| Research Article | ||
Open Vet. J.. 2025; 15(10): 5304-5311 Open Veterinary Journal, (2025), Vol. 15(10): 5304-5311 Research Article Pathological investigation for effect of trichloroethylene on rat liverNoora Nadhim Ali* and Zainab J. Mohammed JawadDepartment of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Baghdad, Baghdad, Iraq *Corresponding Author: Noora Nadhim Ali. Department of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Baghdad, Baghdad, Iraq. Email: Noura.ali2207p [at] covm.uobaghdad.edu.iq Submitted: 17/03/2025 Revised: 27/08/2025 Accepted: 15/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
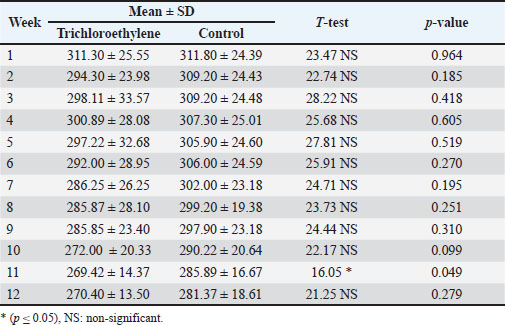
AbstractBackground: Trichloroethylene, a halocarbon solvent commonly used as a degreaser, is a major environmental hazard. Recent research reveals that continuous exposure to trichloroethylene (TCE) contributes to the development of liver injury. TCE toxicology is particularly important because the liver is one of the most sensitive organs to toxic chemicals due to its metabolic processes. Aim: To investigate the potential harm that TCE might have caused to the liver of rats. Methods: 20 Healthy adult male Wister albino rats were divided into two groups. Each group had 10 rats. One group was treated with TCE (0.2 ml/100 gm) [B.W of rat] orally administered by gavage to induce liver injury. The control group was given corn oil for 12 weeks. The rats were sacrificed after a 12-week treatment period, blood samples were taken to separate serum for liver enzymes test, and liver tissue samples were fixed immediately in 10% neutral-buffered formalin. Results: Clinical symptoms, body weight changes, liver enzymes, and liver pathology (macro and micro) observed were examined. In terms of body weight, the control group increased significantly more than the TCE group, which decreased. According to the liver enzyme results, there was no significant increase in ALP but a considerable increase in AST and ALT enzymes. The results of liver pathology showed that the TCE group caused severe fatty changes of hepatocytes in the form of cytoplasmic lipid droplets, degenerative changes with proliferation of hepatocytes with necrosis, hyperplasia of the bile ducts, constricted central vein, proliferation of Kupffer cells, apoptotic cells, and dilated sinusoids, while the control group experienced mild degenerative changes of hepatocytes. The current findings demonstrated the extensive damage that TCE caused to the liver tissue and enzymes. Conclusion: TCE caused hepatic damage in male rats after a long duration of exposure. Keywords: Trichloroethylene, Pathology, Liver, Male rats. IntroductionTrichloroethylene (TCE) is a widely used organic solvent in a variety of commercial and industrial fields. It is used as an intermediate chemical in the production of chemical compounds such as polyvinyl chloride, in the automotive and metal industries, as well as in adhesives, lubricants, paints, varnishes, paint removers, insecticides, cold steel cleaners, grease, oil, strong fats, wax, and tar, in the fabric and aerospace industries, and as a coolant for thermo-transfer (ATSDR, 2019; Slunge et al., 2022). The Environmental Protection Agency (EPA) has recognized TCE as a widespread environmental pollutant, and it was listed sixteenth on the U.S. Agency for Toxic Substances and Disease Registry’s Priority List of Hazardous Substances. (Chiu et al., 2013; EPA, 2017). The air pollution levels in metropolitan areas are typically below 1 µg/m³, while contaminated sites might have significantly higher levels. The U.S. EPA reports that indoor air concentrations are typically below 2 µg/m³, while occupational exposures can be much higher (EPA, 2011). Groundwater concentrations can range from less than 1 µg/l to thousands of µg/l in contaminated places. TCE has been identified as a major contaminant in industrial groundwater by the EPA (2016). The maximum contamination limit (MCL) for TCE in drinking water is 5 µg/l, according to the EPA (2016). Halogenated organic solvents, including TCE, are rarely disposed of or buried in soil in the United States and many other countries. However, TCE is widely used and causes human exposure through inhalation of polluted air at work, skin contact with contaminated