| Research Article | ||
Open Vet. J.. 2025; 15(2): 1024-1031 Open Veterinary Journal, (2025), Vol. 15(2): 1024-1031 Research Article Potential toxic elements in edible shrimp and other edible parts: A health risk assessmentSaad Ibrahim Al-Sultan1, Sabry Mohamed El-Bahr2,3*, Wageh Sobhy Darwish4, Ahmed M.A. Meligy5,6, Mahmoud El Sebaei2,7, Mahmoud H. A. Mohamed8,9, Ayman Megahed10 and Radwa Rasheed Elzawahry111Department of Public Health, College of Veterinary Medicine, King Faisal University, Al Hofuf, Saudi Arabia 2Department of Physiology, Biochemistry and Pharmacology, College of Veterinary Medicine, King Faisal University, Al Hofuf, Saudi Arabia 3Department of Biochemistry, Faculty of Veterinary Medicine, Alexandria University, Alexandria, Egypt 4Food Control Department, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt 5Department of Clinical Sciences, Central Diagnostic Lab., College of Veterinary Medicine, King Faisal University, Al-Hufof, Saudi Arabia 6Physiology Department, Institute (PPRI), Agricultural Research Center (ARC), Giza, Egypt 7Department of Biochemistry and Chemistry of Nutrition, Faculty of Veterinary Medicine, Mansoura University, Mansoura, Egypt 8Department of Clinical Sciences, College of Veterinary Medicine, King Faisal University, Al Hofuf, Saudi Arabia 9Department of Avian and Rabbit Diseases, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt 10Department of Veterinary Public Health, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt 11Educational Veterinary Hospital, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt *Corresponding Author: Sabry Mohamed El-Bahr. Department of Physiology, Biochemistry and Pharmacology, College of Veterinary Medicine, King Faisal University, Al Hofuf, Saudi Arabia. Email: sabryelbahr [at] hotmail.com Submitted: 31/12/2024 Accepted: 07/02/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
AbstractBackground: Pollutant pollution caused by heavy metals is considered a significant problem worldwide. Consuming food, drinking water, and breathing in air are the primary routes through which heavy metals enter the human body. Shellfish are a source of heavy metals for humans, despite being a significant source of protein derived from animals, vitamins, and trace minerals. The influence of seasons on the heavy metal burden in shellfish has received less attention than it should have. Aim: The purpose of this research was to determine the levels of harmful metal residues, including lead (Pb), cadmium (Cd), mercury (Hg), and arsenic (As), present in the edible parts of crab and shrimp intended for human consumption. A risk evaluation for human health was also computed, in addition to the predicted daily intakes that were calculated. Method: A collection of crab and shrimp samples was made near the coasts of the Red Sea in Suez city, Egypt, throughout the winter and summer seasons. Atomic absorption spectrometry (AAS) was used to detect heavy metals such as lead and cadmium, while cold vapor AAS was used to measure arsenic and mercury. Additional computations were made regarding daily intakes as well as potential hazards. Results: The findings of this research indicated that the toxic elements lead, cadmium, arsenic, and mercury were present in the edible tissues of shrimp and crab samples that were collected from Egypt. Compared with shrimp, crabs in particular displayed larger levels of these components in their bodies. When the samples collected in the summer were compared with those obtained in the winter, it was found that the heavy metal burden in the samples was much higher in the winter. Conclusion: When the potential health hazards associated with consuming such shellfish were estimated, it is possible that the Egyptian population was put in danger due to the presence of harmful metals. Therefore, it is suggested that you reduce the amount of crab and shrimp that you consume on a daily basis, particularly during winter. Keywords: Heavy metals, Crab, Shrimp, Health risk assessment, Season, Red Sea. IntroductionShrimp and crab are new and excellent providers of high biological value proteins (19.2%), water (75.3%), carbohydrates (3.6%), omega-3 polyunsaturated fatty acids, minerals (calcium and phosphorus), vitamins (D), and more. Sidwell (1981) and Hafez et al. (2022) found that crustaceans had high protein levels of alanine (8.1%), arginine (7.7%), aspartic acid (10.7%), glutamic acid (4.5%), and lysine (5.5%). Shellfish and fish consumption are rising worldwide, especially in Egypt, as red meat consumption declines. In addition, crustaceans are exposed to xenobiotics, such as heavy metals, throughout their life. Heavy metals such as mercury, lead, cadmium, and arsenic have no physiological uses. These metals have significant health risks (Thompson and Darwish, 2019). Darwish et al. (2016) reported that lead decreased children’s intelligence and led to child death. Lead and mercury were neurotoxins that harmed mental health, according to Cunningham and Saigo (1997). The US Environmental Protection Agency classifies Cd as a B1 carcinogen (IARC, 2016). Nishijo et al. (2017) and Elhelaly et al. (2022) also indicated that Cd is the reason for itai-itai disease, which causes renal failure and osteomalacia. High dosages cause organ damage and skin keratosis (Morshdy et al., 2013, 2019). Because these dangerous substances remain in the food and environment, public health and food safety regulators must monitor their levels. Lead, cadmium, mercury, and arsenate can disturb aquatic ecosystems and harm humans through the food chain (Ismahene and El Hadi, 2012; Atia et al., 2018). Recent studies have focused on how climate change and seasonal variations affect environmental heavy metal burdens. However, the seasonal effects of heavy metal deposits on Egyptian shrimp and crab are not well documented. Shellfish can absorb higher amounts of heavy metals because they are a strong indicator of heavy metal pollution in water systems. Hence, keeping an eye on the potential damage to people and calculating the amounts of heavy metals in these animals’ tissues and surroundings provide a glimpse into the present pollution situation (Guerra-Garcı´a et al., 2010). With this background, we set out to determine the amounts of lead, mercury, cadmium, and arsenate that remained in the edible portions of red sea crab and shrimp in the summer and winter. We also estimated the daily intake of these potentially harmful substances from crab and shrimp, as well as the risks to human health. Material and MethodsCollection of samplesIn the winter of 2021–2022, 30 samples of crab (Portunus pelagicus) and shrimp (Penaeus semisulcatus) were collected from Suez City (29°10′N, 32°54′E). In the summer of 2022, 60 samples were obtained from the same location. After collection, samples were promptly transported to the laboratory in labeled ice boxes. Upon arrival, the nonedible portions of the shellfish that were to be researched were removed. Then, 5 ml of an acid digestion solution was used to macerate 1 g of each sample (Darwish et al., 2015). Three milliliters of nitric acid (HNO3) at a 65% concentration (Merck, Darmstadt, Germany) and two milliliters of perchloric acid (HCLO4) at a 70% concentration (Merck, Darmstadt, Germany) were used in this solution. Overnight, at room temperature, the substance was allowed to stand in falcon tubes. The tubes were then subjected to a water bath that was heated to 70°C for 3 hours, with vigorous shaking applied every hour. After diluting the tubes with 20 ml of deionized water and allowing them to cool to room temperature, filter paper (Watt man No. 42) was used for the filtration process. The filtrate was kept at room temperature until testing for heavy metals. The standards and blanks were prepared using the same methods and chemicals used for wet digestion. Analytical proceduresA Perkin Elmer® PinAAcleTM 900T atomic absorption spectrophotometer (Shelton, CT) was used to assess As and Hg levels. For Pb and Cd, a graphite furnace was also used in conjunction with hydride generation/cold-vapor atomic absorption spectroscopy. All of the analyses were finished at the central laboratory of Zagazig University’s Faculty of Veterinary Medicine in Egypt. Quality assuranceThe reference material DORM-3 (which is a fish muscle, imported from the National Research Council, Canada) was used to ensure the accuracy and reliability of the heavy metal testing procedures. The recovery rates ranged from 80% to 110%. The metals examined had the following detection limits (µg/g ww): 0.05 for lead, 0.005 for cadmium, 0.02 for arsenate, and 0.2 for mercury. Wet weight parts per million for lead, cadmium, arsenic, and mercury were recorded. Estimated daily intake (EDI)Data on heavy metal levels in different shellfish species, fish consumption habits, and adult Egyptian body weight were used to derive the estimated daily intake (EDI). To obtain the EDI (µg/kg/day) for heavy metals, the following formula was used, which was derived from the Human Health Evaluation Manual (US Environmental Protection Agency, EPA) (US EPA, 2010): EDI=Cm × FIR / BW. Where EDI is the EDI, Cm is the measured metal concentration; FIR is the shellfish ingestion rate of 48.57 g/day (Food and Agricultural Organization, 2003); and BW is the body weight of Egyptian adults, which was estimated at 70 kg. Health risk assessmentThe non-cancer risk of heavy metal consumption by Egyptians who eat shellfish was evaluated. The US EPA (1989) recommendations were followed when conducting the risk assessment. To ensure that the EDI did not cause cancer, it was compared with the recommended reference doses (RfD) for lead, mercury, and cadmium, which are 4E-03, 1E-03, 3E-04, and 5E-04 mg/kg/d, respectively. HR=EDI / RfD, Here, HR is the hazard ratio (HR), EDI is the EDI; RfD is the suggested RfD. The risk of combined pollutants was also evaluated using a hazard index (HI). The HI was created by applying the equation below: HI=∑ HRi, Here, each metal is represented by i. While a result of 1 implies no risk of negative health impacts, an HR and/or HI value of >1 suggests a potential risk to human health.
