| Research Article | ||
Open Vet. J.. 2025; 15(2): 965-976 Open Veterinary Journal, (2025), Vol. 15(2): 965-976 Research Article The mitigating effect of dietary β-glucan against fipronil-induced intoxication in Nile Tilapia (Oreochromis niloticus): Histopathological, immunological, hematological, and biochemical analysisFawiziah Khalaf Alharbi*Department of Biology, College of Science, Qassim University, Buraydah, Saudi Arabia *Corresponding Author: Fawiziah Khalaf Alharbi. Department of Biology, College of Science, Qassim University, Buraydah, Saudi Arabia. Email: hrbief [at] qu.edu.sa Submitted: 30/12/2024 Accepted: 07/02/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
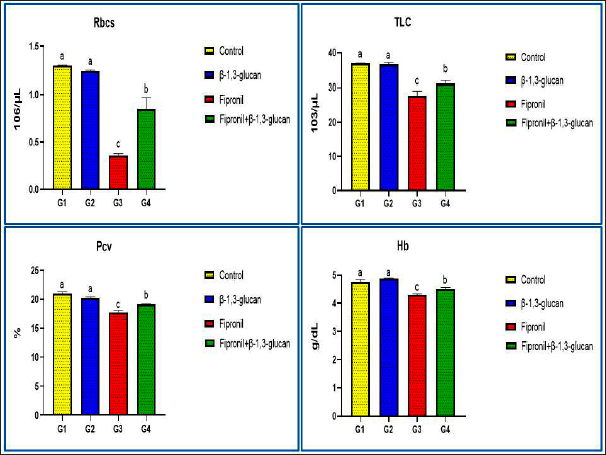
AbstractBackground: Due to its remarkable effectiveness against a wide range of pests and insects at very low concentrations, the broad-spectrum fipronil pesticide is currently gaining popularity in the agricultural, public health, and international industries. However, the stressor effects of fipronil insecticides cause ecological disruption, growth retardation, immunosuppression, and higher fish mortality rates. Both animals and humans have demonstrated the effectiveness of prebiotics such as β-1,3-glucan in their diets. Aquaculture has recently increased in use because of its potential to control diseases, compete with environmental stresses, and promote fish growth. Aim: The goal of this study was to determine how dietary β-1,3-glucan can protect Nile Tilapia (Oreochromis niloticus) fish from fipronil’s harmful effects. Methods: We randomly divided 240 fish into four equal groups. As a control, the first group (G1) was fed a standard diet. A 0.1% dose of -1, 3-glucan was added to G2. Fipronil was added to G3 at a concentration of 2.8 mg/l (1/10 96 h LC50). At the indicated concentrations, G4 was combined with β-1, 3-glucan, and fipronil. Alterations in vital signs, metabolic profiles, immunological responses, blood counts, and any histological abnormalities in the liver or spleen of the fish were investigated and recorded. Results: The fipronil-exposed group exhibited slow mobility, respiratory discomfort, and increased mucus secretion. Several blood markers, like immunoglobulin M and lysozyme, were found to be significantly lower. On the other hand, the levels of aspartate aminotransferase, alanine aminotransferase, urea, creatinine, and cortisol in the serum were significantly higher. Liver histopathology revealed hemorrhagic blood vessels, steatosis in hepatocytes, hydropic degeneration, and widespread necrosis. Furthermore, we noted serious splenic parenchymal necrosis, hemorrhagic red pulp, white pulp depletion, and hemosiderosis. Histological changes were slowed by G4, which had β-1,3-glucan and fipronil. Moreover, it increases blood markers and physical activity levels. Conclusion: The results show that β-1,3-glucan is an effective dietary supplement for Nile tilapia, and it improves their health, increases their immunity, and neutralizes fipronil contaminants in fish farming. Keywords: Fipronil, β-1,3-glucan, Toxicity, Nile Tilapia, Histopathological, Immunological, Biochemical. IntroductionDue to the severe decline in biodiversity that has occurred in recent decades, freshwater ecosystems are among the most fragile on Earth (Geist, 2011). From a pragmatic and financial perspective, fish are an excellent protein source for poor nations (Soliman and Yacout, 2016). According to Doak and Morris (2010), fish ecosystems are under threat from a variety of stressors, including chemical contamination, nutrient fluctuations, climate change, habitat loss, exploitation, acidification, and biological invasion. A major contributor to chemical stress is the widespread and careless use of pesticides, especially in agriculture, which contaminates water sources and poses a threat to aquatic life (Barbieri, 2009; Schäfer et al., 2011). When pesticides pollute waterways to dangerous levels, they kill fish or cause them to starve to death by killing food sources. Researchers have also shown that many toxicants can damage aquatic organism tissues and slow down their reproduction and growth (Kalavathy et al., 2001; Srivastav et al., 2002). With its broad spectrum of action, fipronil is a new insecticide that belongs to the N-phenylpyrazole class. The International Union of Pure and Applied Chemistry refers to the chemical compound known as fipronil is referred to as (±)-5-amino. In addition to being referred to as -1-(2,6-dichloro-α,α,α-trifluoro-p-tolyl)-, P4-trifluoromethyl sulfinyl pyrazole-3-carbonitrile is known as according to Tomlin (2006). The Environmental Protection Agency (EPA) of the United States has identified fipronil as a potential substitute for organophosphate compounds (U.S. EPA, 2002; Chiovarou and Siewicki, 2008). As a result of its lipophilicity and persistency properties, fipronil has recently garnered much attention and is utilized extensively in agriculture, public health, and international markets (Mulrooney et al., 1998; Tingle et al., 2003). This is because it is effective against a wide variety of pests at very low concentrations. One application not related to agriculture is the management of veterinary pests (Jennings et al., 2002). It was demonstrated by Tomlin (2006) and U.S. EPA (1996) that fipronil is effective against a wide range of insects. On the other hand, Zhang et al. (2018) reported that fipronil has been linked to a number of negative health effects in both humans and animals. There are a number of impacts, one of which is the encouragement of neuronal cell damage, which ultimately results in death through the production of reactive oxygen species. Furthermore, compared with fipronil, the metabolites sulfone and sulfide are significantly more hazardous (Wang et al., 2019). The effects of fipronil are quite noticeable in a number of fish species. Researchers found that fipronil can have genotoxic and cytotoxic effects, weaken the immune system, and stop growth in amounts below the level needed to kill. It can also have other effects that are not fatal. The harmful effects differ according to the type of fish and the method of exposure. In water, fipronil induces changes in the