materials, and consumption of TCE-contaminated food and drink. Consequently, there is a constant risk to human health (Mayans et al., 2021). Because of its widespread use, TCE is one of the most important environmental contaminants that can introduce both occupational and public health problems (Lash, 2024). TCE has been used extensively and for a long time, making it a prevalent environmental pollutant that can be found in food, groundwater, and oxygen (Wu and Schaum, 2000; Horzmann et al., 2020). There is a lot of interest in the possible toxicological effects of TCE, a solvent that is used extensively in industrial applications, especially with regard to liver health. Understanding the histopathological effects of TCE in rat models is essential for deciphering the intricacies of hepatic injury because TCE is predominantly processed in the liver. Because of their physiological resemblance to humans, rats are frequently used in toxicological studies, making them an excellent tool for evaluating the effects of TCE exposure. As a key organ in detoxification, the liver experiences some pathological alterations in response to xenobiotic injury. Therefore, by using histological analysis to examine liver tissue, researchers may spot particular changes such as inflammation, necrosis, and fibrosis, which help to clarify the mechanisms by which TCE works (Kakodia et al., 2024). Biochemical and histological changes are part of the complex underlying mechanisms of liver damage caused by TCE exposure in rats. Hepatocyte damage is mostly caused by TCE metabolites, especially dichloroacetate, which cause oxidative stress and the production of reactive oxygen species (Famurewa et al., 2023; Kakodia et al., 2024). TCE induces liver cancer in mice but not in rats (Yang et al., 2023). The liver is an essential organ for vertebrate metabolism (Shuai et al., 2016; Luo et al., 2022). Although the molecular mechanism of TCE-induced liver damage remains unknown, prolonged exposure to TCE can result in significant liver damage (Zhang et al, 2018). Systemic circulation, or the gastrointestinal portal vein, is essential for the transformation and detoxification of substances that enter the body. As a result, the hazardous compounds included in these chemicals may have harmful effects on the liver (Woolbright and Jaeschke, 2017). Therefore, this study aimed to evaluate the hepatotoxic effects of TCE, identify histopathological alterations caused by TCE exposure, and understand the possible processes of TCE-induced liver damage. Material and MethodsExperiment animalsAdult Wister Albino male rats were used in this study (n=20) with age of about two to three months and body weight ranged between 120 and 150 g. This study was conducted at the “University of Baghdad - College of Veterinary Medicine”, where the animals were bred two weeks before the study. The animals were housed in cages with dimensions of 20*30*50 cm3 and an average of five rats per cage with wood shared for bedding. To allow them to acclimate to the ideal breeding conditions of 22°C ± 3°C with a 12/12 hour (light/dark) cycle. Feed pellets and drinking water were freely accessible for the animals and were provided throughout the duration of the experiment (Hafes, 1970; Mustafa and Zainab, 2024). No significant difference was observed in food and water intake between the control and TCE groups. Experimental DesignFirst experiment: (Acute toxicity):10 adult male rats were used for the LD50 study for TCE using the Up and down method (Dixon, 1980). Second experiment: The effect of chronic exposure to TCE in 20 adult male rats for 90 days was divided into two groups as follows: Group 1: 10 adult rats treated orally and daily with corn oil as Control negative group by gavage needle. Group 2: 10 adult rats treated orally then TCE ( 110 LD50, daily for 90 days. Blood samples were collected after 3 months at the end of the experiment. Cardiac punctures were used to collect blood samples from animals anesthetized (chloroform) by inhalation (Hameed and Hassan, 2022; Khalaf and Salih, 2023). Blood sample (2 ml) from each group. Kept in a cool place prior to centrifugation for 15 minutes at 3,000 rpm. Serum was stored in an Eppendorf tube at −20°C. Until used for estimation of liver enzyme levels