| Research Article | ||
Open Vet. J.. 2025; 15(10): 5230-5237 Open Veterinary Journal, (2025), Vol. 15(10): 5230-5237 Research Article Serological, molecular, and clinical diagnosis of caprine brucellosis in Tikrit, IraqJassim Mohamed Suleiman*, Abdulkader Hadi Kandori and Omar-Althani Shareef SaedDepartment of Internal and Preventive Medicine, College of Veterinary Medicine, University of Tikrit, Tikrit, Iraq *Corresponding Author: Jassim Mohamed Suleiman. Department of Internal and Preventive Medicine, College of Veterinary Medicine, University of Tikrit, Tikrit, Iraq. Email: drjassimms1980 [at] tu.edu.iq Submitted: 14/04/2025 Revised: 30/08/2025 Accepted: 15/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
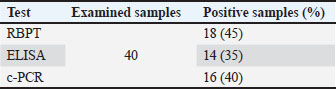
AbstractBackground: Brucellosis is a prevalent and significantly endemic cause of abortion in goat. Among the various diagnostic methods used for the confirmatory diagnosis of the causing agent, the standard polymerase chain reaction (c-PCR) method is more accurate and appropriate for diagnosing Brucella spp. infection in goats than serological tests such as RBPT and ELISA. The current study compared molecular and serological tests with the diagnosis of Brucella spp. Aim: This study used the enzyme-linked immunosorbent assay (ELISA) and the Rose Bengal Plate Test (RBPT) on blood serum to determine the prevalence of brucellosis in goats in Tikrit, Iraq. In addition, c-PCR was conducted on whole blood samples for Brucella spp. detection of investigation the kind to Brucella spp. using the technique of multiplex polymerase chain reaction (m-PCR) and to comparative the c-PCR methodology with RBPT and indirected enzyme-linked immunosorbent as approaches to illness diagnosis. Methods: This study inspected 40 aborted goats of different ages gathered from various areas of Tikrit, Iraq, from November 2023 to March 2024. Molecular analysis included the collection of a 1 ml blood sample from the jugular vein in an anticoagulant EDTA-coated plastic tube, as well as 2–4 ml blood in an anticoagulant-free glass gel tube. In order to detection Brucella antibodies at the serum using the RBPT and indirect ELISA test, these tubes were centrifuged for 5 minutes at 3,000 rpm. Meanwhile, PCR was used on the complete blood samples to identify Brucella species. Results: Using RBPT and ELISA of blood serum and the traditional PCR test, the overall prevalences of brucellosis in abortion goats were 40% (16), 35% (14), and 45% (18) out of 40, respectively. The multiplex PCR method used to primary identification of Brucella spp. showed that prevalence of B. melitensis was 27.5% (11 out of 40), with positive band in 731 bp and B. Abortus was 12.5% (5 of 40), with positive bands at 498 bp, indicating the prevalence of Brucella spp. According to the Kappa value of 0.218, the present research revealed a difference between RBPT and the c-PCR technique in the diagnosis of Brucella spp. at abortion goat, and accuracy, specificity, and sensitivity of RBPT were 87.5%, 83.3%, and 40%, respectively, when comparative to the technique of c-PCR. Accorded to Kappa value of 0.216, the present study also found an similarity between ELISA and the c-PCR technique at the accuracy, specificity, and sensitivity and of ELISA were 81.2, 95%, and 95%, respectively, when compared with the technique of c-PCR. The study concluded that Brucellosis is widespread and important for inducing abortion in goats in Tikrit city, and the technique of c-PCR is more dependable and suitable for the detection Brucella spp. infection of goats than RBPT and ELISA. Conclusion: The study concluded that B. mentensis and B. abortus are important causes to abortion of goats in some areas of Tikrit city. The tests of serologically used in this study were the RBPT most sensitive, whereas the indirect ELISA test was the most specific, and the technique of c-PCR was more dependable and suitable for the detection Brucella spp. infection of goats than the serological tests RBPT and ELISA. Keywords: Molecular, Serological, Goat brucellosis. IntroductionThe gram-negative bacteria that resemble coccobacilli (short rods) cause brucellosis. It does not produce spores and is not motile. The synthesis of H2S and urease, sensitive to dyes, cell wall antigen, and phage sensitive are characteristics that distinguish the various species. Some principal species have biovars. Brucella species are approximately 0.6 to 1.5 μm by 0.5 to 0.7 μm, facultative intracellular gram-negative coccobacilli that do not form spores and capsules, or flagella (Di Bonaventura et al., 2021). Brucella neotomae, B. abortus, B. ovis, B. melitensis, B. canis, B. suis, B. pinnipedialis, B. ceti, B. microti, B. vulpis, B. Papionis, and B. inopinata are among the