gills of Caspian white fish (Ardeshir et al., 2017), as well as changes in the skin color and behavior of juvenile brown shrimp (Al-Badran et al., 2018). These changes were observed in both white fish and brown shrimp. Bownik and Szabelak (2021) explained the physiological and behavioral changes in Daphnia magna. According to the findings of numerous studies, fipronil is extremely harmful to Nile tilapia. According to Guedes et al. (2020), when it accumulates in the tissue of fish, it causes the fish to die and causes cell and gene damage. Several researchers, including Ansoar-Rodríguez et al. (2015), Correia et al. (2017), and AnvariFar et al. (2018), have reached a consensus that the species Oreochromis niloticus, also known as Nile tilapia, is an effective method for identifying contaminants in the environment. Some researchers, like Piner and Uner (2012), Ansoar-Rodríguez et al. (2016), Jia et al. (2019), and Meng et al. (2019), have reported that this species has been utilized for the purpose of identifying stress proteins and DNA fragmentation. These molecular biomarkers provide valuable information regarding the extent of pesticide toxicity. There are a number of negative effects of fipronil on the hematological, biochemical, histopathological, and other growth performance indicators of Nile tilapia. In addition, it alters the expression of genes involved in apoptosis, stress, inflammation, and antioxidants (Fadl et al., 2022). Araújo and Blasco (2019) found that fish populations drop, disease immunity drops, and predator avoidance drops when exposed to repeated exposure to pesticide pollutants. Pollutants have an indirect impact on human health and aquaculture. People who eat seafood can absorb some contaminants. There is growing concern about the potential hazardous effects of pesticides on fish tissues for commercial purposes, especially because Nile tilapia is consumed by people in large quantities. It is important to identify natural compounds that can be added to fish food to improve their health and immunity. This is necessary to prevent fish from getting sick and transmitting pollution to humans. Animal and human studies have demonstrated the efficacy of prebiotics in food. Recently, they have been in the spotlight in aquaculture for their potential to boost the development of farmed fish, compete with various environmental stresses, and manage diseases. Welker et al. (2007) say that β-Glucans have been widely used as a prebiotic in aquaculture to help many types of fish live longer, grow faster, and convert more food into energy. Based on the information provided, the introduction of fipronil insecticide into rivers can cause damage to ecosystems. This can, in turn, affect physiological parameters, stunt growth, depress the immune system, and increase the mortality rate of fish. Oreochromis niloticus, often known as Nile tilapia, is a tropical freshwater fish that many nations enjoy eating. Pesticides have the potential to harm these fish, which are among the most farmed in the world. Consequently, safeguarding humans from the tissue remnants of these contaminants and minimizing their negative effects on Nile tilapia culture is of utmost importance. We think that giving Nile tilapia immunomodulatory substances like β-1,3-glucan prebiotics would improve their health by blocking the effects of fipronil stressors, strengthening their natural defenses, stopping changes that are not healthy, and so on. The goal of this study was to determine if dietary β-1,3-glucan could protect Nile tilapia (O. niloticus) against fipronil poisoning. This was done by measuring different immunological, biochemical, and hematological parameters and looking at the fish’s tissues. Rather than spending a fortune on harmful pesticides, this approach would improve fish health while shielding fish eaters from harmful residues in fish tissues. Materials and MethodsChemicalsWe sourced 20% fipronil from YongNong Biosciences Co., Ltd. in China. Technical grade Fipronil (C12H4Cl2F6N4OS) produced by Bio Quest International Private Limited in Mumbai, India, contained equal amounts of the γ and β isomers. It was 99.1% pure. The dose used in the present study was based on a previous report (El-Murr et al., 2015).We combined the basic diet components with dissolved beta-glucan: 1-glucan and 3-glucan (Sigma, USA), in phosphate-buffered saline (PBS). The mixture was then added to the components at a rate of 0.1% before palletization. A previous study (Nikl et al., 1992) was used to guide the feeding period, and β-1, 3 glucan dose was used in this study. Fish management and maintenanceTwo hundred forty O. niloticus with an average body weight of 35 ± 1.0 g were used in this investigation. The fish used in this study were from a private farm in Abbassah, Sharkia Province, Egypt. The fish did not appear to have any external parasites or skin blemishes, suggesting that they were in excellent health. The first step in treating fish for fungal infections and external parasites is to soak them in a salt bath with 2.5% NaCl for 5 minutes. The glass aquaria was equipped with aerators and thermostatically regulated heaters. The water in each aquarium was chlorine-free and measured 80 x 30 x 40 cm. After the fish had acclimated to the laboratory setting for two weeks, the experiments began, lasting a total of two months. We fed them a baseline diet consisting of 30% crude protein. We fed the fish 5% of their body weight in dry matter three times a day. Over the course of the experiment, the water’s average values were as follows: temperature 25.5 ± 2.0 1C, pH 6.4 ± 0.2, dissolved oxygen 5.1 ± 2.0 mg/l, non-ionized ammonia 0.8 ± 0.01 µg/l, and nitrite 0.06 ± 0.01 mg/l. Experimental protocolWe separated the fish into four distinct groups, each of which contained twenty fish in each aquarium. A conventional meal was given to the fish in the first group (G1), which served as the control group. There were no supplements or other interventions administered to the fish in this group. Furthermore, the second group, denoted as G2, was provided with a conventional meal containing 0.1% β-1, 3-glucan. In the third group (G3), participants were given a conventional meal while