| Research Article | ||
Open Vet. J.. 2026; 16(1): 372-383 Open Veterinary Journal, (2026), Vol. 16(1): 372-383 Research Article First characterization of somatic proteins of trematodes of the family Paramphistomidae by SDS-PAGE isolated from cattle from the Cajamarca region, PeruCharito Jennyfer Fernandez-Mendoza1, Antony Tayca-Saldaña1, Medali Cueva-Rodríguez1*, Fernando Aliaga-Tambo1, Claudia Rodríguez-Ulloa2, Wuesley Alvarez-García3, Carlos Quilcate-Pairazamán4 and Marco Cabrera-González11Laboratorio de Biotecnología en Sanidad Animal, Programa Nacional de Bovinos, Estación Experimental Baños del Inca, Dirección de Desarrollo Tecnológico Agrario, Instituto Nacional de Innovación Agraria (INIA), Cajamarca, Perú 2Escuela de Biología y Biotecnología, Facultad de Ciencias de la Salud, Universidad Nacional de Cajamarca, Cajamarca, Perú 3Estación Experimental de los Baño del Inca, Dirección de Desarrollo Tecnológico Agrario, Instituto Nacional de Innovación Agrario, Cajamarca, Perú 4Dirección de Desarrollo Tecnológico Agrario, Instituto Nacional de Innovación Agrario, La molina, Lima, Perú *Corresponding Author: Medali Cueva-Rodríguez. Laboratorio de Biotecnología en Sanidad Animal, Programa Nacional de Bovinos, Estación Experimental Baños del Inca, Dirección de Desarrollo Tecnológico Agrario, Instituto Nacional de Innovación Agraria (INIA), Cajamarca, Perú. Email: mcuevar [at] unc.edu.pe Submitted: 09/02/2025 Revised: 25/11/2025 Accepted: 20/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: Paramphistomosis is a gastrointestinal parasitic disease of worldwide distribution, with higher prevalence in tropical and subtropical regions. Its biological cycle has a chronic phase caused by adult trematodes that adhere to the rumen mucosa and a highly pathogenic phase caused by immature trematodes, which induce acute parasitic gastroenteritis. Studies related to the parasite are very scarce in the region of Cajamarca, Peru, regarding the characterisation of antigenic proteins, considering that this parasitosis is endemic and has emerging characteristics. Aim: The study aimed to characterise somatic proteins of adult forms of Paramphistomidae in cattle by electrophoresis, Sodium dodecyl sulphate-polyacrylamide gel electrophoresis. Methods: For this purpose, 630 adult parasites were collected from the rumen and reticulum of cattle from the processing centre of Cajamarca, Peru. Results: The number of protein bands in the paramphistomid somatic crude extract was variable depending on the concentration of the protein analyzed. Thus, concentration 2 showed the highest number of protein bands (21 bands) with molecular weights (MW) within a variable range of 15 kDa–119 kDa. The results of ITS-2 gene sequencing, after alignment in NCBI’s Basic Local Alignment Search Tool of the obtained consensus sequences, show 100% similarity to sequences belonging to the species Calicophoron microbothrioides. Conclusion: Twenty-one protein bands from the somatic extract were characterized. The MWs of these bands ranged from 15 kDa to 119 kDa. This study may help carry out alternative control programmes, such as developing vaccines, choice of appropriate drugs due to the therapeutic failures expressed by this parasite. In addition, molecular identification based on ITS-2 gene sequencing revealed a 100% sequence similarity with Calicophoron microbothrioides. Keywords: Cattle, Calicophoron microbothrioides, Somatic extract, SDS-PAGE, Protein. IntroductionParamphistomosis is an emerging gastrointestinal parasitic disease caused by trematodes of the family Paramphistomidae Fischoeder, 1901 (Eduardo, 1983; Huson, 2017). These parasites require freshwater pulmonate molluscs of the families Planorbidae, Bulinidae, and Lymnaeidae as intermediate hosts (Cordero et al., 1999) and exhibit a broad spectrum of definitive hosts from domestic ruminants such as cattle, goats, and sheep to wild ruminants (Horak, 1971). They have even been documented in camelids (El-Khabaz et al., 2019) and in humans by Gastrodiscoides hominis Lewis and McConnall, 1876 (Dutt, 1972; Kumar,1980; Sah, 2019). Adult parasites reside in the rumen and reticulum of ruminants; however, in other species, they may be located in the large intestine or parenteral sites of ruminants, pigs, and horses (Ghatani et al., 2024). This disease has a global distribution, with higher prevalence in tropical and subtropical regions (Taylor et al., 2007), although it also occurs in temperate zones (Nikander and Saari, 2007). The most affected areas include Africa, Eastern Europe, Asia, Australia, and Russia (Rolfe, 1991; Arias, 2011). In particular, high prevalences of 77% have been reported in Ireland (Martinez-Ibeas et al., 2016), 75.2% in Sarawak-Malaysia (Harizt et al., 2021), 59% in Wales (Jones et al., 2017), 56.25% in Pakistan (Rizwan et al., 2022), and 53.8% in Northern Ireland (Atcheson et al., 2022). Also, in some regions of the UK, the prevalence of paramphistomosis is higher than that of fasciolosis (Huson et al., 2017). It has also been reported in several Latin American countries, such as Mexico (Hernández-Hernández et al., 2023), Brazil (Lopes et al., 2024), Colombia (Arroyo et al., 2022), Chile (Cerda et al., 2019), Peru (Cueva-Rodríguez et al., 2024), among others. The highly pathogenic phase of the disease is caused by migrating immature trematodes, which induce acute parasitic gastroenteritis and can cause mortality (Tehrani, 2015; O’Shaughnessy et al., 2018). The chronic phase, caused by adult stags attached to the rumen mucosa, reticulum, or omasum (Torrel et al., 2015) is less pathogenic but can cause necrosis of rumen papillae, mechanical damage to the rumen wall, ruminal atony, decreased production, etc. (Alzieu, 2007; Fuertes, 2015; Rizwan, 2022). It is also essential to economic losses, affecting cattle’s dairy and meat productivity (Torrel, 2015; Ali et al., 2022). Diagnosis of this parasitosis remains challenging; it relies on ineffective coproparasitological examinations in situations of low parasite load or during the prepatent