| Research Article | ||
Open Vet. J.. 2025; 15(2): 954-964 Open Veterinary Journal, (2025), Vol. 15(2): 954-964 Research Article Role of lactoferrin in the treatment of E. coli-induced bovine mastitisMohamed Ibrahim Mostafa1, Saber Ali Saad2, Asmaa Elsayed Mohammed3*, Elsaid Mohamed Mohamed Saafan4 and Amal Mohamed El-Sayed51Department of Bacteriology, Animal Health Research Institute, Agriculture Research Center (ARC), Damanhur branch, Egypt 2Department of Food Hygiene, Animal Health Research Institute, Agriculture Research Center (ARC), Damanhur branch, Egypt 3Department of Bacteriology, Animal Health Research Institute, Agriculture Research Center (ARC), Sohag, Egypt 4Department of Food Hygiene, Animal Health Research Institute, Agriculture Research Center (ARC), Mansoura branch, Egypt 5Department of Food Hygiene and Control, Faculty of Veterinary Medicine, Aswan University, Egypt *Corresponding Author: Asmaa Elsayed Mohammed. Department of Bacteriology, Animal Health Research Institute, Agriculture Research Center (ARC), Sohag, Egypt. Email: dr_asmaa_lab [at] yahoo.com Submitted: 30/12/2024 Accepted: 07/02/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
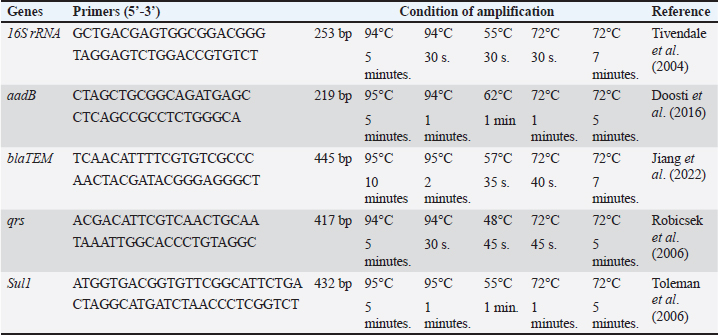
AbstractBackground: Bovine mastitis (BM) is a costly disease in the dairy industry. It is associated with reduced milk production and changes in milk composition and quality. Escherichia coli is the most important pathogen causing BM. Aim: This study aimed to assess the efficacy of lactoferrin (LF) as an enhanced agent when combined with marbofloxacin for the treatment of E. coli BM. Methods: Eighty lactating cattle and buffaloes, 40 each, aged 4–6 years suffering from BM underwent clinical examination. Milk samples were collected for bacteriological examination. The recovered E. coli isolates were subjected to serological, and polymerase chain reaction (PCR) analysis. BM cases were allocated into two groups: group (A) was treated with marbofloxacin combined with LF, and group (B) was treated with marbofloxacin alone. Results: Fifty E. coli isolates were recovered. Six serogroup were identified: O55, O103, O26, O11, O44, and O124. All isolates were sensitive to marbofloxacin, while there was resistance to gentamycin and ampicillin (26% each), cefquinome (14%), cefixime (10%), and SuLFamethoxazole and trimethoprim (4%). The molecular identification of E. coli strains revealed that the 16S rRNA gene was present in all identified E. coli strains (100%). All isolates resistant to antibiotics encoded BlaTEM, aadB, and Sul1, whereas the qnrS gene was not detected in any E. coli isolate. The broth dilution method did not detect growth when the marbofloxacin (6 µg/ml) was combined with LF (2 mg/ml). Group (A) showed significant improvements compared with group (B), with no recurrence rate. Conclusion: LF can serve as a significant cotreatment agent for treating BM. As antibiotic resistance increases, LF-based drugs could play an important role in providing sustainable, effective alternatives for dairy animals. Keywords: Bovine mastitis, Lactoferrin, E. coli, Marbofloxacin. IntroductionBovine mastitis (BM) is a mammary gland inflammation caused by physical trauma or bacterial invasion. It is the most prevalent and economically significant infectious disease in dairy farms worldwide (Sharun et al., 2021). Escherichia coli is the most prevalent Gram-negative pathogen causing clinical mastitis in dairy animals (Gao et al., 2017). Coliform mastitis causes high morbidity, and only 30%–50% of animals can regain their full production (Kutila et al., 2004) despite supportive and antibacterial therapy (Singh et al., 2024). Management of BM is challenging because it is polygenic. Antibiotics are considered the cornerstone of treatment for BM. The efficiency of treating animals with clinical mastitis depends on the use of appropriate antimicrobials that eradicate pathogenic bacteria from udder tissue. Broad-spectrum antimicrobial drugs, namely, 3rd generation cephalosporins and fluoroquinolones, are prescribed for treating coliform BM. These drugs can be administered via intramammary, intramuscular, or direct intravenous injections (Abd-ELFatah et al., 2024). Although antimicrobial therapy is associated with rapid bacterial elimination and a high rate of survival in animals, it can be a significant cause for spreading antibiotic-resistant bacteria to humans (Fatemi et al., 2024). The search for noninvasive therapeutic options that do not produce resistance is mandatory (Marques-Bastos et al., 2023). Some alternatives, such as peptides, glycoproteins, and nanoparticles, have been used and proven effective (Saeed et al., 2023). Lactoferrin (LF) is a glycoprotein naturally produced by polymorphonuclear neutrophils and epithelial cells. The protein is secreted by these cells at high levels in response to the inflammatory process. It has both bacteriostatic and bactericidal effects against a wide range of pathogens (Le et al., 2024). The function of LF in mammary gland immunity is to modify and regulate macrophage, lymphocyte, and neutrophil function in response to nonspecific resistance to infection. LF increases the permeability of bacterial cell membranes and destroys the outer membrane of Gram-negative pathogens by binding to its lipopolysaccharide (LPS) and inhibiting its damaging