| Research Article | ||
Open Vet. J.. 2025; 15(10): 5294-5303 Open Veterinary Journal, (2025), Vol. 15(10): 5294-5303 Research Article Investigation of the parasite Sarcocystis spp. in rabbit by conventional and molecular methodsEnas Saadi Hussein1*, Eman Daham Hadi2 and Sura Salim Aghwan11Department of Microbiology, College of Veterinary Medicine, University of Mosul, Mosul, Iraq 2Department of Internal and Preventive Medicine, College of Veterinary Medicine, University of Mosul, Mosul, Iraq *Corresponding Author: Enas Saadi Hussein. Department of Microbiology, College of Veterinary Medicine, University of Mosul, Mosul, Iraq. Email: bachtar.bakrie [at] brin.go.id Submitted: 04/03/2025 Revised: 25/08/2025 Accepted: 08/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
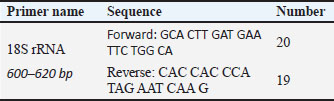
AbstractBackground: Sarcocystosis is an intracellular protozoan infection that causes mild gastrointestinal signs such as diarrhea and nervous signs such as Seizures, hind-limb weakness, circling behavior, and stargazing in many different hosts that result from contamination of grass and water with feces of final hosts. Aim: To diagnose the infection with Sarcocystis spp. In imported and local rabbits in Mosul City, Iraq. Methods: For the estimation, the incidence of Sarcocystis spp. in rabbits in Mosul, Iraq. Specimens were collected from rabbits across October 2023 and March 2024. All muscle samples were analyzed using classic microscopically examination, enzymatic digestion (pepsin and trypsin), staining with Giemsa, and molecular detecting. Results: Macroscopic and microscopic examination of the infected tissue revealed the presence of tissue cysts containing bradyzoites within the muscles. The overall infection rate was 70.1%. Both male and female are similarly vulnerable to Sarcocystis spp. infection, despite some differences in percentages. 2–4 months old Showed had the highest infection rate (47.5%) while 2 years old infection rate reach to (35%), and 1–1.5-year-olds had the lowest rate (17.5%). Six positive samples for pepsin and trypsin digestion technique were selected for for extraction of DNA and restrained to polymerase chain reaction (PCR) utilizing definite primers for exaggeration of sectional fragment of nucleotide sequence of the small ribosomal RNA gene (18SrRNA). The sequences were registered in National Center for Biotechnology Information (NCBI) and then compared with gene sequence data from NCBI by means of phylogenetic tree analysis, which revealed sequence identity with NCBI-BLAST, Sarcocystis halieti (No. EES1: LC830205), Sarcocystis japonica (No. ESS2: LC830206), Sarcocystis speeri (ESS3 LC830207) and Sarcocystis zamani (ESS4 LC830208), confirming the results with isolates from Norway, Japan, and Australia. Conclusion: Our study indicated that the PCR is the most accurate diagnostic test used to confirm the diagnosis of S. halieti, S. japonica, S. speeri, and S. zamani isolates, revealing genetic diversity and classification. The phylogenetic tree analysis demonstrated similarities between local and registered isolates, indicating potential genetic similarities. Keywords: Rabbits, Molecular, Sarcocystosis, Mosul, Iraq. IntroductionSarcocystosis is an infectious disease caused by the sarcocystis genus (Levine, 2018). It is one of the most ordinary pathogenic parasites for a spacious assortment of hosts, including humans and various farm animals (Fazly Ann et al., 2014; Taylor et al., 2016). The sexual phase (gametogenesis) occurs in intestines of the carnivorous ultimate host, while the asexual phase (cell division) arises in the muscles of the herbivorous intermediate host. During the schizogony phase, the cysts are called sarcocysts or Miescher’s tubes, as they become visible in the skeletal muscle fibers Özbek and Özbek (2000). Acute sarcocystosis in animal intermediate hosts causes encephalitis, myelitis, and a predisposition to hemorrhage. In pregnant animals, it can cause fetal death, preterm birth, and abortion (Tenter, 1995; Caspari et al., 2010; Fadel and Mustafa, 2023). Mild and chronic sarcocystosis leads to reduced weight and fur quality (Tenter, 1995; Mohammed. et al., 2022). Moreover, noteworthy behavioral deviations have been detected in affected animals (Reiner et al., 2009). Chronic cases of the disease result in substantial economic losses, particularly in meat production due to cyst formation (Mustafa et al., 2021; Fadel and Mustafa, 2023). Rabbits infected with Sarcocystis naturally manifested a neurological indication, the supreme prevelance of which is ataxia, giving them a “drunken” exterior. Infected rabbits may stagger when hopping, tremors assay, and possibly will collapse. Other indications take in posterior limb impairment, seizures, enclosure manners, revolving, and stargazing. These indications can gradually go downhill after a while, making initial findings and cure substantial (Fadel and Mustafa, 2023). Sarcocystis may infected and spreader in many animals firstly the rabbits, posing public health risks as a food source. They can also act as an infection reservoir for cats and carry environmentally resistant oocysts (Motamedi et al., 2011; Hussein and Shukur, 2020 a,b). For years, interspecies diagnosis of the Sarcocystis genus relied on electron microscopy due to the absence of visible differences between species under accessible magnification, making light microscopy ineffective (Mohammed et al., 2022). In many cases, serological diagnostic methods lack sensitivity. Various molecular procedures have been devised and employed to discover, recognize Sarcocystis species, and evaluate genetic variety across different hosts. Today, molecular diagnostics are the most commonly used, cost-effective, and time-efficient methods worldwide for distinguishing species (Stojecki et al., 2012; Mohammed et al., 2022; Mohammed et al., 2024). The study objective is to identify Sarcocystis species infecting rabbits in Mosul city using traditional and molecular techniques [polymerase chain reaction (PCR), sequencing, and phylogenetic analysis] to improve disease control, especially attributable to the dearth of investigation to Sarcocystis in rabbits. Materials and MethodsAnimals and sample collectionFifty-seven wild and domestic rabbits (Belgian Rabbit, British Dwarf Rabbit, and Himalayan Rabbit), aged