genus’s at least twelve species (Kurmanov et al., 2022; Teshome et al., 2022). Microorganisms are housed in the host cells. Most bacterial infections in goats and sheep are caused by B. melitensis and are expelled in different fluids, including milk, urine, semen, and vaginal secretions. Bacteria are also found in placental tissues, amniotic fluid, and aborted fetuses. The vaginal shedding of the causative organisms after parturition or miscarriage usually takes three weeks, although it can take up to 2 months (Promsatit et al., 2024). Brucellosis is an infectious bacterial disease of goats, which is considered very important for public health and animal production (Anonymous, 2001; Constable et al., 2017). The Brucella infection is an important bacterial diseases that cause miscarriage in flocks of sheep and goats in the majority of countries, which results in enormous financial losses. The illness is typified by the presence of epididymitis and orchitis in male samples, placentitis in pregnant females, and uterine discharges in females. In livestock, the illness showed up as sterility, feeble lambs and children, stillbirths, and abortions in the last third of pregnancy. Owing to its high morbidity, it also causes a significant loss of production. Although brucellosis is widespread globally, it remain a substantial threat to low and middle-income nations (Al-mashhadany, 2021; Abd et al., 2025). The clinical diagnosis of the disease is challenging because of its wide range of clinical presentations. Thus, the isolation or demonstration of an immune response to its antigens must be employed to directly confirm the diagnosis (Rasheed et al., 2019; Ebid et al., 2020). The distribution and density of Brucella in the sample are key determinants of bacteriological method effectiveness. Because not all sick animals develop enough antibody and because cross-reactivity with other Bacterial species may cause false-negative result, serological tests are not always conclusive. Culture techniques also tend to be ineffective because of the significant time required and the attendant dangers of working with pathogenic microbes (Radostits et al., 2007). In addition, not all infection animals develop detectable antibodies, and false-negative results could arise from interference with other bacteria, rendering serological techniques uncertain (Ilhan et al., 2008). Even when the samples contain very little Brucella, the PCR approach has been utilized as a sensitive and accurate assay to the detected of Brucella spp. from variety of clinically sample, including semen, milk, blood, and aborted fetuse. But these methods are insufficient (Anonymous, 2001; Dehkordi et al., 2012). Using specific primers, the aimed of the present study to determine whether the species-specific PCR assay is a reliable diagnostic technique for detecting Brucella spp. DNA in goats’ whole blood following miscarriage. Materials and MethodsEthical approvalThe Tikrit University Research Ethics Committee has approved this investigation. With the consent of the owners and the local veterinarian, all samples, including blood samples, were taken in accordance with standard protocols without causing any harm to the animals. Animals and samplesFrom November 2023 to March 2024, 40 aborted goats of different ages from various areas of Tikrit, Iraq, were used in this study. For molecular analysis, taken blood samples 1 ml from the jugular vein were placed in an anticoagulant EDTA plastic tube, while 2–4 ml were stored in free-anticoagulant gel tube and centrifugation for 5 minutes at 3,000 rpm to extract serum samples in order to use the Rose-Bengal plate test to identify Brucella antibodies in serum and indirect ELISA test. In contrast, entire blood samples were subjected to the (PCR) to identify Brucella species. Use a single pair of primers tailored to the Brucella species strains. All whole blood and Eppendorf tubes of serum were stored frozen at −20°C until use (Coles, 1986; Charan and Biswas, 2013). Sample analysis using serologyRose-Bengal test (RBPT)The RBPT antigen was used for test serum samples (Alton et al., 1988). On a white ceramic plate, 30 μl the serum and 30 μl of RBPT antigen were mixed gently. After 4 minutes of plate agitation, agglutination was deemed positive. To compare the results, negative and positive control sera were used. The i-ELISA test is an indirect enzyme-linked immunosorbent assayFor the quantification of individual Ab against brucellosis, indirect ELISA tests were conducted following the manufacturer instruction (SunLong Biotech, China). The ELISA Microplate Reader (BioTek, USA) was used to measure fluorescence at an optical density (OD) of 450 nm. After calculating the test efficiency, the samples were deemed