being exposed to 2.8 mg/l (1/10 96 h LC50) of fipronil. For the fourth group of fish, which we refer to as G4, a basic meal consisting of 0.1% 1,3-β-glucan was administered prior to their exposure to fipronil at the concentrations that were discussed before. The fish were observed on a regular basis to check for any changes in coloration, abnormal behavior, clinical signs, or respiratory distress. This was done to ensure that the fish were in good health. Both the mortality rate and the postmortem lesions found on the fish that were impacted were reported. Sample collectionAt the end of the experiment, blood samples were collected by gently shaking a medical syringe that had been washed with Ethylenediamine tetraacetic acid (EDTA) solution (an anticoagulant) and puncturing caudal blood arteries. We then used the blood sample for hematological analysis. We drew serum blood without using an anticoagulant and stored it at −20°C until we measured biochemical and immunological parameters. After this, we beheaded the fish and preserved their livers and spleens in neutral buffered formalin for subsequent histological analysis. Humoral immunological studiesLysozyme test: The turbidometric technique was used to measure serum lysozyme concentrations (Schultz, 1987). We used an Enzyme-linked immunosorbent assay (ELISA) kit, C (alog Number CSB-E12045Fh (96 tests) to measure immunoglobulin M (IgM), CUSABIO BIOTECH CO., LTD. The assessment involves several biochemical variables. Blood tests were performed for cortisol, aspartate aminotransferases (AST), and alanine aminotransferases (ALT), urea, and creatinine. We estimated the following hematological parameters: red blood cell (RBC) count, packed cell volume (PCV), hemoglobin, and total leukocyte count. Histopathological examinationLiver and spleen samples were collected and quickly fixed in 10% buffered neutral formalin solution for at least 48 hours prior to histopathological analysis. The samples were then processed histologically, which included dehydrating them in ethanol grades from 70% to 100%, cleaning them in xylene, and embedding them in paraffin. Following the steps outlined by Suvarna et al. (2018), 5-micron-thick paraffin slices were prepared and regularly stained with hematoxylin and eosin dyes before examination under a microscope. Statistical analysisThe statistical analysis presented the results as the mean ± SEM. To compare the four treatment groups for the various biochemical indicators, we utilized a one-way analysis of variance as well as Tukey’s Honestly Significant Difference test. To demonstrate the statistical significance of our findings, we used a p-value of 0.05. GraphPad Prism 8.0.2 and the Statistical Package for the Social Sciences, version 24.0 (both created by IBM Corporation and located in Armonk, New York) were used to perform data analysis and visualization. Ethical approvalThe Institutional Animal Care and Use Committee at Zagazig University in Egypt has assessed and approved the study protocol. They also assigned the approval number (ZU—IACUC; No. ZU-IACUC/ 2/F / 297 / 2024). ResultsMortality rates in different Nile Tilapia (O. niloticus) groupsIn the third group exposed to fipronil, half of the Nile tilapia exposed to fipronil perished. The fourth group, which received β 1,3-glucan in addition to fipronil, experienced a mortality rate of 5%. There was no mortality in either the control group (G1) or the group that received β-1,3-glucan supplements (G2). Clinical signs and postmortem lesionsThe third generation of fipronil-exposed fish showed no response to reflex tests, sluggish swimming, or respiratory distress. In addition, a thick coating of mucus engulfed the entire body. The gills also looked pale because there was a lot of mucus secreted from them. The postmortem examination revealed congestion of all internal organs and an enlarged gallbladder. On the other hand, the G4 group, which received both β-1,3-glucan and fipronil, exhibited better physiological functions. They had a strong defense against capture, had no clinical symptoms, had no breathing problems, and had a normal weight. The fish in the G2 group that were given β-1,3-glucan had the most physiological health benefits and gained the most body weight among the four groups that were studied. The fish in the G1 control group, on the other hand, had no unusual symptoms and weighed healthy weights. Hematological evaluationAll hematological parameters measured in the third group of fish exposed to fipronil decreased significantly. These included the numbers of RBCs, PCV, hemoglobin, and total leucocyte counts (TLCs). On the other hand, all of the hematological parameters measured increased significantly in the groups that were treated with β-1,3-glucan, either by itself (G2) or with fipronil (G4) (Fig. 1).
Fig. 1. Showing the effect of fipronil and β-1,3-glucan on some hematological parameters; RBCs (red blood cells), total leucocytes counts, packed cell volume, hemoglobin for all experimental groups. According to Tukey’s Honestly test, the different letters (a–c) were significantly different between treatments at the p < 0.05 level.
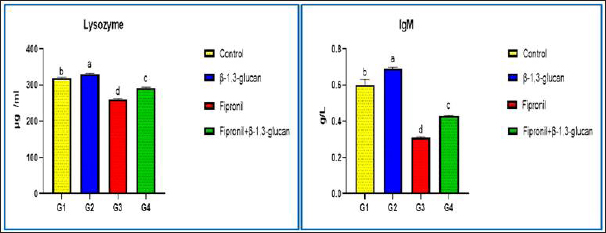
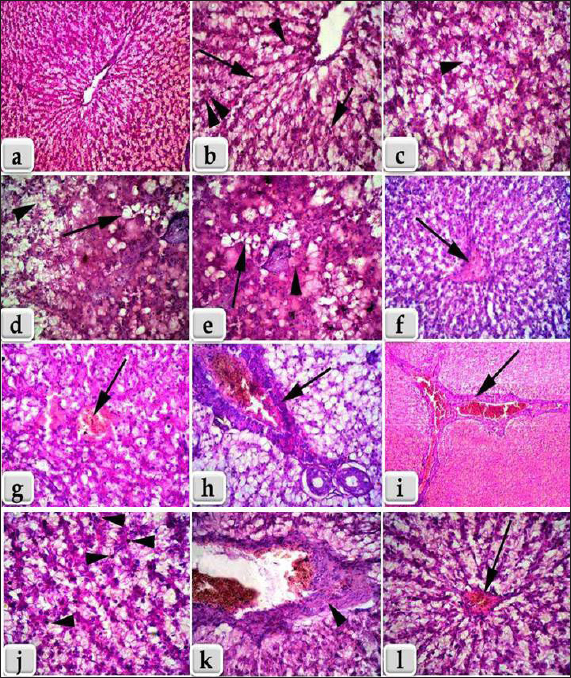
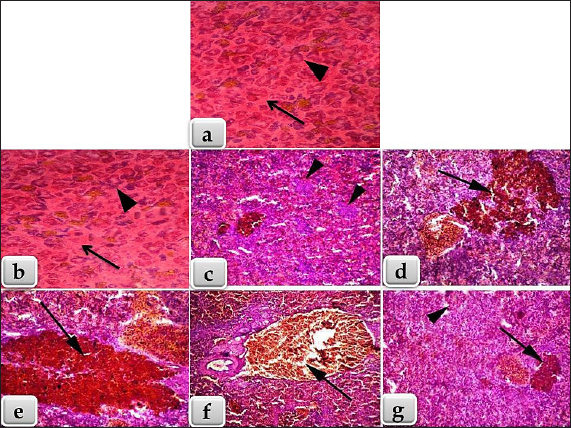
Fig. 2. Showing the effect of fipronil and β-1,3-glucan on some immunological parameters; lysozyme and immunoglobulin M for all experimental groups. According to Tukey’s Honestly test, the different letters (a–d) were significantly different between treatments at the p < 0.05 level. Humoral immunological parametersThe amounts of lysozymes and IgM in G2 that received β-1,3-glucan were much higher than those in the control group. Fipronil significantly decreased the amounts of IgM and lysozyme in G3 fish compared with G1 and G2. Although G4 was given β-1,3-glucan and fipronil as supplements, lysozymes, and IgM levels were slightly lower, but they were still approximately the same as in the control fish (Fig. 2). Biochemical parametersCompared with the G1 control group, the G2 groups that were treated with β-1,3-glucan alone, and the G4 groups that were treated with both β-1,3-glucan and fipronil, the levels of ALT and AST in the blood of