effects. Low LF levels in clinical BM may be inadequate for preventing bacterial proliferation in the udder tissue (Yassin and Abd Elhady, 2023). LF combined with antibiotics has a synergistic effect against BM. With an optimum dosage and duration of therapy, LF can help bacterial clearance from the animal mammary gland while simultaneously inhibiting the toxic properties of LPS (Diarra et al., 2002c). The objective of the current study was to assess the efficacy of LF as an enhanced agent when combined with marbofloxacin for the reduction of bacterial resistance to this antibiotic in E. coli BM. Material and MethodsStudy sampleThis study included 80 lactating cattle and buffaloes (40 each) from dairy farms aged 4–6 years. All animals had clinical mastitis. The study was conducted from January 2022 to October 2024 at the El-Beheira Governorate, Egypt. Clinical examination of animalsClinical examination of animals was conducted following Constable et al. (2016) to identify the clinical features of mastitis. California mastitis test (CMT)Equal volumes of CMT reagent and milk (2 ml from each quarter) were mixed thoroughly in a cup and then gently rotated for 10 seconds. The results were recorded and categorized into score 0 (negative), score 1 (trace), 2 (weakly positive), 3 (positive), and 4 (strongly positive) (Moroni et al., 2006). Collection of milk samplesMilk samples were collected for bacteriological examination after cleaning and disinfecting the animal teats with soap, water, and alcohol (70%) (Kerro Dego and Tareke, 2003). The first jet of milk from each quarter of the udder was discarded. Then, 5–10 ml was taken in a sterile vial. The samples were labeled with the identification number of the affected animal and the date of collection. The samples were then brought to the Animal Health Research Institute Laboratory, Damanhur Laboratory, Egypt, for bacteriological examination. Isolation, enumeration, and identification of E. coliMilk samples from Clinical BM cases underwent centrifugation at 3,000 rpm for 20 minutes using a laboratory centrifuge (Sigma 1-7). After discarding the cream and supernatant, the resulting sediment was streaked onto MacConkey agar (Oxoid, UK) and EMB agar (Oxoid, UK), which were incubated at 37°C for 18–24 hours and subsequently examined for bacterial growth. Escherichia coli colonies on MacConkey’s agar appeared as pink or red colonies lactose fermenter, while on EMB they appeared as bluish black colonies with a green metallic sheen. (Zinnah et al., 2007). E.coli enumeration10-fold serial dilutions of milk samples were prepared using sterile peptone water. From the prepared dilutions 100 µl was plated on EMB agar. The inoculated plates were incubated at 37°C for 18–24 hours. The bacterial count was recorded from triple readings (Malt, 2013). Microscopic examination The direct film was prepared from pure culture and stained with gram stain to observe the morphological characteristics of E. coli, which is gram-negative short rod bacilli, that occur singly or in pairs (Cheesbrough, 2006). Biochemical confirmation of E. coli Pure cultures of a single colony from EMB agar were streaked onto nutrient agar (Oxoid, UK) for purification and then incubated at 37°C for 24 hours. Oxidase (Merk, Germany), triplicate sugar iron (Oxoid, UK), Indole, Methyl red (Oxoid, UK), Voges-Proskauer (Oxoid, UK), and Citrate tests (Oxoid, UK) were performed (De Boer and Heuvelink, 2000). Serotyping of E. coli: A slide agglutination test was performed to identify E. coli using polyvalent and monovalent antisera was done (da Silva et al., 2001). Antibiotic sensitivity testing of E. coli:The 50 isolates of E. coli were examined for antibiograms using the disc diffusion method against six antibiotics (Oxoid). The antibiotic discs used were marbofloxacin (5 μg), cefixime (5 μg), suLFamethoxazole and trimethoprim (25 μg), gentamycin (10 μg), ampicillin (10 μg), and cefquinome (30 μg). The results were interpreted according to CLSI (He et al., 2024). Molecular detection of antibiotic-resistance genesDNA extraction and amplification DNA extraction was performed using the QIAamp DNA Mini kit (Qiagen, Germany, GmbH) with modifications based on the manufacturer’s recommendations. Briefly, 200 µl of the sample suspension was incubated with 10 µl of proteinase K and 200 µl of lysis buffer at 56°C for 10 minutes. After incubation, 200 µl of 100% ethanol was added to the lysate. The samples were subsequently washed and centrifuged according to the manufacturer’s instructions. The nucleic acid was then eluted with 100 µl of elution buffer. The oligonucleotide primers used were supplied by Metabion (Germany) (Table 1). The 50 recovered E. coli isolates were confirmed by polymerase chain reaction (PCR) amplification of the 16S rRNA gene (Tivendale et al., 2004). Then, we screened for antibiotic resistance genes: BlaTEM gene for detection of β-lactam resistance (Jiang et al., 2022), Sul1 encoding sulfonamide resistance (Toleman et al., 2006), aadB mediating gentamycin resistance (aminoglycosides) (Doosti et al., 2016), and qnrs encoding fluoroquinolone resistance (Robicsek et al., 2006). PCR amplification was conducted in an applied biosystem 2,720 thermal cycler (Life Technologies, Germany). Table 1. Primers sequences, target genes, amplicon sizes, and cycling conditions.