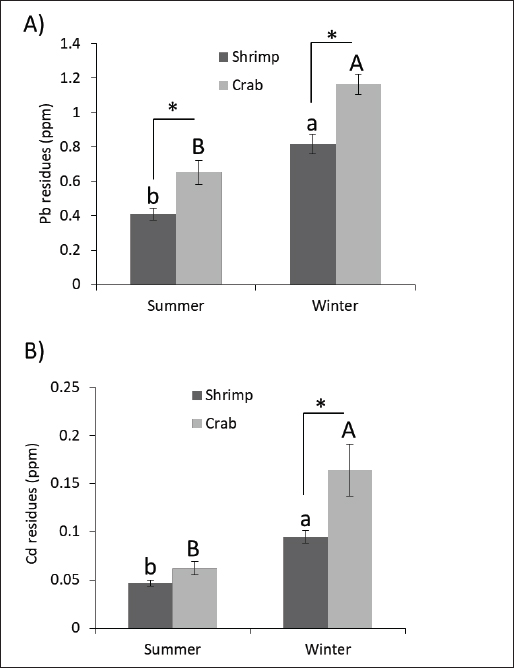
Fig. 1. Residual lead and cadmium concentrations in shrimp and crab collected during summer and winter. (A) Lead (Pb) (B) Cadmium (Cd) residual concentrations (ppm) in the examined shrimp and crab during the summer and winter seasons (n=15 for each species). Columns carrying different superscript letter is significantly different at p < 0.05. Statistical analysisTukey–Kramer Honest significant difference tests were used to ascertain whether a difference was statistically significant (p < 0.05). JMP software (SAS Institute, Cary, NC) was used for statistical analyses. ResultsThe mean ± SE residues of Pb in the shrimp and crab under examination were as follows 0.41 ± 0.03 and 0.65 ± 0.07 in the summer and 0.82 ± 0.06 and 1.16 ± 0.06 in the winter, as shown in Figure 1A. The mean residual Cd concentrations (ppm/ww) in the edible tissue of the shellfish are shown in Figure 1B. The mean Cd content in crabs was significantly higher, especially in the winter (0.16 ± 0.03), compared to shrimp (0.09 ± 0.006), crab (0.06 ± 0.007), and shrimp samples (0.05 ± 0.003) in the summer. The residual As concentrations in the edible tissue of crabs and shrimp are displayed in Figure 2A based on the obtained data. The data clearly show that As residues in crabs were significantly higher in winter (20.09 ± 0.92 ppm/ww) than in summer (14.20 ± 0.92 ppm/ww). Shrimp samples gathered in winter (16.45 ± 1.23 ppm/ww) had higher concentrations than those gathered in summer (12.22 ± 1.02 ppm/ww). In this investigation, total mercury was examined, and Figure 2B shows the results. Hg concentrations in the studied shrimp showed a mean value of 0.37 ± 0.03 ppm/ww in the summer and 0.67 ± 0.05 ppm/ww in the winter. In winter, the mean concentration of total mercury in crabs was 0.83 ± 0.03, whereas in summer, it was 0.43 ± 0.04.
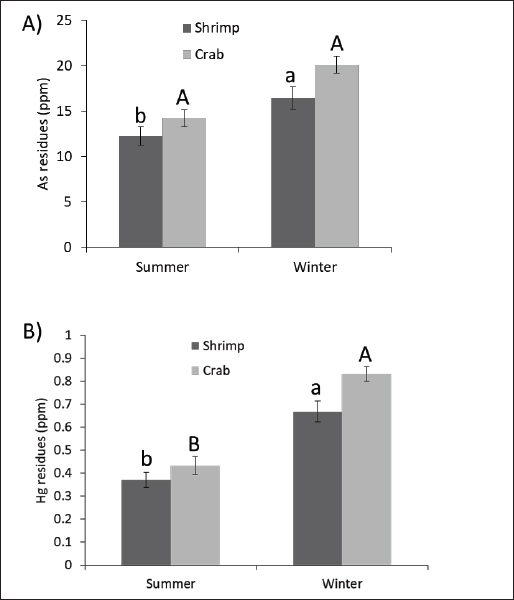
Fig. 2. Residual arsenic and mercury concentrations in shrimp and crab during the summer and winter. (A) Arsenic (As) (B) residual mercury (Hg) concentrations (ppm) in the examined shrimp and crab during the summer and winter seasons (n=15 for each species). Columns carrying different superscript letter is significantly different at p < 0.05. Dietary intake and risk assessment for the examined metals are presented in Tables 1 and 2. DiscussionShellfish are good indicators of heavy-metal pollution because they can collect large quantities. However, these animals provide vitamins, microelements, and animal-derived proteins, especially for coastal residents. Thus, food and environmental hygienists must evaluate residual metal levels, especially poisonous levels, analyze human health concerns, and explore the relationship between metal contamination and harvest season. The results show Pb levels in summer and winter shrimp and crab muscle tissue. The crab had much higher Pb concentrations than shrimp. Our edible shellfish tissue Pb levels matched those found in Andalusia (Southern Spain) fish and shellfish species by Olmedo et al. (2013) and Egyptian shellfish by Atia et al. (2018). Vázquez-Boucard et al. (2014) discovered higher Pb levels (7.2–9.9 ppm) in the edible tissue of Sinaloa and Sonora sentinel oysters. Silva da Araújo et al. (2016) recorded lower Pb levels in edible tissues of mussels, crabs, and shrimp collected from Aratu Bay, Brazil. According to Atia et al. (2018), Egyptian shellfish have similar Cd levels. The study revealed low mean Cd residues in shellfish, unlike Baudrimont et al. (2005), who observed high Cd levels in edible tissues of oysters and clams from the Nord Medoc salt marshes (Gironde estuary, France). According to Silva da Araújo et al. (2016), crabs from Aratu Bay in Brazil have lower Cd levels in their edible tissue. This study found arsenic in edible shellfish tissue, which was consistent with that found