period of infection. Early detection in the subclinical phase is, therefore, essential for timely treatment (Wang et al., 2006). There is currently no effective vaccine against paramphistomosis, which exacerbates the problem. The Cajamarca region, located in the northern highlands of Peru, stands out as the country’s leading dairy basin, contributing 17% of national fresh milk production (INEI, 2012; Torrel, 2015). However, the region faces the challenge of being an endemic area for this little-studied disease, with a reported prevalence of paramphistomosis at 34.2%, exceeding that of Fasciola hepatica Linnaeus, 1758, which is recorded at 15.3% in the district of Cajamarca (Torrel et al., 2023). The spread of this parasite is influenced by several factors, including temperature, humidity, rainfall, and abundant vegetation (Torrel, 2015; Rafiq, 2022). In addition, treatments are ineffective due to inappropriate administration of fasciolicides by farmers because of the similarity of symptomatology between paramphistomosis and fascioliasis (Howell et al., 2020). Proteomics plays a crucial role in analysing proteins expressed by parasites. Information derived from proteomic approaches to parasite-host interaction, such as identifying antigenic proteins, is crucial, as these proteins can act as biomarkers in diagnosis, drug targets, and vaccine candidates (Van der Ree and Mutapi, 2015). Due to their immunogenicity, somatic proteins are particularly relevant in helminths (Sripa et al., 2017). For example, fatty acid binding proteins (FABPs) in Fasciola gigantica Cobbold, 1855, have been shown to induce damage to the parasite tegument through FABPs-based vaccines, which in turn triggers an effective immune response in the host (Sirisriro et al., 2002). In the somatic extract of Gigantocotyle explanatum Creplin, 1847, glutathione S-transferase (GST) is found in high concentrations and is an effective diagnostic antigen (Rehman et al., 2020). Several somatic proteins have been identified in paramphistomids, such as Calicophoron daubneyi. Among them, FABPs are prominent in the somatic proteome, along with dehydrogenases/reductases, globins, and GST (Huson et al., 2018). However, knowledge of the somatic protein profile of parasites of the family Paramphistomidae remains limited, particularly in Calicophoron microbothrioides Price and McIntosh, 1944, which is the species responsible for paramphistomosis in Cajamarca (Torrel et al., 2015). In this context, the present study aimed to characterise the somatic proteins of adult forms of parasites of the Paramphistomidae family in cattle by Sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) electrophoresis. This will provide a baseline to optimise the diagnostic and control capacity of the disease and contribute to the generation of vaccines. Materials and MethodsLocation of the studyThe study was conducted at the Cajamarca cattle processing centre located in the Iscoconga village centre (7° 12’ 04.6" S, 78° 26’ 57.4" W), Llacanora district, Cajamarca region, Peru (Fig. 1). A total of 630 adult parasites were collected from the rumen and reticulum of cattle.
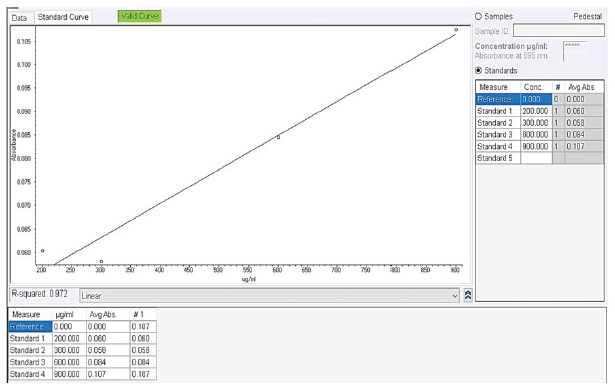
Fig. 1. Geographical location of the Cajamarca livestock processing centre. Iscoconga village centre, Cajamarca, Peru. Using ArcGIS (free version 10.8, Harvard, USA). Collection of parasitesAccording to the methodology described by (Abdolahi et al., 2016), parasites were washed five times with phosphate-buffered saline (PBS, pH 7.2) (Thermo Scientific™, USA) and transferred to 50 ml tubes (Falcon®, USA) containing PBS. Subsequently, they were transported at 37°C to the Laboratory of Biotechnology in Animal Health of the Baños del Inca Agrarian Experimental Station of the National Institute of Agrarian Innovation (INIA). Somatic protein extractionSomatic protein extraction was performed according to the protocol determined by Cabrera et al. (2020). 10, 20, 30, and 50 parasites were distributed in individual 2 ml microtubes (Eppendorf®, Germany) and preserved in an ice bucket. The microtubes were immersed in liquid nitrogen at −195°C for approximately 20 seconds, repeating this process three times. Subsequently, the microtubes were fragmented in a mortar, and the contents were crushed to a paste-like consistency. The paste was collected in new 2 ml microtubes (Eppendorf®, Germany), 1.5 ml PBS was added, and homogenised in a vortex (VWR®, Vortex Mini 230V, USA). Centrifuged at 14,000 g, 4°C, for 10 minutes (Eppendorf®, 5430 R, Germany), the supernatant was discarded, and the pellet was allowed to dry at room temperature. The pellet was then suspended in BugBuster Protein Extraction Reagent® (Novagen®, USA) according to the manufacturer’s specifications and incubated on a slow-shaking platform (Labnet International Inc, USA) for 20 minutes at room temperature. One mM phenylmethylsulfonyl fluoride (Sigma®, USA) was added as a protease inhibitor and incubated for 20 minutes at room temperature. Insoluble cell debris was removed by centrifugation at 16,000 g, 4°C, for 20 minutes. The supernatant was placed into a new 2 ml microtube (Eppendorf®, Germany). The protein was stored at −20°C for further analysis. Protein quantificationThe protein concentration of the somatic extracts was determined using the modified Bradford method (Bradford, 1976) with a NanoDrop™ 2000 spectrophotometer (Thermo Scientific, USA). The assay was performed using the Bio-Rad concentrated dye reagent (USA), according to the manufacturer’s instructions, and bovine serum albumin (BSA; Spinreact®, Spain) as the standard protein. The dye reagent was diluted at a 1:4 ratio, and 3–5 BSA dilutions within the linear range of the method (200, 300, 600, and 900 µg/ml) were prepared. Each dilution was mixed with the reagent and incubated for 5 minutes before measuring absorbance at 595 nm to construct the calibration curve (Fig. 2). The test samples were processed under the same conditions, and their absorbance was measured using the NanoDrop™ 2,000 to determine protein concentration.