Analysis of PCR products At room temperature, the products of PCR were separated by electrophoresis on 1.5% agarose gels (Applichem, Germany, GmbH) in 1× TBE buffer at 5 V/cm. For gel analysis, a gene ruler 100-bp ladder (Fermentas, Thermo, Germany), melilot 100-bp and 100-bp plus ladders (Qiagen, Gmbh, Germany), and Genedirex 50-bp DNA ladder RTU (Cat. No. DM012-R500) were used to determine the fragment sizes. The gel was photographed (Alpha Innotech, Biometra) and analyzed by computer software. Detection of minimum inhibitory concentration (MIC) of marbofloxacin and LF using the Broth dilution method:Preparation of bacterial inoculum Escherichia coli strain O55 was sub-cultured onto EMB agar plates and incubated at 37°C for 24 hours. 3–4 colonies were selected, inoculated in tryptic soy broth, and then incubated at 37°C for 2–6 hours. Suspension turbidity was balanced to coordinate with 0.5 McFarland standards and then diluted to obtain a concentration of 105 CFU/ml approximately (Owuama, 2017). LF preparation Pure LF (Sigma Chemical Co.) was purchased. The following concentrations of LF solution were prepared, (5, 4, 3, 2, and 1 mg/ ml) in distilled water, sterilized by a 0.45-mm filter, and freshly used (Al Habty and Ali, 2023). Preparation of marbofloxacin antibiotic Marbofloxacin was diluted to different concentrations (5, 6, 7, 7.5, 9, 9.5, 9.8, 10, and 15 μg/ml) in distilled water in test tubes. 100 μl of bacterial concentration 105 CFU/ml were added to the prepared LF and marbofloxacin concentrations. The inoculated tubes and the positive control tube (tubes containing broth only without any treatment) were incubated at 37°C for 24 hours. To determine the MIC, 100 μl of each clear tube was spread onto EMB agar and incubated at 37°C for 24 hours. The bacterial count was also recorded. The MIC is the lowest concentration of an antimicrobial agent that suppresses microbial growth (90%. The minimum bactericidal concentration (MBC) is the lowest antimicrobial concentration required to kill microorganisms. The mean MIC was calculated from triple readings for each test (Andrews, 2001). The MICs of both LF and marbofloxacin were added to 100 μl of bacterial concentration 105 CFU/ml in a sterile tube to assess the synergistic effect of marbofloxacin and LF. Treatment of infected patientsThe 50 BM cases were allocated randomly in two groups (A) and (B), each consisting of 25 cases. Group (A) received marbofloxacin (marbocyl ®) (1 cm/50 kg bw) for 3 days, and LF (2 gm intramammary every 12 hours) for 3 days. Group (B) received marbofloxacin (marbocyl ®) 1 cm / 50-kg bw) for 3 days. The two groups were injected with the nonsteroidall anti-inflammatory drug flunixin meglumine (finadyne®) at a dose of 1 cm/50 kg bw for three days. Follow-up of cases to evaluate treatment outcomeThe two groups were evaluated during the first week and two months after treatment to determine the efficacy of the treatment as follows: Clinical examination: systemic clinical signs were monitored (Heart rate, rectal temperature, rumen motility, appetite, and general attitude). The udder was palpated for soreness, swelling, and hardness. The milk was inspected for clotting, color changes, and sourness at each milking. CMT was performed. Bacteriological examination: E. coli enumeration and bacterial counts were recorded. Statistical analysisThe obtained data were statistically analyzed to calculate the mean, SD, and p values using the independent sample t test and SPSS program according to (Snedecor and Cochran, 1980). Ethical approvalThis study was approved by the Institutional Ethics Committee and was conducted in accordance with local laws and regulations. The study was approved by the Medical Research Ethics Committee, Faculty of Medicine, Sohag University (OHRP #: IRB00013006) and was performed according to the Interdisciplinary Principles and Guidelines for the Use of Animals in Research, Testing, and Education by the New York Academy of Sciences, Ad Hoc Animal Research Committee. Table 2. Serotypes of E. coli strains isolated from BM animals.
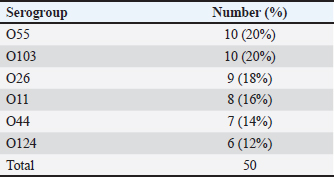
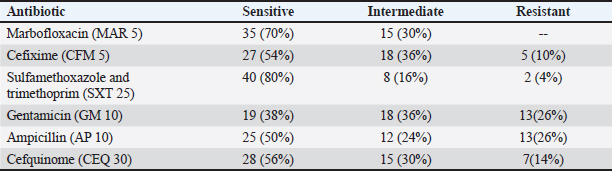
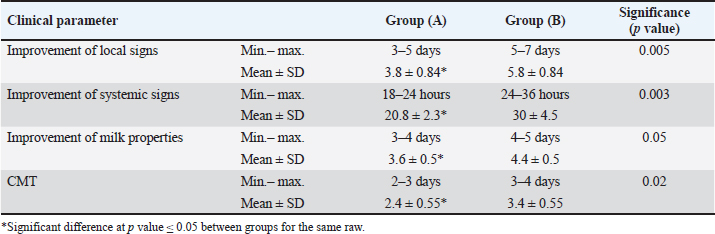
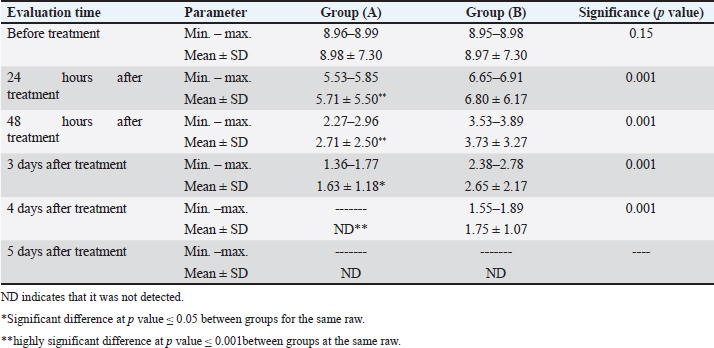
ResultsOf the 80 examined BM animals, 50 were positive for E. coli (62.5%). Thirty East coli strains were isolated from buffaloes and 20 E. coli strains were isolated from cattle. Six serogroups were identified: O55 and O103 (20% each), followed by O26 (18%) and O124 (12%) (Table 2). Antibiotic sensitivity testing of 50 E. coli isolates revealed that all isolates were sensitive to marbofloxacin. The isolates showed resistance to gentamicin and ampicillin (26% each), cefquinome (14%), cefixime (10%), and SuLFamethoxazole and trimethoprim (4%) (Table 3). Molecular identification by PCR confirmed that all 50 isolates of E. coli were positive for the 16S rRNA gene. All beta-lactam-resistant isolates carried the BlaTEM gene. All suLFonamide-resistant isolates encode Sul1. All aminoglycoside-resistant isolates carried the aadB gene. The qnrS gene was not detected in any E. coli isolates (Fig. 1). The broth dilution method revealed that the MIC of marbofloxacin was 6 µg/ml with a bacterial count of 4.16 log cfu/ml and MBC was 7 µg/ml, whereas the MIC of LF was 2 mg/ml with a bacterial count of 4.41 log cfu/ml and MBC of 3 mg/ml. No growth was detected when the marbofloxacin (6 µg/ml) was combined with LF (2 mg/ml) (Table 4). Group (A), which received LF intramammary infusion with marbofloxacin, showed significant improvement in BM compared with group (B), which received marbofloxacin alone. Local soreness and edema in the udder improved within 3–5 days in group (A) and 5–7 days in group (B). Systemic signs improved within 18 to 24 hours in group (A), and within 24 to 36 hours in group (B). Milk properties returned to normal levels within 3–4 days in group (A), and 4–5 days in group (B). CMT returned to the normal level after 2–3 days in group (A), and 3–4 days in group (B) (Table 5). There was a significant decrease in the E. coli count in group (A) compared with group (B). The E. coli count could not be detected after 4 days in group (A) and after 5 days in group (B) (Table 6). Re-evaluation of BM cases after 2 months revealed no recurrence in group (A), whereas recurrence occurred in three cases in group (B). Table 3. Antimicrobial resistance profiles of E. coli isolated from BM animals.
