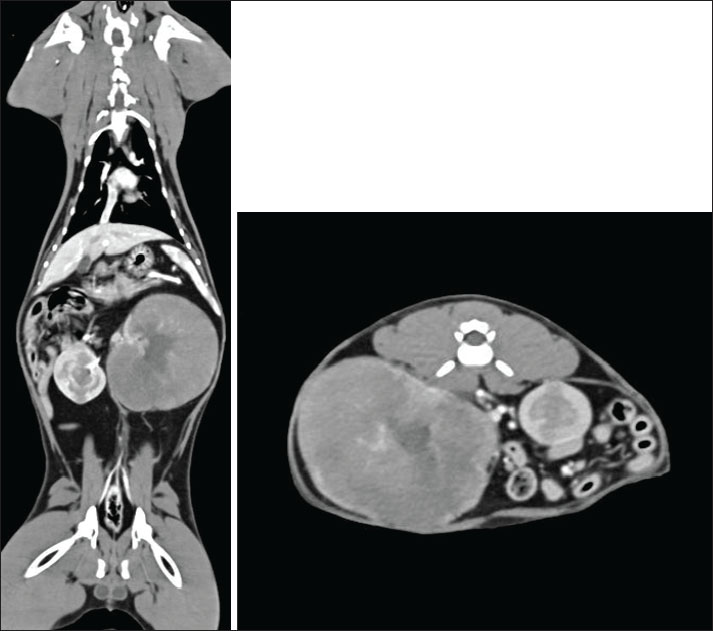
Fig. 1. Soft tissue window CT scan of the left coronal and right axis. The left kidney is markedly enlarged and measures approximately 80 mm in width, 65 mm in height, and 90 mm in length. It has a markedly deformed structure with no cortical medullary definition and a large lobulated expansile appearance. The cortex and medullary show reduced contrast enhancement, whereas the irregular peripheral rim of the tissue shows increased contrast enhancement. It retains a central area resembling a renal pelvis, where the left renal artery and vein are seen to enter; however, no accumulation of contrast medium is seen. The left kidney has a mass effect with severe lateral displacement of the intestines into the right abdomen. It also causes craniolateral displacement of the spleen, which is curled in the left cranial abdomen. The right kidney is mildly enlarged, measuring approximately 5 cm in length. It has several expansile rounded lesions within the cortex and medulla that mildly deform the external contour of the kidney. They are similar to attenuation to the medulla precontrast (approximately 40 HU) but show markedly reduced contrast enhancement.
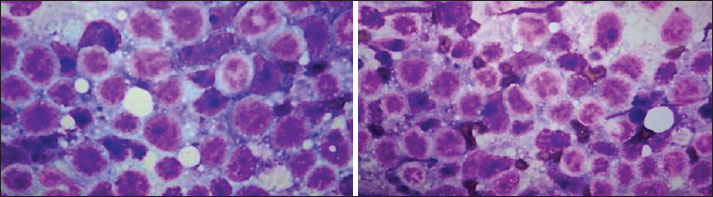
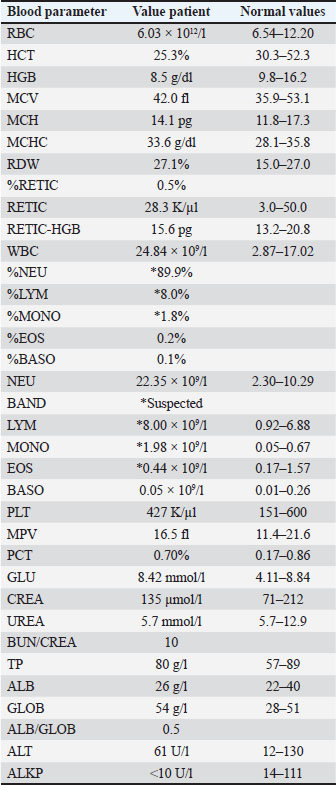
Fig. 2. 100x magnification of the fine-needle aspirate originating from the left kidney mass. The sample is highly cellular and contains large lymphocytes,2-3 times the size of a neutrophil. The lymphoid population is dominated by large lymphocytes with scant dark blue cytoplasm. A few punctate clear cytoplasmic vacuoles were Present in some cells. The nuclei are round and have fine reticular chromatin and one or more Prominent nucleoli. No normal renal tissue is seen. No evidence of inflammation or infection. The finding of a predominance of larger lymphocytes in the tissue is consistent with the findings of Large cell lymphoma. Table 1. Hematology when the cat was diagnosed with renal lymphoma. Abnormalities found were neutrophilia and monocytosis.