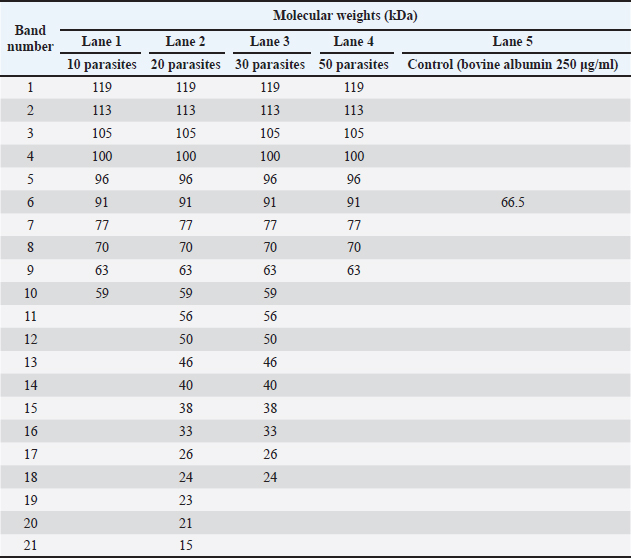
Fig. 2. Calibration curve for the spectrophotometric determination of protein concentration in crude somatic extracts of adult Paramphistomidae parasites using the Bradford method. Sodium dodecyl sulphate-polyacrylamide gel electrophoresisProtein extracts were analysed by SDS-PAGE as described by Cabrera et al. (2020) and Alonso et al. (2020) with slight modifications. A 12% resolving gel (30% acrylamide solution, acrylamide 29.2 g, bisacrylamide 0.8 g; 1.5M TRIS buffer, pH 8.8, 5.6 ml; distilled water 3.1 ml; 10% SDS 150 µl; 10% ammonium persulfate 100 µl; TEMED 40 µl) and a 5% (30% acrylamide solution 1.7 ml; 0.5M TRIS buffer, pH 6.8, 1.25 ml; distilled water 7.03 ml.; 10% SDS 100 µl.; 10% ammonium persulfate 150 µl.; TEMED 30 µl) concentration gel was prepared. Proteins were denatured at 95°C for 5 minutes in a water bath (Techne, Tempette TE-10D, UK), then prepared in a 1:1 ratio with sample buffer. Subsequently, A running buffer of pH 8.8 was used (TRIS Base 3.03 g, glycine 14.42 g, SDS 1 g, distilled water 1,000 ml), 10 μl of the broad-spectrum protein marker Spectra™ Multicolor (Thermo Scientific, USA), and 29 μl of each sample were loaded into the corresponding well. Additionally, 29 μl of bovine albumin at a concentration of 250 µg/ml, with a molecular weight (MW) of ~ 66.5 kDa, was used as a control (Spinreact®, Spain). The electrophoretic run was carried out using the vertical gel electrophoresis system (Labnet International Inc, ENDURO™ VE10, USA) at a constant voltage of 50 V for 30 minutes for the concentration gel and at 120 V for 2 hours and 50 minutes for the resolution gel. Silver stainingGel staining was performed with the commercial Silver Stain Plus™ kit (Bio-Rad, USA), following the manufacturer’s specifications. Molecular weight determinationThe somatic protein bands obtained on the gel were analysed to establish their MW according to the methodology described by (Matsumoto et al., 2019). Then, the relative mobility value (Rf) of the standard marker bands and each problem band was calculated in relation to the migration distance of the dye front: Rf= protein migration distance (cm) ÷ gel front migration distance (cm) Subsequently, the MW of the standard protein marker were plotted on a base ten logarithmic scale as a function of relative mobility (log10 MW versus Rf), and the regression equation was generated as follows: y= mx + b. Finally, the Rf -value of the protein of interest was interpolated in the obtained equation to determine its approximate MW. SequencingTwo adult parasites were randomly selected for molecular identification. polymerase chain reaction products were sent to the Genomics Laboratory of the Directorate of Genetic Resources and Biotechnology of the National Institute of Agricultural Innovation-INIA (Lima, Peru), and bidirectional sequencing was performed using the Sanger method (3,500 Series Genetic Analyzer Biosystem Applied). Statistical analysisStatistical analysis was performed using Graph Pad Prism 9.3.1 software (Prism Software, Irvine, CA, USA). The Kolmogorov–Smirnoff test demonstrated the data’s normality. Analysis of variance tests followed by a t-test were used to compare groups. To compare MW with the band frequency of parasites. Statistical differences were considered significant when the p-value was less than 0.05. Ethical approvalThe research was not submitted to an ethics committee for animal experimentation because the human-animal interaction was only limited to the extraction of samples of adult parasites from the rumen and reticulum of already benefited animals after inspection by well-trained veterinarians from the Faculty of Veterinary Sciences of the National University of Cajamarca, Peru. ResultsTaking into account the little information on the characterization of the parasite in the area, it was necessary to include some morphometric identification parameters reported by Torrel and Paz (2015), for its identification in slaughtered cattle, such as its location in the rumen, the pink or bright red colour, the conical shape with concave ventral face and convex dorsal face, the approximate size of 4–15 mm long by 2–5 mm wide, and the presence of an oral sucker and a sucker in the back surrounded by a bulge. The presence of an oral sucker and a sucker in the posterior part surrounded by a protuberance, having isolated 630 parasites isolated in an aseptic way. Morphological identification of the 630 parasites collected aseptically and preserved in PBS (pH 7.2, 37°C) was mainly located in the rumen of slaughtered cattle, which were pink or bright red in colour, conical in shape with a concave ventral face and a convex dorsal face, and were approximately 4–15 mm long by 2–5 mm wide; they also had an oral sucker and a sucker on the back surrounded by a protuberance (Cabrera et al., 2020). Four concentrations of somatic extract (1: 500 µg/ml, n=10; 2: 726.5 µg/ml, n=20; 3: 1,979.5 µg/ml, n=30 and 4: 2,000 µg/ml, n=50) were evaluated by liquid nitrogen (−195°C) for better expression and separation of protein bands by SDS-PAGE electrophoresis, showing that concentration 2 showed 21 protein bands compared to the other protein concentrations. The somatic crude extract was obtained using liquid nitrogen (−195°C), having achieved four working concentrations with a different number of parasites, which allowed the identify the appropriate concentration for better expression and separation of protein bands by SDS-PAGE technique; these samples were analysed through the colorimetric technique for protein quantification (595 nm), so we had in concentration 1: 500 µg/ml (n=10), concentration 2: 726.5 µg/ml (n=20), concentration 3: 1,979.5 µg/ml (n=30) and concentration 4: 2,000 µg/ml (n=50). In this context, by means of the SDS-PAGE method with silver staining, it was observed that the number of protein bands of the paramphistomid somatic crude extract varied according to the concentration of the protein analysed, Thus, concentration 2 showed the highest number of protein bands (21 bands) with MW within a variable range of 15–119 kDa, in relation to the other concentrations analysed. Thus, concentration 1 showed 10 bands (59–119 kDa), concentration 3 showed 18 bands (24–119 kDa), and concentration 4 showed 9 bands (63–119 kDa) (Table 1). Table 1. Protein molecular masses of paramphistomids somatic extract, according to protein concentrations and number of parasites.