by Spectrophotometers Al-Kurdy and Kazaal (2024). Preparation of TCE doseThe TCE dose was determined using the Dixon approach using the following equation: LD50=xf + kd Xf=last dose administrated K=value from appendix D=difference between dose levels (Dixon, 1980). TCE was freshly prepared by being dissolved in distilled water, diluting 1 ml of TCE (1 Emulsifiable Concentrate) in 99ml of distilled water, where doses were given for 10 adult male rats. This test was conducted by sequentially administering a single dose to each animal at intervals of 24 h. Animals that died within 24 hours after showing clinical signs of poisoning were included. According to a predetermined dose progression factor, the dose was decreased after each death and increased after each survival. Determination of serum ALT activity (mg/dl)Kinetic determination of alanine aminotransferase (ALT) activity according to (Gella et al., 1985; Young, 1997). Determination of serum AST activity (mg/dl)Kinetic determination of aspartate aminotransferase (AST) activity according to the method of Gella et al. (1985), Ng (2002). Determination of serum alkaline phosphatase (ALP) activity (mg/dl)The activity of Alkaline phosphatase (ALP) was measured according to (Tietz et al., 1983; Burtis and Ashwood, 1999). Processing and histopathological examination of liver tissueRats were immediately scarified at the end of the experiment, after being given chloroform anesthesia. The liver samples were fixed in 10% neutral-buffered formalin and prepared for histology in line with a standardized procedure (Bancroft and Gamble, 2008). The fixed tissues were quickly placed under water to remove formaldehyde, to using increasing concentrations of alcohol, cleared by xylene, and dipped in paraffin wax by histokinate. Finally, the tissues were cut into 4–5 mm sections using a microtome, fixed on the slides, and stained with hematoxylin and eosin. The slides were examined under a light microscope, and photomicrographs were taken for control and treatment groups. Determination of body weightEach animal’s body weight was measured before the study began, once a week while it was underway, and before it was sacrificed. The body weight of each animal was recorded during the last several days of the exposure. The proportion of body weight before and after TCE exposure. Statistical analysisThe Statistical Packages for the Social Sciences (SPSS) (2019) program was used to detect the effect of different groups in the study parameters. The T-test was used to significantly compare between the means in this study. Ethical approvalEthical approval was granted through the local committee of the animal care and use at the College of Veterinary Medicine, University of Baghdad (Number 1388 / P.G at 2972024). ResultsClinical singsThe overall appearance, eating, drinking activities, and movement of the TCE group showed reduced eating and drinking after 4 weeks of experiment, and showed roughness and change in hair color and emaciation. Body weightThe TCE group showed statistically significant decrease (p ≤ 0.05) in body weight during 12 weeks compared with the control group (Table 1). Table 1. Comparison between and control groups in Weight with difference weeks.
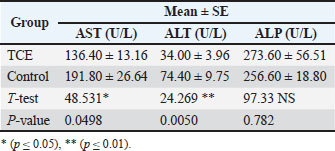
Liver enzymesIn Table 2, the results show significant increased (p ≤ 0.05) in the mean AST levels between the groups. The TCE group (136.40 U/l) was compared with the control (191.80 U/l), whereas the mean level of ALT level of the TCE group (34.00U/l) significantly increased compared with the control group (74.40 U/l), showed high significant increased (p ≤ 0.01), while the results of ALP revealed non-significant change between the TCE and control groups. Table 2. Comparison between the TCE and the control groups in Liver enzyme.
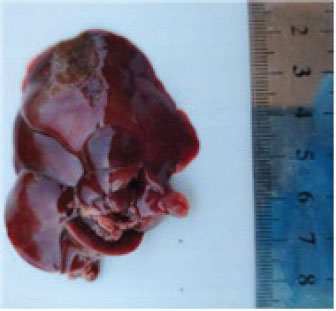
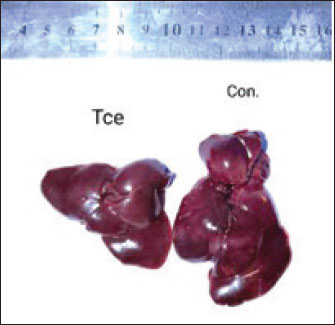
Pathological changesGross appearanceAt the end of the experiment, the animals were sacrificed for the gross changes observed, which are shown in Figure 1. The liver of the TCE group showed atrophy and dark congested area, while at Figure 2, the liver showed atrophy and congestion of the TCE liver group compared with the control group.
Fig. 1. liver atrophy and dark congested area of TCE group.