Table 2. Biochemistry when the cat was diagnosed with renal lymphoma. The only abnormalities were an increased urea and decreased ALKP.
Table 3. Urine analysis including Urine Protein to Creatinine ratio when the cat was diagnosed with renal lymphoma. Only proteinuria was found.
Twenty weeks after the start of chemotherapy, during the maintenance injection protocol of vincristine and cyclophosphamide, anorexia and lethargy were reported by the patient. Examination revealed moderate dyspnea with paradoxical breathing and a fever of 40°C. Bloodwork revealed mild nonregenerative anemia 25.3% (30.3–52.3), neutrophilia 22.35 × 109/l (2.30–10.29), and mild hyperglobulinemia 54 g/l (28–51) (Table 4). Table 4. Bloodwork from cats when FIP was suspected. Nonregenerative anemia, neutrophilia with band neutrophils and hyperglobulinemia .
Thoracic radiographs revealed a large amount of pleural effusion. Therapeutic thoracocentesis was performed, and the fluid was submitted for evaluation. Fluid analysis showed an exudate with a high protein count, and a proteinaceous background with neutrophilic and macrophage inflammation; negative aerobic culture for bacteria (Fig. 3). A PCR test for feline coronavirus was performed on the fluid, and a positive result was obtained (Table 5). Table 5. Examination results from the fluid collected from the thorax. The fluid is an exudate, and the PCR test result for FIP is positive, which diagnoses the cat.
Echocardiography and abdominal ultrasonography performed at this point revealed no signs of cardiac arrest. Cardiomyopathy that could have led to CHF or other abnormalities suggestive of lymphoma recurrence. A combination of blood tests, fluid analysis, and exclusion of other abnormalities using ultrasound confirmed the suspicion of FIP. Dexamethasone administered intravenously at 0.2 mg/kg q 24 hours was started, while the FeCoV PCR test result was pending. After a short initial clinical improvement, pleural effusion reoccurred 2 days later, and therapeutic thoracocentesis was repeated. Following the confirmation of FIP by PCR, daily subcutaneous (SQ) injections of GS441524 were administered at a dose of 10 mg/kg for 12 weeks. Mild discomfort and irritation during SQ injection were the only side effects reported by the owner. Dexamethasone treatment was stopped once FIP was confirmed, and nucleoside analogs were started. Marked clinical improvement was noticed after GS441524 was started with no clinical or abnormalities detected at 1–3–6–12 months rechecks. At the time this manuscript was written almost 5 years after the initial diagnosis of large-cell renal lymphoma, the cat is doing well without any clinical signs of lymphoma or recurrence of FIP. Ethical approvalThis study involved the use of client-owned animals only, following the internationally recognized high standards of individual clinical veterinary patient care. Ethical approval from a committee was not required. DiscussionLymphoma is the most common cancer affecting domestic cats, with an average age of 11 years (Pedersen et al., 2019). Pediatric or juvenile lymphoma is rare in cats; however, it has been reported (Williams et al., 2021; Rogato and Tanis, 2023). Recently, young cats with lymphoma tended to have mediastinal or disseminated forms, with only 15% renal origin (Rogato and Tanis, 2023). The incidence of renal lymphoma was 3.6% of all lymphoma cases in one study, where patients as young as 1-year-old were also reported to be affected (Williams et al., 2021). Although age difference is not assessed, it is always a prognostic factor; cats with renal lymphoma treated with chemotherapy have a median survival of 203 days, with 3 of 27 cats surviving longer than 2 years (Williams et al., 2021). This means that some cats could be cured of renal lymphoma. Unfortunately, these long-surviving cats were not reported, making it impossible to assess the