O. niloticus G3 animals exposed to fipronil were greater in the G3 animals. In addition, the levels of both blood cortisol and renal function markers, specifically creatinine and urea, increased. In Figure 3, it is evident that the incorporation of β-1,3-glucan and fipronil into G4 resulted in a favorable impact, resulting in a reduction of cortisol levels as well as all liver and kidney indicators that were examined. Histopathology examinationThis study showed that the liver and spleen histoarchitecture did not change significantly in the G1 group, control group, or the group that was given β-glucan. This was based on histological examination. Regarding the histoarchitecture of all the chosen organs, the G3 organ with fipronil showed the most significant pathological alterations. Moreover, all of the chosen organs displayed normal and intact histoarchitecture in the G4 that was supplemented with β-1,3-glucan and fipronil although the effect was minor. The livers of Nile Tilapia (O. niloticus) fish in the G1 control and G2 groups that had β-glucan added to them did not show any signs of disease. We found the liver normal, homogeneous, and undamaged. Hepatocytes were distributed around the edges of the hepatic lobules in a plate or cord pattern that started at the central vein and went outwards. Due to the extremely fine, loose connective tissue, the hepatic lobulations were difficult to visualize (Fig. 4a). Higher magnification reveals a sponge-like appearance in the liver parenchyma, primarily composed of large, irregular, polygonal hepatocytes. Large, solitary, central, or subcentral spherical nuclei, conspicuous nucleoli, and occasional binucleation are typical features of these hepatocytes. A pale or vacuolar region is often present in the nucleus. There is a lot of glycogen in hepatocytes; thus, their cytoplasm is pale and uniform. In addition, normal sinusoidal structures loaded with erythrocytes laterally separated the hepatic cords. Figure 4b shows the presence of melanomacrophage centers (MMCs) and Kupffer cells. The third group of Nile Tilapia had livers with widespread necrosis, many lymphocytes, and very few erythrocytes. Figures 4c and d shows areas of necrotic tissue with focally enlarged necrotic cells, pyknosis, and karyorrhexis of their nuclei. There were vacuolations (vacuolar degeneration) and widespread hydropic degeneration in the hepatocytes, as well as disorganization of the hepatic cord. We observed deterioration of the liver’s cellular architecture, along with massive distention, vacuolation, and microvesicular fatty degeneration. The study also revealed steatosis or fatty alterations within the cytoplasm of hepatocytes (Fig. 4d and e). Because proteins clumped together, hepatocytes had cytoplasm that was both homogeneous and acidophilic, indicating severe coagulative necrosis. A few vacuolated hepatocytes had a few eosinophilic hyalinized globules, which are sometimes called Mallory bodies, in their cytoplasm (Fig. 4d and e). Severe congestion in the hepatic blood vessels, especially the central vein (Fig. 4f), sinusoids, and hemorrhages (extravasated RBCs) among the hepatic cells were observed (Fig. 4g). Figure 4h and i further shows that the portal blood vessels were severely congested and hemorrhaging. Figure 4j also shows the presence of lymphocytic infiltration throughout the liver tissue. Additionally, we noted fibrous tissue proliferation and severe bile duct thickening (Fig. 4k). The researcher found areas of long-lasting inflammation marked by the growth of fibrous connective tissue and the presence of cells that cause inflammation. Meanwhile, Figure 4l shows that the fourth group, which received a combination of fipronil and 1,3-β-glucan, had normal and undamaged hepatic parenchyma, regular hepatic cords, and minor congestion inside the central vein. As shown in Fig. 5a and b, the spleens of Nile Tilapia from the G1 control group and the G2 group that was given β-glucan did not exhibit any abnormalities. There was normal, undamaged parenchyma in the spleens; white pulp and red pulp as well as activated MMCs that stained darkly. The third group of Nile tilapia that was exposed to fipronil had moderate to severe necrosis of the splenic parenchyma. This showed uasdepletion in the white pulp, lymphatic nodules, and hemorrhage in the red pulp, and a loss of normal architecture in some areas (Fig. 5c). In severe haemosidriosis, red blood cells break down and hemosiderin pigment leaks out. The pigment then accumulated, creating vast patches of golden yellow inside the splenic parenchyma (Fig. 5d and e). Furthermore, severe congestion and hemorrhage were noted within the splenic blood vessels in the red pulp (Fig. 5f). Meanwhile, Figure 5g also shows that the fourth group had normal and intact splenic parenchyma, accompanied by mild depletion of white pulp and small patches of hemosiderin pigment (Fig. 5g).
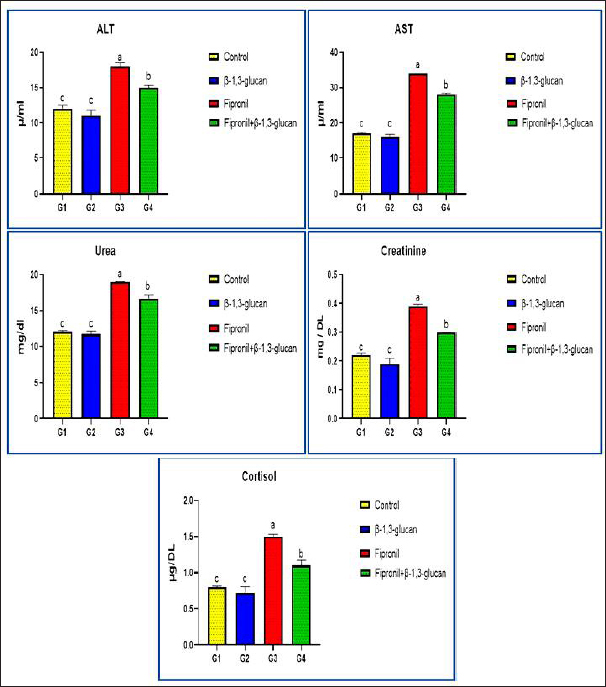
Fig. 3. Showing the effect of fipronil and β-1,3-glucan on some biochemical parameters; alanine aminotransferase and aspartate aminotransferase as liver function markers, urea and creatinine as kidney function markers, and cortisol for all experimental groups. According to Tukey’s Honestly test, the different letters (a–c) were significantly different between treatments at the p < 0.05 level. DiscussionAs the top predators in most water habitats, fish are also the most vulnerable to pollution. Besides adding harmful chemicals to fish meat and fish products, heavy metal contaminants can make fish less able to smell, swim poorly, have slower metabolisms, hurt important organs, and weaken their immune systems, all of which can raise their risk of getting sick or dying. Therefore, it is critical to increase resistance to contaminants like fipronil that could affect fish farming via streams and the environment. Improving the health and disease resistance of cultured fish is the goal of functional feeding, a new aquaculture paradigm that seeks to create balanced meals with feed additives (Li and Iii, 2004). This study examined what happens when Nile tilapia are exposed to fipronil and how adding β-1,3-glucan to their food can help lessen these adverse effects.