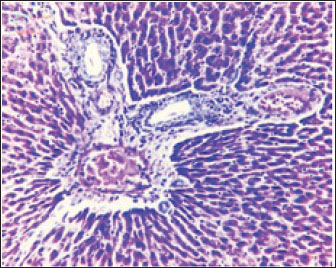
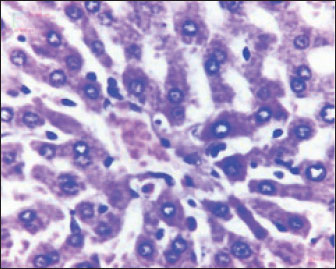
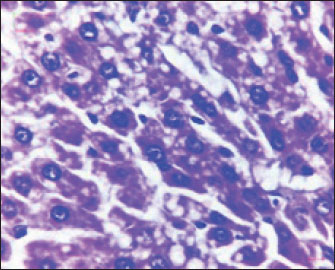
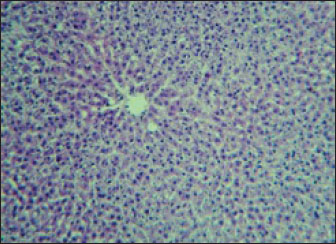
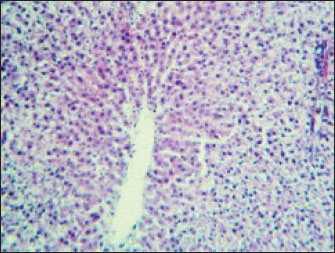
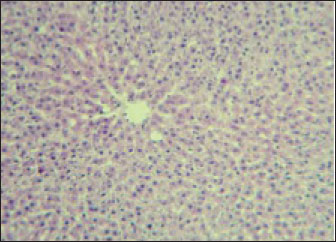
Fig. 2. Liver showed atrophy and congestion in the TCE group compare with the control group. Histopathological change in the liverThe histopathological results in the TCE group showed that the treatment with TCE for 90 days ( 110 LD50daily) resulted in the infiltration of inflammatory cells, mainly lymphocytes, in portal area with mild fibrosis, hyperplasia of the bile duct (Fig. 3) showed congested central vein, proliferation of Kupffer cells, apoptotic cells and, with dilated sinusoids, while the (Fig. 4) showed severe fatty changes of hepatocytes in the form of cytoplasmic lipid droplets (microvascular steatosis) with proliferation of Kupffer cells. (Fig. 5) showed degenerative changes with necrosis of hepatocytes with proliferation also (Fig. 6). Histopathological section of the liver (group + with oil) showing mild degenerative changes of hepatocytes. In Fig. 7, the histopathological section of the liver (c +group/90 days) showing mild degenerative changes of hepatocytes with few inflammatory cells infiltration (Fig. 8). Finally, in Fig. 9, histological section of the liver (control group) with normal structure.
Fig. 3. Histopathological section of the liver (TCE group/90 days) showing infiltration of inflammatory cells, mainly lymphocytes, in the portal area with mild fibrosis and hyperplasia of the bile duct (H&E stain 100X).
Fig. 4. Histopathological section of the liver (TCE group, 90 days) showing congested central vein, proliferation of Kupffer cells, apoptotic cells and dilated sinusoids (H&E stain 400X).
Fig. 5. Histopathological section of the liver (TCE group/90 days) showing severe fatty changes in hepatocytes in the form of cytoplasmic lipid droplets (microvascular steatosis) with proliferation of Kupffer cells (H&E stain 400X).
Fig. 6. Histopathological section of the liver (TCE group/90 days) showing degenerative changes with necrosis of hepatocytes and proliferation of Kupffer cells ( H&E stain 400X).
Fig. 7. Histopathological section of the liver (group + with oil) showing mild degenerative changes in hepatocytes (H&E stain 100X).
Fig. 8. Histopathological section of the liver (c + group/90 days) showing mild degenerative changes of hepatocytes with few inflammatory cell infiltrations (H&E stain 100X).