between 3 and 4 weeks, were bought from commercial markets in Mosul, Iraq. Internal organs (heart, lungs, liver, spleen, esophagus, and skeletal muscles) were collected from each rabbit after their slaughtered and forwarded to the Parasitology Laboratory at the Veterinary Medicine College, Mosul University. Tissue samples obtained from each organ were inspected macroscopically for detected the cysts (Odening 1996; Motamedi et al., 2011; Fazly Ann et al., 2014). Enzyme digestion and microscopic examinationA modified version of the Dubey et al. (1989) method was utilized for muscle digestive, as described below: 100 g of a muscle mixture was crushed then digestedby enzymes for 30 minutes at 37°C in 100 ml of digestive medium containing 1.3 g of pepsin (Merck), 3.5 ml HCl, and 2.5 g of sodium chloride in 500 ml of distilled water. Trypsin digestion: The organs were digested with trypsin as described by (Dubey, 1989; Motamedi et al., 2011) After digestive, the mixing was centrifuged for 3 minutes at 3500 rpm and the sediment was stained using Gimsa stain and examined microscopically for parasite identify (Fazly Ann et al., 2014); after that washed two time for DNA extraction. Trypsin digestion of cattle diaphragm muscles had been accomplished in accordance with the optimized protocols of (Chiesa et al., 2013; Verma et al., 2017) Ensuing trypsin digestion of 20 g each in 50 ml of digestion solution [trypsin 1:250, 2.5 g/l phosphate buffered saline (PBS)] for 16 hours at 37°C in an incubator with stirring, the suspension was centrifuged for 5 minutes at 7,000 rpm, following which the pellet was resuspended in 5 ml PBS and centrifuged for 3 minutes at 5,000 rpm. The resulting pellet was resuspended in 5 ml PBS, and 300 µl of the suspension was used for DNA extraction. DNA extractionDNA was obtained from Sarcocystis-infected tissues via the Geneaid DNA extraction kit. To migrate and detect the DNA, 1% agarose gel was prepared. For this concentration, 0.5 g of agarose powder was dissolved in 50 ml of 1X TBE, and 3 ml of safe red dye were added. This was done utilizing a warmth provenance using constant stirring till ebullition, then drop it till cool down near a temperature of 50°C–60°C before proceeding with the next experiment (Mohammed et al., 2024). Diagnosis of Sarcocystis spp. by molecular reactionThe DNA diluted by using TE buffer for the reaction; the gene primers from (Geneaid) were mixed with the pre-prepared components in small tubes and ended the volume to 20 µl using distilled water. At that time, the solution mixed for 3–5 seconds to prepare it for the reaction. A thermocycler was used to perform the reactions, following a specific program for each reaction. The samples were placed in wells containing agarose gel with DNA and subjected to electrophoresis and a procedure that separates molecules according to electrical charge and size, for approximately 60 hours (Hamidinejat et al., 2014; Pestechian et al., 2021). Molecular detection for the Sarcocystis spp.The nucleotide sequences represents and types of primers at the molecular discovery of Sarcocystis spp. (Table 1). By using universal primer during amplification, the intention region is the 18S rRNA. Based on this, the sarcocyst was diagnosed. For the reaction, 4 µl added of DNA along with 1 µl of each primer to the reaction mixture. Table 1. The nucleotide sequences represents and types of primers at the molecular discovery of Sarcocystis spp.
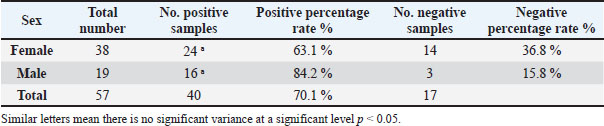
The steps show for amplifying 18S rRNA, detailing each stage of the reaction, including denaturation, annealing, and final extension to ensure amplification. The tubes were placed in a thermocycler to perform the reactions as described (Hamidinejat et al., 2014; Pestechian et al., 2021): Initial denaturation: At 95°C for 6 minutes to disruption the hydrogen bonds in the DNA and separate the strands; Repeated denaturation (35 cycles): Performed at 95°C for 45 seconds. This stage is crucial to keep the strands separated for effective binding; Annealing (Repeats 35 times): After denaturation, the reaction proceeds with annealing at 58°C for 1 minute, allowing the primers to collabrate specifically to their supplementary sequences in use single-stranded DNA; Extension (Repeats 35 times): The extension step follows, where the heat is elevated near 72°C used for 1 minute. At this best heating, Taq polymerase manufactures new DNA strands by increment nucleotides to the primer-bound regions; Final Extension: After completing 35 rotations, an eventual extension next to 72°C used for 5 minutes, assure that any remaining single-stranded DNA is fully extended. This detailed PCR program ensures the efficient amplification of the target 18S rRNA region, facilitating the molecular identification of Sarcocystis spp. Nucleotide sequencing of amplified fragments based on DNA sequencing technologyThe nucleotide sequences of the specimen (Sarcocystis spp.) under study were determined. The PCR products of the aforementioned sampling, along with the primers for the resulting fragment, were sent for sequencing using the Psomagen device in the USA. The gene sequences were compared with documented gene sequences in the National Center for Biotechnology Information (NCBI), and the outcomes were investigated via the BLAST program. Following this, the NCBI BLAST (BLASTn) from NCBI (http://www.ncbi.nlm.nih.gov) was used to compare the sequences with other sequences that were available in GenBank. With MEGA11 software, the Likelihood method on the Tamura-Nei model and bootstrap analysis with 1,000 resamplings. Statistical analysisChi-square tests were calculated using SPSS version 19 to determine the divergence in the pervasiveness of Sarcocystis spp. Variances with a p value <0.05 were measured statistically significant. Ethical approvalEthical approval was gained from the Ethics Committee or the Academic Committee of the Microbiology Department, Veterinary Medicine College, Mosul University, on October 1, 2023. ResultsThe Table 2 shows the infection rate of Sarcocystis spp. in rabbits from Mosul city, categorized by the gender of the rabbits. A whole of 57 rabbits were tested, with 40 rabbits 70.2% testing positive for infection. Table 2. The infection rate with Sarcocystis spp. in rabbits in Mosul city according to the sex of rabbit.
