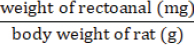| Research Article | ||
Open Vet. J.. 2026; 16(1): 384-395 Open Veterinary Journal, (2026), Vol. 16(1): 384-395 Research Article The effect of topical and oral combination of ethanolic extract of Graptophyllum pictum leaf on the inflammation level of rectoanal tissue in hemorrhoid-induced ratDesak Gede Budi Krisnamurti1*, Annia Zhafarina Dalilati2, Ani Retno Prijanti3 and Christian Marco Hadi Nugroho41Departement of Medical Pharmacy, Drug Discovery Research Cluster, IMERI, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia 2Master’s Programme in Biomedical Sciences, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia 3Departement of Biochemistry and Molecular Biology, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia 4Animal Health Research and Diagnostic Unit Medika Satwa Laboratoris, Bogor, Indonesia *Corresponding Author: Desak Gede Budi Krisnamurti. Department of Pharmacy, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia. Email: desak.gede [at] ui.ac.id Submitted: 18/02/2025 Revised: 30/11/2025 Accepted: 29/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: Hemorrhoids are a pathological condition characterized by abnormal dilation of blood vessels and swelling in the rectoanal region, often accompanied by inflammation. The ethanolic extract of purple leaf (Graptophyllum pictum) has demonstrated potential anti-inflammatory properties, which are attributed to its rich content of bioactive compounds, including flavonoids, tannins, and saponins. Aim: This research aims to assess the potential of topically applied and orally administered purple leaf extract in regulating inflammation in the rectoanal tissue of hemorrhoid-induced rats using 10% Croton oil. Methods: Following a 3-day induction period with 10% Croton oil, the rats were subjected to specific therapeutic regimens for 7 days. Subsequently, the rectoanal tissue was isolated and divided for histopathological analysis and assessment of immunological parameters. tTumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and Transforming Growth Factor Beta 1 (TGF-β1) cytokine levels were quantified using ELISA, while interleukin-10 (IL-10) messenger RNA (mRNA) expression was determined using qRT-PCR. The histopathological observation of the rectoanal and calculation of the rectoanal coefficient were conducted. Results: The application of 10% Croton oil induced a significant elevation of TGF-β1 and TNF-α, but not IL-6, in the negative control group compared to the normal and treated group. The combined topical and oral administration of purple leaf extract reduced TNF-α, IL-6, and TGF-β1 cytokines, and significantly elevated the IL-10 mRNA expression in the rectoanal tissue. However, purple leaf extract did not appear to reduce the rectoanal coefficient. Conclusion: The presence of diverse active compounds, in purple leaf extract notably flavonoids, is thought to modulate tissue inflammation and facilitate tissue repair. Keywords: Purple Leaf, Inflammation, Flavonoid, Hemorrhoids. IntroductionHemorrhoids are characterized by the abnormal dilation of veins in the anal canal, disintegration of connective tissue, and prolapse of anal cushion tissue. (Lohsiriwat, 2012; Margetis, 2019; De Marco and Tiso, 2021) Hemorrhoids are a prevalent condition globally. It is estimated that approximately 4.4% of the worldwide population suffers from hemorrhoids. (Kibret et al., 2021) In Indonesia, the number of hemorrhoid sufferers was recorded at 12.5 million in 2008, with an anticipated increase to 20.3 million by 2020 among individuals aged 45–65 years.(Putri, 2022). Hemorrhoids frequently manifest as asymptomatic; nonetheless, they induce discomfort in patients, characterized by bleeding and pain during bowel movement. Additionally, there is a notable association between hemorrhoids and the incidence of colorectal adenoma, signifying an augmented susceptibility to colorectal cancer among individuals with hemorrhoids. (Toyoshima et al., 2023) Common symptoms experienced by individuals with hemorrhoids include rectal bleeding, prolapsed anal tissue, anal pruritus, and pain culminating in a burning sensation (Dönmez et al., 2020). The precise etiology of hemorrhoids remains undetermined; however, several studies stated that various risk factors, including inadequate fiber consumption, constipation, pregnancy, heredity, and obesity, may influence them. (Peery et al., 2015; De Marco and Tiso, 2021) Furthermore, smoking and alcohol intake are believed to be associated with an elevated risk of developing hemorrhoids. Research indicates a correlation between limited physical activity and the incidence of hemorrhoids. Regular physical activity, such as walking, swimming, or running, is indispensable for fortifying pelvic musculature, enhancing circulation, and promoting regular bowel movements (De Marco and Tiso, 2021). In this study, inflammation and hemorrhoids were induced by applying 10% croton oil. The utilization of croton oil for the induction of hemorrhoids has been well-documented in numerous prior studies. (Hutagalung, 2019; Parish Budiono et al., 2021; Zhang et al., 2021; Kusumawati et al., 2022; Prasetyo et al., 2023) Croton oil, extracted from the seeds of the Croton tiglium plant, has a rich historical tradition in traditional Chinese medicine, where it has been employed for the treatment of various ailments. Notably, it is recognized for its high concentration of phorbol esters, particularly phorbol 12-tiglate 13-decanoate. Moreover, Croton oil contains several other phorbol ester compounds, including 12-O-tetradecanoylphorbol-13-acetate (TPA), 4-deoxy-4a-phorbol diester, and phorbol monoester. The administration of low doses of croton oil has shown efficacy in the management of various conditions, notably including constipation, abdominal pain, and intestinal infections. Furthermore, Croton oil has demonstrated anti-tumor and analgesic properties, along with the ability to inhibit the proliferation of bacteria and fungi (Niu et al., 2020; Zhang et al. 2022). Croton oil is markedly toxic and irritable in elevated concentrations, eliciting pronounced inflammatory responses. The TPA compound inherent in croton oil has been observed to induce histamine release from mast cells in vivo (Sagi-Eisenberg et al., 1985). As an inflammatory mediator, histamine plays a pivotal role in instigating vasodilation, elevating cell permeability, and acts as a chemoattractant. Consequently, the elevated histamine levels lead to the extravasation of immune cells into rectoanal tissue, thus culminating in edema and an escalated state of tissue inflammation. This upsurge in inflammation is characterized by the upregulation of various inflammatory markers, encompassing proinflammatory cytokines, chemokines, acute-phase proteins, and matrix metalloproteinases (MMPs) (Branco et al., 2018). Purple leaf (Graptophyllum pictum), an herbaceous plant originating from Papua New Guinea and extensively cultivated in various regions of Asia, has garnered substantial attention for its therapeutic potential in hemorrhoid treatment (United States Department of Agriculture: Natural Resources Conservation Services, 2021). This plant harbors a rich reservoir of bioactive compounds, including flavonoids, tannins, anthraquinones, and saponins, demonstrating