Using the SDS-PAGE method with silver staining, it was observed that the number of protein bands of the paramphistomids somatic crude extract was variable according to the concentration of the protein analysed. Thus, concentration 2 showed the highest number of protein bands (21 bands) with MW within a variable range of 15 kDa to 119 kDa; about the other concentrations analysed, in concentration 1, 10 bands were separated (59 kDa–119 kDa), concentration 3, 18 bands were separated (24 kDa–119 kDa). In concentration 4, 9 bands were separated (63 kDa–119 kDa) (Table 1). When analysing the protein bands on the polyacrylamide gel, a total of 9 standard bands were observed that appeared at the four concentrations used, with a variable range between 63 kDa and 119 kDa (63, 70, 77, 91, 96, 100, 105, 113, and 119 kDa) (Fig. 3).
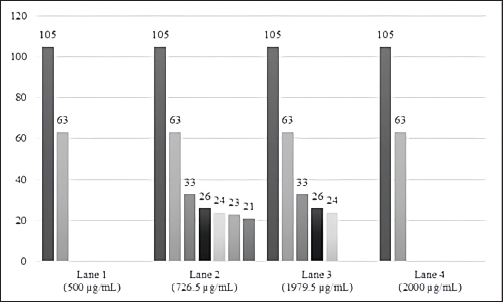
Fig. 3. Silver-stained somatic protein profile of different numbers of adult paramphistomids analysed by SDS-PAGE and their corresponding protein concentration. MW presents the molecular weight standards; lane 1 (500 µg/ml, n=10); lane 2 (726.5 µg/ml, n=20); lane 3 (1,979.5 µg/ml, n=30); lane 4 (2,000 µg/ml, n=50) and lane 5 (bovine albumin 250 μg/ml). Full-length gels are presented in Supplementary Figures: 1 (a); 2 (b, e); 2 (c, f) and 2 (d). Band intensity reflects the relative abundance or concentration of the protein present at a specific molecular weight. Regarding the intensity of the protein bands in the polyacrylamide gel of the different concentrations of paramphistomid somatic crude extract used, 7 protein bands were observed with MWs ranging from 21 kDa–105 kDa (105, 63, 33, 26, 24, 23, and 21 kDa). In addition, 2 protein bands were shown at the four concentrations used with MWs of 63 kDa and 105 kDa respectively; while 3 bands were expressed at concentration 2 (726.5 µg/ml) and 3 (1,979.5 µg/ml), with a MW of 33 kDa, 26 kDa and 24 kDa, thus only the bands with low MW of 23 kDa and 21 kDa resulted from concentration 2 (726.5 µg/m,) (Fig. 4).
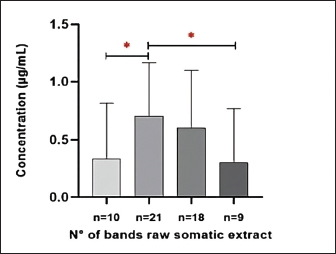
Fig. 4. Intensity of the protein bands of paramphistomids somatic crude extract, stained with silver, according to the different protein concentrations and number of parasites analysed. Lane 1 (63, 105 kDa); lane 2 (21, 23, 24, 26, 33, 63, 105 kDa); lane 3 (24, 26, 33, 63, 105 kDa); lane 4 (63, 105 kDa). A significant difference was also found in the concentration of the somatic crude extract protein (µg/ml) and the number of separated bands (kDa) of paramphistomids (p < 0.05), as shown in Figure 5.
Fig. 5. Statistical analysis of paramphistomids somatic crude extract protein band. The asterisk (*) indicates a significant difference (p < 0.05). The results of ITS-2 gene sequencing, after alignment in NCBI’s Basic Local Alignment Search Tool (BLAST) (https://blast.ncbi.nlm.nih.gov/Blast.cgi) of the obtained consensus sequences, show 100% similarity to sequences belonging to the species C. microbothrioides (Fig. 6). In addition to 100% coverage and a low e-value (2e–138), confirming its membership in the family Paramphistomidae and within the subclade formed by isolates belonging to the genus Calicophoron spp. The ITS-2 gene is a highly conserved molecular marker useful for identifying some families and even genera of nematodes; however, it is less sensitive in identifying closely related species. Therefore, highly similar sequences belonging to other species, such as Paramphistomum cervi, may appear in the alignment analysis. For this reason, multilocus analyses are recommended for greater specificity.
Fig. 6. Molecular phylogenetic analysis of Calicophoron microbothrioides by Sanger sequencing. DiscussionIn Cajamarca, the region with the highest fresh milk production in Peru (INEI, 2012), paramphistomiasis has received very little attention in terms of research and control, despite its high prevalence due to the presence of favorable environmental factors (temperature and humidity) (Torrel-Pajares et al., 2022). While previous studies exist on parasitic incidence and the correlation between parasite load and the presence of adult forms, the molecular characterization of these trematodes remains limited, hindering the development of more sensitive diagnostic techniques (Coronado et al., 2023). In this context, the molecular identification performed provides important evidence supporting the taxonomic classification of the analyzed paraphistomids. Sequencing of the ITS-2 region followed by BLAST alignment revealed 100% similarity to C. microbothrioides. This is consistent with previous studies reporting that ITS-2 is a suitable conserved molecular marker for family- and genus-level discrimination in trematodes. However, given its limited resolution in closely related species, this explains the occurrence of highly similar sequences from taxa such as Paramphistomum cervi in alignment analyses. Therefore, while ITS-2 sequencing is useful for molecular confirmation, multilocus approaches that improve species-level resolution should be considered. This could strengthen the interpretation of subsequent proteomic analyses. Currently, the diagnosis of paramphistomiasis is based primarily on fecal examination, a technique with low sensitivity, especially in the acute phase of infection. In contrast, the application of proteomic approaches to other trematodes such as Fasciola hepatica has allowed the identification of candidate antigens for immunology and vaccination (Johnston et al., 2009). Therefore, understanding the antigenic profile of paramphistomids is fundamental, as this is a necessary step for the future development of more precise diagnostic strategies and for laying the groundwork for vaccine development. The