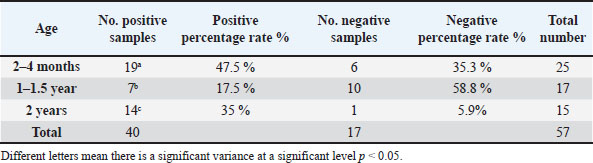
Out of 38 females, 24 were infected, representing an infection rate of 63.1%. While 19 male rabbits, 16 were infected, with a lower infection rate of 84.2%. The table also notes that similar letters indicate no significant variance at the significance level of p < 0.05, meaning that while there are numerical differences between male and female infection rates, they are not statistically significant. This implies that both male and female rabbits in Mosul city are similarly vulnerable to Sarcocystis spp. infection, despite some differences in percentages. Table 3 displays the transmission percentage of Sarcocystis spp. in rabbits from Mosul city according to their age groups. The data includes 40 infected rabbits, with the following age-related infection trends: (2 years old). This group had the highest infection rate, with 14 out of 15 rabbits (93.3%) testing positive. While the group (2–4 months old) showed an infection rate of 76%, with 19 out of 25 rabbits infected. And the lowest infection rate was observed in this group (1–1.5 years old), where 7 out of 17 rabbits (41.1%) were infected. Table 3. The infection rate with Sarcocystis spp. in Mosul city according to the age of rabbit.
Table 3 indicates a significant variation in infection rates across different age groups, as shown by the different letters (a, b, c, d) associated with each group. The p < 0.05 means that the age of the rabbits significantly influences the likelihood of infection, with older rabbits (2 years) being more prone to infection than younger ones. Table 4 illustrates the results of enzyme digestion and the detection of parasites in rabbit tissues from Mosul city. Pepsin and trypsin enzymes were used to carry out the digestion, and the outcomes indicate a substantial variation in each enzyme’s ability to identify bradyzoites stage of Sarcocystis species in the infected rabbits. Table 4. The tissues digestion with the digestive enzymes.
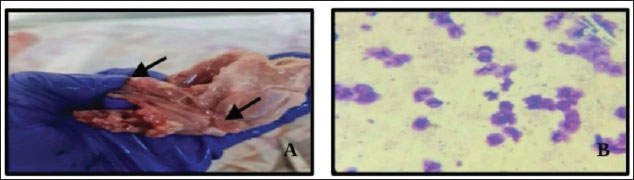
Pepsin enzyme treatmentA parasite test was performed on all 57 tissue samples (100%) that were subjected to pepsin treatment. Trypsin enzyme treatementOnly 13 out of 57 tissue samples (32.5%) treated with trypsin showed positive results for the presence of parasites This implies that trypsin was less effective compared to pepsin in identifying the parasites. The different letters (a, b) point toward a significant variance between the results of pepsin and trypsin enzyme treatments at a significance level of p < 0.05. This means that pepsin is significantly more effective in detecting Sarcocystis spp. in the infected rabbit tissues than trypsin. Macroscopic examination of the infected tissue samples discovered the presence of cysts in the tissues containing bradyzoites within the muscles. Internal to the rabbit body, the cysts can reach bigger than 5 mm in length (Fig. 1). (A) Macroscopic examination of the infected tissue samples revealed the attendance of tissue cysts containment bradyzoites within the muscles, appearing as banana-shaped bradyzoites. The average length of the bradyzoite stage is 4–5 mm, with a width of 1.2 ± 1 mm. (B) Bradyzoites stage of sarcocystis spp. in rabbits was discovered in thin tissue smears stained with Giemsa and photographed at 100× magnification.
Fig. 1. Tissues revealed cysts containing bradyzoites within the muscle of rabbits. (A) Macrocysts in muscle. (B) Bradyzoites stage of sarcocystis spp in thin tissue impression stained with Giemsa at 100×. Figure 2 appears to show an agarose gel electrophoresis result, typically used to separate DNA fragments for analysis after a PCR reaction. The bands seen in each lane indicate the presence of amplified DNA fragments. The clear, distinct bands means a successful amplification of DNA, while variations in the intensity or clarity of the bands could provide insights into the quality or quantity of the DNA. In this context, the gel is likely being utilized to approve the occurrence of Sarcocystis species DNA after PCR amplification from infected rabbit tissue. The results would help identify which samples were successfully amplified for further sequencing or analysis.
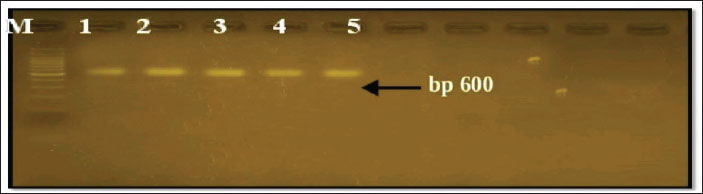
Fig. 2. DNA extraction from tissue samples. Figure 3 shows the PCR result, the amplification of Sarcocystis spp. DNA, specifically targeting the 18s rRNA gene region, resulting in a product size of 600 base pairs (bp).