antioxidant, anticancer, wound-healing, antimicrobial, and analgesic properties. Studies have established the capacity of flavonoids to impede the expression of pro-inflammatory cytokines and elevate superoxide dismutase (SOD) levels in experimental animals (Serra et al., 2015; Parish Budiono et al., 2021). Numerous studies have demonstrated that the anti-inflammatory properties of flavonoids are linked to the inhibition of nuclear factor kappa B (NF-κB) activation. This inhibition consequently leads to a reduction in the signaling of pro-inflammatory genes, such as interleukin-1 (IL-1), tumor necrosis factor-alpha (TNF-α), and interleukin-6 (IL-6) (Georgescu et al., 2016). Tannins found in purple leaf extract are recognized for their astringent properties as well as their anti-inflammatory effects. These qualities aid in wound healing by protecting inflamed tissue from drying out and further inflammation. Additionally, tannins bind to the superficial protein layer of the skin and mucosa, forming a protective barrier that helps prevent water loss. They are also noted for their antibacterial properties and their role in promoting re-epithelialization and collagen organization at the wound site (Twumasi et al., 2019). Moreover, based on prior studies, oral administration of purple leaf extract has exhibited efficacy in attenuating the pro inflammatory cytokines TNF-α, IL-1β, IL-6, and matrix metalloproteinase-9 (MMP-9) in hemorrhoidal mice (Hutagalung, 2019; Parish Budiono et al., 2021; Kusumawati et al., 2022). Nonetheless, the combined administration of purple leaf extract via both topical and oral routes is suggested to yield superior therapeutic efficacy compared to oral administration alone. Oral delivery promotes systemic modulation of inflammatory processes and cytokine regulation, whereas topical formulations, such as creams, provide localized anti-inflammatory effects and protect tissue at the site of rectoanal injury. This study aims to assess the potential benefit of Graptophyllum pictum extract administered both topically and orally to modulate inflammation and repair rectoanal tissue in hemorrhoidal-induced rats. This study evaluates the potential benefits of administering Graptophyllum pictum extract both topically and orally to modulate inflammatory cytokines IL-6, TNF-α, and Transforming Growth Factor Beta 1 (TGF-β1), and aid in the repair of rectoanal tissue in a model of hemorrhoidal-induced rats by modulating the inflammatory response of interleukin-10 (IL-10) mRNA. To our knowledge, this is the first investigation into the combined topical and oral administration of ethanolic extract from G. pictum (purple leaf) in a rat model induced by 10% croton oil. While previous studies have highlighted the oral anti-inflammatory effects of G. pictum and provided comparative analyses of various topical concentrations, we are also the first to present data demonstrating how the combination of topical and oral administration G. pictum extract modulates inflammation by inducing the expression of TGF-β1 cytokine and IL-10 mRNA. Materials and MethodsHemorrhoids inductionThe rats were induced with hemorrhoids by applying 10% Croton oil to the rectoanal area using a sterile cotton bud for approximately 20 seconds. Croton oil (Sigma-Aldrich PCode 1003518147) was prepared by mixing with water, pyridine (Supelco EMSURE CAS No. 110-86-1), and diethyl ether within the ratio 1:4:5:10. The induction was carried out for 3 consecutive days, and the daily changes were observed and documented. This study has received approval for the use of experimental animals from the health. Administration of therapyTherapy was administered to the positive control group (10% Croton oil-induced rats + Venosmil gel), dose 1 (10% Croton oil-induced rats + combination of topical purple leaf topical (2.5%) and per oral 100 mg/kg BW per oral administration), dose 2 (10% croton oil-induced rats + combination of topical purple leaf topical (5%) and per oral 100 mg/kg BW per oral administration), and dose 3 (10% croton oil-induced rats + combination of topical purple leaf topical (10%) and per oral 100 mg/kg BW) as per the categorization in Table 1. Table 1. Groups of experimental rats.
Termination and rectoanal isolationOn the 10th day of the experiment, the experimental animals were anesthetized using ketamine. Surgical procedures were then carried out to isolate the rectoanal organ, and the measurement of length and weight of the rectoanal rats were recorded. A segment of the organ was preserved in 10% formalin for preparation and (HE) staining, while another part was stored in a freezer for subsequent total protein, qPCR, and ELISA tests. Measurement of TNF-α, IL-6, and TGF-β1 cytokine with ELISAThe rectoanal organ was homogenized with a PBS solvent at pH 7.4 and then centrifuged for 10 minutes at a speed of 10,000 × g. Before the ELISA test, the Bradford test was conducted to determine the total protein levels in the rectoanal area. The Bradford test was performed following the available protocol, and the ELISA test was carried out according to the protocol from ELK Biotechnology for TNF-α (ELK1387 Lot: 21111640773) and IL-6 (ELK1157 Lot: 21111640425), and FineTest for TGF-β1 (Lot: FN240628). Measurement of IL-10 mRNA Expression using quantitative reverse transcription-polymerase chain reaction (qRT-PCR)IL-10 and iNOS mRNA expression was measured using the qRT-PCR method according to protocols available from Toyobo Bio-Tech. The forward and reverse primers for the IL-10 and β-actin genes are shown in Table 2. Table 2. Forward and reverse primer for IL-10 and β-actin.
Histopathological analysis of the rectoanal tissueRectoanal tissue preserved in 10% formalin and was processed using the Formalin-Fixed Paraffin-Embedded Block technique followed by hematoxylin-eosin staining. The coefficient of the rectoanal was calculated to examine the edema. Calculations are carried out using the formula:
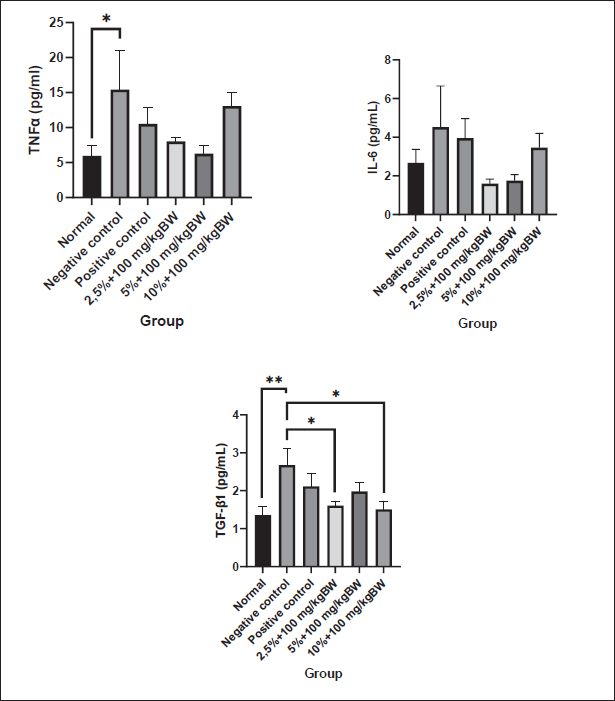
Data analysisThe data were analyzed using the SPSS version 26 software, with the normality of data distribution determined through the application of the Shapiro-Wilk test. Subsequently, data conforming to normal distribution underwent the One-way Analysis of Variants (ANOVA) test to ascertain significant differences among the treatment groups. The results of the ANOVA test were presented in a mean ± SEM graph and visualized using the GraphPad Prism 9.0 software. Ethical approvalFaculty of Medicine Universitas Indonesia-RSUPN Dr. Cipto Mangunkusumo no. KET-1270/UN2.F1/ETIK/PPM.00.02/2023, dated: 2-9 Sept 2023. ResultsProinflammatory cytokines analysisThe levels of the pro-inflammatory cytokines TNF-α, IL-6, and TGF-β1 in mouse rectoanal tissue were measured using the ELISA method. The results were then normalized by the total protein in the tissue obtained from the Bradford assay. After statistical analysis, it was found that TNF-α, IL-6, and TGF-β1 levels were normally distributed (p > 0.05) as shown in Figure 1. One-way ANOVA and post hoc LSD tests were conducted based on this finding. The statistical tests revealed a significant increase in TNF-α levels in the negative control group (15.42 ± 5.55 pg/ml) compared to the normal controls (5.97 ± 1.51 pg/ml), but there was no significant difference compared to the positive control (10.53 ± 2.37 pg/ml).