relationship between young age and survival. Nevertheless, in one large study using CHOP-based chemotherapy, the anatomical form of lymphoma, approximately 30% of cats that achieved a complete response reported to live for 2 or more years; however, renal lymphoma tended to have the shortest survival compared with other forms (Collette et al., 2016). The cat reported here is still alive, 4 years after renal lymphoma diagnosis without a reported relapse. As said by Williams et al. (2021) and Collette et al. (2016), a cure is a possibility in cases that achieve a long-term, durable complete response. However, retrospective case reports have a limitation, and there is a need for larger cohort studies. Although multiagent chemotherapy is the therapy of choice for renal lymphoma, comparing the most commonly used chemotherapy protocols, such as COP (vincristine, cyclophosphamide, and prednisolone) and CHOP (vincristine, cyclophosphamide, and doxorubicin), is rare. Nonetheless, it appears that cats that achieve complete response tend to live for a long time, independent of the protocol used (Bernardo Marques, F. Ponce and Beaudu-Lange, 2024). In this case, a modified COP protocol with vinblastine addition (COVP protocol) was used. This is routinely used by the clinic of the lead author (Pui Yung Anna et al., 2025). Feline infectious peritonitis is an immune-mediated disease secondary to systemic infection of macrophages by a mutant feline coronavirus strain (Thayer et al., 2022). Before the use of nucleoside analogs such as remdesivir (GS5734) or GS441524, a median survival of 9 days was expected in cats with FIP (Ritz et al., 2007). Treatment with GS441524 proved to be effective against FIP in both the short and long term, with no confirmed relapses during a 12-month follow-up period and a 96% survival rate at 6 months in cats that survived beyond the first 48 hours (Coggins et al., 2023; Zwicklbauer et al., 2023). In this report, SQ GS441524 was used. Although very effective, daily SQ injections of nucleoside analogs are associated with pain, vocalization, and injection site reactions, making their administration challenging at times, as reported in our case (Green et al., 2023; Taylor et al., 2023; Negash et al., 2024). SQ GS441524 administration in the present study case was chosen due to the lack of availability of both oral form and information at the time of the efficacy of oral GS441524. Oral administration is now widely available and is the treatment of choice used by the authors, as shown by long-term follow-up of cat remissions (Krentz et al., 2021; Zwicklbauer et al., 2023). Stressful events during a young age, including surgical procedures such as spaying and neutering, have been associated with FIP development (Pedersen, 2009). Stress suppresses the immune system and increases the risk of FIP via a higher rate of viral replication mutation (Riemer et al., 2016). Long-term chemotherapy can contribute to immune defense, but further research is needed to confirm its correlation and predisposition for FIP. Following recovery from immunosuppression, immune reconstitution might have contributed to the following: the development of FIP. This is known to occur because of a change in the T helper responses to inflammation after discontinuation of immunosuppressive drugs (Sueki et al., 2019). Positive FCoV result from ascites fluid has been anecdotally reported in a young cat following chemotherapy for jejunal lymphoblastic lymphosarcoma (Borku et al., 2017). The specificity of the RT RNA PCR test on effusion is 95.8%, and the sensitivity is 68.6% (Felten et al., 2017). Hematological abnormalities are very common in cats with FIP. The following may be seen but may not always be present: nonregenerative anemia, microcytosis, lymphopenia, neutrophilia, thrombocytopenia, hyperproteinemia, hyperglobulinemia, hypoalbuminemia, hyperbilirubinemia, azotemia, and