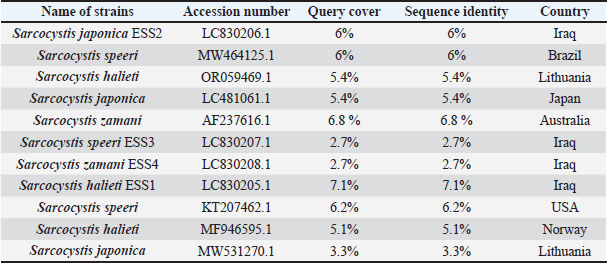
Fig. 3. PCR result for Sarcocystis spp. Based on the 18srRNA region and a reaction product of 600 bp.to the positive samples. The bands visible on the agarose gel indicate that the PCR successfully amplified the 600 bp fragment, which corresponds to the expected size for the 18s rRNA region of Sarcocystis spp. DNA. The presence of clear bands in the lanes indicate a positive detection of the parasite in the analyzed samples. The BLAST alignment result shows a 100% match between our query sequence and the reference sequence of Sarcocystis halieti (small subunit ribosomal RNA gene, GenBank accession: MH130211.1). All 600 base pairs are identical, with no gaps or mismatches. The alignment indicates a perfect match, as evidenced by the E-value of 0.0 and a bit score of 1,164, confirming a highly significant alignment. This submits that the organism in our sample is either S. halieti or a closely related species established on the 18S rRNA gene region. Phylogenetic Tree for Analyzing Sarcocystis spp.: S. halieti (No. EES1: LC830205), S. japonica (No. ESS2: LC830206), Sarcocystis speeri (ESS3 LC830207), and Sarcocystis zamani (ESS4 LC830208), established on 18SrRNA Gene Sequence in local rabbit isolates from Mosul city that shown in Table 5. Table 5. Homology using BLAST n between the local sequence of Sarcocystis.spp. (ESS1, ESS2, ESS3, ESS4) and other genotypes.
Construction methodAnalysis methodThe analysis was conducted using the Non-weighted Pair Grouping technique by way of Arithmetic Mean (UPGMA) in MEGA version 6.0. This method is typically used to construct phylogenetic trees based on genetic sequence data. Studied isolates
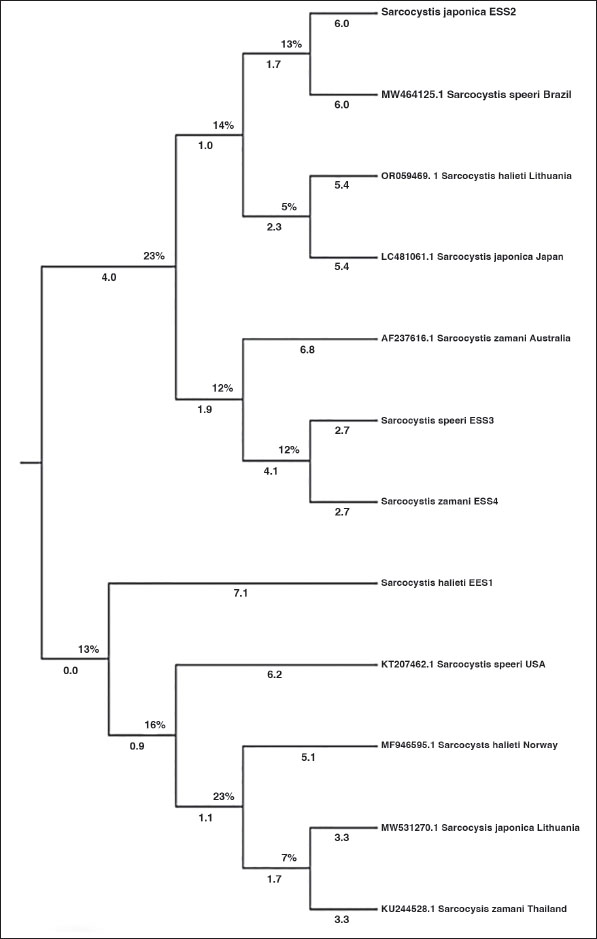
Analysis resultsGenetic diversityThe phylogenetic tree analysis indicates genetic diversity among local Sarcocystis isolates and other registered isolates in databases. Genetic Classification S. halieti (No. EES1: LC830205), S. japonica (No. ESS2: LC830206), S. speeri (ESS3 LC830207), and S. zamani (ESS4 LC830208). This analysis aids in the classification of genetic species, contributing to the understanding of biological diversity and the evolution of Sarcocystis spp. across different regions. Figure 4 will provide a visual representation of the phylogenetic tree, illustrating the relationships between different isolates based on the 18SrRNA gene sequence, which aids in understanding the kinship and genetic diversity among them.