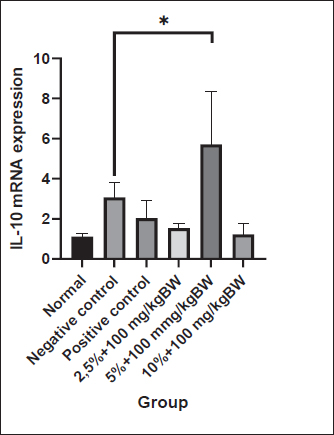
Fig. 1. Level of proinflammatory cytokines of the rectoanal tissue. The pro-inflammatory cytokine levels were measured using ELISA after 3 days of hemorrhoid induction and 7 days of Graptophyllum pictum topical and oral treatment. The data were shown as mean ± standard error (SEM). The asterisk(s) showed significant differences based on the LSD posthoc test (*p < 0.05; **p < 0.005). In the cohort of experimental animals administered a combination of topical and oral purple leaf extract at dosages 1 and 2, there was a noticeable inclination toward a reduction in the TNF-α/total protein levels in the rectoanal tissue in comparison to the negative control group. The recorded levels were 8.04 ± 0.54 pg/ml and 6.29 ± 1.22 pg/ml, respectively, albeit without reaching statistical significance. This trend contrasts with the cohort administered the same combination at dose 3, where the tissue TNF-α levels exhibited a persistent elevation (13.07 ± 1.98 pg/ml). In the One-way ANOVA test on IL-6 levels in rectoanal tissue, no significant differences were observed in the treatment groups (p > 0.05). However, it is worth noting that 10% croton oil induction for 3 days tended to increase IL-6 levels in the negative control animal group (4.52 ± 2.13 pg/ml) compared to the normal control animal group (2.67 ± 0.70 pg/ml) and positive control (3.95 ± 1.00 pg/ml). Upon examining the graph, it becomes evident that experimental animals receiving a combination of topical and oral purple leaf extract at dose 1 and dose 2 showed slightly lower IL-6 levels than the negative control (1.59 ± 0.23 pg/ml and 1.74 ± 0.31 pg/ml), although this difference was not statistically significant. In contrast, the group of experimental animals receiving a combination of topical and oral purple leaf extract at dose 3 displayed slightly higher IL-6 levels than those at dose 1 and dose 2 (3.45 ± 0.74 pg/ml). The application of 10% croton oil resulted in a significant increase in TGF-β1 cytokine levels in the negative control animals (2.68 ± 0.45 pg/ml) as compared to the normal animal group (1.36 ± 0.23 pg/ml) (p < 0.005). Following the combined topical and oral administration of purple leaf extract, a noteworthy reduction in TGF-β1 levels was observed, particularly at dose 1 (1.61 ± 0.12 pg/ml) and dose 3 (1.51 ± 0.19 pg/ml). Notably, the administration of a combination of topical and oral purple leaf extract at dose 2 tended to decrease TGF-β1 levels (1.98 ± 0.23 pg/ml) in comparison to the negative control group, although not statistically significant. Expression of IL-10 mRNAAccording to the measurements using qRT-PCR, results were obtained as shown in the graph in Figure 2. The cycle threshold was obtained, which was then calculated using the Livak equation to determine the relative expression of IL-10 mRNA. The results of the Shapiro-Wilk normality test on the relative expression of IL-10 mRNA showed that the data were normally distributed (p > 0.05) and continued with one-way ANOVA and post hoc least significant difference tests. The results of the LSD post hoc test stated that there was a significant difference (p < 0.05), as shown in the graph.
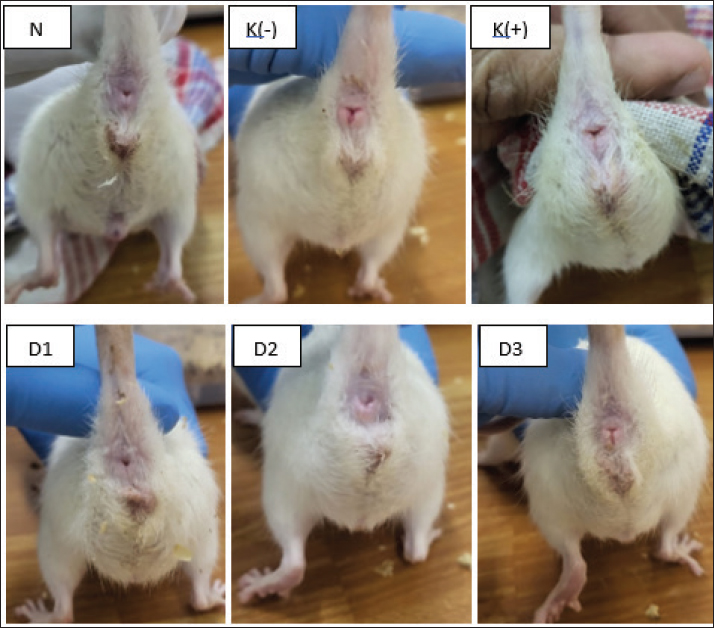
Fig. 2. Relative expression of IL-10 mRNA of the rectoanal tissue. The expression of IL-10 mRNA was measured using qRT–PCR after 3 days of hemorrhoid induction and 7 days of Graptophyllum. pictum topical and oral treatment. The data were shown as mean ± standard error (SEM). The asterisk showed significant differences based on the LSD posthoc test (*p < 0.05). Although the data were not significantly different, there was a trend towards an increase in the mean level of IL-10 mRNA expression in experimental animals in the negative control group (3.07 ± 0.73) when compared with animals in the normal group (1.12 ± 0.15) and positive control (2.03 ± 0.88). The combination of topical and oral administration of purple leaf extract at a dose of 2 (5% topical + 100 mg/kgBW oral) can increase the IL-10 mRNA expression level compared to the negative control significantly, to 5.71 ± 2.66. The mean ratio of IL-10 mRNA expression in experimental animals in dose group 2 was significantly higher than that in experimental animals in dose group 1 (1.53 ± 0.22) and dose 3 (1.23 ± 0.53). The mean ratio of IL-10 mRNA expression in experimental animals in dose group 2 was also significantly higher than in the normal control and positive control animal groups. Level of rectoanal edemaOver three consecutive days, the examination of rectoanal edema in rats induced by applying 10% Croton oil was conducted, followed by continued monitoring during the treatment phase. Upon macroscopic evaluation, it was observed that following the 3-day administration of 10% Croton oil, pronounced swelling and concurrent bleeding manifested in the rectoanal region of rats. Subsequent administration of purple leaf extract at doses 1, dose 2, and dose 3 demonstrated mitigating effects on both the swelling and the bleeding in the rectoanal region as depicted in Figure 3.