Fig. 1. Gel electrophoresis and PCR amplification for E. coli. Lane (L): DNA marker; lane (P): positive control; lane (N): negative control, (bp) base pair. (A): 1, 2, 3,4,5,6 lanes are positive for the 16S rRNA gene (253 bp). (B): 1, 2, 3,4,5,6 lanes are positive for the BlaTEM gene (445 bp). (C): 1, 2, 3,4,5,6 lanes are positive for aadB gene (219 bp). (D): 1, 2, 3,4,5,6 lanes are negative for qnrS gene (417bp). (E): 1 and 2 lanes are positive for Sul1 gene (432 bp). DiscussionBM is considered among the most common diseases in farm animals, resulting in a reduction in dairy milk consumption and high economic losses (Morales-Ubaldo et al., 2023). Escherichia coli is the major causative agent of clinical BM (Nery Garcia et al., 2024). In the environment, E. coli can invade the udders via the teat canal and cause intramammary infection (Burvenich et al., 2003). In the present study, out of the 80 examined BM animals, there were 50 positive samples for E. coli (62.5%) lower results were obtained by Hameed et al. (2008), Ali et al. (2011), Bhanot et al. (2012), Lamey et al. (2013), who observed E. coli at 15.38%, 16.18%, 16.3%, and 18.47%, respectively, in BM animals. The variations between the results of different reports may be due to the sample size, geographical distribution, management practice, and sanitary measures (Barua et al., 2014). Table 4. MIC of marbofloxacin and LF.
Table 5. Comparison of clinical outcomes between the treatment groups.
Table 6. Comparison of E. coli counts among the treatment groups.
Buffaloes possess certain traits that can increase the risk of mastitis. Their udder is more pendulous than that of cattle (Moroni et al., 2006). In this study, 30 E. coli strains were isolated from buffaloes and 20 E. coli strains were isolated from cattle. In the current study, six serogroups were identified: O55 and O103 (20% each), followed by O26 (18%), and the least was O124 (12%). These results are similar to those obtained by Mohamed et al. (2011) showed that the most prevalent E. coli serogroup from persistent BM cases were O55 (19.2%), O119 (12.3%), O114 (10.5%), O26 (7%), O111 (15.8%), O124 (12.3%), O157 (7%), and O44 (3.5%). Antibiotics are considered the first-choice treatment for BM (El-Sayed and Kamel, 2021). The antibiotic sensitivity test is widely used in clinical investigations to determine the most effective antimicrobial drug for therapy (Singh et al., 2018). The antibiotic sensitivity testing of 50 E. coli isolates revealed that all isolates were sensitive to marbofloxacin. The isolates showed resistance to gentamicin and ampicillin (26% each), cefquinome (14%), cefixime (10%), and sulfamethoxazole and trimethoprim (4%). This finding agrees with Rahimiyan et al. (2021) w, who recommended the use of marbofloxacin as an effective drug in the treatment of East coli bone marrow infections. Fairbrother et al. (2015) and Martínez-Vázquez et al. (2018) concluded that 20% of the isolates were resistant to more than two groups of antibiotics and 20%–33% of E. coli isolates from BM cases were resistant to at least one drug. Yu et al. (2020) reported that all the isolated E. coli strains expressed multi-resistance to antimicrobial agents. The abuse of antibiotics leads to antibiotic resistance in pathogenic strains. These resistances are coded by specific genes that may be located on the bacterial chromosome, plasmids, transposons, or gene cassettes that bind to integrons; thus, they are easily transmitted through isolates (Daka et al., 2012). In this study, the molecular identification of antibiotic-resistance genes of E. coli strains revealed that all beta-lactam-resistant isolates possessed the BlaTEM gene. All sulfonamide-resistant isolates encode Sul1. All aminoglycoside-resistant isolates carried the aadB gene. The qnrS gene was not detected in any E. coli isolates. Similarly, Pishtiwan and Khadija (2019) detected blaTEM in 81% of E. coli isolates. Arabi et al. (2015) also detected Sul1 in 81% of E. coli isolates. In contrast, Yu et al. (2008) detected the qnrS gene in 14 (6%) E. coli isolates. Previous in vitro mastitis studies revealed that LF had an antibacterial effect against some principal udder pathogens, especially E. coli (Kutila et al., 2003), and it can prevent sequelae related to the use of antimicrobial drugs (Jenssen and Hancock, 2009; Yen et al., 2011). In this study, we detected the antibacterial activity of LF as a cotreatment with marbofloxacin using the broth dilution method, which revealed that the MIC of marbofloxacin was 6 µg/ml with a bacterial count of 4.16 log cfu/ml and MBC was 7 µg/ml, while the MIC of LF was 2 mg/ml with a bacterial count of 4.41 log cfu/ml and MBC was 3 mg/ml. No growth was detected when the marbofloxacin (6 µg/ml) was combined with LF (2 mg/ml). This finding agrees with that reported by Moravian et al. (2014), who found that all concentrations of bovine LF significantly inhibited Gram-negative bacteria such as Salmonella, P. aeruginosa, and E. coli growth. Diarra et al. (2002b) reported that combining LF with antimicrobial agents yielded better outcomes than combining LF with antimicrobials or LF alone. Kell et al. (2020) reported that LF can be used as an intramammary infusion to protect the udder with exogenous LF against new infections during the involution stage when the animal’s own LF level is still low. LF has bacteriostatic or bactericidal effects and induces innate immunity in the host. Bukhari et