positive if the OD value was greater than or equal to CUT OFF. The cut-off value (CUT OFF) was 0.299. The kit is intended to the identifications of antibodies against Brucella melitensis. Additionally, it minimizes cross-reaction with other Gram-negative bacterial (Al-Marzooqi et al., 2022). PCR assaysDNA extractionThe Brucella spp. of genomic DNA was extracted from 40 aborted goat whole blood samples: use a commercial DNA extraction kit (AddBio, Korea). According to manufacturer instruction, using NanoDrop (BioDrop, England), the concentrations of extraction DNA, regard at a wavelength of 450 nm, range between 20 and 65 ng/μl. By calculate the ratio of (A440–A460 nm), the purity was found to be between 1.61 and 1.74 (Green and Sambrook, 2019). DNA amplificationThree primer combinations were employed to target the IS711 gene for B. ovis [O-R (5′- CGG GTT CTG GCA CCA TCG TCG -3′)], B. melitensis [M-R (5′- AAA TCG CGT CCT TGC TGG TCT GAA -3′)], B. Abortus [A-R (5′- GAC GAA CGG AAT TTT TCC AAT CCC -3′)] and Brucella spp. [IS-F: (5′-TCG CGA TCA CTT AAG GGC CTT CAT-3′)]were prepared using the Ready to use AccuPower® PCR PreMix Kit (AddBio, Korea) as designed by Halling and Bricker (1994). The MasterMix tube was prepared to a total volume of 25 μl. PCR-reaction was carried out in a thermal cycler using the Optimase ProtocolWriterTM online to achieve an optimal situation for every primer. Brucella melitensis detection by electrophoresis in 1.5% agarose gel stained with ethidium bromide at 223, 498, 731, and 976 bp, respectively. A comparison of the methods employed in this investigationThe concordance between the c-PCR method and the microscopic examination (ME) of blood smears was ascertained by the kappa value. When the kappa number is less than zero, there is no similarity between two tests. If the Kappa value falls between 0.0 and 0.20, the similarity is minor; if it falls between 0.21 and 0.40, it is fair; if it falls between 0.41 and 0.60, it is moderate; if it falls between 0.61 and 0.80, it is substantial; and if it falls between 0.81 and 1, it is nearly perfect. Furthermore, the gold standard PCR (c-PCR) method’s sensitivity, specificity, and accuracy were contrasted with those of RBPT and ELISA. (Baratloo et al., 2015). Statistical analysisSPSS 16.0 was used for statistical analysis. McNemar’s chi-square test was used to assess the goodness-of-fit between RBPT and PCR, and the kappa statistic was used to measure their agreement, with P value of 0.05 being statistical significantly (Viera and Garrett, 2005). ResultsAccording to RBPT and I-ELISA of blood serum and the traditional PCR method, the total incidence of brucellosis in aborted goats in Tikrit city was 45% (18 out of 40), 35% (14 out of 40), and 40% (16 out of 40) in the current study (Table 1). Brucella spp. primary identification. Table 1. Brucellosis prevalence in goats using the Rose Bengal plate test, indirect ELISA test, and c-PCR technique.
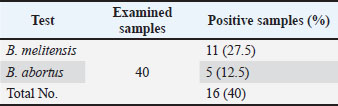
Brucella melitensis prevalence was 27.5% (11 out of 40), with positive bands at 731 bp, while B. Abortus prevalence was 12.5% (5 out of 40), with positive bands at 498 bp, according to the multiplex PCR technique results (Figs 1, 2), prevalence of the types of Brucella spp. (Table 2).
Fig. 1. B. Abortus prevalence was 12.5% (5 out of 40), according to the multiplex PCR method, with positive bands at 498 bp..
Fig. 2. Multiplex PCR indicated that the prevalence of B. melitensis was 27.5 %, with the positive bands at 731 bp. Table 2. Prevalence of Brucella spp. types in aborted goats in Tikrit city using multiplex polymerase chain reaction (n=40).
Using the achieved Kappa value of 0.218, this study found that a small extent of agreement was existent between RBPT and c-PCR test in Brucella spp. detection of abortions from goats and the accuracy, specificity, and sensitivity of RBPT were 40%, 83.3%, and 87.5%, respectively, when tested in comparison with c-PCR method (Table 3). The specificity, sensitivity, and accuracy of RBPT for diagnosing Brucella are established based on the comparison of the Kappa value between the c-PCR and RBPT techniques. Table 3. The sensitivity, accuracy, and specificity of RBPT in identifying Brucella were determined by comparing the kappa values of c-PCR and RBPT methods.
Based on the Kappa value of 0.216, this research also indicated slight agreement between ELISA and the c-PCR method in the diagnosis of Brucella spp. from goats’ abortions, and the accuracy, specificity, sensitivity and of ELISA were 81.2, 95%, and 95%, respectively, compared with the c-PCR method (Table 4). Table 4. ELISA’s sensitivity, specificity, and accuracy in identifying Brucella are determined by comparing the kappa values of ELISA and c-PCR methods.