Fig. 9. Histological section of the liver (control group) with normal stricture (H&E stain 10X). DiscussionExposure of rats to TCE modifies their liver enzymes through a variety of intricate biochemical processes that are largely fueled by metabolites of the chemicals’. TCE undergoes metabolism to produce a number of metabolites, such as dichloroacetic acid (DCA) and trichloroacetic acid (TCA), both of which have been linked to liver carcinogenesis and the accompanying changes in enzymes. Hepatotoxicity may be exacerbated by the potential of these metabolites to increase oxidative stress and trigger inflammatory reactions. According to research, DCA and TCA cause hepatocyte damage by interfering with regular cellular communication pathways. This ultimately results in notable alterations in liver enzyme function, which is essential for detoxification procedures (Capinha, 2024). The observed decrease in eating and drinking, as well as emaciation and hair roughness after 4 weeks of exposure, suggests systemic toxicity and overall health deterioration in the experimental subjects, most likely due to the effects of TCE. Yoo et al. (2008) found that exposure to TCE induced weight loss, decreased food intake, and neurobehavioral abnormalities in experimental rats, indicating systemic toxicity. Abdel-Mageed et al. (2012) reported hair and skin alterations linked to systemic metabolic disturbances in rats exposed to specific toxins. Shipp et al. (2010) described how systemic toxins can cause weight loss and metabolic abnormalities, which are frequent in toxicological investigations involving chemical exposure. Body weight reductionIn this experiment, body weight showed significant decrease at the end of the experimental period after TCE exposure in the G1 is it in this study compared with the control group (G2). The results of this study agreed with a previous study (Stermer et al., 2019). Moreover, continuous exposure to TCE has been shown to reduce in body weight gain in rats. This effect is age-dependent and is more pronounced with prolonged exposure. Liver enzymesLiver enzymes are essential for many metabolic processes in the liver. They are also important for liver function testing (Kumarand and Malviya, 2024) because high serum levels of these enzymes might be a symptom of liver dysfunction or damage. Serum enzyme levels are considerable to be indicators of overall health, especially in relation to hepatocyte injury and stress. (Nwankwo et al., 2019; Abdul-Azeez and Mutlag, 2023; Taqa and Sultan, 2024). Any changes in the sensitive biomarkers ALP and ALT considered a diagnosis of hepatic damage because they are cytoplasmic in location and are released into the circulation after cellular damage (Jawad et al., 2018; Gabriel et al., 2019; Ahmed and Mohammed, 2022). The liver is highly susceptible to damage following TCE exposure. In cases of acute and chronic TCE poisoning, rats frequently exhibit symptoms indicative of hepatic injury, such as jaundice; scleral icterus; elevated alanine transaminase (ALT), aspartate transaminase (AST) levels; and hepatomegaly. Multiple epidemiological studies have demonstrated that TCE exposure can lead to liver injury (Yamazaki et al., 2002; Zhu et al., 2024) found minimal changes in liver enzymes after low-dose or short-term TCE exposure, whereas Kawamoto (1984) found liver enzyme elevation only after high-dose or chronic exposure. The histopathological results of this study were in agreement with those of Gupta and Kumar (2022), who concluded that the liver of TCE-treated rat showed hyperplasia and hypertrophy and nuclei of different shapes and sizes, whereas the liver of the control group was normal in these studies. In contrast, the results of this study showed mild degenerative changes of hepatocytes, due to the long duration of the treatment with oil. Significant information about the hepatotoxic effects of trichloroethene is revealed by examining histological alterations in rat livers after exposure. Exposure to trichloroethene causes a variety of liver changes that are suggestive of severe hepatic dysfunction, such as bile duct hyperplasia, necrosis, and