Fig. 4. Phylogenetic tree analysis of Sarcocystis spp. established on 18SrRNA gene sequence in local rabbit isolates from Mosul city. DiscussionSarcocystosis is a usual parasitic disease that affects a broad variety of household animals, and some species can cause significant commercial damages by make happen both clinical and subclinical illness (Dubey et al., 2016; Konell et al., 2019; Mostafa et al., 2021a,b; Fadel and Mustafa, 2023). Sarcocystis can institute prominent into binary phases, with microscopic and large cysts able to pervade various structures of body in intermediate host (Faraj and Kawan, 2012; Rudaitytė-Lukošienė et al., 2018; Jawad et al., 2023). The study of “Sarcocystosis in Rabbits,” reviewed largely by Vandy Foss focused either on the cyst phase or its prevalence in rabbits established on detecting the cyst phase. Whilst microscopic and large cysts were set up in the muscles of cottontail rabbits (Crum and Prestwood, 1977; Pritt et al., 2012; Prakas et al., 2020) and demonstrated the clear presence of sarcocystic cysts in the muscles of wild cottontail rabbits. The outcomes discovered the coexist of microscopic cysts embedded in the esophagus (Shekarforoush et al., 2005). The study also displays the attendance of various microscopic cysts in various organs (Nasr et al., 2005; Serfilippi et al., 2020). The difference in cyst size could be attributed to the time of life of the cyst and its duration within the intermediate host. The characteristics of the bradyzoite (cystozoite) look like in dissimilar shapes and dimensions, resembling a banana figure with a slightly sharp anterior end and a curvy posterior end containing (Serfilippi et al., 2020; Ahmed and Albakri, 2021; Faraj et al., 2020 This study is consistent with other studies that showed the infection rate was around 75% in mice. Similarly, another study on rabbits reported the same result, as they were raised in cages used for cat breeding, leading to the transmission of infection to the rabbits, where the infection rate reached 100% (Munday et al., 1980; Hussein and Shukur, 2020 a,b). The results of some researchers showed different infection rates, with 50% of rabbits being infected with Sarcocystis (Hussein and Shukur, 2020 a,b). Another study discovered that 36% of rabbits had indirect antibodies (Černa et al., 1981; Serflippe et al., 2020). This result is consistent with another study that showed the presence of infection in the brain and muscles (Alsaadi et al., 2020). The molecular PCR method successfully diagnosed Sarcocystis spp. (600 bp). Some researchers have informed Sarcocystis spp. in different hosts using molecular process (Dakhil et al., 2017; Al-Saadi et al., 2020; Pestechian et al., 2021; Hamidinejat et al., 2014; Alfalahy et al., 2022) reported Sarcocystis in sheep in Karbala province Al-saadi (2020) and that Sarcocystis tenella and Sarcocystis arieticanis are the supreme major pathogenic cysts formed within sheep. Dakhil et al. (2017) and van Praag (2014) registered the total averages of S. fusiformis in addition S. Moulei in ruminant. The results of this molecular study may differ from those of other molecular studies due to the different methods used for extracting genetic DNA (Faraj et al., 2019; 2020), the several elements affecting amplicon manufacture, the obscurity of the parasite in the body structures, otherwise its lose owing to chilly and repeated study. The inspector’s expertise and the quantity of specimen also possess a significant effect continuously the infection percentage. DNA sequencing in addition to phylogenetic investigation are among the supreme important methods for classifying different pathogenic infection species and comparing the pathogen strain (Barham et al., 2005; Dubey et al., 2006) whether it is a parasite, virus, bacteria, otherwise other organism, by way of comparable besides various strains spreading worldwide (Dubey, 1980; Dubey et al., 2011; Jassim et al., 2021). At the current study, five PCR specimens are obtained, and four of them are confirmed and had been examined utilizing NCBI-BLAST assay of Sarcocystis strains then paralleled by means of additional Sarcocystis species. The sequencing in the gene bank provides insight into the new strain of Sarcocystis spp. in Iraq’s Mosul city, aiding in controlling this disease. The DNA sequencing results from rabbits from various regions of Mosul were examined using references to the 18SrRNA gene of the Sarcocystis parasite, including S. halieti, S. japonica, S. zamani, and S. speeri. Likeness and divergence between the Iraqi strain and strains from nearby nations and far-off regions of the world were revealed by the phylogenetic tree results. The results of the analysis showed a significant degree of similarity between the local isolates of Sarcocystis; isolates from Norway and Japan were shown to be closely related to isolates of S. halieti (No. EES1: LC830205) and S. japonica (No. ESS2: LC830206), respectively. The isolates shared a close relationship with Australian S. zamani (ESS4 LC830208) and S. speeri (ESS3 LC830207) isolates