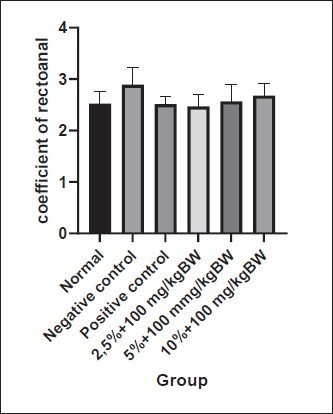
Fig. 3. Macroscopic appearance of the rectoanal of rats on the seventh day of therapy. Findings indicate the presence of edema and bleeding in the anorectal region of rats in the negative control rats compared to normal rats. After 7 days of Graptophyllum pictum treatment, the bleeding appeared to have ceased, although a slight edema had still appeared. N: untreated rats, K(-): 10% Croton oil-induced rats without therapy, K(+): 10% Croton oil-induced rats+Venosmil gel, D1: 10% Croton oil-induced rats+combination of purple leaf extract topical (2,5%)+ per oral 100 mg/kgBW, D2: 10% Croton oil-induced rats+combination of purple leaf extract topical (5%) and per oral 100 mg/kg BW, D3: 10% Croton oil-induced rats + combination of purple leaf extract topical (10%) and per oral 100 mg/kg BW. Statistically, to determine the level of edema in the rectoanal rats, the rectoanal coefficient was calculated. The rectoanal coefficient is the ratio between the rectoanal weight in milligrams and the body weight in g of the rat. An increase in the rectoanal coefficient value indicates the presence of edema in the rectoanal tissue of rats. Based on the normality test, it was found that the rectoanal coefficient data were normally distributed (p > 0.05); therefore, the one-way ANOVA test was continued. Induction of 10% croton oil for 3 days in the negative control group tended to increase the rectoanal coefficient (2.89 ± 0.33) compared to the normal control group (2.52 ± 0.24), although there was no statistically significant difference. The group of experimental animals given a combination of topical and oral purple leaf extract at doses 1 (2.47 ± 0.23), 2 (2.56 ± 0.32), and 3 (2.68 ± 0.23) showed a tendency to decrease rectoanal coefficient compared to the negative control group, although not statistically significantly different (p > 0.05), as shown in Figure 4.
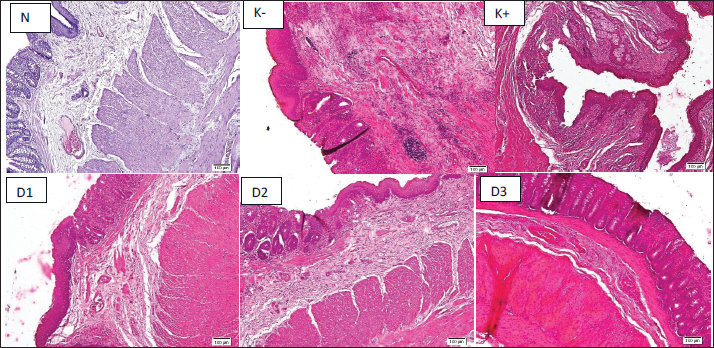
Fig. 4. The coefficient of represents the level of rectoanal edema. Thecoefficient of rectoanal was calculated after 3 days of hemorrhoid induction and 7 days of Graptophyllum. pictum topical and oral treatment. Data were shown as mean ± standard error (SEM). Although the data were not statistically significant, there were noticeable changes were observed in the negative control group compared to the normal group. The histopathological analysis of the rectoanal region in the experimental animals is delineated in Figure 5. Upon the administration of 10% croton oil, discernible necrosis was evident in the rectoanal tissue of the experimental animals in the negative control group in comparison to the normal group, particularly affecting the mucosa and submucosa layers. Furthermore, there was a notable infiltration of mononuclear cells in the submucosa layer of the rectoanal tissue of the negative control group mice, indicating an escalated inflammatory response following the administration of 10% croton oil. Subsequent to the combined oral and topical administration of purple leaf extract, a reduction in mononuclear cell infiltration in the submucosa layer was observed, although necrosis remained discernible.
Fig. 5. Histopathology features of rectoanal region show necrosis of the submucosal layer. The histopathology of rectoanal was observed after 3 days of hemorrhoid induction and 7 days of Graptophyllum pictum topical and oral treatment. The control negative group showed a noticeable massive infiltration of inflammatory cells into the submucosa of the rectoanal compared to the normal group. Treatment with Graptophyllum pictum appeared to reduce mononuclear infiltration and further tissue inflammation. Discussion10% croton oil was used to induce hemorrhoids in animal models, according to studies conducted by (Prasetyo et al., 2023), Budiono et al. (Parish Budiono, 2021), (Ayun et al., 2020), and (Kusumawati et al., 2022) with modification. In previous studies, 6% croton oil was commonly used to induce hemorrhoids in experimental animal models. However, in this study, a higher concentration of 10% croton oil was employed. In studies conducted by Prasetyo et al. (2023); Budiono et al. (2021); Ayun et al. (2020); and Kusumawati et al. (2022) used 10% croton oil was utilized to induce hemorrhoids in animal models, with some modifications. While previous research typically employed a 6% concentration of croton oil for inducing hemorrhoids in experimental animals, this study opted for a higher concentration of 10%. Preliminary optimization results indicated that the 10% concentration produced more consistent and pronounced signs of rectoanal inflammation, such as swelling and bleeding, without resulting in excessive tissue damage or mortality. The administration of croton oil was conducted over a period of three consecutive days, with daily documentation of the process. Croton oil is extracted from the seeds of the Croton tiglium plant, which is part of the Euphorbiaceae family (Zhang et al. (2022). This oil contains phorbol ester compounds that can induce inflammation by activating Protein Kinase C, which triggers an enzymatic cascade leading to inflammatory reactions, including the release of histamine from immune cells, particularly mast cells. Previous research has noted an increase in the number of mast cells in patients with hemorrhoids (Taweevisit et al., 2008). Croton oil is commonly used to create experimental animal models of inflammation and hemorrhoids due to its cytotoxic properties, particularly in high doses, which can induce cell death. In cases where cell death is caused by trauma or exposure to toxic chemical compounds, the inflammatory signaling pathway can be activated through the release of damage-associated molecular patterns (DAMPs). These DAMPs are then recognized by various inflammatory receptors, such as Toll-like receptors, NOD-like receptor, and Receptor for Advanced Glycation End Products. Subsequently, initiating the expression of inflammatory mediators. Consequently, this signaling activation leads to the infiltration of immune cells into the tissue, increased expression of pro-inflammatory cytokines and chemokines, and ultimately tissue damage. (Rock and Kono, 2008) In daily observations, the application of 10% Croton oil for 3 consecutive days caused bleeding and swelling in the rectoanal region of rats. This indicates inflammation in the rectoanal organ. Croton oil 10% can significantly increase the level of the cytokine TNF-α in the rectoanal organs of experimental animals in the negative control group compared to the normal experimental group. Experimental animals were induced by 10% croton oil and then continued with therapy using a