in fish and crustaceans from the Pak Pa-Nang Estuary and its catchment area in Southern Thailand, as well as in shrimp and crab specimens from Egypt by Atia et al. (2018). Kucuksezgin et al. (2014) found lower levels of arsenic in the eastern Aegean Sea marine biota from Izmir Bay. According to Krishnakumar et al. (2016), western Arabian Gulf shrimp and bivalves (clams and oysters) have high arsenic contents (11–134 and 16–118 ppm). Our clams and oysters have substantially lower Hg levels than those from the Nord Medoc salt marshes in the Gironde estuary, France (Baudrimont et al., 2005). Our investigation found mercury residues similar to those found in oysters from subtropical coastal lagoons in the southeastern Gulf of California, USA, with a mean mercury level of 0.38 ± 0.17 (Páez-Osuna and Osuna-Martínez, 2015). Atia et al. (2018) reported similar mercury levels in Egyptian shellfish. Table 1. Percentage of samples exceeding the maximum permissible limits.
Table 2. EDI, HR, and HI of Pb, C, As, and Hg due to consumption of shrimp and crab in the summer and winter seasons.
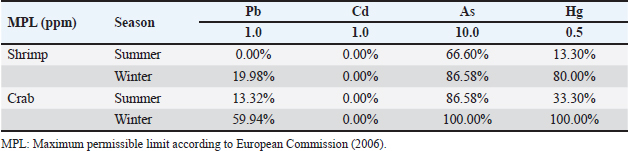
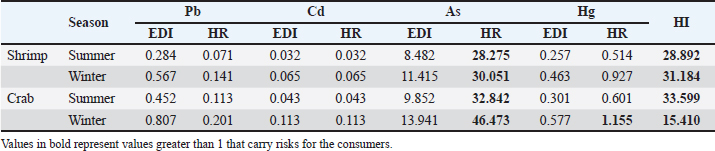
The variations in the metal concentrations analyzed in our research compared with those in other publications may indicate variations in local environmental pollution conditions. Moreover, variations among species in their xenobiotic metabolizing enzymes and capacities for detoxifying these hazardous metals may account for variations in their capacities to collect distinct metals (Ikenaka et al., 2015). The winter months had considerably greater levels of metals than the summer months. This difference in heavy metal loads could be explained by increased precipitation and environmental pollution during winter. The seasonal influence of metal loads in fish and shellfish was validated by Rajeshkumar et al. (2018), which is consistent with this premise. Examining the health risks associated with consuming shellfish and other food items before they are introduced to consumers constitutes a significant responsibility of food and environmental hygiene. To measure EDI, HI, and HR, the metal concentrations in the meat of these species were compared to the international maximum permissible limits (MPL). This allowed the assessment of the health risks associated with the presence of metals in shellfish. The percentage of tested samples that exceeded the European Commission’s (2006) maximum permissible limits (MPL)—1.0 ppm for lead, 1.00 ppm for mercury, 10.0 ppm for arsenic, and 0.5 ppm for mercury—is displayed in Table 1. The Pb content was higher than MPL in crab (13.32% and 59.94% in the summer and winter, respectively) and shrimp (0% and 19.98%, respectively) across the two seasons. Every shellfish sample analyzed fell below the Cd MPL. For residues, 86.58% and 100% of crab samples tested in the summer and winter, respectively, exceeded the MPL; similarly, in shrimp samples tested in the two seasons, 66.60% and 86.58%, respectively, exceeded the MPL. However, during the summer and winter, these percentages were 33.30% and 100% for crab and 13.30% and 80% for shrimp (Table 1). The EDI (µg/kg Bwt/day), HR, and HI of the hazardous metals under investigation as a result of consumption of crab and shrimp are reported in Table 2. The Pb EDI values in crab samples varied from 0.452 in summer to 0.807 in winter; similarly, for shrimp, they ranged from 0.287 in summer to 0.567 in winter. In terms of Cd, these values varied from 0.043 to 0.113 in crab and from 0.032 to 0.065 in shrimp. In contrast, the EDI values for As in crab and shrimp varied from 9.852 to 13.941 and 8.482 to 11.415, respectively; the values for Hg varied from 0.301 to 0.577 in crab and from 0.257 to 0.463 in shrimp, according to Table 2. It is noteworthy that the HR values of Cd and Pb did not exceed