from NCBI-BLAST. This closeness and difference in outcomes between nations is caused by the variation in the degree of similarity between globally unique strains in populations distant from Iraq and isolated strains in Iraq: S. halieti, S. japonica, and S. zamani (Hamidinejat et al., 2014; Pestechian et al., 2021). Importance of the StudySpecies investigationThis research has significant implications for public health and agriculture, as Sarcocystis spp. may contribute to diseases in animals and humans. Contribution to databasesThese studies help improve genetic databases, facilitating future research in this field. ConclusionThis work contributes to our understanding of genetic species and their evolutionary interactions with other isolates by providing important information about the genetic diversity of local Sarcocystis isolates in Mosul. Further, examined isolates of S. halieti, S. Japonica, and S. zamani, demonstrating genetic variety and categorization. relationships between the local and registered isolates were shown by the phylogenetic tree analysis, suggesting possible genetic relationships. The potential implications of this work for public health and agriculture make it significant, since Sarcocystis spp. may be involved in disorders for humans and animals, as well as in its enhancement of genetic databases for future studies. For the first time, S. halieti, S. japonica, S. zamani, and S. speeri in rabbits in Iraq have been identified thanks to molecular study. AcknowledgmentsThe authors would gratitude the Integrated Laboratory for Research and Testing, University of Mosul, for provided the services rendered for this study. Conflict of interestThe authors declare that they have no conflict of interest. FundingThis study was supported by grants from the Directorate of Research, University of Mosul Thesis Recognition Grant 2024. Authors’ contributionsConceptualized and Supervision: E.S.H., E.D.H., and S.S.A, Methodology: E.S.H., S.S.A., Writing-original draft: E.S.H., S.S.A; Writing-review and editing: E.S.H., E.D.H., and S.S.A.; Visualized: E.S.H., E.D.H., and S.S.A. All authors have read and approved the final manuscript. Data availabilityAll data supporting the findings of this study are available within the manuscript, and no additional data sources are required. ReferencesAhmed, A.M. and Albakri, H.S. 2021. Phynotypic and genotypic identification of Eimeria species in backyard chicken in Nineveh governorate, Iraq. Iraqi J. Vet. Sci. 35(2), 41–46; doi:10.33899/ijvs.2021.130487.1834 Al-Amery, F. 2020. Microscopic and molecular diagnosis of Ascaridia spp. in domestic pigeons (Columba livia domestica) in Baghdad city, Iraq. Iraqi J. Agric. Sci. 51(4), 1220–1225. Al-Amery, A.M., Al-Waely, T.N. and Faraj, A.A. 2023. Phenotypic and genotypic study of sarcocystosis in Iraqi domestic goats (Capra hircus). Iraqi J. Vet. Sci. 37(Supplement I), 29–35. Jawad, H.H. and Jassem, G.A. 2023. Traditional, histopathological and molecular diagnosis of sarcocytosis in slaughtered sheep in Al-Diwaniyah province, Iraq. Iraqi J. Vet. Sci. 37(4), 871–875; doi:10.33899/ijvs.2023.138763 Alfalahy, R.I., Al-Amery, A.M. and Faraj, A.A. 2022. Molecular study of Oestrus ovis larvae infesting in sheep in Baghdad city. Iraqi J. V. Sci. 36(1), 41–45; doi:10.33899/ijvs.2022.135053.2438 Jassim, A., Alfatlawi, M.A., Jarad, N.I. and Klaif, S.F. 2021. Clinical and molecular identification of ruling Theileria annulata strains in cattle calves in Al-Diwaniyah province, Iraq. Iraqi J. Vet. Sci. 35(1), 115–119; doi:10.33899/ijvs.2020.126429 Al-Saadi, S.A., Al-Mussawi, K.A. and Muhammed, H.A. 2020. Molecular identification of Sarcocystis species infection in sheep in Karbala governorate-Iraq. Med. Legal Update. 20(1), 889–895; doi:10.37506/v20/i1/2020/mlu/194708 Barham, M., Stützer, H., Karanis, P., Latif, B.M. and Neiss, W.F. 2005. Seasonal variation in Sarcocystis species infections in goats in northern Iraq. Parasitology 130(2), 151–156. Caspari, K., Grimm, F., Kühn, N., Claire Caspari, N. and Basso, W. 2010. First report of naturally acquired clinical sarcocystosis in a pig breeding stock. Vet. Parasitol. 177, 175–178; doi:10.1016/j.vetpar.2010.11.019 Černa, Z., Louckova, M., Nedvdova, H. and Vavra, J. 1981. Spontaneous and experimental infection of domestic rabbits by Sarcocystis cuniculi Brumpt, 1913. Folia Parasitologica (PRAHA). 28, 313–318. Chiesa, F., Muratore, E., Dalmasso, A. and Civera, T. 2013. A new molecular approach to assess the occurrence of Sarcocystis spp. in cattle and products thereof: preliminary data. Ital. J. Food Saf. 2, 148–151. Crum, J.M. and Prestwood, A.K. 1977. Transmission of Sarcocystis leporum from a cottontail rabbit to domestic cat. J. Wildlife Dis. 13, 174 -175. Dakhil, H.G., Abdallah, B.H. and Abdallah, F.A. 2017. Molecular identification of Sarcocystis fusiformis and S. moulei infecting water buffaloes (Bubalus bubalis) in southern Iraq. World J. Pharm. Res. 6(3), 215–229; doi:10.20959/wjpr20173-8013 Dubey, J.P. 1980. Mouse pathogenicity of Toxoplasma gondii isolated from a goat. Am. J. Vet. Res. 41, 427–429. Dubey, J.P. and Lindsay, D.S. 2006. Neosporosis, toxoplasmosis, and sarcocystosis in ruminants. Vet. Clinics North Amer. Food Anim. Pract. 