DiscussionA hazardous zoonotic disease that is common throughout the world is brucellosis. It results in infertility and abortion. The facultative, gram-negative intracellular bacteria are the cause of the illness (El-Sharkawy et al., 2019; Albayati and Shareef, 2023). 16 (40%) goats that were aborted in Tikrit exhibited Brucella spp. infections, included B. abortus and B. Melitensis, at ratios of 11 (27.5) and 5 (12.5), respectively, according to the current study results. This agrees with the findings of anonymous (2001), who indicated that the main causative agent of brucellosis at small ruminant is B. Melitensis. Keeping different livestock species together may be linked to the examing of mixed B. melitensis and B. abortus infections (Gharekhani et al., 2016). According to a report (ZareBidaki et al., 2022), the presence of various and unvaccinated animal on the farm, the aging of the animal, the prevalence of miscarriage, and the interaction of cattle with wild animals are strong risk factors for brucellosis. In Egypt’s dairy farms that have been brucellosis-free, the discovery of B. melitensis and B. abortus in blood revealed how stray dogs contribute to the diseases comeback. The findings prompted the veterinary authorities to start organizing strategies to prevent this illness in dogs. To address the interface between canine brucellosis and goats (Hamdy et al., 2023). Our study showed more significant differences of ELIZA compared with RBPT, which was consistent with the results of ELISA being more specific, sensitive, and confirmatory of the test results than RBPT (Rahman et al., 2013). The results of RBPT were in similarity with those of a previous study (Leyla et al., 2003), after performing RBPT on aborted goat serum, which yielded 66.7%. Although the findings of this investigation were similar to those of Olufemi et al. (2018), 39 out of 50 samples had a positive RBPT result. This was explained by the disparity in the animal’s age and sample characteristics. Despite not matching the researcher’s findings (Constable et al., 2017; Gusi et al., 2019), the RBPT was used as a diagnostic screen test because it was quick and simple to use and yielded results, particularly when the numbers were high, because it detects antibodies. RBPT is known as the most widely used serological test because of its ease, speed, and low cost. It also detects all types of antibodies, although it gives false positive and negative results (Akowongo and Kakooza, 2022). It is used to identify early infection in herds because it has greater sensitivity to immunoglobulin IgM, of which there is a significant percentage at the start of the infection (Gusi et al., 2019). However, one of the disadvantages of this test is that it gives false positive results by a percentage of (1%–3%). In addition, it does not distinguish vaccinated animals from infected ones (Corbel, 1997; Li et al., 2021). This was significantly higher than that recorded by Ilyas (2019), representing 9.69% of the goats in Northern Iraq’s Duhok and Sumel Districts. The percentage showed in this study was also higher than that reported by Saleem et al. (2004), which was 13% in goats using the RBPT in Nineveh Governorate, and 5.5% in aborted goats using the RBPT reported by Hadad and Al-azawy (1991). This difference between the results may be due to a difference in the period during which samples were collected from the lamb, the number of bacteria present in the lamb, and the immune status. The susceptibility of animals to brucellosis depends on several factors, including disease spread, grazing strategy, number of animals, and population density (Almashhadany, 2021; Thuamsuwan et al., 2023). Using RBPT and ELISA, aborted goats showed higher positivity than non-aborted goats. This is because positive serological tests are associated with more abortions than cases that have not yet been aborted. The aborted antibodies also interact less with the ELISA test compared to any aborted condition, and this can be explained by the gradual increase in the number of antibodies after the abortion, whereas the condition occurs in non-aborted cases (Peck et al., 2018). It is mostly chronic and has been aborted some time ago, and antibodies are detected. IgG is used in this test because it remains in chronic cases, unlike IgM, which decreases significantly in late infection (Nielsen and Yu, 2010). The researcher also pointed out that isolating Brucella is considered a golden way to give accurate results because serological tests do not give actual results for the spread of Brucella because the number of antibodies gradually diminishes. In goats infected with Brucella, the immune response starts 7–9 weeks before miscarriage or delivery and lasts for 30 weeks. A significant portion of samples yield positive RBPT results within 24–48 