combination of topical and oral purple leaf extract at dose 1 (2.5% topical + 100 mg/kg BW orally) and dose 2 (5% topical and 100 mg/kg BW orally) showed there was a tendency to decrease of TNF-α levels compared to the negative control animal group. This is in line with previous research, where oral administration of purple leaf extract can reduce the levels of TNF-α, Cyclooxygenase-2 (COX-2), and IL-6 in the blood serum of mice induced by hemorrhoids. (Riwanto et al., 2020) However, the combination of oral and topical administration of purple leaf extract at a dose of 3 (10% topical + 100 mg/kg BW) did not appear to reduce TNF-α expression in the rectoanal of rats. Another pro-inflammatory cytokine measured in this study was (IL-6), which plays a pivotal role in the response to cell or tissue damage that directly promotes inflammation. Administration of 10% croton oil to the negative control group of experimental animals tended to increase IL-6 cytokine levels compared to the normal control group, although it was not statistically significant. Even though there was no statistically significant difference, the experimental animal group received a therapy of purple leaf extract, topically and orally. The treatment was given at dose 1 (2.5% topical + 100 mg/kgBW) and dose 2 (5% topical + 100 mg/kgBW). In these animals, there seemed to be a reduction in the expression level of IL-6 in the rectoanal tissue. Among the experimental animals given a combination of topical and oral purple leaf extract, dose 2 (5% topical + 100 mg/kg BW) showed the lowest level of IL-6 cytokine expression compared to dose 1 (2.5% topical + 100 mg/kg BW) and dose 3 (10% topical + 100 mg/kg BW). The results of this research are in line with that research by (Prasetyo et al., 2023), which stated that the oral administration of purple leaf extract to hemorrhoidal mice was proven to significantly reduce the levels of pro-inflammatory cytokines IL-6 and COX-2. Inflammatory cytokines, such as TNF-α and IL-6, play a vital role in triggering inflammation by activating immune cells, including macrophages and neutrophils. Additionally, neutrophils facilitate tissue degradation, particularly of collagen, through the secretion of MMP-9. This degradation can affect surrounding tissues, such as the ligament of Treitz and the muscularis mucosa, ultimately leading to the prolapse of the anal cushion. (Budiono et al, 2021) This study also identified the expression of TGF-β1 cytokines in the rectoanal region. TGF-β1, a prominent isoform of the TGF-β family cytokines, is notably expressed in the skin. This cytokine is involved in diverse aspects of cellular regulation, particularly within the immune system, encompassing functions such as regulating proliferation, migration, and differentiation, and activating or suppressing various cell subsets. (Sabbadini et al., 2021) TGF-β1, in addition to its anti-inflammatory role, serves as a chemoattractant for monocytes, neutrophils, and lymphocytes, while also playing a regulatory role in the differentiation of Th0 cells into Th17.(Mangan et al., 2006) Similar to TNF-α, the application of 10% croton oil also led to a significant increase in TGF-β1 levels in the negative control animal group compared to the normal group. Following the administration of purple leaf extract for seven consecutive days, a significant decrease in TGF-β1 levels was observed, particularly evident at dose 1 (2.5% topical + 100 mg/kgBW) and dose 3 (10% topical + 100 mg/kgBW). Conversely, experimental animals in dose group 2 (5% topical + 100 mg/kgBW) exhibited a tendency toward elevated TGF-β1 levels. TGF-β1 is considered to be a key mediator in the regulation of MMP and its inhibitor, Tissue Inhibitor Metalloproteinase, in the development of venous insufficiency. It is also recognized for its role in indirectly promoting angiogenesis by increasing the expression of Vascular Endothelial Growth Factor. At the cellular level, elevated levels of TGF-β1 lead to heightened endothelial permeability, resulting in the migration of immune cells into the tissue. As far as we know, there is a lack of prior research on the role of TGF-β1 in regulating inflammation in hemorrhoids and the potential of purple leaf extract in modulating TGF-β1 expression. (Serralheiro et al., 2017; Serralheiro et al., 2018). In addition to measuring pro-inflammatory parameters, this study also assessed the expression of IL-10 mRNA as an anti-inflammatory indicator. The goal of this study was to evaluate the potential of the active compounds in purple leaf extract to enhance the production of anti-inflammatory cytokines, which play a role in reducing inflammation and promoting tissue repair. IL-10 is a cytokine expressed by various immune cells, including CD4+ T cells, macrophages, monocytes, natural killer cells, mast cells, and granulocytes, in vivo. It is also produced by other cells, such as keratinocytes and epithelial cells, in response to inflammation or cellular damage. IL-10 is known to have anti-inflammatory properties by inhibiting NF-κB pathway to produce inflammatory cytokines such as IL-1, TNF-α, and IL-6. (Subramanian Iyer and Cheng, 2012) Furthermore, IL-10 is associated with tissue healing and regeneration by promoting extracellular matrix production, especially type 1 collagen, promoting fibroblast migration, and production of hyaluronan, and recruiting endothelial progenitor cells to form new blood vessels in the wound site (King et al., 2014). Administration of topical and oral combination therapy of purple leaf extract at dose 1 (2.5% topical + 100 mg/kgBW) did not appear to increase IL-10 mRNA expression compared to the negative control. However, in the group of animals given dose 2 (5% topical + 100 mg/kgBW), there was a significant increase in IL-10 mRNA expression levels compared with the negative control, and tended to be higher than dose 1 (2.5% topical + 100 mg/kgBW) and dose 3 (10% topical + 100 mg/kgBW). The high levels of IL-10 mRNA in dose group 2 did not appear to be in line with the expression of the pro-inflammatory cytokines IL-6 and TNF-α, which tended to be lower in dose group 2 (5% topical + 100 mg/kgBW) than in dose groups 1 (2.5% topical + 100 mg/kgBW) and 3 (10% topical + 100 mg/kgBW). It is assumed that administering purple leaf extract orally and topically to hemorrhoid experimental animals can inhibit the expression of pro-inflammatory cytokines while also inducing the expression of the IL-10 gene. However, the potential of the active compounds in purple leaf extract to induce anti-inflammatory cytokines still needs to be further investigated. Croton oil induction for 3 consecutive days appeared to cause