hepatocyte vacuolization. Interestingly, a dose-dependent link appears between the intensity of histopathological alterations and the length and concentration of exposure (Santos et al., 2022). An immunological reaction to TCE-induced liver injury is demonstrated by leukocyte infiltration and portal inflammation. This immune response may cause more tissue damage and interfere with natural liver repair processes, which may worsen liver disease (Andani et al., 2023; Yuming et al., 2024). AcknowledgmentWe acknowledge the University of Baghdad for providing the tools needed to complete this study and Baghdad Laboratories for providing the technical assistance needed to carry it out the study. FundingThis research received no specific grant. Authors’ contributionsNoora Nadhim Ali: Data curation; formal analysis; investigation, methodology; review and editing. Prof. Zainab Jamal Mohammed Jawad: Writing - original draft. Conflict of interestAccording to the authors, there are no conflicts of interest. Data availabilityThis article includes all of the data produced or examined during the course of this study. ReferenceAbdul-Azeez, Z.M. and Mutlag, S.H. 2023. Possible protective anticancer effect of ethanol fraction of Iraqi Hibiscus tiliaceus L. leaves extract on diethylnitrosamine-induced hepatocarcinogenesis in male rats. Iraqi J. Pharm. Sci. 32(Suppl.), 145–155. Abdel-Mageed, A.M., Abo-Elkhair, A.E. and Kamel, M.I. 2012. Toxic effects of some environmental pollutants on skin and hair of laboratory animals. Toxicol. Rep. 1, 125–132. Agency for Toxic Substances and Disease Registry (ATSDR). 2019. Toxicological profile for trichloroethylene. U.S. Department of Health and Human Services, Georgia. Ahmed, R.M. and Mohammed, A.K. 2022. Role of sodium butyrate supplement on reducing hepatotoxicity induced by lead acetate in rats. Iraqi J. Vet. Med. 46(2), 29–35. AL-Kurdy, M.J. and Kazaal, M.A. 2024. Antioxidant and hepatoprotective effects of chitosan on high fructose induced liver damage in Albino rats. Iraqi J. Sci. 4220–4229. Andini, A., Veterini, L., Wikurendra, E. A., Syafiuddin, A., Rimasari, D. and Aida, M.N. 2023. Histopathological findings of the liver and kidneys of wistar rat exposed to insect repellent. Bali Med. J. 12(2), 1330–1333. Bancroft, J.D. and Gamble, M. 2008. Theory and practice of histology techniques. . 6th ed., Edinburgh, NY: Churchill Livingstone Elsevier. Burtis. and Ashwood. 1999. Tietz textbook of clinical chemistry. 2nd ed., Philadelphia, PA: Saunders. Capinha, L.M.S. 2024. Application of new approach methodologies in molecular toxicology: A case study on metabolism and cellular stress responses from glutathione conjugation products of trichloroethylene. Chiu, W.A., Jinot, J., Scott, C.S., Makris, S.L., Cooper, G.S., Dzubow, R.C., Bale, A.S., Evans, M.V., Guyton, K.Z., Keshava, N., Lipscomb, J.C., Barone, S., Fox, J.F., Gwinn, M.R., Schaum, J. and Caldwell, J.C. 2013. Human health effects of trichloroethylene: key findings and scientific issues. Environ. Health Perspect. 121(3), 303–311. Deferme, L. 2015. Oxidative stress responses in hepatocarcinogenesis: Unravelling the mechanisms using a toxicogenomics approach. Dixon, W.J. 1980. Efficient analysis of experimental observations. Annu. Rev. Pharmacol. Toxicol. 20, 441–462. Environmental Protection Agency (EPA, USA). 2017. Risk management for trichloroethylene(TCE). Available via https://www.epa.gov/assessing-and-managing-chemicals-under-tsca/risk-management-trichloroethylene-tce EPA. 2016. Contaminant Candidate List 4 (CCL 4). U.S. Environmental Protection Agency. Famurewa, A.C., Ekeleme-Egedigwe, C.A., Ogbu, P.N., Ajibare, A.J., Folawiyo, M.A., Obasi, D.O. and Narayanankutty, A. 2023. Morin hydrate downregulates inflammation-mediated nitric oxide overproduction and potentiates antioxidant mechanism against anticancer drug doxorubicin oxidative hepatorenal toxicity in rats. Avicenna J. Phytomed. 13(5), 475. Gabriel, U.U., Edori, O.S. and Egobueze, E.C. 2019. Plasma enzymes and electrolytes in Heterobranchus bidorsalis treated with cypermethrin. Biochem. Anal. Biochem. 