hours following an abortion or parturition (Kakooza et al., 2022). The study showed that blood serum may be used to quickly and accurately diagnose Brucella germs using the polymerase chain reaction (PCR) approach. This is consistent with Ameen et al. (2023) and the use of previously described primers. A comparative analysis was conducted using blood cells and blood serum to assess the effectiveness of PCR technology. As the researcher demonstrated, PCR can detect 30–80 femtogram (fg) of the DNA genetic material of Brucella bacteria, making it a highly sensitive and specific technology due to its ability to detect small amounts of DNA (ElTahir et al., 2018). From observing the result of the PCR reaction test using the different primers that were applied to the samples under study from the positive sera for the RBPT and ELISA tests for serum, the aim of using the first primer, IS711gene, IS-F, was to detect the genus Brucella, which includes the following three types: (A-R) B. abortus, (M-R) B. melitensis, and (O-R) B. Ovis. This primer is present in the Brucella genus. Therefore, when bands of replicated DNA appear on the agarose gel, evidence of the absence of Brucella at the samples, and when the bands appear when using the second primer, it is characterized by the B. melitensis, as it is evidence that the type of Brucella belongs to the type B. melitensis, and this agrees with most researchers. Therefore, the main cause of abortion in goats is Brucella infection, especially type B. melitensis (Saleem et al., 2023). The PCR technique, especially using species-specific primers, is an appropriate and rapid method for diagnosing Brucella bacteria, through which the genus and specific type of bacteria can be known for study and their identity determined, even if they are in a mixture containing large groups of bacterial pathogens and even if they are close to each other in antigenic composition. This explains the accuracy and sensitivity of PCR in detecting pathogens that other techniques cannot detect (Hamdy et al., 2023). The efficiency of the primer used lies in the proportion of the nitrogenous bases it contains, especially the C + G type, relying on the bonds between G and C, which are stronger than T and A. Thus, the connection between the primer and its complementary site on the DNA template is stronger; therefore, the connection forms a clear band. Any replication is good, and this explains why the primers used in the application give good results, especially during the replication period (Hassan et al., 2025). Hinić et al. (2008) stated that the IS711 gene was used to distinguish between the Brucella genera B. abortus, and B. melitensis. Brucella strains were analyzed, and all PCR reaction produced PCR product that was associated with Brucella species in all cases, whereas no amplification with any of the primers was observed for non-Brucella species. It is important to improve PCR and DNA extraction protocols to increase the sensitivity of the convention tissue PCR method. In the current study on aborted goat and cow fetuses, different target genes for B. melitensis showed the highest sensitivity in tissue, semen, milk, and blood comparative with convention bacterial isolation, using different circumstances of optimization of PCR protocols, which is consistent with (Çiftci et al., 2017). In terms of sensitivity, the study’s findings were in close agreement with the values of 79.12% and 88% reported by Zakaria (2018) and Verma et al., (2017), respectively. Compared with RBPT, which detects antibodies, PCR is more accurate, dependable, and capable of detecting antigens. It was discovered that the sensitivity of RBPT was higher than that of ELISA (Jain et al., 2012). The study findings, sensitivity, and specificity of the ELISA test for abortion goats were 81.2% and 95%, respectively, when the PCR device was utilized as an evaluation instrument. The study’s sensitivity rate was nearly identical to the 47.7% sensitivity attained by Verma et al., 2017. If the PCR apparatus was employed as a tool for assessment, the results obtained by Zakaria (2018) were also similar in terms of sensitivity, as it was 55.49% in aborted goats, but the specificity and positive and negative prediction values differed, reaching 52.08%, 81.45%, and 23.58%, respectively. The result of this study was also similar to those of Mukherjee et al. (2007), as the specificity reached 81% when using PCR as an evaluation tool and differed in the positive and negative prediction values, which amounted to 83.3% and 55.5%, respectively. The results showed that the sensitivity was 98.2% and the specificity was 100%, which differed from the results reported by Peck et al. (2018). These findings also differed from those reported by Gompo et al. (2021), who found that the sensitivity of the