Fig. 4. (a, b): Section of Nile Tilapia (Oreochromis niloticus) liver of the G1; control and G2 supplemented with β-glucan. a) showing normal, homogenous, intact hepatic parenchyma with normal central vein. b) Higher magnification of figure a showing normal and distinct radiated hepatic cords from the central vein to the periphery of the hepatic lobules (long arrow) with normal hepatocytes of irregular polygonal shaped cells with single, central, large vesicular nucleus and pale cytoplasm with slightly vacuolations (single arrow head), large like kupffer cells (short arrow) and melanomacrophage centers (double arrow heads).(c–k): Liver of fish of the G3 exposed to fipronil, c) showing diffuse necrosis, hydropic degenerations and vacuolations within the hepatocytes with disorganization of the hepatic cords (arrow head). d) showing diffuse microvesicular fatty degenerations , pyknotic central nuclei and pale cytoplasm with sever vacuolations (arrow), beside diffuse area of hydropic degeneration (arrow head). e) showing sever coagulative necrosis with homogeneous and acidophilic cytoplasm, due to the coagulation of cytoplasmic proteins (arrow head) and diffuse microvesicular fatty degenerations (arrow). f) showing sever congestion and hemorrhage of the central vein (arrow). g) showing congestion, hemorrhage and extravasated erythrocytes in between the hepatocytes (arrow). h, i) showing sever congestion and hemorrhage in the portal blood vessels. j) showing sever necrosis of hepatocytes infiltrated with numerous lymphocytes (arrow head). k) showing sever thickening of the bile duct with fibrous tissue proliferation (arrow head).(l): liver of fish of the G4 supplemented with 1,3 β-glucan and fipronil showing normal, intact hepatic parenchyma and hepatic cord, but with mild to moderate congestion within the central vein (arrow). Stain: All (hematoxylin and eosin). Magnification power: All = X 400, except for a, f = X 100, and i = X 40.
Fig. 5. (a) Section of Nile Tilapia (Oreochromis niloticus) G1 spleen; control group showing normal, intact splenic parenchyma; white pulp (arrow heads) and red pulp (arrow).(b) Section of Nile Tilapia (O. niloticus) G2 spleen supplemented with β-glucan showing normal, intact splenic parenchyma; white pulp (arrow heads) and red pulp (arrow).(c–f) Section of Nile Tilapia spleen of the G3 exposed to fipronil, c) showing necrosis of the splenic parenchyma; white and red pulps with some loss of normal architecture and depletion of the white pulp (arrow head). d, e) showing severe hemorrhage; hemolysis of RBCs and escape of large amount of haemosidrin pigment that accumulated forming large golden yellow patches of haemosiderin pigments within the splenic parenchyma (arrow). f) showing severe congestion and hemorrhage within the splenic blood vessels; red pulp (arrow).(g) Section of Nile Tilapia spleen of the G4 supplemented with 1,3 β-glucan and fipronil showing normal, intact splenic parenchyma but with mild depletion of white pulp (arrow head) and mild haemosidriosis represented in small golden yellow patches of haemosiderin pigments within the splenic parenchyma (arrow). Stain: All (hematoxylin and eosin). Magnification power: All = X 100, except for a and b = X 40. The results revealed that G3 exposed to fipronil experienced a maximum mortality rate of 50%. The mortality rate was reduced to 5% in the G4 group that got β1,3-glucan + fipronil, which had a positive effect. Elmowalid et al. (2023) found that in catfish (Clarias gariepinus), the third group exposed to lead nitrate and fipronil had the highest death rate (45%. This conclusion is consistent with our own findings. On the other hand, in the fourth group, which received 1,3-glucan along with a combination of fipronil and lead nitrate, 5% of the participants died. A study by El-Murr et al. (2015) found that fish exposed to 0.014 mg/l of fipronil for 4 days had a 53% mortality rate. Fish exposed to 0.0042 and 0.002 mg/l for 10 weeks had rates of 21% and 8% mortality, respectively. The fish in the fipronil-exposed group (G3) had a slow escape reaction, moved slowly, had trouble breathing, and had a thick layer of mucus covering their bodies. A problem with the central nervous system caused by fipronil infection could be one reason for the symptoms and high death rates observed in fish. Gibbons et al. (2015) reported that this disruption occurs because fipronil stops chloride ions from moving through Gamma-aminobutyric acid (GABA) and glutamate-gated chloride channels. This condition, in turn, causes muscular exhaustion, nervous manifestation, and respiratory distress. However, when G4 were given β-1,3-glucan along with fipronil, their physiological responses were better. They reacted more quickly when they were caught; they did not have any clinical symptoms; their weight was normal, and they did not have any trouble breathing. The results of Elmowalid et al. (2023) in catfish (C. gariepinus) corroborated these findings. They found that fipronil and lead had negative toxic effects on catfish when supplemented with 1,3 β-glucan at a dose of 0.1%. In addition, giving the fish β-1,3-glucan supplements made their overall health, and the mortality rate decreased from 45% to 5% during the 2-month study period. In addition, the groups that were given β-1,3-glucan in their food did not experience any strange physical changes, like losing weight, moving in strange ways, discolored skin, or changes in the shape of their spines (Nikl et al., 1992). Hematological parameters were measured. In the G3 fish that were exposed to fipronil, all of the parameters measured decreased significantly compared with the groups that were treated with β-1,3-glucan. These parameters included RBC count, PCV, hemoglobin, and TLC. Ghisi et al. (2011) first noticed that fipronil damages the production of erythrocytes in the silver catfish Rhamdia quelen. They discovered that erythrocytes are hurt at very low concentrations of fipronil (0.0002 mg/l, or 0.2 µg/l). El-Murr et al. (2015) also discovered that fish exposed to fipronil had lower hemoglobin and total leukocyte counts compared to the control group. Based on Clasen et al. (2012), this could be because hemoglobin changes quickly into methemoglobin or because fipronil produces oxygen radicals, which are harmful and cause oxidative stress. In addition, Gupta et al. (2012) discovered that a sublethal dose (1/10th LC50) of Fipronil significantly