8(380), 2161–1009. Gella, F.J., Olivella, T., Cruz-Pastor, M., Arenas, J., Moreno, R., Durban, R. and Gomez, J.A. 1985. A simple procedure for routine determination of aspartate aminotransferase and alanine aminotransferase with pyridoxal phosphate. Clinica. Chim. Acta. 153(3), 241–247. Gupta, S. and Kumar, D. Histopathological studies on the liver of rats exposed to toluene and trichloroethylene pre-treated with phenobarbital. Hafez, E.S.E. 1970. Reproduction and breeding techniques for laboratory animals. Hameed, H.A. and Hassan, A.F. 2022. The prophylactic anti-inflammatory effect of omega-7 against paracetamol-induced liver injury in rats. Iraqi J. Vet. Med. 46(2), 43–47. Horzmann, K.A., Portales, A.M., Batcho, K.G. and Freeman, J.L. 2020. Developmental toxicity of trichloroethylene in zebrafish (Danio rerio). Environ. Sci. Processes &. Impacts 22, 728–739. Jawad, B.M., Musa, T.N. and Ali, A.J. 2018. Determination of deltamethrin residues in mandarin orange and studying its physiological effects on male mice blood profile. Iraqi J. Agricult. Sci. 49(6), 1160. Kakodia, A.K. and Awasthi, S. 2024. Kerosene: risk assessment. Hazardous chemicals: overview, toxicological profile, challenges, and future perspectives, p: 219. Kawamoto T. 1984. Metabolism of trichloroethylene in the isolated perfused rat liver. Yamaguchi Igaku 33, 527. Kumar, S. and Malviya, R. 2024. Bioprinting of hepatic tissue using 3D technology: transitioning beyond laboratory models to real-world applications in medical treatments. Appl. Mater. Today 39, 102307. Lash, L.H. 2024. Trichloroethylene: an update on an environmental contaminant with multiple health effects. Annu. Rev. Pharmacol. Toxicol. 65, 65, 507–527. Lou, H., Yang, J., Liu, Q. and Yang, Q. 2024. Hazards of trichloroethylene on the liver in animals: a systematic review and meta-analysis. Emerg. Contaminants 100338. Luo, Y., Lu, H., Peng, D., Ruan, X., Eugene Chen, Y. and Guo, Y. 2022. Liver-humanized mice: a translational strategy to study metabolic disorders. J. Cellular Physiol. 237, 489–e506. Mustafa Khalaf, M. and Salih, R.A. 2023. Investigating the potential hepatoprotective effect of quercetin in male rats following acute exposure to cyclophosphamide. Iraqi J. Vet. Med. 47(2), 23–30. Mayans, B., Camacho-Arévalo, R., García-Delgado, C., Alcántara, C., Nägele, N., Antón-Herrero, R. and Eymar, E. 2021. Mycoremediation of soils polluted with trichloroethylene: first evidence of pleurotus genus effectiveness. Appl. Sci 11(4), 1354. Ng, V. 2002. Effects of disease on clinical laboratory tests, Vol. 1 and 2. Eds., Young, D.S and Friedman, R.B. Washington, DC: AACC Press, vol. 48(4), pp: 682–683. Nwankwo, R.C., Ibegbu, M.D., Onyekwelu, K.C., Ejezie, C.S., Ikekpeazu, J.E. and Ejezie, F.E. 2019. Biochemical and histopathological effects of sub-acute exposure of albino rats to fumigants—dichlorvos and cypermethrin. Interdiscipl. Toxicol. 12(4), 180. Q Mustafa, M. and Jm Jawad, Z. 2024. Evaluating the hepatoprotective potential of ginger ethanolic extract against lambda-cyhalothrin-induced toxicity in male rats. Iraqi J. Vet. Med. 48(2), 26–31; doi:10.30539/r5qyq534 Santos, R. M. B., Monteiro, S. M. V., Cortes, R. M. V., Pacheco, F. A. L., & Fernandes, L. F. S. (2022). Seasonal differences in water pollution and liver histopathology of Iberian barbel (Luciobarbus bocagei) and Douro nase (Pseudochondrostoma duriense) in an agricultural watershed. Water, 14(3), 444. Available via https://www.semanticscholar.org/paper/80ba02d86323a0d3bd862b9e16c1a0b7eebd2c64 Shipp, A.M., Smith, J. and Lee, R. 2010. Toxicological assessment of chemical mixtures: effects on body weight and metabolism. Toxicol. Appl. Pharmacol. 250(2), 183–189. Shuai, Z., Leung, M.W., He, X., Zhang, W., Yang, G., Leung, P.S. and Eric Gershwin, M. 2016. Adaptive immunity in the liver. Cell. Mol. Immunol. 13, 354–368. Slunge, D., Andersson, I. and Sterner, T. 2022. Reach authorisation and the substitution of hazardous chemicals: the case of trichloroethylene. J. Cleaner Prod. 364, 132637; doi:10.1016/j.jclepro.2022.132637 SPSS. 2019. Statistical Packages of Social Sciences – SPSS/IBM Statistics 26 step. 16th ed; doi:10.4324/9780429056765 Stermer, A.R., Klein, D., Wilson, S.K., Dalaijamts, C., Bai, C.Y., Hall, S.J., Madnick, S., Bianchi, E., Chiu, W.A. and Boekelheide, K. 2019. Differential toxicity of water versus gavage exposure to trichloroethylene in rats. Environ. Toxicol. Pharmacol. 