ELISA test was 5.5% when PCR was used as the assessment technique. In vitro agglutination test results produce false-negative findings for early infection (Constable et al., 2017). The present study findings indicate that the c-PCR and RBPT techniques have a high degree of agreement (Kappa value of 0.190), although the c-PCR methodology demonstrates poor accuracy and higher specificity and sensitivity. RBPT is the most sensitive and quick screening test for detecting animal Brucella infections, and can provide outcomes that are both falsely positive and falsely negative. In this study, a PCR and a serological technique for brucellosis detection were compared (Kazemi et al., 2008). Even in cases where there are few Brucella present, PCR has proven to be a sensitive and accurate method for the detection Brucella species in several clinical samples (Ilhan et al., 2008). ConclusionsThe study concluded that B. mentensis and B. abortus are important causes of abortion in goats in some areas of Tikrit city. The tests of serologically used in this study were the RBPT most sensitive, whereas the indirect ELISA test was the most specific, and the technique of c-PCR was more dependable and suitable for the detection Brucella spp. infection of goats than the serological tests RBPT and ELISA. AcknowledgmentThis study was supported by the College of Veterinary Medicine/University of Tikrit. Thanks to all goats owner for their collaboration. Conflict of interestNo conflicts of interest were disclosed by the writers. FundingNo grant was received for this study, and it has been supported by self-funding. Authors’ contributionsOmar-Althani Shareef Saed was in charge of animal care, clinical parameters, and blood sample collection, while Dr. Jassim Mohamed Suleiman served as the article’s corresponding author. Abdulkader Hadi Kandori was in charge of the research management and study observations. Each author contributed equally to the article’s writing. Data availabilityAll data are provided in the manuscript. ReferencesAbd, A.A., Al-Juhaishi, O.A. and Jumma, Q.S. 2025. Effects of sulpiride on the reproductive system of male rats after puberty. World Vet. J. 15(1), 42–48. Akowongo, C.J. and Kakooza, S. 2022. Exposure to Brucella species In goats and sheep in Karenga district, Uganda, diagnosed using the modified Rose Bengal method. Zoonotic. Dis. 2(3), 163–171. Albayati, O.A.S. and Shareef, R. 2023. Clinical, hematological and serological aspect of rams and aborted ewes with brucellosis. Tikrit J. Vet. Sci. 1(2), 19 -24. Al-Marzooqi, W., Elshafie, I.E., Al-Toobi, A., Al-Hamrashdi, A., Al-Kharousi, K. and ElTahir, Y. 2022. Seroprevalence and risk factors of brucellosis in ruminants in Dhofar Province, Southern Oman. Vet. Med. Int. 12(1), 1–7. Almashhadany, D.A. 2021. Diagnosis of brucellosis in sheep and goats raw milk by fast and reliable techniques. Iraqi J. Vet. Sc. 35(4), 663–668. Alton, G.G., Jones, L.M., Angus, R.D. and Verger, J.M. 1988. Techniques for the treatment of Brucellosis Institute National de le Recherché Agronomique. Uni. Paris, 75007. Ameen, A.M., Abdulaziz, N.S. and Ghaffar, N.M. 2023. Molecular detection and associated risk factors of Brucella melitensis in aborted sheep and goats in duck province, Iraq. Pathogens 12(4), 544. Anonymous, C.W. 2001. Brucellosis in goats and goats European Commission. Scientific Committee on Animal Health and Welfare. Doc. SANCO. 32(4), 33–40. Baratloo, A., Hosseini, M., Negida, A. and El Ashal, G. 2015. Part 1: simple definition and calculation of accuracy, sensitivity, and specificity. Spring 3(2), 48–49. Charan, J. and Biswas, T. 2013. How to calculate sample size for different study designs in medical research. Indian J. Psychol. Med. 35(2), 121–126. Çiftci, A., İça, T., Savaşan, S., Sareyyüpoğlu, B., Akan, M. and Diker, K.S. 2017. Evaluation of PCR methods for detecting Brucella strains in culture and tissues. Rop. Anim. Health. Prod. 49(4), 755–763. Coles, E.H. 1986. Veterinary clinical pathology. 4th ed., Philadelphia: Saunders Company, pp: 12–4. Constable, P.D., Hinchcliff, K.W., Done, S.H. and Grunberg, W. 2017. Veterinary medicine: a textbook of cattle diseases horses, goat, pigs and goats. 