protein profile of paramphistomids, molecularly characterized as C. microbothrioides, obtained by SDS-PAGE in this study, showed 21 protein bands between 15 and 119 kDa at an optimal concentration of 726.5 µg/ml. This is comparable to that reported by Anuracpreeda et al., (2016), who observed 26 bands with MWs between 12 and 170 kDa in Paramphistomum gracile. Likewise, somatic proteins with MWs between 10 and 150 kDa have been reported in Paramphistomum cervi (Abdullah et al., 2019). In the case of Paramphistomum epiclitum, 15 bands have been reported with weights ranging from 8 to 169.3 kDa (Jyoti et al., 2014). These differences in band size and number are related to the variability of species analyzed in each study and are probably due to genomic heterogeneity (Mattison et al., 1994). In addition, the differences could be related to interspecific variations, differences in the extracts used, the number of parasites processed, or gel conditions. In comparison with other trematodes, Dezhabad et al. (2023) reported 12 protein bands with variable MWs ranging from 26 to 263 kDa for Fasciola hepatica. Similarly, Abdolahi et al. (2016) conducted a study with Fasciola hepatica and F. gigantica, revealing 30 protein bands ranging from 18 kDa to 180 kDa, with band intensities between 22.5 kDa, 27 kDa, 31 kDa, and 62 kDa MW. This differs from the findings in this study, possibly due to the difference in trematode species. The literature reports that the interaction between a parasite and its host is crucial in the parasite’s protein expression (De Vera et al., 2009). Therefore, variations in the parasite’s protein electrophoretic profile may exist depending on the definitive host due to the impact of its immune response. Thus, host-type variability has been reported to induce changes in the protein electrophoretic profile of Schistosoma japonicum (Hong et al., 2011). Similarly, De Vera et al. (2009) found the existence of exclusive bands in the protein electrophoretic profile of Fasciola hepatica and F. gigantica for Bubalus bubalis, unlike Bos taurus. Similarly, Paramphistomum epiclitum has been observed, for which 16 protein bands between 14 and 76 kDa have been reported in parasites isolated from goats and sheep (Jadav et al., 2018), and for buffalo, 23 bands between 14 and 115.5 kDa (Hussain, 2013). Protein characterization of the crude somatic extract of adult paramphistomids was performed using polyacrylamide gel electrophoresis. A significantly higher number of protein bands (n=21) were observed at a concentration of 726.5 µg/ml compared to the other concentrations tested (500 µg/ml, 1,979.5 µg/ml, and 2,000 µg/ml). This difference is possibly related to the saturation of the polyacrylamide gel pore diameter and protein migration. Finally, it is important to note that further research is needed to identify the antigenicity, immunogenicity, and immunodominance of the proteins identified in this study. Some of the identified bands may be related to proteins that have been previously reported. Thus, the purified 16 kDa immunogenic protein from Paramphistomum gracile is a protein with high specificity that does not exhibit cross-reactivity with other parasites Anuracpreeda et al., (2016) ConclusionThe results of the present study revealed a profile of 21 protein bands from the somatic extract of adult forms of parasites of the family Paramphistomidae in cattle by SDS-PAGE electrophoresis technique, this finding suggests that at a protein concentration of 726. 5 µg/ml a greater number of protein bands can be exhibited which presented a MW within a range of 15–119 kDa (15, 21, 23, 24, 26, 33, 33, 38, 40, 46, 50, 56, 56, 59, 63, 70, 77, 91, 96, 100, 105, 113 and 119 KDa), it is necessary to mention that these proteins obtained from local parasites of Paramphistomidae should be characterized in more detail to determine their probable antigenic potential as candidates for local vaccines, due to the phenomenon of anthelmintic resistance that the parasite expresses to the drugs used in its control by the farmers of the Region; Furthermore, they can be used for diagnostic tests based on antigens of native E/S products, taking into consideration the little information available in the Region regarding the parasite in terms of its prevention and control. In addition, molecular identification based on ITS-2 gene sequencing revealed a 100% sequence similarity with C. microbothrioides. AcknowledgmentsThis research was funded by “Proyecto de Mejoramiento Genético Nacional-PROMEG CUI 2432072" and to Subdirección de Biotecnología del Instituto Nacional de Innovación Agraria-INIA-Lima, Peru. Conflict of interestThe authors declare that they have no conflicts of interest. FundingThis research was partially funded by the "Proyecto de Mejoramiento Genetico Nacional-PROMEG CUI 2432072". Authors’ contributionsThe study was conceptualized by MCG. Methodology CJFM and ATS. Validation FAT. Formal analysis, MCG and CRU. Internal research WAG. Resources CQP. Original draft writing MCG. Drafting, revising, and editing, MCR and MCG. Project administration CQP. All authors have read and accepted the published version of the manuscript. Data availabilityData supporting the results and conclusions of this research are available from the corresponding author, MCG, ATS, and CJFM upon reasonable request. ReferencesAbdolahi Khabisi, S. and Sarkari, B. 2016. Detection of Fasciola hepatica and Fasciola gigantica common and uncommon antigens, using rabbit hyper immune serum raised against their excretory-secretory and somatic antigens. J. Parasit. Dis. 40(4), 1552–1557. Abdullah, I., Tak, H. and Gani, S.A. 2019. Protein profile of somatic antigens of Paramphistomum cervi. IJZAB 4(6), 