decreased the number of red blood cells, white blood cells, and hemoglobin in caprinus carpio fry after 45 days of exposure. Abouelghar et al. (2020) also discovered that medium and high doses of fipronil lowered the hemoglobin, platelets, red blood cells, and white blood cells in mice after 28 days of exposure compared with the control group. Based on the analysis of immunological markers, the group exposed to fipronil had much lower IgM and lysozyme levels. Compared with the control group and the group that was exposed to fipronil, the group that was given β-1,3-glucan had much higher levels of lysozymes and IgM. This suggests that the immune system was affected by fipronil in a way that dietary 1,3-glucan could help. Elmowalid et al. (2023) conducted a study on catfish (C. gariepinus) and found that fipronil had an immunosuppressive effect. Researchers observed a significant decrease in the levels of interleukin-1 (IL-1), interleukin-6 (IL-6), and IgM. They also observed a drop in the expression of genes that encode these proteins. In addition, the catfish that were given β-1,3-glucan had significantly higher amounts of IL-1β, IL-6, and IgM in their plasma compared with the control group. Another study that supported these results was conducted by Gupta et al. (2014). They showed that fipronil weakens fish immune systems by lowering levels of lysozyme and nitroblue tetrazolium in the blood of Cyprinus carpio fry exposed to a low dose of fipronil (1/10th LC50 for 96 hours) for 45 days. Clasen et al. (2012) reported the same results after administering C. carpio 0.65 mg/l fipronil for 7, 30, and 90 days. They discovered that exposure altered the immune status of various common carp tissues, altered the antioxidant profile, and elevated oxidative stress parameters. Fipronil changes the activity of catalase and superoxide dismutase (SOD), which could be one reason for its negative effect on the immune system. Enzyme SOD provides the best defense against oxidative stress and is the first to react to free radicals (Van der Oost et al., 2003). The biochemical parameters of G3 of O. niloticus exposed to fipronil showed a clear rise in serum levels of urea and creatinine, liver function markers (ALT and AST), and cortisol. This result was compared to the control group and the groups that were treated with β-1,3-glucan. These results exactly match those of El-Murr et al. (2015), who found that fipronil exposure increased serum levels of ALT and AST in Nile tilapia (O. niloticus) by a large amount. It is well-known that fipronil, like many harmful substances, can change the physiological and biochemical responses of aquatic creatures by affecting their metabolic enzyme profiles (U.S. EPA, 1996). There may be more AST in the liver because the pesticide affects Kreb’s cycle oxidation or because it makes enzymes work harder in response to insecticide stress (Arshad et al., 2007). Therefore, the stress effect and liver damage caused by the pesticide fipronil may explain the large increases in AST and ALT levels in the liver observed in this study. Gupta et al. (2012) placed C. carpio fry in water containing 0.0428 mg/l fipronil for 45 days. This is about 1/10th of the LC50 for 96 hours. The fish exhibited increased serum cortisol levels. Another possible explanation for the high urea and creatinine levels is that fipronil has an effect on the ability of the kidneys to detox. When we looked at the histopathology, our results showed that the spleen and liver histoarchitecture changed significantly in the group exposed to fipronil. Fat degeneration, hydropic degeneration, vacuolation in the hepatocytes, disorganization of the hepatic cords, lymphocytic infiltration in the hepatic parenchyma, and severe congestion and bleeding in the portal blood vessels were some of the changes that occurred. The splenic parenchyma also presented with considerable necrosis, loss of white pulp, bleeding red pulp, and severe hemosiderosis. However, these changes in histology were lessened by the G4 that had β-1,3- glucan and fipronil. In a study on Nile tilapia, El-Murr et al. (2015) discovered that the fipronil-exposed group had focal areas of necrosis in the hepatic parenchyma, severe congestion in the hepatic blood vessels, diffuse hydropic degenerations and vacuolations in the hepatocytes, and diffuse fatty change. Current theories support these findings. A study by Elmowalid et al. (2023) also showed that in catfish (C. gariepinus), the group given fipronil and lead had hemosiderosis, red pulp hemorrhagic areas, blood vessel dilation, and splenic parenchymal congestion. There was also significant white pulp depletion and necrosis; congestion of the red pulp and lymphoid depletion were observed in the group that received a low dose of fipronil, according to a new study on O. niloticus. The group that received a high dose of fipronil had fewer lymphoid cells and more severe necrosis of lymphoid and melanomacrophage cells. This happened regardless of the dose (Fadl et al., 2022). Additionally, the effects were not very strong, but the histoarchitecture of some organs in the group that took β-1,3-glucan and a mix of fipronil and lead looked normal and not hurt. Based on these results, β-glucan appears to be a potential dietary supplement for catfish that can improve their health, cause changes in their histopathology, boost their immunity, and neutralize fipronil and lead pollutants in their culture (Elmowalid et al., 2023). ConclusionSeeing Nile Tilapia (O. niloticus) exposed to fipronil led us to believe that adding β-glucan to their food can improve their health, stop changes in their tissues, and boost their immune system. Therefore, we highly recommend incorporating β-glucan in the diets of farmed fish whenever there is sufficient evidence of its positive effects in an effort to enhance general well-being. AcknowledgmentsThe author would like to thank the Deanship of Graduate Studies and Scientific Research at Qassim University, Saudi Arabia, for financial support (QU-APC-2025). Conflict of interestNo competing interests. Data availabilityAll data generated during the current study are included in this manuscript. ReferencesAbouelghar, G.E., El-Bermawy, Z.A. and Salman, H.M.S. 2020. Oxidative stress, hematological and biochemical alterations induced by sub-acute exposure to fipronil (COACH®) in albino mice and ameliorative effect of selenium plus vitamin