al. (2016) advocated that LF in combination with antibiotics can improve udder defense mechanisms and inhibit bacterial proliferation. In the present study, we assessed the synergistic effect of LF as a cotreatment agent with marbofloxacin in the treatment of BM animals. BM cases were divided randomly into two groups. Group (A) received 2 g of LF every 12 hours for 3 days, in combination with marbofloxacin (1 cm/50 kg bw) for 3 days. Group (B) was treated with marbofloxacin 1 cm / 50 kg bw) for 3 days. The two groups were injected with flunixin meglumine (finadyne®) at a dose of 1 cm/50 kg. In this study, statistical analysis revealed a significant difference in posttreatment clinical parameters in group (A) compared to group (B). Group (A) showed rapid improvement in both local and systemic signs of BM. The time needed for the milk properties to return to normal levels in group (A) was less than that in group (B). CMT regained normal levels in group (A) earlier than in group (B). There was also a significant decrease in the E. coli count in group (A) compared with group (B). The E. coli count could not be detected after 4 days in group (A) and after 5 days in group (B). These results agreed with Kutila (2004) who used LF in treated BM cases and reported that systemic manifestations improved within 2–3 days, and local manifestations within 1 week. Bacteria were eliminated from enrofloxacin-treated animals within an average of 3.8 days and from LF-treated animals within 5.8 days. These results were in accordance with Petitclerc et al. (2007), who studied the effect of LF in combination with antibiotics in mastitis treatment, and Lacasse et al. (2008), who concluded that the combination of LF and antibiotics increases the cure rate as LF binds to the bacterial membrane through LPS, facilitating antibiotic penetration, and destroys the bacterial cell. Diarra et al. (2002a) demonstrated a synergistic effect of LF combined with antibiotics for BM treatment. Ongena et al. (2024) suggested that LF binding to lipid A in gram-negative bacteria causes discontinuity of the outer membrane and releases LPS, resulting in altered cell membrane permeability. Receptors for the N-terminal region of LF were noticed on the surface of some microorganisms. The binding of LF to these receptors causes cell death in Gram-negative bacteria due to disruption in its cell wall. In this study, 2 g of LF was infused into the infected udder to increase LF levels approximately to those present in mastitic milk; these levels are within the effective range. Low levels do not play therapeutic roles. High levels can produce moderate-to-severe inflammatory reactions in udder quarters and may even disturb or damage the normal function of the epithelial cells of the udder (Kell et al., 2020). Intramammary LF infusion might cause local reactions in the udder quarters. This could be due to a foreign body reaction of the udder to the exogenous protein. This can be attributed to the contamination of LF with endotoxins, which induce an inflammatory reaction. The dose of LF (2 gm) used in this study contained insignificant amounts of endotoxins, which could cause mild local reactions (Izquierdo-Vega et al., 2023). Therefore, we decided to use LF at 12-h intervals for 3 days to prolong the period of therapeutic LF levels in milk so that LF would better eliminate bacteria and LPS. The future perspectives of LF in treating BM are promising, with potential uses ranging from antimicrobial anti-inflammatory therapies to immune modulation and prevention. Future studies are needed to better understand the optimal use, dosing, and delivery mechanisms of LF in BM management. With antibiotic resistance becoming an increasing concern, LF-based drugs could play an important role in providing sustainable, effective alternatives for dairy animals. ConclusionBM is one of the most prevalent diseases in farm animals, leading to reduced dairy milk production and significant economic losses. E. coli is one of the primary causative agents of BM. Incorporating LF as a cotreatment with antibiotics, such as marbofloxacin, could limit the growth of E. coli bacteria better than marbofloxacin alone, thus enhancing cure rates. Conflicts of interestNone. FundingThis research received no specific grant from public, commercial, or nonprofit funding agencies. Authors’ contributionsAll authors made substantial contributions to all the following: substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data, acknowledgment of drafting the article or revising it critically for important intellectual content ; final approval of the version to be published; and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Mohamed Ibrahim Mostafa: Study sample collection, clinical examination of animals, collection of milk samples, isolation, and identification of E. coli, antibiotic sensitivity testing of E. coli, molecular detection of antibiotic-resistance genes, detection of MIC of marbofloxacin and LF by broth dilution method, treatment of the infected cases, follow-up of the cases for evaluation of the treatment outcome, and statistical analysis. Saber Ali Saad: Study sample collection, clinical examination of animals, collection of milk samples, treatment of infected cases, follow-up of the cases for evaluation of treatment outcome, and statistical analysis. Asmaa Elsayed Mohammed: Study sample collection, clinical examination of animals, collection of milk