275–277. Ali, R., Afshan, K., Fuentes, M.V. and Firasat, S. 2022. Serological diagnostic potential of the 38-72 kDa somatic antigen of Gastrothylax crumenifer in buffaloes using the Indirect Enzyme-Linked Immunosorbent Assay. J. Hellenic Vet. Med. Soc. 73(2), 4105–4112. Alonso Villela, S.M., Kraïem, H., Bouhaouala-Zahar, B., Bideaux, C., Aceves Lara, C.A. and Fillaudeau, L. 2020. A protocol for recombinant protein quantification by densitometry. Microbiol 9, 1175–1182. Alzieu, J.P. and Dorchies, P. 2007. Réémergence de la paramphistomose bovine en France: synthèse des connaissances actuelles épidémiologiques, physiopathologiques et diagnostiques. Bull. Acad. Vet. Fr. 160(2), 93–99. Anuracpreeda, P., Chawengkirttikul, R. and Sobhon, P. 2016. Antigenic profile, isolation and characterization of whole body extract of Paramphistomum gracile. Parasite Immunol. 38(7), 431–438. Arias, M., Lomba, C., Dacal, V., Vázquez, L., Pedreira, J., Francisco, I., Piñeiro, P., Cazapal-Monteiro, C., Suárez, J.L., Díez-Baños, P., Morrondo, P., Sánchez-Andrade, R. and Paz-Silva, A. 2011. Prevalence of mixed trematode infections in an abattoir receiving cattle from northern Portugal and north-west Spain. Vet. Rec. 168(15), 408. Arroyo, M.I., Gómez, L., Hernández, C., Agudelo, D., Galván-Diaz, A.L. and Velásquez, L.E. 2022. Prevalencia de Fasciola hepatica y Paramphistomidae en bovinos de doble propósito en una hacienda del trópico bajo andino colombiano. Rev. Med. Vet. Zoot. 69(1), 9–32. Atcheson, E., Lagan, B., Mccormick, R., Edgar, H., Hanna, R.E.B., Rutherford, N.H., Mcevoy, A., Huson, K.M., Gordon, A., Aubry, A., Vickers, M., Robinson, M.W. and Barley, J.P. 2022. The effect of naturally acquired rumen fluke infection on animal health and production in dairy and beef cattle in the UK. Front. Vet. Sci. 9, 968753. Bradford, M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254. Cabrera, M., Chávez, S. and Ravines, J. 2020. Manual: protocolos de laboratorio en la elaboración de vacunas para el control inmunológico de Fasciola hepatica en rumiantes. Peru, Lima: Martínez Compañón Editores. Cerda, C., Veloso-Frías, J., Lobos-Chávez, F., Oyarzún-Ruiz, P., Henríquez, A., Loyola, M., Fuente, M.C.S.D.L., Ortega, R., Letelier, R. and Landaeta-Aqueveque, C. 2019. Morphological and molecular identification with frequency analysis of Calicophoron microbothrioides in central Chile. Rev. Bras. Parasitol. Vet. 28(4), 582–591. Cordero Del Campillo, M., Rojo, F.A., Martínez, A.R., Sanchez, M.C., Hernandez, S., Navarrete, I., Diez, P., Quiroz, H. and Carvalho, M. 1999. Parasitosis del aparato digestivo. In Parasitología Veterinaria, Eds., González, F.L. Madrid, España: McGraw-Hill, pp: 195–259. Coronado, J., Torrel-Pajares, T. and Vargas-Rocha, L. 2023. Relationship between number of adult rumen fluke (Calicophoron microbothrioides) and eggs per gram of feces in culled dairy cattle in Peru: a pilot study. Rev. Colomb. Cienc. Pecu. 37(2), 113–121. Cueva-Rodríguez, M., Torrel, T., Hobán, C., Alvarez-García, W., Mejía, F. and Vargas-Rocha, L. 2024. Prevalence of Fasciola hepatica and Calicophoron spp. in extensively reared cattle in the Florida district (Amazonas), Peru. Rev. Mex. Cienc. Pecu. 15(2), 376–392. De Vera, M., Sato, K. and Oyong, G. 2009. Comparison of protein profile of co-existing Fasciola hepatica and Fasciola gigantica parasite in Bos taurus (cattle) and Bubalus bubalis (Philippine water buffalo). J. Protozool. Res. 19(1), 1–9. Dezhabad, A., Dalimi, A., Hoghooghi Rad, N. and Madani, R. 2023. Secretory Excretory and Somatic Immunogenic Antigens Profiles of Adult Fasciola spp. Arch. Razi. Inst. 78(1), 435–443. Dutt, S.C. and Srivastava, H.D. 1972. The life history of Gastrodiscoides hominis (Lewis and McConnel, 1876) Leiper, 1913--the amphistome parasite of man and pig. J. Helminthol. 46(1), 35–46. Eduardo, S.L. 1983. The taxonomy of the family Paramphistomidae Fischoeder, 1901 with special reference to the morphology of species occurring in ruminants. III. Revision of the genus Calicophoron Näsmark. Syst. Parasitol. 5(1), 25–79. El-Khabaz, K.A.S., Abdel-Hakeem, S.S. and Arfa, M.I. 2019. Protozoan and helminthes parasites endorsed by imported camels (Camel dromedaries) to Egypt. J. Parasit. Dis. 43(4), 607–615. Fuertes, M., Pérez, V., Benavides, J., González-Lanza, M.C., Mezo, M., González-Warleta, M., Giráldez, F.J., Fernández, M., Manga-González, M.Y. and Ferreras, M.C. 2015. Pathological changes in cattle naturally infected by Calicophoron daubneyi adult flukes. Vet. Parasitol. 209(3–4), 188–196. Ghatani, S., Toledo, R. and Fried, B. 2024. Amphistomes. Digenetic Trematodes. Cham, Switzerland: Springer, pp: 323–347. Hernández-Hernández, J.C., González-Garduño, R., Ortiz-Pérez, D.O., Villa-Mancera, A., Arias-Vázquez, M.S. and Paz-Silva, A. 2023. Prevalence of flukes (Fasciola hepatica and paramphistomids) in cattle in south-eastern Mexico. Helminthologia 60(2), 141–151. Hong, Y., Peng, J., Jiang, W., Fu, Z., Liu, J., Shi, Y., Li, Z. and Lin, J. 2011. Proteomic Analysis of Schistosoma japonicum Proteins that are Differentially Expressed Among Hosts Differing in Their Susceptibility to the Infection. MCP. 10(8), M110.006098. Horak, I.G. 1971. Paramphistomiasis of domestic ruminants. Adv. Parasitology. 9, 33–72. Howell, A.K. and Williams, D.J.L. 2020. The Epidemiology and Control of Liver Flukes in Cattle and Sheep. Vet. Clin. North Am. Food Anim. Pract. 36(1), 109–123. Huson, K.M., Morphew, R.M., Allen, N.R., Hegarty, M.J., Worgan, H.J., Girdwood, S.E., Jones, E.L., Phillips, H.C., Vickers, M., Swain, M., Smith, D., Kingston-Smith, A.H. and Brophy, P.M. 2018. Polyomic tools for an emerging livestock parasite, the rumen fluke Calicophoron daubneyi; identifying shifts in rumen functionality. Parasit. Vectors. 