E. Environ. Sci. Pollut. Res. Int. 27(8), 7886–900. Al-Badran, A.A., Fujiwara, M., Gatlin, D.M. and Mora, M.A. 2018. Lethal and sub-lethal effects of the insecticide fipronil on juvenile brown shrimp Farfantepenaeus aztecus. Sci. Rep. 8(1),10769. Ansoar-Rodríguez, Y., Christofoletti, C.A., Correia, J.E., de Souza R.B., Moreira-de-Sousa, C., Marcato, A.C., Bueno, O.C., Malaspina, O., Silva-Zacarin, E.C. and Fontanetti, C.S. 2016. Liver alterations in Oreochromis niloticus (Pisces) induced by insecticide imidacloprid: histopathology and heat shock protein in situ localization. J. Environ. Sci. Health B. 51(12), 881–7. Ansoar-Rodrıguez, Y., Christofoletti, C.A., Marcato, A.C., Correia, J.E., Bueno, O.C., Malaspina, O. and Fontanetti, C.S. 2015. Genotoxic potential of the insecticide imidacloprid in a non-target organism (Oreochromis niloticus-Pisces). J. Environ. Prot. 6(12), 1360–7. AnvariFar, H., Amirkolaie, A.K., Jalali, A.M., Miandare, H.K., Sayed, A.H., Üçüncü, S.İ., Ouraji, H., Ceci, M. and Romano, N. 2018. Environmental pollution and toxic substances: cellular apoptosis as a key parameter in a sensible model like fish. Aquat. Toxicol. 204, 144–59. Araújo, C.V.M. and Blasco, J. 2019. Spatial avoidance as a response to contamination by aquatic organisms in nonforced, multicompartmented exposure systems: a complementary approach to the behavioral response. Environ. Toxicol. Chem. 38, 312–20. Ardeshir, R.A., Zolgharnein, H., Movahedinia, A., Salamat, N. and Zabihi, E. 2017. Comparison of waterborne and intraperitoneal exposure to fipronil in the Caspian white fish (Rutilus frisii) on acute toxicity and histopathology. Toxicol. Rep. 4, 348–57. Arshad, N., Shabbir, G., Aleem, S. and Arshad, M. 2007. GOT is one of the enzymes, which gives valuable diagnostic information for a number of disease conditions. Asian J. Exp. Sci. 21(2), 239–248. Barbieri, E. 2009. Effect of 2,4-D herbicide(2,4-dichlorophenoxyaceticacid) on oxygen consumption and ammonium excretion of juveniles of Geophagus brasiliensis (Quoy & Gaimard, 1824) (Osteichthyes, Cichlidae). Ecotoxicology 18(1), 55–60. Bownik, A. and Szabelak, A. 2021. Short-term effects of pesticide fipronil on behavioral and physiological endpoints of Daphnia magna. Environ. Sci. Pollut. Res. 28, 33254–64. Chiovarou, E.D., and Siewicki, T.C. 2008. Comparison of storm intensity and application timing on modeled transport and fate of six contaminants. Sci. Total. Environ. 389, 87–100. Clasen, B., Loro, V.L., Cattaneo, R., Moraes, B., Lópes, T., de Avila, L.A., Zanella, R., Reimche, G.B. and Baldisserotto, B. 2012. Effects of the commercial formulation containing fipronil on the non-target organism Cyprinuscarpio: implications for rice fish cultivation. Ecotoxicol. Environ. Saf. 77, 45–51. Correia, J.E., Christofoletti, C.A., Ansoar-Rodrıguez, Y., Guedes, T.A. and Fontanetti, C.S. 2017. Comet assay and micronucleus tests on Oreochromis niloticus (Perciforme: Cichlidae) exposed to raw sugarcane vinasse and to phisicochemical treated vinasse by pH adjustment with lime (CaO). Chemosphere 173, 494–501. Doak, D.F. and Morris, W.F. 2010. Demographic compensation and tipping points in climate-induced range shifts. Nature 467, 959–62. Elmowalid, G.A., Ghonimi, W.A.M., Abd Allah, H.M., Abdallah, H., El-Murr, A. and Abdelwahab, A.M. 2023. β-1,3-glucan improved the health and immunity of juvenile African catfish (Clarias gariepinus) and neutralized the histological changes caused by lead and fipronil pollutants. BMC Vet. Res. 19(1), 45. El-Murr, A.E., Imam, T.S., Hakim, Y. and Ghonimi, W.A.M. 2015. Histopathological, immunological, hematological and biochemical effects of fipronil on Nile Tilapia (Oreochromis niloticus). J. Veterinar. Sci. Technol. 6, 252. Fadl, S.E., Elbialy, Z.I., Abdo, W., Saad, A.H., Aboubakr, M., Abdeen, A., Elkamshishi, M.M., Salah, A.S., El-Mleeh, A., Almeer, R., Aleya, L., Abdel-Daim, M.M., Najda, A. and Abdelhiee, E.Y. 2022. Ameliorative effect of Spirulina and Saccharomyces cerevisiae against fipronil toxicity in Oreochromis niloticus. Ecotoxicol. Environ. Saf. 242, 113899. Geist, J. 2011. Integrative fresh water ecology and biodiversity conservation. Ecol. Indic. 11, 1507–16. Ghisi, D.C., Ramsdorf, W.A., Vinícius, M., Ferraro, M., Almeida, M.I.M., Ribeiro, C.A.d.O. and Cestari, M.M. 2011. Evaluation of genotoxicity in Rhamdia quelen (Pisces, Siluriformes) after sub-chronic contamination with Fipronil. Environ. Monit. Assess. 180, 589–99. Gibbons, D., Morrissey, C. and Mineau, P. A. 2015. review of the direct and indirect effects of neonicotinoids and fipronil on vertebrate wildlife. Environ. Sci. Pollut. Res. 22, 103–18. Guedes, T.A., Moreira-de-Sousa, C., Lima, H.M.S., Grella, T.C., Socolowski, P.C. and Fontanetti, C.S. 2020. Cytoprotective and anti-apoptotic action of HSP70 stress protein in Oreochromis niloticus exposed to residual dilutions of insecticides with fipronil and ethiprole. J. Environ. Sci. Health B. 55, 687–93. Gupta, S.K., Pal, A.K., Sahu, N.P., Saharan, N., Mandal, S.C., prakash, C., Akhtar, M.S. and Prusty, A.K. 2012. Dietary microbial levan ameliorates stress and augments immunity in Cyprinuscarpio fry (Linnaeus, 1758) exposed to sublethal toxicity of Fipronil. Aquac. Res. 45(5), 893–906. Gupta, S.K., Pal, A.K., Sahu, N.P., Saharan, N., Prakash, C., Akhtar, M.S. and Kumar, S. 2014. Haemato-biochemical responses in Cyprinus carpio (Linnaeus, 1758) fry exposed to sub-lethal concentration of a phenylpyrazole insecticide, fipronil. Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. 84, 113–22. Jennings, K.A., Keller, A.R.J., Atieh, B.H., Doss, R.B. and Gupta, R.C. 2002. Human exposure to fipronil from dogs treated with Frontoline. Vet. Hum. Toxicol. 44(5), 301–3. Jia, R., Li, Y., Cao, L., Du, J., Zheng, T., Qian, H., Gu, Z., Jeney, G., Xu, P. and Yin, G. 2018. Antioxidative, anti-inflammatory and hepatoprotective effects of resveratrol on oxidative stress induced liver samage in Tilapia (Oreochromis niloticus). Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 215, 56–66. Kalavathy, K., Sivakumar, A.A. and Chandran, R. 2001. Toxic effect of the pesticide Dimethoate on the fish Sarotherodon mossambicus. J. Ecol. Res. Bioconserv. 