68, 1–3; doi:10.1016/j.etap.2019.02.003 Taqa, G.A. and Sultan, O.N. 2024. Physiological and histological effects of apigenin and luteolin on cytarabine-injected rats. Baghdad Sci. J. 21(1), 1. Tietz, N.W., Burtis, C.A., Duncan, P., Ervin, K., Petitclerc, C.J., Rinker, A.D., Shuey, D. and Zygowicz, E.R. 1983. A reference method for measurement of alkaline phosphatase activity in human serum. Clin. Chem. 29(5), 751–761. U.S. Environmental Protection Agency (EPA). 2011. Toxicological review of trichloroethylene (TCE) (EPA/635/R-10/003F). Woolbright, B. and Jaeschke 2017. The impact of sterile inflammation in acute liver injury. J. Clin. Translational Res. 3,170–188. Wu, C. and Schaum, J. 2000. Exposure assessment of trichloroethylene. Environ. Health Perspect. 108, 359–363. Yamazaki, H., Smith, J., Lee, R. and Kim, S. 2002. Biochemical and histopathological effects of trichloroethylene in rats. Environ. Toxicol. Pharmacol. 12(2), 89–98. Yang, M., Zhu, W., Lv, Y., Jiang, B., Jiang, C., Zhou, X. and Wu, L. 2023. A dual-responsive ratiometric indicator designed for in vivo monitoring of oxidative stress and antioxidant capacity. Chem. Sci. 14(45), 12961–12972. Yoo, Y.M., Lee, S.H., Kim, H.S. and Kim, S.H. 2008. Trichloroethylene induces neurobehavioral and neurochemical alterations in rats. Arch. Toxicol. 82(12), 937–946. Young, D.S. 1997. Effects of drugs on clinical laboratory tests. J. Anal. &. Clin. Biochem. 34(6), 579–581. Zhang, C., Yu, Y., Yu, J.F., Li, B.D., Zhou, C.F., Yang, X.D., Wang, X., Wu, C., Shen, T. and Zhu, Q.X. 2018. Viral mimic polyinosine-polycytidylic acid potentiates liver injury in trichloroethylene-sensitized mice: viral-chemical interaction as a novel mechanism. Ecotoxicol. Environ. Saf. 155, 101–108. Zhu, L., Jia, X., Xie, H., Zhang, J. and Zhu, Q. 2024. Trichloroethylene exposure, multi-organ injury, and potential mechanisms: a narrative review. Sci. Total Environ. 946, 174029. | ||
| How to Cite this Article |
| Pubmed Style Ali NN, Jawad ZJM. Pathological investigation for effect of trichloroethylene on rat liver. Open Vet. J.. 2025; 15(10): 5304-5311. doi:10.5455/OVJ.2025.v15.i10.47 Web Style Ali NN, Jawad ZJM. Pathological investigation for effect of trichloroethylene on rat liver. https://www.openveterinaryjournal.com/?mno=247810 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.47 AMA (American Medical Association) Style Ali NN, Jawad ZJM. Pathological investigation for effect of trichloroethylene on rat liver. Open Vet. J.. 2025; 15(10): 5304-5311. doi:10.5455/OVJ.2025.v15.i10.47 Vancouver/ICMJE Style Ali NN, Jawad ZJM. Pathological investigation for effect of trichloroethylene on rat liver. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5304-5311. doi:10.5455/OVJ.2025.v15.i10.47 Harvard Style Ali, N. N. & Jawad, . Z. J. M. (2025) Pathological investigation for effect of trichloroethylene on rat liver. Open Vet. J., 15 (10), 5304-5311. doi:10.5455/OVJ.2025.v15.i10.47 Turabian Style Ali, Noora Nadhim, and Zainab Jamal Mohammed Jawad. 2025. Pathological investigation for effect of trichloroethylene on rat liver. Open Veterinary Journal, 15 (10), 5304-5311. doi:10.5455/OVJ.2025.v15.i10.47 Chicago Style Ali, Noora Nadhim, and Zainab Jamal Mohammed Jawad. "Pathological investigation for effect of trichloroethylene on rat liver." Open Veterinary Journal 15 (2025), 5304-5311. doi:10.5455/OVJ.2025.v15.i10.47 MLA (The Modern Language Association) Style Ali, Noora Nadhim, and Zainab Jamal Mohammed Jawad. "Pathological investigation for effect of trichloroethylene on rat liver." Open Veterinary Journal 15.10 (2025), 5304-5311. Print. doi:10.5455/OVJ.2025.v15.i10.47 APA (American Psychological Association) Style Ali, N. N. & Jawad, . Z. J. M. (2025) Pathological investigation for effect of trichloroethylene on rat liver. Open Veterinary Journal, 15 (10), 5304-5311. doi:10.5455/OVJ.2025.v15.i10.47 |