11(1), 617. Huson, K.M., Oliver, N.A.M. and Robinson, M.W. 2017. Paramphistomosis of Ruminants: an Emerging Parasitic Disease in Europe. Trends Parasitol. 33(11), 836–844. Hussain, K. 2013. Immunological and molecular characterization of common amphistomes infecting ruminants, Master’s Thesis, Indian Veterinary Research Institute (Deemed University). Instituto Nacional de Estadística e Informática (INEI). 2012. IV Censo Nacional Agropecuario 2012. Jadav, M.M., Kumar, N., Das, B. and Solanki, J.B. 2018. Immunodiagnostic potency of homologous antigens for natural Paramphistomum epiclitum infection in small ruminants in plate and paper enzyme linked immunosorbent assay. Indian J. Anim. Res. 52(1), 83–91. Johnston, M.J.G., Macdonald, J.A. and Mckay, D.M. 2009. Parasitic helminths: a pharmacopeia of anti-inflammatory molecules. Parasitology 136(2), 125–147. Jones, R.A., Brophy, P.M., Mitchell, E.S. and Williams, H.W. 2017. Rumen fluke (Calicophoron daubneyi) on Welsh farms: prevalence, risk factors and observations on co-infection with Fasciola hepatica. Parasitol 144(2), 237–247. Jyoti., Prasad, A. and Singh, N.K. 2014. Evaluation of antibody response to various developmental stage specific somatic antigens of Paramphistomum epiclitum in Goats. Biomed. Res. Int. 2014, 505484. Kumar, V. 1980. The digenetic trematodes, Fasciolopsis buski, Gastrodiscoides hominis and Artyfechinostomum malayanum, as zoonotic infections in South Asian countries. Ann. Soc. Belg. Med. Trop. 60(4), 331–339. Lopes, A.S., López-Hernández, D. and Pinto, H.A. 2024. Morphological and molecular data confirm the occurrence of Paramphistomum leydeni (Trematoda: paramphistomidae) in ruminants from Southern Brazil. Vet. Parasitol. Reg. Stud. Rep. 52, 101050. Martinez-Ibeas, A.M., Munita, M.P., Lawlor, K., Sekiya, M., Mulcahy, G. and Sayers, R. 2016. Rumen fluke in Irish sheep: prevalence, risk factors and molecular identification of two paramphistome species. BMC. Vet. Res. 12(1), 143. Matsumoto, H., Haniu, H. and Komori, N. 2019. Determination of Protein Molecular Weights on SDS-PAGE. Methods. Mol. Biol. 1855, 101–105. Mattison, R.G., Abidi, S.M.A., Nizami, W.A. and Hanna, R.E.B. 1994. Preliminary studies in numerical systematics of the Paramphistomoidea (Digenea) from domestic Artiodactyla of Northern India. Syst. Parasitol. 27, 81–103. Harizt, A.M., Malahubban, M., Syed-Hussain, S.S., Ramanoon, S.Z., Sadiq, M.B., Sarbini, S., Hassan, M.N., Mustafa, S., Aziz, N.A.A., Isa, N.M.M., Nating, W.R., Asut, R. and Kamaludeen, J. 2021. Gastrointestinal parasitic infections of buffaloes (Bubalus bubalis) in Sarawak Borneo: Prevalence, risk factors, and farming practices. Trop. Biomed. 38(3), 318–326. Nikander, S. and Saari, S. 2007. Notable seasonal variation observed in the morphology of the reindeer rumen fluke (Paramphistomum leydeni) in Finland. Rangifer 27, 47–57. O’Shaughnessy, J., Garcia-Campos, A., Mcaloon, C.G., Fagan, S., De Waal, T., Mcelroy, M., Casey, M., Good, B., Mulcahy, G., Fagan, J., Murphy, D. and Zintl, A. 2018. Epidemiological investigation of a severe rumen fluke outbreak on an Irish dairy farm. Parasitol 145(7), 948–952. Rafiq, N., Ayaz, S., Niaz, S., Haleem, S., Ullah, R., Bari, A., Bourhia, M. and Ali, E.A. 2022. Changes in the prevalence of natural Paramphistomum Cercariae Infection in Indoplanorbis and Lymnaea Intermediate hosts influenced by meteorological factors. J. Trop. Med. 2022, 8719834. Rehman, A., Ullah, R., Khan, M.A.H. and Abidi, S.M.A. 2020. Glutathione-S-transferase: an important diagnostic antigen of liver amphistome Gigantocotyle explanatum, infecting the Indian water buffalo. Acta Trop. 205, 105400. Rizwan, H.M., Usman, M., Naeem, M.A., Farid, M.U., Younus, M., Sajid, M.S., Tahir, U.B., Luqman, N., Abbas, H., Ateeq, M.K., Taseer, M.S.A. and Asif, M. 2022. Prevalence of Ruminant Paramphistomosis and Comparative Histopathology of the Infected Rumens in Narowal District, Punjab, Pakistan. Helminthologia 59(4), 377–384. Rolfe, P.F., Boray, J.C., Nichols, P. and Collins, G.H. 1991. Epidemiology of paramphistomosis in cattle. Int. J. Parasitol. 21(7), 813–819. Sah, R., Acosta, L. and Toledo, R. 2019. A case report of human gastrodiscoidiasis in Nepal. Parasitol. Int. 71, 56–58. Sirisriro, A., Grams, R., Vichasri-Grams, S., Ardseungneon, P., Pankao, V., Meepool, A., Chaithirayanon, K., Viyanant, V., Tan-Ariya, P., Upatham, E.S. and Sobhon, P. 2002. Production and characterization of a monoclonal antibody against recombinant fatty acid binding protein of Fasciola gigantica. Vet. Parasitol. 105(2), 119–129. Sripa, J., Laha, T. and Sripa, B. 2017. Characterization and functional analysis of fatty acid binding proteins from the carginogenic liver fluke, Opisthorchis viverrini. Parasitol. Internat. 66(4), 419–425. Taylor, M.A., Coop, R.L. and Wall, R.L. 2007. Veterinary parasitology. 3.ª ed. London, UK: Wiley-Blackwell. Tehrani, A., Javanbakht, J., Khani, F., Hassan, M.A., Khadivar, F., Dadashi, F., Alimohammadi, S. and Amani, A. 2015. Prevalence and pathological study of Paramphistomum infection in the small intestine of slaughtered ovine. J. Parasit. Dis. 39(1), 100–106. Torrel, S., Rojas-Moncada, J., Saldaña, K., Silva, M., Gallardo, I., Cadenillas, R.D.P., Alfaro, D., Irigoín, C., Murga-Moreno, C.A. and Vargas-Rocha, L. 2023. Trematodos del ganado vacuno lechero al pastoreo en Cajamarca, Perú: fasciola hepatica y Calicophoron microbothrioides. Rev. Investig. Vet. Perú 34(4), e24296. Torrel, T.S., Paz, A. 2015. Paramphistomosis en Bovinos y Ovinos en Cajamarca. Cajamarca: Martínez Compañón Editores. Torrel-Pajares, T.S., Murga-Moreno, C.A., Infante-Mendo, M.S., Vargas-Rocha, L.A., Burga-León, J.E.J. and Rojas-Moncada, J.D.D. 2022. Occurrence of paramphistomosis (Trematoda: digenea) in sheep in northern Peru. Vet. Parasitol. Reg. Stud. Rep. 34, 100763. Van Der Ree, A.M. and Mutapi, F. 2015. The helminth parasite proteome at the host-parasite interface - Informing diagnosis and control. Exp. Parasitol. 157, 48–58. Wang, C.R., Qiu, J.H., Zhu, X.Q., Han, X.H., Ni, H.B., Zhao, J.P., Zhou, Q.M., Zhang, H.W. and Lun, Z.R. 2006. Survey of helminthes in adult sheep in Heilongjiang Province, People’s Republic of China. Vet. Parasitol. 140(3–4), 378–382. Supplementary Figure