swelling and bleeding in the rectoanal area of the rats and appeared to decrease after topical and oral administration of purple leaf extract therapy for 7 days. This result is in line with a previous study (Kusumawati et al., 2022). Rectoanal coefficient calculations were carried out to determine the level of edema in the rectoanal tissue of rats induced by hemorrhoids due to the extravasation of inflammatory cells into the rectoanal tissue. Administration of 10% croton oil tended to increase the rectoanal coefficient, as shown by the negative control group, although it was not statistically significant compared to the normal controls. The increase in the rectoanal coefficient value in the hemorrhoidal rat is in line with the research by Zhang et al. (2021); Azeemuddin et al. (2014); and Hutagalung (2019). The increase of the rectoanal coefficient indicates a tendency for increased migration of immune cell to the rectoanal tissue due to induction by croton oil. The rectoanal coefficient, which tends to be higher in the negative control of animals, was in line with the expression levels of the pro-inflammatory cytokines IL-6, TNF-α, and TGF-β1, which also tend to be high in this group when compared with normal controls. 10% croton oil also appeared to cause necrosis in the negative control group compared to the normal group, which is in line with research by Kusumawati et al. (2022), which found necrosis, thickening of the mucosal layer, and infiltration of immune cells in the hemorrhoids-induced rats (Kusumawati et al., 2022). Croton oil, renowned for its high irritancy, may induce detriment to normal cell integrity, resulting in tissue inflammation. Concomitant administration of purple leaf extract yielded a notable reduction in tissue necrosis, particularly evident at doses 1 and 2, with a diminished effect observed at dose 3. These findings indicate a potential abatement of inflammation in experimental animals with hemorrhoids through the oral administration of purple leaf extract in conjunction with topical therapy. The flavonoids, saponins, tannins, and various other active compounds contained in purple leaf extract are believed to inhibit the expression of various inflammatory markers produced by leukocytes. Flavonoids and saponins play a role in inhibiting the NFκB signaling pathway, which can produce pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6. Furthermore, flavonoids and steroids are also known to inhibit the expression of adhesion molecules on endothelial cells induced by TNF-α and IL-1 through the NFκB signaling pathway, thereby inhibiting extravasation and excessive activation of immune cells (Moses et al., 2014; Song et al., 2018; Al-Khayri et al., 2022; Jing et al., 2022; Zhong et al., 2022). Previous studies have indicated the beneficial effects of orally administered purple leaf extract in reducing inflammation markers in rats with hemorrhoidal conditions. These benefits encompass a reduction in the extravasation of polymorphonuclear cells to the rectoanal tissue and a decrease in the rectoanal coefficient. Nevertheless, to the best of our knowledge, the present study constitutes the first investigation into the potential advantages of both oral and topical administration of purple leaf extract in mitigating inflammation and promoting expedited wound healing through the modulation of inflammatory and anti-inflammatory profiles. Many antioxidants, including flavonoids, enhance IL-10 production by activating the STAT3 signaling pathway in various immune cells, such as B cells and monocytes. Flavonoids are also recognized for their capacity to reduce the production of inflammatory cytokines by modulating the expression of the JAK/STAT pathway, which further facilitates IL-10 production in immune cells (Carlini et al., 2023). Additionally, tannins have been shown to enhance IL-10 production by modulating the NF-κB pathway, ultimately leading to the inhibition of pro-inflammatory cytokines. Tannins also regulate antioxidant enzyme activity by activating the Nrf2 pathway, which promotes an anti-inflammatory environment and aids in wound healing. Furthermore, the activation of Nrf2 results in macrophage polarization toward the M2 subset, which suppresses the inflammatory M1 subset. (Wang and He, 2022) The combination of oral and topical therapy in the management of hemorrhoids offers complementary benefits that address both the systemic and local aspects of the disease, enhancing overall therapeutic outcomes. The dual-route approach is expected to have a synergistic effect, where systemic and local actions reinforce each other, promoting more effective resolution of inflammation and faster tissue repair. ConclusionThe oral and topical administration of purple leaf extract (Graptophyllum pictum) to hemorrhoidal rats showed no statistically significant decrease in pro-inflammatory cytokine (TNF-α, IL-6) expression levels, and rectoanal edema. However, the treatment did result in increased IL-10 mRNA expression levels, decreased TGF-β1 levels, and reduced tissue necrosis, indicating a potential for promoting the healing of rectoanal tissue in hemorrhoidal-induced rats. AcknowledgmentWe also would like to thank Balai Besar Pengujian Standar Instrumen Veteriner (BBPSI Veteriner) and Medika Satwa Laboratoris for providing facilities and assistance in this research. Conflict of interestThe authors declare that there is no conflict of interest. FundingThis research funded by Universitas Indonesia through Publikasi Terindeks International (PUTI) Grants with contract No. NKB-645/UN2.RST/HKP.05.00/2023 Authors' contributionDGBK: conceptualization, data analysis and interpretation, and writing the manuscript. AZD: data collection, analysis, and interpretation, and manuscript writing. ARP: data interpretation, writing, and editing of the manuscript. CMHN: data collection and sharing of the manuscript. All authors are approved the final manuscript for publications. Data availabilityAll data supporting this findings are mentioned in the manuscript. ReferencesAl-Khayri, J.M., Sahana, G.R., Nagella, P., Joseph, B.V., Alessa, F.M. and Al-Mssallem, M.Q. 2022. Flavonoids as potential anti-inflammatory moleculesPotential Anti-Inflammatory Molecules: a reviewReview. Molecules 27(9), 1–24; doi:10.3390/molecules27092901 Ayun, N. Q., Kusmardi, Nurhuda, and Elya, B. 2020. Anti-inflammation of soursop leaves (Annona muricata l.) against hemorrhoids in mice induced by croton oil. Journal of Physiology, 86, 69–79.. Pharmacognosy Journal, 12(4), 784–792; doi: 10.5530/pj.2020.12.112 Azeemuddin, M., Viswanatha, G.L., Rafiq, M., Thippeswamy, A.H., Baig, M.R., Kavya, K.J., Patki, P.S. and Shyam, R. 2014. An Improved Experimental Model of Hemorrhoids in Rats: evaluation of Herbal Formulation’s Antihemorrhoidal ActivityAntihemorrhoidal Activity of an Herbal Formulation. ISRN. Pharmacol. 