samples, isolation, and identification of E. coli, antibiotic sensitivity testing of E. coli, molecular detection of antibiotic-resistance genes, detection of MIC of marbofloxacin and LF, treatment of the infected cases, follow-up of the cases for evaluation of the outcome of treatment, statistical analysis, and journal correspondence. Elsaid Mohamed Mohamed Saafan: Study sample collection, clinical examination of animals, collection of milk samples, treatment of the infected cases, follow-up of the cases for evaluation of the treatment outcome, statistical analysis, and statistical analysis. Amal Mohamed El-Sayed: Study sample collection, clinical examination of animals, collection of milk samples, treatment of infected cases, follow-up of the cases for evaluation of treatment outcome, and statistical analysis. Data availabilityData are available upon request. ReferencesAbd-ELFatah, E.B., El-Emam, A., Mohamed, M., Shehata, A.A., Elsheikh, H.E. and Bakrash, M.M. 2024. Assessment of potential treatments for clinical mastitis caused by Escherichia coli in cattle using clinical and biochemical markers. Benha. Vet. Med. J. 47(1), 5–11. Al Habty, S.H. and Ali, D.N. 2023. Efficiency of lactoferrin to eradicate multidrug resistant Staphylococcus aureus isolated from some dairy products. Adv. Anim. Vet. Sci. 11(1), 35–44. Ali, M., Ahmad, M., Muhammad, K. and Anjum, A. 2011. Prevalence of sub-clinical mastitis in dairy buffaloes of Punjab, Pakistan. J. Anim. Plant. Sci. 21(3), 477–480. Andrews, J.M. 2001. Determination of minimum inhibitory concentrations. J. Antimicrob. Chemother. 48(1), 5–16. Arabi, H., Pakzad, I., Nasrollahi, A., Hosainzadegan, H., Jalilian, F.A., Taherikalani, M., Samadi, N. and Sefidan, A.M. 2015. Sulfonamide resistance genes (sul) m in extended spectrum beta-lactamase (ESBL) and non-ESBL producing Escherichia coli isolated from Iranian hospitals. Jundishapur. J. Microbiol. 8(7), e19961. Barua, M., Prodhan, M.A.M., Islam, K., Chowdhury, S., Hasanuzzaman, M., Imtiaz, M.A. and Das, G.B. 2014. Sub-clinical mastitis prevalent in dairy cows in Chittagong district of Bangladesh: detection by different screening tests. Vet. World. 7(7), 438–488. Bhanot, V., Chaudhri, S., Bisla, R. and Singh, H. 2012. Retrospective study on prevalence and antibiogram of mastitis in cows and buffaloes of eastern Haryana. Indian J. Anim. Res. 46(2), 160–163. Bukhari, S., Mir, M. and Baba, R.A. 2016. Bovine mastitis: novel protein treatment strategy. Camel. Int. J. Vet. Sci. 4(2), 15–25. Burvenich, C., Van Merris, V., Mehrzad, J., Diez-Fraile, A. and Duchateau, L. 2003. Severity of E. coli mastitis is mainly determined by cow factors. Vet. Res. 34(5), 521–564. Cheesbrough, M. 2006. District laboratory practice in tropical countries, part 2. Cambridge University Press. Constable, P.D., Hinchcliff, K.W., Done, S.H. and Grünberg, W. 2016. Veterinary medicine: a textbook of the diseases of cattle, horses, sheep, pigs and goats. Elsevier Health Sciences. da Silva, Z.N., da Cunha, A.S., Lins, M.C., Carneiro, L.d.A., Almeida, A.C.D.F. and Queiroz, M.L. 2001. Isolation and serological identification of enteropathogenic Escherichia coli in pasteurized milk in Brazil. Revista. Saude. Publica. 35, 375–379. Daka, D., Gsilassie, S. and Yihdego, D. 2012. Antibiotic-resistant Staphylococcus aureus isolated from cow’s milk in the Hawassa area, south Ethiopia. Ann. Clin. Microbiol. Antimicrob. 11, 1–6. De Boer, E. and Heuvelink, A. 2000. Methods for the detection and isolation of shiga toxin-producing Escherichia coli. J. Appl. Microbiol. 88(1), 133S–143S. Diarra, M., Petitclerc, D. and Lacasse, P. 2002a. Effect of lactoferrin in combination with penicillin on the morphology and the physiology of Staphylococcus aureus isolated from bovine mastitis. J. Dairy. Sci. 85(5), 1141–1149. Diarra, M., Petitclerc, D. and Lacasse, P. 2002b. Response of Staphylococcus aureus isolates from bovine mastitis to exogenous iron sources. J. Dairy. Sci. 85(9), 2141–2148. Diarra, M.S., Petitclerc, D. and Lacasse, P. 2002c. Effect of lactoferrin in combination with penicillin on the morphology and the physiology of (Staphylococcus aureus) isolated from bovine mastitis. J. Dairy. Sci. 85(5), 1141–1149. Doosti, A., Mahmoudi, E., Jami, M.S. and Mokhtari-Farsani, A. 2016. Prevalence of aada1, aada2, aadb, stra and strb genes and their associations with multidrug resistance phenotype in Salmonella Typhimurium isolated from poultry carcasses. Thai J. Vet. Med. 46(4), 691–697. El-Sayed, A and Kamel, M. 2021. Bovine mastitis prevention and control in the post-antibiotic era. Trop. Anim. Health. Prod. 53, 1–16. Fairbrother, J.H., Dufour, S., Fairbrother, J.M., Francoz, D., Nadeau, É. and Messier, S. 2015. Characterization of persistent and transient Escherichia coli isolates recovered from clinical mastitis episodes in dairy cows. Vet. Microbiol. 176(1–2), 126–133. Fatemi, F., Alizadeh Sani, M., Noori, S.M.A. and Hashemi. M. 2024. Status of antibiotic residues in milk and dairy products of Iran: a systematic review and meta-analysis. J. Environ. Health. Sci. Eng. 22(1), 31–51. Gao, J., Barkema, H.W., Zhang, L., Liu, G., Deng, Z., Cai, L., Shan, R., Zhang, S., Zou, J., Kastelic, J.P. and Han B. 2017. Incidence of clinical mastitis and distribution of pathogens on large Chinese dairy farms. J. Dairy. Sci. 100(6), 4797–4806. Hameed, S., Arshad, M., Ashraf, M., Avais, M. and Shahid, M. 2008. Prevalence of common mastitogens and their antibiotic susceptibility in Tehsil Burewala, Pakistan. Pak. J. Agri. Sci. 45(2), 227–246. He, N., Yang, X., Haque, A., Chen, J., Guo, Y., Li, J., Yao, L., Zhuo, C., Wang, J. and Wang, Y. 2024. Practice of standardization of clsi m45 a3 antimicrobial susceptibility testing of infrequently isolated or fastidious bacteria strains isolated from blood specimens in Guangdong province 2017–2021. Front. Microbiol. 