increased liver enzyme activity [Felten and Hartmann (2019)]. The albumin-to-globulin ratio can be used to predict FIP diagnosis. Several cutoffs have been made to rule in (<0.4) or rule out (>0.6–0.8) FIP (Felten and Hartmann, 2019). The development of a debilitating condition, such as renal lymphoma and immunosuppression caused by chemotherapeutic agents, might have played a role in the FIP development in this case. To the authors’ knowledge, this is the first report documenting the development of FIP following the successful treatment of renal lymphoma. Regardless of the reason for the two potentially fatal pathologies, the current case report highlights the need for effective therapeutic options to be available and long-term remissions, or even a potential cure, are possible if appropriate therapies and care. AcknowledgmentsNone. FundingThe authors have not declared a specific grant from any funding source for this research from public, commercial, or not-for-profit agencies. Authors’ contributionsI.B and A.G produced the original draft of the manuscript. A.A., A.G., and I.B. revised the manuscript. All authors have read and approved the final version of the manuscript. Conflict of interestThe author declares that there is no conflict of interest. Data availabilityAll data supporting this study’s findings are available within the manuscript. ReferencesAmbrosini, F., Garcia, M., Silva, V.G.E., Rossa, A.P., Alves, T.L., Sousa, R.S.D., Albernaz, V.G.P., Sousa, M.G. and Stedile, S.T.D.O. 2021. Primary renal lymphoma in two cats: case report. Arch. Vet. Sci. 26, 107–116. Argyle, D.J. and Pecceu, E. 2016. Canine and feline lymphoma: challenges and opportunities for treatment creating a paradigm shift. Vet. Comp. Oncol. 14, 1–7. Bernardo Marques, G., Ponce, F., Beaudu-Lange, C., Beguin, J., Boissy, L., Chavalle, T., Denoeux, P., Diop, N., Ferro, L., Floch, F., Lajoinie, M., Sayag, D., Barrett, L. and Chamel, G 2024. High-grade Feline and Large granular lymphocyte alimentary lymphomas treated with COP- or CHOP-based treatment chemotherapy: a multi-center retrospective study of 57 patients. Vet. Comp. Oncol. 22, 186–197. Borku, M.K., Kara, E. and Kurtdede, A. 2017. Application of chemotherapy on a cat with lymphoblastic lymphosarcoma. Turkish J. Vet. Res. 1, 29–33. Coggins, S.J., Norris, J.M., Malik, R. Govendir, M., Hall, E., Kimble, B.F. and Thompson, M. 2023. Outcomes of treatment of feline cats infectious peritonitis using parenterally administered remdesivir, with or without transition to orally administered GS-441524. J. Vet. Intern. Med. 37, 1772–1783. Collette, S.A., Allstadt, S.D., Chon, E.M. Vernau, W., Smith,A.N., Garrett, L.D., Choy, K., Rebhun, R.B., Rodriguez Jr, C.O. and Skorupski, K.A. 2016. Treatment of intermediate- to high-grade lymphoma with a modified University of Wisconsin-Madison protocol: 119 Cases (2004-2012). Vet. Comp. Oncol. 14(Suppl 1), 136–146. Dickinson, P.J., Bannasch, M., Thomasy, S.M., Murthy, V.D., Vernau, K.M., Liepnieks, M., Montgomery, E., Knickelbein, K.E., Murphy, B. and Pedersen, N.C. 2020. Antiviral treatment using the adenosine nucleoside analogue GS-441524 in cats with clinically diagnosed neurological feline infectious peritonitis. J. Vet. Intern. Med. 34, 1587–1593. Felten, S. and Hartmann, K. 2019. Diagnosis of feline infectious peritonitis: a review of the literature current literature. Viruses 11, 1068. Felten, S., Leutenegger, C.M., Balzer, H.J., Pantchev, N., Matiasek, K., Wess, G. and Egberink H Hartmann. 