Fig. 1. MW. Molecular weight standards in the range of 10–260 kDa. (Lane 1, Lane 4). Silver-stained somatic protein profile of different numbers of adult paramphistomids analyzed by SDS-PAGE. L1 (n=10 paramphistomids/500 μg/ml); L2 (n=20 paramphistomids/726.5 μg/ml); L3 (n=50 paramphistomids); L4 (n=10 paramphistomids, different sample); L5 and L6 (n=20 paramphistomids, different sample); C1 (bovine albumin 125 μg/ml); C2 (bovine albumin 250 μg/ml); C3 (bovine albumin 500 μg/ml); MW represents molecular weight standards. L1 (500 μg/ml, lane 1 in Fig. 1) and L3 (2,000 μg/ml, lane 4 in Fig. 1). (Lane 2, Lane 5) Perfil proteico somático teñido con plata de 20 paramphistomidos adultos analizados mediante SDS-PAGE. L1-L10 (n=20 paramphistomidos, muestras diferentes), C (albúmina bovina), MW presenta los estándares de peso molecular. L7 (726.5 μg/ml, carril 2 en Fig. 2) y C (250 μg/ml, carril 5 en Fig. 1). (Lane 3) Silver-stained somatic protein profile of 20 and 30 adult paramphistomids analyzed by SDS-PAGE. L1-L7 (n=20 paramphistomids, different samples), L8-L10 (n=30 paramphistomids, different samples), C (bovine albumin 500 μg/ml), MW shows molecular weight standards. L9 (1979.5 μg/ml, lane 3 in Fig. 1). | ||
| How to Cite this Article |
| Pubmed Style Fernandez-mendoza CJ, Tayca-saldaña A, Cueva-rodríguez M, Aliaga-tambo F, Rodríguez-ulloa C, Alvarez-garcía W, Quilcate-pairazamán C, Cabrera-gonzález M. First characterization of somatic proteins of trematodes of the family Paramphistomidae by SDS-PAGE isolated from cattle from the Cajamarca region, Peru. Open Vet. J.. 2026; 16(1): 372-383. doi:10.5455/OVJ.2026.v16.i1.34 Web Style Fernandez-mendoza CJ, Tayca-saldaña A, Cueva-rodríguez M, Aliaga-tambo F, Rodríguez-ulloa C, Alvarez-garcía W, Quilcate-pairazamán C, Cabrera-gonzález M. First characterization of somatic proteins of trematodes of the family Paramphistomidae by SDS-PAGE isolated from cattle from the Cajamarca region, Peru. https://www.openveterinaryjournal.com/?mno=241960 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.34 AMA (American Medical Association) Style Fernandez-mendoza CJ, Tayca-saldaña A, Cueva-rodríguez M, Aliaga-tambo F, Rodríguez-ulloa C, Alvarez-garcía W, Quilcate-pairazamán C, Cabrera-gonzález M. First characterization of somatic proteins of trematodes of the family Paramphistomidae by SDS-PAGE isolated from cattle from the Cajamarca region, Peru. Open Vet. J.. 2026; 16(1): 372-383. doi:10.5455/OVJ.2026.v16.i1.34 Vancouver/ICMJE Style Fernandez-mendoza CJ, Tayca-saldaña A, Cueva-rodríguez M, Aliaga-tambo F, Rodríguez-ulloa C, Alvarez-garcía W, Quilcate-pairazamán C, Cabrera-gonzález M. First characterization of somatic proteins of trematodes of the family Paramphistomidae by SDS-PAGE isolated from cattle from the Cajamarca region, Peru. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 372-383. doi:10.5455/OVJ.2026.v16.i1.34 Harvard Style Fernandez-mendoza, C. J., Tayca-saldaña, . A., Cueva-rodríguez, . M., Aliaga-tambo, . F., Rodríguez-ulloa, . C., Alvarez-garcía, . W., Quilcate-pairazamán, . C. & Cabrera-gonzález, . M. (2026) First characterization of somatic proteins of trematodes of the family Paramphistomidae by SDS-PAGE isolated from cattle from the Cajamarca region, Peru. Open Vet. J., 16 (1), 372-383. doi:10.5455/OVJ.2026.v16.i1.34 Turabian Style Fernandez-mendoza, Charito Jennyfer, Antony Tayca-saldaña, Medali Cueva-rodríguez, Fernando Aliaga-tambo, Claudia Rodríguez-ulloa, Wuesley Alvarez-garcía, Carlos Quilcate-pairazamán, and Marco Cabrera-gonzález. 2026. First characterization of somatic proteins of trematodes of the family Paramphistomidae by SDS-PAGE isolated from cattle from the Cajamarca region, Peru. Open Veterinary Journal, 16 (1), 372-383. doi:10.5455/OVJ.2026.v16.i1.34 Chicago Style Fernandez-mendoza, Charito Jennyfer, Antony Tayca-saldaña, Medali Cueva-rodríguez, Fernando Aliaga-tambo, Claudia Rodríguez-ulloa, Wuesley Alvarez-garcía, Carlos Quilcate-pairazamán, and Marco Cabrera-gonzález. "First characterization of somatic proteins of trematodes of the family Paramphistomidae by SDS-PAGE isolated from cattle from the Cajamarca region, Peru." Open Veterinary Journal 16 (2026), 372-383. doi:10.5455/OVJ.2026.v16.i1.34 MLA (The Modern Language Association) Style Fernandez-mendoza, Charito Jennyfer, Antony Tayca-saldaña, Medali Cueva-rodríguez, Fernando Aliaga-tambo, Claudia Rodríguez-ulloa, Wuesley Alvarez-garcía, Carlos Quilcate-pairazamán, and Marco Cabrera-gonzález. "First characterization of somatic proteins of trematodes of the family Paramphistomidae by SDS-PAGE isolated from cattle from the Cajamarca region, Peru." Open Veterinary Journal 16.1 (2026), 372-383. Print. doi:10.5455/OVJ.2026.v16.i1.34 APA (American Psychological Association) Style Fernandez-mendoza, C. J., Tayca-saldaña, . A., Cueva-rodríguez, . M., Aliaga-tambo, . F., Rodríguez-ulloa, . C., Alvarez-garcía, . W., Quilcate-pairazamán, . C. & Cabrera-gonzález, . M. (2026) First characterization of somatic proteins of trematodes of the family Paramphistomidae by SDS-PAGE isolated from cattle from the Cajamarca region, Peru. Open Veterinary Journal, 16 (1), 372-383. doi:10.5455/OVJ.2026.v16.i1.34 |