22(3), 645–671; doi:10.1016/j.cvfa.2006.08.001 Dubey, J.P., Moré, G., Van Wilpe, E., Calero-Bernal, R., Verma, S.K. and Schares, G. 2016. Sarcocystis rommeli, n. sp. (Apicomplexa: sarcocystidae) from cattle (Bos taurus) and its differentiation fromSarcocystis hominis. J. Euk. Microbiol. 63(1), 62–68; doi:10.1111/jeu.12248 Dubey, J.P., Passos, L.M., Rajendran, C., Ferreira LR Gennari SM. and Su, C. 2011. Isolation of viable Toxoplasma gondii from feral guinea fowl (Numida meleagris and domestic rabbits (Oryctolagus cuniculus) from Brazil. J. Parasitol. 97(5), 842–845; doi:10.1645/GE-2728.1 Dubey, J.P., Speer, C.A. and Charleston, W.A. 1989. Ultrastructural differentiation between Sarcocystis of Sarcocystis hirsuta and S. hominis. Vet. Parasitol. 34(34), 153–157. Faraj, A.A., Hade, B.F. and Al-Amery, A.M. 2019. Conventional and molecular study of Babesia spp. of natural infection in dragging horses at some areas of Baghdad city, Iraq. Iraqi J. Agric. Sci. 50(3), 909–915. Faraj, A.A. and Al- Amery, A.M. 2020. Microscopic and molecular diagnosis of Ascaridia spp. In Domestic Pigeons (Columba livia domestica) in Baghdad city, Iraq Iraqi J. Agric. Sci. 51(4), 1220–1225; doi:10.36103/ijas.v51i4.1101 Fadel, M.A. and Mustafa, K.A. 2023. The anti-inflammatory effect of allopurinol and diclofenac in chick’s model. Iraqi J. Vet. Sci. 37(3), 547–553; doi:10.33899/ijvs.2023.138108.2769 Faraj, A.A. and Kawan, M.H. 2012. Detection of Sarcocystosis in some wild birds. Iraqi J. Vet. Med. 3(2), 65–70. Fazly Ann, Z., Muhamad Syamsul Naim, W.O.B. Wan., Normaziah S Geethamalar. and Iswadi , M. Ramlan. Modified digestion technique for detection of sarcocystis bradyzoites. In Proceedings of the 1st ARCAP & 35th Malaysian Society of Animal Production (MSAP) Annual Conference, Riverside Majestic Hotel, Kuching, Sarawak, Malaysia, p 4. Hamidinejat, H., Moetamedi, H., Alborzi, A. and Hatami, A. 2014. Molecular detection of Sarcocystis species in slaughtered sheep by PCR–RFLP from south-western of Iran. J. Parasitic Dis. 38, 233–237. Hussein, S.N. and Shukur, M.S. 2020. In vivo experimental infection of sarcosporidiosis and toxoplasmosis of rabbits in Duhok Province, Kurdistan region, Iraq. Int. J. Res. Med. Sci. 8(10), 3467–347.4 Hussein, S.N. and Shukur, M.S. 2020. In vivo experimental infection of sarcosporidiosis and toxoplasmosis of rabbits in Duhok Province, Kurdistan region, Iraq. Int. J. Res. Med. Sci. 8(10), 3467–3474. Jawad, H.H. and Jassem, G.A. 2023. Traditional, histopathological and molecular diagnosis of sarcocytosis in slaughtered sheep in al-diwaniyah province, Iraq. 37(4), 871–5; doi:10.33899/ijvs.2023.138763.2835 Jassim, A., Alfatlawi, M. A., Jarad, N. I. and Klaif, S. F. 2021. Clinical and molecular identification of ruling Theileria annulata strains in cattle calves in Al-Diwaniyah province, Iraq. Iraqi J. Agric. Sci. 35(1), 115–119. Konell, A.L., Sato, A.P., Stival, M., Malaguini, N.P., Anjos, A.D., Ferreira, R.F. and Locatelli-Dittrich, R. 2019. Serosurvey of Toxoplasma gondii, Sarcocystis sp. and Neospora caninum in geese (Anser sp.) from urban parks and captivity. Rev. Brasil. Parasitol. 28(2), 221–228; doi:10.1590/S1984-29612019042 Levine, N.D. 2018, The Protozoan Phylum Apicomplexa; vol 2. CRC Press, Boca Raton, FL. Mohammed, N.H., Khalil, L.Y. and Suleiman, E.G. 2024. Detection of Sarcocystis spp in imported frozen and slaughtered chickens by different techniques in Mosul city. Iraqi J. Vet. Sci. 38(4), 925–932; doi:10.33899/ijvs.2024.150772.3719 Mohammed, R.G.,., Dragh, M.A. and M. H, H. 2022. First Molecular Identification of Sarcocystis Buffalonis in Goats Worldwide in Misan Governorate, Iraq. P. J. M. H. S. 16(10), 715; doi:10.53350/pjmhs221610 Mostafa, E.S., Alhayali, N.S. and Suleiman, E.G. 2021a. Pathological and molecular study of ovine diaphragms naturally infected by Sarcosystis spp. Iraqi J. Vet. Sci. 35(4), 749–755; doi:10.33899/ijvs.2021.128327.1570 Mostafa, E.S., Alhayali, N.S. and Suleiman, E.G. 2021b. Pathological and molecular study of ovine diaphragms naturally infected by Sarcosystis spp. Iraqi J. Vet. Sci. 35(4), 749–755; doi:10.33899/ijvs.2021.128327 Motamedi, G.R., Dalimi, A., Nouri, A. and Aghaeipour, K. 2011. Ultrastructural and molecular characterization of Sarcocystis isolated from camel (Camelus dromedarius) in Iran. Parasitology Res. 108(4), 949–954. Munday, B.L., Smith, D.D. and Frenkel, J.K. 1980. Sarcocystis and related organisms in Australian wildlife: iv. Studies on Sarcocystis cuniculi in European Rabbits (Oryctolagus cuniculus). J. Wildlife Dis. 16(2), 201–204. Nasr, S., Hussen, E. and Soad, M. 2005. Prevalence of sarcocystis spp. in sheep and goats and its effect on some blood constituents in sharkia province. Vet. Rec. 156(13), 418. Odening K.,Stolte M., Bockhardt I., 1996. “On the diagnostics of Sarcocystis in cattle: sarcocysts of a species unusual for Bos taurus in a dwarf zebu,” Veterinary Parasitology, vol. 66, no. 1-2, pp. 