2019. Sensitivity and specificity of real-time reverse transcriptase the polymerase chain reaction (PCR) for detecting feline coronavirus mutations in effusion and serum/plasma of cats to diagnose feline infectious peritonitis. BMC Vet. Res. 2(2), 219–222. Green, J., Syme, H. and South, T. 2023. Thirty-two cats with effusive or non-effusive feline infectious peritonitis treated with remdesivir and GS-441524. J. Vet. Intern. Med. 37, 1784–1793. Gülersoy, E., Balıkçı, C., Kısmet, E. Gunal, I., Sahan, A., Guclu, M.A. and Ok, M 2023. Renal ultrasonography findings in cats with Feline Infectious Peritonitis. Van Vet. J. 34, 63–69. Kipar, A., May, H., Menger, S., Weber, M., Leukert, W. and Reinacher, M. 2005. Morphologic features and the development of granulomatous vasculitis in infectious peritonitis in felines. Vet. Pathol. 42, 321–330. Krentz, D., Zenger, K., Alberer, M., Felten, S., Bergmann, M., Dorsch, R., Matiasek, K., Kolberg, L., Hofmann-Lehmann, R., Meli, M.L., Spiri, A.M., Horak, J., Weber, S., Holicki, C.M., Groschup, M.H., Zablotski, Y., Lescrinier, E., Koletzko, B., Von Both, U. and Hartmann, K. 2021. Curing Cats with Feline Infectious Peritonitis with an Oral Multi-Component Drug Containing GS-441524. Viruses 13, 2228. Lee, P.Y.A., Horta, R., Sze, C.N. and Giuliano, A. 2025. Use of cyclophosphamide, vincristine, prednisolone and vinblastine for the treatment of large cell lymphoma in cats. J. Vet. Intern. Med. 39(2), e700661-9 Lewis, K.M. and O’Brien, R.T. 2010. Abdominal ultrasonographic findings associated with feline infectious peritonitis: a retrospective review of 16 patients. J. Am. Anim. Hosp. Assoc. 46, 152–160. Moore, A. 2013. Extranodal lymphoma in the cat prognostic factors and treatment options. J. Feline Med. Surg. 15, 379 -390. Müller, T.R., Penninck, D.G., Webster, C.R. and et al. 2023. Abdominal ultrasonographic findings in cats with feline infectious peritonitis: an update. J. Feline Med. Surg. 25, 1098612X. Negash, R., Li, E., Jacque, N., Novicoff, W. and Evans, J.M.S 2024. Owner’s experience and veterinary involvement with unlicensed GS-441524 treatment of feline infectious peritonitis: a prospective cohort study. Front. Vet. Sci. 11, 1377207. Pedersen, N.C. 2009. Feline infectious peritonitis virus infection: 1963–2008. J. Feline Med. Surg. 11, 225–258. Pedersen, N.C., Perron, M., Bannasch, M., Montgomery, E., Murakami, E., Liepnieks, M. and Liu, H. 2019. Efficacy and safety of nucleosides analog GS-441524 for the treatment of naturally occurring feline infectious peritonitis. J. Feline Med. Surg. 21, 271–281. Riemer, F., Kuehner, K.A., Ritz, S. Sauter-Louis, C. and Hartmann, K. 2016. Clinical and laboratory characteristics of cats with feline infectious peritonitis: a retrospective study of 231 confirmed cases (2000-2010). J. Feline Med. Surg. 18, 348–356. Ritz, S., Egberink, H. and Hartmann, K. 2007. Effect of feline IFN-omega on survival time and quality of life of cats with infectious peritonitis. J. Vet. Intern. Med. 21, 1193–1197. Rogato, F., Tanis, J.B., Pons Gil, B., Pittaway, C., Johnston, C.A. and Guillen, A. 2023. Clinical characterization and long-term survival of pediatric and juvenile lymphoma in cats: 33 cases (2008-2022). J. Small Anim. Pract. 64, 788–796. Sato, Y., Fujino., Chino, J., Takahashi, M., Fukushima, K., Goto-Koshino, Y., Uchida, K., Ohno, K. and Tsujimoto, H. 2014. Prognostic anatomical and morphological analyses classification of feline lymphoma. J. Vet. Med. Sci. 76, 807–811. Slaviero, M., Cony, F.G., Da Silva, R.C., De Lorenzo, C., A de Almeida, B., Bertolini, M., Driemeier, D., Pavarini, S.P. and Sonne, L. 2024. Pathological findings and feline pattern analysis infectious peritonitis in the cat respiratory tract. J. Comp. Pathol. 210, 15–24. Sueki, H., Mizukawa, Y. and Aoyama, Y. 2018. Immune reconstitution in inflammatory syndrome in non-HIV immunosuppressed patients. J. Dermatol. 