11th ed., Saunders, Canada, pp: 1630–738. Corbel, M.J. 1997. Brucellosis: an overview. Emerg. Infect. Dis. 3, 213–221. Dehkordi, F.S., Saberian, S. and Momtaz, H. 2012. Detection and segregation of Brucella abortus and Brucella melitensis in Aborted Bovine, Ovine, Caprine, Buffaloes, and Camelid Fetuses using conventional and real-time polymerase chain reactions. Thai J. Vet. Med. 42, 13–20. Di Bonaventura, G., Angeletti, S., Ianni, A., Petitti, T. and Gherardi, G. 2021. Microbiological laboratory diagnosis of human Brucellosis. Pathogens 10(12), 1623. Ebid, M., El Mola, A. and Salib, F. 2020. Seroprevalence of brucellosis in sheep and goats in the Arabian Gulf region. Vet. World 13(8), 1495–1509. El-Sharkawy, H., Ahmed, S.A., Salem, M.A.M., Soliman, S.M. and Soliman, H.S. 2019. Some epidemiological studies on sheep and goats Brucellosis in Egypt. Thesis Ph.D. Cairo University, Egypt. ElTahir, Y., Al Toobi, A.G., Al-Marzooqi, W., Mahgoub, O., Jay, M., Corde, Y. and Johnson, E.H. 2018. Serological, cultural and molecular evidence of Brucella melitensis infection in goats in Al Jabaal Al Akhdar, Sultanate of Oman. Vet. Med. Sci. 4(3), 190–120. Gharekhani, J., Rasouli, M., Abbasi-Doulatshahi, E., Bahrami, M., Hemati, Z. and Rezaei, A. 2016. Sero-epidemiological survey of brucellosis in small ruminants in Hamedan province, Iran. Anim. Res. 3(4), 399–405. Gompo, T.R., Shah, R., Tiwari, I. and Gurung, Y.B. 2021. Sero-epidemiology and associated risk factors of brucellosis among sheep and goat populations in South Western Nepal: a comparative study. BMC Vet. Res. 17, 132. Green, M.R. and Sambrook, J. 2019. Polymerase chain reaction (PCR). Cold Spring Harbor Protocols 6(6), 095109. Gusi, A.M., Bertu, W.J., De Miguel, M.J., Dieste-Pérez, L., Smits, H.L. and Muñoz, P.M. 2019. Comparison of the performance of lateral flow immunochromatography, iELISA, and Rose Bengal tests for the diagnosis of cattle, sheep, goat, and swine brucellosis. PLos Negl. Trop. Dis. 13(6), 7509. Hadad, J.J. and Al-azawy, Z.S. 1991. Incidence of brucellosis in sheep and goats in the Ninevah province, Yemen. Iraqi J. Vet. Sci. 4(1), 27–33. Halling, S.M. and Bricker, B.J. 1994. Characterization and occurrence of two repeated palindromic DNA elements of Brucella spp. Bru-RS1 and Bru-RS2. Mol. Microbiol. 14(4), 681–689. Hamdy, M.E.R., Abdel-Haleem, M.H., Dawod, R.E., Ismail, R.I., Hazem, S.S., Fahmy, H.A. and Abdel-Hamid, N.H. 2023. First seroprevalence and molecular identification report of Brucella canis among dogs in Greater Cairo region and Damietta Governorate of Egypt. Vet. World 16(1), 229–238. Hassan, S.U., Khan, F.A., Saddique, U., Shahid, M., Shah, S.S. and Ullah, N. 2025. Sero-molecular prevalence of Brucella melitensis in local small ruminants in Khyber Pakhtunkhwa, Pakistan. PLos One 20(2), e0315206. Hinić, V., Brodard, I., Thomann, A., Cvetni., Makaya, P.V., Frey, J. and Abril, C. 2008. Identification and differentiation of Brucella melitensis, B. abortus, B. suis, B. ovis, B. canis, and B. neotomae suitable for both conventional and real-time polymerase chain reaction systems. J. Microbiol. Methods 75(2), 75–378. Ilhan, Z., Solmaz, H., Aksakal, A., Gulhan, T., Ekin, I. and Boynukara, B. 2008. Detection of Brucella melitensis DNA in the milk of sheep after abortion by PCR assay. Arch. Med. Vet. 40, 141–146. Ilyas, N.M. 2019. Seroprevalence of Brucellosis in Sheep and Goats in Duhok and Sumel Districts of Northern Iraq. Int. J. Sci. Technol. Res. 5, 37–41. Jain, N., Boyle, S.M. and Sriranganathan, N. 2012. Effect of exogenous erythritol on Brucella growth and survival. Vet. Microbiol. 160(3–4), 513–516. Kakooza, S., Watuwa, J., Ipola, P.A., Munyiirwa, D.F.N., Kayaga, E., Nabatta, E., Mahero, M., Ssajjakambwe, P. and Kaneene, J.B. 2022. Seromonitoring of brucellosis in goats and sheep slaughtered at an abattoir in Kampala, Uganda. J. Vet. Diagn. Invest. 34(6), 964–967. Kazemi, B., Yousefi Namin, S.A., Dowlatshahi, M., Bandepour, F. and Kafilzadeh, L. 2008. Detection of brucella by peripheral blood PCR and comparison with culture and serological methods in suspected cases. Public Health 37, 96–102. Kurmanov, B., Zincke, D., Su, W., Hadfield, T.L., Aikimbayev, A., Karibayev, T., Berdikulov, M., Orynbayev, M., Nikolich, M.P. and Blackburn, J.K. 2022. Assays for identification and differentiation of Brucella species: a review. Microorganisms 10(8), 1584. Leyla, G., Kadri, G. and Ümra, O.K. 2003. Comparison of polymerase chain reaction and bacteriological culture for the diagnosis of goat brucellosis using aborted fetus samples. Vet. Microbiol. 93(1), 53–61. Li, Y., Tan, D., Xue, S., Shen, C., Ning, H., Cai, C. and Liu, Z. 2021. Prevalence, distribution and risk factors for brucellosis infection in goat farms in Ningxiang, China. BMC Vet. Res. 17, 39. Mukherjee, F., Jain, J., Patel, V. and Nair, M. 2007. Multiple genus-specific markers in polymerase chain reaction (PCR) assays improve the specificity and sensitivity of brucellosis diagnosis in field animals. J. Med. Microbiol. 6(10), 1309–1316. Nielsen, K. and Yu, W.L. 2010. Serological diagnosis of Brucellosis. Sec. Biol. Med. Sci. 1, 65–89. Olufemi, O.T., Dantala, D.B., Shinggu, P.A., Dike, U.A., Otolorin, G.R. and Amama, F.I. 2018. Seroprevalence of brucellosis and associated risk factors among indigenous goat breeds in Wukari, Taraba State, Tanzania. Niger. J. Pathog. 