one. In contrast, the value varied from 0.514 to 1.155 for Hg and from 28.275 to 46.473 for As. In every shellfish tested in this investigation, the HI values for the four hazardous elements (Pb, Cd, As, and Hg) exceeded one, with values ranging from 15.410 to 33.599 (Table 2). According to EDI, HR, and HI, a higher dietary intake of numerous toxic metals, such as arsenic and mercury, was observed. This finding suggests potential risks and hazards; the end consumers of crustaceans and bivalve mollusks were found to be the most at risk. According to Whyte et al. (2009), Jiang et al. (2016), Raknuzzaman et al. (2016), and Atia et al. (2018), these values are equivalent to those recorded in New Zealand, China, Bangladesh, and Egypt. According to Ogwuegbu and Muhanga (2005), lead is linked to a decrease in hemoglobin production, renal failure, reproductive, and cardiovascular systems, as well as neurological symptoms. A number of poisoning cases, particularly those involving children, have been reported in several countries, such as Nigeria, China, and Zambia (Ajumobi et al., 2014; Xu et al., 2014; Yabe et al., 2014). These instances have been linked to lead in a number of places. Additionally, they found that lead induced significant cytotoxicity in human liver cells (Darwish et al., 2016). It is possible for humans to develop lung diseases such as pneumonia and bronchitis if they are exposed to high levels of cerium. Cd exposure that is not considered chronic can also have adverse effects on the kidneys and bones, according to Young (2005). There is a wide spectrum of gastrointestinal problems that can be caused by acute or subacute As exposure. These illnesses can vary from mild diarrhea and cramping in the abdomen to severe hemorrhagic gastroenteritis, followed by shock. The As was also the cause of several adverse health effects, including skin lesions, cancers, heart diseases, diabetes, respiratory disorders, neurological symptoms, and damage to development and reproduction, as stated by Feng et al. (2013). Mercury and its chemicals are exceedingly toxic to your health. The failure of the kidneys is one of the acute health effects of high exposure to inorganic mercury. There have also been cases of allergic skin reactions that occurred as a result of receiving mercury. The lungs may experience adverse effects due to mercury exposure. Researchers have also found a connection between mercury exposure and nervous symptoms such as tremors and increased excitability. Among the severe symptoms that people who are at high risk of mercury exposure present with, chest discomfort and pneumonia are among the most common (Food and Agricultural Organization/World Health Organization, 2002). ConclusionAccording to the findings of this study, lead, Cd, As, and Hg were found in the edible tissues of shrimp and crabs that were collected from Egypt. Compared to shrimp, crabs had significantly higher quantities of these components. Compared with the samples taken during the summer season, the heavy metal loads in the samples that were evaluated were much higher in the samples that were collected during the winter season. After performing a calculation to determine the potential health risks associated with the intake of such shellfish, it was discovered that the Egyptian population may be exposed to various dangers associated with hazardous metals. Therefore, it is recommended to cut back on the amount of crab and shrimp that one consumes on a daily basis, especially during winter. AcknowledgmentThis work was supported through the research group support track by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia (Grant No. 1811007). Conflicts of interestNone. Author contributionsAll authors contributed equally. Data availabilityAll data will be available upon reasonable