19–24. Özbek, A. and Özbek, E. 2000. Morphological and Histochemical Features of a Sarcocyst Located in the Skeletal Muscle of a Rabbit. Türkiye Parazitoloji Dergisi 24(3), 259–263. Pestechian, N., Yousefi, H.A., Kalantari, R., Jafari, R., Khamesipour, F., Keshtkar, M. and Esmaeilifallah, M. 2021. Molecular and microscopic investigation of Sarcocystis species isolated from sheep muscles in Iran. J. Food Qual. 2021, 1–6; doi:10.1155/2021/5562517 Prakas, P., Strazdaitė-Žielienė, Z., Januškevičius, V., Chiesa, F., Baranauskaitė, A., Rudaitytė-Lukošienė, E., Servienė, E., Petkevičius, S. and Butkauskas, D. 2020. Molecular identification of four Sarcocystis species in cattle from Lithuania, including S. hominis, and development of a rapid molecular detection method. Parasit. Vectors 13(1), 610; doi:10.1186/s13071-020-04473-9 Pritt, S., Cohen, K. and Sedlacek, H. 2012. Parasitic diseases.In The laboratory rabbit, guinea pig, hamster, and other rodents. Suckow, M.A., Stevens, K.A. and Wilson, R.P Waltham, MA: Elsevier, pp: 415–46. https://doi.org/10.1016/B978-0-12-380920 Reiner, G., Hübner, K. and Hepp, S. 2009. Suffering in diseased pigs as expressed by behavioural, clinical and clinical-chemical traits, in a well-defined parasite model. Appl. Anim. Behav. Sci. 118, 222–231; doi:10.1016/j.applanim.2009.02.010 Rudaitytė-Lukošienė, E., Prakas, P., Butkauskas, D., Kutkienė, L., Vepštaitė-Monstavičė, I. and Servienė, E. 2018. Morphological and molecular identification of Sarcocystis spp. from the sika deer (Cervus nippon), including two new species Sarcocystis frondea and Sarcocystis nipponi. Parasitol. Res. 117(5), 1305–1315; doi:10.1016/j.parint.2018.08.006 Serfilippi, L.M., Saladino, B.H. and Spainhour Ch, B. 2020. Case Report Sarcocystis Infection in Laboratory Rabbits. Am. Assoc. For Lab. Anim. Sci. 2020. . 70(3), 300–301; doi:10.30802/AALAS-CM-19-000113 Shekarforoush, S.S., Razavi, S.M., Dehghan, S.A. and Sarihi, K. 2005. Prevalence of Sarcocystis pecies in slaughtered goats in Shiraz, Iran. Vet. Rec. 156(13), 418; doi:10.1136/vr.156.13.418 Stojecki, K., Karamon, J., Sroka, J. and Cencek, T. 2012. Molecular diagnostics of Sarcocystis spp. Infections. J. Vet. Scie. 15(3), 589–596. Taylor MA, Coop RL, Wall RL, 2016.Veterinary parasitology.,4th ed. Wiley Blackwell; doi:10.1002/9781119073680. Tenter, A.M. 1995. Current research on Sarcocystis species of domestic animals. Int. J. Parasitol. 25, 1311–1330; doi:10.1016/0020-7519 van Praag E. 2014. Toxoplasmosis, an unrecognized parasitosis in rabbits 2014.10.13140/2.1.3251.9368. Available via http://www.medrabbit.com/EN/NeurologyToxoplasma/Toxo_rab_en.pdf Verma, S.K., Lindsay, D.S., Grigg, M.E. and Dubey, J.P. 2017. Isolation, culture and cryo preservation of Sarcocystis species. Curr. Protoc. Microbiol. 45, 11–127. | ||
| How to Cite this Article |
| Pubmed Style Hussein ES, Hadi ED, Aghwan SS. Investigation of the parasite Sarcocystis spp. in rabbits by conventional and molecular methods. Open Vet. J.. 2025; 15(10): 5294-5303. doi:10.5455/OVJ.2025.v15.i10.46 Web Style Hussein ES, Hadi ED, Aghwan SS. Investigation of the parasite Sarcocystis spp. in rabbits by conventional and molecular methods. https://www.openveterinaryjournal.com/?mno=245757 [Access: January 24, 2026]. doi:10.5455/OVJ.2025.v15.i10.46 AMA (American Medical Association) Style Hussein ES, Hadi ED, Aghwan SS. Investigation of the parasite Sarcocystis spp. in rabbits by conventional and molecular methods. Open Vet. J.. 2025; 15(10): 5294-5303. doi:10.5455/OVJ.2025.v15.i10.46 Vancouver/ICMJE Style Hussein ES, Hadi ED, Aghwan SS. Investigation of the parasite Sarcocystis spp. in rabbits by conventional and molecular methods. Open Vet. J.. (2025), [cited January 24, 2026]; 15(10): 5294-5303. doi:10.5455/OVJ.2025.v15.i10.46 Harvard Style Hussein, E. S., Hadi, . E. D. & Aghwan, . S. S. (2025) Investigation of the parasite Sarcocystis spp. in rabbits by conventional and molecular methods. Open Vet. J., 15 (10), 5294-5303. doi:10.5455/OVJ.2025.v15.i10.46 Turabian Style Hussein, Enas Saadi, Eman Daham Hadi, and Sura Salim Aghwan. 2025. Investigation of the parasite Sarcocystis spp. in rabbits by conventional and molecular methods. Open Veterinary Journal, 15 (10), 5294-5303. doi:10.5455/OVJ.2025.v15.i10.46 Chicago Style Hussein, Enas Saadi, Eman Daham Hadi, and Sura Salim Aghwan. "Investigation of the parasite Sarcocystis spp. in rabbits by conventional and molecular methods." Open Veterinary Journal 15 (2025), 5294-5303. doi:10.5455/OVJ.2025.v15.i10.46 MLA (The Modern Language Association) Style Hussein, Enas Saadi, Eman Daham Hadi, and Sura Salim Aghwan. "Investigation of the parasite Sarcocystis spp. in rabbits by conventional and molecular methods." Open Veterinary Journal 15.10 (2025), 5294-5303. Print. doi:10.5455/OVJ.2025.v15.i10.46 APA (American Psychological Association) Style Hussein, E. S., Hadi, . E. D. & Aghwan, . S. S. (2025) Investigation of the parasite Sarcocystis spp. in rabbits by conventional and molecular methods. Open Veterinary Journal, 15 (10), 5294-5303. doi:10.5455/OVJ.2025.v15.i10.46 |