2018, 5257926. Peck, M.E., Chanachai, K., Alexander, B.H. and Bender, J.B. 2018. Seroprevalence of brucellosis in goats and sheep in Thailand: results from the 2013–2015 Thai National Brucellosis Surveillance System from 2013 to 2015. Transbound. Emerg. Dis. 65(3), 799–805. Promsatit, S., Heingraj, S. and Pumipuntu, N. 2024. Investigation of outbreaks and risk factors for brucellosis in goat and sheep farms in central Thailand. One Health 10(1), 125–132. Radostits, O.M., Gay, C., Blood, C.D. and Hinchclift, W.K. 2007. Veterinary medicine, textbook of the diseases of cattle, sheep, pigs, goats and horses. 10th ed., London, UK: ELBS Bailliere Tindall, pp: 963–94. Rahman, A.A., Saegerman, C., Berkvens, D., Fretin, D., Gani, M.O., Ershaduzzaman, M. and Emmanuel, A. 2013. Bayesian estimation of true prevalence, sensitivity, and specificity of indirect enzyme-linked immunosorbent assay, Rose Bengal Test, and slow aggregation test for the diagnosis of brucellosis in sheep and goats in Bangladesh. Prev. Vet. Med. 110(2), 242–252. Rasheed, S.T., Younis, L.S. and Abid, A.A. 2019. Effect of G(129)R polymorphism in growth differentiation factor 9 gene on Awassi ewes that breed out of season. Malaysian J. Biochem. Mol. Biol. 22(2), 69–73. Saleem, A.N., Rhaymah, M.S. and Shamoo, G.N. 2004. Isolation and prevalence of ovine and caprine brucellosis. Iraqi J. Vet. Sci. 18(1), 31–38. Teshome, D., Sori, T., Banti, T., Kidane, G., Wieland, B. and Alemayehu, G. 2022. Prevalence and risk factors of Brucella spp. infection in goats in the pastoral area of Borana, Southern Oromia, Ethiopia. Small Ruminant Res. 206, 106594. Thuamsuwan, N., Chanachai, K., Ekgatat, M., Srisai, P., Prarakamawongsa, T. and Rukkwamsuk, T. 2023. Risk factors associated with brucellosis seropositivity in goat farms of Sing Buri Province, Thailand. World Vet. J. 1, 191–193. Verma, A.K., Kumar, A. and Ahmed, I. 2017. Comparative efficacy of serological diagnostic methods and polymerase chain reaction evaluation for bovine brucellosis diagnosis. Iranian J. Vet. Res. 18(4), 279. Viera, A.J. and Garrett, J.M. 2005. Understanding interobserver agreement: the kappa statistic. Med. Res. Ser. 37(5), 360–363. Zakaria, A.M. 2018. Comparative assessment of sensitivity and specificity of the rose bengal test and modified in-house ELISA using the IS711 TaqMan real-time PCR assay as a gold standard for the diagnosis of bovine Brucellosis. Biomed. Pharmacol. J. 11(2), 951–957. ZareBidaki, M., Allahyari, E., Zeinali, T. and Asgharzadeh, M. 2022. Occurrence and risk factors of brucellosis in domestic animals: an artificial neural network approach. Trop. Anim. Health Prod. 54(1), 1–13. | ||
| How to Cite this Article |
| Pubmed Style Suleiman JM, Kandori AH, Saed OS. Serological, molecular, and clinical diagnosis of caprine brucellosis in Tikrit, Iraq. Open Vet. J.. 2025; 15(10): 5230-5237. doi:10.5455/OVJ.2025.v15.i10.39 Web Style Suleiman JM, Kandori AH, Saed OS. Serological, molecular, and clinical diagnosis of caprine brucellosis in Tikrit, Iraq. https://www.openveterinaryjournal.com/?mno=252674 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.39 AMA (American Medical Association) Style Suleiman JM, Kandori AH, Saed OS. Serological, molecular, and clinical diagnosis of caprine brucellosis in Tikrit, Iraq. Open Vet. J.. 2025; 15(10): 5230-5237. doi:10.5455/OVJ.2025.v15.i10.39 Vancouver/ICMJE Style Suleiman JM, Kandori AH, Saed OS. Serological, molecular, and clinical diagnosis of caprine brucellosis in Tikrit, Iraq. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5230-5237. doi:10.5455/OVJ.2025.v15.i10.39 Harvard Style Suleiman, J. M., Kandori, . A. H. & Saed, . O. S. (2025) Serological, molecular, and clinical diagnosis of caprine brucellosis in Tikrit, Iraq. Open Vet. J., 15 (10), 5230-5237. doi:10.5455/OVJ.2025.v15.i10.39 Turabian Style Suleiman, Jassim Mohamed, Abdulkader Hadi Kandori, and Omar-althani Shareef Saed. 2025. Serological, molecular, and clinical diagnosis of caprine brucellosis in Tikrit, Iraq. Open Veterinary Journal, 15 (10), 5230-5237. doi:10.5455/OVJ.2025.v15.i10.39 Chicago Style Suleiman, Jassim Mohamed, Abdulkader Hadi Kandori, and Omar-althani Shareef Saed. "Serological, molecular, and clinical diagnosis of caprine brucellosis in Tikrit, Iraq." Open Veterinary Journal 15 (2025), 5230-5237. doi:10.5455/OVJ.2025.v15.i10.39 MLA (The Modern Language Association) Style Suleiman, Jassim Mohamed, Abdulkader Hadi Kandori, and Omar-althani Shareef Saed. "Serological, molecular, and clinical diagnosis of caprine brucellosis in Tikrit, Iraq." Open Veterinary Journal 15.10 (2025), 5230-5237. Print. doi:10.5455/OVJ.2025.v15.i10.39 APA (American Psychological Association) Style Suleiman, J. M., Kandori, . A. H. & Saed, . O. S. (2025) Serological, molecular, and clinical diagnosis of caprine brucellosis in Tikrit, Iraq. Open Veterinary Journal, 15 (10), 5230-5237. doi:10.5455/OVJ.2025.v15.i10.39 |