2014, 1–7; doi:10.1155/2014/530931 Branco, A.C.C.C.C., Yoshikawa, F.S.Y., Pietrobon, A.J. and Sato, M.N. 2018) Role of histamine in modulating immune response and inflammation. In Mediators of Inflammation (Vol. 2018). Hindawi Limited; doi:10.1155/2018/9524075 Budiono, B., Bernadus., Presetyo, S., Sigit., Riwanto, I., Ignatius., Sulistyaningsih, S., Sulistyaningsih. and EEriawan, N. 2021. Graptophyllum pictum Extract for treating in the Treatment of Experimental Hemorrhoids: effects on Vascular Leakage and Matrix Metalloproteinase-9 Levels. Open Access Macedonian J. Med. Sci. 9, 1785–1789; doi:10.3889/oamjms.2021.7763 Carlini, V., Noonan, D.M., Abdalalem, E., Goletti, D., Sansone, C., Calabrone, L. and Albini, A. 2023. The multifaceted nature of IL-10: regulation, role in immunological homeostasis, and its relevance to cancer, COVID-19, and post-COVID-19 conditions. Front. Immunol. 14, 1161067; doi:10.3389/fimmu.2023.1161067 De Marco, S. and Tiso, D. 2021. Lifestyle and risk factors in hemorrhoidal diseaseRisk Factors in Hemorrhoidal Disease. Front. Surg. (Vol. 8). 8, 1–5; doi:10.3389/fsurg.2021.729166 Dönmez , C., Yalçın, F.N., Boyacıoğlu., Korkusuz, P., Akkol, E.K., Nemutlu, E., Balaban, Y.H. and Çalışkan, U.K. 2020. From nutrition to medicine: assessing hemorrhoid healing activity of Solanum melongena L. and its major chemicals via in vivo experimental models. J. Ethnopharmacology , 261; doi:10.1016/j.jep.2020.113143 Georgescu, C., Frum, A., Virchea, L.I., Sumacheva, A., Shamtsyan, M., Gligor, F.G., Olah, N.K., Mathe, E. and Mironescu, M. 2022. Geographic variability of berry phytochemicals with antioxidant and antimicrobial properties. Molecules 27(15), 4986; doi: 10.3390/molecules27154986 1-18 Hutagalung, M.S.B. 2019. Phlebotrophic Effect of Graptophyllum Pictum (L.) Griff on Hemorrhoids in Experimental Wistar. J. Biomed. Translational Res. 5(1), 1; doi:10.14710/jbtr.v5i1.3704 Jing, W., Xiaolan, C., Yu, C., Feng, Q. and Haifeng, Y. 2022. Pharmacological effects and mechanisms of TAtannic acid. In Biomedicine and Pharmacotherapy (Vol. 154). Elsevier Masson s.r.l. https://doi.org/10.1016/j.biopha.2022.113561 Kibret, A.A., Oumer, M. and Moges, A.M. 2021. Prevalence and associated factors of hemorrhoids among adult patients visiting the surgical outpatient department in the University of Gondar Comprehensive Specialized Hospital, Northwest Ethiopia. PLos One. 16, e0249736; doi:10.1371/journal.pone.0249736 King, A., Balaji, S., Le, L.D., Crombleholme, T.M. and Keswani, S.G. 2014. Regenerative wound healingWound Healing: the role of interleukin-10Role of Interleukin-10. Adv. Wound. Care. 3(4), 315–323; doi:10.1089/wound.2013.0461 Kusumawati, I., Rullyansyah, S., Rohmania, R., Rizka, A.F., Hestianah, E.P. and Matsunami, K. 2022. Histomorphometric study of ethanolic extract of Graptophyllum pictum (L.) Griff. leaves on croton oil-induced hemorrhoid mice: a traditional Javanese traditional antihemorrhoid anti-hemorrhoid herb in Java. J. Ethnopharmacology 284;, doi:10.1016/j.jep.2021.114765 Lohsiriwat, V. 2012. Hemorrhoids: from basic pathophysiology to clinical management. World J. Gastroenterol. 18(17), 2009–2017; doi:10.3748/wjg.v18 Mangan, P.R., Harrington, L.E., O'Quinn, D.B., Helms, W.S., Bullard, D.C., Elson, C.O., Hatton, R.D., Wahl, S.M., Schoeb, T.R. and Weaver, C.T. 2006. Transforming growth factor-β induces development of the Th17 lineage. Nature 441(7090), 231–234; doi:10.1038/nature04754 Margetis. 2019. The pathophysiology of hemorrhoids. Ann. Gastroenterol. 32(3), 264–272; doi:10.20524/aog.2019.0355 Moses, T., Papadopoulou, K.K. and Osbourn, A. Metabolic and functional diversity of saponins, biosynthetic intermediates, and semi-synthetic derivatives. In Critical Reviews in Biochemistry and Molecular Biology,2014 Informa Health careHealthcare, 49(6), pp 439–462;. doi:10.3109/10409238.2014.953628 Niu, Q.L., Sun, H., Liu, C., Li, J., Liang, C.X., Zhang, R.R., Ge, F.R. and Liu, W. 2020. Croton tiglium essential oil compounds have antiproliferativeanti-proliferative and pro-apoptotic effects in A549 lung cancer cell lines. PLos One. 15(5), e0231437; doi:10.1371/journal.pone.0231437 O’Bryan, M.K., Gerdprasert, O., Nikolic-Paterson, D.J., Meinhardt, A., Muir, J.A., Foulds, L.M., Phillips, D.J., De Kretser, D.M. and Hedger, M.P. 2005. Cytokine profiles in the testes of lipopolysaccharide-treated rats treated with lipopolysaccharide reveal localized suppression of inflammatory responses. Am. J. Physiol-Regulatory Physiol. Integr. Physiol.288(6), 57–67; doi:10.1152/ajpregu.00651.2004 Parish Budiono, B., Adi Presetyo, S., Riwanto, I., Sulistyaningsih, S. and Nugroho, E.A. 2021. Graptophyllum pictum Extract for treating the Treatment of Experimental Hemorrhoids: effects on Vascular Leakage and Matrix Metalloproteinase-9 Levels. Open Access Macedonian J. Med. Sci. 9(B), 1785–1789; doi:10.3889/oamjms.2021.7763 Peery, A.F., Sandler, R.S., Galanko, J.A., Bresalier, R.S., Figueiredo, J.C., Ahnen, D.J., Barry, E.L. and Baron, J.A. 2015. Risk factors for hemorrhoids on screening colonoscopy. PLos One. 2015(9), 10; doi:10.1371/journal.pone.0139100 Prasetyo, S.A., Prajoko, Y.W., Nugroho, E.A., Dharmana, E., Susilaningsih, N. and Riwanto, I. 2023. Gratophyllum pictum (L.) Griff extract as an anti-inflammatory agent Extract as Anti-Inflammatory on Wistar rats with experimental hemorrhoids Rats With Experimental Hemorrhoids. Int. Surg. 107(2), 76–82; doi:10.9738/INTSURG-D-18-00039.1 Putri, G. 2022. Tunda BAB, Wasir Mengancam. Available via https://yankes.kemkes.go.id/view_artikel/403/tunda-bab-wasir-mengancamhttps://yankes.kemkes.go.id/view_artikel/403/tunda-bab-wasir-mengancam Riwanto, I., Nugroho, E.A., Susilaningsih, N., Prajoko, Y.W., Budiono, P., Prasetyo, S.A. and Azhar, A. 2020. Antioxidant and anti-inflammatory effects of Graptophyllum pictum (L.) Griff extract: study on SOD and COX-2 serum of experimental hemorrhoids. Med. Hosp. 7(2), 422–426. Rock , K.L. and Kono, H. 2008. Inflammatory The inflammatory response to cell death. In Annual Review of Pathology: mechanisms of Disease. ED., Moses. UK,3, pp: 99–126. https://doi.org/10.1146/annurev.pathmechdis.3.121806.151456 Sabbadini, F.F., Bertolini, M.M., De Matteis, S.S., Mangiameli, D.D., Contarelli, S.S., Pietrobono, S. and Melisi, D. 2021. MultifacetedThe multifaceted role of TGF-β in gastrointestinal tumors. Cancers 13(16), 1–24; doi:10.3390/cancers13163960 Sagi-Eisenberg, R., Foreman, J.C. and Shelly, R. 1985. Histamine release induced by histone and phorbol ester from rat peritoneal mast cellscells. Eur. J. Pharmacol. 