2, 27–32. Li, P. and Iii, D.M.G. 2004. Strategies of dietary supplementation for health management of hybrid striped bass morone chrysops X M. Saxatilis: a review of research at Texas A & M University. Rev. Lit. Arts Am. 95–108. Meng, S.L., Liu, T., Chen, X., Qiu, L.P., Hu, G.D., Song, C., Fan, L., Zheng, Y., Chen, J.Z. and Xu, P. 2019. Effect of chronic exposure to methomyl on tissue damage and apoptosis in testis of Tilapia (Oreochromis niloticus) and recovery pattern. Bull. Environ. Contam. Toxicol. 102, 371–6. Mulrooney, J.E., Wolfenbarger, D.A., Howard, K.D., Goli, D. and Goli, D. 1998. Efficacy of ultra low volume and high volume applications of fipronil against the boll weevil. J. Cotton Sci. 2, 110–6. Nikl, L., Albright, L. J. and Evelyn, T.P.T. 1992. No TitleImmunostimulants hold promise in furunculosis prevention. Bull Aquac. Assoc. 1, 49–52. Piner, P. and Uner, N. 2012. Oxidative and apoptotic effects of lambda- € cyhalothrin modulated by piperonyl butoxide in the liver of Oreochromis niloticus. Environ. Toxicol. Pharmacol. 33, 414–20. Schäfer, R.B., vonderohe, P.C., Kühne, R., Schüürmann, G. and Liess, M. 2011. Occurrence and toxicity Of 31 organic pollutants in large rivers of north Germany over adecade (1994 to 2004). Environ. Sci. Technol. 45, 6167–74. Schultz, A. 1987. Methods in clinical chemistry. St. Louis, MO: The C.V. Mosby Co., pp: 742–6. Soliman, N.F. and Yacout, D.M.M. 2016. Aquaculture in Egypt: status, constraints and potentials. Aquac. Int. 24, 1201–27. Srivastav, A.K., Srivastava, S.K., Mishra, D. and Srivastav, S.K. 2002. Ultimobranchial gland of freshwater catfish, Heteropneustes fossilis in response to deltamethrin treatment. Bull. Environ. Contam. Toxicol. 68, 584–91. Suvarna, S.K., Layton, C. and Bancroft, J.D. 2018. Bancroft’s theory and practice of histological techniques, 8th ed. New York, NY and London, UK: Churchill Livingstone, pp: 83–92. Tingle, C.C., Rother, J.A., Dewhurst, C.F., Lauer, S. and King, W.J. 2003. Fipronil: environmental fate, ecotoxicology, and human health concerns. Rev. Environ. Contam. Toxicol. 176, 1–66. Tomlin, C.D.S. 2006. The pesticide manual, a world compendium, 14th ed. Hampshire, UK: British Crop Protection Council, pp: 462–4. U.S. EPA. 1996. New pesticide fact sheet -fipronil; EPA 737-F-96-005. United State Environmental Protection Agency, Washington, DC: U.S. Environmental Protection Agency, Office of Prevention, Pesticides and Toxic Substances, Office of Pesticide Programs, U.S. Government Printing Office, pp: 1–10. U.S. EPA. 2002. Interim reregistration eligibility decision for chlorpyrifos. In Prevention, pesticides and toxic substances, EPA 738-R-01-007. Washington, DC. Van der Oost, R., Beyer, J. and Vermeulen, N.P. 2003. Fish bioaccumulation and biormarkers in environmental risk assessment: a review. Environ. Toxicol. Pharmacol. 13, 57–149. Wang, K., Vasylieva, N., Wan, D., Eads, D.A., Yang, J., Tretten, T., Barnych, B., Li, J., Li, Q.X., Gee, S.J., Hammock, B.D. and Xu, T. 2019. Quantitative detection of fipronil and fipronil-sulfone in sera of black-tailed prairie dogs and rats after oral exposure to fipronil by camel single-domain antibody-based immunoassays. Anal Chem. 91, 1532–40. Welker, T.L., Lim, C., Yildirim-Aksoy, M., Shelby, R. and Klesius, P.H. 2007. Immune response and resistance to stress and Edwardsiella ictaluri challenge in channel catfish, Ictalurus punctatus, fed diets containing commercial whole-cell yeast or yeast subcomponents. J. World Aquac. Soc. 38, 24–35. Zhang, B., Zhang, L., He, L., Yang, X., Shi, Y., Liao, S., Yang, S., Cheng, J. and Ren, T. 2018. Interactions of fipronil within fish and insects: experimental and molecular modeling studies. J. Agric. Food Chem. 66, 5756–61. | ||
| How to Cite this Article |
| Pubmed Style Fawiziah Khalaf Alharbi. The mitigating effect of dietary β-glucan against fipronil-induced intoxication in Nile Tilapia (Oreochromis niloticus): Histopathological, immunological, hematological, and biochemical analysis. Open Vet. J.. 2025; 15(2): 965-976. doi:10.5455/OVJ.2025.v15.i2.45 Web Style Fawiziah Khalaf Alharbi. The mitigating effect of dietary β-glucan against fipronil-induced intoxication in Nile Tilapia (Oreochromis niloticus): Histopathological, immunological, hematological, and biochemical analysis. https://www.openveterinaryjournal.com/?mno=240423 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.45 AMA (American Medical Association) Style Fawiziah Khalaf Alharbi. The mitigating effect of dietary β-glucan against fipronil-induced intoxication in Nile Tilapia (Oreochromis niloticus): Histopathological, immunological, hematological, and biochemical analysis. Open Vet. J.. 2025; 15(2): 965-976. doi:10.5455/OVJ.2025.v15.i2.45 Vancouver/ICMJE Style Fawiziah Khalaf Alharbi. The mitigating effect of dietary β-glucan against fipronil-induced intoxication in Nile Tilapia (Oreochromis niloticus): Histopathological, immunological, hematological, and biochemical analysis. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 965-976. doi:10.5455/OVJ.2025.v15.i2.45 Harvard Style Fawiziah Khalaf Alharbi (2025) The mitigating effect of dietary β-glucan against fipronil-induced intoxication in Nile Tilapia (Oreochromis niloticus): Histopathological, immunological, hematological, and biochemical analysis. Open Vet. J., 15 (2), 965-976. doi:10.5455/OVJ.2025.v15.i2.45 Turabian Style Fawiziah Khalaf Alharbi. 2025. The mitigating effect of dietary β-glucan against fipronil-induced intoxication in Nile Tilapia (Oreochromis niloticus): Histopathological, immunological, hematological, and biochemical analysis. Open Veterinary Journal, 15 (2), 965-976. doi:10.5455/OVJ.2025.v15.i2.45 Chicago Style Fawiziah Khalaf Alharbi. "The mitigating effect of dietary β-glucan against fipronil-induced intoxication in Nile Tilapia (Oreochromis niloticus): Histopathological, immunological, hematological, and biochemical analysis." Open Veterinary Journal 15 (2025), 965-976. doi:10.5455/OVJ.2025.v15.i2.45 MLA (The Modern Language Association) Style Fawiziah Khalaf Alharbi. "The mitigating effect of dietary β-glucan against fipronil-induced intoxication in Nile Tilapia (Oreochromis niloticus): Histopathological, immunological, hematological, and biochemical analysis." Open Veterinary Journal 15.2 (2025), 965-976. Print. doi:10.5455/OVJ.2025.v15.i2.45 APA (American Psychological Association) Style Fawiziah Khalaf Alharbi (2025) The mitigating effect of dietary β-glucan against fipronil-induced intoxication in Nile Tilapia (Oreochromis niloticus): Histopathological, immunological, hematological, and biochemical analysis. Open Veterinary Journal, 15 (2), 965-976. doi:10.5455/OVJ.2025.v15.i2.45 |