15, 1335169. Izquierdo-Vega, J.A., Castillo-Juarez, R.J., Sánchez-Gutiérrez, M., Ares, M.A. and Miguel, A. 2023. A mini-review of enteroaggregative with a specific target on the virulence factors controlled by the aggr master regulator. Pol. J. Microbiol. 72(4), 347–354. Jenssen, H. and Hancock, R.E.W. 2009. Antimicrobial properties of lactoferrin. Biochimie. 91(1), 19–29. Jiang, Y.H., Xin, W.G., Yang, L.Y., Ying, J.P., Zhao, Z.S., Lin, L.B., Li, X.Z. and Zhang, Q.L. 2022. A novel bacteriocin against Staphylococcus aureus from Lactobacillus paracasei isolated from Yunnan traditional fermented yogurt: purification, antibacterial characterization, and antibiofilm activity. J. Dairy. Sci. 105(3), 2094–2107. Kell, D.B., Heyden, E.L. and Pretorius, E. 2020. The biology of lactoferrin, an iron-binding protein that can help defend against viruses and bacteria. Front. Immunol. 11, 1221. Kerro Dego, O. and Tareke, F. 2003. Bovine mastitis in selected areas of southern Ethiopia. Trop. Anim. Health. Prod. 35, 197–205. Kutila, T. Pyörälä, S., Saloniemi, H. and Kaartinen, L. 2003. Antibacterial effect of bovine lactoferrin against udder pathogens. Acta Vet. Scand. 44(1), 35. Kutila, T. 2004. Lactoferrin and citrate concentrations at drying-off and during early mammary involution of dairy cows. J. Vet. Med. 50(7), 350–353. Kutila, T., Suojala, L., Lehtolainen, T., Saloniemi, H., Kaartinen, L., Tähti, M., Seppälä, K. and Pyörälä, S. 2004. The efficacy of bovine lactoferrin in the treatment of cows with experimentally induced Escherichia coli mastitis. J. Vet. Pharmacol. Ther. 27(4), 197–202. Lacasse, P., Lauzon, K., Diarra, M. and Petitclerc, D. 2008. Utilization of lactoferrin to fight antibiotic-resistant mammary gland pathogens. J. Anim. Sci. 86(13), 66–71. Lamey, A., Ammar, A.M., Zaki, E., Khairy, N., Moshref, B.S. and Refai, M.K. 2013. Virulence factors of Escherichia coli isolated from recurrent cases of clinical and subclinical mastitis in buffaloes. Int. J. Microbiol. Res. 4(1) 86–94. Le, H.Q., Kim, S.K. and Yoon, J.W. 2024. Bovine mastitis-associated Escherichia coli. J. Food Hyg. Saf. 39(3), 181–190. Malt. 2013. ‘ISO 6496: 1999. Microbiological analysis Malt samples for microbiological testing were prepared by dilution method in conformity with standard LVS EN ISO 6887-1. 1999, 6887-4: 2004. Res. Rural. Dev. 1: 117. Marques-Bastos, S.L.S, Coelho, M.L.V., de Sousa Santos, I.N., Moreno, D.S.A., Barrias, E.S., De Mendonça, J.F.M., Mendonça, L.C., Lange, C.C., de Paiva Brito, M.A.V. and do Carmo de Freire Bastos, M. 2023. Effects of the natural antimicrobial peptide aureocin a53 on cells of Staphylococcus aureus and Streptococcus agalactiae involved in bovine mastitis in the excised teat model. World. J. Microbiol. Biotechnol. 39(1), 5. Martínez-Vázquez, A.V., Rivera-Sánchez, G., Lira-Méndez, K., Reyes-López, M.A. and Bocanegra-García, V. 2018. Prevalence, antimicrobial resistance and virulence genes of Escherichia coli isolated from retail meat in Tamaulipas, Mexico. J. Global. Antimicrob. Resist. 14, 266–272. Mohamed, S., Ammar, M., Aisha, R.A., Mona, A., Hakim, A., Bakry, M., Azza, S. and Eraqi, M. 2011. Molecular and virulence characterization of Escherichia coli strains isolated from persistent bovine mastitis. J. Am. Sci. 7, 614–624. Moradian, F., Sharbafi, R. and Rafiei, A.J. 2014. Lactoferrin, isolation,purification and antimicrobial effects. J. Atom Bioeng. 3, 203–206. Morales-Ubaldo, A.L., Rivero-Perez, N., Valladares-Carranza, B., Velázquez-Ordoñez, V., Delgadillo-Ruiz, L. and Zaragoza-Bastida, A. 2023. Bovine mastitis, a worldwide impact disease: prevalence, antimicrobial resistance, and viable alternative approaches. Vet. Anim. Sci. 21, 100306. Moroni, P., Rossi, C.S., Pisoni, G., Bronzo, V., Castiglioni, B. and Boettcher, P. 2006. Relationships between somatic cell count and intramammary infection in buffaloes. J. Dairy. Sci. 89(3), 998–1003. Nery Garcia, B.L., Dantas, S.T.A, da Silva Barbosa, K., Mendes Mitsunaga, T., Butters, A., Camargo, C.H. and Nobrega, D.B. 2024. Extended-spectrum beta-lactamase-producing Escherichia coli and other antimicrobial-resistant gram-negative pathogens isolated from bovine mastitis: one. Health. Perspect. Antibiot. 13(5), 391. Ongena, R., Dierick, M., Vanrompay, D., Cox, E. and Devriendt, B. 2024. Lactoferrin impairs pathogen virulence through its proteolytic activity. Front. Vet. Sci. 11, 1428156. Owuama, C.I. 2017. Determination of minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) using a novel dilution tube method. Afr. J. Microbiol. Res. 11(23), 977–980. Petitclerc, D., Lauzon, K., Cochu, A., Ster, C., Diarra, M.S. and Lacasse, P. 2007. Efficacy of a lactoferrin-penicilllin combination to treat β-lactam-resistant Staphylococcus aureus mastitis. J. Dairy. Sci. 90(6), 2778–2787. Pishtiwan, A.H. and Khadija, K.M. 2019. Prevalence of blatem, blashv, and blactx-m genes among esbl-producing Klebsiella pneumoniae and Escherichia coli isolated from thalassemia patients in Erbil, Iraq. Mediterr. J. Hematol. Infect. Dis. 11(1), e2019041. Rahimiyan, J., Shayegh, J. and Golzari, A. 2021. Effect of marbofloxacin on mastitis caused by Escherichia coli in dairy cows. Vet. Clin. Pathol. 