request. ReferencesAjumobi, O.O., Tsofo, A., Yango, M., Aworh, M.K., Anagbogu, I.N., Mohammed, A., Umar-Tsafe, N., Mohammed, S., Abdullahi, M., Davis, L., Idris, S., Poggensee, G., Nguku, P., Gitta, S. and Nsubuga, P. 2014. High concentration of blood lead levels among young children in Bagega community, Zamfara—Nigeria and the potential risk factor. Pan. Afr. Med. J. 18, 1–14. Atia, A.S., Darwish, W.S. and Zaki, M.S. 2018. Monitoring of heavy metal residues, metal-metal interactions, and the effect of cooking on the metal load in shellfish. J. Anim. Plant Sci. 28, 732–743. Baudrimont, M., Schäfer, J., Marie, V., Maury-Brachet, R., Bossy, C., Boudou, A. and Blanc, G. 2005. Geochemical survey and metal bioaccumulation of three bivalve species (Crassostrea gigas, Cerastoderma edule and Ruditapes philippinarum) in the Nord Medoc salt marshes (Gironde estuary, France). Sci. Total Environ. 337, 265–280. Cunningham, W.P. and Saigo, B.W. 1997. Environmental science a global concern, 4th ed. New York, NY: WMC Brown Publisher, p: 389. Darwish, W.S., Hussein, M.A., El-Desoky, K.I., Ikenaka, Y., Nakayama, S., Mizukawa, H. and Ishizuka, M. 2015. Incidence and public health risk assessment of toxic metal residues (cadmium and lead) in Egyptian cattle and sheep meats. Int. Food Res. J. 22, 1719–1726. Darwish, W.S., Ikenaka, Y., Nakayama, S.M., Mizukawa, H. and Ishizuka, M. 2016. Constitutive effects of lead on Aryl hydrocarbon receptor gene battery and protection by β-carotene and ascorbic acid in human HepG2 Cells. J. Food Sci. 81, T275–T281. Elhelaly, A.E., Elbadry, S., Eltanani, G.S., Saad, M.F., Darwish, W.S., Tahoun, A.B. and Abdellatif, S.S. 2022. Residual contents of the toxic metals (lead and cadmium), and the trace elements (copper and zinc) in the bovine meat and dairy products: residues, dietary intakes, and their health risk assessment. Toxin Rev. 41, 968–975. European Commission. 2006. Commission regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuff. Off. J. Eur. Union. Document 32006R1881. http://data.europa.eu/eli/reg/2006/1881/oj Feng, H., Gao, Y., Zhao, L., Wei, Y., Li, Y. and Wei, W. 2013. Biomarkers of renal toxicity caused by exposure to arsenic in drinking water. Environ. Toxicol. Pharmacol. 35, 495–501. Food and Agricultural Organization. 2003. Nutrition country profiles—EGYPT. Rome, Italy: FAO. Available via http://www.fao.org/docrep/017/aq037e/aq037e.pdf Food and Agricultural Organization/World Health Organization. 2002. Human exposure to mercury in fish in mining areas in the Philippines. Marrakech, Morocco: FAO/WHO Global Forum of Food Safety Regulators Marrakech. Guerra-Garcı´a, J.M., Ruiz-Tabares, A., Baeza-Rojano, E., Cabezas, M.P., Dı´az-Pavo´n, J.J., Pacios, I., Maestre, M., Gonza´ lez, A.R., Espinosa, F. and Garcı´a Go´mez, J.C. 2010. Trace metals in Caprella (Crustacea: Amphipoda). A new tool for monitoring pollution in coastal areas? Ecol. Indicators, 10, 734–743. Hafez, A.E.S.E., Elbayomi, R.M., El Nahal, S.M., Tharwat, A.E. and Darwish, W. 2022. Potential hazards associated with the consumption of crustaceans: the Egyptian scenario. J. Adv. Vet. Res. 12, 811–814. IARC. 2016. IARC monographs on the identification of carcinogenic hazards to humans. Lyon, France: IARC. Available via https://monographs.iarc.fr/agents-classified-by-the-iarc/ Ikenaka, Y., Nakayama, S.M., Oguri, M., Saengtienchai, A., Mizukawa, H., Kobayashi, J., Darwish, W.S. and Ishizuka, M. 2015. Are red gourami (Colisa labiosa) low xenobiotic metabolizers? Elucidation of in vivo pharmacokinetics of pyrene as a model substrate. Environ. Toxicol. Pharmacol. 39, 1148–1153. Ismahene, G. and El Hadi, K.M. 2012. Assessment of heavy metal concentrations (Lead, Cadmium and Zinc) in three Crustacean species fished for in two regions of eastern Algeria. Ann. Biol. Res. 3, 2838–2842. Jiang, H., Qin, D., Chen, Z., Tang, S., Bai, S. and Mou, Z. 2016. Heavy metal levels in fish from Heilongjiang river and potential health risk assessment. Bull. Environ. Contam. Toxicol. 