45(1), 3–9. Tasker, S. 2018. Diagnosis of feline infectious peritonitis: update on evidence supporting available tests. J. Feline Med. Surg. 20, 228 -243. Tasker, S., Addie, D.D., Egberink, H. Hofmann-Lehmann, R., Hosie, M.J., Truyen, U., Belak, S., Boucraut-Baralon, C., Frymus, T., Lloret, A., Marsilio, F., Pennisi, M.G., Thiry, E., Mostl, K. and Hartmann, K. 2023. Feline infectious peritonitis: european advisory board on cat disease guidelines. Viruses 15, 1847. Taylor, S.S., Coggins, S., Barker, E.N.Gunn-Moore, D., Jeevaratnam, K., Norris, J.M., Hughes, D., Stacey, E., MacFarlance, L., O’Brien, C., Korman, R., McLauchlan, G., Torres, X.S., Taylor, A., Bongers, J., Castro, L.E., Foreman, M., McMurrough, J., Thomas, B., Royaux, E., Saiz, I.C., Bertoldi, G., Harlos, C., Work, M., Prior, C., Sorrell, S., Malik, R. and Tasker, S. 2023. Retrospective study and outcome of 307 cats with a sex with feline infectious peritonitis treated with legally sourced veterinary preparations of remdesivir and GS-441524 (2020–2022). J. Feline Med. Surg. 25, 1098612X. Thayer, V., Gogolski, S., Felten, S. Hartmann, K., Kennedy, M. and Olah, G.A. 2022. AAFP/EveryCat feline infectious peritonitis diagnostic guidelines. J. Feline Med. Surg. 24, 905–933. Twomey, L.N. and Alleman, A.R. 2005. Cytodiagnosis of feline lymphoma. Compendium 27, 17–31. Williams, A.G., Hohenhaus, A.E. and Lamb, K.E. 2021. Incidence and treatment of feline renal lymphoma: 27 cases. J. Feline Med. Surg. 23, 936–944. Zwicklbauer, K., Krentz, D., Bergmann, M. et al. 2023. Long-term follow-up of cats in complete remission after treatment of feline infectious peritonitis with oral GS- 441524. J. Am. Coll. Cardiol. 2, 41524. | ||
| How to Cite this Article |
| Pubmed Style Beekhuis I, Serrano G, Almendros A, Giuliano A. Successful treatment and long-term follow-up of a young cat with feline infectious peritonitis and renal lymphoma. Open Vet. J.. 2025; 15(10): 5383-5390. doi:10.5455/OVJ.2025.v15.i10.56 Web Style Beekhuis I, Serrano G, Almendros A, Giuliano A. Successful treatment and long-term follow-up of a young cat with feline infectious peritonitis and renal lymphoma. https://www.openveterinaryjournal.com/?mno=239304 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.56 AMA (American Medical Association) Style Beekhuis I, Serrano G, Almendros A, Giuliano A. Successful treatment and long-term follow-up of a young cat with feline infectious peritonitis and renal lymphoma. Open Vet. J.. 2025; 15(10): 5383-5390. doi:10.5455/OVJ.2025.v15.i10.56 Vancouver/ICMJE Style Beekhuis I, Serrano G, Almendros A, Giuliano A. Successful treatment and long-term follow-up of a young cat with feline infectious peritonitis and renal lymphoma. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5383-5390. doi:10.5455/OVJ.2025.v15.i10.56 Harvard Style Beekhuis, I., Serrano, . G., Almendros, . A. & Giuliano, . A. (2025) Successful treatment and long-term follow-up of a young cat with feline infectious peritonitis and renal lymphoma. Open Vet. J., 15 (10), 5383-5390. doi:10.5455/OVJ.2025.v15.i10.56 Turabian Style Beekhuis, Ilse, Goncalos Serrano, Angel Almendros, and Antonio Giuliano. 2025. Successful treatment and long-term follow-up of a young cat with feline infectious peritonitis and renal lymphoma. Open Veterinary Journal, 15 (10), 5383-5390. doi:10.5455/OVJ.2025.v15.i10.56 Chicago Style Beekhuis, Ilse, Goncalos Serrano, Angel Almendros, and Antonio Giuliano. "Successful treatment and long-term follow-up of a young cat with feline infectious peritonitis and renal lymphoma." Open Veterinary Journal 15 (2025), 5383-5390. doi:10.5455/OVJ.2025.v15.i10.56 MLA (The Modern Language Association) Style Beekhuis, Ilse, Goncalos Serrano, Angel Almendros, and Antonio Giuliano. "Successful treatment and long-term follow-up of a young cat with feline infectious peritonitis and renal lymphoma." Open Veterinary Journal 15.10 (2025), 5383-5390. Print. doi:10.5455/OVJ.2025.v15.i10.56 APA (American Psychological Association) Style Beekhuis, I., Serrano, . G., Almendros, . A. & Giuliano, . A. (2025) Successful treatment and long-term follow-up of a young cat with feline infectious peritonitis and renal lymphoma. Open Veterinary Journal, 15 (10), 5383-5390. doi:10.5455/OVJ.2025.v15.i10.56 |