113. Serra R Serra., Gallelli, L.L., Grande, R.R., Amato, B.B., De Caridi G, Sammarco., G, G.., Sammarco, G., Ferrari, F.F., Butrico, L.L., Gallo, G.G., Rizzuto, A.A. and De Franciscis S, Sacco. 2015. Hemorrhoids and matrix metalloproteinases: a multicenter study on the predictive role of biomarkers. Surg. (United. States). 159(2), 487–494; doi:10.1016/j.surg.2015.07.003 Serralheiro, P., Cairrão, E., Maia, C.J., João, M., Almeida, C.M.C. and Verde, I. 2017. Effect of TGF-beta1 on MMP/TIMP and TGF-beta1 receptors in great saphenous veins and its significance on chronic venous insufficiency. Phlebology 32(5), 334–341; 1–14; doi:10.1177/0268355516655067 Serralheiro, P., Novais, A., Cairrão, E., Maia, C., Costa Almeida, C.M. and Verde, I. 2018. Variability of MMP/TIMP and TGF-β1 receptors throughout chronic venous disease clinical progressionthe clinical progression of chronic venous disease. Int. J. Mol. Sci. 19(1), doi:10.3390/ijms19010006 Song, D., Zhao, J., Deng, W., Liao, Y., Hong, X. and Hou, J. 2018. Tannic acid inhibits NLRP3 inflammasome-mediated IL-1β production via blocking NF-κB signaling in macrophagesmacrophages. Biochem. Biophysical Res. Commun. 503(4), 3078–3085; doi:10.1016/j.bbrc.2018.08.096 Subramanian Iyer, S. and Cheng, G. 2012. Role of Interleukin-10Interleukin 10 Transcriptional Regulation in Inflammation and Autoimmune Disease. Taweevisit, T., Wisadeopas, N., Phumsuk, U. and Thorner, P.S. 2008. Increasedincreased mast cell density in hemorrhoid venous blood vessel. Med. J. 49(12), 977 Toyoshima, O., Nishizawa, T., Yoshida, S., Matsuno, T., Miyoshi, K., Naito, E., Shiomi, C., Uozumi, T., Fujishiro, M. and Saito, Y. 2023. Hemorrhoids as a risk factor for colonoscopy-induced colorectal adenomascolorectal adenomas on colonoscopy. Endoscopy. Int. Open. 11(05), E497–E503; doi:10.1055/a-2062-9443 Twumasi, M.A., Edmund, E., Priscilla, M.K., Mariam, B.G.E., Merlin, M.L.K. and Eric, W. 2019. Pharmacognostic studies of the leaves, stem and root of capparis erythrocarpos isert (Capparaceae). Pharmacognosy J. 11(1), 112–118. doi: 10.5530/pj.2019.1.19 United States Department of Agriculture: Natural Resources Conservation Services. 2021. Classification for Kingdom Plantae Down to Species Graptophyllum pictum (L.) Griffith. https://plants.usda.gov/home/classification/48895 Wang, L. and He, C. 2022. Nrf2-mediated anti-inflammatory polarization of macrophages as therapeutic targets for osteoarthritis. Front. Immunol. 13, 967193; doi:10.3389/fimmu.2022.967193 Zhang, L., Wu H, S.H., -S.., Chen, Y., Guo X, J.X., -J.., Wang, L., Wang C, Y.C., -Y.., Zhang J, H.J., -H.. and Tian, Y. 2006. Role of nitric oxide in Toll-like receptor 2 and 4 mRNA expression in the liver of rats with acute hemorrhagic necrotizing pancreatitis. World J. Gastroenterol. 12(3), 485–488. Zhang, T.T., Liu, Z.Z., Sun, X.X., Liu, Z.Z., Zhang, L.L., Zhang, Q.Q., Peng, W. and Wu, C. 2022. Botany, traditional uses, phytochemistry, and pharmacologicalpharmacological and toxicological effects of C.Croton tiglium Linn.: a comprehensive reviewreview. J. Pharm. Pharmacol. 74(8), 1061–1084; doi:10.1093/jpp/rgac040 Zhong, R., Miao, L., Zhang, H., Tan, L., Zhao, Y., Tu, Y., Angel Prieto, M., Simal-Gandara, J., Chen, L., He, C. and Cao, H. 2022. Anti-inflammatory activity of flavonols via inhibiting MAPK and NF-κB signaling pathways in RAW264.7 macrophages. Curr. Res. Food. Sci. 5, 1176–1184; doi:10.1016/j.crfs.2022.07 | ||
| How to Cite this Article |
| Pubmed Style Krisnamurti DGB, Dalilati AZ, Prijanti AR, Nugroho CMH. The effect of topical and oral combination of ethanolic extract of Graptophyllum pictum leaf on the inflammation level of rectoanal tissue in hemorrhoid-induced rat. Open Vet. J.. 2026; 16(1): 384-395. doi:10.5455/OVJ.2026.v16.i1.35 Web Style Krisnamurti DGB, Dalilati AZ, Prijanti AR, Nugroho CMH. The effect of topical and oral combination of ethanolic extract of Graptophyllum pictum leaf on the inflammation level of rectoanal tissue in hemorrhoid-induced rat. https://www.openveterinaryjournal.com/?mno=243436 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.35 AMA (American Medical Association) Style Krisnamurti DGB, Dalilati AZ, Prijanti AR, Nugroho CMH. The effect of topical and oral combination of ethanolic extract of Graptophyllum pictum leaf on the inflammation level of rectoanal tissue in hemorrhoid-induced rat. Open Vet. J.. 2026; 16(1): 384-395. doi:10.5455/OVJ.2026.v16.i1.35 Vancouver/ICMJE Style Krisnamurti DGB, Dalilati AZ, Prijanti AR, Nugroho CMH. The effect of topical and oral combination of ethanolic extract of Graptophyllum pictum leaf on the inflammation level of rectoanal tissue in hemorrhoid-induced rat. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 384-395. doi:10.5455/OVJ.2026.v16.i1.35 Harvard Style Krisnamurti, D. G. B., Dalilati, . A. Z., Prijanti, . A. R. & Nugroho, . C. M. H. (2026) The effect of topical and oral combination of ethanolic extract of Graptophyllum pictum leaf on the inflammation level of rectoanal tissue in hemorrhoid-induced rat. Open Vet. J., 16 (1), 384-395. doi:10.5455/OVJ.2026.v16.i1.35 Turabian Style Krisnamurti, Desak Gede Budi, Annia Zhafarina Dalilati, Ani Retno Prijanti, and Christian Marco Hadi Nugroho. 2026. The effect of topical and oral combination of ethanolic extract of Graptophyllum pictum leaf on the inflammation level of rectoanal tissue in hemorrhoid-induced rat. Open Veterinary Journal, 16 (1), 384-395. doi:10.5455/OVJ.2026.v16.i1.35 Chicago Style Krisnamurti, Desak Gede Budi, Annia Zhafarina Dalilati, Ani Retno Prijanti, and Christian Marco Hadi Nugroho. "The effect of topical and oral combination of ethanolic extract of Graptophyllum pictum leaf on the inflammation level of rectoanal tissue in hemorrhoid-induced rat." Open Veterinary Journal 16 (2026), 384-395. doi:10.5455/OVJ.2026.v16.i1.35 MLA (The Modern Language Association) Style Krisnamurti, Desak Gede Budi, Annia Zhafarina Dalilati, Ani Retno Prijanti, and Christian Marco Hadi Nugroho. "The effect of topical and oral combination of ethanolic extract of Graptophyllum pictum leaf on the inflammation level of rectoanal tissue in hemorrhoid-induced rat." Open Veterinary Journal 16.1 (2026), 384-395. Print. doi:10.5455/OVJ.2026.v16.i1.35 APA (American Psychological Association) Style Krisnamurti, D. G. B., Dalilati, . A. Z., Prijanti, . A. R. & Nugroho, . C. M. H. (2026) The effect of topical and oral combination of ethanolic extract of Graptophyllum pictum leaf on the inflammation level of rectoanal tissue in hemorrhoid-induced rat. Open Veterinary Journal, 16 (1), 384-395. doi:10.5455/OVJ.2026.v16.i1.35 |