97, 536–542. Krishnakumar, P.K., Qurban, M.A., Stiboller, M., Nachman, K.E., Joydas, T.V., Manikandan, K.P., Mushir, S.A. and Francesconi, K.A. 2016. Arsenic and arsenic species in shellfish and finfish from the western Arabian Gulf and consumer health risk assessment. Sci. Tot. Environ. 566–567, 1235–1244. Kucuksezgin, F., Gonul, L.T. and Tasel, D. 2014. Total and inorganic arsenic levels in some marine organisms from Izmir Bay (Eastern Aegean Sea): a risk assessment. Chemosphere 112, 311–316. Morshdy, A.E., Darwish, W.S., Daoud, J.R.M. and Sebak, M.A.M. 2019. Estimation of metal residues in Oreochromis niloticus and Mugil cephalus intended for human consumption in Egypt: a health risk assessment study with some reduction trials. J. Consum. Prot. Food Saf. 14, 81–91. Morshdy, A.E., Hafez, A.E., Darwish, W.S., Hussein, M.A. and Tharwat, A.E. 2013. Heavy metal residues in canned fishes in Egypt. Jpn. J. Vet. Res. 61, S54–S57. Nishijo, M., Nambunmee, K., Suvagandha, D., Swaddiwudhipong, W., Ruangyuttikarn, W. and Nishino, Y. 2017. Gender-specific impact of cadmium exposure on bone metabolism in older people living in a cadmium-polluted area in Thailand. Int. J. Environ. Res. Public Health, 14, 401. Olmedo, P., Pla, A., Hernández, A.F., Barbier, F., Ayouni, L. and Gil, F. 2013. Determination of toxic elements (mercury, cadmium, lead, tin and arsenic) in fish and shellfishsamples. Risk assessment for the consumers. Environ. Int. 59, 63–72. Ogwuegbu, M.O.C. and Muhanga, W. 2005. Investigation of lead concentration in the blood of people in the copper belt province of Zambia. J. Environ. 1, 66–75. Páez-Osuna, F. and Osuna-Martínez, C.C. 2015. Bioavailability of cadmium, copper, mercury, lead, and zinc in subtropical coastal lagoons from the southeast Gulf of California using mangrove oysters (Crassostrea corteziensis and Crassostrea palmula). Arch. Environ. Contam. Toxicol. 68, 305–316. Rajeshkumar, S., Liu, Y., Zhang, X., Ravikumar, B., Bai, G. and Li, X. 2018. Studies on seasonal pollution of heavy metals in water, sediment, fish and oyster from the Meiliang Bay of Taihu Lake in China. Chemosphere 191, 626–638. Raknuzzaman, M., Ahmed, M.K., Islam, M.S., Habibullah-Al-Mamun, M., Tokumura, M., Sekine, M. and Masunaga, S. 2016. Trace metal contamination in commercial fish and crustaceans collected from coastal area of Bangladesh and health risk assessment. Environ. Sci. Pollut. Res. Int. 23, 17298–17310. Sidwell, V.D. 1981. Chemical and nutritional composition of finfishes, whales, crustaceans, mollusks, and their products. Silver Spring, MD: US Department of Commerce, National Oceanic and Atmospheric Administration, National Marine Fisheries Service, vol. 55. Silva da Araújo, C.F., Lopes, M.V., Vaz Ribeiro, M.R., Porcino, T.S., Vaz Ribeiro, A.S., Rodrigues, J.L., do Prado Oliveira, S.S. and Menezes-Filho, J.A. 2016. Cadmium and lead in seafood from the Aratu Bay, Brazil and the human health risk assessment. Environ. Monit. Assess. 188, 259. Thompson, L.A. and Darwish, W.S. 2019. Environmental chemical contaminants in food: review of a global problem. J. Toxicol. 2019, 2345283. US EPA. 1989. Risk assessment guidance for superfund, Vol 1. EPA/540/1-89/002. Washington, DC: Office of Emergency and Remedial Response, US EPA. US EPA. 2010. Integrated risk information system (IRIS). Cadmium (CASRN-7440-43-9). Available via http://www.epa.gov/iris/subst/0141.htm Vázquez-Boucard, C., Anguiano-Vega, G., Mercier, L. and Rojas del Castillo, E. 2014. Pesticide residues, heavy metals, and DNA damage in sentinel oysters Crassostrea gigas from Sinaloa and Sonora, Mexico. J. Toxicol. Environ. Health A. 77, 169–176. Whyte, A.L., Raumati Hook, G., Gail, E., Greening, G.E., Gibbs-Smith, E. and Gardner, J.P. 2009. Human dietary exposure to heavy metals via the consumption of greenshell mussels ( Perna canaliculus Gmelin 1791) from the Bay of Islands, northern New Zealand. Sci. Tot. Environ. 