14(56), 381–388. Robicsek, A., Strahilevitz, J., Sahm, D., Jacoby, G. and Hooper, D. 2006. Qnr prevalence in ceftazidime-resistant Enterobacteriaceae isolates from the United States. Antimicrob. Agents. Chemother. 50(8), 2872–2874. Saeed, S.I., Vivian, L., Zalati, C.S.C., Sani, N.I.M., Aklilu, E., Mohamad, M., Noor, A.A.M., Muthoosamy, K. and Kamaruzzaman, N.F. 2023. Antimicrobial activities of graphene oxide against biofilm and intracellular Staphylococcus aureus isolated from bovine mastitis. BMC Vet. Res. 19(1), 10. Sharun, K., Dhama, K., Tiwari, R., Gugjoo, M.B., Iqbal Yatoo, M., Patel, S.K., Pathak, M., Karthik, K., Khurana, S.K. and Singh, R. 2021. Advances in therapeutic and managemental approaches of bovine mastitis: a comprehensive review. Vet. Quart. 41(1), 107–136. Singh, A., Chhabra, D., Sikrodia, R., Shukla, S., Sharda, R. and Audarya, S. 2018. Isolation of E. coli from bovine mastitis and their antibiotic sensitivity pattern. Int. J. Curr. Microbiol. Appl. Sci.7(10), 11–18. Singh, B., Mal, G., Kalra, R.S. and Marotta, F. 2024. Probiotics against veterinary infectious diseases. In Probiotics as live biotherapeutics for veterinary and human health, 1: functional feed and industrial applications. Springer, pp: 391–407. Snedecor, G. and Cochran, W. 1980. Statistical method 7th ed. Ames, Iowa, USA: The Iowa State University Press, pp: 39–63. Tivendale, K.A., Allen, J.L., Ginns, C.A., Crabb, B.S. and Browning, G.F. 2004. Association of iss and iucA, but not tsh, with plasmid-mediated virulence of avian pathogenic Escherichia coli. Infect. Immun. 72(11), 6554–6560. Toleman, M.A., Bennett, P.M. and Walsh, T.R. 2006. Common regions eg orf 513 and antibiotic resistance: is 91-like elements evolving complex class 1 integrons. J. Antimicrob. Chemother. 58(1), 1–6. Yassin, S.A. and Abd Elhady, H.A. 2023. Utilization of lactoferrin to inhibit E. coli and S. aureus isolates from milk and kariesh cheese. Iraqi. J. Vet. Sci.37(4), 853–863. Yen, C.C., Shen, C.J., Hsu, W.H., Chang, Y.H., Lin, H.T., Chen, H.L. and Chen, C.M. 2011. Lactoferrin: an iron-binding antimicrobial protein against Escherichia coli infection. Biometals. 24(4), 585–594. Yu, P., Zhou, X., Li, Z. and Yan, Y. 2020. Inactivation and change of tetracycline-resistant escherichia coli in secondary effluent by visible light-driven photocatalytic process using ag/agbr/g-c3n4. Sci. Total. Environ. 705, 135639. Yue, L., Jiang, H.X., Liao, X.P., Liu, J.H., Li, S.J., Chen, X.Y., Chen, C.X., Lü, D.H. and Liu, Y.H. 2008. Prevalence of plasmid-mediated quinolone resistance qnr genes in poultry and swine clinical isolates of Escherichia coli. Vet. Microbiol. 132(3–4), 414–420. Zinnah, M., Bari, M., Islam, M., Hossain, M., Rahman, M., Haque, M., Babu, S., Ruma, R. and Islam, M. 2007. Characterization of Escherichia coli isolated from samples of different biological and environmental sources. Bangladesh J. Vet. Med. 5, 25–32. | ||
| How to Cite this Article |
| Pubmed Style Mostafa MI, Saad SA, Mohammed AE, Saafan EMM, El-sayed AM. Role of lactoferrin in the treatment of E. coli-induced bovine mastitis. Open Vet. J.. 2025; 15(2): 954-964. doi:10.5455/OVJ.2025.v15.i2.44 Web Style Mostafa MI, Saad SA, Mohammed AE, Saafan EMM, El-sayed AM. Role of lactoferrin in the treatment of E. coli-induced bovine mastitis. https://www.openveterinaryjournal.com/?mno=240422 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.44 AMA (American Medical Association) Style Mostafa MI, Saad SA, Mohammed AE, Saafan EMM, El-sayed AM. Role of lactoferrin in the treatment of E. coli-induced bovine mastitis. Open Vet. J.. 2025; 15(2): 954-964. doi:10.5455/OVJ.2025.v15.i2.44 Vancouver/ICMJE Style Mostafa MI, Saad SA, Mohammed AE, Saafan EMM, El-sayed AM. Role of lactoferrin in the treatment of E. coli-induced bovine mastitis. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 954-964. doi:10.5455/OVJ.2025.v15.i2.44 Harvard Style Mostafa, M. I., Saad, . S. A., Mohammed, . A. E., Saafan, . E. M. M. & El-sayed, . A. M. (2025) Role of lactoferrin in the treatment of E. coli-induced bovine mastitis. Open Vet. J., 15 (2), 954-964. doi:10.5455/OVJ.2025.v15.i2.44 Turabian Style Mostafa, Mohamed Ibrahim, Saber Ali Saad, Asmaa Elsayed Mohammed, Elsaid Mohamed Mohamed Saafan, and Amal Mohamed El-sayed. 2025. Role of lactoferrin in the treatment of E. coli-induced bovine mastitis. Open Veterinary Journal, 15 (2), 954-964. doi:10.5455/OVJ.2025.v15.i2.44 Chicago Style Mostafa, Mohamed Ibrahim, Saber Ali Saad, Asmaa Elsayed Mohammed, Elsaid Mohamed Mohamed Saafan, and Amal Mohamed El-sayed. "Role of lactoferrin in the treatment of E. coli-induced bovine mastitis." Open Veterinary Journal 15 (2025), 954-964. doi:10.5455/OVJ.2025.v15.i2.44 MLA (The Modern Language Association) Style Mostafa, Mohamed Ibrahim, Saber Ali Saad, Asmaa Elsayed Mohammed, Elsaid Mohamed Mohamed Saafan, and Amal Mohamed El-sayed. "Role of lactoferrin in the treatment of E. coli-induced bovine mastitis." Open Veterinary Journal 15.2 (2025), 954-964. Print. doi:10.5455/OVJ.2025.v15.i2.44 APA (American Psychological Association) Style Mostafa, M. I., Saad, . S. A., Mohammed, . A. E., Saafan, . E. M. M. & El-sayed, . A. M. (2025) Role of lactoferrin in the treatment of E. coli-induced bovine mastitis. Open Veterinary Journal, 15 (2), 954-964. doi:10.5455/OVJ.2025.v15.i2.44 |