407, 4348–4355. Xu, J., Sheng, L., Yan, Z. and Hong, L. 2014. Blood lead and cadmium levels of children: a case study in Changchun, Jilin Province, China. West Ind. Med. J. 63, 29–33. Yabe, J., Nakayama, S.M., Ikenaka, Y., Yohannes, Y.B., Bortey-Sam, N., Oroszlany, B., Muzandu, K., Choongo, K., Kabalo, A.N., Ntapisha, J., Mweene, A., Umemura, T. and Ishizuka, M. 2014. Lead poisoning in children from townships in the vicinity of a lead-zinc mine in Kabwe, Zambia. Chemosphere 119C, 941–947. Young, R.A. 2005. Toxicity profiles: toxicity summary for cadmium, risk assessment information system. Knoxville, TN: University of Tennessee Available via rais.ornl.Gov/tox/profiles/cadmium.html | ||
| How to Cite this Article |
| Pubmed Style Al-sultan SI, El-bahr SM, Darwish WS, Meligy AM, Sebaei ME, Mohamed MHA, Megahed A, Elzawahry RR. Potential toxic elements in edible shrimp and other edible parts: A health risk assessment. Open Vet. J.. 2025; 15(2): 1024-1031. doi:10.5455/OVJ.2025.v15.i2.50 Web Style Al-sultan SI, El-bahr SM, Darwish WS, Meligy AM, Sebaei ME, Mohamed MHA, Megahed A, Elzawahry RR. Potential toxic elements in edible shrimp and other edible parts: A health risk assessment. https://www.openveterinaryjournal.com/?mno=240734 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.50 AMA (American Medical Association) Style Al-sultan SI, El-bahr SM, Darwish WS, Meligy AM, Sebaei ME, Mohamed MHA, Megahed A, Elzawahry RR. Potential toxic elements in edible shrimp and other edible parts: A health risk assessment. Open Vet. J.. 2025; 15(2): 1024-1031. doi:10.5455/OVJ.2025.v15.i2.50 Vancouver/ICMJE Style Al-sultan SI, El-bahr SM, Darwish WS, Meligy AM, Sebaei ME, Mohamed MHA, Megahed A, Elzawahry RR. Potential toxic elements in edible shrimp and other edible parts: A health risk assessment. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 1024-1031. doi:10.5455/OVJ.2025.v15.i2.50 Harvard Style Al-sultan, S. I., El-bahr, . S. M., Darwish, . W. S., Meligy, . A. M., Sebaei, . M. E., Mohamed, . M. H. A., Megahed, . A. & Elzawahry, . R. R. (2025) Potential toxic elements in edible shrimp and other edible parts: A health risk assessment. Open Vet. J., 15 (2), 1024-1031. doi:10.5455/OVJ.2025.v15.i2.50 Turabian Style Al-sultan, Saad Ibrahim, Sabry Mohamed El-bahr, Wageh Sobhy Darwish, Ahmed M.a. Meligy, Mahmoud El Sebaei, Mahmoud H. A. Mohamed, Ayman Megahed, and Radwa Rasheed Elzawahry. 2025. Potential toxic elements in edible shrimp and other edible parts: A health risk assessment. Open Veterinary Journal, 15 (2), 1024-1031. doi:10.5455/OVJ.2025.v15.i2.50 Chicago Style Al-sultan, Saad Ibrahim, Sabry Mohamed El-bahr, Wageh Sobhy Darwish, Ahmed M.a. Meligy, Mahmoud El Sebaei, Mahmoud H. A. Mohamed, Ayman Megahed, and Radwa Rasheed Elzawahry. "Potential toxic elements in edible shrimp and other edible parts: A health risk assessment." Open Veterinary Journal 15 (2025), 1024-1031. doi:10.5455/OVJ.2025.v15.i2.50 MLA (The Modern Language Association) Style Al-sultan, Saad Ibrahim, Sabry Mohamed El-bahr, Wageh Sobhy Darwish, Ahmed M.a. Meligy, Mahmoud El Sebaei, Mahmoud H. A. Mohamed, Ayman Megahed, and Radwa Rasheed Elzawahry. "Potential toxic elements in edible shrimp and other edible parts: A health risk assessment." Open Veterinary Journal 15.2 (2025), 1024-1031. Print. doi:10.5455/OVJ.2025.v15.i2.50 APA (American Psychological Association) Style Al-sultan, S. I., El-bahr, . S. M., Darwish, . W. S., Meligy, . A. M., Sebaei, . M. E., Mohamed, . M. H. A., Megahed, . A. & Elzawahry, . R. R. (2025) Potential toxic elements in edible shrimp and other edible parts: A health risk assessment. Open Veterinary Journal, 15 (2), 1024-1031. doi:10.5455/OVJ.2025.v15.i2.50 |